Abstract
A hemiparasitic plant, Striga hermonthica (Del.) Benth and soil nitrogen stress are the key constraints to maize (Zea mays L.) productivity in sub-Saharan Africa, where commonly cultivated maize is the normal endosperm type that is deficient in provitamin A, tryptophan and lysine (PVATL). Seventy-six extra-early maize inbreds with provitamin A, tryptophan, and lysine qualities (TZEEIORQ) were developed to address these constraints, and four checks were assessed under Striga, low and high nitrogen conditions at three locations in Nigeria. The inbreds were further genotyped with two beta-carotene hydroxylase 1 (crtRB1) markers, and their seeds were quantified for provitamin A content. Significant (p < 0.01) genetic variations were observed for grain yield and other agronomic attributes of the inbreds under varying environmental conditions. Levels of PVATL for the inbreds ranged from 2.21–10.95 µg g−1, 0.04–0.08%, and 0.19–0.39%, respectively. Beta-carotene marker, crtRB1-3′TE, was polymorphic and grouped the inbreds into two. The marker was effective in identifying inbreds with moderate provitamin A content. Inbreds TZEEIORQ 5, TZEEIORQ 52, and TZEEIORQ 55 exhibited resistance to Striga, tolerance to nitrogen stress with moderate levels of PVATL and could be invaluable sources of favorable alleles for breeding nutritionally improved maize varieties with resistance/tolerance to Striga and soil nitrogen stress.
1. Introduction
Maize (Zea mays L.) is one of the major cereal crops in sub-Saharan Africa (SSA). The contribution of maize to human calorific intake is 50%, 30% and 15% for southern, eastern, and West and Central Africa (WCA), respectively [1]. Normal endosperm maize, known to be lacking in provitamin A [2], tryptophan and lysine (PVATL) [3,4], is a major component of the food fed to babies (from two to three-month-old) and children of preschool age; this is usually done with no supplements in many developing countries [5]. In SSA, vitamin A deficiency (VAD) has been reported in about 40% and 15% of children and pregnant women, respectively [6,7]. VAD causes night blindness, increased childhood mortality, delayed growth, and a depressed immune system [8,9]. On the other hand, tryptophan and lysine are important building blocks of protein required by humans and monogastric [10]. Tryptophan deficiency causes a reduction in food intake, reduction in growth rate, impairment of skeletal development, increased pain sensitivity, increased aggression and anxiety [11], while lysine deficiency results in fatigue and reduction in growth rate, among others.
Varieties of maize (in the late, intermediate, early and extra-early maturity groups) that can mitigate the effects of VAD [12,13,14] and protein malnutrition [15,16] have been developed. However, no maize hybrid or variety with adequate provitamin A, tryptophan, and lysine contents with extra-early characteristic (matures around 80–85 days), Striga resistance and tolerance to nitrogen stress is yet to be developed and commercialized in WCA. With a population growth rate greater than 2% for many SSA countries, the need for maize has been predicted to triple by 2050 [17]. The current maize grain yield in farmers’ fields in the subregion is abysmally low to meet the projected demand. The mean maize grain yield in Nigeria is 1.8 t ha−1, while 2.4 t ha−1 has been reported for SSA; these values are lower than the 5.6 t ha−1 mean grain yield of the crop all over the world [18]. The lower yield of maize in the subregion is attributed to a combination of factors, namely: Striga hermonthica parasitism [19], stem borer attacks [20], low and declining soil nitrogen [21], recurrent drought [22], and more recently armyworm invasion [23]. Reduction in grain yield of maize due to nitrogen stress can be as high as 52% [24], while drought can result in 34% yield loss [25]. Striga hermonthica alone can cause about 53.7% [26] to 100% [27] yield losses, armyworms about 47% [28] and stem borers about 21% [29]. The widespread cultivation of maize with normal endosperm features and low grain yield of the crop is expected to exacerbate the nutritional problems in SSA.
Striga hermonthica is widespread in the savannas of WCA, where environmental conditions are considered excellent for maize production. Maize yield losses due to Striga stress varied from 30–90%, and the parasite can cause total crop failure where the infestation is severe, compelling farmers to abandon their fields [19]. Adetimirin and colleagues [26] identified ears per plant as the primary component of yield most severely affected under Striga infestation. Effects of Striga are most severe in the soils with low nutrients, particularly nitrogen, [30] which is a key constraint in major maize-producing areas of WCA [31,32]. Oikeh and colleagues [33] reported that most farmers in WCA grow maize under low nitrogen stress. This is because the soils of the area are inherently low in nitrogen, and many farmers cannot afford inorganic fertilizers to augment the low soil nitrogen [34]. About 50% reduction in maize yield has been reported due to nitrogen deficiency [35]. Therefore, developing improved maize with Striga resistance, tolerance to nitrogen stress, extra-earliness, provitamin A, and quality protein maize traits offers a sustainable and economic strategy to combat Striga and soil nitrogen stresses while improving human nutrition and health in WCA.
Seventy-six maize-inbred lines have been developed by the International Institute of Tropical Agriculture (IITA) with a view to breeding extra-early maize hybrids/varieties that combine improved levels of PVATL and tolerance/resistance to multiple stresses in SSA. However, the reactions of these newly developed inbreds have not been thoroughly investigated under Striga and nitrogen stress. Although the inbreds with appropriate modification of endosperm for tryptophan and lysine were repeatedly selected for using lightbox [16] and kernels with relatively deep orange color were assumed to have increased provitamin A levels, information on the per se PVATL concentration of each of the inbreds is lacking. The information will facilitate selecting suitable inbreds as parents in hybrid breeding programs in the subregion.
The deep orange kernel color in maize, though presumed to correlate with provitamin A levels, has been reported not to be sufficiently indicative of the levels of beta-carotene [2,36]. Safawo and colleagues [2] reported that using high-performance liquid chromatography (HPLC) is very costly in breeding maize with increased provitamin A and proposed that marker-assisted selection could be more efficient than using only maize kernel color for beta-carotene content—A major provitamin A carotenoid. Beta-carotene hydroxylase 1 (crtRB1-3′TE and crtRB1-5′TE) is one of the three major genetic markers, which play an important role during the accumulation of beta-carotene in the endosperm of maize [37]. The crtRB1-3′TE marker is a favorable DNA marker in maize for effecting an increase in the level of beta-carotene from 2 to about 10-fold in the kernels [38,39]. This study, therefore, aimed at developing and identifying Tropical Zea extra-early provitamin A quality protein maize inbred (TZEEIORQ) lines that possess tolerance/resistance to Striga and tolerance to nitrogen stress and determine the usefulness of beta-carotene hydroxylase 1 (crtRB1-3′TE and crtRB1-5′TE) in identifying inbreds with high kernel provitamin A content.
2. Materials and Methods
2.1. Germplasm
In 2007, IITA initiated a breeding program to develop varieties of maize that combine drought tolerance, tolerance to low nitrogen stress, Striga resistance and high PVALT for WCA. A variety of extra-early maize possessing Striga resistance and quality protein traits (with both yellow and orange endosperm color) was crossed to Syn-KU1409/DES/1409 (OR2), a donor of beta-carotene alleles. The resulting cross was backcrossed to multiple stress-resistant varieties, TZEE-Y STR QPM. Following the backcrossing that was aimed at introgressing genes for increased provitamin A content of maize, a total of 76 inbred lines were developed after seven cycles of selfing and selection for agronomically desirable traits.
