Abstract
Aloe vera is an economic crop due to the presence of a gel-like substance inside the leaves, which contains polysaccharides, vitamins, proteins, fats, and amino acids. There is an ongoing need for this plant and its products for diverse food and pharmaceutical industries. To meet good agricultural practices, we studied the effect of manure application (0%, 10%, and 20% of v/v of soil) and foliar application with distilled water, salicylic acid (SA, 13.8 mg L−1 equivalent to 100 µM), cerium oxide nanoparticles (CeO2-NPs, 50 mg L−1), and cerium oxide salicylic acid nanoparticles (CeO2: SA-NPs; 100 µM + 50 mg L−1) on the growth and physiological responses of Aloe vera, and a factorial experiment was arranged based on a completely randomized design with three replications at Azarbaijan Shahid Madani University, Iran. The results showed that foliar application of CeO2: SA-NPs with the soil-based application of 10% and 20% manure increased plant dry weight and aloin content. The highest harvest index, economic yield, shoots-to-roots ratio, gel dry weight, chlorophyll a and b content, phenolics content of gel, and leaf and gel flavonoids’ content, as well as potassium, magnesium, zinc, and iron content, were obtained by the CeO2: SA-NPs × 20% manure treatment combination. The 10% and 20% manure and foliar application with CeO2-NPs, SA, and CeO2: SA-NPs increased the phenolics content of leaves compared to the control. Manure application (20%) positively affected the plant’s phosphorus content. The results revealed that co-application of soil-based cow manure and foliar CeO2: SA-NPs stimulated the growth and physiological responses of Aloe vera, and the results would be helpful to the agricultural extension sections and pioneer farmers.
1. Introduction
Nanotechnology is a branch of science that works with particles at a range of 1–100 nanometers in size. The small size of these particles has made it possible to use them in various fields of science such as medicine, the environment, and agriculture. Due to their small size, nanomaterials have unique electronic, optical, magnetic, and mechanical properties. These characteristics make them functionally different from their conventional forms. Cerium oxide belongs to the group of lanthanides (atomic number 58), which is one of the candidates to mitigate many environmental problems (due to their high specific surface area). Cerium oxide does this by scavenging oxygen free radicals, preventing the growth of some microorganisms, and having catalytic and antioxidant properties [1,2]. Salicylic acid is a phenolic compound with several functions in the plant, such as hormonal effects, growth stimulation, and defense responses under the biotic and abiotic stressful environments, inducing pest and disease tolerance and increasing secondary metabolites biosynthesis [3]. In a study on tomatoes, the application of cerium oxide increased yield, soluble solids content, and calcium content in the fruits [4]. Research in yarrow [3] and apple [5] found that salicylic acid raised the yield, chlorophyll, and phenolics content, as well as the plant root growth potential. Plants need adequate amounts of nitrogen, phosphorus, potassium, and sulfur to pursue their growth and metabolic activities (photosynthesis, respiration, biosynthesis of organic acids, nitrogen fixation, and enzymatic activities). Today, to increase plant yield, chemical fertilizers are widely used, which in the long term can cause environmental pollution. The results of a study conducted on Aloe vera showed that the overuse of chemical fertilizers harmed the quality of plants [6]. To compensate, in addition to chemical fertilizers, organic fertilizers must be utilized to prevent nutrient deficiencies in the soil [7]. The application of animal manure has an essential role in soil nutrient supply, soil health, and improving the soil’s physical and chemical properties. Due to its high porosity, livestock manure improves soil water-holding capacity, soil aeration, and drainage, increases the organic carbon ratio in the soil, and by supplying the plant nutrients, plays an essential role in plant growth and product quality [8,9]. A study on Aloe vera found that poultry manure increased plant yield combined with chemical fertilizers [7].
Aloe vera is an evergreen plant with thick, watery leaves from the lily family [6]. The leaf gel of the aloe plant contains more than 200 active ingredients such as polysaccharides (glucomannan and galactan), vitamins, proteins, fats, and amino acids. The gel has several therapeutic properties such as anti-wrinkle and anti-tumor effects, burn treatment, and strengthening the immune system (due to the presence of anthracene compounds) [6,10]. Aloe vera is a valuable plant in the food, pharmaceutical, cosmetic, and health industries. Due to the tremendous climatic diversity of Iran, some parts of the country are suitable to cultivate this valuable medicinal plant. Therefore, the present study aimed to evaluate the application of animal manure and foliar treatments with salicylic acid, cerium oxide nanoparticles, and cerium oxide salicylic acid nanoparticles (CeO2: SA-NPs) on the growth and some physiological traits of Aloe vera in the hope to develop a cultivation area for this plant, especially within stress-prone environments.
2. Materials and Methods
2.1. Plant Material and Experimental Setup
To assess the growth and physiological responses of Aloe vera L. to soil-based cow manure use and the application of foliar salicylic acid (SA), cerium oxide nanoparticles (CeO2-NPs), and cerium oxide salicylic acid nanoparticles (CeO2: SA-NPs), this experiment was conducted as a factorial experiment based on a randomized complete design with three replications at the Azarbaijan Shahid Madani University, East Azarbaijan province, Iran (37°48′26.66″ N and 45°56′33.70″ E). The first factor was the soil cow manure (no-manure, 10 and 20% v/v of soil) use, and the second factor was the foliar application of distilled water (DW), SA (13.8 mg L−1 equivalent to 100 µM) [11], CeO2-NPs (50 mg L−1) [12], and cerium oxide salicylic acid nanoparticles (CeO2: SA-NPs, 100 µM + 50 mg L−1), and the control of our experiment was no-manure × foliar application of distilled water. Six-month-old plants provided by a local supplier were used for the experiment. Disinfection of plant roots was performed with 10% sodium hypochlorite solution for 20 min. The plantlets were then transferred to 5 L pots containing the desired volume of soil and manure treatments. The soil and manure physicochemical characteristics are summarized in Table 1.

Table 1.
Physico-chemical characteristics of soil and cow manure.
To adapt the plants to the greenhouse conditions, plants were kept in pots for two months. They were irrigated with tap water during this period. Greenhouse conditions during plant maintenance were as follows: light intensity was about 450 µmol m−2 s−1 as common ambient light, temperature regime was 25 and 20 °C in the day and night, and the relative humidity was 65%. After 60 days of planting, the first foliar treatment was done in the 5–7 leaves stage, and the second foliar spraying was applied two weeks later. Two months after the last foliar treatment, plants were harvested to evaluate the desired traits.
2.2. CeO2 Synthesis
The CeO2-NPs were synthesized via a easy sonochemical procedure. The precursor materials used in this research were Ce(NO3)3 hexahydrate and urea. For the synthesis, 0.05 M of Ce(NO3)3, 6H2O was dissolved in 17 mL of water and ultra-sonicated for 20 min. Then, 20 g of urea was added to the solution and allowed to sonicate for 2 h. Subsequently, we centrifuged the solution (8000 rpm/15 min) and washed it with water and ethanol to remove the unreacted materials. Finally, the precipitate material was dried at 50 °C overnight. Later, the dried powder was calcined at 850 °C for 5 h.
2.3. Preparation of CeO2-NPs and Salicylic Acid Solution (CeO2: SA-NPs)
The CeO2-NPs were mixed with salicylic acid by molar ratios (1:0, 1:1, 1:2, 2:1, and 0:1) with 0, 50, and 100 µm. For example, 500 mL solution of CeO2-NPs (50 µm) and salicylic acid (50 µm) (1:1) was fabricated by a sonication bath at 60 °C for 1 h and then treated by a pulse probe sonication for 1 h to form a clear solution without any suspended particles.
2.4. TEM
Figure 1 (left) represents the TEM image of fabricated CeO2: SA-NCs. As shown, the successful octahedron nanoparticles are around 30–80 nm, which agrees with the DLS data presented in Figure 1 (right).

Figure 1.
TEM image of sonochemical synthesis of CeO2-SA nanoparticles (left), and DLS analysis of CeO2-SA nanoparticles (right).
2.5. Fresh and Dry Weight of Plants and Gel, Total Plant Yield, Economic Yield, and Harvest Index
At the time of harvest, the leaves were cut from the crown, and after transfer to the laboratory, the fresh and dry weight (drying at 40 °C until constant weight) of the leaves was measured using a digital scale (BB141, Beco, Geesthacht, Germany). After removing the outer layers of the leaf, the inside gel was separated with a sharp knife and its fresh and dry weight was recorded. The plant economic yield, total plant yield, and harvest index were calculated using the below equations [13,14]:
Economic yield = dry weight of gel (g m−2)
Total plant yield or biological yield = dry weight of leaves and roots (g m−2)
Harvest Index = Economic yield/Biological yield × 100
2.6. Chlorophyll Content
Chlorophylls a and b were quantified by Prochazkova et al. [15] with a spectrophotometer (T80+, Beijing, China) at 645 and 665 nm. For this, 0.5 g of leaf samples were extracted by dimethyl sulfoxide for 4 h at 65 °C, and chlorophylls a and b were calculated as mg g−1 FW.
2.7. Total Soluble Solids Content (°Brix)
The filtered leaf extracted juice was used to determine the total soluble solids (TSS) content by a digital refractometer (Erma, Tokyo, Japan).
2.8. Total Phenolics Content
For measurement of total phenolics content, as described by Kim et al. [16], 0.5 g of the fresh leaf sample was homogenized using acidic methanol and centrifuged at 12,000 rpm for 15 min. Then, 1.59 mL of distilled water, 100 μL of 10% Folin–Ciocalteu reagent, and 20 μL of the supernatant were combined and kept for 10 min. Finally, 300 μL of 7.5% sodium carbonate was added to the mixture and stored in the dark for 2 h. The final data are expressed as mg g−1 FW.
2.9. Total Flavonoids Content
To measure the flavonoids content according to the method of Quettier-Deleu et al. [17], 0.5 g of fresh leaf sample was smoothed in methanol 80% and centrifuged at 14,000× g rpm for 15 min. Then, 200 μL of the supernatant, 600 μL of 95% methanol, 40 μL of 10% aluminum chloride, 40 μL of 1 M potassium acetate, and 1120 μL of distilled water were admixed and stored at room temperature for 40 min. The absorbance was detected at 415 nm and expressed as mg g−1 FW.
2.10. Aloin Content of the Gel
To evaluate the gel’s aloin content, the leaf gel was first separated, and after passing through a strainer; 1% Ca(OH)2 was added. The samples were then centrifuged. The supernatant from the centrifuge was discarded. The remaining solution contained aloin. The compound concentration was expressed as mg kg−1 against aloin as standard [18]. The solution was scanned in the UV range of 200–400 nm.
