Abstract
Rye (Secale cereale L.) cultivation has a long history in the Alpine region, where local ecotypes have been honed through generations of human selection. Despite the introduction of commercial varieties during the 20th century, traditional ecotypes are believed to persist in Valtellina, especially in the medium/upper valley (Teglio and Bormio). Our study aims to identify and preserve these local rye ecotypes by comparing them to commercial varieties. We examined 14 rye accessions through population genetic analyses (by using eight SSR makers), agronomic trials (germplasm characters, plant traits, and yields), and phytochemical analyses (total phenolic compound, flavonoids, and the radical scavenging activity). Two commercial varieties were used—one from Poland, previously genetically characterized (C_POL_P1; i.e., P1 ancestry), and one with an unknown origin. Additionally, we analyzed eleven accessions from Valtellina (medium and upper valley) and one from another alpine valley in South Tirol (Laimburg; LAI). Molecular markers analysis revealed the presence of two distinct genetic lineages: the first (P1) comprised all Teglio accessions and the commercial one from Poland, while the second (P2) grouped the two alpine accessions from Bormio (upper Valtellina) and South Tirol, along with a commercial one of uncertain origin. The clusters P2 exhibited significantly lower average values of the number of alleles (z = 2.03; p < 0.05), Shannon index (z = 2.35; p < 0.05), and expected heterozygosity (z = 1.97; p < 0.05) than in P1. Significant differences were observed in plant traits and agronomic performance between P1 and P2 accessions, with the latter displaying superior traits. For instance, the seed weight of germplasm was the highest in P2 (H(chi2) = 22.44; p < 0.001). Phytochemical analyses did not reveal a clear pattern between genetic lineages but identified some very well-performing Valtellina landraces in terms of scavenging activity. Characterizing these local rye varieties helps establish a short food supply chain to support Valtellina’s mountain economy. The medium valley (P1) and upper valley (P2) varieties have the potential for agroecological valorization, benefiting local farmers. Phytochemical analyses identified better accessions within P1 and P2 for nutraceutical use and commercialization. Additionally, selecting and conserving resilient ecotypes can address future challenges from extreme climate conditions.
1. Introduction
The cultivation of rye (Secale cereale L.) has been documented since ancient times in the Alpine region, where it has contributed to maintaining a traditional rural economy [1]. Indeed, this species has traditionally been used to supply flour, fodder, and straw material for building roofs and producing baskets. The broader cultivation of rye in less favorable environments, in comparison to other cereals, can be attributed to its remarkable ability to adapt to infertile sandy soils, extreme temperatures, and periods of drought [2].
Historically, rye cultivation began around 12,000 B.C. in Anatolia, Asia [3]. In Europe, rye cultivation dates back to the Bronze Age but became widespread in the Iron Age [4,5,6]. It is believed that rye was introduced to Europe from Southwestern Asia through Russia and later to Central and Western Europe, with a possible second route through Turkey and the Balkan Peninsula [7]. Rye may have initially been seen as a weed in wheat and barley fields and only later adapted to new conditions, performing well in colder climates [8]. In the Alpine region, rye likely transitioned from a weed to a crop during the pre-Roman Iron Age [6]. Its widespread cultivation began in the early Middle Ages when Europeans started farming steep slopes and poor soils [9]. Pollen cores from the region show the presence of S. cereale since then [10].
Recent studies based on different molecular data (Diversity Array Technology, microsatellites, and SNP) revealed a relatively low genetic diversity in rye accessions (both historical and modern) collected across the world. They suggested widespread germplasm exchange and a common genetic background of rye from different geographic regions [11,12]. In detail, historical accessions retrieved from rye gene banks exhibit divergence from modern accessions developed by European breeding companies, and a distinction of Southwestern Asian/South European populations from those of the rest of the European ones seems to be confirmed [11,12,13].
Crop genetic erosion over the last century has been globally documented for the primary food plants (including cereals) and has been attributed to the wide use of modern crop cultivars. These are genetically homogeneous and are cultivated in monoculture over large geographic areas [14]. The literature has extensively documented the significant decline in landraces and the diversity of local crop varieties over the past century up to the present day [14]. In S. cereale, the replacement of local landraces with modern commercial varieties has been described as one of the main causes of the loss of genetic diversity [15]. Genetic diversity of crops facilitates the production of varieties with specific traits to increase tolerance to abiotic and biotic stresses, and, finally, food quality and security [16,17]. For this reason, there has been a growing effort to preserve local landraces and crop varieties through ex situ collections, as well as the promotion of traditional agricultural practices. Indeed, landraces are widely recognized to have unique local agroecological and socio-economic functions, also offering important genetic resources in plant breeding programs [18], to support the mountain economy [19], and for nutritional and nutraceutical purposes [20]. Nutritional and nutraceutical aspects are particularly relevant for rye, given its substantial content of minerals, vitamins, and bioactive compounds. Several scientific studies attribute a favorable role to the non-starch polysaccharides found in rye, as they are rich in preventing cardiovascular diseases, obesity, and cancer [21,22]. Over the decades, the interest in the cultivation of local varieties of cereals (i.e., landraces) has increased in Europe [23]. In this context, European regulations of seed have imposed restrictions allowing seed exchange only on a limited scale [24]. Local varieties of cereal products have also become ever more popular among European consumers [25]. In the Alpine region, multifunctional agriculture practiced by small-scale farmers can play a strategic role in the conservation of slopes and terraced areas of mountain valleys [26,27]. Therefore, recovering the cultivation of traditional cereals like rye can favor local sustainable development and economy and reduce land abandonment. Considering all these historical, genetic, and socio-economic aspects, we conducted a multi-disciplinary study in Valtellina (Central Alps), where the cultivation of rye has drastically declined over the last century due to land abandonment. Specifically, to avoid genetic crop erosion and favor socio-economic benefits for the local community, the aims of this research were as follows:
- -
- Characterize the genetic structure through varietal genetic analyses based on codominant microsatellite (SSR) markers of 12 local landraces of rye collected from small-scale farmers in Valtellina (Central Alps), one alpine accession, and two commercial modern accessions from Poland and Russia;
- -
- Characterize the rye landraces from a nutraceutical point of view to explore the possibility of enhancing their value in the framework of the local economy.