2.2. Research Locations, Experimental Design, and Field Management
Evaluation of the 76 inbreds and the four extra-early normal endosperm checks (Supplementary Materials Table S1) was carried out under artificial Striga environment at Abuja (9°15′ N, 7°20′ E, 1700 mm annual precipitation, 300 m altitude) in 2016 and Mokwa (9°18′ N, 5°4′ E, 1100 mm annual precipitation, 457 m altitude) in 2017; the two locations are found in the southern Guinea savanna agroecological zone of Nigeria. Ethylene gas was injected into the soil following land preparation—To rid the soil of Striga seeds native to it. The gas was applied 12 cm deep into the soil. This activity was repeated at intervals of 100 cm to ensure good coverage of the field with the gas. Seeds of Striga obtained from the fields previously planted to sorghum were used for artificial infestation, following the procedure described by [31]. Well-sieved sand and the Striga seeds were mixed carefully by weight in the ratio 99:1. Before planting maize seeds per hill, each hill was infested with Striga seed-sand mixture (8.5 g containing about 5000 germinable seeds of Striga hermonthica). At four weeks after planting, NPK fertilizer (NPK 15–15–15) was applied at the rate of 30 kg ha−1 of K2O, P2O5 and N to the established plants. All other unwanted plants, apart from Striga, were controlled by hand-pulling.
In addition, the 76 inbreds and the four checks were evaluated in adjacent blocks in high and low soil nitrogen environments both at Ile-Ife (7°28′ N, 4°33′ E, 1350 mm annual precipitation, 244 m altitude) in the rainforest agroecological zone in 2016 and Mokwa in 2017. The soils at Mokwa and Ile-Ife are Luvisol and Alfisol, respectively [40]. Depletion of N from the low-N fields at Mokwa and Ile-Ife was achieved through regular planting of maize for many years and removing the stover following every harvest. At both locations, soil samples obtained (with a soil auger) before land preparation from zero to fifteen-centimeter depth were subjected to analysis. The total potassium (K), phosphorus (P) and nitrogen (N) contents of the soils were determined by Kjeldahl digestion and colorimetric method [41]. The soil analyses at Ile-Ife and Mokwa used in this study are the same as reported by [42]. Following the soil test, the total N available in the high- and low-N plots were augmented with urea to 90 and 30 kg N ha−1, respectively. Nitrogen fertilizer was applied, in two equal splits, under nitrogen experiments at 2 and 4 weeks after planting (WAP). In addition, 60 kg K ha−1 as muriate of potash (K2O) and 60 kg P ha−1 as single superphosphate (P2O5) were applied to the two N treatments at 2 WAP.
A 10 × 8 alpha lattice design, replicated two times, was used for all evaluations in the Striga, high- and low-N trials. Single row plots, 3 m long, were used under the Striga experiment. Within-row spacing was 0.40 m, while between-row spacing was 0.75 m. However, in 2016, single -ow plots 4 m long were used during evaluations in high- and low nitrogen fields. Three maize kernels were sown per hill, and the seedlings thinned to two per hill at 2 WAP. In low- and high-N fields, weeds were controlled by applying atrazine and gramoxone, supplemented with hand weeding. Fall armyworms (Spodoptera frugiperda) were controlled by using ampligo at 300 mL ha−1. Ampligo contained 100 g per liter of chlorantraniliprole and 50 g per liter of lambda-cyhalothrin.
2.3. Collection of Data on Various Characters
Data collection was carried out on plants per plot. The traits measured include days to 50% anthesis (DA) and 50% silking (DS), respectively determined as the total number of days from sowing to the time 50% of the plants had shed pollen and showed silk extrusion. Anthesis-silking interval (ASI) was calculated by subtracting DA from DS. Plant height (PLHT) represented the distance from the first tassel branch to the base of the plant, while ear height (EHT) was recorded as the distance from the node bearing the topmost ear (in prolific lines) to the base of the maize ear. In addition, stalk lodging (SLPER) was recorded as the proportion of plants with the broken stalk at (or below) the node bearing the uppermost ear. Ear aspect (EASP) was determined on a scale of 1–9, where 1 = uniform, large, clean and well-filled ears, and 9 = variable, small, rough and poorly filled ears [43]. Husk cover was assessed on a scale of 1–9, where 1 = tightly arranged husks extending beyond the tip of the ear and 9 = husks loosely arranged with ear tip exposed. The number of ears per plant (EPP) was calculated as the ratio of the total number of harvested ears for each plot to the number of harvested plants in the plot. For the high nitrogen and Striga environments, grain yield was calculated from ear weight, on the assumption of 80% shelling percentage, adjusted to 15% moisture content.
Plant aspect (PASP), assessed only under low- and high-nitrogen conditions, was scored on a scale of 1–9 using plant type and overall appeal, where 1 = excellent plant type and 9 = poor plant type. Stay-green characteristic (STGR) was determined under low soil nitrogen alone on a scale of 1–9, where 1 = nearly all foliage were lush green, and 9 = practically all foliage were dead. Determination of grain yield under low-N involved shelling of harvested ears per plot, weighing of the kernels and measuring of grain moisture content. Afterward, grain yield was calculated, per plot, using grain weight adjusted to 15% moisture content.
Additional data collected under Striga infestation were: host plant damage (by Striga) syndrome rating at 8 and 10 WAP, an indication of Striga tolerance [27,40] and the number of emerged Striga plants, which is indicative of resistance to Striga. Host plant damage was scored per plot on a scale of 1–9, where 1 = no visible damage, suggesting plant with normal growth and high tolerance to Striga, and 9 = severe damage or total collapse of the plant, indicating high susceptibility to Striga [19,44].
2.4. Identification of Beta-Carotene Rich Inbreds Using Allele-Specific Beta-Carotene Markers-crtRB1-3′TE and crtRB1-5′TE.
Leaf samples for each of the inbred genotypes were accumulated from seven to eight plants at 3 WAP. Thereafter, the samples were lyophilized using Free Zone 18 liter console dry system (Labconco Inc., Missouri, USA). Genomic DNA was extracted from the lyophilized samples using a DNA extraction protocol, modified cetyl trimethyl ammonium bromide (CTAB), described by [45]. The following markers, according to [37], were used to identify crtRB1-3′TE: (i) the forward primer (F) (5′ACACCACATGGACAAGTTCG 3′), (ii) the first reverse primer (R1) (5′ACACTCTGGCCCATGAACAC 3′) and (iii) the second reverse primer (R2) (5′ACAAGCAATACAGGGGACCAG3′). In addition, crtRB1-5′TE was identified with: (i) the forward primer (F) (5′TTAGAGCCTCGACCCTCTGTG 3′) and (ii) the reverse primer (R) (5′AATCCCTTTCCATGTTACGA 3′). A polymerase chain reaction (PCR) was conducted in 25 µL volume for each of the functional markers. The quantity of genomic DNA, beta-carotene DNA markers and other reaction mixtures used are, as shown in Table 1. The thermocycler standard cycling conditions provided by [37] were used for the PCR. Resolution of amplicons was carried out on 2% agarose gel. DNA bands were viewed on a UV Transilluminator.

