Simple Summary
The association of screening mammography with breast cancer (BC) was investigated in cases with a hereditary predisposition unexplained by BRCA1 or BRCA2 and unrelated controls. Participants reported their lifetime mammography exposures in a questionnaire. Additionally, germline rare deleterious or predicted deleterious variants (D-PDVs) were investigated in 113 DNA repair genes. No association was found between having been exposed to mammograms (never vs. ever) and BC. However, when considering the number of mammograms, an increase in BC risk of 4% (95% CI: 1–6%) per additional exposure was found. When women were grouped according to their D-PDV carrier status and the estimated associated BC risk, mammograms doubled the BC risk of women carrying a D-PDV in a gene associated with BC with an odds ratio (OR) < 0.9, as compared to those carrying a D-PDV in a gene with an OR > 1.1. Even though mammographic screening reduces the risk of mortality from BC, the identification of populations more or less susceptible to ionizing radiation may be clinically relevant.
Abstract
Background: Women with a familial predisposition to breast cancer (BC) are offered screening at earlier ages and more frequently than women from the general population. Methods: We evaluated the effect of screening mammography in 1552 BC cases with a hereditary predisposition to BC unexplained by BRCA1 or BRCA2 and 1363 unrelated controls. Participants reported their lifetime mammography exposures in a detailed questionnaire. Germline rare deleterious or predicted deleterious variants (D-PDVs) in 113 DNA repair genes were investigated in 82.5% of the women and classified according to the strength of their association with BC. Genes with an odds ratio (OR) < 0.9 was assigned to the Gene Group “Reduced”, those with OR ≥ 0.9 and ≤1.1 to Group “Independent”, and those with OR > 1.1 to Group “Increased”. Results: Overall, having been exposed to mammograms (never vs. ever) was not associated with BC risk. However, an increase in BC risk of 4% (95% CI: 1–6%) per additional exposure was found under the assumption of linearity. When grouped according to D-PDV carrier status, mammograms doubled the BC risk of women carrying a D-PDV in Group “Reduced”, as compared to those carrying a D-PDV in Group “Increased”. Conclusions: Our study is the first to investigate the joint effect of mammogram exposure and variants in DNA repair genes other than BRCA1 and BRCA2 in women at high risk of BC; therefore, further studies are needed to verify our findings. Even though mammographic screening reduces the risk of mortality from BC, the identification of populations that are more or less susceptible to ionizing radiation may be clinically relevant.
1. Introduction
Women with a genetic predisposition to breast cancer (BC) may be at increased risk of cancer after exposure to ionizing radiation. Screening mammography is associated with exposure of the breast to a small dose of radiation (i.e., the regulatory limit set at 3.0 mGy for each view of an average-sized breast and two views of each breast), which has not been found to increase BC risk in the general population [1]. However, women who are at increased genetic risk might be more sensitive to the DNA-damaging effect of ionizing radiation, in particular women carrying a deleterious variant in a gene involved in the repair of DNA damages like BRCA1 and BRCA2 [2]. Moreover, women with a family history of BC are offered screening at earlier ages and at more frequent intervals than women from the general population. Assuming there is no lower threshold dose in which radiation exposure does not cause damage, repeated exposure to ionizing radiation from screening mammographic procedures may increase the risk of BC, and the possible benefit of mammography screening could be reduced due to the risk of radiation-induced tumors.
While exposures to diagnostic ionizing radiation to the chest excluding mammograms have been studied in the population with a genetic predisposition to BC [2,3,4,5,6,7,8], few studies have investigated the effect of mammograms in such populations, and these studies were essentially conducted among BRCA1/2 pathogenic variant carriers [2,4,9,10,11,12,13]. Therefore, we evaluated the effect of radiation exposure from mammography screening on BC risk in women with a genetic predisposition to BC and tested negative for a BRCA1/2 pathogenic variant. Furthermore, we evaluated whether carrying a rare deleterious or predicted deleterious variant (D-PDV) (i.e., loss-of-function or missense variant with a predicted deleterious effect, phred CADD score ≥ 20 [14]) in 113 DNA repair genes (other than BRCA1/2) previously selected [15,16] modifies the association between mammography screening exposures and BC risk.