- -
- Carry out agronomic analyses to estimate the productivity of the landraces and improve their short food supply chain.
2. Materials and Methods
2.1. The Study Species
Rye (Secale cereale L.) is an out-crossing, wind-pollinated cereal cultivated in world temperate regions, especially in Central, Eastern, and Northern Europe [11], where it is the second most used cereal for producing bread, after wheat [5]. The species exhibits good resistance to both biotic and abiotic stresses. In particular, its cultivation requires low soil fertility and water supply during the germination phases. Rye is reported to prefer sandy soils, but it is also able to grow in heavy clays and poorly drained soils. From a climatic point of view, it can thrive in cool weather conditions and germinate at temperatures as low as 1 °C, surviving periods of snow cover as well [28,29].
2.2. Plant Materials
Plant material of S. cereale consisted of 14 accessions (Table 1; Figure 1) collected from seed stocks during 2019:
- -
- Ten at the village of Teglio in Valtellina (T_1–T_10);
- -
- One in upper Valtellina (Up_V);
- -
- One population of alpine origin certified at the Laimburg Research Centre, an experimental station devoted to mountain agriculture in the Bolzano province (LAI);
- -
- One commercial accession from Poland (C_POL_P1) that was already analyzed in the study of Targońska et al. [12] (i.e., Danko farm, accession SZK44) where it was included in the group of the ancestry population P1 clustering accession from Central and Western Europe. The second cluster (ancestry population P2) of the study [12] included accessions from Southwest Asia, South Europe, and in the Balkans;
- -
- Another is from a local mill of Teglio (C_UN); about 20 years ago, a local farmer started producing rye from a commercial seed stock, originating from Russia.

Table 1.
Information on rye accessions analyzed in this study.
Table 1.
Information on rye accessions analyzed in this study.
| Population | Abbreviation | Sample Size (n) | Farmer | Origin |
|---|---|---|---|---|
| Teglio_1 | T_1 | 20 | Motalli | Teglio, Valtellina, Italy |
| Teglio_2 | T_2 | 20 | Mazzucchelli | Triangia (SO), Valtellina, Italy |
| Teglio_3 | T_3 | 14 | Finotti | Teglio, Valtellina, Italy |
| Teglio_4 | T_4 | 20 | Fanchi | Teglio, Valtellina, Italy |
| Teglio_5 | T_5 | 20 | Fanchetti | Teglio, Valtellina, Italy |
| Teglio_6 | T_6 | 19 | Marchetti | Teglio, Valtellina, Italy |
| Teglio_7 | T_7 | 20 | Arrondini | Teglio, Valtellina, Italy |
| Teglio_8 | T_8 | 20 | Saini | Teglio, Valtellina, Italy |
| Teglio_9 | T_9 | 16 | Pelacchi | Teglio, Valtellina, Italy |
| Teglio_10 | T_10 | 20 | De Filippi | Teglio, Valtellina, Italy |
| Upper_Valley | Up_V | 20 | Martinelli | Valdidentro, Valtellina, Italy |
| Laimburg | LAI | 20 | Experimental Centre | Laimburg, Adige Valley, Italy |
| Comm_Poland_P1 | C_POL_P1 | 19 | Commercial seeds | Poland (Danko farm, accession SZK44) and analyzed by Targońska et al. (2016) [12] |
| Comm_Uncertain | C_UN | 20 | Maxenti | Russia, (not coded) uncertain |
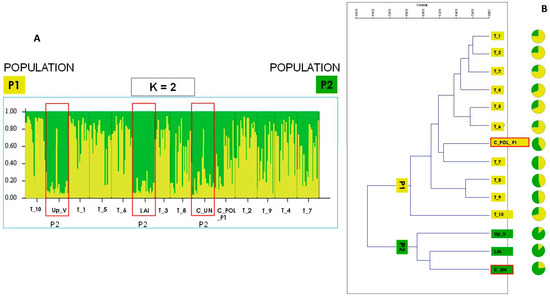
Figure 1.
(A) Results of the ΔK calculation (K = 2) with the bar diagram represent the proportion of ancestry in each of the K populations; accession assigned to the ancestry population P2 are highlighted in red. (B) Cluster analysis based on Nei’s genetic (UPGMA) distance between S. cereale accessions associated with results of the STRUCTURE analysis; accessions of commercial origin are highlighted in red.
The gene pool of each accession was constituted by a sample of 14–20 randomly chosen plants (see Table 1) germinated and grown in a growth chamber at the University of Milano-Bicocca at room temperature (20 °C).
PCR products were resolved using capillary electrophoresis with an ABI PRISM 3130 automatic sequencer Genetic Analyzer (Thermo Fisher Scientific, Waltham, MA, USA), Pop-7 polymer (Thermo Fisher Scientific, Waltham, MA, USA), and GeneScanTM 500 LIZTM as a size standard (Thermo Fisher Scientific, Waltham, MA, USA). Allele sizes were scored using GeneMapper software 4.0 (Thermo Fisher Scientific, Waltham, MA, USA). The resulting capillary sequencer electropherograms were visually inspected for clarity and the number of obtained fragments.
2.3. Genetic Analysis
Genomic DNA was isolated using the DNeasy Plant Mini Kit (Qiagen, Hilden, Germany) according to the instructions of the manufacturer. The integrity of DNA extracted from the samples was assessed by electrophoresis on a 1% agarose gel/1 × TAE containing 1 × Sybr Safe DNA gel stain (Life Technologies, Carlsbad, CA, USA). Subsequently, the quality and quantity of the isolated DNA were then determined from absorbance measurements.