Table 1.
Cocktail and reaction mix used for polymerase chain reaction. a Primer 3 R was not used under crtRB1-5′TE.
Photographs of the bands were taken and then scored for absence or presence of the favorable allele of crtRB1-3′TE gene (allele 1) and favorable allele of crtRB1-5′TE gene (allele 2) [39].
2.5. Generation and Processing of Seed Samples of Inbreds for Carotenoid, Tryptophan, and Lysine Analyses
The first two and last two plants in each plot, under high nitrogen conditions at Mokwa and Ile-Ife in 2016, were self-pollinated to develop seed samples of S8 lines that were used for carotenoids [46] tryptophan and lysine analyses. The ears were harvested at each location, dried, processed and stored at 4 °C [12]. Thereafter, two samples, each containing 60 kernels, were prepared for analyses; the first sample was used for carotenoids analysis, while the second was used for lysine and tryptophan analyses. Analyses for carotenoids, lysine and tryptophan were carried out at CIMMYT, Mexico. Samples of 20–30 maize kernels of each inbred were frozen at −80 °C until when required for analysis, at which time they were ground to (0.5 µm) powder. Carotenoids analysis was carried out using ultra-high-performance liquid chromatography (UPLC) (Waters, Milford, MA, USA) Apex Track. It involved extraction, separation, and quantification by UPLC using protocols described by [47]. Beta-carotene (13-cis, all-trans and 9-cis isomers), beta-cryptoxanthin, zeaxanthin and lutein were measured. Overall, provitamin A content of each inbred line was calculated thus: beta-carotene (13-cis + all-trans + 9-cis) + 0.5 (beta-cryptoxanthin) [46]. The amount of lysine and tryptophan in whole grains of the inbred lines were determined as reported by [47]; briefly, Kjeldahl apparatus was used to grind and de-fat whole grain sample for each inbred line, followed by the addition of papain to hydrolyze the protein. A purple coloration was induced with the addition of a combination of glacial acetic acid and H2SO4. The deepness/concentration of the induced color was quantified using a spectrophotometer at 560 nm. The percent tryptophan content of each inbred line was then obtained from the reading of the spectrophotometer converted to percent tryptophan. Two measurements were taken for each inbred line.
2.6. Statistical Analysis
Prior to statistical analyses, log transformation was carried out to achieve homogeneity of variances for data collected on Striga (hostplant) damage rating and the number of emerged Striga plants. Year–location–treatment combination was considered a test environment [48]. Analysis of variance (ANOVA) was first carried out for each research condition (Striga, low and high soil nitrogen). Thereafter, a combined ANOVA was carried out across the six environments for yield and other characters. The performance of the inbreds was determined under Striga, low nitrogen and across all research environments using the following base indices:
- (i)
- Striga base index = (2YLI + EPP − SDR1 − SDR2 − 0.5ESP1 − 0.5ESP2).where YLI = yield of maize in Striga-infested plots (kg ha−1), EPP = number of ears per plant, SDR1 and SDR2 = Striga damage ratings at 8 and 10 WAP, ESP1 and ESP2 = number of emerged Striga plants at 8 and 10 WAP [49];
- (ii)
- Low nitrogen base index = (2YLDL + EPP − (PASP + ASI + EASP + STGREN)).where YLDL = yield of low nitrogen plots (kg ha−1), EPP = ears per plant, PASP = plant aspect, ASI = anthesis-silking interval, EASP = ear aspect, STGREN = stay-green characteristic [50].
- (iii)
- Multiple-character—base-index = (2YLD + EPP − 0.5ESP1 − 0.5ESP2 − SD1 − SD2 − (PASP + EASP + STGREN)) [5]where YLD = yield across six research environments, EPP = number of ears per plant across research environments, ESP1 = number of emerged Striga plants at 8 WAP under Striga-infested environment, ESP2 = number of emerged Striga plants at 10 WAP under Striga-infested environment, SD1 = Striga damage rating at 8 WAP under Striga-infested environment, SD2 = Striga damage rating at 10 WAP under Striga-infested environment, PASP = plant aspect across high- and low nitrogen environments, EASP = ear aspect across six research environments, STGREN = stay-green characteristic under low nitrogen environment [5].
Adjusted means for each character of each genotype were standardized to minimize the effects of the different scales used to measure them. In each case, a positive base index value suggested that the inbred was tolerant to that stress, whereas negative values were indicative of the susceptibility of the inbreds to the stress [5]. Chi-squared analysis was carried out to determine if the groups formed from the results of the molecular screening were associated with the amount of provitamin A in the inbreds determined by HPLC. In addition, stepwise regression of total provitamin A on provitamin A carotenoids was carried out.
3. Results
3.1. Mean Squares of Inbreds Evaluated in Striga-Infested, Low- and High Nitrogen Conditions
Results of ANOVA across six research conditions (two environments each of Striga infestation, low and high nitrogen) showed significant (p < 0.01) environment, inbred and inbred × environment variance for all traits except inbred × environment interaction mean square for EPP (Table 2). ANOVA under Striga-infested environments indicated significant (p < 0.01) environment, inbred and inbred × environment interaction variance for grain yield and other traits (Table 3). Similarly, the ANOVA under low nitrogen revealed significant (p < 0.01) environment, inbred and inbred × environment interaction variance for all characters, except environment variance for STGR (Table 3). ANOVA under high nitrogen environments indicated significant (p < 0.01) environment, inbred, and inbred × environment interaction mean squares for all traits, except environment mean squares for EPP and PASP, and inbred × environment interaction variance for PLHT, EHT and EPP (Table 3).

Table 2.
Mean squares for grain yield and other agronomic traits of extra-early provitamin A quality protein maize inbreds evaluated across six (low-N, Striga-infested and high-N) environments at Ile-Ife, Mokwa and Abuja in Nigeria in 2016 and 2017.

Table 3.
Mean squares for grain yield and other agronomic traits of extra-early provitamin A quality protein maize inbreds evaluated under Striga, low-N and high-N environments at Abuja, Mokwa and Ile-Ife in Nigeria in 2016 and 2017.
3.2. Performance of Inbreds for Grain Yield and Other Agronomic Traits and Identification of Striga-Resistant/Tolerant and Nitrogen Stress-Tolerant Inbreds
Out of the 80 inbreds evaluated under Striga, 34 were resistant/tolerant to Striga based on their positive Striga base index values (Table 4). Eighteen of the 34 lines had higher Striga base index values than the best check-TZEEI 73. The index values for the top ranking 15 lines ranged from 3.97 to 11.34 compared to 3.38 obtained for TZEEI 73 (Table 4). A total of 41 lines were identified as low nitrogen tolerant based on their low nitrogen base index values (Table 5). Of these lines, 12 had higher low nitrogen base index values (4.93–9.28) than TZEEI 73 (4.87)—The best Striga and low nitrogen tolerant checks were: TZEEIORQ 57, TZEEIORQ 21, TZEEIORQ 64, TZEEIORQ 53, TZEEIORQ 43, TZEEIORQ 55, TZEEIORQ 63, TZEEIORQ 20, TZEEIORQ 51, TZEEIORQ 42, TZEEIORQ 52, and TZEEIORQ 14 (Table 5).