2. Methods
2.1. Study Population
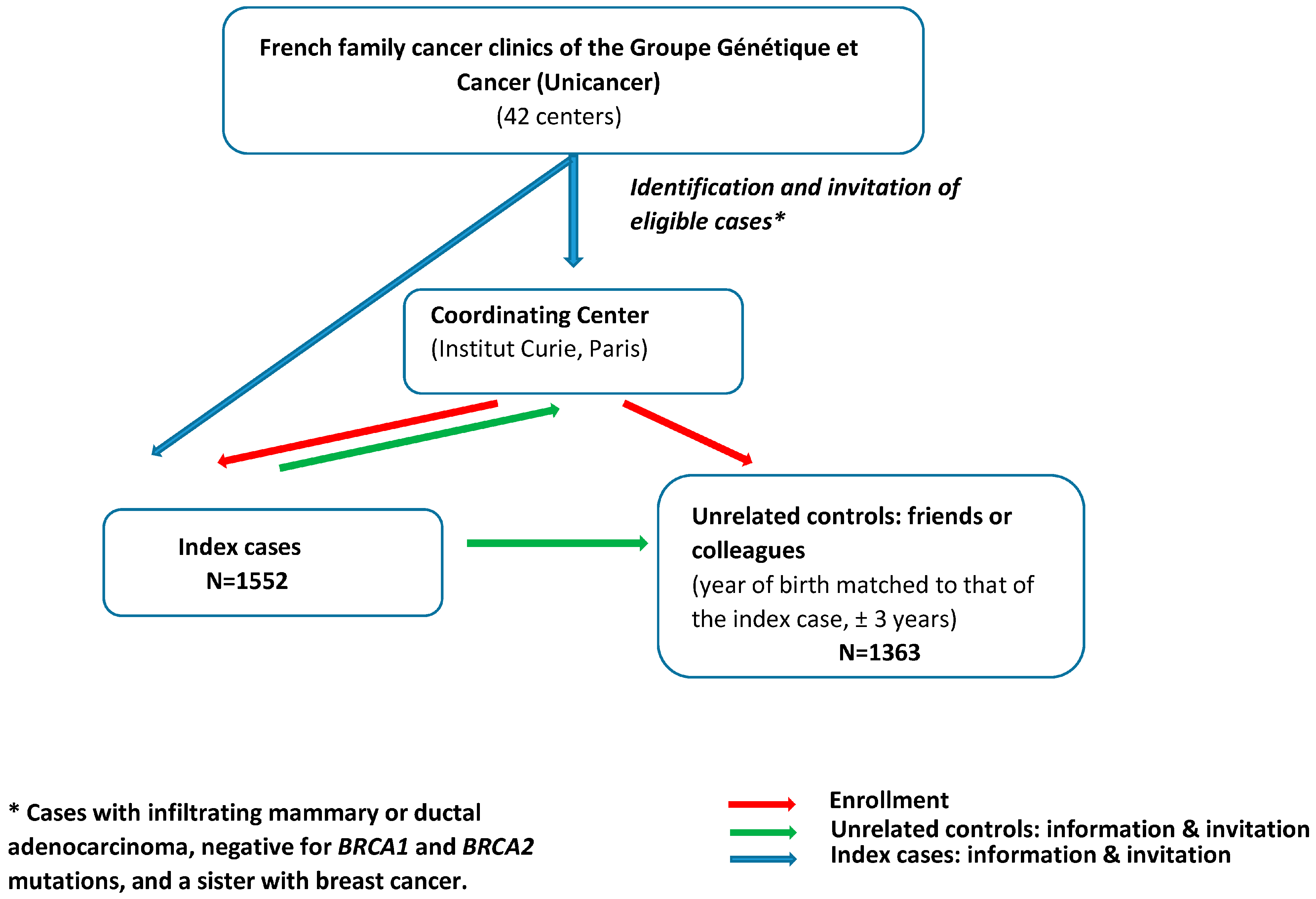
The study population involved cases and unrelated controls enrolled in GENESIS between February 2007 and December 2013, a national study initially set up to investigate genetic factors involved in the susceptibility to BC other than BRCA1/2 [17]. Index cases were identified through the French family cancer clinics of the Groupe Génétique et Cancer (Unicancer) (i.e., 42 centers) and were eligible when diagnosed with infiltrating mammary or ductal adenocarcinoma, negative for BRCA1 and BRCA2 mutations, and had a sister with BC (Figure 1). The mutation screening strategy was similar for all the clinics (cf. Supplemental Methods). The unrelated controls were selected from among the unaffected friends and/or colleagues of the cases. The year of birth of controls was matched to that of the case (±3 years). All women completed a questionnaire on environmental, lifestyle, and reproductive factors and family history of cancer. Only women reporting European ancestry (i.e., over 95% of the study population) were considered for this study.
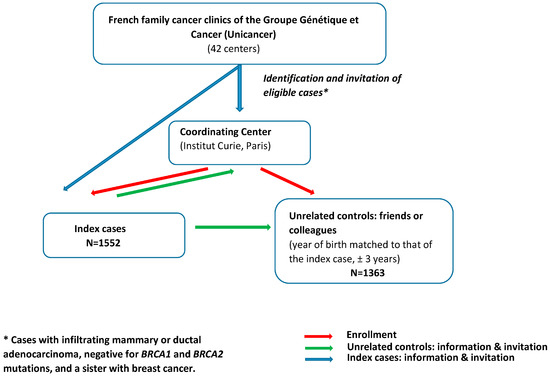
Figure 1.
Recruitment process in GENESIS study.
2.2. Screening Mammography
Participants reported their history of screening mammography in a detailed questionnaire at the time of their recruitment. To exclude mammograms that could have been performed because of BC diagnosis, we considered exposures that occurred up to one year prior to BC diagnosis for cases and one year prior to the date of questionnaire completion for controls. Variables considered in the analyses were ever vs. never exposed, lifetime number of screening mammograms, age at first mammography, and timing since the first mammography, i.e., the delay in years between the age at first mammography and the age at BC diagnosis for cases and the age at interview for the controls (i.e., age at censuring).
We excluded 52 women who underwent radiotherapy for a benign disease more than one year prior to age at censoring (2.19% cases; 1.23% controls). Among cases, we also excluded 10 women (0.63%) who underwent radiotherapy for a cancer other than BC before their BC diagnosis.
2.3. Variants in DNA Repair Genes
Contribution of germline rare D-PDVs (with minor allele frequency <0.5% in controls) in 113 DNA repair genes in familial BC was previously assessed by performing targeted sequencing of their entire coding sequence in 1207 cases and 1199 controls from the GENESIS study. Detailed information on the selection of genes, sequencing procedure, and variants filtering and annotation is described in Girard et al. [16]. Loss-of-function and missense variants with a phred CADD score ≥ 20 as a predictor of deleterious effect [14] and with minor allele frequency <0.5% in controls were selected and defined as D-PDV. Sequencing data were available for 82.5% of the women investigated in the present study, and no difference in the distribution of the characteristics such as education level, smoking, or ionizing exposures was observed between the subsets of cases and controls with and without sequencing data (see Supplemental Table 1 in Ribeiro-Guerra et al. [15]).
Because study participants carried very few rare D-PDVs per gene (pool of variants for each gene ranged between 0% and 4.1% in controls), we grouped the genes according to the value of their association with BC, i.e., the odds-ratio (OR) point estimate obtained in the study by Girard et al. [16] (whatever the degree of significance) and classified them as follows: Group “Reduced” including genes with OR < 0.9; Group “Independent”, including genes with 0.9 ≤ OR ≤ 1.1; and Group “Increased”, including genes with OR > 1.1. An individual could be assigned to more than one group if carrying several variants in genes belonging to different groups [15]. Results of the association tests per gene were first published [16]. Gene group assignation is defined in Ribeiro-Guerra et al. [15] and reported in Supplemental Table S1 to ease the reading. Additionally, we hypothesized that variants might have additive effects and built a Variant Risk Score (VRS = β1x1 + β2x2 +…+ β113x113 with β the per DNA repair gene minor allele mean effect, i.e., log OR for BC, and x equals 1 or 0 for being carrier or non-carrier, respectively) and performed analysis using the above bounds for ORs.