All samples were analyzed with 8 specific microsatellite markers available for S. cereale (SSR: SCM2, SCM9, SCM43, SCM98, SCM152, SCM164, SCM166, SCM180; see Supplementary Table S1) and selected from a pool of 20 primers already tested by Targońska et al. [12].
Multiplex PCR amplification was optimized to be performed in a 10 μL reaction volume containing 2–10 ng of genomic DNA, 2 μL Hot FIREPol Multiplex Mix (Solis Biodyne, Tartu, Estonia), double distilled water, and respective concentrations of forward and reverse primers each (Supplementary Table S1). We used the following cycling protocol on a TC-412 Programmable Thermal Controller (Techne, St Neots, UK). Before the first cycle, a prolonged denaturation step (95 °C for 12 min) was included followed by 35 cycles with 95 °C for 20 s, 60 °C for 50 s, and 72 °C for 120 s. The last cycle was a 5 min extension at 72 °C.
2.4. Seed Characteristics and Agronomic Analysis
Image analysis software GrainScan v. 1.0 [30] was used to study the seed type of the different accessions from morphometric and colorimetric points of view. GrainScan can measure various parameters by returning grain size and color values (RGB) from images obtained by scanning 100 seeds at 300 dpi resolution (as suggested by the instruction manual).
To assess the performance of the different accessions and genotypes resulting from the genetic analyses, we performed field trials, adopting a randomized block experimental design with four repetitions and 7 × 3 m plots. Due to a lack of space, we were able to only replicate the 7 accessions from Teglio (T_3, T_4, T_6, T_7, T_8, T_9, T_10). The two alpine accessions (LAI, UP_V) and one commercial (C_UN) were sowed in only one replicate. For this reason, statistical comparisons were not performed for all the accessions but only for varieties identified by the genetic analyses.
In particular, we measured the following:
- -
- Plant height (cm);
- -
- Seed weight of 1000 seeds (g);
- -
- Seed color;
- -
- Crop yield (kg/ha).
2.5. Phytochemical Analysis
Phytochemical analyses were performed on the same subset of accessions and genotypes investigated for agronomic performance to compare plant materials grown in the same field conditions and cultivated with the same techniques (see Supplementary Table S2).
Total phenolic compound (TPC), total flavonoid content (TFC), and radical scavenging activity (RSA) were determined in whole grains of rye taking into consideration that many bioactive compounds are concentrated in the bran of the rye grain and only in low amounts in the flour endosperm [31,32]. TPC, TFC, and RSA are among the most used phytochemical indicators for characterizing nutraceutical performances of crop plants [33].
After being milled from mature rye grains, flour (0.15 g) was extracted in 2.25 mL of 80% (v/v) methanol and 0.1% HCl, with stirring for 12 h, and then centrifugated for 20 min (7830 rpm × g at 20 °C). The obtained supernatant was recovered and used for the TPC, TFC, and RSA determinations according to the methods reported below.
The extraction was performed at least in triplicate for each sample.
Antioxidant activity in vitro/radical scavenging activity (RSA): the antioxidant activity of the extracts was evaluated using the scavenging effect on 2,2-diphenyl-1-pycrylhydrazil radical (DPPH•) according to the method of Brand-Williams et al. [34], modified for the microplate screening assay.
Briefly, 25 μL of extracts/standard/MeOH (blank) was mixed with 100 μL of a 50 μM solution of DPPH in 60/40 MeOH/Acetate buffer (0.1 M, pH 5). The samples were incubated in the dark for 30 min, and absorbance was measured at 515 nm. The percentage (%) of radical scavenging activity was calculated using the following equation: RSA% = [(A 0 − A s)/A 0] × 100; where A 0 is the absorbance of the blank and A s is the absorbance of the sample at 515 nm. IC50 values refer to the concentration of the samples/standards required to scavenge 50% DPPH free radical. The scavenging activity was expressed as equivalents of Trolox equivalents (Trolox equiv. or TEAC) or Ascorbic acid equivalents (AA equiv. or AEAC) per kilogram of grain, where TEAC = IC50 Trolox/IC50 sample, and AEAC = IC50 Ascorbic Acid/IC50 sample.
Total phenolic compound (TPC): the total phenolic contents of the extracts were determined using the method based on the Folin–Ciocalteu reagent [35] modified for the microplate screening system.
Briefly, 20 μL of Folin–Ciocalteu reagent was added to 20 μL of each extract/standard/MeOH (blank). After mixing, samples were kept in the dark for 5 min, then 200 μL of an aqueous solution of Na2CO3 (10% w/v) was added by mixing. Samples were incubated in the dark for 90 min and absorbance was measured at 760 nm. The total phenolic content was expressed as mg of gallic acid equivalents (GA equiv.) per kilogram of grain.
Total flavonoid content (TFC): the total flavonoid contents of the extracts were determined according to Krošlák et al. [36] protocol modified for the microplate screening system. Briefly, 20 μL of a solution of AlCl3 (5% w/v in MeOH) or 20 μL of MeOH as blank was added to 100 μL of each extract/standard. Samples were incubated in the dark for 10 min and absorbance was measured at 415 nm. The total flavonoid content was expressed as mg of quercetin equivalents (Q equiv.) per kilogram of grain.
2.6. Data Analyses
Concerning the SSR analysis, the percentage of polymorphic loci (P), number of observed and expected alleles (Na and Ne, respectively), Shannon information index (I), observed heterozygosity (HO), expected heterozygosity (HE) across the populations, inbreeding coefficient, and estimate of gene flow (Nm = 1 − GST/(4GST) were calculated using in Genalex software version 6.5 [37]. The F-statistics (FST) of Wright [38], inbreeding coefficients (FIS = (HE − HO)/HE), and significance of deviations of FIS from zero using permutation tests were calculated for each population using FSTAT [39].
