Biomarkers and Management of Cholangiocarcinoma: Unveiling New Horizons for Precision Therapy
Simple Summary
Abstract
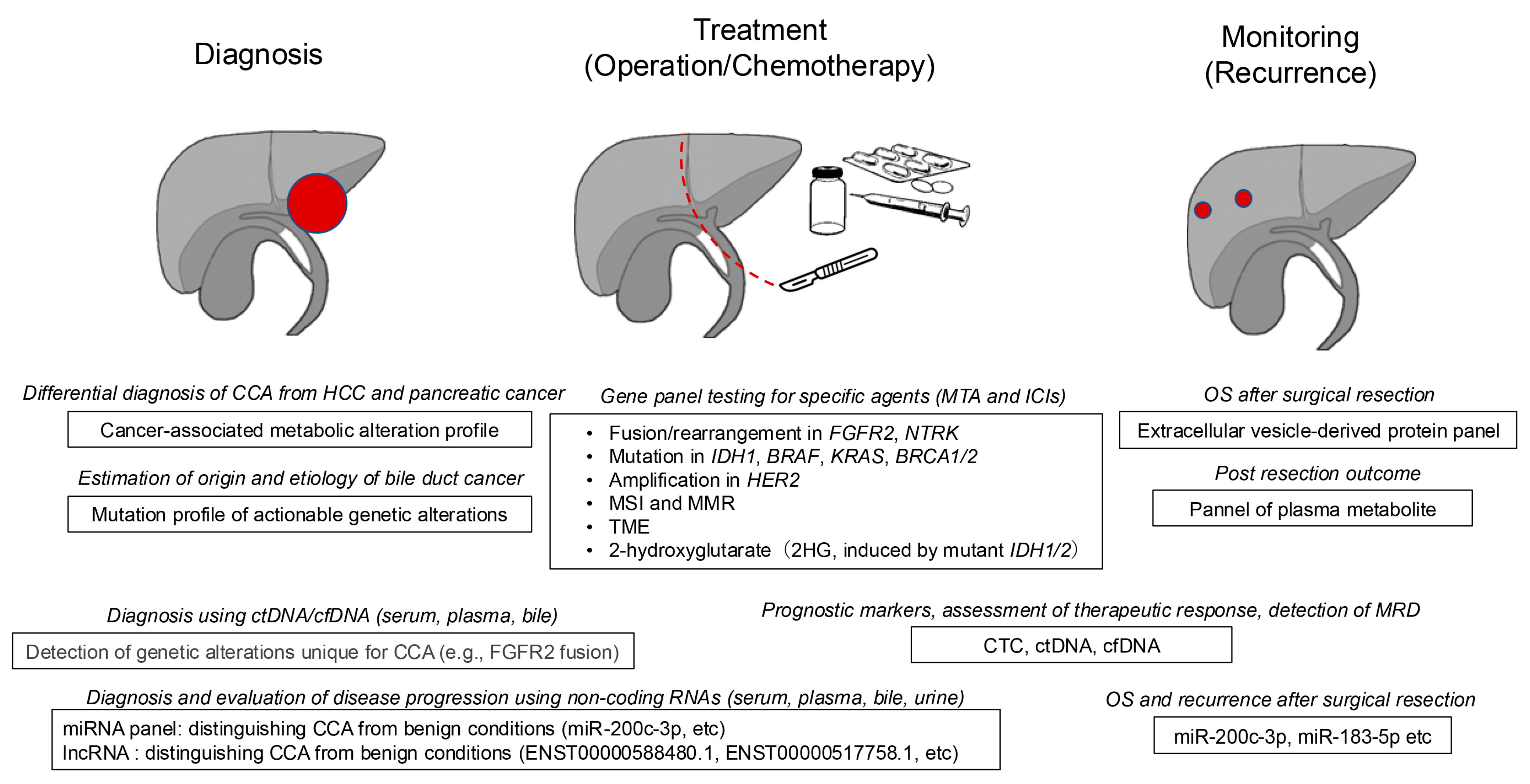
1. Introduction
2. Serum and Plasma Biomarkers for Cholangiocarcinoma
2.1. Conventional Serum and Plasma Biomarkers
2.2. Serum and Plasma Biomarkers Under Development
2.3. Cancer-Associated Metabolites
3. Genetic Aberrations in Cholangiocarcinoma
3.1. Etiologies, Pathologies, and Genetic Alterations in the Small-Duct Type of Intrahepatic Cholangiocarcinoma
3.2. Etiologies, Pathologies, and Genetic Alterations in the Large-Duct Type of Intrahepatic, Perihilar, and Distal Cholangiocarcinoma
3.3. Cancer Panels and Companion Diagnostics for Cholangiocarcinoma
4. Molecular-Targeted Therapies and Biomarkers
4.1. FGFR2 Gene Fusions/Rearrangements
4.2. Mutations in the IDH Gene
4.3. Activating Mutations in the KRAS and BRAF Genes
4.4. HER2 Gene Amplification/Overexpression
4.5. Other Biomarkers for Molecular-Targeted Agents
5. Emerging Biomarkers and Their Future Perspectives
5.1. Tumor Cells and Cell-Free DNA in Peripheral Blood
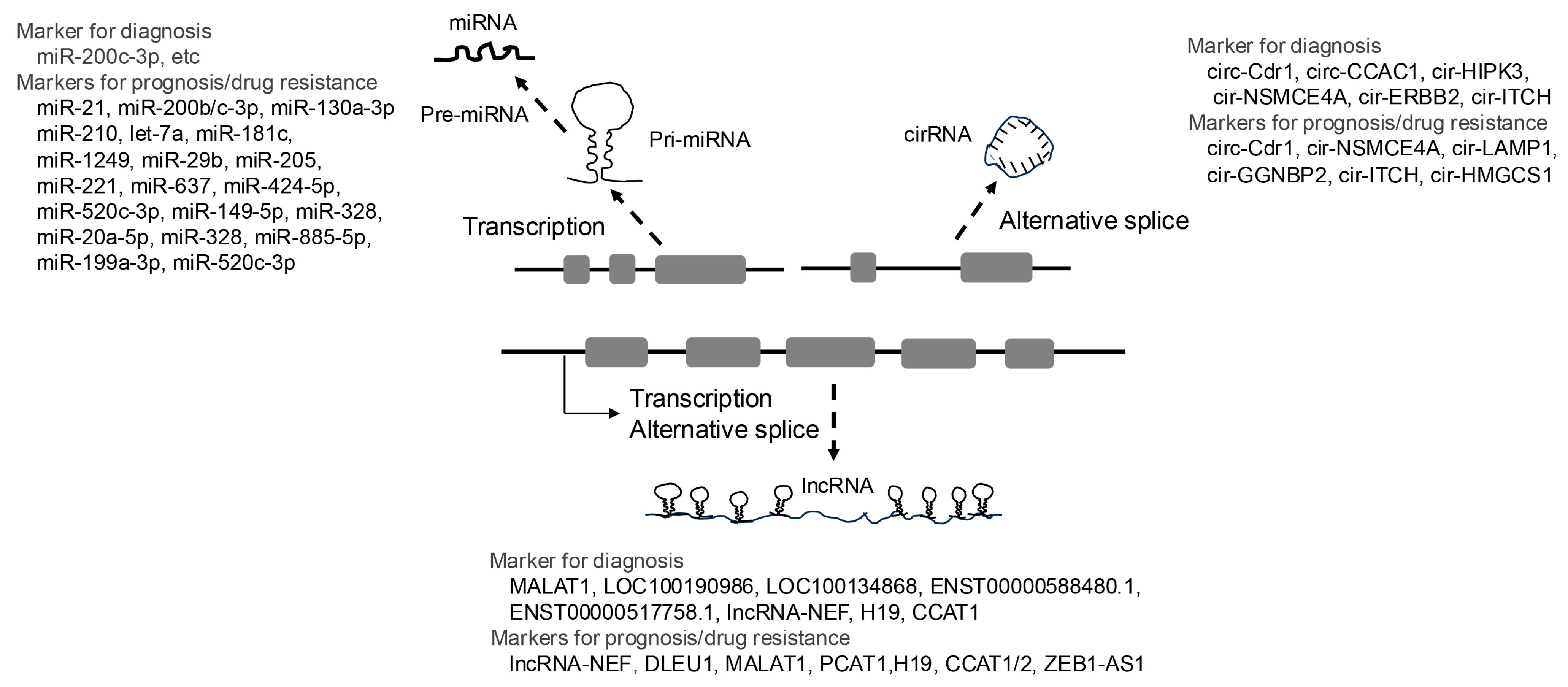
5.2. Non-Coding RNAs
5.2.1. MicroRNAs
5.2.2. LncRNAs
5.2.3. CircRNAs
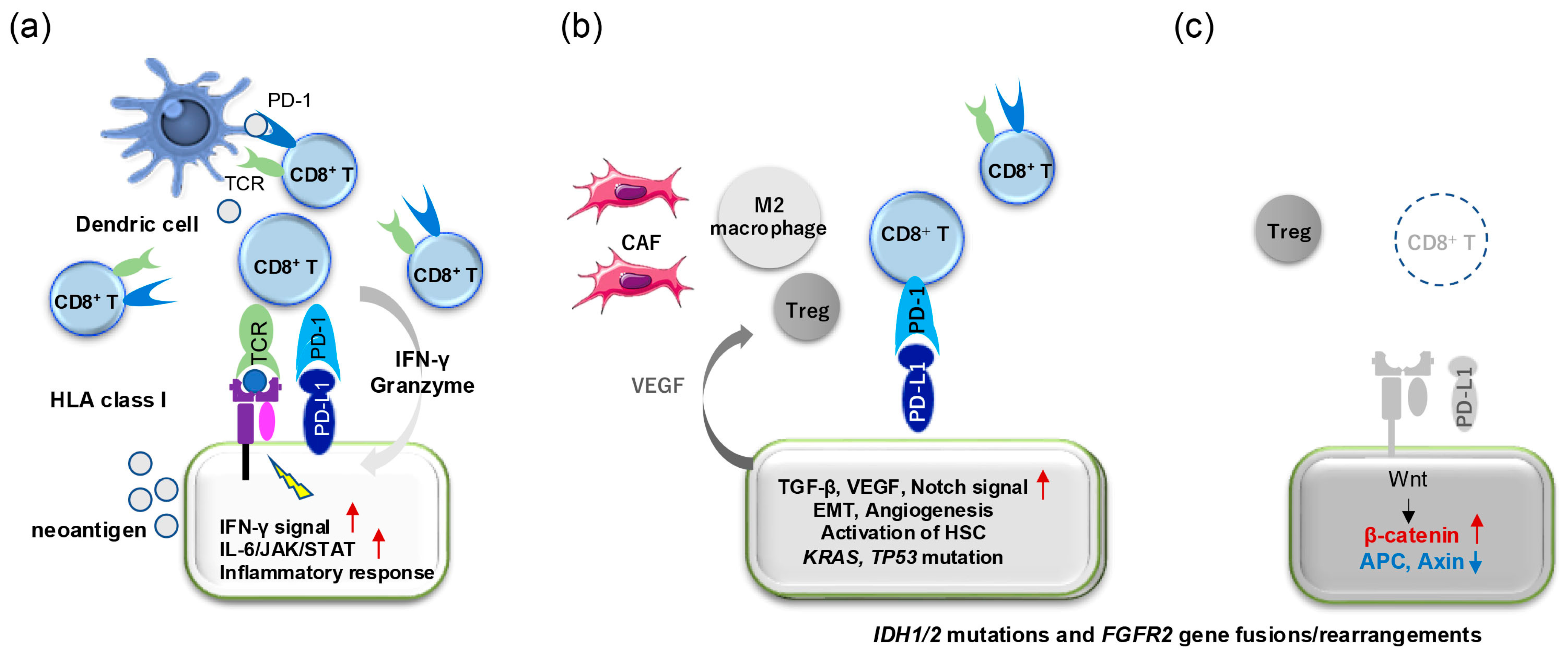
6. Tumor Immune Microenvironment and Treatment Using Immune Checkpoint Inhibitors
7. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| CA19-9 | Carbohydrate antigen 19-9 |
| CCA | Cholangiocarcinoma |
| CEA | Carcinoembryonic antigen |
| cfDNA | Cell-free DNA |
| ctDNA | Circulating tumor DNA |
| dCCA | Distal CCA |
| CA19-9 | Carbohydrate antigen 19-9 |
| CTCs | Circulating tumor cells |
| DDR | DNA damage response |
| dMMR | Mismatch repair deficiency |
| EpCAM | Epithelial cell adhesion molecules |
| HBV | Hepatitis B virus |
| HCCs | Hepatocellular carcinomas |
| iCCA | Intrahepatic CCA |
| ICIs | Immune checkpoint inhibitors |
| IL-6 | Interleukin-6 |
| MMP-7 | Matrix metalloproteinase-7 |
| MTAs | Molecular-targeted agents |
| NGS | Next-generation sequencing |
| ORR | Objective response rate |
| OS | Overall survival |
| PARP | Poly ADP-ribose polymerase |
| PFS | Progression-free survival |
| TILs | Tumor-infiltrating lymphocytes |
| TME | Tumor immune microenvironment |
References
- Knox, J.J.; McNamara, M.G.; Bazin, I.S.; Oh, D.Y.; Zubkov, O.; Breder, V.; Bai, L.Y.; Christie, A.; Goyal, L.; Cosgrove, D.P.; et al. A phase III randomised study of first-line NUC-1031/cisplatin vs. gemcitabine/cisplatin in advanced biliary tract cancer. J. Hepatol. 2025; in press. [Google Scholar]
- Shroff, R.T.; King, G.; Colby, S.; Scott, A.J.; Borad, M.J.; Goff, L.; Matin, K.; Mahipal, A.; Kalyan, A.; Javle, M.M.; et al. SWOG S1815: A Phase III Randomized Trial of Gemcitabine, Cisplatin, and Nab-Paclitaxel Versus Gemcitabine and Cisplatin in Newly Diagnosed, Advanced Biliary Tract Cancers. J. Clin. Oncol. 2025, 43, 536–544. [Google Scholar]
- Vogel, A.; Saborowski, A.; Wenzel, P.; Wege, H.; Folprecht, G.; Kretzschmar, A.; Schutt, P.; Jacobasch, L.; Ziegenhagen, N.; Boeck, S.; et al. Nanoliposomal irinotecan and fluorouracil plus leucovorin versus fluorouracil plus leucovorin in patients with cholangiocarcinoma and gallbladder carcinoma previously treated with gemcitabine-based therapies (AIO NALIRICC): A multicentre, open-label, randomised, phase 2 trial. Lancet Gastroenterol. Hepatol. 2024, 9, 734–744. [Google Scholar] [PubMed]
- Takahara, N.; Nakai, Y.; Isayama, H.; Sasaki, T.; Morine, Y.; Watanabe, K.; Ueno, M.; Ioka, T.; Kanai, M.; Kondo, S.; et al. A prospective multicenter phase II study of FOLFIRINOX as a first-line treatment for patients with advanced and recurrent biliary tract cancer. Investig. New Drugs 2023, 41, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Lamarca, A.; Palmer, D.H.; Wasan, H.S.; Ross, P.J.; Ma, Y.T.; Arora, A.; Falk, S.; Gillmore, R.; Wadsley, J.; Patel, K.; et al. Second-line FOLFOX chemotherapy versus active symptom control for advanced biliary tract cancer (ABC-06): A phase 3, open-label, randomised, controlled trial. Lancet Oncol. 2021, 22, 690–701. [Google Scholar] [PubMed]
- Sasaki, T.; Isayama, H.; Nakai, Y.; Ito, Y.; Yasuda, I.; Toda, N.; Kogure, H.; Hanada, K.; Maguchi, H.; Sasahira, N.; et al. A randomized phase II study of gemcitabine and S-1 combination therapy versus gemcitabine monotherapy for advanced biliary tract cancer. Cancer Chemother Pharmacol. 2013, 71, 973–979. [Google Scholar]
- Valle, J.; Wasan, H.; Palmer, D.H.; Cunningham, D.; Anthoney, A.; Maraveyas, A.; Madhusudan, S.; Iveson, T.; Hughes, S.; Pereira, S.P.; et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N. Engl. J. Med. 2010, 362, 1273–1281. [Google Scholar] [CrossRef]
- Izquierdo-Sanchez, L.; Lamarca, A.; La Casta, A.; Buettner, S.; Utpatel, K.; Klumpen, H.J.; Adeva, J.; Vogel, A.; Lleo, A.; Fabris, L.; et al. Cholangiocarcinoma landscape in Europe: Diagnostic, prognostic and therapeutic insights from the ENSCCA Registry. J. Hepatol. 2022, 76, 1109–1121. [Google Scholar]
- Bowlus, C.L.; Arrive, L.; Bergquist, A.; Deneau, M.; Forman, L.; Ilyas, S.I.; Lunsford, K.E.; Martinez, M.; Sapisochin, G.; Shroff, R.; et al. AASLD practice guidance on primary sclerosing cholangitis and cholangiocarcinoma. Hepatology 2023, 77, 659–702. [Google Scholar]
- Liang, B.; Zhong, L.; He, Q.; Wang, S.; Pan, Z.; Wang, T.; Zhao, Y. Diagnostic Accuracy of Serum CA19-9 in Patients with Cholangiocarcinoma: A Systematic Review and Meta-Analysis. Med. Sci. Monit. 2015, 21, 3555–3563. [Google Scholar] [CrossRef]
- Macias, R.I.R.; Cardinale, V.; Kendall, T.J.; Avila, M.A.; Guido, M.; Coulouarn, C.; Braconi, C.; Frampton, A.E.; Bridgewater, J.; Overi, D.; et al. Clinical relevance of biomarkers in cholangiocarcinoma: Critical revision and future directions. Gut 2022, 71, 1669–1683. [Google Scholar]
- Tot, T. Adenocarcinomas metastatic to the liver: The value of cytokeratins 20 and 7 in the search for unknown primary tumors. Cancer 1999, 85, 171–177. [Google Scholar] [CrossRef]
- Dhanasekaran, R.; Suzuki, H.; Lemaitre, L.; Kubota, N.; Hoshida, Y. Molecular and immune landscape of hepatocellular carcinoma to guide therapeutic decision-making. Hepatology 2023, 81, 1038–1057. [Google Scholar] [CrossRef] [PubMed]
- Tot, T. Identifying colorectal metastases in liver biopsies: The novel CDX2 antibody is less specific than the cytokeratin 20+/7- phenotype. Med. Sci. Monit. 2004, 10, BR139–BR143. [Google Scholar]