2.4. Statistical Analyses
To assess the association between mammography exposure and BC risk, we used logistic regression models adjusted for variables described in Supplemental Methods.
We assessed associations between mammography exposure and BC by DNA repair gene group and used likelihood ratio tests to test for heterogeneity and multiplicative interaction. Additionally, we adjusted for other gene groups when the analysis was stratified by gene group.
We assessed heterogeneity between ER tumor status using a multinomial logistic regression model and tested the equality of coefficients between equations by the difference between the log-likelihoods.
All p-values using Z-tests were two-sided, and a 5% level of significance was used. All analyses were performed using Stata software version 14 [18].
3. Results
Characteristics of the study population are described in Table 1. “Most of the cases were prevalent with a mean delay between diagnosis and interview of 8.3 years (SD: ±7.1). The mean age at BC diagnosis was 50.2 years (SD: ±9.3) and the mean age at interview for the controls was 55.8 years (SD: ±9.9)” as described in Ribeiro-Guerra et al. [15].

Table 1.
Characteristics of GENESIS participants.
Compared to controls, index cases were more likely to have a basic education level, lower body mass index, and as expected, a stronger family history of BC at censure. Regarding birth cohort, cases were more likely to be born before 1945 than controls. Among the subset of participants who had been sequenced for the 113 DNA repair genes (74.1%), 20% had no D-PDV, 31% carried 1 D-PDV, 26%, 2 D-PDVs, and 23% carried 3 or more D-PDVs. Cases were more likely to carry a variant in a gene from the “Increased” risk group than controls (57.7% and 42.6%, respectively) (Table 1). Mammography exposure characteristics are described in Table 2. More controls had mammograms than cases (91.9% and 81.9%, respectively). The mean number of exposures was similar for cases and controls, and the first exposure occurred earlier for cases than for controls (38.9 years and 43.1 years, respectively).

Table 2.
Mammography exposure characteristics.
As a whole, we did not find an association between BC and having been exposed to mammograms when measured as ever vs. never (OR = 0.99; 95% CI: 0.69–1.43) (Table 3) or when categorized according to the number of mammograms. However, when the number of mammograms was considered as a continuous variable, under the assumption of linearity, we found a significant trend in BC risk (OR = 1.04; 95% CI: 1.01–1.06) with an increased risk of 4% by additional exposure. We did not find a significant association between BC and age at the first mammogram exposure and time since first exposure although we found an OR point estimate increasing as the age at first exposure decreases from age 40 to age 30 (OR = 1.52; 95% CI: 0.91–2.53). Among exposed women, under the assumption of linearity, we found a significant decrease in BC risk of 2% per additional year for age at first exposure (95% CI: 1–4%).

Table 3.
Effect of lifetime mammography exposure (any exposure) on breast cancer risk according to the number of exposures, the age at first exposure, and time since first exposure.
When analyses were performed according to the estrogen (ER) status of the tumor (Table 4), there were no differences in ORs between ER− and ER+ tumors for almost all mammogram exposure variables except for the number of mammograms, with a significant trend observed for women with ER+ tumors (8% per additional exposure, 95% CI: 4–11%).

Table 4.
Effect of lifetime mammography exposure (any exposure) on breast cancer risk according to the number of exposures, the age at first exposure, and time since first exposure by ER status of the breast tumor.
As expected, having at least one D-PDV in a gene from Group “Reduced” is associated with a significant decrease in BC risk of 0.62 (95% CI: 0.50–0.77) compared to not having D-PDV in a gene from Group “Reduced”; having at least one D-PDV in a gene from Group “Independent” is associated with a non-significant BC risk of 1.02 (95% CI: 0.84–1.25), and having at least one D-PDV in a gene from Group “Increased” is associated with a significant increased BC risk of 1.98 (95% CI: 1.62–2.42) compared to not having D-PDV in a gene from Group “Increased” (Table 5). When stratifying women according to the group of altered genes, we found a difference in the association between having been exposed to mammograms and BC but not in the expected direction. The effect of mammograms’ exposure on BC risk was significantly higher for women carrying at least one variant in Group “Reduced” than for those carrying at least one variant in the other groups and especially in Group “Increased” (Pint = 0.02) (Table 6) (ever vs. never: “Reduced”(OR<0.9): OR = 2.17; “Independent”(0.9≤OR≤1.1): OR = 1.25; “Increased”(OR>1.1): OR = 0.92).

Table 5.
Main effect of variant carrier status on breast cancer.

Table 6.
Effect of lifetime mammography exposure (any exposure) on breast cancer risk according to the number of exposures, the age at first exposure, and time since first exposure by DNA repair gene group.