2.11. Elemental Composition
The leaves’ mineral content was analyzed by the method described in detail by Chrysargyris et al. [19]. Na+ and K+ were quantified by the flame photometric method (Corning, 410, London, UK). Zn and Fe contents of plant tissues were traced by atomic absorption spectroscopy (Shimadzu, AA6300, Tokyo, Japan) by Honarjoo et al. [20].
2.12. Experimental Design and Data Analysis
The experiment was conducted as a factorial based on the completely randomized design with three replications. Mean comparisons were done by the least significant difference test (LSD) at 1% and 5% probability levels. The normality of the data was tested by SPSS (version 20), and in case of any abnormal data, the Chi-square conversion test was conducted. Pearson’s correlation and cluster dendrogram heat maps were plotted in R software (R foundation for statistical computing Version 4.1.2).
3. Results
3.1. Growth Parameters
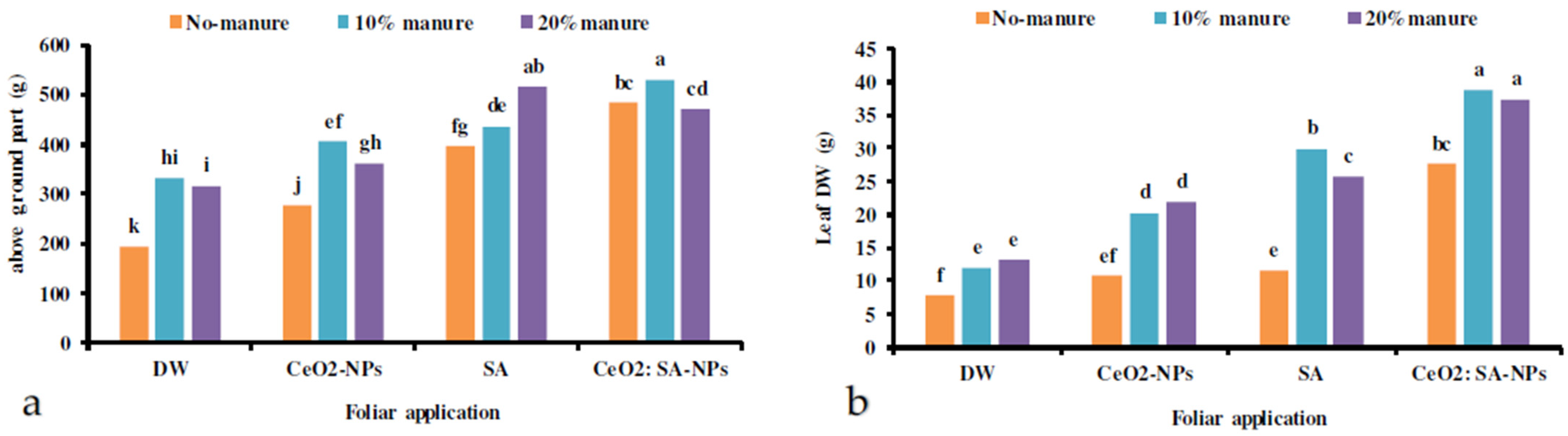
The results showed that the plants’ fresh and dry weight was influenced by the co-application of manure and foliar spray of CeO2-NPs, SA, and CeO2: SA-NPs (Table 2). The lowest fresh and dry weight was observed in the control treatment (Figure 2). The treatments caused an improvement in the fresh and dry weight gain of plants. The treatment effects varied on the above-ground weight. Animal manure (10%) × CeO2: SA-NPs and 20% manure × SA foliar application caused the highest fresh weight gain of the plants (Figure 2a). The highest dry weight of leaves was obtained in the foliar application with CeO2: SA-NPs and 10% and 20% animal manure (Figure 2b). Leaf diameter, leaf length, and the number of leaves were affected by the independent effects of experimental treatments (Table 2). Foliar application of cerium oxide and CeO2: SA-NPs increased the leaves’ diameter (Table 3). Leaf length and leaf number were influenced by CeO2: SA-NPs (Table 3). However, CeO2: SA-NPs increased leaf length by 27% compared to the control treatment (Table 3). Moreover, manure application significantly improved leaf length, and 20% manure increased the trait up to 38% compared to the control. Both levels of manure increased the number and diameter of leaves compared to the control treatment (Table 4). Root fresh weight was influenced by 20% animal manure × CeO2-NPs foliar application (Table 5). The shoot dry weight/root dry weight ratio was influenced by 20% animal manure × CeO2: SA-NPs (Table 5).

Table 2.
ANOVA for the effect of animal manure (0%, 10%, and 20% v/v) and foliar applications (salicylic acid, CeO2-nanoparticles, and CeO2: SA-nanoparticles) on Aloe vera L. growth characteristics.
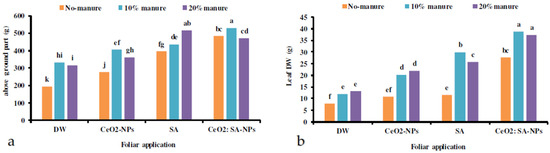
Figure 2.
The interaction effects of animal manure application (no-manure, 10%, and 20% v/v) and foliar spray of distilled water (DW), salicylic acid (SA), cerium oxide nanoparticles (CeO2-NPs), and cerium oxide salicylic acid nanoparticles (CeO2: SA-NPs) on the above-ground parts (a) and leaf dry weight (b) of Aloe vera L. plants. Significant differences among treatments are indicated by the different Latin letters.

Table 3.
The effects of salicylic acid (SA), CeO2-NPs, and CeO2: SA-NPs foliar treatments and 10% and 20% of soil-based animal manure use on leaves’ number and diameter, and leaf length of Aloe vera L. plants.

Table 4.
Interaction effects of soil-based animal manure (0%, 10%, and 20% v/v) use and foliar applications (no-foliar, SA, CeO2-nanoparticles, and CeO2: SA-nanoparticles) on Aloe vera L. plants’ growth and physiological traits.

Table 5.
Interaction effects of soil-based animal manure (0%, 10%, and 20% v/v) use and foliar applications (no-foliar, SA, CeO2-nanoparticles, and CeO2: SA-nanoparticles) on Aloe vera L. plants’ chlorophyll a and b as well as total soluble solids (TSS) content.
3.2. Fresh and Dry Weight of Plants and Gel
The interactions of manure × foliar applications significantly influenced these traits. The 20% manure and CeO2: SA-NPs and CeO2-NPs treatments increased the gel FW of leaves (Table 5). The highest gel DW was obtained in 20% manure with CeO2: SA-NPs, which showed an 81% increase compared to the control (Table 5). The lowest gel FW and DW were obtained in the control plants (Table 4).
No-manure × SA foliar spray increased the gel fresh weight/leaf fresh weight ratio, but all treatments used in this experiment influenced the gel dry weight/leaf dry weight ratio (Table 4).
3.3. Chlorophyll Content
Co-application of animal manure × foliar treatments significantly improved chlorophyll content. The highest Chl a and b content was obtained in the foliar CeO2: SA-NPs × 20% manure. Chl a and b content in plants under 20% manure × CeO2: SA-NPs improved by 64% and 70% compared to the control, respectively (Table 5).
3.4. Total Soluble Solids Content (°Brix)
Interactions of treatments significantly affected TSS content. Thus, the foliar application of CeO2-NPs, CeO2: SA-NPs × (20%) manure, and (20%) manure × no-foliar treatment increased the TSS of the plants. The highest and the lowest data were observed in the treatments of 20% manure × CeO2-NPs, and CeO2: SA-NPs and control, respectively (Table 5).
3.5. Total Plant Yield and Economic Yield
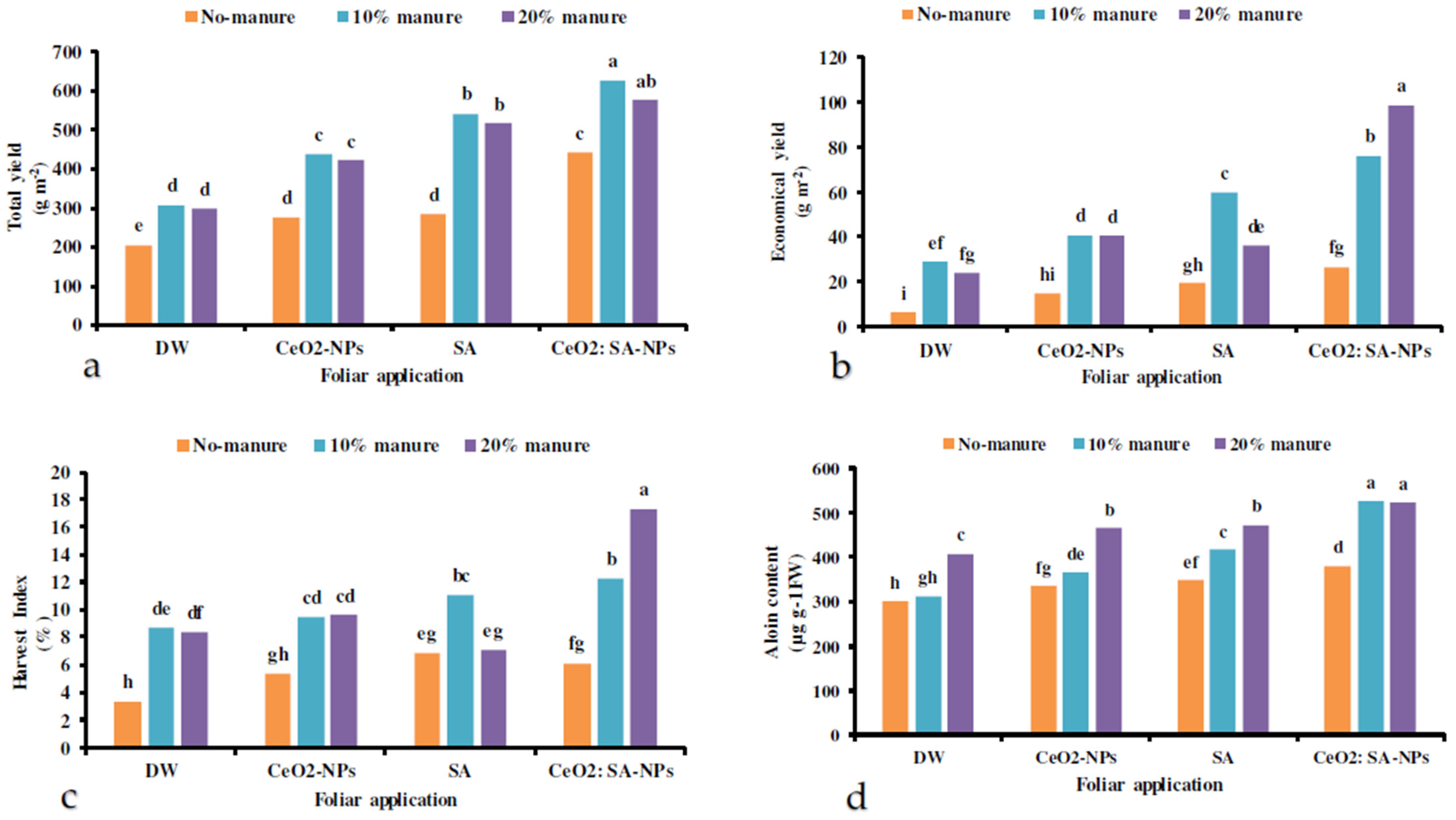
Foliar application of CeO2-NPs, SA, and CeO2: SA-NPs × manure significantly impacted the yield of Aloe vera (Table 6). Manure (10% and 20%) × CeO2: SA-NPs attained the highest total yield, while the lowest was observed in the control (Figure 3a). The foliar application treatments × manure significantly influenced the economic yield (Table 7). The interaction of CeO2: SA-NPs × 20% manure significantly increased the economic yield of the plant compared to the control. The 20% manure × foliar CeO2: SA-NPs treatment improved the economic yield by 29% compared to the 10% manure × CeO2: SA-NPs treatment. Contrarily, the lowest economic yield was recorded in control plants (Figure 3b).