The ancestry of S. cereale samples was estimated with a Bayesian approach to model the structure of the different populations (i.e., accessions) in STRUCTURE version 2.3.4 [40,41]. Allele frequencies of the different S. cereale accessions were assumed to be correlated, which is a realistic model for populations that are likely to be similar due to common migration events and/or shared ancestry. To individuate the number of clusters, 20 independent runs of K (K = 1–14) were carried out with an admixture model, 1,000,000 Markov chain Monte Carlo (MCMC) iterations, and a 100,000 burn-in period (LOCPRIOR selection; estimate λ). Results of STRUCTURE were plotted according to the K value with the highest log-likelihood. These calculations were performed by processing the STRUCTURE result files in Structure Harvester version 0.6.94 [42].
Analysis of molecular variance (AMOVA) was performed using Genalex to estimate genetic differentiation within and among populations. The significance of the estimates was obtained through 999 data replications.
Differences in phytochemical contents (TPC, TFC, RSA) between accessions or ancestry populations were tested utilizing Analysis of Variance (ANOVA), with Tukey’s post hoc test to assess pairwise differences or by non-parametric tests (Mann–Whitney). When necessary, variables were log-transformed to achieve normality of model residuals. Correlation analyses between phytochemical contents and parameters relating to genetic variability were performed using Spearman’s correlation index.
3. Results
3.1. Genetic Analysis and Population Structure
A total of 57 alleles were generated by the 8 SSR markers across the 14 accessions of rye. The number of alleles detected per SSR primer pairs varied between 5 (SCM9, SCM98, and SCM164) and 11 (SCM43), with an average of 7.1 alleles per loci. A total of 5 rare alleles (8.77% of the total) were detected in this study. For the investigated loci, polymorphism information content (PIC), an estimator of the allelic diversity for a specific locus, ranged from 0.12 to 0.63 with an average of 0.37 (Table 2). The highest values of PIC were observed in 2 SSR loci, SCM2 (0.63) and SCM43 (0.51), then exhibited their utility in discriminating genotypes (see Supplementary Table S1).

Table 2.
Population genetic variability across the analyzed accessions. Na: observed number of alleles; Ne: effective number of alleles; I: Shannon index; Ho = observed heterozygosity; uHe: unbiased expected heterozygosity; F: inbreeding coefficient.
The percentage of polymorphic loci was 100% for all accessions. Across accessions, the number of alleles (Na) varied from 4.12 in T_3 to 5.50 in T_2, while the expected number of alleles (Ne) varied from 2.56 in LAI to 3.28 in T_7. The Shannon information index (I) exhibited the highest value in T_4 (I = 1.31) and the lowest one in LAI (I = 1.07). Across accessions, the observed heterozygosity (HO) varied from 0.48 in T_10 to 0.69 in T_9 with an average value of 0.61 (Table 2), while expected heterozygosity (HE) varied from 0.58 in LAI to 0.68 in T_4 and T_6 with an average value of 0.65. The inbreeding coefficient (F) exhibited values around 0 for all accessions indicating an equilibrium between homozygotes and heterozygotes (Table 2).
STRUCTURE analysis estimated the highest mean log-likelihood at K = 2 (ln P(K) (–5898.7)). This indicated that the studied accessions of S. cereale are subdivided into two distinct genetic clusters. Results were produced using an Admixture model suggesting that individuals may have mixed ancestors from different populations (Figure 1). The cluster analysis associated with the ancestry of populations resulting from STRUCTURE (pie graphs) revealed that all the Teglio accessions (T_1–T_10) were related and formed a separate cluster along with the commercial variety C_POL_P1 (originating from North-East Europe, Poland). On the other hand, the alpine accession of LAI, commercial accession (C_UN), and the Up_V, form the upper valley of Valtellina formed a separate cluster.
The overall genetic differentiation among populations (FST) was 0.072 while gene flow (Nm) was 3.64. The AMOVA (analysis of molecular variance) highlighted that most of the genetic variability (94%) was allocated within populations, while a minimal, but significant, proportion of the variation was present between populations (6%).
The occurrence between the two clusters was also highlighted by significantly lower average values of Na (z = 2.03; p < 0.05), I (z = 2.35; p < 0.05), and uHe (z = 1.97; p < 0.05) in P2 than in P1 applying the Mann–Whitney test.
3.2. Seed Characteristics and Agronomic Analysis
The shape of the seed (germplasm) collected from the different farmers was different between populations grouped according to P1 and P2 genetic ancestry (Figure 2). Seed weight of germplasm was the highest in P2 (H(chi2) = 22.44; p < 0.001). Similarly, the seed area and the ratio between seed axes were highest in P2 (Figure 2; Seed area: H(chi2) = 73.92, p < 0.001; Am/AM: H(chi2) = 135.80, p < 0.001). RGB colors exhibited significant differences in red, green, and blue components (R: H(chi2) = 152.2, p < 0.001; G: H(chi2) = 13.48, p < 0.001; B: H(chi2) = 9.34, p < 0.01).
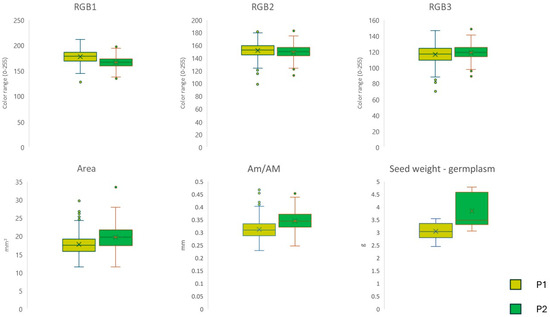
Figure 2.
Differences in seed shape between P1 and P2 ancestry populations based on RGB color components, seed area (mm2), the ratio between seed axes, and seed weight (g).
In Figure 3, the results of agronomic trials are shown. Overall, the accessions LAI, UP_V, and C_UN exhibited better performance than the accession from Teglio (Figure 3). Indeed, the productivity of rye ranged from 2449 kg/ha (in T_3) to 4479 kg/ha in (LAI). Plant height was the highest in the accessions of Teglio (especially T_9 and T_7 with a height of 195 cm) and lowest in LAI and UP_V (height of 145 cm). Therefore, accessions from Teglio were more subject to lodging.