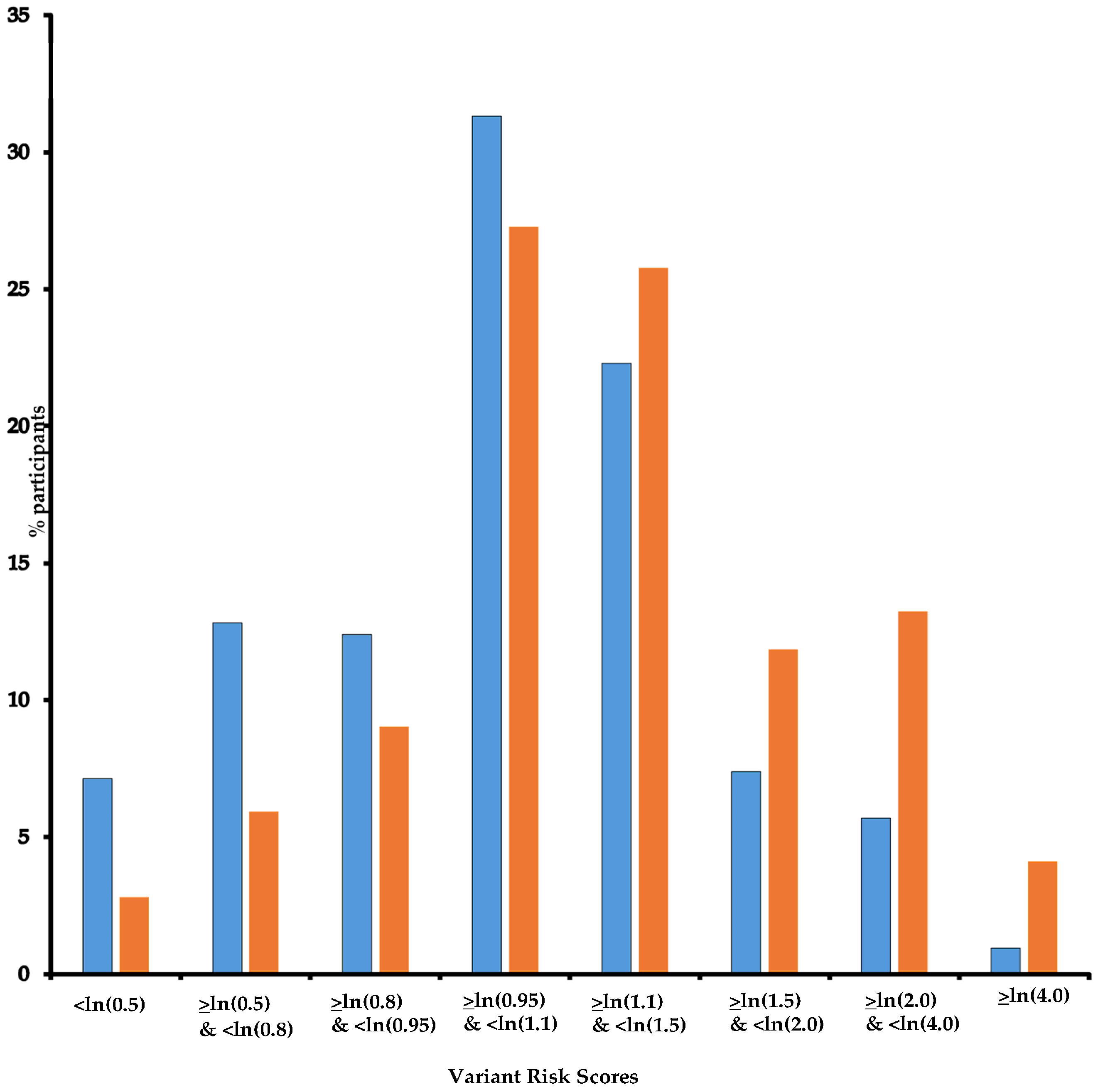
In the three groups, there was no significant association with the categorized number of mammograms although a significant trend was found, with an increased risk of 6% (95% CI: 2–10%) by additional exposure for both Group “Independent” and Group “Increased”. Additionally, we found significant multiplicative interactions for Group “Increased” (Pint = 0.020 and 0.019 for 1 to 4 and 5 to 9 exposures, respectively). Having been first exposed before age 30 increased the risk of BC significantly only in Group “Reduced” with an OR of 3.16 (95% CI: 1.01–9.89). For the three other age classes at first exposure, we found significant interactions for Group “Increased” (Pint = 0.019, 0.021, and 0.057, respectively). As for the whole study population, we did not find an association between BC and time since first exposure in any gene group. Additionally, as ~80% of participants carried more than one D-PDV (Table 1) and could be categorized into more than one DNA repair gene group, we hypothesized that variants might have additive effects and built a DNA repair VRS (Figure 2) and estimated the risk of BC per quintile of VRS. As expected, BC risk increased as VRS increased (Table 7). When stratifying women according to VRS quintiles, results for association between mammograms and BC confirmed the trend observed in the previous analyses, i.e., a decrease in BC risk associated with mammograms as DNA repair VRS increased (<ln(0.8): OR = 2.56; ≥ln(0.8) & <ln(0.95): OR = 3.88; ≥ln(0.95) & <ln(1.1): OR = 2.29; ≥ln(1.1) & <ln(1.5): OR = 1.24; ≥ln(1.5): OR = 0.30; Pint = 0.006) (Table 7). The same trend is observed as VRS increased when first exposed after age 40 or before age 40, but with point estimates higher for the latest group of women (Table 7).
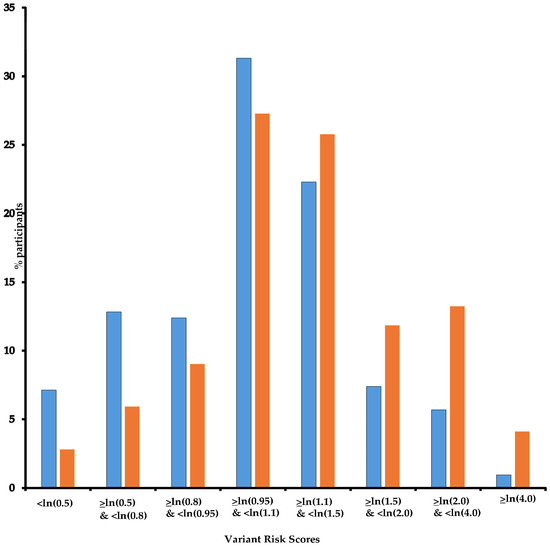
Figure 2.
DNA repair Variant Score Risk distribution. Legend: Horizontal axis: Variant Risk Scores; vertical axis: % participants; Blue: controls; Orange: cases. Note: For each participant, a Variant Risk Score (VRS = β1x1 + β2x2 +…+ β113x113) was built with βi per DNA repair gene minor allele mean effect, i.e., ln odds ratio (OR) for breast cancer, and xi equals 1 or 0 for being carrier of the i-th DNA repair gene minor allele or not, respectively.

Table 7.
Effect of mammography exposure according to the DNA repair Variant Score Risk.
Because we used a priori bounds for ORs for defining the DNA repair gene groups, we performed several sensitivity analyses using different bounds, and we found similar trends in the difference in the BC risk between groups (Supplemental Table S2).