Table 6.
ANOVA for the effect of soil-based animal manure (no-manure, 10%, and 20% v/v) use and foliar applications (no-foliar, salicylic acid, cerium oxide nanoparticles, and CeO2: SA-nanoparticles) on Aloe vera L. plants’ aloin content and yield traits.
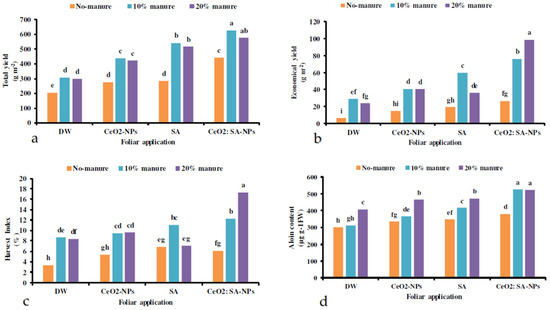
Figure 3.
The interaction effects of soil-based animal manure application (no-manure, 10%, and 20% v/v) and foliar spray of distilled water (DW), salicylic acid (SA), cerium oxide nanoparticles (CeO2-nanoparticles), and cerium oxide salicylic acid nanoparticles (CeO2: SA-nanoparticles) on the total yield (a), economic yield (b), harvest index (c), and aloin content (d) of Aloe vera L. plants. Significant differences among treatments are indicated by the different Latin letters on bars.

Table 7.
ANOVA for the effects of soil-based animal manure (0%, 10%, and 20% v/v) use and foliar applications (no-foliar, salicylic acid, cerium oxide nanoparticles, and CeO2: SA-nanoparticles) on Aloe vera L. plants’ phenolics and flavonoids content as well as elemental content.
3.6. Harvest Index
Foliar application of CeO2-NPs, SA, and CeO2: SA-NPs × manure significantly influenced the harvest index of Aloe vera L. (Table 7). The lowest harvest index was observed in the control treatment, while the highest was obtained with CeO2: SA-NPs × 20% animal manure. Under the CeO2: SA-NPs treatment, the application of manure (20%) increased the harvest index by 25% compared to the application of 10% manure (Figure 3c).
3.7. Aloin Content of the Gel
The co-application of manure and foliar treatments had a significant effect on the aloin content of plants (Table 6). The 10% and 20% manure × CeO2: SA-NPs foliar applications led to the aloin content of 525 and 524 µg g−1 Fwt, respectively. The aloin content of control plants was the lowest recorded one between all the treatments (Figure 3d).
3.8. Total Phenolics and Total Flavonoids Content of Gel and Leaves
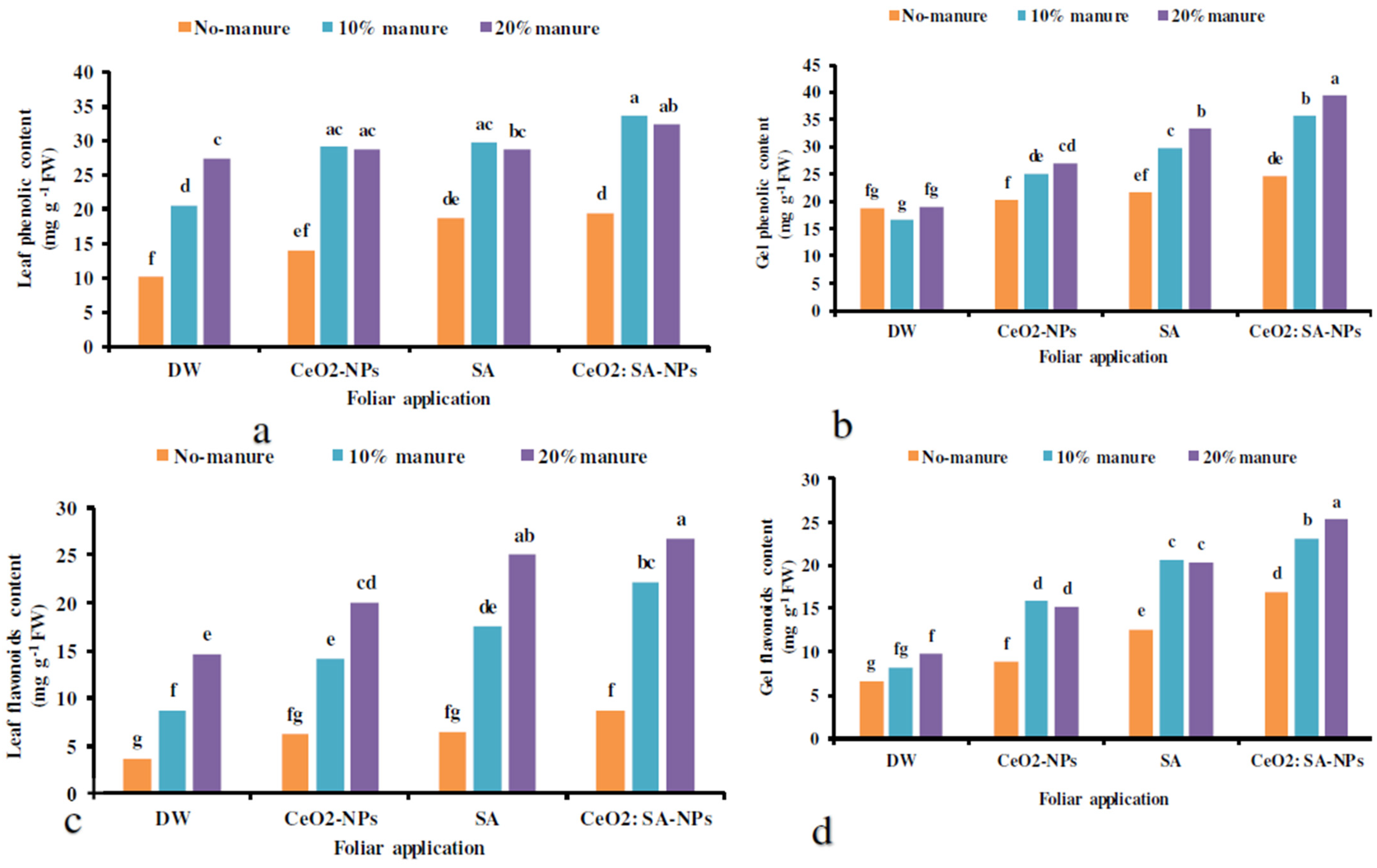
The results showed that the total phenolics and flavonoids content of leaves and gels was significantly affected by the interactions of the experimental treatments (Table 7). Manure application (10% and 20%) × foliar spraying of CeO2-NPs and CeO2: SA-NPs and 10% manure × SA spray increased the total phenolics content of leaves compared to the control (Figure 4a). The highest phenolics and flavonoids content of gel was obtained in the treatment of manure (20%) × CeO2: SA-NPs (Figure 4b,d). The uppermost leaves’ flavonoids content was observed in manure (20%) × foliar application of CeO2: SA-NPs and SA foliar spray (Figure 4c). The application of 20% manure × CeO2: SA-NPs caused an 84% increase in gel flavonoids content compared to the control (Figure 4d).
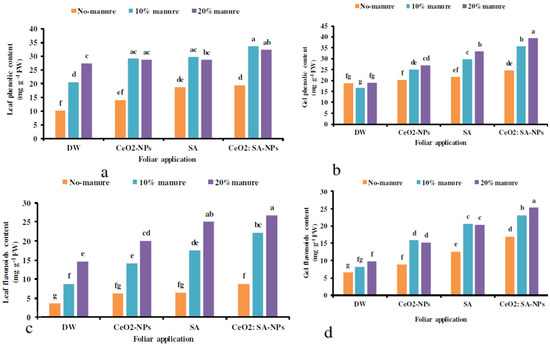
Figure 4.
The interaction effects of soil-based animal manure application (0%, 10%, and 20% v/v) and foliar use of distilled water (DW), salicylic acid (SA), cerium oxide nanoparticles (CeO2-nanoparticles), and cerium oxide salicylic acid nanoparticles (CeO2: SA-nanoparticles) on the phenolics content of the leaves (a) and gel (b) as well as the flavonoids content of the leaf (c) and gel (d) of Aloe vera L. plants. Significant differences among treatments are indicated by the different Latin letters on bars.