- Uenishi, T.; Yamazaki, O.; Tanaka, H.; Takemura, S.; Yamamoto, T.; Tanaka, S.; Nishiguchi, S.; Kubo, S. Serum cytokeratin 19 fragment (CYFRA21-1) as a prognostic factor in intrahepatic cholangiocarcinoma. Ann. Surg. Oncol. 2008, 15, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chen, W.; Liang, P.; Hu, W.; Zhang, K.; Shen, S.; Chen, J.; Zhang, Z.; Chen, B.; Han, Y.; et al. Serum CYFRA 21-1 in Biliary Tract Cancers: A Reliable Biomarker for Gallbladder Carcinoma and Intrahepatic Cholangiocarcinoma. Dig. Dis. Sci. 2015, 60, 1273–1283. [Google Scholar] [CrossRef]
- Leelawat, K.; Narong, S.; Wannaprasert, J.; Ratanashu-ek, T. Prospective study of MMP7 serum levels in the diagnosis of cholangiocarcinoma. World J. Gastroenterol. 2010, 16, 4697–4703. [Google Scholar] [CrossRef]
- Sirica, A.E. Matricellular proteins in intrahepatic cholangiocarcinoma. Adv. Cancer Res. 2022, 156, 249–281. [Google Scholar]
- Loosen, S.H.; Roderburg, C.; Kauertz, K.L.; Pombeiro, I.; Leyh, C.; Benz, F.; Vucur, M.; Longerich, T.; Koch, A.; Braunschweig, T.; et al. Elevated levels of circulating osteopontin are associated with a poor survival after resection of cholangiocarcinoma. J. Hepatol. 2017, 67, 749–757. [Google Scholar] [CrossRef]
- Goydos, J.S.; Brumfield, A.M.; Frezza, E.; Booth, A.; Lotze, M.T.; Carty, S.E. Marked elevation of serum interleukin-6 in patients with cholangiocarcinoma: Validation of utility as a clinical marker. Ann. Surg. 1998, 227, 398–404. [Google Scholar] [CrossRef]
- Matull, W.R.; Andreola, F.; Loh, A.; Adiguzel, Z.; Deheragoda, M.; Qureshi, U.; Batra, S.K.; Swallow, D.M.; Pereira, S.P. MUC4 and MUC5AC are highly specific tumour-associated mucins in biliary tract cancer. Br. J. Cancer 2008, 98, 1675–1681. [Google Scholar] [CrossRef]
- Lapitz, A.; Azkargorta, M.; Milkiewicz, P.; Olaizola, P.; Zhuravleva, E.; Grimsrud, M.M.; Schramm, C.; Arbelaiz, A.; O’Rourke, C.J.; La Casta, A.; et al. Liquid biopsy-based protein biomarkers for risk prediction, early diagnosis, and prognostication of cholangiocarcinoma. J. Hepatol. 2023, 79, 93–108. [Google Scholar] [PubMed]
- Banales, J.M.; Inarrairaegui, M.; Arbelaiz, A.; Milkiewicz, P.; Muntane, J.; Munoz-Bellvis, L.; La Casta, A.; Gonzalez, L.M.; Arretxe, E.; Alonso, C.; et al. Serum Metabolites as Diagnostic Biomarkers for Cholangiocarcinoma, Hepatocellular Carcinoma, and Primary Sclerosing Cholangitis. Hepatology 2019, 70, 547–562. [Google Scholar] [PubMed]
- Macias, R.I.R.; Munoz-Bellvis, L.; Sanchez-Martin, A.; Arretxe, E.; Martinez-Arranz, I.; Lapitz, A.; Gutierrez, M.L.; La Casta, A.; Alonso, C.; Gonzalez, L.M.; et al. A Novel Serum Metabolomic Profile for the Differential Diagnosis of Distal Cholangiocarcinoma and Pancreatic Ductal Adenocarcinoma. Cancers 2020, 12, 1433. [Google Scholar] [CrossRef]
- Urman, J.M.; Herranz, J.M.; Uriarte, I.; Rullan, M.; Oyon, D.; Gonzalez, B.; Fernandez-Urien, I.; Carrascosa, J.; Bolado, F.; Zabalza, L.; et al. Pilot Multi-Omic Analysis of Human Bile from Benign and Malignant Biliary Strictures: A Machine-Learning Approach. Cancers 2020, 12, 1644. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Shu, M.; Liao, J.; Liang, R.; Liu, S.; Kuang, M.; Peng, S.; Xiao, H.; Zhou, Q. Identification and validation of a plasma metabolomics-based model for risk stratification of intrahepatic cholangiocarcinoma. J. Cancer Res. Clin. Oncol. 2023, 149, 12365–12377. [Google Scholar] [CrossRef]
- Nakamura, H.; Arai, Y.; Totoki, Y.; Shirota, T.; Elzawahry, A.; Kato, M.; Hama, N.; Hosoda, F.; Urushidate, T.; Ohashi, S.; et al. Genomic spectra of biliary tract cancer. Nat. Genet. 2015, 47, 1003–1010. [Google Scholar]
- Javle, M.; Bekaii-Saab, T.; Jain, A.; Wang, Y.; Kelley, R.K.; Wang, K.; Kang, H.C.; Catenacci, D.; Ali, S.; Krishnan, S.; et al. Biliary cancer: Utility of next-generation sequencing for clinical management. Cancer 2016, 122, 3838–3847. [Google Scholar] [CrossRef]
- Jusakul, A.; Cutcutache, I.; Yong, C.H.; Lim, J.Q.; Huang, M.N.; Padmanabhan, N.; Nellore, V.; Kongpetch, S.; Ng, A.W.T.; Ng, L.M.; et al. Whole-Genome and Epigenomic Landscapes of Etiologically Distinct Subtypes of Cholangiocarcinoma. Cancer Discov. 2017, 7, 1116–1135. [Google Scholar] [CrossRef]
- Wardell, C.P.; Fujita, M.; Yamada, T.; Simbolo, M.; Fassan, M.; Karlic, R.; Polak, P.; Kim, J.; Hatanaka, Y.; Maejima, K.; et al. Genomic characterization of biliary tract cancers identifies driver genes and predisposing mutations. J. Hepatol. 2018, 68, 959–969. [Google Scholar]
- Umemoto, K.; Yamamoto, H.; Oikawa, R.; Takeda, H.; Doi, A.; Horie, Y.; Arai, H.; Ogura, T.; Mizukami, T.; Izawa, N.; et al. The Molecular Landscape of Pancreatobiliary Cancers for Novel Targeted Therapies From Real-World Genomic Profiling. J. Natl. Cancer Inst. 2022, 114, 1279–1286. [Google Scholar]
- Nishida, N.; Aoki, T.; Morita, M.; Chishina, H.; Takita, M.; Ida, H.; Hagiwara, S.; Minami, Y.; Ueshima, K.; Kudo, M. Non-Inflamed Tumor Microenvironment and Methylation/Downregulation of Antigen-Presenting Machineries in Cholangiocarcinoma. Cancers 2023, 15, 2379. [Google Scholar] [CrossRef] [PubMed]
- Nishida, N.; Kudo, M. Genetic/Epigenetic Alteration and Tumor Immune Microenvironment in Intrahepatic Cholangiocarcinoma: Transforming the Immune Microenvironment with Molecular-Targeted Agents. Liver Cancer 2024, 13, 136–149. [Google Scholar] [CrossRef] [PubMed]
- Verlingue, L.; Malka, D.; Allorant, A.; Massard, C.; Ferte, C.; Lacroix, L.; Rouleau, E.; Auger, N.; Ngo, M.; Nicotra, C.; et al. Precision medicine for patients with advanced biliary tract cancers: An effective strategy within the prospective MOSCATO-01 trial. Eur. J. Cancer 2017, 87, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Verdaguer, H.; Sauri, T.; Acosta, D.A.; Guardiola, M.; Sierra, A.; Hernando, J.; Nuciforo, P.; Miquel, J.M.; Molero, C.; Peiro, S.; et al. ESMO Scale for Clinical Actionability of Molecular Targets Driving Targeted Treatment in Patients with Cholangiocarcinoma. Clin. Cancer Res. 2022, 28, 1662–1671. [Google Scholar] [CrossRef]
- Mosele, F.; Remon, J.; Mateo, J.; Westphalen, C.B.; Barlesi, F.; Lolkema, M.P.; Normanno, N.; Scarpa, A.; Robson, M.; Meric-Bernstam, F.; et al. Recommendations for the use of next-generation sequencing (NGS) for patients with metastatic cancers: A report from the ESMO Precision Medicine Working Group. Ann. Oncol. 2020, 31, 1491–1505. [Google Scholar] [CrossRef]
- Park, J.O.; Feng, Y.H.; Su, W.C.; Oh, D.Y.; Keam, B.; Shen, L.; Kim, S.W.; Liu, X.; Liao, H.; Qing, M.; et al. Erdafitinib in Asian patients with advanced solid tumors: An open-label, single-arm, phase IIa trial. BMC Cancer 2024, 24, 1006. [Google Scholar] [CrossRef]
- Goyal, L.; Meric-Bernstam, F.; Hollebecque, A.; Valle, J.W.; Morizane, C.; Karasic, T.B.; Abrams, T.A.; Furuse, J.; Kelley, R.K.; Cassier, P.A.; et al. Futibatinib for FGFR2-Rearranged Intrahepatic Cholangiocarcinoma. N. Engl. J. Med. 2023, 388, 228–239. [Google Scholar] [CrossRef]