We also performed a sensitivity analysis that included in the “Increased” Group only the genes that were found significantly (or borderline) associated with BC in our population [16] (i.e., ATM, CHEK2, PALB2, FANCM, MAST1) or confirmed as pathogenic or predicted to be pathogenic for BC by international consortia [19,20] (i.e., PALB2, TP53, CHEK2, PTEN, ATM, BARD1, MSH6, RAD51C, RAD51D). Only a few participants had never been exposed to mammography among those D-PDV carriers (28 and 24, respectively), and similar point estimates are however observed (ever vs. never: OR = 0.32; 95% CI: 0.07–1.54 and OR = 0.22; 95% CI: 0.04–1.23, respectively).
Finally, the magnitude and direction of the effect estimates based on analyses using an extra class for missing data or a multiple imputation strategy were similar (Table 3 and Table 8, Supplemental Tables S2 and S3).

Table 8.
Effect of lifetime mammography exposure (any exposure) on breast cancer risk according to the number of exposures, the age at first exposure, and time since first exposure by DNA repair gene group (Multiple Imputation).
4. Discussion
Overall, we found that mammography exposure, measured as ever vs. never, is not associated with the risk of developing a BC in women with a hereditary predisposition to BC unexplained by a BRCA1 or BRCA2 pathogenic variant. However, this risk increases with the number of mammograms, with an increase of 4% for each additional exposure. An increased risk is also suggested when first exposure occurs before age 40. To our knowledge, only three studies investigated diagnostic radiation exposure to the chest among women with a hereditary predisposition to BC and almost all without BRCA1/2 pathogenic variant [5,15,21], and only one considered mammograms by assessing the effect of age at first exposure and did not find evidence for an increased BC risk for women exposed before 37 years of age compared to women first exposed between the ages of 40 and 46 years [21]. Only four studies assessed the effect of a history of mammography among BRCA1/2 pathogenic variant carriers [2,4,9,10,11,12,13] and only one found an increased point estimate with a hazard ratio of 1.43 for women first exposed before age 30 [2], which is in line with our findings.
Surprisingly, our findings suggested that radiation delivered by mammograms double the risk of BC in women carrying a D-PDV in a DNA repair gene negatively associated with the risk of BC, while such an exposure appears not to be associated with BC in women carrying a D-PDV in a gene associated with an increased risk of BC. This finding is counterintuitive and not in line with our previous study [15], where we found a difference in the association between having been exposed to chest X-ray and BC with a significantly higher risk for women carrying at least one D-PDV in DNA repair genes associated with an increased risk of BC.
A possible weakness of our study is the use of nonrandomly selected friends or colleagues as controls. However, this approach offered the advantage of making it easier to identify suitable controls compared to random selection from the general population, and a higher comparability for unmeasured factors with, however, a likely indication bias for mammograms. Indeed, a strong family history of BC could be an indication for mammographic screening, especially at a young age. We investigated this potential bias away from the null by looking at the distribution of the family history by category of age at first exposure and did not find difference (chi2 = 23.3, df = 23, p = 0.438) (Supplemental Table S4).
However, to take into account potential confounders and indication biases, we adjusted the analyses for numerous variables like age at censure, birth cohort, being eligible for national screening, and the number of BC in the family at censure.
Because the mammography technique has evolved over time, as has the level of dose received for each image, we are aware that an estimation of the doses received at each examination according to when and where the examination was carried out would have generated more precise exposure data. However, retrospective studies are not able to trace and standardize mammography procedures through medical record reviews because of the difficulties in accessing medical records. For this reason, information on lifetime mammograms’ exposures was self-reported with accompanying potential recall biases and exposure misclassification (see also Supplemental Table S6). Therefore, even if some studies have shown that self-reported mammography data were widely reliable for epidemiologic research [22,23], we cannot totally rule out such a bias. Thus, to avoid the potential effect of exposure misclassification, indication, and recall biases, we performed case-only analyses and assessed the interaction effects between the groups of D-PDVs and mammograms’ exposure (under the assumption of independence). We found an ORinteraction = 1.62 (p = 0.10) between mammograms’ exposure and DNA repair genes potentially decreasing BC risk, ORinteraction = 0.84 (p = 0.48) between mammograms’ exposure and those with no effect, and ORinteraction = 0.68 (p = 0.14) between mammograms exposure and those with increasing BC risk). Even though not statistically significant, these results are in line with the observed trend in our case–control analyses.
One strength of our study is that it was conducted in a homogeneous sample of high-risk women and population controls with detailed lifetime information on mammograms. Our study is unique in that sequencing data for 113 DNA repair genes are available for an important subset of the study population [16]. Indeed, our study is the first to investigate not only the effect of mammograms but also the joint effect of mammogram exposure and D-PDVs in DNA repair genes in women at high risk of BC. We cannot exclude potential biases first due to the classification of the genes according to the ORs calculated in the same population and second due to a classification based on point estimates with, for many of them, insufficient power to characterize precisely their effects. However, the latter potential misclassification should be differential according to gene groups to lead to the observed differential association with mammograms, which is unlikely. Moreover, defining the group of genes with a point estimate falling in a large range of ORs around 1 as the group of genes “not associated with BC” might have an impact on the findings. Therefore, we performed numerous sensitivity analyses performing case-only analysis, changing the boundaries and assuming additivity of the effects of genes, and we found similar trends in the difference in the BC risk between groups.
Finally, the observed difference in the association between mammography and BC risk between the DNA repair gene groups cannot be explained by potential differences in characteristics like age at the diagnostic or educational level or even the number of BC in the family at censure (Supplemental Table S5).
Our exploratory findings showed that mammogram exposure (ever vs. never) does not increase BC risk, even among women carrying a rare D-PDV in a DNA repair gene potentially increasing BC risk, except when first exposure occurred before age 30. Surprisingly, a D-PDV in a DNA repair gene potentially decreasing BC risk may increase sensibility to mammograms’ exposure.
The evaluation of low doses in epidemiological studies is challenged by many potential limitations like lack of statistical power, unheeded time dependency, misclassification in exposure assessment, inappropriate evaluation of confounding risk factors, and failure to take hormesis into account [24]. Therefore, we acknowledge that both the lifetime numbers of mammograms as a surrogate for X-ray doses received at breasts and the potential misclassification of the D-PDVs might weaken our observations. However, our study is the first to investigate the joint effect of mammogram exposure and D-PDV in DNA worth checking our findings in further studies on larger populations, ideally with prospectively recorded detailed exposure data and evaluating other genes that could modify radiation sensitivity.