3.9. Elemental Composition
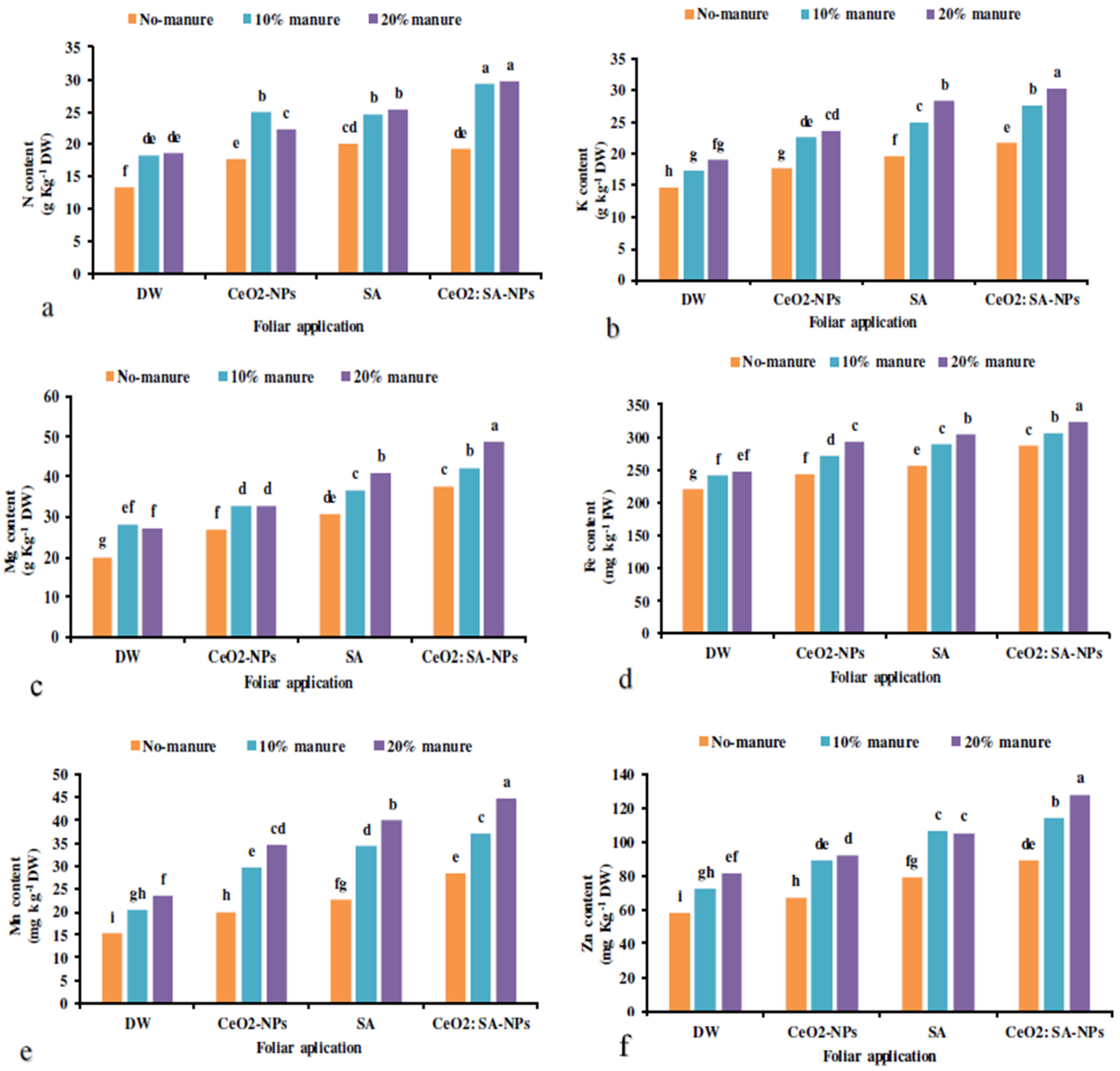
The manure × foliar application treatments significantly increased the elemental content except for the P and Ca content of plants (Table 7). The highest N content was obtained in the manure application (10% and 20%) × foliar utilization of CeO2: SA-NPs (Figure 5a). Furthermore, CeO2: SA-NPs × animal manure (20%) attained the highest K content. The lowest K content was obtained in controls (Figure 5b). The lowest Mg content was recorded for control, while the highest (62% more) was traced at animal manure (20%) × CeO2: SA-NPs (Figure 5c).
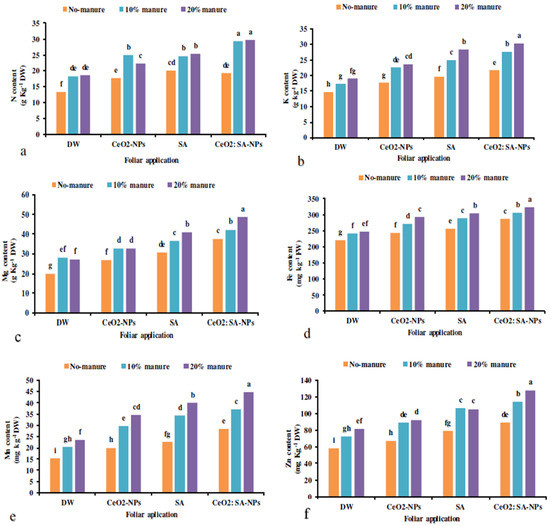
Figure 5.
The interaction effects of soil-based animal manure application (0%, 10%, and 20% v/v) and foliar spray of distilled water (DW), salicylic acid (SA), cerium oxide nanoparticles (CeO2-nanoparticles), and cerium oxide salicylic acid nanoparticles (CeO2: SA-nanoparticles) on the elemental content of Aloe vera plants (a–f). Significant differences among treatments are indicated by the different Latin letters.
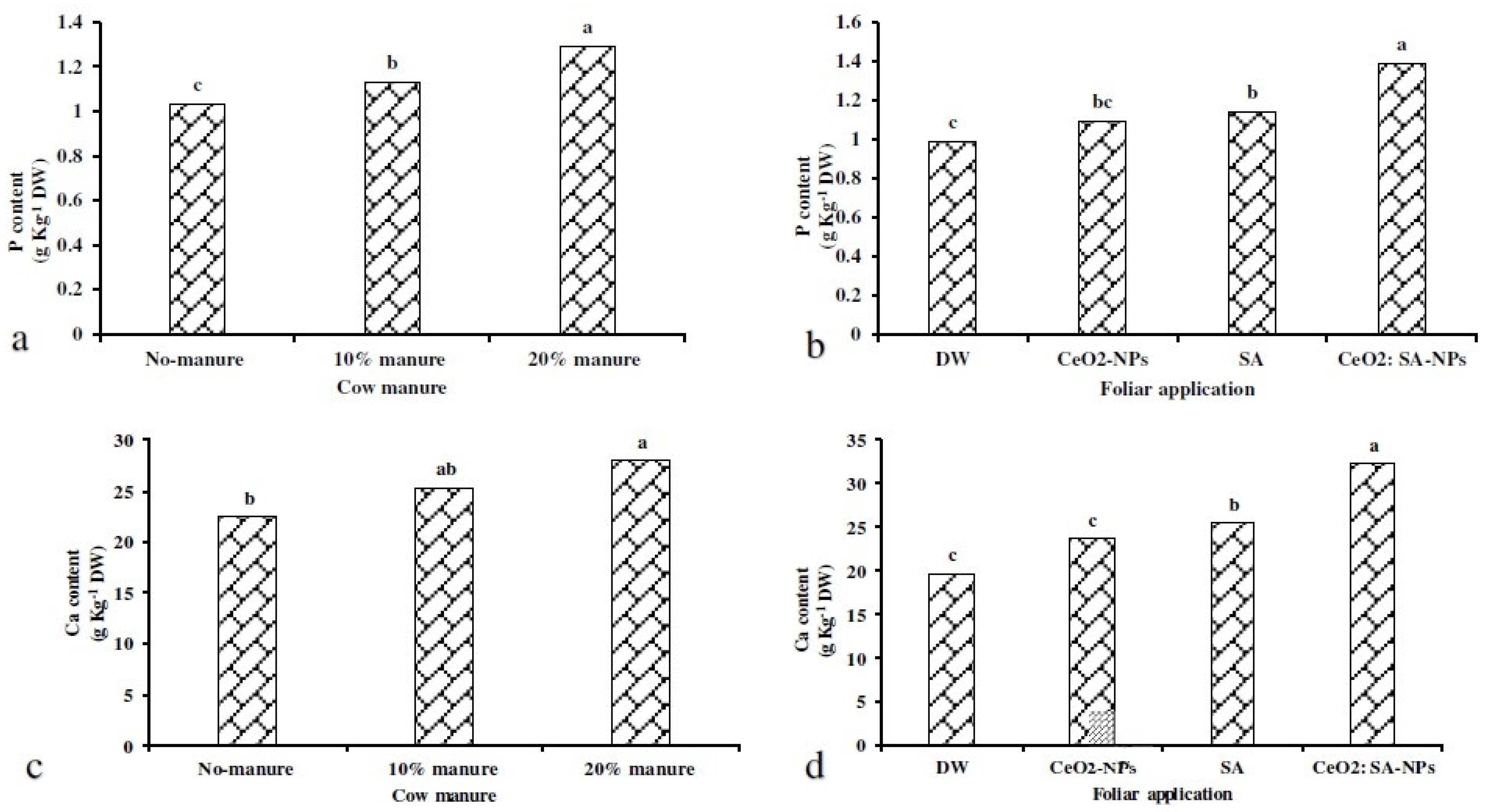
The 20% manure treatment caused a 24% increase in P content compared to the control (Figure 6a). Moreover, CeO2: SA-NPs foliar application caused a 40% increase in P content compared to the control (Figure 6b). Ca content was affected by the independent effects of experimental treatments (Figure 6c,d). The application of cow manure (10% and 20%) increased the Ca content compared to the control (Figure 6c). Foliar application of CeO2: SA-NPs increased the Ca content by 68% compared to the control (Figure 6d).
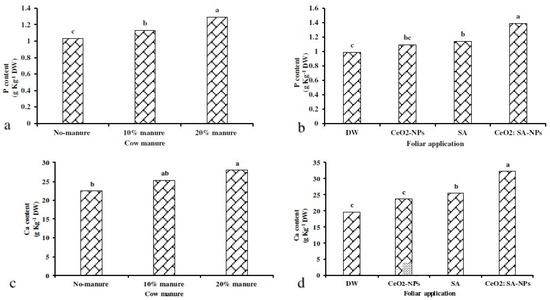
Figure 6.
The effects of soil-based animal manure application (0%, 10%, and 20% v/v) on P (a) and Ca (c) content, and the foliar spray of distilled water (DW), salicylic acid (SA), cerium oxide nanoparticles (CeO2-nanoparticles), and cerium oxide salicylic acid nanoparticles (CeO2: SA-nanoparticles) on P (b) and Ca (d) content of Aloe vera L. plants. Significant differences among treatments are indicated by the different Latin letters.