Table 4.
Grain yield and other agronomic traits of 76 extra-early provitamin A quality protein maize inbreds with four checks evaluated under Striga-infested environments at Abuja and Mokwa in Nigeria in 2016 and 2017.

Table 5.
Grain yield and other agronomic traits of 76 extra-early provitamin A/QPM inbreds with four checks evaluated across low-N environments at Ile-Ife and Mokwa between 2016 and 2017 in Nigeria.
Averaged over inbred lines, grain yield was 1064 kg ha−1 under Striga-infested environment, 1257 kg ha−1 under low nitrogen and 2120 kg ha−1 under high nitrogen; thus, compared to high nitrogen plots, grain yield reduction due to Striga and low nitrogen averaged 49.8% and 40.7%, respectively (Table 6). The number of ears per plant for these environments was 0.7, 0.7, and 0.8, respectively. Grain yield across the environments ranged from 658 kg ha−1 for TZEEIORQ 16 to 2337 kg ha−1 for TZEEIORQ 63 with an average of 1492 kg ha−1, while ears per plant ranged from 0.3 for TZEEIORQ 16 to 1.3 for TZEEIORQ 57 with a mean of 0.7. Ear aspect was lowest for TZEEIORQ 62 and TZEEIORQ 64 (3.2) and highest for TZEEIORQ16 (6.9) (Table 7). There were no significant differences in grain yield among the top five ranking inbreds (TZEEIORQ 57, TZEEIORQ 63, TZEEIORQ 42, TZEEIORQ 55, and TZEEIORQ 64), identified as resistant/tolerant to both Striga and nitrogen stresses based on the multiple character base index, and the best check TZEEI 73 (Table 6). Inbred TZEEIORQ 57 had the highest mean value (1.3) for ears per plant across research environments, but it had the lowest mean values for stay-green characteristic (1.7) under low nitrogen stress and Striga (host–plant) damage rating at 8 WAP (2.7) under Striga condition. The expression of these desirable attributes by the inbred contributed to its excellent performance across research environments.

Table 6.
Grain yield and other agronomic traits of 20 best and 5 worst extra-early maturing provitamin A quality protein maize inbreds evaluated under Striga-infested, low-N and high-N environments.

Table 7.
Tropical Zea extra-early maturing provitamin A quality protein maize inbreds with and without favorable allele for provitamin A marker crtRB1-3′TE and corresponding levels of beta carotene and total provitamin A as determined by high-performance liquid chromatography.
3.3. Molecular Screening and Biochemical Analyses of Inbreds Derived from Tropical Zea Extra-Early Striga-Resistant Maize Population Improved for Provitamin A and Quality Protein Maize Properties
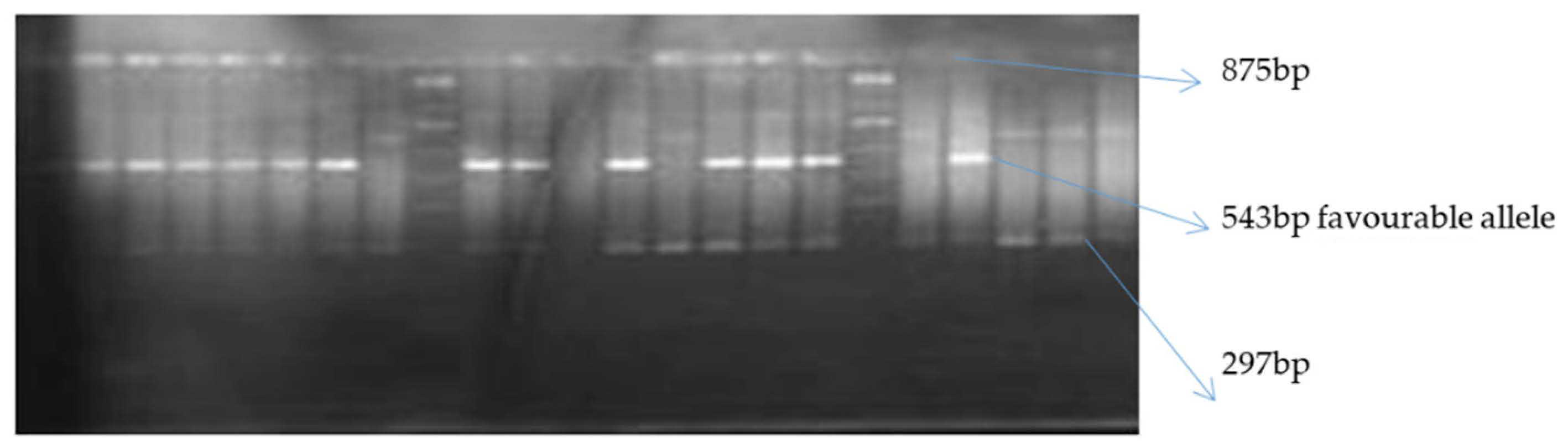
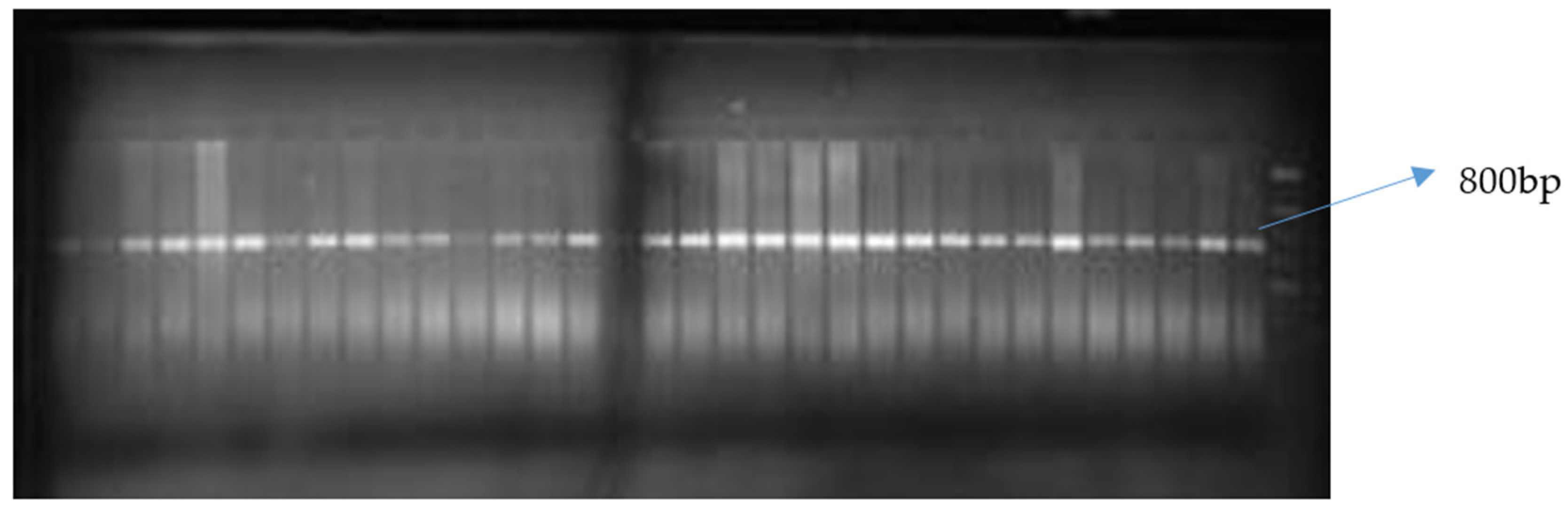
Of the two allele-specific provitamin A markers used, only crtRB1-3′TE was polymorphic among the inbred lines (Figure 1 and Figure 2). The polymorphic marker differentiated the 76 biofortified lines into two groups (Table 7). The first group comprised 16 inbred lines with the favorable allele of crtRB1-3′TE, while the second group of 60 inbred lines was without the favorable provitamin A allele. HPLC did not detect beta carotene and provitamin A in two of the 16 samples possessing the favorable allele of crtRB1-3′TE (Table 7). Levels of provitamin A in all the inbred lines analyzed ranged from 2.21 µg g−1 for TZEEIORQ 27 to 10.95 µg g−1 for TZEEIORQ 54 with an average of 6.18 µg g−1. A total of 12 of the 16 inbreds with the favorable allele of crtRB1-3′TE marker had provitamin A levels greater than the mean provitamin A value (Table 7). Two inbred lines (TZEIORQ 64 and TZEEIORQ 73) with the favorable provitamin A allele had lower levels of provitamin A than the average provitamin A value for the inbred lines analyzed (Table 7). Chi-squared analysis showed a significant association (p < 0.01) between the desirable provitamin A marker allele and the provitamin A content of the inbred lines (Table 8). Stepwise regression of provitamin A carotenoids (beta-carotene and beta-cryptoxanthin) on the overall provitamin A in the inbreds showed significant contributions, of 81.9% and 18.3% for beta-carotene and beta-cryptoxanthin, respectively, to the levels of total provitamin A in the inbreds studied (Table 9). Tryptophan content varied from 0.04% in TZEEIORQ 53 to 0.08% in TZEEIORQ 72 with an average of 0.05%, while lysine content ranged from 0.19% in TZEEIORQ 50 to 0.39% in TZEEIORQ 74 with a mean of 0.27% (Table 10). In all, 39 inbred lines had provitamin A levels above 6.18 µg g−1; these inbred lines showed different combinations of Striga tolerance/resistance, low nitrogen stress tolerance, provitamin A, tryptophan and lysine contents (Table 7 and Table 10). Three lines viz. TZEEIORQ 55, TZEEIORQ 5 and TZEEIORQ 52 combined high provitamin A content (>10.0 µg g−1), tolerance/resistance to Striga and low nitrogen stress with improved tryptophan and lysine contents—Not lower than the average values of 0.05 and 0.27%, respectively (Table 7 and Table 10). An inbred line with a positive base index value was identified as resistant/tolerant to the stress, while a negative base index value indicated susceptibility to the stress.
Figure 1.
Gel image of crtRB1-3′TE PCR product resolved on a 2% agarose gel indicating polymorphism of the marker for beta carotene allele.
Figure 2.
Gel image of crtRB1-5′TE PCR product resolved on a 2% agarose gel indicating monomorphism of the marker for beta carotene allele.

Table 8.