5. Conclusions
Our results showed that mammogram exposure (ever vs. never) does not increase BC risk, even among women carrying a D-PDV in a DNA repair gene potentially increasing BC risk, except when first exposure occurred before age 30. Surprisingly, D-PDVs in a DNA repair gene potentially decreasing BC risk may increase sensitivity to mammograms’ exposure. Further studies on larger populations are needed to verify our findings and to evaluate other genes that could modify radiation sensitivity. Even though the current data are clear in demonstrating that mammographic screening thanks to early diagnosis significantly reduces the risk of mortality from BC [25,26], identification of sub-populations that are more or less susceptible to ionizing radiation is much more important and clinically relevant.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/cancers17071062/s1, Supplemental methods. Table S1. Association of rare coding variants with breast cancer, for the 113 DNA repair genes sequenced in the GENESIS population [15,16]. Table S2. Sensitivity analyses with varying bounds of ORs for the definition of genetic variant group: effect of lifetime mammography exposure (any exposure) on breast cancer risk according to the number of exposures, the age at first exposure and time since first exposure. Table S3. Sensitivity analyses with varying bounds of ORs for the definition of genetic variant group: effect of lifetime mammography exposure (any exposure) on breast cancer risk according to the number of exposures, the age at first exposure and time since first exposure (Multiple Imputation). Table S4. Number of breast cancer in the family at censure according to age at mammography first exposure. Table S5. Characteristics of GENESIS participants according to DNA repair gene groups. Table S6. Cross table between lifetime number of mammograms and lifetime number of views. References [18,27,28,29,30] are cited in the Supplementary Materials.
Author Contributions
Conceptualization, N.A.; data curation, M.R.-G. and M.-G.D.; formal analysis, M.R.-G. and N.A.; funding acquisition, F.L., D.S.-L. and N.A.; investigation, S.E.-M., D.L.G., J.B., N.M., M.B. and E.C.; methodology, N.A.; resources, C.A.-L., S.A.-B., P.B., V.B., B.B., O.C., M.C., J.C., C.C., I.C., C.D., H.D., A.F., S.F.-F., J.-P.F., M.G.-V., P.G., S.G., L.G., C.L., S.L.-D., J.-M.L., M.L., A.L., E.L., C.M.M., I.M., S.N., C.N., P.P., L.V.-B., F.S., J.T., A.T. and D.S.-L.; supervision, N.A.; writing—original draft, M.R.-G. and N.A.; writing—review and editing, M.R.-G., M.-G.D., F.L., D.S.-L. and N.A. All authors have read and agreed to the published version of the manuscript.
Funding
Brazilian National Council for the Improvement of Higher Education—CAPES, for the fellowship (process number: BEX 5852/15-3) to M.R. Guerra. Financial support for GENESIS was provided by the Ligue Nationale contre le Cancer (3 grants: PRE05/DSL and PRE07/DSL to D. Stoppa-Lyonnet; PRE11/NA to N. Andrieu), the French National Institute of Cancer (INCa, Grant b2008-029/LL-LC), and the comprehensive cancer center SiRIC (Site de Recherche Intégrée sur le Cancer: Grant INCa-DGOS-4654) to N. Andrieu. Sequencing was supported by the France Génomique National infrastructure, funded as part of the «Investissements d’Avenir» program managed by the Agence Nationale pour la Recherche (ANR-10-INBS-09), the Centre National de Recherche en Génomique Humaine (CNRGH), CEA, the Ligue Comité de Paris (grant RS15/75-78), and the Fondation ARC pour la recherche sur le cancer (grant PJA 20151203365).
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the appropriate ethics committee (Comité de Protection des Personnes Ile-de-France III, 3 October 2006, agreement no. 2373).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data underlying this article will be shared upon reasonable request to the corresponding author.
Acknowledgments
We wish to thank the genetic epidemiology platform (the PIGE, Plateforme d’Investigation en Génétique et Epidémiologie: S. Eon-Marchais, M. Marcou, D. Le Gal, L. Toulemonde, J. Beauvallet, N. Mebirouk, E. Cavaciuti, A. Fescia), the biological resource center (C. Verny-Pierre, L. Barjhoux, V. Sornin, N. Mebirouk, F. Lesueur), and all the GENESIS collaborating cancer clinics (Clinique Sainte Catherine, Avignon: H. Dreyfus; Hôpital Saint Jacques, Besançon: M-A. Collonge-Rame; Institut Bergonié, Bordeaux: M. Longy, A. Floquet, E. Barouk-Simonet; CHU, Brest: S. Audebert; Centre François Baclesse, Caen: P. Berthet; Hôpital Dieu, Chambéry: S. Fert-Ferrer; Centre Jean Perrin, Clermont-Ferrand: Y-J. Bignon; Hôpital Pasteur, Colmar: J-M. Limacher; Hôpital d’Enfants CHU—Centre Georges François Leclerc, Dijon: L. Faivre-Olivier; CHU, Fort de France: O. Bera; CHU Albert Michallon, Grenoble: D. Leroux; Hôpital Flaubert, Le Havre: V. Layet; Centre Oscar Lambret, Lille: P. Vennin †, C. Adenis; Hôpital Jeanne de Flandre, Lille: S. Lejeune-Dumoulin, S. Manouvier-Hanu; CHRU Dupuytren, Limoges: L. Venat-Bouvet; Centre Léon Bérard, Lyon: C. Lasset, V. Bonadona; Hôpital Edouard Herriot, Lyon: S. Giraud; Institut Paoli-Calmettes, Marseille: F. Eisinger, L. Huiart; Centre Val d’Aurelle—Paul Lamarque, Montpellier: I. Coupier; CHU Arnaud de Villeneuve, Montpellier: I. Coupier, P. Pujol; Centre René Gauducheau, Nantes: C. Delnatte; Centre Catherine de Sienne, Nantes: A. Lortholary; Centre Antoine Lacassagne, Nice: M. Frénay, V. Mari; Hôpital Caremeau, Nîmes: J. Chiesa; Réseau Oncogénétique Poitou Charente, Niort: P. Gesta; Institut Curie, Paris: D. Stoppa-Lyonnet, M. Gauthier-Villars, B. Buecher, A. de Pauw, C. Abadie, M. Belotti; Hôpital Saint-Louis, Paris: O. Cohen-Haguenauer; Centre Viggo-Petersen, Paris: F. Cornélis; Hôpital Tenon, Paris: A. Fajac; GH Pitié Salpétrière et Hôpital Beaujon, Paris: C. Colas, F. Soubrier, P. Hammel, A. Fajac; Institut Jean Godinot, Reims: C. Penet, T. D. Nguyen; Polyclinique Courlancy, Reims: L. Demange †, C. Penet; Centre Eugène Marquis, Rennes: C. Dugast; Centre Henri Becquerel, Rouen: A. Chevrier, T. Frebourg ††, J. Tinat, I. Tennevet, A. Rossi; Hôpital René Huguenin/Institut Curie, Saint Cloud: C. Noguès, L. Demange †, E. Mouret-Fourme; CHU, Saint-Etienne: F. Prieur; Centre Paul Strauss, Strasbourg: J-P. Fricker, H. Schuster; Hôpital Civil, Strasbourg: O. Caron, C. Maugard; Institut Claudius Regaud, Toulouse: L. Gladieff, V. Feillel; Hôpital Bretonneau, Tours: I. Mortemousque; Centre Alexis Vautrin, Vandoeuvre-les-Nancy: E. Luporsi; Hôpital de Bravois, Vandoeuvre-les-Nancy: P. Jonveaux; Gustave Roussy, Villejuif: A. Chompret †, O. Caron). We wish to pay tribute to Olga M. Sinilnikova who was one of the initiators and principal investigators of GENESIS and who died prematurely on 30 June 2014. † Deceased prematurely. †† Suddenly passed away on 13 March 2021.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| BC | breast cancer |
| BRCA1/2 | BRCA1 or BRCA2 |
| CI | confidence interval |
| D-PDV | deleterious or predicted deleterious variant |
| DNA | deoxyribonucleic acid |
| OR | odds ratio |
| VRS | variant risk score |
References
- IARC Working Group on the Evaluation of Cancer-Preventive Strategies. Breast Cancer Screening; World Health Organization: Geneva, Switzerland, 2016.
- Pijpe, A.; Andrieu, N.; Easton, D.F.; Kesminiene, A.; Cardis, E.; Noguès, C.; Gauthier-Villars, M.; Lasset, C.; Fricker, J.P.; Peock, S.; et al. Exposure to diagnostic radiation and risk of breast cancer among carriers of BRCA1/2 mutations: Retrospective cohort study (GENE-RAD-RISK). BMJ 2012, 345, e5660. [Google Scholar] [PubMed]
- Andrieu, N.; Easton, D.F.; Chang-Claude, J.; Rookus, M.A.; Brohet, R.; Cardis, E.; Antoniou, A.C.; Wagner, T.; Simard, J.; Evans, G.; et al. Effect of chest X-rays on the risk of breast cancer among BRCA1/2 mutation carriers in the international BRCA1/2 carrier cohort study: A report from the EMBRACE, GENEPSO, GEO-HEBON, and IBCCS Collaborators’ Group. J. Clin. Oncol. 2006, 24, 3361–3366. [Google Scholar] [PubMed]
- Gronwald, J.; Pijpe, A.; Byrski, T.; Huzarski, T.; Stawicka, M.; Cybulski, C.; van Leeuwen, F.; Lubiński, J.; Narod, S.A. Early radiation exposures and BRCA1-associated breast cancer in young women from Poland. Breast Cancer Res. Treat. 2008, 112, 581–584. [Google Scholar]
- John, E.M.; Phipps, A.I.; Knight, J.A.; Milne, R.L.; Dite, G.S.; Hopper, J.L.; Andrulis, I.L.; Southey, M.; Giles, G.G.; West, D.W.; et al. Medical radiation exposure and breast cancer risk: Findings from the Breast Cancer Family Registry. Int. J. Cancer 2007, 121, 386–394. [Google Scholar]
- John, E.M.; McGuire, V.; Thomas, D.; Haile, R.; Ozcelik, H.; Milne, R.L.; Felberg, A.; West, D.W.; Miron, A.; Knight, J.A.; et al. Diagnostic chest X-rays and breast cancer risk before age 50 years for BRCA1 and BRCA2 mutation carriers. Cancer Epidemiol. Biomarkers Prev. 2013, 22, 1547–1556. [Google Scholar]
- Lecarpentier, J.; Noguès, C.; Mouret-Fourme, E.; Stoppa-Lyonnet, D.; Lasset, C.; Caron, O.; Fricker, J.P.; Gladieff, L.; Faivre, L.; Sobol, H.; et al. Variation in breast cancer risk with mutation position, smoking, alcohol, and chest X-ray history, in the French National BRCA1/2 carrier cohort (GENEPSO). Breast Cancer Res. Treat. 2011, 130, 927–938. [Google Scholar]
- Geoffroy-Perez, B.; Janin, N.; Ossian, K.; Laugé, A.; Stoppa-Lyonnet, D.; Andrieu, N. Variation in breast cancer risk of heterozygotes for ataxia-telangiectasia according to environmental factors. Int. J. Cancer 2002, 99, 619–623. [Google Scholar] [CrossRef]
- Giannakeas, V.; Lubinski, J.; Gronwald, J.; Moller, P.; Armel, S.; Lynch, H.T.; Foulkes, W.D.; Kim-Sing, C.; Singer, C.; Neuhausen, S.L.; et al. Mammography screening and the risk of breast cancer in BRCA1 and BRCA2 mutation carriers: A prospective study. Breast Cancer Res. Treat. 2014, 147, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Narod, S.A.; Lubinski, J.; Ghadirian, P.; Lynch, H.T.; Moller, P.; Foulkes, W.D.; Rosen, B.; Kim-Sing, C.; Isaacs, C.; Domchek, S.; et al. Screening mammography and risk of breast cancer in BRCA1 and BRCA2 mutation carriers: A case-control study. Lancet Oncol. 2006, 7, 402–406. [Google Scholar]