- Bibeau, K.; Feliz, L.; Lihou, C.F.; Ren, H.; Abou-Alfa, G.K. Progression-Free Survival in Patients With Cholangiocarcinoma With or Without FGF/FGFR Alterations: A FIGHT-202 Post Hoc Analysis of Prior Systemic Therapy Response. JCO Precis Oncol. 2022, 6, e2100414. [Google Scholar] [CrossRef]
- Javle, M.; Roychowdhury, S.; Kelley, R.K.; Sadeghi, S.; Macarulla, T.; Weiss, K.H.; Waldschmidt, D.T.; Goyal, L.; Borbath, I.; El-Khoueiry, A.; et al. Infigratinib (BGJ398) in previously treated patients with advanced or metastatic cholangiocarcinoma with FGFR2 fusions or rearrangements: Mature results from a multicentre, open-label, single-arm, phase 2 study. Lancet Gastroenterol. Hepatol. 2021, 6, 803–815. [Google Scholar] [CrossRef]
- Bekaii-Saab, T.S.; Valle, J.W.; Van Cutsem, E.; Rimassa, L.; Furuse, J.; Ioka, T.; Melisi, D.; Macarulla, T.; Bridgewater, J.; Wasan, H.; et al. FIGHT-302: First-line pemigatinib vs. gemcitabine plus cisplatin for advanced cholangiocarcinoma with FGFR2 rearrangements. Future Oncol. 2020, 16, 2385–2399. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Sahai, V.; Hollebecque, A.; Vaccaro, G.; Melisi, D.; Al-Rajabi, R.; Paulson, A.S.; Borad, M.J.; Gallinson, D.; Murphy, A.G.; et al. Pemigatinib for previously treated, locally advanced or metastatic cholangiocarcinoma: A multicentre, open-label, phase 2 study. Lancet Oncol. 2020, 21, 671–684. [Google Scholar] [PubMed]
- Makawita, S.; Abou-Alfa, G.K.; Roychowdhury, S.; Sadeghi, S.; Borbath, I.; Goyal, L.; Cohn, A.; Lamarca, A.; Oh, D.Y.; Macarulla, T.; et al. Infigratinib in patients with advanced cholangiocarcinoma with FGFR2 gene fusions/translocations: The PROOF 301 trial. Future Oncol. 2020, 16, 2375–2384. [Google Scholar]
- Mazzaferro, V.; El-Rayes, B.F.; Droz Dit Busset, M.; Cotsoglou, C.; Harris, W.P.; Damjanov, N.; Masi, G.; Rimassa, L.; Personeni, N.; Braiteh, F.; et al. Derazantinib (ARQ 087) in advanced or inoperable FGFR2 gene fusion-positive intrahepatic cholangiocarcinoma. Br. J. Cancer 2019, 120, 165–171. [Google Scholar] [PubMed]
- Abou-Alfa, G.K.; Macarulla, T.; Javle, M.M.; Kelley, R.K.; Lubner, S.J.; Adeva, J.; Cleary, J.M.; Catenacci, D.V.; Borad, M.J.; Bridgewater, J.; et al. Ivosidenib in IDH1-mutant, chemotherapy-refractory cholangiocarcinoma (ClarIDHy): A multicentre, randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 2020, 21, 796–807. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, A.; Bhargava, P.; Srinivas, S.; Kapoor, A.; Mishra, B.K.; Gupta, A.; Mandavkar, S.; Kannan, S.; Chaugule, D.; Patil, R.; et al. Bevacizumab Erlotinib Switch Maintenance in Chemo-Responsive Advanced Gallbladder and Cholangiocarcinoma (BEER BTC): A Multicenter, Open-Label, Randomized, Phase II Trial. J. Clin. Oncol. 2024, 42, 3218–3227. [Google Scholar]
- Amin, N.E.L.; Hansen, T.F.; Fernebro, E.; Ploen, J.; Eberhard, J.; Lindebjerg, J.; Jensen, L.H. Randomized Phase II trial of combination chemotherapy with panitumumab or bevacizumab for patients with inoperable biliary tract cancer without KRAS exon 2 mutations. Int. J. Cancer 2021, 149, 119–126. [Google Scholar] [CrossRef]
- Leone, F.; Marino, D.; Cereda, S.; Filippi, R.; Belli, C.; Spadi, R.; Nasti, G.; Montano, M.; Amatu, A.; Aprile, G.; et al. Panitumumab in combination with gemcitabine and oxaliplatin does not prolong survival in wild-type KRAS advanced biliary tract cancer: A randomized phase 2 trial (Vecti-BIL study). Cancer 2016, 122, 574–581. [Google Scholar]
- Malka, D.; Cervera, P.; Foulon, S.; Trarbach, T.; de la Fouchardiere, C.; Boucher, E.; Fartoux, L.; Faivre, S.; Blanc, J.F.; Viret, F.; et al. Gemcitabine and oxaliplatin with or without cetuximab in advanced biliary-tract cancer (BINGO): A randomised, open-label, non-comparative phase 2 trial. Lancet Oncol. 2014, 15, 819–828. [Google Scholar] [CrossRef]
- Lee, J.; Park, S.H.; Chang, H.M.; Kim, J.S.; Choi, H.J.; Lee, M.A.; Jang, J.S.; Jeung, H.C.; Kang, J.H.; Lee, H.W.; et al. Gemcitabine and oxaliplatin with or without erlotinib in advanced biliary-tract cancer: A multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2012, 13, 181–188. [Google Scholar]
- Cecchini, M.; Pilat, M.J.; Uboha, N.; Azad, N.S.; Cho, M.; Davis, E.J.; Ahnert, J.R.; Tinoco, G.; Shapiro, G.I.; Khagi, S.; et al. Olaparib in treatment-refractory isocitrate dehydrogenase 1 (IDH1)- and IDH2-mutant cholangiocarcinoma: Safety and antitumor activity from the phase 2 National Cancer Institute 10129 trial. Cancer 2025, 131, e35755. [Google Scholar]
- Azad, N.; Hu, Z.; Sahin, I.; Iyer, R.; Aranha, O.; Hochster, H.; Pathak, P.; Paulson, A.S.; Kalyan, A.; Liao, C.Y.; et al. COMPANION-002 A clinical trial of investigational drug CTX-009 plus paclitaxel vs. paclitaxel in second line advanced BTC. Future Oncol. 2024, 20, 2241–2248. [Google Scholar] [CrossRef] [PubMed]
- Subbiah, V.; Kreitman, R.J.; Wainberg, Z.A.; Gazzah, A.; Lassen, U.; Stein, A.; Wen, P.Y.; Dietrich, S.; de Jonge, M.J.A.; Blay, J.Y.; et al. Dabrafenib plus trametinib in BRAFV600E-mutated rare cancers: The phase 2 ROAR trial. Nat. Med. 2023, 29, 1103–1112. [Google Scholar] [CrossRef] [PubMed]
- Valle, J.W.; Wasan, H.; Lopes, A.; Backen, A.C.; Palmer, D.H.; Morris, K.; Duggan, M.; Cunningham, D.; Anthoney, D.A.; Corrie, P.; et al. Cediranib or placebo in combination with cisplatin and gemcitabine chemotherapy for patients with advanced biliary tract cancer (ABC-03): A randomised phase 2 trial. Lancet Oncol. 2015, 16, 967–978. [Google Scholar] [CrossRef]
- Ohba, A.; Morizane, C.; Kawamoto, Y.; Komatsu, Y.; Ueno, M.; Kobayashi, S.; Ikeda, M.; Sasaki, M.; Furuse, J.; Okano, N.; et al. Trastuzumab Deruxtecan in Human Epidermal Growth Factor Receptor 2-Expressing Biliary Tract Cancer (HERB.; NCCH1805): A Multicenter, Single-Arm, Phase II Trial. J. Clin. Oncol. 2024, 42, 3207–3217. [Google Scholar] [CrossRef] [PubMed]
- Harding, J.J.; Piha-Paul, S.A.; Shah, R.H.; Murphy, J.J.; Cleary, J.M.; Shapiro, G.I.; Quinn, D.I.; Brana, I.; Moreno, V.; Borad, M.; et al. Antitumour activity of neratinib in patients with HER2-mutant advanced biliary tract cancers. Nat. Commun. 2023, 14, 630. [Google Scholar] [CrossRef]
- Harding, J.J.; Fan, J.; Oh, D.Y.; Choi, H.J.; Kim, J.W.; Chang, H.M.; Bao, L.; Sun, H.C.; Macarulla, T.; Xie, F.; et al. Zanidatamab for HER2-amplified, unresectable, locally advanced or metastatic biliary tract cancer (HERIZON-BTC-01): A multicentre, single-arm, phase 2b study. Lancet Oncol. 2023, 24, 772–782. [Google Scholar] [CrossRef]