Foliar applications × animal manure use increased the content of microelements compared to the control (Table 7). Manure application (20%) × CeO2: SA-NPs foliar use increased the Fe (Figure 5d), Mn (Figure 5e), and Zn (Figure 5f) content of Aloe vera L. The lowest content of Fe, Mn, and Zn was observed in the control treatment. Foliar application of CeO2: SA-NPs × manure (20%) attained the highest content of Zn, Mn, and Fe content with an 11%, 17%, and 11% increase compared to control, respectively (Figure 6d).
3.10. Multivariate Analysis of Soil-Based Animal Manure Application × Foliar Use of Salicylic Acid, Cerium Oxide-NPs, and CeO2: SA-NPs on Aloe vera Plants
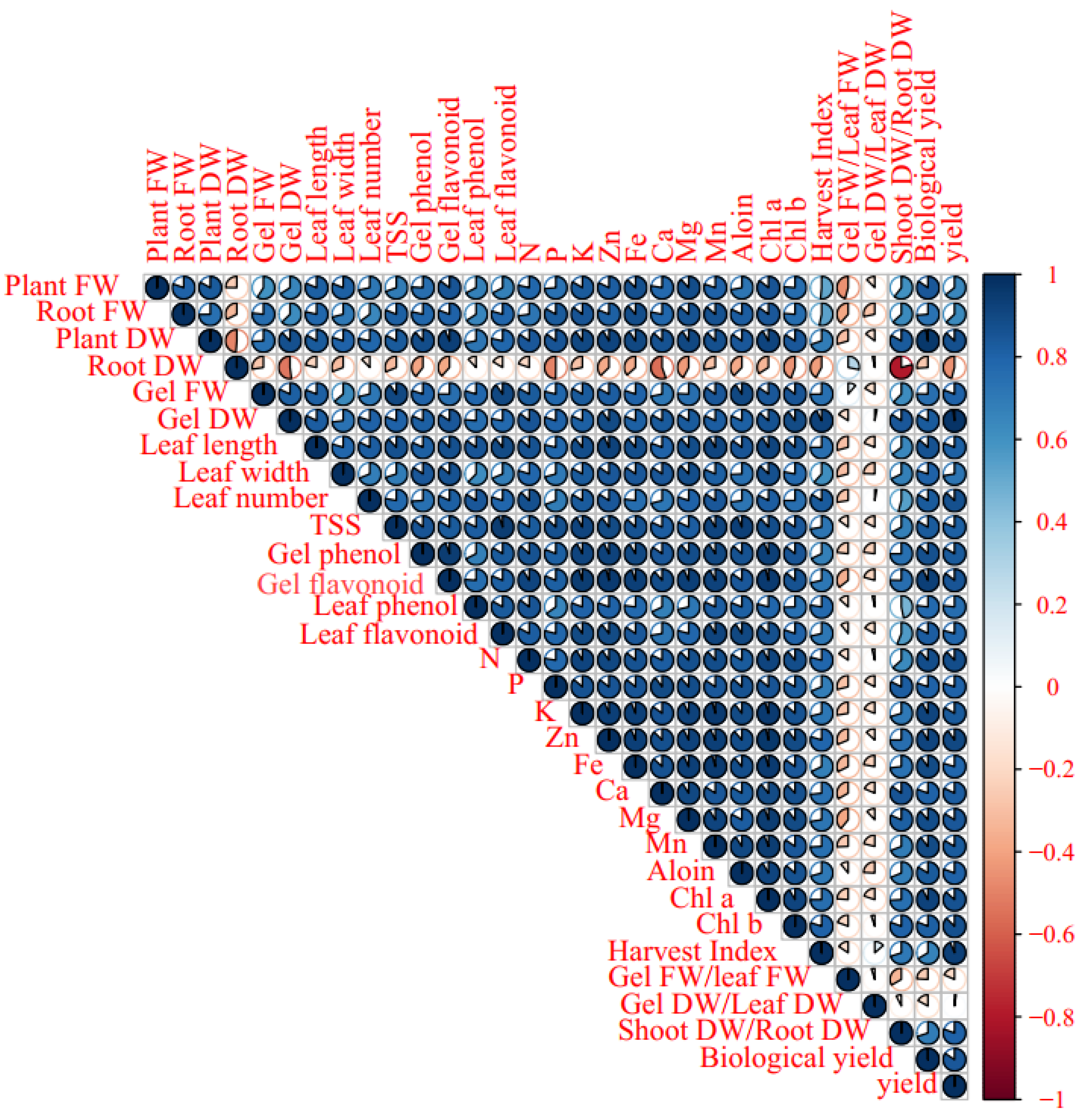
The Pearson’s correlation heat map of morphological, biochemical, and elemental traits showed a significant positive correlation among leaf fresh weight, root fresh weight, leaf dry weight, gel fresh weight, gel dry weight, leaf length, leaf width, leaf number, TSS, gel and leaf phenolics, flavonoids content, and N, P, K, Zn, Fe, Ca, Mg, Mn, aloin, Chl a, and Chl b content, as well as harvest index, shoot dry weight, root dry weight, and biological yield. However, root dry weight negatively correlated to Ca content, gel dry weight, and shoot dry weight/root dry weight. Otherwise, the traits have no significant correlation with gel fresh weight/leaf fresh weight and gel dry weight/leaf dry weight (Figure 7).
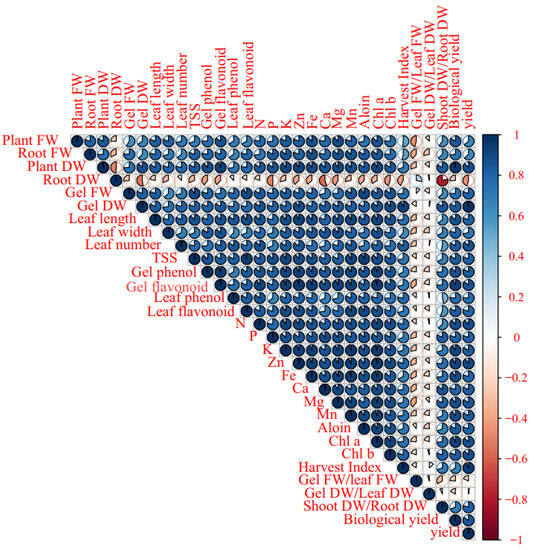
Figure 7.
Pearson’s correlation heat map of soil-based animal manure use × foliar applications (distilled water, salicylic acid, cerium oxide nanoparticles, and CeO2: SA-nanoparticles) on the growth, physiological responses, and elemental content of Aloe vera L. plants. FW and DW refer to fresh and dry weight, respectively.
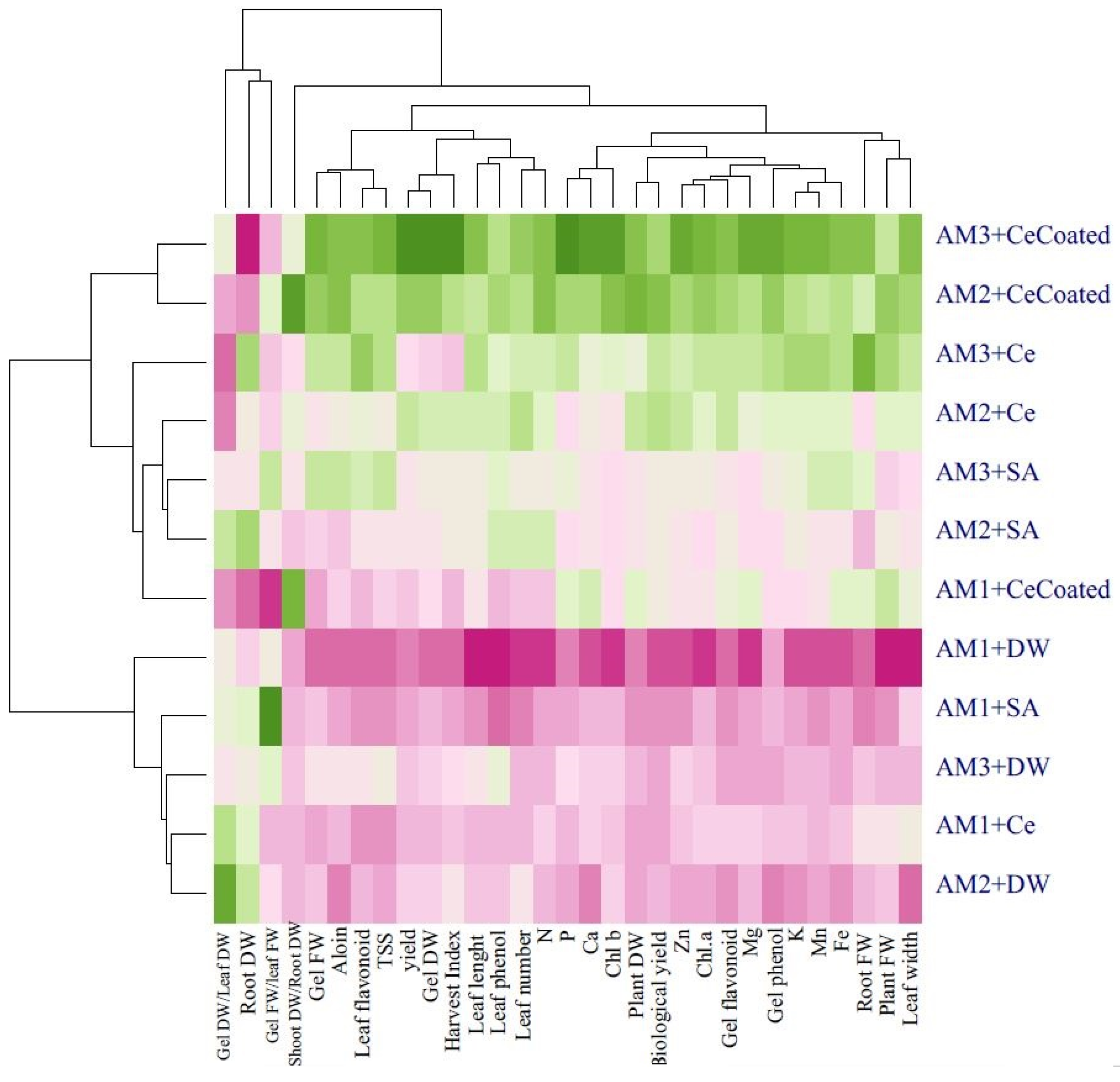
Heat map analysis for the treatment combinations showed that the following traits had positive compliance with animal manure × foliar application treatments: plant fresh and dry weight, root fresh weight, gel fresh and dry weight, leaf length, leaf width, leaf number, TSS, gel and leaf phenolics, and flavonoids content; N, P, K, Zn, Fe, Ca, Mg, Mn, aloin, Chl a, and Chl b content; and the harvest index, shoot dry weight/root dry weight, and biological yield (Figure 8).