Presence or absence of desirable allele of beta-carotene hydroxylase gene (crtRB1-3′TE) in the 76 extra-early provitamin A quality protein maize inbreds and provitamin A content of the inbred lines determined by high-performance liquid chromatography.

Table 9.
Regression of provitamin A carotenoids on total provitamin A content of extra-early provitamin A quality protein maize inbreds analyzed using high-performance liquid chromatography.

Table 10.
Levels of tryptophan and lysine in the extra-early provitamin A quality protein maize inbreds with the most stable check and their responses to Striga and low-N, determined by Striga and low-N base index values.
4. Discussion
In the moist savanna of WCA, where maize is considered to have great potential, Striga hermonthica [19], soil nitrogen stress [21], and recurrent drought [22] limit maize production and productivity. In the present study, Striga hermonthica reduced grain yield by 49.8%, while the average yield loss due to nitrogen stress was 40.7%. These stresses present unique, challenging conditions as evidenced by the significant variations obtained for grain yield and the majority of the research environments’ characteristics. Abiotic and biotic stresses in the WCA savanna often occur simultaneously in farmers’ fields [31], resulting in geometric losses in yield. In an earlier study, [26] reported a 53.7% reduction in maize grain yield due to Striga stress alone and 85% yield loss when in combination with drought stress. Although the grain yield reduction obtained under Striga infestation in this study is lower than the 68% yield reduction reported by [51], it is higher than the 39% reported by [49]. In addition, the 40.7% yield reduction under low nitrogen stress in this study is higher than the 35% reduction in yield reported by [32]. The differences in yield reduction observed in this study and those earlier reported could be attributed to the dissimilarities in the genotypes evaluated, severity and uniformity of the stresses and other management practices.
Of the nutrient elements in tropical soils, nitrogen is the most restraining [21,32]. Low soil nitrogen is widespread in WCA, where average fertilizer application is 6.11 kg ha−1, an amount lower than the 8.89 kg ha−1 average fertilizer consumption in SSA [52], a consequence of non-availability and/or non-affordability by the predominant resource-limited farmers in the area [34]. The cultivation of extra-early maize varieties is one of the strategies for mitigating end-of-season drought. Consequently, developing extra-early maturing maize germplasm, which combines tolerance/resistance to Striga and nitrogen stress, is an important approach for improving maize productivity under the biotic and abiotic stresses of low nitrogen, Striga and drought in WCA. The incidence of Striga in farmlands is erratic and often influenced by environmental factors. Maize cultivars developed for the savanna of WCA, therefore—in addition to showing resistance/tolerance to these stresses—must also demonstrate capability for high-performance in nonstress environments.
In the current study, the inbreds TZEEIORQ 55, TZEEIORQ 52 and TZEEIORQ 5 were among the thirteen most promising extra-early provitamin A quality protein maize inbreds identified across the research environments (evidenced by their relatively high and positive multiple-character base index values). Coupled with their performance under each of the stresses, these inbreds showed tolerance/resistance to Striga, tolerance to low nitrogen, and better performance in stress-free environments. The inbreds also had moderate to relatively high levels of tryptophan and lysine. The consistently higher base index values of TZEEIORQ 55 than those of TZEEI 73, in each and across environments, indicated the outstanding performance of the inbred across the research conditions. The different base indices used in this study integrated several important traits under the respective stresses. For example, under Striga, high yield, reduced host–plant damage (tolerance index), and reduced number of emerged Striga plants (resistance index) were important for sustainability. While tolerance ensured high yield and low host damage, resistance reduced the number of emerged parasites and buildup of the seeds of Striga in the soil [44,52].
The significant genotype × environment interaction obtained in the current study indicated that the genotypes varied in their response patterns to each of the stresses and nonstress conditions. In effect, performance under the conditions in any of the research environments cannot be used to extrapolate performance in other environments. These results justified our approach of developing a considerable number of lines from the source population and screening the lines for their responses under the different stresses and in stress-free environments, thus allowing identifying lines that possessed alleles for both tolerance/resistance to the stresses and good performance under nonstress conditions. Ifie [53] reported significant genotype × environment interaction for yield and other traits of 100 early-maturing maize inbreds studied under Striga and low nitrogen environments. Similarly, Akaogu and colleagues [49] observed significant genotype × environment interaction for many characters of 90 extra-early yellow maize inbreds in Striga-free and Striga-infested environments. The similarity in the results of this study and those of the previous authors suggests that the environments where the maize genotypes were evaluated might be similar.