- Javle, M.; Borad, M.J.; Azad, N.S.; Kurzrock, R.; Abou-Alfa, G.K.; George, B.; Hainsworth, J.; Meric-Bernstam, F.; Swanton, C.; Sweeney, C.J.; et al. Pertuzumab and trastuzumab for HER2-positive, metastatic biliary tract cancer (MyPathway): A multicentre, open-label, phase 2a, multiple basket study. Lancet Oncol. 2021, 22, 1290–1300. [Google Scholar] [CrossRef]
- Maruki, Y.; Morizane, C.; Arai, Y.; Ikeda, M.; Ueno, M.; Ioka, T.; Naganuma, A.; Furukawa, M.; Mizuno, N.; Uwagawa, T.; et al. Molecular detection and clinicopathological characteristics of advanced/recurrent biliary tract carcinomas harboring the FGFR2 rearrangements: A prospective observational study (PRELUDE Study). J. Gastroenterol. 2021, 56, 250–260. [Google Scholar] [CrossRef]
- Nishida, N. The role of FGFR inhibitors in the treatment of intrahepatic cholangiocarcinoma-unveiling the future challenges in drug therapy. Hepatobiliary Surg. Nutr. 2023, 12, 790–794. [Google Scholar] [CrossRef]
- Speckart, J.; Rasmusen, V.; Talib, Z.; GnanaDev, D.A.; Rahnemai-Azar, A.A. Emerging Therapies in Management of Cholangiocarcinoma. Cancers 2024, 16, 613. [Google Scholar] [CrossRef]
- Goyal, L.; Meric-Bernstam, F.; Hollebecque, A.; Valle, J.W.; Morizane, C.; Karasic, T.B.; Abrams, T.A.; Furuse, J.; Kelley, R.K.; Cassier, P.A.; et al. Plain language summary of the FOENIX-CCA2 study: Futibatinib for people with advanced bile duct cancer. Future Oncol. 2024, 20, 2811–2822. [Google Scholar]
- Borger, D.R.; Goyal, L.; Yau, T.; Poon, R.T.; Ancukiewicz, M.; Deshpande, V.; Christiani, D.C.; Liebman, H.M.; Yang, H.; Kim, H.; et al. Circulating oncometabolite 2-hydroxyglutarate is a potential surrogate biomarker in patients with isocitrate dehydrogenase-mutant intrahepatic cholangiocarcinoma. Clin. Cancer Res. 2014, 20, 1884–1890. [Google Scholar] [PubMed]
- Marabelle, A.; Le, D.T.; Ascierto, P.A.; Di Giacomo, A.M.; De Jesus-Acosta, A.; Delord, J.P.; Geva, R.; Gottfried, M.; Penel, N.; Hansen, A.R.; et al. Efficacy of Pembrolizumab in Patients with Noncolorectal High Microsatellite Instability/Mismatch Repair-Deficient Cancer: Results From the Phase II KEYNOTE-158 Study. J. Clin. Oncol. 2020, 38, 1–10. [Google Scholar] [PubMed]
- Marabelle, A.; Fakih, M.; Lopez, J.; Shah, M.; Shapira-Frommer, R.; Nakagawa, K.; Chung, H.C.; Kindler, H.L.; Lopez-Martin, J.A.; Miller, W.H., Jr.; et al. Association of tumour mutational burden with outcomes in patients with advanced solid tumours treated with pembrolizumab: Prospective biomarker analysis of the multicohort, open-label, phase 2 KEYNOTE-158 study. Lancet Oncol. 2020, 21, 1353–1365. [Google Scholar] [PubMed]
- Mavroeidi, I.A.; Burghofer, J.; Kalbourtzis, S.; Taghizadeh, H.; Webersinke, G.; Piringer, G.; Kasper, S.; Schreil, G.; Liffers, S.T.; Reichinger, A.; et al. Understanding homologous recombination repair deficiency in biliary tract cancers: Clinical implications and correlation with platinum sensitivity. ESMO Open 2024, 9, 103630. [Google Scholar]
- Hong, D.S.; DuBois, S.G.; Kummar, S.; Farago, A.F.; Albert, C.M.; Rohrberg, K.S.; van Tilburg, C.M.; Nagasubramanian, R.; Berlin, J.D.; Federman, N.; et al. Larotrectinib in patients with TRK fusion-positive solid tumours: A pooled analysis of three phase 1/2 clinical trials. Lancet Oncol. 2020, 21, 531–540. [Google Scholar]
- Doebele, R.C.; Drilon, A.; Paz-Ares, L.; Siena, S.; Shaw, A.T.; Farago, A.F.; Blakely, C.M.; Seto, T.; Cho, B.C.; Tosi, D.; et al. Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours: Integrated analysis of three phase 1-2 trials. Lancet Oncol. 2020, 21, 271–282. [Google Scholar]
- Lin, D.; Shen, L.; Luo, M.; Zhang, K.; Li, J.; Yang, Q.; Zhu, F.; Zhou, D.; Zheng, S.; Chen, Y.; et al. Circulating tumor cells: Biology and clinical significance. Signal Transduct Target Ther. 2021, 6, 404. [Google Scholar] [CrossRef]
- Yang, J.D.; Campion, M.B.; Liu, M.C.; Chaiteerakij, R.; Giama, N.H.; Ahmed Mohammed, H.; Zhang, X.; Hu, C.; Campion, V.L.; Jen, J.; et al. Circulating tumor cells are associated with poor overall survival in patients with cholangiocarcinoma. Hepatology 2016, 63, 148–158. [Google Scholar] [CrossRef]
- Liang, K.H.; Yeh, T.S.; Wu, R.C.; Yeh, C.N.; Yeh, C.T. GALNT14 genotype is associated with perineural invasion, lymph node metastasis and overall survival in resected cholangiocarcinoma. Oncol. Lett. 2017, 13, 4215–4223. [Google Scholar]
- Pan, Y.; Shao, S.; Sun, H.; Zhu, H.; Fang, H. Bile-derived exosome noncoding RNAs as potential diagnostic and prognostic biomarkers for cholangiocarcinoma. Front Oncol. 2022, 12, 985089. [Google Scholar] [CrossRef] [PubMed]
- Uson Junior, P.L.S.; Majeed, U.; Yin, J.; Botrus, G.; Sonbol, M.B.; Ahn, D.H.; Starr, J.S.; Jones, J.C.; Babiker, H.; Inabinett, S.R.; et al. Cell-Free Tumor DNA Dominant Clone Allele Frequency Is Associated With Poor Outcomes in Advanced Biliary Cancers Treated With Platinum-Based Chemotherapy. JCO Precis Oncol. 2022, 6, e2100274. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Xin, H.; Zhou, Z.; Hu, Z.; Sun, R.; Yao, N.; Sun, Q.; Borjigin, U.; Wu, X.; Fan, J.; et al. Tumor-derived exosomes induce immunosuppressive macrophages to foster intrahepatic cholangiocarcinoma progression. Hepatology 2022, 76, 982–999. [Google Scholar] [CrossRef]
- Lurje, I.; Czigany, Z.; Bednarsch, J.; Gaisa, N.T.; Dahl, E.; Knuchel, R.; Miller, H.; Ulmer, T.F.; Strnad, P.; Trautwein, C.; et al. Genetic Variant of CXCR1 (rs2234671) Associates with Clinical Outcome in Perihilar Cholangiocarcinoma. Liver Cancer 2022, 11, 162–173. [Google Scholar] [CrossRef]
- Shu, L.; Li, X.; Liu, Z.; Li, K.; Shi, A.; Tang, Y.; Zhao, L.; Huang, L.; Zhang, Z.; Zhang, D.; et al. Bile exosomal miR-182/183-5p increases cholangiocarcinoma stemness and progression by targeting HPGD and increasing PGE2 generation. Hepatology 2024, 79, 307–322. [Google Scholar] [CrossRef]
- Iyer, R.V.; Pokuri, V.K.; Groman, A.; Ma, W.W.; Malhotra, U.; Iancu, D.M.; Grande, C.; Saab, T.B. A Multicenter Phase II Study of Gemcitabine, Capecitabine, and Bevacizumab for Locally Advanced or Metastatic Biliary Tract Cancer. Am. J. Clin. Oncol. 2018, 41, 649–655. [Google Scholar] [CrossRef]
- Frundt, T.; von Felden, J.; Krause, J.; Heumann, A.; Li, J.; Riethdorf, S.; Pantel, K.; Huber, S.; Lohse, A.W.; Wege, H.; et al. Circulating tumor cells as a preoperative risk marker for occult metastases in patients with resectable cholangiocarcinoma. Front. Oncol. 2022, 12, 941660. [Google Scholar] [CrossRef]
- Gopinathan, P.; Chiang, N.J.; Bandaru, A.; Sinha, A.; Huang, W.Y.; Hung, S.C.; Shan, Y.S.; Lee, G.B. Exploring Circulating Tumor Cells in Cholangiocarcinoma Using a Novel Glycosaminoglycan Probe on a Microfluidic Platform. Adv. Healthc. Mater. 2020, 9, e1901875. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.L.; Huang, C.J.; Tsai, Y.C.; Chiang, N.J.; Huang, Y.S.; Hung, S.C.; Shan, Y.S.; Lee, G.B. An integrated microfluidic system for automatic detection of cholangiocarcinoma cells from bile. Lab. Chip. 2024, 24, 375–382. [Google Scholar] [CrossRef]