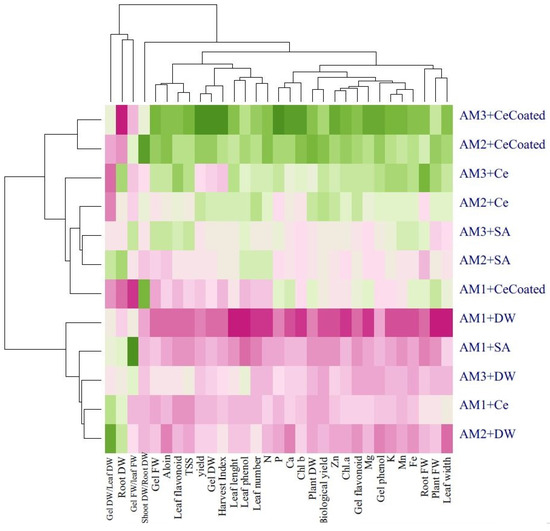
Figure 8.
Heat map of cluster analysis dendrogram for the effects of soil-based animal manure use and foliar application (distilled water, salicylic acid, cerium oxide nanoparticles, and CeO2:SA-nanoparticles) on the growth, physiological responses, and elemental content of Aloe vera L. plants. FW and DW refer to fresh and dry weight, respectively. AM1 = without animal manure), AM2 = 10% animal manure, and AM3 = 20% animal manure. SA is salicylic acid), Ce is cerium oxide nanoparticles, and CeCoated is CeO2: SA-nanoparticles. The green color and the purple color show the positive and the negative response of the evaluated traits to the treatments, respectively. Moreover, the darker green indicates more positive response, and the darker purple indicates more negative responses.
Cluster analysis and heat map dendrograms (Figure 8 and Figure 9) revealed two main groups in the evaluated characteristics of the Aloe vera plants. Group 1 contained the following: plant fresh and dry weight, root fresh weight, gel fresh and dry weight, leaf length, leaf width, leaf number, TSS, gel and leaf phenolics, and flavonoids content; N, P, K, Zn, Fe, Ca, Mg, Mn, aloin, Chl a, and Chl b content; and harvest index, shoot dry weight/root dry weight, biological yield, and plant yield. Moreover, group 2 contained the following: gel fresh weight/leaf fresh weight and root dry weight. In addition, cluster analysis of heat maps for treatments showed three main groups. Group 1 used 10% and 20% of animal manure with CeO2: SA-NPs, group 2 used the application 10% and 20% of animal manure with CeO2-NPs and SA as well as CeO2: SA-NPs without animal manure, and group 3 included foliar spray of CeO2: SA-NPs without animal manure, and 10% and 20% of animal manure applications alone.
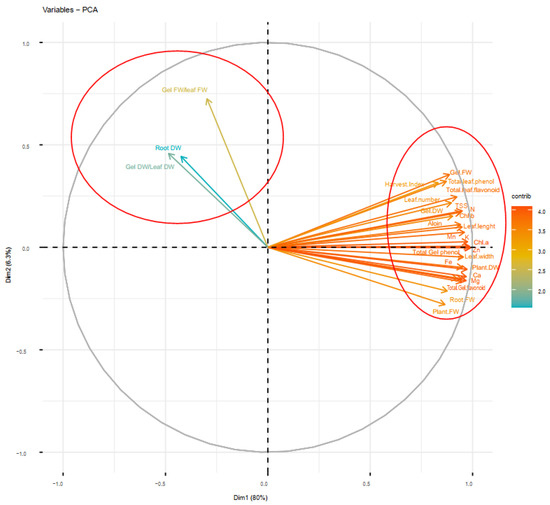
Figure 9.
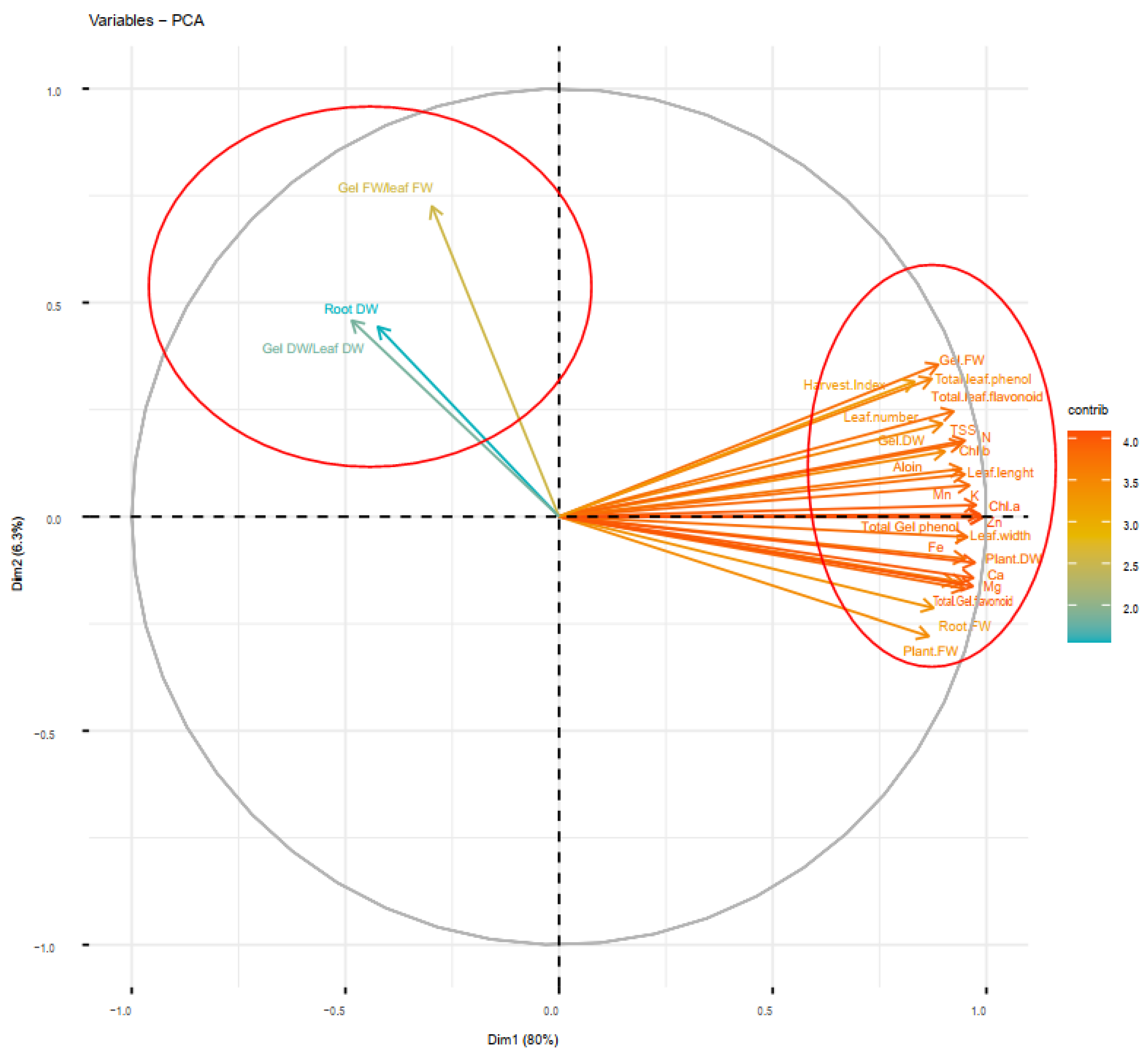
Principal component analysis (PCA) of soil-based animal manure and foliar applications (distilled water, salicylic acid, cerium oxide nanoparticles, and CeO2: SA-nanoparticles) on the growth, physiological responses, and elemental content of Aloe vera L. plants. FW and DW refer to fresh and dry weight, respectively.
The principal component analysis (PCA) delineates that the evaluated features were classified under two main principal components (PC1 and PC2) explaining 86.3% of the total variance. Most of the traits were classified as PC1, and thus they revealed a major part of the variance (80%), while a minor part of variance (6.3%) was classified as PC2. Additionally, the PCA approved the results of the Pearson’s correlation and cluster analysis, so that the dominant traits explaining the majority of total variance were included in PC1. However, PC2 only included root dry weight, gel fresh weight/leaf fresh weight, and gel dry weight (Figure 9).
4. Discussion
The widespread use of chemical fertilizers in agricultural production systems damages the environment, causes pollution of water resources, and reduces the soil microorganism population. Nanotechnology helps to improve agricultural production by increasing the nutrients’ efficiency and simultaneously reducing the chemical fertilizers used [1,2,21]. Numerous studies on the effect of cerium oxide on plants have shown that this compound plays an important role in promoting the physiological responses, gene expression [2,22], photosynthetic efficiency [23], and the oxidative potential of plants, especially under stressful environmental conditions [24,25], and additionally enhances water use efficiency [26], improves enzymatic activity, and helps in the absorption of the nutrients [2], and thus improves plant growth and yield [27,28]. The particular physical and chemical properties of cerium oxide make it an exceptional substance in manipulating the physiological activities of plants at low concentrations [2]. Organic fertilizers and foliar application of CeO2: SA-NPs impacted aloe plants’ yield, chlorophyll content, and other physiological traits. Improving the activity of optical receptors and, hence, the elevated electron transfer in photosynthetic components, improves photosynthesis efficacy and ultimately the plants’ productivity [29]. Studies have shown that cerium oxide at low concentrations improved photosynthesis, root growth, and the soluble solids content of Medicago arborea [30] and marigold [12]. Mineral elements have inevitable roles in plants’ growth, quality attributes, and productivity. Our results also confirmed the previous data regarding the increase in nutrients’ uptake due to cerium oxide and salicylic acid treatments [31,32,33]. The application of 500 mgL−1 of cerium oxide reduced the content of zinc, iron, manganese, and calcium, but with low concentrations, it improved the absorption of those elements in Salvia mittiollhiza [34]. Kalisz et al. [28] found that foliar application of cerium oxide increased the content of phenolics, flavonoids, chlorophyll, and antioxidants in lettuce and sweet pepper. Cerium oxide seems to act as a catalyst in chlorophyll biosynthesis, increasing light energy absorption and electron transfer from PSI to PSII [35,36]. The results showed the positive effect of CeO2: SA-NPs with animal manure on the content of phenolics, flavonoids, and chlorophyll of Aloe vera, which indicates the effectiveness of the nanoparticles treatment in the present study.