- Berrington de Gonzalez, A.; Berg, C.D.; Visvanathan, K.; Robson, M. Estimated risk of radiation-induced breast cancer from mammographic screening for young BRCA mutation carriers. J. Natl. Cancer Inst. 2009, 101, 205–209. [Google Scholar]
- Goldfrank, D.; Chuai, S.; Bernstein, J.L.; Ramon, Y.; Cajal, T.; Lee, J.B.; Alonso, M.C.; Diez, O.; Baiget, M.; Kauff, N.D.; et al. Effect of mammography on breast cancer risk in women with mutations in BRCA1 or BRCA2. Cancer Epidemiol. Biomark. Prev. 2006, 15, 2311–2313. [Google Scholar]
- Canet, M.L.; Harbron, R.; Thierry-Chef, I.; Cardis, E. Cancer effects of low to moderate doses of ionising radiation in young people with cancer predisposing conditions: A systematic review. Cancer Epidemiol. Biomark. Prev. 2022, 31, 1871–1889. [Google Scholar]
- Kircher, M.; Witten, D.M.; Jain, P.; O’Roak, B.J.; Cooper, G.M.; Shendure, J. A general framework for estimating the relative pathogenicity of human genetic variants. Nat. Genet. 2014, 46, 310–315. [Google Scholar]
- Ribeiro, G.M.; Coignard, J.; Eon-Marchais, S.; Dondon, M.G.; Le Gal, D.; Beauvallet, J.; Mebirouk, N.; Belotti, M.; Caron, O.; Gauthier-Villars, M.; et al. Diagnostic chest X-rays and breast cancer risk among women with a hereditary predisposition to breast cancer unexplained by a BRCA1 or BRCA2 mutation. Breast Cancer Res. 2021, 23, 79. [Google Scholar] [CrossRef]
- Girard, E.; Eon-Marchais, S.; Olaso, R.; Renault, A.L.; Damiola, F.; Dondon, M.G.; Barjhoux, L.; Goidin, D.; Meyer, V.; Le Gal, D.; et al. Familial breast cancer and DNA repair genes: Insights into known and novel susceptibility genes from the GENESIS study, and implications for multigene panel testing. Int. J. Cancer 2019, 144, 1962–1974. [Google Scholar]
- Sinilnikova, O.M.; Dondon, M.G.; Eon-Marchais, S.; Damiola, F.; Barjhoux, L.; Marcou, M.; Verny-Pierre, C.; Sornin, V.; Toulemonde, L.; Beauvallet, J.; et al. GENESIS: A French national resource to study the missing heritability of breast cancer. BMC. Cancer 2016, 16, 13. [Google Scholar]
- StataCorp. Stata: Release 14. Statistical Software (2015); StataCorp LP: College Station, TX, USA, 2015. [Google Scholar]
- Breast Cancer Association Consortium; Dorling, L.; Carvalho, S.; Allen, J.; González-Neira, A.; Luccarini, C.; Wahlström, C.; Pooley, K.A.; Parsons, M.T.; Fortuno, C.; et al. Breast Cancer Risk Genes—Association Analysis in More than 113,000 Women. N. Engl. J. Med. 2021, 384, 428–439. [Google Scholar]
- Hu, C.; Hart, S.N.; Gnanaolivu, R.; Huang, H.; Lee, K.Y.; Na, J.; Gao, C.; Lilyquist, J.; Yadav, S.; Boddicker, N.J.; et al. Population-Based Study of Genes Previously Implicated in Breast Cancer. N. Engl. J. Med. 2021, 384, 440–451. [Google Scholar]
- Evans, D.G.; Kotre, C.J.; Harkness, E.; Wilson, M.; Maxwell, A.J.; Howell, A. No strong evidence for increased risk of breast cancer 8–26 years after multiple mammograms in their 30s in females at moderate and high familial risk. Br. J. Radiol. 2016, 89, 20150960. [Google Scholar] [CrossRef] [PubMed]
- Pijpe, A.; Mulder, R.L.; Manders, P.; Van Leeuwen, F.E.; Rookus, M.A. Validation study suggested no differential misclassification of self-reported mammography history in BRCA1/2 mutation carriers. J. Clin. Epidemiol. 2011, 64, 1434–1443. [Google Scholar]
- Walker, M.J.; Chiarelli, A.M.; Mirea, L.; Glendon, G.; Ritvo, P.; Andrulis, I.L.; Knight, J.A. Accuracy of Self-Reported Screening Mammography Use: Examining Recall among Female Relatives from the Ontario Site of the Breast Cancer Family Registry. Int. Sch. Res. Not. 2013, 2013, 810573. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Fontana, L.; Santocono, C.; Guarino, D.; Laudiero, M.; Calabrese, E.J. The challenges of defining hormesis in epidemiological studies: The case of radiation hormesis. Sci. Total Environ. 2023, 902, 166030. [Google Scholar] [CrossRef] [PubMed]
- Dauer, L.T.; Brooks, A.L.; Hoel, D.G.; Morgan, W.F.; Stram, D.; Tran, P. Review and evaluation of updated research on the health effects associated with low-dose ionising radiation. Radiat. Prot. Dosim. 2010, 140, 103–136. [Google Scholar] [CrossRef]
- Lewanski, C.R.; Gullick, W.J. Radiotherapy and cellular signalling. Lancet Oncol. 2001, 2, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Van Buuren, S.; Boshuizen, H.C.; Knook, D.L. Multiple imputation of missing blood pressure covariates in survival analysis. Stat. Med. 1999, 18, 681–694. [Google Scholar] [CrossRef]
- Raghunathan, T.E. What do we do with missing data? Some options for analysis of incomplete data. Annu. Rev. Public Health 2004, 25, 99–117. [Google Scholar] [CrossRef]
- Little, R.J.A.; Rubin, D.B. Statistical Analysis with Missing Data, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2002. [Google Scholar]
- Rubin, D.B.; Schenker, N. Multiple imputation in health-care databases: An overview and some applications. Stat. Med. 1991, 10, 585–598. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).