- Rompianesi, G.; Di Martino, M.; Gordon-Weeks, A.; Montalti, R.; Troisi, R. Liquid biopsy in cholangiocarcinoma: Current status and future perspectives. World J. Gastrointest Oncol. 2021, 13, 332–350. [Google Scholar] [CrossRef]
- Lamarca, A.; Kapacee, Z.; Breeze, M.; Bell, C.; Belcher, D.; Staiger, H.; Taylor, C.; McNamara, M.G.; Hubner, R.A.; Valle, J.W. Molecular Profiling in Daily Clinical Practice: Practicalities in Advanced Cholangiocarcinoma and Other Biliary Tract Cancers. J. Clin. Med. 2020, 9, 2854. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Taniguchi, H.; Ikeda, M.; Bando, H.; Kato, K.; Morizane, C.; Esaki, T.; Komatsu, Y.; Kawamoto, Y.; Takahashi, N.; et al. Clinical utility of circulating tumor DNA sequencing in advanced gastrointestinal cancer: SCRUM-Japan GI-SCREEN and GOZILA studies. Nat. Med. 2020, 26, 1859–1864. [Google Scholar] [CrossRef] [PubMed]
- Wintachai, P.; Lim, J.Q.; Techasen, A.; Lert-Itthiporn, W.; Kongpetch, S.; Loilome, W.; Chindaprasirt, J.; Titapun, A.; Namwat, N.; Khuntikeo, N.; et al. Diagnostic and Prognostic Value of Circulating Cell-Free DNA for Cholangiocarcinoma. Diagnostics 2021, 11, 999. [Google Scholar] [CrossRef]
- Hashimoto, T.; Nakamura, Y.; Oki, E.; Kobayashi, S.; Yuda, J.; Shibuki, T.; Bando, H.; Yoshino, T. Bridging horizons beyond CIRCULATE-Japan: A new paradigm in molecular residual disease detection via whole genome sequencing-based circulating tumor DNA assay. Int. J. Clin. Oncol. 2024, 29, 495–511. [Google Scholar] [CrossRef] [PubMed]
- Mody, K.; Cleary, S.P. A Review of Circulating Tumor DNA in Hepatobiliary Malignancies. Front. Oncol. 2018, 8, 212. [Google Scholar] [CrossRef]
- Ettrich, T.J.; Schwerdel, D.; Dolnik, A.; Beuter, F.; Blatte, T.J.; Schmidt, S.A.; Stanescu-Siegmund, N.; Steinacker, J.; Marienfeld, R.; Kleger, A.; et al. Genotyping of circulating tumor DNA in cholangiocarcinoma reveals diagnostic and prognostic information. Sci. Rep. 2019, 9, 13261. [Google Scholar] [CrossRef]
- Okamura, R.; Kurzrock, R.; Mallory, R.J.; Fanta, P.T.; Burgoyne, A.M.; Clary, B.M.; Kato, S.; Sicklick, J.K. Comprehensive genomic landscape and precision therapeutic approach in biliary tract cancers. Int. J. Cancer 2021, 148, 702–712. [Google Scholar] [CrossRef]
- Berchuck, J.E.; Facchinetti, F.; DiToro, D.F.; Baiev, I.; Majeed, U.; Reyes, S.; Chen, C.; Zhang, K.; Sharman, R.; Uson Junior, P.L.S.; et al. The clinical landscape of cell-free DNA alterations in 1671 patients with advanced biliary tract cancer. Ann. Oncol. 2022, 33, 1269–1283. [Google Scholar] [CrossRef]
- Kurzrock, R.; Aggarwal, C.; Weipert, C.; Kiedrowski, L.; Riess, J.; Lenz, H.J.; Gandara, D. Prevalence of ARID1A Mutations in Cell-Free Circulating Tumor DNA in a Cohort of 71,301 Patients and Association with Driver Co-Alterations. Cancers 2022, 14, 4281. [Google Scholar] [CrossRef]
- Choi, W.J.; Ivanics, T.; Gravely, A.; Gallinger, S.; Sapisochin, G.; O’Kane, G.M. Optimizing Circulating Tumour DNA Use in the Perioperative Setting for Intrahepatic Cholangiocarcinoma: Diagnosis, Screening, Minimal Residual Disease Detection and Treatment Response Monitoring. Ann. Surg. Oncol. 2023, 30, 3849–3863. [Google Scholar] [CrossRef]
- Vedeld, H.M.; Folseraas, T.; Lind, G.E. Detecting cholangiocarcinoma in patients with primary sclerosing cholangitis—The promise of DNA methylation and molecular biomarkers. JHEP Rep. 2020, 2, 100143. [Google Scholar] [CrossRef] [PubMed]
- Klein, E.A.; Richards, D.; Cohn, A.; Tummala, M.; Lapham, R.; Cosgrove, D.; Chung, G.; Clement, J.; Gao, J.; Hunkapiller, N.; et al. Clinical validation of a targeted methylation-based multi-cancer early detection test using an independent validation set. Ann. Oncol. 2021, 32, 1167–1177. [Google Scholar] [CrossRef]
- Andersen, L.B.; Mahler, M.S.K.; Andersen, R.F.; Jensen, L.H.; Raunkilde, L. The Clinical Impact of Methylated Homeobox A9 ctDNA in Patients with Non-Resectable Biliary Tract Cancer Treated with Erlotinib and Bevacizumab. Cancers 2022, 14, 4598. [Google Scholar] [CrossRef]
- Kasi, P.M.; Lee, J.K.; Pasquina, L.W.; Decker, B.; Vanden Borre, P.; Pavlick, D.C.; Allen, J.M.; Parachoniak, C.; Quintanilha, J.C.F.; Graf, R.P.; et al. Circulating Tumor DNA Enables Sensitive Detection of Actionable Gene Fusions and Rearrangements Across Cancer Types. Clin. Cancer Res. 2024, 30, 836–848. [Google Scholar] [CrossRef]
- Castro-Oropeza, R.; Melendez-Zajgla, J.; Maldonado, V.; Vazquez-Santillan, K. The emerging role of lncRNAs in the regulation of cancer stem cells. Cell Oncol. 2018, 41, 585–603. [Google Scholar]
- Liang, Z.; Zhu, B.; Meng, D.; Shen, X.; Li, X.; Wang, Z.; Li, L. Down-regulation of lncRNA-NEF indicates poor prognosis in intrahepatic cholangiocarcinoma. Biosci. Rep. 2019, 39, BSR20181573. [Google Scholar]
- Meng, F.; Henson, R.; Lang, M.; Wehbe, H.; Maheshwari, S.; Mendell, J.T.; Jiang, J.; Schmittgen, T.D.; Patel, T. Involvement of human micro-RNA in growth and response to chemotherapy in human cholangiocarcinoma cell lines. Gastroenterology 2006, 130, 2113–2129. [Google Scholar] [PubMed]
- Shen, L.; Chen, G.; Xia, Q.; Shao, S.; Fang, H. Exosomal miR-200 family as serum biomarkers for early detection and prognostic prediction of cholangiocarcinoma. Int. J. Clin. Exp. Pathol. 2019, 12, 3870–3876. [Google Scholar]
- Asukai, K.; Kawamoto, K.; Eguchi, H.; Konno, M.; Asai, A.; Iwagami, Y.; Yamada, D.; Asaoka, T.; Noda, T.; Wada, H.; et al. Micro-RNA-130a-3p Regulates Gemcitabine Resistance via PPARG in Cholangiocarcinoma. Ann. Surg. Oncol. 2017, 24, 2344–2352. [Google Scholar]
- Peng, F.; Jiang, J.; Yu, Y.; Tian, R.; Guo, X.; Li, X.; Shen, M.; Xu, M.; Zhu, F.; Shi, C.; et al. Direct targeting of SUZ12/ROCK2 by miR-200b/c inhibits cholangiocarcinoma tumourigenesis and metastasis. Br. J. Cancer 2013, 109, 3092–3104. [Google Scholar]
- Carotenuto, P.; Hedayat, S.; Fassan, M.; Cardinale, V.; Lampis, A.; Guzzardo, V.; Vicentini, C.; Scarpa, A.; Cascione, L.; Costantini, D.; et al. Modulation of Biliary Cancer Chemo-Resistance Through MicroRNA-Mediated Rewiring of the Expansion of CD133+ Cells. Hepatology 2020, 72, 982–996. [Google Scholar] [CrossRef] [PubMed]
- Silakit, R.; Kitirat, Y.; Thongchot, S.; Loilome, W.; Techasen, A.; Ungarreevittaya, P.; Khuntikeo, N.; Yongvanit, P.; Yang, J.H.; Kim, N.H.; et al. Potential role of HIF-1-responsive microRNA210/HIF3 axis on gemcitabine resistance in cholangiocarcinoma cells. PLoS ONE 2018, 13, e0199827. [Google Scholar] [CrossRef]