While breeding for improved levels of provitamin A in maize, several workers have identified and used different molecular markers linked to provitamin A carotenoids [37,39]. Of the two provitamins A markers used in the present study, only crtRB1-3′TE was polymorphic among the inbreds. In a previous study, [12] reported polymorphism for both markers. The variation in the results of this study and that of [12] might be attributed to differences in the genetic materials used for the studies. The allele 1 of crtRB1-3′TE has been reported to bring about a 2 to 10-fold favorable increase in kernel beta-carotene in maize [38,39]. The range of provitamin A levels (2.21–10.95 µg g−1) and the average beta-cryptoxanthin (5.25 µg g−1) observed in the present study are comparable to values (provitamin A levels = 3.01–11.90 µg g−1; average beta-cryptoxanthin = 4.23 µg g−1) obtained by [54]. The similarity in our results is suggestive of the fact that the inbreds used in this study may be genetically related, concerning provitamin A, to those studied by [54]. The significant chi-squared value obtained between the results of the molecular markers and provitamin A content of the inbred lines indicates that the marker was associated with the levels of provitamin A in the inbreds. This suggests that the marker was effective in identifying inbreds with increased levels of provitamin A.
All the inbreds used in the current study, with or without the favorable marker alleles, had lower provitamin A content than the HarvestPlus target of 15 µg g−1 [55]. The lines with relatively high content of provitamin A in this study (7.50–10.25 µg g−1) can, therefore, be regarded as being moderate in provitamin A. Of the 60 inbred lines without the favorable marker allele, seven had provitamin A content in the range 7.50–10.25 µg g−1. This suggests the involvement and effectiveness, in some of the inbred lines, of other provitamin A carotenoid(s) apart from the one linked to the favorable allele of the crtRB1-3′TE. The results of the stepwise regression analyses of the provitamin A carotenoids on the total provitamin A levels in the inbreds revealed that β-cryptoxanthin (with half the vitamin A activity of β-carotene) also contributed significantly to the increased levels of total provitamin A in the inbred lines with or without the favorable allele of crtRB1-3′TE. Similar to our result is the finding of [54], who observed a strong positive relationship between β-cryptoxanthin and provitamin A concentration in maize.
Inbreds TZEEIORQ 55, TZEEIORQ 72 and TZEEIORQ 74 had relatively high levels of tryptophan and lysine, indicating that they possess quality protein properties. Kostadinovic [56] reported a similar range of 0.06–0.08% tryptophan for 13 maize lines. In general, inbreds TZEEIORQ 58, TZEEIORQ 55, TZEEIORQ 5, TZEEIORQ 52, TZEEIORQ 57, TZEEIORQ 62, TZEEIORQ 72, TZEEIORQ 59 and TZEEIORQ 54 had the highest levels of PVATL.
The lines developed in the present study, which are the first set of extra-early maize lines with combined resistance/tolerance to Striga and tolerance to nitrogen stress and moderate levels of PVATL, showed exploitable genetic variation for these traits. In addition to their use in developing open-pollinated maize varieties/hybrids for increasing maize productivity in WCA, the lines offer promise for addressing the prevalent problems of VAD and protein deficiency in the subregion. Opportunities exist to further improve the levels of these nutrients in the inbreds through selection.
5. Conclusions
Exploitable genetic variability exists for grain yield and other agronomic characters of the TZEEIORQ lines studied under Striga, low- and high-nitrogen soil conditions. The beta-carotene marker, crtRB1-3′TE, was polymorphic and grouped the inbreds into two. The marker was effective in identifying inbreds with moderate provitamin A content. Inbred lines TZEEIORQ 55, TZEEIORQ 52 and TZEEIORQ 5 combined resistance/tolerance to Striga and nitrogen stress with improved performance under high nitrogen conditions. These inbreds are invaluable pools of favorable alleles in breeding for extra-earliness, Striga resistance, nitrogen stress tolerance, and PVATL.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/agronomy11050891/s1, Table S1. Pedigree of 76 extra-early maturing provitamin A quality protein maize inbreds derived from a tropical Zea Striga resistant provitamin A quality protein maize population along with four checks used in this study.
Author Contributions
Conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, software, original draft, review & editing, S.A.O.; conceptualization, data curation, investigation, methodology, funding acquisition, project administration, resources, supervision, validation, review & editing, B.B.-A.; supervision, validation, review and editing, V.O.A.; data curation, formal analysis, methodology, investigation, review and editing, N.U.; Conceptualization, funding acquisition, methodology, resources, review and editing, M.G. All authors contributed to manuscript revision, as well as read and approved the submitted version. All authors have read and agreed to the published version of the manuscript.
Funding
The information presented in this article is a part of the PhD research of the first author funded by the Pan African University Institute of Life and Earth Sciences (Including Health and Agriculture), University of Ibadan, Ibadan, Nigeria and the Drought Tolerant Maize for Africa Project of the Bill and Melinda Gates Project (OPP1134248), IITA, Ibadan, Nigeria. The funding bodies did not participate in the design, collection, analysis, interpretation of data, and writing the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets used/or analyzed during the current study, as well as the materials used, are available from the corresponding author on reasonable request.
Acknowledgments
The authors, especially the first author, are grateful to the organizations that funded the PhD research. The technical supports by the staff of the Maize Improvement Program and Bioscience Center of IITA, in the field and laboratory, respectively, are appreciated.
Conflicts of Interest
The authors declare that they have no competing interests.
References
- Adebayo, M.A.; Menkir, A. Combining ability of adapted and exotic drought-tolerant maize inbred lines under full irrigation and rainfed conditions in Nigeria. J. Crop Improv. 2015, 29, 117–130. [Google Scholar] [CrossRef]
- Safawo, T.; Senthil, N.; Raveendran, M.; Vellaikumar, S.; Ganesan, K.N.; Nallathambi, G.; Saranya, S.; Shobhana, V.G.; Abirami, B.V.; Gowri, E. Exploitation of natural variability in maize for β-Carotene content using HPLC and gene specific markers. Electron. J. Plant Breed. 2010, 1, 548–555. [Google Scholar]
- Nuss, E.T.; Tanumihardjo, S.A. Quality protein maize for Africa: Closing the protein inadequacy gap in vulnerable populations. Adv. Nutr. 2011, 2, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Chua, H.D.; Le, N.Q. Improving nutritional quality of plant proteins through genetic engineering. Curr. Genom. 2016, 17, 220–229. [Google Scholar]
- Badu-Apraku, B.; Fakorede, M.A.B.; Talabi, A.O.; Oyekunle, M.; Akaogu, I.C.; Akinwale, R.O.; Annor, B.; Melaku, G.; Fasanmade, Y.; Aderounmu, M. Gene action and heterotic groups of early white quality protein maize inbreds under multiple stress environments. Crop Sci. 2016, 56, 183–199. [Google Scholar] [CrossRef]
- Aguayo, V.M.; Baker, S.K. Vitamin A deficiency and child survival in sub-Saharan Africa: A reappraisal of challenges and opportunities. Food Nutr. Bull. 2005, 26, 348–355. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Global prevalence of vitamin A deficiency in populations at risk 1995–2005. In WHO Global Database on Vitamin A Deficiency; World Health Organization: Geneva, Switzerland, 2009; Available online: http://whqlibdoc.who.int/publications/2009/9789241598019_eng.pdf (accessed on 10 November 2015).