Phenolics and flavonoids are secondary metabolites that play essential roles in the reaction between cells and their environment and even in the plants’ enzymatic activities [37]. The flavonoids accumulation in the plants is controlled by the nutrients’ availability and environmental factors [38]. Results on the application of organic fertilizers alone or in combination with chemical fertilizers in Cephalaria syriaca [39], Saffron [40], Aloe vera [27], and tomato [41] showed the increased photosynthetic pigment content, nutrients content (iron, zinc, and copper), phenolics and flavonoids content and hence the improved plant yield. The combined application of chemical and organic fertilizers increases the activity of acid phosphatase and alkaline phosphatase around the roots, which powerfully stimulate the uptake of phosphorus, nitrogen, zinc, copper, and iron by the plant [42,43]. Organic fertilizers have significant functions in the soil’s nutrient supply and sustainability, the soil water storage capacity, and even soil aeration, and thus, help to improve plant growth [39]. Salicylic acid is a phenolic compound and growth regulator with crucial functions in photosynthesis, plant growth, seed germination, nutrient uptake, stomatal movements, enzymatic activity, and gene expression. Salicylic acid increases pigment content and the activity of RuBisco and PEP carboxylase enzymes. Increasing the mentioned activities improves photosynthesis potential and plant growth and yield [5,44]. Salicylic acid affects the activity of enzymes involved in nitrogen metabolism, plays an essential role in nitrate reductase activity, and influences plant proteins content and composition [44]. In a study in cucumber [33] and Portulaca oleraceae [45], salicylic acid treatment increased plants’ yield, TSS, and chlorophyll content. Low concentrations of salicylic acid stimulate H2O2 production. H2O2 availability increases phenylalanine ammonia-lyase activity and thus increases the content of phenolic compounds. Salicylic acid prevents the oxidation of fatty acids, fortifies membranes’ integrity, and hence stimulates growth parameters [46]. In the present study, salicylic acid improved the growth and physiological responses of the plant. However, based on the results, CeO2: SA-NPs were more effective on the studied traits when combined with animal manure application. Research on saffron [40] and Aloe vera [27,47] found that organic fertilizer increased flower yield and chlorophyll content. In two separate studies on aloe plants, it was found that the co-application of poultry manure with chemical fertilizer stimulated gel biosynthesis and the content of leaf gel [27,47]. The presence of numerous secondary metabolites, especially aloin in the Aloe vera plant improves the economic and medicinal value of the plant [27]. The increase in aloin content with animal manure use is due to the improvements in the soil ecosystem which stimulates the growth attributes and gel biosynthesis. Similar results were reported in the present experiment concerning aloin content via the application of animal manure and foliar use of CeO2: SA-NCs. In Faba bean, the use of organic and chemical fertilizers increased the content of copper, manganese, iron, potassium, nitrogen, calcium, magnesium, and phosphorus [43,48]. Animal manure use under field conditions increases the activity of soil microbial population, improves soil water content, the availability of nutrients, adjusts pH, and even improves soil structure leading to promoted plant growth and productivity [49,50,51]. In a study on chickpea, it was found that the plant’s access to sufficient water and nutrients during the grain-filling stage increased the harvest index. The present results also showed the increased plant nutrient uptake due to animal manure and foliar applications. Furthermore, the results revealed the effectiveness of animal manure application and foliar treatments on increasing the harvest index and economic yield of Aloe vera.
The widespread use of nanoparticles in modern agriculture is an excellent opportunity to reach the sustainable agricultural goals. The exceptional physical and chemical properties of cerium oxide nanoparticles have led to their widespread use in agriculture. Cerium oxide application under salinity stress increased the expression of the stress-responsive genes that regulate the biosynthesis of antioxidant enzymes in the glutathione pathway [52]. Nanoparticles play an important role in regulating the expression of upstream genes of catalase, peroxidase, phosphatase, and superoxide dismutase. The enzymes produced play fundamental roles in protecting the plant against environmental stresses.
Moreover, nanoparticles protect the photosynthetic apparatus against free radicals produced in response to the stress factors. Cerium oxide nanoparticles improve photosynthesis potential by enhancing the availability of the nutrients needed for photosynthesis and improving water use efficiency. Furthermore, studies have shown that nanoparticles mediate the expression of genes involved in DNA function, hormone biosynthesis, and photosynthesis, which ultimately impact plant growth and productivity [2].
5. Conclusions
The results showed the positive effects of animal manure and foliar treatments on the growth and physiological traits of Aloe vera. The application of 10% and 20% manure × foliar treatment with CeO2: SA-NPs improved most of the growth and physiological characteristics of Aloe vera. Considering that the tropical regions of Iran have a good potential for Aloe vera cultivation, the results from the present study can be used to advise the agricultural extension sections to develop cultivation areas with this high-valued crop, especially with saline and drought-prone areas.
Author Contributions
Conceptualization, M.B.H., L.V.M. and M.A.A.; data curation, R.B. and L.V.M.; formal analysis and methodology, M.B.H., L.V.M. and F.R.; project administration, M.B.H. and L.V.M.; visualization, M.B.H., L.V.M., F.R., K.K. and M.S.; writing—original draft, M.B.H., L.V.M. and F.R.; writing—review and editing, M.B.H. and M.S. All authors have read and agreed to the published version of the manuscript.
Funding
This study was financially supported by the Azarbaijan Shahid Madani University, Iran.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All new research data are presented in this contribution.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Singh, K.R.B.; Nayak, V.; Sarkar, T.; Singh, R.P. Cerium oxide nanoparticles: Properties, biosynthesis and biomedical application. R. Soc. Chem. Adv. 2020, 10, 27194–27214. [Google Scholar] [CrossRef]
- Prakash, V.; Peralta-Videa, J.; Tripathi, D.K.; Ma, X.; Sharma, S. Recent insight in to the impact, fate and transport of cerium oxide nanoparticles in plants-soil continuum. Ecotoxicol. Environ. Saf. 2021, 221, 112403. [Google Scholar] [CrossRef] [PubMed]
- Gorni, P.H.; Pacheco, A.C. Growth promotion and elicitor activity of salicylic acid in Achillea millefolium L. Afr. J. Biotechnol. 2016, 15, 657–665. [Google Scholar] [CrossRef]
- Barrios, A.C.; Medina-Velo, I.A.; Zuverza-Mena, N.; Dominguez, O.E.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Nutritional quality assessment of tomato fruits after exposure to uncoated and citric acid coated cerium oxide CeO2:SA-NCs, bulk cerium oxide, cerium acetate and citric acid. Plant Physiol. Biochem. 2017, 110, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Gacnik, S.; Veberic, R.; Hudina, M.; Marinovic, S.; Halbwirth, H.; Mikulic-petkovsek, M. Methyl salicylic acid affect quality and phenolic apple fruit three weeks before the harvest. Plants 2021, 10, 1807. [Google Scholar] [CrossRef] [PubMed]
- Lakshmi, C.S.; Sekhar, C. Importance of medicinal and aromatic plants and their response to organic source. J. Phsrm. Phytochem. 2018, 7, 3296–3301. [Google Scholar]
- Chowdhury, T.; Chowdhury, M.A.H.; Rahman, M.A.; Nahar, K.; Chowdhury, M.T.I.; Khan, M.S.I. Response of Aloe vera to inorganic and organic fertilization in relation to leaf biomass yield and postharvest fertility of soil. Bulg. J. Agric. Sci. 2020, 26, 346–354. [Google Scholar]
- Jahan, M.; Koocheki, A.R.; Ghorbani, R.; Nassiri-Mahallati, M.; Salari, M.D. The effects of manure application and branch management methods on some agroecological aspects of summer squash (Cucurbit pepo L.) in a low input cropping system. Int. J. Agric. Sci. 2013, 2, 428–434. [Google Scholar]
- Darini, M.T.; Sulistyaningsih, E. Combination of cow manure rate and different sources of nitrogen humite on the nutritional content and yield of Aloe vera L. plant in sandy soil. Int. J. Adv. Sci. Eng. Inf. Technol. 2020, 10, 1631. [Google Scholar] [CrossRef]
- Farrokhi, E.; Koocheki, A.; Nasiri Mahallati, M.; Khodemi, R. The effect of manure, chemical and biological fertilizers on Aloe vera growth in Boushehr province. J. Agroecol. 2018, 1, 1–21. [Google Scholar]
- Fathi, S.; Kharazmi, M.; Najafian, S. Effects of salicylic asid foliar application on morpho physiological traits of purslane (Portulaea olaracea L.) under salinity stress condition. J. Plant Physiol. Breed. 2019, 9, 1–9. [Google Scholar]
- Tumburu, L.; Andersen, C.P.; Rygiewicz, P.T.; Reichman, J.R. Molecular and physiological responses to titanium dioxide and cerium oxide CeO2: SA-NCs in Arabidopsis. Environ. Toxicol. Chem. 2017, 36, 71–82. [Google Scholar] [CrossRef] [PubMed]
- El Naim, A.M.; Abdalla, A.A.; Ahmed, M.F.M.; Taha, M.B. Biological yield and harvest index of faba bean (Vicia faba) as affected by different agro-ecological environments. World J. Agric. Res. 2015, 3, 78–82. [Google Scholar]