- Lu, M.; Qin, X.; Zhou, Y.; Li, G.; Liu, Z.; Geng, X.; Yue, H. Long non-coding RNA LINC00665 promotes gemcitabine resistance of Cholangiocarcinoma cells via regulating EMT and stemness properties through miR-424-5p/BCL9L axis. Cell Death Dis. 2021, 12, 72. [Google Scholar] [CrossRef] [PubMed]
- Obata, T.; Tsutsumi, K.; Ueta, E.; Oda, T.; Kikuchi, T.; Ako, S.; Fujii, Y.; Yamazaki, T.; Uchida, D.; Matsumoto, K.; et al. MicroRNA-451a inhibits gemcitabine-refractory biliary tract cancer progression by suppressing the MIF-mediated PI3K/AKT pathway. Mol. Ther. Nucleic Acids 2023, 34, 102054. [Google Scholar] [CrossRef] [PubMed]
- Tokuhisa, A.; Tsunedomi, R.; Kimura, Y.; Nakajima, M.; Nishiyama, M.; Takahashi, H.; Ioka, T.; Kobayashi, S.; Eguchi, H.; Nagano, H. Exosomal miR-141-3p Induces Gemcitabine Resistance in Biliary Tract Cancer Cells. Anticancer Res. 2024, 44, 2899–2908. [Google Scholar] [CrossRef]
- Du, H.; Hou, S.; Zhang, L.; Liu, C.; Yu, T.; Zhang, W. LncRNA FALEC increases the proliferation, migration and drug resistance of cholangiocarcinoma through competitive regulation of miR-20a-5p/SHOC2 axis. Aging 2023, 15, 3759–3770. [Google Scholar] [CrossRef]
- Zhang, L.; Ma, D.; Li, F.; Qiu, G.; Sun, D.; Zeng, Z. Lnc-PKD2-2-3/miR-328/GPAM ceRNA Network Induces Cholangiocarcinoma Proliferation, Invasion and 5-FU Chemoresistance. Front. Oncol. 2022, 12, 871281. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, W.; Chen, L.; Chen, W.; Xu, S.; Tang, L.; Yang, Y.; Li, Q.; Jiang, Q.; Miao, L. The ATO/miRNA-885-5p/MTPN axis induces reversal of drug-resistance in cholangiocarcinoma. Cell Oncol. 2021, 44, 907–916. [Google Scholar] [CrossRef]
- Li, Q.; Xia, X.; Ji, J.; Ma, J.; Tao, L.; Mo, L.; Chen, W. MiR-199a-3p enhances cisplatin sensitivity of cholangiocarcinoma cells by inhibiting mTOR signaling pathway and expression of MDR1. Oncotarget 2017, 8, 33621–33630. [Google Scholar] [CrossRef]
- Li, J.; Jiang, X.; Li, Z.; Huang, L.; Ji, D.; Yu, L.; Zhou, Y.; Cui, Y. SP1-induced HOXD-AS1 promotes malignant progression of cholangiocarcinoma by regulating miR-520c-3p/MYCN. Aging 2020, 12, 16304–16325. [Google Scholar] [CrossRef]
- Kopp, F.; Mendell, J.T. Functional Classification and Experimental Dissection of Long Noncoding RNAs. Cell 2018, 172, 393–407. [Google Scholar]
- Lapitz, A.; Arbelaiz, A.; O’Rourke, C.J.; Lavin, J.L.; Casta, A.; Ibarra, C.; Jimeno, J.P.; Santos-Laso, A.; Izquierdo-Sanchez, L.; Krawczyk, M.; et al. Patients with Cholangiocarcinoma Present Specific RNA Profiles in Serum and Urine Extracellular Vesicles Mirroring the Tumor Expression: Novel Liquid Biopsy Biomarkers for Disease Diagnosis. Cells 2020, 9, 721. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Wang, Y.; Nie, J.; Li, Q.; Tang, L.; Deng, X.; Wang, F.; Xu, B.; Wu, X.; Zhang, X.; et al. The diagnostic/prognostic potential and molecular functions of long non-coding RNAs in the exosomes derived from the bile of human cholangiocarcinoma. Oncotarget 2017, 8, 69995–70005. [Google Scholar]
- Li, J.; Jiang, X.; Xu, Y.; Kang, P.; Huang, P.; Meng, N.; Wang, H.; Zheng, W.; Wang, H.; Wang, Z.; et al. YY1-induced DLEU1/miR-149-5p Promotes Malignant Biological Behavior of Cholangiocarcinoma through Upregulating YAP1/TEAD2/SOX2. Int. J. Biol. Sci. 2022, 18, 4301–4315. [Google Scholar]
- Shi, J.; Li, X.; Zhang, F.; Kong, L.; Zhang, X.; Cheng, Y.; Guan, Q.; Cao, X.; Zhu, W.; Ou, K.; et al. The Plasma LncRNA Acting as Fingerprint in Hilar Cholangiocarcinoma. Cell Physiol. Biochem. 2018, 49, 1694–1702. [Google Scholar] [CrossRef]
- Zhang, F.; Wan, M.; Xu, Y.; Li, Z.; Leng, K.; Kang, P.; Cui, Y.; Jiang, X. Long noncoding RNA PCAT1 regulates extrahepatic cholangiocarcinoma progression via the Wnt/beta-catenin-signaling pathway. Biomed. Pharmacother. 2017, 94, 55–62. [Google Scholar] [PubMed]
- Xu, Y.; Wang, Z.; Jiang, X.; Cui, Y. Overexpression of long noncoding RNA H19 indicates a poor prognosis for cholangiocarcinoma and promotes cell migration and invasion by affecting epithelial-mesenchymal transition. Biomed. Pharmacother. 2017, 92, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Han, B.W.; Ye, H.; Wei, P.P.; He, B.; Han, C.; Chen, Z.H.; Chen, Y.Q.; Wang, W.T. Global identification and characterization of lncRNAs that control inflammation in malignant cholangiocytes. BMC Genom. 2018, 19, 735. [Google Scholar]
- Jiang, X.M.; Li, Z.L.; Li, J.L.; Zheng, W.Y.; Li, X.H.; Cui, Y.F.; Sun, D.J. LncRNA CCAT1 as the unfavorable prognostic biomarker for cholangiocarcinoma. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 1242–1247. [Google Scholar]
- Bai, J.G.; Tang, R.F.; Shang, J.F.; Qi, S.; Yu, G.D.; Sun, C. Upregulation of long non-coding RNA CCAT2 indicates a poor prognosis and promotes proliferation and metastasis in intrahepatic cholangiocarcinoma. Mol. Med. Rep. 2018, 17, 5328–5335. [Google Scholar]
- Jiang, X.; Li, J.; Wang, W.; Hu, Z.; Guan, C.; Zhao, Y.; Li, W.; Cui, Y. AR-induced ZEB1-AS1 represents poor prognosis in cholangiocarcinoma and facilitates tumor stemness, proliferation and invasion through mediating miR-133b/HOXB8. Aging 2020, 12, 1237–1255. [Google Scholar] [CrossRef] [PubMed]
- Angenard, G.; Merdrignac, A.; Louis, C.; Edeline, J.; Coulouarn, C. Expression of long non-coding RNA ANRIL predicts a poor prognosis in intrahepatic cholangiocarcinoma. Dig. Liver Dis. 2019, 51, 1337–1343. [Google Scholar] [CrossRef]
- Wang, X.P.; Song, J.; Liu, G.T.; Wang, J.J.; Guo, H.F. Upregulation of gastric adenocarcinoma predictive long intergenic non-coding RNA promotes progression and predicts poor prognosis in perihilar cholangiocarcinoma. Oncol. Lett. 2018, 16, 3964–3972. [Google Scholar] [CrossRef]
- Hu, Z.; Huang, L.; Wang, W.; Guan, C.; Zhao, Y.; Liu, L.; Jiang, X. Long Non-coding RNA FOXD2-AS1 Promotes Proliferation, Migration, and Invasion in Cholangiocarcinoma Through Regulating miR-760/E2F3 Axis. Dig. Dis. Sci. 2022, 67, 546–558. [Google Scholar]
- Jiang, X.M.; Li, Z.L.; Li, J.L.; Xu, Y.; Leng, K.M.; Cui, Y.F.; Sun, D.J. A novel prognostic biomarker for cholangiocarcinoma: CircRNA Cdr1as. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 365–371. [Google Scholar] [PubMed]
- Xu, Y.; Leng, K.; Yao, Y.; Kang, P.; Liao, G.; Han, Y.; Shi, G.; Ji, D.; Huang, P.; Zheng, W.; et al. A Circular RNA, Cholangiocarcinoma-Associated Circular RNA 1, Contributes to Cholangiocarcinoma Progression, Induces Angiogenesis, and Disrupts Vascular Endothelial Barriers. Hepatology 2021, 73, 1419–1435. [Google Scholar] [CrossRef]