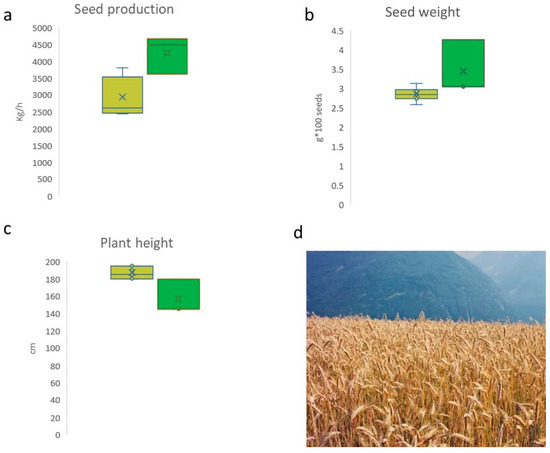
Figure 3.
Results of agronomic trials about seed production (a), seed weight (b), and plant height (c). Field of rye in Valtellina (d). Light green: P1 ancestry; green: P2 ancestry.
The seed weight (100 seeds) produced in the trial ranged from 30.04 g (in T_6) to 36.61 g (in C_UN).
3.3. Phytochemical Analysis
The contents in total phenolic compounds (TPC), flavonoids (TFC), and the radical scavenging activity (RSA) for the accessions tested in the Teglio experimental field during summer 2021 are shown in Figure 4.
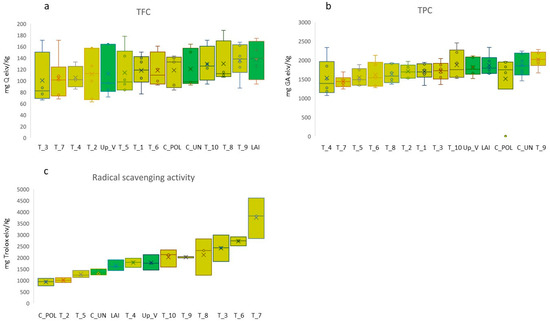
Figure 4.
Box plots of total phenolic compounds (a), total content of flavonoids (b), and radical scavenging activity (c) in the tested rye accessions (see Table 1 for abbreviation). Box plots are ordered according to increasing median values.
The highest content of TPC was found in accession T_9 (2015.9 ± 87.80), followed by T_10 (1872.6 ± 159.33) and C_UN (1872.1 ± 125.32), while the lowest was registered for T_7 (1436.6 ± 63.59). However, the statistical analysis did not show significant differences in TPC among the accessions (p = n.s.) and between the two genetic clusters P1 and P2. The same result was obtained for TFC: the highest content was found in accession LAI (137.0 ± 17.45), followed by T_9 (136.1 ± 15.15) and T_10 (129.2 ± 15.89), while the lowest was registered for accession T_3 (100.5 ± 23.91), but no significant differences in TFC between accessions were highlighted by the analysis (p = n.s.). Differently, statistical differences among accessions were recorded for RSA (H = 31.48; p < 0.01). The highest RSA was expressed by the accession T_7 (3756.2 ± 514.0), followed by T_6 (2720.6 ± 114.34) and T_3 (2416.8 ± 335.02), while the lowest was registered for C_UN (1351.7 ± 80.05). The analysis showed significantly higher RSA of T_7 with respect to C_UN, LAI, UP_V, T_4, T_8, T_9, T_10, and of T_6 with respect to C_UN (Figure 3c). RSA was accession specific and, on average, no statistical difference was observed between the two P1 and P2 clusters.
Correlation analyses between phytochemical contents (TFC, TPC, and RSA) and parameters relating to genetic population variability (Table 2) only exhibited negative relations between TPC and I (rs = 0.696; p < 0.01) and between TPC and uHe (rs = 0.695) p < 0.01).
4. Discussion
Our research on the recovery of rye cultivation in an alpine valley (Valtellina, Central Alps) indicated that the investigated local varieties (landraces) are worthy of conservation due to their genetic, nutraceutical, and agronomic characteristics.
Our genetic analysis revealed the presence of two distinct genetic lineages (ancestral populations) in the study area: the first cluster (P1) grouped all Teglio accessions (T) and the commercial one from Poland (C_POL_P1); the second cluster (P2) grouped the two alpine accessions, Up_V and LAI, and the commercial one of uncertain origin, C_UN. These results agree with the study by Targońska et al. [12], who, in turn, revealed the presence of two ancestral populations: (a) a first cluster (P1) mostly including populations from Central and Western Europe; (b) a second cluster (P2) mostly including populations from Southwestern Asia, South Europe, and the Balkans. Among our accessions, one (Comm_Poland_P1) was already analyzed in the study by Targońska et al. [12] (i.e., Comm_Poland_P1 = Danko farm, accession SZK44) and was included in the group of the ancestral population of Central and Western Europe (i.e., P1). Hence, it is highly likely that the second cluster of our analysis corresponds to the other cluster identified by Targońska et al. [12], including accessions from Southwestern Asia, South Europe, and the Balkans (i.e., P2).
The presence of two distinct ancestries suggests different historical cultivation paths of rye in Valtellina, especially between the upper and lower valleys. Indeed, the upper valley has historically been separated from the rest of Valtellina, and in ancient documents, it was reported as the County of Bormio with distinct economy and jurisdiction [43,44]. Particularly, since the Middle Ages, the medium Valtellina, with a warmer climate, was mainly cultivated with vines, while the upper Valtellina, with less favorable environmental conditions, was principally cultivated with rye [45,46]. From a genetic point of view, the two ancestral populations verified in Valtellina exhibit quite different patterns, with the accessions from the upper valley exhibiting lower levels of allelic and genetic diversities. In other contexts, this unbalanced genetic differentiation with unequal levels of erosion has been explained by long-term environmental isolation [47]. On one side, the lower genetic diversity of the upper valley population (i.e., P2 ancestry) can be attributed to the genetic loss of ancient lineages already observed in rye or other cereals [48,49]. On the other hand, the higher genetic diversity observed in the medium Valtellina (at Teglio) can be explained by more intense germplasm exchange, likely with populations from Central and Western Europe, for historical and economic reasons (see [50]).