- West, K.P., Jr.; Darnton-Hill, I. Vitamin A deficiency. In Nutrition and Health in Developing Countries, 2nd ed.; Semba, R.D., Bloem, M.W., Eds.; Nutrition and Health Series; Humana Press: Totowa, NJ, USA, 2008; pp. 377–434. [Google Scholar]
- Muthayya, S.; Hyu Rah, J.; Sugimoto, J.; Roos, F.; Kraemer, K.; Black, R. The global hidden hunger indices and maps: An advocacy tool for action. PLoS ONE 2013, 8, e67860. [Google Scholar] [CrossRef]
- Huang, S.; Adams, W.; Zhou, Q.; Malloy, K.; Voyles, D.; Anthiny, J.; Kriz, A.; Luethy, M. Improving the nutritional quality of maize protein by expressing sense and antisense genes. J. Agric. Chem. 2004, 52, 1958–1964. [Google Scholar] [CrossRef]
- Moehn, S.; Pencharz, P.B.; Ball, R.O. Lessons learned regarding symptoms of tryptophan deficiency and excess from animal requirement studies. Am. Soc. Nutr. 2012, 142, 2231S–2235S. [Google Scholar] [CrossRef]
- Azmach, G.; Gedil, M.; Menkir, A.; Spillane, C. Marker-trait association analysis of functional gene markers for provitamin A levels across diverse tropical yellow maize inbred lines. BMC Plant Biol. 2013, 13, 227. Available online: http://www.biomedcentral.com/1471-2229/13/227 (accessed on 15 July 2015). [CrossRef]
- Badu-Apraku, B.; Oyekunle, M.; Fakorede, M.A.B.; Vroh, I.; Akinwale, R.O.; Aderounmu, M. Combining ability, heterotic patterns and genetic diversity of extra-early yellow inbreds under contrasting environments. Euphytica 2013, 192, 413–433. [Google Scholar] [CrossRef]
- Menkir, A.; Maziya-Dixon, B.; Mengesh, W.; Rocheford, T.; Alamu, E.O. Accruing genetic gain in pro-vitamin A enrichment from harnessing diverse maize germplasm. Euphytica 2017, 213, 105. [Google Scholar] [CrossRef]
- Fan, X.M.; Tan, J.; Yang, J.Y.; Chen, H.M. Combining ability and heterotic grouping of ten temperate, subtropical, and tropical quality protein maize inbreds. Maydica 2004, 49, 267–272. [Google Scholar]
- Krivanek, A.F.; De Groote, H.; Gunaratna, N.S.; Diallo, A.O.; Friesen, D. Breeding and disseminating quality protein maize (QPM) for Africa. Afr. J. Biotechnol. 2007, 6, 312–324. [Google Scholar]
- Van Ittersum, M.K.; van Bussel, L.G.J.; Wolf, J.; Grassini, P.; van Wart, J.; Guilpart, N.; Claessens, L.; de Groot, H.; Wiebe, K.; Mason-D’Croz, D.; et al. Can Sub-Saharan Africa Feed Itself? Proc. Natl. Acad. Sci. USA 2016, 113, 14964–14969. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization Statistics (FAOSTAT). FAOSTAT Database 2017. Available online: http://www.fao.org/faostat/en/#data (accessed on 12 December 2017).
- Badu-Apraku, B.; Annor, B.; Oyekunle, M.; Akinwale, R.O.; Fakorede, M.A.B.; Talabi, A.O.; Akaogu, I.C.; Melaku, G.; Fasanmade, Y. Grouping of early maturing quality protein maize inbreds based on SNP markers and combining ability under multiple environments. Field Crop. Res. 2015, 183, 169–183. [Google Scholar] [CrossRef]
- Ajala, S.O.; Kling, J.G.; Menkir, A. Full-sib family selection in maize populations for tolerance to low soil nitrogen. J. Crop Improv. 2012, 26, 581–598. [Google Scholar] [CrossRef]
- Edmonds, D.E.; Abreu, S.L.; West, A.; Caasi, D.R.; Conley, T.O.; Daft, M.C.; Desta, B.; England, B.B.; Farris, C.D.; Nobles, T.J.; et al. Cereal nitrogen use efficiency in sub-Saharan Africa. J. Plant Nutr. 2009, 2, 2107–2122. [Google Scholar] [CrossRef]
- Meseka, S.K.; Menkir, A.; Ajala, S. Genetic analysis of performance of maize inbred lines under drought stress. J. Crop Improv. 2011, 25, 521–539. [Google Scholar] [CrossRef]
- Day, R.; Abrahams, P.; Bateman, M.; Beale, T.; Clottey, V.; Cock, M.; Colmenarez, Y.; Corniani, N.; Early, R.; Godwin, J.; et al. Fall armyworm: Impacts and implications for Africa. Outlooks Pest Manag. 2017, 213, 105. [Google Scholar] [CrossRef]
- Meseka, S.K.; Menkir, A.; Ibrahim, A.E.S.; Ajala, S.O. Genetic analysis of performance of maize inbred lines selected for tolerance to drought under low nitrogen. Maydica 2006, 51, 487–495. [Google Scholar]
- Azeez, J.O.; Chikoye, D.; Kamara, A.Y.; Menkir, A.; Adetunji, M.T. Effect of drought and weed management on maize genotypes and the tensiometric soil water content of an euric nitisol in south western Nigeria. Plant Soil 2005, 276, 61–68. [Google Scholar] [CrossRef]
- Adetimirin, V.O.; Aken’Ova, M.E.; Kim, S.K. Effects of Striga hermonthica on yield components in maize. J. Agric. Sci. 2000, 135, 185–191. [Google Scholar] [CrossRef]
- Fajemisin, J.M. The Revolutionary Trend of Maize in Nigeria: My Memoir; Phaloray Book Works: Oluyole Estate, Nigeria, 2014; Volume 6, p. 118. [Google Scholar]
- Kassie, M.; Wossen, T.; De Groot, H.; Tefera, T.; Sevgan, S.; Balew, S. Economic impacts of fall armyworm and its management strategies: Evidence from southern Ethiopia. Eur. Rev. Agric. Econ. 2020, 47, 1473–1501. [Google Scholar] [CrossRef]
- De Groot, H. Maize yield losses from stemborers in Kenya. Insect Sci. Its Appl. 2002, 22, 89–96. [Google Scholar] [CrossRef]
- Adetimirin, V.O.; Kim, S.K.; Aken’Ova, M.E. Expression of mature plant resistance to Striga hermonthica in maize. Euphytica 2000, 115, 149–158. [Google Scholar] [CrossRef]
- Kim, S.K.; Adetimirin, V.O. Responses of tolerant and susceptible maize varieties to timing and rate of nitrogen under Striga hermonthica infestation. J. Agron. 1997, 89, 38–44. [Google Scholar] [CrossRef]
- Abe, A.; Adetimirin, V.O.; Menkir, A.; Moose, S.P.; Olaniyan, A.B. Performance of tropical maize hybrids under conditions of low and optimum levels of nitrogen fertilizer application—Grain yield, biomass production and nitrogen accumulation. Maydica 2013, 58, 141–150. [Google Scholar]