- Wang, S.; Hu, Y.; Lv, X.; Li, B.; Gu, D.; Li, Y.; Sun, Y.; Su, Y. Circ-0000284 arouses malignant phenotype of cholangiocarcinoma cells and regulates the biological functions of peripheral cells through cellular communication. Clin. Sci. 2019, 133, 1935–1953. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Chen, Z.; Li, Z.; Li, S.; Wen, Z.; Cao, L.; Chen, Y.; Xue, P.; Li, H.; Zhang, D. Tumor-associated macrophages promote cholangiocarcinoma progression via exosomal Circ_0020256. Cell Death Dis. 2022, 13, 94. [Google Scholar] [CrossRef]
- Xu, Y.; Gao, P.; Wang, Z.; Su, Z.; Liao, G.; Han, Y.; Cui, Y.; Yao, Y.; Zhong, X. Circ-LAMP1 contributes to the growth and metastasis of cholangiocarcinoma via miR-556-5p and miR-567 mediated YY1 activation. J. Cell Mol. Med. 2021, 25, 3226–3238. [Google Scholar] [CrossRef]
- Li, H.; Lan, T.; Liu, H.; Liu, C.; Dai, J.; Xu, L.; Cai, Y.; Hou, G.; Xie, K.; Liao, M.; et al. IL-6-induced cGGNBP2 encodes a protein to promote cell growth and metastasis in intrahepatic cholangiocarcinoma. Hepatology 2022, 75, 1402–1419. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, Y.; Wang, W.; Yu, S.; Liu, L.; Sun, D.; Li, W.; Jiang, X. Upregulation of circ_0059961 suppresses cholangiocarcinoma development by modulating miR-629-5p/SFRP2 axis. Pathol. Res. Pract. 2022, 234, 153901. [Google Scholar] [CrossRef]
- Xu, Y.P.; Dong, Z.N.; Wang, S.W.; Zheng, Y.M.; Zhang, C.; Zhou, Y.Q.; Zhao, Y.J.; Zhao, Y.; Wang, F.; Peng, R.; et al. circHMGCS1-016 reshapes immune environment by sponging miR-1236-3p to regulate CD73 and GAL-8 expression in intrahepatic cholangiocarcinoma. J. Exp. Clin. Cancer Res. 2021, 40, 290. [Google Scholar]
- Oh, D.Y.; Ruth He, A.; Qin, S.; Chen, L.T.; Okusaka, T.; Vogel, A.; Kim, J.W.; Suksombooncharoen, T.; Ah Lee, M.; Kitano, M.; et al. Durvalumab plus Gemcitabine and Cisplatin in Advanced Biliary Tract Cancer. NEJM Evid. 2022, 1, EVIDoa2200015. [Google Scholar] [PubMed]
- Kelley, R.K.; Ueno, M.; Yoo, C.; Finn, R.S.; Furuse, J.; Ren, Z.; Yau, T.; Klumpen, H.J.; Chan, S.L.; Ozaka, M.; et al. Pembrolizumab in combination with gemcitabine and cisplatin compared with gemcitabine and cisplatin alone for patients with advanced biliary tract cancer (KEYNOTE-966): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2023, 401, 1853–1865. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.M.; Huang, X.Y.; Wu, D.; Sun, H.C.; Liang, F.; Ji, Y.; Chen, Y.; Yang, G.H.; Lu, J.C.; Meng, X.L.; et al. Toripalimab combined with lenvatinib and GEMOX is a promising regimen as first-line treatment for advanced intrahepatic cholangiocarcinoma: A single-center, single-arm, phase 2 study. Signal Transduct Target Ther. 2023, 8, 106. [Google Scholar] [CrossRef]
- Job, S.; Rapoud, D.; Dos Santos, A.; Gonzalez, P.; Desterke, C.; Pascal, G.; Elarouci, N.; Ayadi, M.; Adam, R.; Azoulay, D.; et al. Identification of Four Immune Subtypes Characterized by Distinct Composition and Functions of Tumor Microenvironment in Intrahepatic Cholangiocarcinoma. Hepatology 2020, 72, 965–981. [Google Scholar] [PubMed]
- Martin-Serrano, M.A.; Kepecs, B.; Torres-Martin, M.; Bramel, E.R.; Haber, P.K.; Merritt, E.; Rialdi, A.; Param, N.J.; Maeda, M.; Lindblad, K.E.; et al. Novel microenvironment-based classification of intrahepatic cholangiocarcinoma with therapeutic implications. Gut 2023, 72, 736–748. [Google Scholar]
- Zhu, C.; Ma, J.; Zhu, K.; Yu, L.; Zheng, B.; Rao, D.; Zhang, S.; Dong, L.; Gao, Q.; Zhang, X.; et al. Spatial immunophenotypes predict clinical outcome in intrahepatic cholangiocarcinoma. JHEP Rep. 2023, 5, 100762. [Google Scholar]
- Diggs, L.P.; Ruf, B.; Ma, C.; Heinrich, B.; Cui, L.; Zhang, Q.; McVey, J.C.; Wabitsch, S.; Heinrich, S.; Rosato, U.; et al. CD40-mediated immune cell activation enhances response to anti-PD-1 in murine intrahepatic cholangiocarcinoma. J. Hepatol. 2021, 74, 1145–1154. [Google Scholar]
- Wabitsch, S.; Tandon, M.; Ruf, B.; Zhang, Q.; McCallen, J.D.; McVey, J.C.; Ma, C.; Green, B.L.; Diggs, L.P.; Heinrich, B.; et al. Anti-PD-1 in Combination With Trametinib Suppresses Tumor Growth and Improves Survival of Intrahepatic Cholangiocarcinoma in Mice. Cell Mol. Gastroenterol Hepatol. 2021, 12, 1166–1178. [Google Scholar]
- Lau, G.; Obi, S.; Zhou, J.; Tateishi, R.; Qin, S.; Zhao, H.; Otsuka, M.; Ogasawara, S.; George, J.; Chow, P.K.H.; et al. APASL clinical practice guidelines on systemic therapy for hepatocellular carcinoma-2024. Hepatol. Int. 2024, 18, 1661–1683. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatocellular carcinoma. J. Hepatol. 2025, 82, 315–374. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Hao, X.; Liu, B.; Liu, S.; Yuan, Y. Bile liquid biopsy in biliary tract cancer. Clin. Chim. Acta 2023, 551, 117593. [Google Scholar] [CrossRef] [PubMed]
- Gou, Q.; Zhang, C.Z.; Sun, Z.H.; Wu, L.G.; Chen, Y.; Mo, Z.Q.; Mai, Q.C.; He, J.; Zhou, Z.X.; Shi, F.; et al. Cell-free DNA from bile outperformed plasma as a potential alternative to tissue biopsy in biliary tract cancer. ESMO Open 2021, 6, 100275. [Google Scholar] [CrossRef]
- Vedeld, H.M.; Grimsrud, M.M.; Andresen, K.; Pharo, H.D.; von Seth, E.; Karlsen, T.H.; Honne, H.; Paulsen, V.; Farkkila, M.A.; Bergquist, A.; et al. Early and accurate detection of cholangiocarcinoma in patients with primary sclerosing cholangitis by methylation markers in bile. Hepatology 2022, 75, 59–73. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, Z.; Chen, L.; Wang, Z.; Zhai, Y.; Qian, P.; Zhao, Y.; Zhu, L.; Jiang, H.; Wu, X.; et al. Liquid Biopsy-Based Accurate Diagnosis and Genomic Profiling of Hard-to-Biopsy Tumors via Parallel Single-Cell Genomic Sequencing of Exfoliated Tumor Cells. Anal. Chem. 2024, 96, 14669–14678. [Google Scholar]
| Trial ID 1 (Study Name) | Phase (Line) | Agents | PEP 2 | Patients | Results | Ref. |
|---|---|---|---|---|---|---|
| NCT04163900 (NuTide:121) | III (1st) | NUC-1031 + cisplatin vs. GC 3 | OS, ORR | 773 advanced BTCs patients | OS for NUC-1031/cisplatin vs. GC was. 9.2 months vs. 12.6 months. ORR for NUC-1031/cisplatin vs. GC was 18.7% vs. 12.4% (p = 0.049). | [1] |
| NCT03768414 (SWOG S1815) | III (1st) | GAP vs. GC | OS | 441 advanced BTCs patients | GAP regimen to the standard GC did not improve OS. GAP vs. GC showed improvement in PFS among patients with GBC. | [2] |
| NCT03043547 (AIO NALIRICC) | II (2nd or later) | Nanoliposomal irinotecan, fluorouracil + leucovorin vs. fluorouracil + leucovorin | PFS | 100 BTC patients with progression on gemcitabine-based therapy | Nanoliposomal irinotecan, fluorouracil + leucovorin did not improve PFS or OS. | [3] |
| jRCTs 031,180,082 4 | II (1st) | Oxaliplatin, irinotecan, leucovorin, fluorouracil | PFS | 35 advanced or recurrent BTC patients | The median PFS and OS were 7.4 and 14.7 months. The study did not meet the PEP. | [4] |