Interestingly, the characteristics of the seeds and plants and their agronomic behavior were similar within the two genetic clusters P1 and P2. This result supports the real existence of two distinct groups of varieties with different origins cultivated in two separate areas of Valtellina (middle and upper Valley). The seed stocks used for genetic analyses and field agronomic trials revealed significant differences in color pattern (RGB) and size (seed area and ratio between main seed axes). In terms of color, P1 exhibited a more brownish hue, while the seeds of P2 appeared more greyish. Previous studies have predominantly attributed differences in seed color among cereal varieties to the human selection of germplasm (artificial allelic segregation) during historical utilization and cultivation [51]. However, some studies have demonstrated differential responses to environmental stressors (e.g., temperature, insulation, salinity, etc.) in the seed with color variants, invoking genetic mechanisms [52,53]. Seed size showed the highest values in P2 varieties. This trait, closely linked to seed weight, is among the principal quality attributes of cereal yield that have been consistently selected during crop domestication and seed exchanges [54]. Additionally, elevation (P2 accessions were collected from localities at higher elevations) might play a role in population differentiation via short-term changes in plant traits through plasticity mechanisms [55,56]. Regarding agronomic trials in the fields, we only reported the main trends. Indeed, reliable results regarding the quantity and quality of yields would require multi-year data collection and numerous replications across different environments [57,58]. Our trials revealed different trends between varieties P1 (including Teglio accessions) and P2 (including upper valley accessions). Particularly, P2 varieties exhibited higher seed production and weight, and lower plant size than P1 varieties.
In a recent study on rye landraces conducted in the western Alps, genotypes collected from higher elevations (i.e., Maira Valley) were characterized by taller and thinner culms but lower yield capacity [2]. This outcome may seem divergent from our own analysis, but it is not unexpected. In fact, rye genotypes can display varying performances influenced by factors such as their origin, historical selection, and growth environment [59]. Nonetheless, in mountain regions, several rye traits across different landraces (e.g., winter hardiness and ripening time) have been reported to be influenced by the elevation of the occurrence where the landraces were collected, indicating a degree of adaptation of rye to climatic conditions [59].
Nevertheless, the extensive historical selection and adaptation to local conditions in Valtellina are apparent when examining the antioxidant properties of different rye accessions cultivated in the same year and location within Valtellina. Differently from agronomic performance and seed/plant characteristics (that were similar within P1 and P2 groups), the antioxidant activity we determined was variety specific. Most of the Valtellina landraces, independently ascribed to the P1 or P2 group, showed a higher radical scavenging activity (RSA) in comparison to the commercial accessions, and in particular T7 showed a four-fold higher RSA than that of the commercial accession Comm_Polland_P1. It agrees with Kulichová et al. [32], who reported considerable differences in the antioxidant properties of 19 rye genotypes. However, the authors ascribed the different radical scavenging properties to the provenience of the genotypes from different parts of Europe, and to their different morphological and agronomical traits. Instead, we can speculate that each Valtellina rye accession adapted to the local conditions by changing the synthesis of some classes of secondary metabolites, usually involved in plant resistance to environmental stresses. Unfortunately, our analyses showed that the content of TPC and TFC did not explain the high RSA found in the Valtellina landraces, as they were similar in all the tested accessions and not significantly correlated to RSA. The negative correlation between TPC and RSA was also evident in rye landraces from the Northwest Italian Alps, which, despite showing a higher content of polyphenols than commercial accessions, did not show a higher RSA [2]. Thus, further studies are needed to discover the antioxidant compounds explaining the high RSA found in some Valtellina landraces. Previous studies suggested that the high antioxidant capacity of rye grains can be related to alkylresolcinols in the bran fraction, whose content is greater than in wheat, oat, and triticale [60,61]. However, these substances are unlikely to be responsible for the high RSA, since these amphiphilic phenolic lipids present an important hydrophobic chain that makes their extraction unlikely under the methanol-based conditions applied in this work.
On the other hand, our results exhibited a negative correlation between phenolic content and genetic variability (expected heterozygosity and Shannon index). This result can be ascribed to the overall lower genetic variability of alpine landraces (P2) compared with the Teglio ones (P2). Multi-genic control can be invoked for the synthesis of phenolic compounds and has been explained by the relationship between their biosynthesis and other metabolic pathways (e.g., carbohydrates) [62]. In this case, the higher elevation of P2 accession can have played a role in their increased content of phenolic compounds.
Implication for the Rural Mountain Economy
In conclusion, recognizing local varieties of cereal species, such as the rye varieties we investigated, can yield several benefits for mountain economies and communities in Valtellina [63]. Indeed, the varieties of the middle (P1 lineage) and upper valley (P2 lineage) may be valorized following agroecological approaches with advantages for local farmers and stakeholders in several respects. In addition, phytochemical results allowed the identification of better accessions to exploit for nutraceutical purposes (e.g., commercialization). These benefits coming from the valorization of local crop varieties are common for different mountain or marginal regions, and crop types:
(a) Conservation of agro-biodiversity: Local varieties embody a rich genetic diversity crucial for preserving resilient and sustainable agricultural systems. Cultivating these varieties contributes to conserving agro-biodiversity in rural mountain areas and mitigating the risk of losing significant genetic resources [64,65].
(b) Income diversification: Local crop varieties offer mountain region farmers an opportunity to diversify their income. By cultivating crop varieties suited to mountain lands and climates, like rye, farmers can mitigate risks and vulnerabilities associated with crop failures or market fluctuations [66]. Moreover, reclaiming marginal and abandoned fields can promote local food chains and agro-tourism, further enhancing income and diversification [27,67].