- Oikeh, S.O.; Horst, W.J. Agro-physiological responses of tropical maize cultivars to nitrogen fertilization in the moist savanna of West Africa. In Plant-Nutrition, Food Security and Sustainability of Agro-Ecosystems; Horst, W.J., Kamh, M., Jibrin, J.M., Chude, V.O., Eds.; Kluwer Academic Publish: Dordrecht, The Netherlands, 2001; pp. 804–805. [Google Scholar]
- Talabi, A.O.; Badu-Apraku, B.; Fakorede, M.A.B. Genetic variances and relationship among traits of an early maturing maize population under drought-stress and low nitrogen environments. Crop Sci. 2017, 57, 1–12. [Google Scholar] [CrossRef]
- Wolfe, D.W.; Henderson, D.W.; Hsiao, T.C.; Alvio, A. Interactive water and nitrogen effects on maize. II. Photosynthetic decline and longevity of individual leaves. Agron. J. 1988, 80, 865–870. [Google Scholar] [CrossRef]
- Muthusamy, V.; Hossain, F.; Thirunavukkarasu, N.; Saha, S.; Agrawal, P.K.; Guleria, S.K.; Gupta, H.S. Genetic variability and interrelationship of kernel carotenoids among indigenous and exotic maize (Zea mays L.) inbreds. Cereal Res. Commun. 2015, 43, 567–578. [Google Scholar] [CrossRef]
- Yan, J.; Kandianis, C.B.; Harjes, E.C.; Bai, L.; Kim, E.H.; Yang, X.; Skinner, D.J.; Fu, Z.; Mitchell, S.; Li, Q.; et al. Rare genetic variation at Zea mays crtRB1 increases β-carotene in maize grain. Nat. Genet. 2010, 42, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Babu, R.; Rojas, N.P.; Gao, S.; Yan, J.; Pixley, K. Validation of the effects of molecular marker polymorphisms in LcyE and crtRB1 on provitamin A concentrations for 26 tropical maize populations. Theor. Appl. Genet. 2013, 126, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Sagare, D.B.; Shetti, P.; Reddy, S.S.; Surender, M.; Pradeep, T. Identification of β-carotene rich maize inbreds using PCR—Based assay for crtRB1-3′TE allele. Int. J. Sci. Nat. 2015, 6, 441–443. [Google Scholar]
- Soil Survey Staff. Soil Taxonomy: A Basic System of Soil Classification for Making and Interpreting Soil Surveys, 2nd ed.; USDA-NRCS Agriculture Handbook, No. 436; U.S. Government Publishing Office: Washington, DC, USA, 1999.
- Bremner, J.M.; Mulvaney, C.S. Nitrogen-total. In Methods of Soil Analysis; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Part 2. Agronomy, No. 9; ASA: Madison, WI, USA, 1982; pp. 595–624. [Google Scholar]
- Oyekale, S.A.; Badu-Apraku, B.; Adetimirin, V.O. Combining ability of extra-early biofortified maize inbreds under Striga infestation and low soil nitrogen. Crop Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K. Breeding maize for Striga tolerance and the development of a field infestation technique. In Combating Striga in Africa; Kim, S.K., Ed.; IITA: Ibadan, Nigeria, 1991; pp. 96–108. [Google Scholar]
- Kim, S.K. Genetics of maize tolerance of Striga hermonthica. Crop Sci. 1994, 34, 900–907. [Google Scholar] [CrossRef]
- Doyle, J.J.; Doyle, J.L. Isolation of plant DNA from fresh tissue. Focus 1990, 12, 13–15. [Google Scholar]
- Suwarno, W.B.; Pixley, K.V.; Palacios-Rojas, N.; Kaeppler, S.M.; Babu, R. Formation of heterotic groups and understanding genetic effects in pro-vitamin A biofortified maize breeding program. Crop Sci. 2014, 54, 14–24. [Google Scholar] [CrossRef]
- Galicia, L.; Nurit, E.; Rosales, A.; Palacios-Rojas, N. Laboratory Protocols 2008: Maize Nutrition Quality and Plant Tissue Analysis Laboratory; CIMMYT: Mexico City, Mexico, 2009. [Google Scholar]
- Ukalski, K.; Klisz, M. Application of GGE biplot graphs in multi-environment trials on selection of forest trees. Folia For. Pol. Ser. A For. 2016, 58, 228–239. [Google Scholar] [CrossRef]
- Akaogu, I.C.; Badu-Apraku, B.; Adetimirin, V.O.; Vroh-Bi, I.; Oyekunle, M.; Akinwale, R.O. Genetic diversity assessment of extra-early maturing yellow maize inbreds and hybrid performance in Striga-infested and Striga-free environments. J. Agric. Sci. 2013, 151, 519–537. [Google Scholar] [CrossRef]
- Badu-Apraku, B.; Akinwale, R.O.; Ajala, S.O.; Menkir, A.; Fakorede, M.A.B.; Oyekunle, M. Relationships among the traits of tropical early maize cultivar in contrasting environments. Agron. J. 2011, 103, 717–729. [Google Scholar] [CrossRef]
- Kim, S.K.; Adetimirin, V.O.; The, C.; Dossou, R. Yield losses in maize due to Striga hermonthica in West and Central Africa. Int. J. Pest Manag. 2002, 48, 211–217. [Google Scholar] [CrossRef]
- Badu-Apraku, B.; Menkir, A.; Lum, A.F. Genetic variability for grain yield and components in an early tropical yellow maize population under Striga hermonthica infestation. J. Crop Improv. 2007, 20, 107–122. [Google Scholar] [CrossRef]
- Ifie, B.E. Genetic Analysis of Striga Resistance and Low Soil Nitrogen Tolerance in Early Maturing Maize (Zea mays L.) Inbred Lines. Ph.D. Thesis, West Africa Centre for Crop Improvement, University of Ghana, Accra, Ghana, 2013. [Google Scholar]
- Venado, R.E.; Owens, B.F.; Ortiz, D.; Lawson, T.; Mateos-Hernandez, M.; Ferruzzi, M.G.; Rocheford, T.R. Genetic analysis of provitamin A carotenoid, β-cryptoxanthin concentration, and relationship with other carotenoids in maize grain (Zea mays L.). Mol. Breed. 2017, 37, 1–18. [Google Scholar] [CrossRef]
- Bouis, H.E.; Saltzman, A. Improving nutrition through biofortification: A review of evidence from HarvestPlus, 2003 through 2016. Glob. Food Secur. 2017, 12, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Kostadinovic, M.; Ignjatovic-Micic, D.; Vancetovic, J.; Ristic, D.; Bozinovic, S.; Stankovic, G.; Drinic, S.M. Development of high tryptophan maize near isogenic lines adapted to temperate regions through marker assisted selection—Impediments and Benefits. PLoS ONE 2016, 11, e0167635. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).