| NCT01926236 (ABC-06) | III (2nd) | FOLFOX vs. symptom control | OS | 162 BTC patients | OS was significantly longer in the FOLFOX group. | [5] |
| ChiCTR-TRC-14004733 5 | II (1st) | Gemcitabine + TS-1 vs. gemcitabine | ORR | 62 advanced BTC patients | The ORR of the combination therapy and the monotherapy were 20.0 and 9.4%, respectively. | [6] |
| NCT00262769 (ABC-02) | III (1st) | CG vs. gemcitabine | OS | 410 advanced BTC patients | OS = 11.7 months in the GC group and 8.1 months in the gemcitabine group (p < 0.001). | [7] |
| Trial ID 1 (Study Name) | Phase (Line) | Agents 2 | PEP 3 | Patients | Results | Ref. |
|---|---|---|---|---|---|---|
| NCT02699606 | II (2nd or later) | Erdafitinib | ORR | 22 advanced CCA patients with FGFR alterations | ORR = 51.7% for patients with FGFR rearrangement 12.5% for patients with FGFR short variants. | [37] |
| NCT02052778 (FOENIX-CCA2) | II (2nd or later) | Futibatinib | ORR | 103 unresectable iCCA patients with FGFR2 fusion/rearrangement | ORR = 42%, with one CR and 42 PRs. | [38] |
| NCT02924376 (FIGHT-202) | II (2nd or later) | Pemigatinib vs. conventional systemic therapy | PFS | 145 advanced CCA patients | PFS = 7.0 months for patients with FGFR2 fusions/rearrangements in the pemigatinib group, which was longer compared with the systemic therapy group. | [39] |
| NCT02150967 (BGJ398) | II (2nd or later) | Infigratinib | ORR | 108 patients with advanced CCA with FGFR2 fusions/rearrangements | ORR = 23 · 1%, with one CR and 24 PRs. | [40] |
| NCT03656536 (FIGHT-302) | III (1st) | Pemigatinib vs. GC | PFS | Advanced CCA patients with FGFR2 rearrangements | Ongoing. | [41] |
| NCT02924376 (FIGHT-202) | II (2nd or later) | Infigratinib | ORR | 147 patients with advanced CCA with/without FGFR2 alterations. | ORR = 35 · 5% in patients with FGFR2 fusions or rearrangements (three CRs and 35 PRs). | [42] |
| NCT03773302 (PROOF 301) | III (1st; One gemcitabine-based therapy is permitted) | Infigratinib vs. GC | PFS | Advanced CCA with FGFR2 rearrangements | PFS = 7.0 for patients with FGFR2 fusions/rearrangements. | [43] |
| NCT01752920 (ARQ 087) | I/II (1st, 2nd, or later) | Derazantinib | Patients with AEs (2nd, ORR) | 29 unresectable iCCA patients with FGFR2 fusion | ORR = 20.7% | [44] |
| NCT02989857 (ClarIDHy) | III (2nd or 3rd) | Ivosidenib vs. placebo | PFS | 185 advanced CCA patients with mutant IDH1 | PFS was significantly improved with ivosidenib. | [45] |
| CTRI/2019/05/019323I 4 (BEER BTC) | II/III (2nd) | Bevacizumab + erlotinib vs. active surveillance | PFS | 98 BTC patients with disease stabilization after 6 months of gemcitabine-based therapy | Bevacizumab + erlotinib improved PFS. | [46] |
| NCT01206049 (GOC-B-P) | II (1st) | Chemotherapy 6 + panitumumab (Arm A) vs. chemotherapy + bevacizumab (Arm B) | PFS | 88 BTC patients without KRAS exon 2 mutation | 42% (arm A) and 53% (arm B) of the patients showed PFS at 6 months; the primary endpoint was not met. | [47] |
| NCT01389414 (Vecti-BIL study) | II (1st) | Panitumumab + GEMOX vs. GEMOX | PFS | 89 advanced BCT patients with wild-type KRAS | No survival differences were observed between the two groups. | [48] |
| NCT00552149 (BINGO) | II (1st) | GEMOX with or without cetuximab | PFS | 150 advanced BTCs patients | 63% and 54% of the patients showed PFS > 4 months in GEMOX + cetuximab and GEMOX alone groups, respectively. | [49] |
| NCT01149122 | III (1st) | GEMOX + erlotinib vs. GEMOX | PFS | 268 metastatic BCT patients | No significant difference in PFS was noted between the two groups. Significantly more patients had an OR in the GEMOX + erlotinib group (p = 0.005). | [50] |
| NCT03212274 (NCI 10129 trial) | II (2nd or later) | Olaparib | ORR | 30 CCA patients with mutant IDH | No objective responses were seen; 27% of the patients showed a PFS of ≥6 months. | [51] |
| NCT05506943 (COMPANION-002) | II (2nd) | CTX-009 + paclitaxel vs. paclitaxel | ORR | 150 advanced BTC patients | CTX-009 with paclitaxel showed an ORR = 37.5%. | [52] |
| NCT02034110 (ROAR trial) | II (Patients with no standard treatment options) | Dabrafenib + trametinib | ORR | 43 advanced BTC patients with BRAFV600E-mutation | ORR = 53%. | [53] |
| NCT00939848 (ABC-03) | II (1st) | GC vs. GC + cediranib | PFS | 124 advanced BTCs patients | PFS = 8.0 months in the cediranib group, 7.4 months in the GC-alone group. | [54] |
| jRCT2091220423 5 (HERB; NCCH1805) | II (2nd or later) | Trastuzumab–Deruxtecan | ORR | 32 unresectable or recurrent BTC with HER2-positive | ORR = 36.4% for HER2-positive disease. ORR = 12.5% for HER2-low disease. | [55] |
| NCT01953926 (SUMMIT) | II (No treatment with any HER2-directed tyrosine kinase inhibitor) | Neratinib | ORR | 25 treatment-refractory BTC patients with HER2 mutations | ORR = 16%. | [56] |
| NCT04466891 (HERIZON-BTC-01) | II (2nd or later) | Zanidatamab | ORR by IHC | 87 BTC patients with HER2-amplified, advanced disease. | ORR= 41 · 3%. | [57] |
| NCT02091141 (MyPathway) | II (2nd) | Pertuzumab + trastuzumab | ORR | 39 treated BTC patients with HER2 amplification/overexpression | ORR = 23%. | [58] |
| Trial ID 1 (Study Name) | Phase (Line) | Agents | PEP 2 | Patients | Results | Ref. |
|---|---|---|---|---|---|---|
| NCT03875235 (TOPAZ-1) | III (1st) | Durvalumab + GC vs. GC | OS | 685 unresectable BTC patients | Durvalumab + GC showed robust and sustained OS benefits. | [134] |
| NCT02628067 (KEYNOTE-158) | II (2nd or later) | Pembrolizumab | ORR | 63 advanced BTC patients | ORR = 0% in TMB-high group, 9% in non-TMB-high group. | [65] |
| NCT04003636 (KEYNOTE-966) | III (1st) | Pembrolizumab + GC vs. GC | OS | 1069 unresectable BTC patients | Significant improvement in OS compared with GC. | [135] |
| NCT03951597 (JS001) | II 3 (1st) | Toripalimab, lenvatinib, and GEMOX | ORR | 30 unresectable iCCA patients | ORR = 80%. Twenty-three achieved PR, and one achieved CR. | [136] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nishida, N. Biomarkers and Management of Cholangiocarcinoma: Unveiling New Horizons for Precision Therapy. Cancers 2025, 17, 1243. https://doi.org/10.3390/cancers17071243
Nishida N. Biomarkers and Management of Cholangiocarcinoma: Unveiling New Horizons for Precision Therapy. Cancers. 2025; 17(7):1243. https://doi.org/10.3390/cancers17071243
Chicago/Turabian StyleNishida, Naoshi. 2025. "Biomarkers and Management of Cholangiocarcinoma: Unveiling New Horizons for Precision Therapy" Cancers 17, no. 7: 1243. https://doi.org/10.3390/cancers17071243
APA StyleNishida, N. (2025). Biomarkers and Management of Cholangiocarcinoma: Unveiling New Horizons for Precision Therapy. Cancers, 17(7), 1243. https://doi.org/10.3390/cancers17071243