(c) Preservation and transmission of traditional knowledge: Cultivating and preserving local crop varieties helps rural mountain communities maintain their traditions of ancient farming practices and cultural heritage (e.g., traditional instruments, mills) and pass on historical knowledge to future generations [68].
(d) Enhancement of food security: The resilience of local varieties to harsh environmental conditions in mountain areas and climatic fluctuations can bolster food security in mountain regions by promoting food self-sufficiency, even in challenging conditions [69].
In the case of this study, these targets are going to be achieved. In fact, the analyses we performed contributed to the creation of a protected label of Municipal Origin Denomination (Denominazione Comunale di Origine, DECO) and to the formation of an association of local farmers to implement a short food chain supply.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/agronomy14081605/s1, Table S1: SSR Primers; Table S2: Phytochemical analyses.
Author Contributions
Conceptualization, R.G. and S.C.; methodology, R.G., E.C., F.G., G.D.M. and B.L.F.; formal analysis, R.G. and E.C.; investigation, R.G., E.C. and G.D.M.; data curation, R.G.; writing—original draft preparation, R.G.; writing—review and editing, R.G., E.C., S.C., F.G., G.M., B.L.F., G.D.M. and G.P.; funding acquisition, S.C. and F.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by REGIONE LOMBARDIA, grant number 2019-NAZ-0046; project: “CONSERVA-Conservazione, gestione ed uso sostenibile delle risorse genetiche di grano saraceno e segale in Valtellina”.
Data Availability Statement
Dataset available on request from the authors.
Acknowledgments
We acknowledge that the seeds used during the project are conserved in the Seed Bank of the Centro Flora Autoctona della Regione Lombadia (Parco Monte Barro). We are grateful to the Municipality of Teglio (especially to the vice-mayor Paolo Pedroli) for the support during the project and to all the local farmers who supplied the seed for the analyses.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Canella, M.; Ardenghi, N.M.G.; Müller, J.V.; Rossi, G.; Guzzon, F. An updated checklist of plant agrobiodiversity of northern Italy. Genet. Resour. Crop Evol. 2022, 69, 2159–2178. [Google Scholar] [CrossRef]
- Sardella, C.; Capo, L.; Adamo, M.; Donna, M.; Ravetto Enri, S.; Vanara, F.; Lonati, M.; Mucciarelli, M.; Blandino, M. The cultivation of rye in marginal Alpine environments: A comparison of the agronomic, technological, health and sanitary traits of local landraces and commercial cultivars. Front. Plant Sci. 2023, 14, 1130543. [Google Scholar] [CrossRef]
- Zohary, D.; Hopf, M.; Weiss, E. Domestication of Plants in the Old World: The Origin and Spread of Domesticated Plants in Southwest Asia, Europe, and the Mediterranean Basin; Oxford University Press on Demand: Oxford, UK, 2012. [Google Scholar]
- Hillman, G. On the Origins of Domestic Rye—Secale cereale: The Finds from Aceramic Can Hasan III in Turkey. Anatol. Stud. 1978, 28, 157–174. [Google Scholar] [CrossRef]
- Schreiber, M.; Himmelbach, A.; Börner, A.; Mascher, M. Genetic diversity and relationship between domesticated rye and its wild relatives as revealed through genotyping-by-sequencing. Evol. Appl. 2019, 12, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Behre, K.-E. The history of rye cultivation in Europe. Veget. Hist. Archaeobot. 1992, 1, 141–156. [Google Scholar] [CrossRef]
- Ma, R.; Yli-Mattila, T.; Pulli, S. Phylogenetic relationships among genotypes of worldwide collection of spring and winter ryes (Secale cereale L.) determined by RAPD-PCR markers. Hereditas 2004, 140, 210–221. [Google Scholar] [CrossRef]
- Preece, C.; Livarda, A.; Christin, P.-A.; Wallace, M.; Martin, G.; Charles, M.; Jones, G.; Rees, M.; Osborne, C.P. How did the domestication of fertile crescent grain crops increase their yields? Funct. Ecol. 2017, 31, 387–397. [Google Scholar] [CrossRef]
- Mucciarelli, M.; Blandino, M.; Capo, L.; Adamo, M. La Segale in Piemonte. Storia di Una Rinascita; Ecomuseo della Segale, Tipolito Europa, Cuneo: Valdieri, Italy, 2022. [Google Scholar]
- Festi, D.; Putzer, A.; Oeggl, K. Mid and late Holocene land-use changes in the Ötztal Alps, territory of the Neolithic Iceman “Ötzi”. Quat. Int. 2014, 353, 17–33. [Google Scholar] [CrossRef]
- Bolibok-Brągoszewska, H.; Targońska, M.; Bolibok, L.; Kilian, A.; Rakoczy-Trojanowska, M. Genome-wide characterization of genetic diversity and population structure in Secale. BMC Plant Biol. 2014, 14, 184. [Google Scholar] [CrossRef]
- Targońska, M.; Bolibok-Brągoszewska, H.; Rakoczy-Trojanowska, M. Assessment of Genetic Diversity in Secale cereale Based on SSR Markers. Plant Mol. Biol. Rep. 2016, 34, 37–51. [Google Scholar] [CrossRef]
- Hagenblad, J.; Oliveira, H.R.; Forsberg, N.E.G.; Leino, M.W. Geographical Distribution of Genetic Diversity in Secale Landrace and Wild Accessions. BMC Plant Biol. 2016, 16, 23. [Google Scholar] [CrossRef] [PubMed]