- Medi, D.; Turan, N.; Issa, K. Evaluation of Aloe vera intercropping on quantitative and qualitative characteristics of Roselle. Bulg. J. Agric. Sci. 2015, 21, 779–783. [Google Scholar]
- Prochazkova, D.; Sairam, R.K.; Srivastava, G.C.; Singh, D.V. Oxidative stress and antioxidant activity as the basis of senescence in maize leaves. Plant Sci. 2001, 161, 765–771. [Google Scholar] [CrossRef]
- Kim, K.H.; Tsao, R.; Yang, R.; Cui, S.W. Phenolic acid profiles and antioxidant activities of wheat bran extracts and the effect of hydrolysis conditions. Food Chem. 2006, 95, 466–473. [Google Scholar] [CrossRef]
- Quettier-Deleu, C.; Gressier, B.; Vasseur, J.; Dine, T.; Brunet, C.; Luyckx, M.; Cazin, M.; Cazin, J.C.; Bailleul, F.; Trotin, F. Phenolic compounds and antioxidant activities of buckwheat (Fagopyrum esculentum Moench) hulls and flour. J. Ethnopharmacol. 2000, 72, 35–40. [Google Scholar] [CrossRef]
- McCarthy, T.J.; Mapp, R.K. A comparative investigation of methods used to estimate aloin and related compounds in Aloes. Planta Med. 1970, 18, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Chrysargyris, A.; Michailidi, E.; Tzortzakis, N. Physiological and biochemical responses of Lavandula angustifolia to salinity under mineral foliar application. Front. Plant Sci. 2018, 9, 489. [Google Scholar] [CrossRef] [PubMed]
- Honarjoo, N.; Hajrasuliha, S.; Amini, H. Comparing three plants in absorption of ions from different natural saline and sodic soils. Int. J. Agric. Crop Sci. 2013, 6, 988–993. [Google Scholar]
- Tumburu, L.; Andersen, C.P.; Rygiewicz, P.T.; Reichman, J.R. Phenotypic and genomic responses to titanium dioxide and cerium oxide nanoparticles in Arabidopsis germinants. Environ. Toxicol. Chem. 2015, 34, 70–83. [Google Scholar] [CrossRef] [PubMed]
- Jahani, S.; Saadatmand, S.; Mahmoodzadeh, H.; Kharari-Nejad, R.A. Effect of foliar application of cerium oxide nanoparticles on growth, photosynthetic pigments, electrolyte leakage, compatible osmolytes and antioxidant enzymes activities of Calendula officinalis L. Biologia 2019, 74, 1063–1075. [Google Scholar] [CrossRef]
- Barrios, A.C.; Rico, C.M.; Trujillo-Reyes, J.; Medina-Velo, I.A.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Effects of uncoated and citric acid coated cerium oxide CeO2: SA-NCs, bulk cerium oxide, cerium acetate, and citric acid on tomato plants. Sci. Total Environ. 2016, 563, 956–964. [Google Scholar] [CrossRef] [PubMed]
- Rossi, L.; Zhang, W.; Lombardini, L.; Ma, X. The impact of cerium oxide CeO2: SA-NCs on the salt stress responses of Brassica napus L. Environ. Pollut. 2016, 219, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Tito, N.; Giraldo, J.P. Anionic Cerium oxide protect plant photosynthesis from abiotic stress by scavenging reactive oxygen species. ACS Nano 2017, 11, 11283–11297. [Google Scholar] [CrossRef]
- Cao, Z.; Rossi, L.; Stowers, C.; Zhang, W.; Lombardini, L.; Ma, X. The impact of cerium oxide CeO2: SA-NCs on the physiology of soybean (Glycine max (L.) Merr.) Under different soil moisture conditions. Environ. Sci. Pollut. Res. 2018, 25, 930–939. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, T.; Chowdhury, A.H.; Qingyue, W.; Enyoh, C.E.; Wang, W.; Islam Khan, S. Nutrient uptake and pharmaceutical compounds of Aloe vera as influenced by the integration of inorganic fertilizer and poultry manure in soil. Heliyon 2021, 7, e07464. [Google Scholar] [CrossRef]
- Kalisz, A.; Huska, D.; Jurkow, R.; Dvorak, M.; Klejdus, B.; Caruso, G.; Sekara, A. Nanoparticles of cerium, iron and silicon oxides change the metabolism of phenols and flavonoids in butterhead lettuce and sweet pepper seeding. Environ. Sci. Nano 2021, 8, 1945–1959. [Google Scholar] [CrossRef]
- Abbas, Q.; Liu, G.; Yousaf, B.; Ali, M.U.; Ullah, H.; Munir, M.A.M.; Rehman, A. Biochar-assisted transformation of engineered-cerium oxide CeO2:SA-NCs: Effect on wheat growth, photosynthetic traits and cerium accumulation. Ecotoxicol. Environ. Saf. 2020, 187, 109845. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Garay, A.; Pintos, B.; Manzanera, J.A.; Lobo, C.; Villalobos, N.; Martín, L. Uptake of CeO2 CeO2:SA-NCs and its effect on growth of Medicago arborea in vitro plantlets. Biol. Trace Elem. Res. 2014, 161, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Rico, C.M.; Majumdar, S.; Duarte-Gardea, M.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Interaction of CeO2: SA-NCs with edible plants and their possible implications in the food chain. J. Agric. Food Chem. 2011, 59, 3485–3498. [Google Scholar] [CrossRef] [PubMed]
- Peralta-Videa, J.R.; Hernandez-Viezcas, J.A.; Zhao, L.; Diaz, B.C.; Ge, Y.; Priester, J.H.; Holden, P.A.; Gardea-Torresdey, J.L. Cerium dioxide and zinc oxide CeO2:SA-NCs alter the nutritional value of soil cultivated soybean plants. Plant Physiol. Biochem. 2014, 80, 128–135. [Google Scholar] [CrossRef]
- Precido-Rangel, P.; Reyes-Perez, J.J.; Ramirez-Rodriguez, S.C.; Salas-Perez, L.; Fortis-Hernandez, M.; Murillo-Amador, B.; Troyo-Diequez, E. Foliar aspersion of salicylic acid improves phenolic and flavonoid compounds, and also the fruit yield in cucumber (Cucumis sativus L.). Plants 2019, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Paryan, S.; Ghorbanpour, M.; Hadian, J. Influence of CeO2-Nanoparticles on morpho-physiological traits and tanshinone contents of roots in Salvia miltiorrihiza Bunge upon foliar and soil application methods. J. Med. Plants 2020, 19, 168–187. [Google Scholar]
- Jurkow, R.; Sękara, A.; Pokluda, R.; Smolen, S.; Kalisz, A. Biochemical response of oakleaf lettuce seedlings to different concentrations of some metal (oid) oxide CeO2: SA-NCs. Agronomy 2020, 10, 997. [Google Scholar] [CrossRef]
- Chen, Y.; Luo, Y.; Qiu, N.; Hu, F.; Sheng, L.; Wang, R.; Cao, F. Ce3+ induces flavonoids accumulation by regulation of pigments, ions, chlorophyll fluorescence and antioxidant enzymes in suspension cells of Ginkgo biloba L. Plant Cell Tissue Organ Cult. 2015, 123, 283–296. [Google Scholar] [CrossRef]
- Kumar, R.; Mathur, M. Sanjana Primary metabolite profiling and antioxidant potential assay from selected plant parts of Gmelina arborea. J. Drug Deliv. Therap. 2019, 9, 376–381. [Google Scholar]
- Kalinova, J.; Vrchotova, N. The influence of organic and conventional crop management, variety and year on the yield and flavonoid level in common buckwheat groats. Food Chem. 2011, 127, 602–608. [Google Scholar] [CrossRef]
- Rahimi, A.; Siavash Moghaddam, S.; Ghiyasi, M.; Heydarzadeh, S.; Ghazizadeh, K.; Popovic-Djordjevic, J. The influence of chemical, organic and biological fertilizers on agrobiological and antioxidant properties of Syrian Cephalaria (Cephalaria syriaca L.). Agriculture 2019, 9, 122. [Google Scholar] [CrossRef]
- Kianimanesh, K.; Lebaschi, M.H.; Jaimand, K.; Abdossi, V.; Tabaei Aghdaei, S.R. The changes in yield, biochemical properties and essential oil componds of saffran (Crocus sativus L.) Plant treated whit organic and inorganic fertilizers under dryland farming system. J. Med. Plant By-Prod. 2020, 10, 37–44. [Google Scholar]
- Antonious, G.; Turlye, E.; Dawood, M. Ascorbic acid, sugars, phenols and nitrates content ration in Tomato grown in animal manure amended soil. Agriculture 2019, 9, 94. [Google Scholar] [CrossRef]
- Behera, B.C.; Singdevsachan, S.K.; Mishra, R.R.; Dutta, S.K.; Thatoi, H.N. Diversity, mechanism and biotechnology of phosphate solubilizing microorganism in mangrove-A review. Biocatal. Agric. Biotechnol. 2014, 3, 97–110. [Google Scholar] [CrossRef]
- Amiri, M.E.; Fallahi, E. Impact of animal manure on soil chemistry, mineral nutrients, yield and Fruit quality in Golden delicious apple. J. Plant Nutr. 2009, 32, 610–617. [Google Scholar] [CrossRef]
- Hayat, Q.; Hayat, S.; Alyemeni, M.N.; Ahmad, A. Salicylic acid-mediated changes in growth, photosynthesis, nitrogen metabolism and antioxidant defense system in Cicer arietinum L. Plant Soil Environ. 2012, 58, 417–423. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Ahamed, K.U.; Khalequzzaman, K.M.; Shamsuzzaman, A.M.M.; Nahar, K. Plant characteristics, growth and leaf yield of Aloe vera L. as affected by prganic manure in pot culture. Aust. J. Crop Sci. 2008, 2, 158–163. [Google Scholar]
- Faiyard, M.N.; Shehata, M.M.; Barsoom, S.W. Response of faba bean grown on sandy soil to organic and inorganic nitrogen fertilization. Egypt. J. Soil Sci. 1991, 31, 343–355. [Google Scholar]
- Gong, W.; Yan, X.; Wang, J.; Hu, T.; Gong, Y. Long-term applications of chemical and organic fertilizers on plant-available nitrogen pools and nitrogen management index. Biol. Fertil. Soils 2011, 47, 767–775. [Google Scholar] [CrossRef]
- Sugihara, S.; Funakawa, S.; Kilasara, M.; Kosaki, T. Dynamics of microbial biomass nitrogen in relation to plant nitrogen uptake during the crop growth period in a dry tropical cropland in Tanzania. Soil Sci. Plant Nutr. 2010, 56, 105–114. [Google Scholar] [CrossRef]
- Rani, R.; Srivastava, O.P. Vermicompost: A potential supplement to nitrogenous fertilizer in rice nutrition. Int. Rice Res. Notes 1997, 22, 30–31. [Google Scholar]
- Silim, S.V.; Saxena, M.C. Adaptation of spring-sown chickpea to the Mediterranean basin. II. Factors influencing yield under drought. Field Crops Res. 1993, 34, 137–146. [Google Scholar] [CrossRef]
- Ma, C.; Chhikara, S.; Xing, B.; Musante, C.; White, J.C.; Dhankher, O.P. Physiological and molecular response of Arabidopsis thaliana (L.) to nanoparticle cerium and indium oxide exposure ACS Sustain. Chem. Eng. 2013, 1, 768–778. [Google Scholar]
- Wanger, G.J. Contact and vacuole/extra-vacuole distribution of neutral sugar free amino acids and anthocyanin in protoplast. Plant Physiol. 1979, 64, 88–93. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).