- Khoury, C.K.; Brush, S.; Costich, D.E.; Curry, H.A.; de Haan, S.; Engels, J.M.M.; Guarino, L.; Hoban, S.; Mercer, K.L.; Miller, A.J.; et al. Crop Genetic Erosion: Understanding and Responding to Loss of Crop Diversity. New Phytol. 2022, 233, 84–118. [Google Scholar] [CrossRef] [PubMed]
- Leoni, V.; Pedrali, D.; Zuccolo, M.; Rodari, A.; Giupponi, L.; Giorgi, A. The Importance of Technical Support in the Return of Traditional Crops in the Alps: The Case of Rye in Camonica Valley. Sustainability 2021, 13, 13818. [Google Scholar] [CrossRef]
- Giupponi, L.; Borgonovo, G.; Panseri, S.; Giorgi, A. Multidisciplinary Study of a Little Known Landrace of Fagopyrum tataricum Gaertn. of Valtellina (Italian Alps). Genet. Resour. Crop Evol. 2019, 66, 783–796. [Google Scholar] [CrossRef]
- Begna, T. Role and economic importance of crop genetic diversity in food security. J. Agric. Sci. Food Technol. 2021, 7, 164–169. [Google Scholar] [CrossRef]
- Ramirez-Villegas, J.; Khoury, C.K.; Achicanoy, H.A.; Diaz, M.V.; Mendez, A.C.; Sosa, C.C.; Kehel, Z.; Guarino, L.; Abberton, M.; Aunario, J.; et al. State of ex situ conservation of landrace groups of 25 major crops. Nat. Plants 2022, 8, 491–499. [Google Scholar] [CrossRef]
- Giupponi, L.; Pilu, R.; Scarafoni, A.; Giorgi, A. Plant Agro-biodiversity Needs Protection, Study and Promotion: Results of Research Conducted in Lombardy Region (Northern Italy). Biodivers. Conserv. 2020, 29, 409–430. [Google Scholar] [CrossRef]
- Singh, S.; Singh, L.; Singh, H.; Sangwan, S. Nutraceutical Potential of Seed and Grain Proteins in Health Promotion. In Grain and Seed Proteins Functionality; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Bondia-Pons, I.; Aura, A.-M.; Vuorela, S.; Kolehmainen, M.; Mykkänen, H.; Poutanen, K. Rye phenolics in nutrition and health. J. Cereal Sci. 2009, 49, 323–336. [Google Scholar] [CrossRef]
- Garutti, M.; Nevola, G.; Mazzeo, R.; Cucciniello, L.; Totaro, F.; Bertuzzi, C.A.; Caccialanza, R.; Pedrazzoli, P.; Puglisi, F. The Impact of Cereal Grain Composition on the Health and Disease Outcomes. Front. Nutr. 2022, 9, 888974. [Google Scholar] [CrossRef]
- Ortman, T.; Sandström, E.; Bengtsson, J.; Watson, C.A.; Bergkvist, G. Farmers’ motivations for landrace cereal cultivation in Sweden. Biol. Agric. Hort. 2023, 39, 247–268. [Google Scholar] [CrossRef]
- Batur, F.; Bocci, R.; Bartha, B. Marketing farmers’ varieties in Europe: Encouraging pathways with missing links for the recognition and support of farmer seed systems. Agronomy 2021, 11, 2159. [Google Scholar] [CrossRef]
- Wendin, K.; Mustafa, A.; Ortman, T.; Gerhardt, K. Consumer awareness, attitudes and preferences towards heritage cereals. Foods 2020, 9, 742. [Google Scholar] [CrossRef] [PubMed]
- Magnani, N.; Struffi, L. Translation sociology and social capital in rural development initiatives. A case study from the Italian Alps. J. Rural Stud. 2009, 25, 231–238. [Google Scholar] [CrossRef]
- de Pasquale, G.; Spinelli, E. The alpine rural landscape as a cultural reserve: The case study of Teglio in Valtellina. Biodivers. Conserv. 2022, 31, 2397–2420. [Google Scholar] [CrossRef]
- Prończuk, M.; Madej, L.; Kolasińska, I. Research for resistance to Microdochium nivale among inbred lines of rye. Plant Breed. Seed Sci. 2003, 48, 83–86. [Google Scholar]
- Clark, A. (Ed.) Managing Cover Crops Profitably, 3rd ed.; Sustainable Agriculture Research and Education (SARE) Program; Diane Publishing: Collingdale, PA, USA, 2007. [Google Scholar]
- Whan, A.P.; Smith, A.B.; Cavanagh, C.R.; Ral, J.-P.F.; Shaw, L.M.; Howitt, C.A.; Bischof, L. GrainScan: A low cost, fast method for grain size and colour measurements. Plant Methods 2014, 10, 23. [Google Scholar] [CrossRef]
- Weselake, R.J.; MacGregor, A.W.; Hill, R.D. Endogenous alpha-amylase inhibitor in various cereals. Cereal Chem. 1985, 62, 120–123. [Google Scholar]
- Kulichová, K.; Sokol, J.; Nemeček, P.; Maliarová, M.; Maliar, T.; Havrlentová, M.; Kraic, J. Phenolic Compounds and Biological Activities of Rye (Secale cereale L.) Grains. Open Chem. 2019, 17, 988–999. [Google Scholar] [CrossRef]
- Shi, L.; Zhao, W.; Yang, Z.; Subbiah, V.; Suleria, H.A.R. Extraction and characterization of phenolic compounds and their potential antioxidant activities. Environ. Sci. Pollut. Res. 2022, 29, 81112–81129. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Hosu, A.; Cristea, V.M.; Cimpoiu, C. Analysis of Total Phenolic, Flavonoids, Anthocyanins and Tannins Content in Romanian Red Wines: Prediction of Antioxidant Activities and Classification of Wines Using Artificial Neural Networks. Food Chem. 2014, 150, 113–118. [Google Scholar] [CrossRef]
- Krošlák, E.; Maliar, T.; Nemeček, P.; Viskupičová, J.; Maliarová, M.; Havrlentová, M.; Kraic, J. Antioxidant and Proteinase Inhibitory Activities of Selected Poppy (Papaver somniferum L.) Genotypes. Chem. Biodivers. 2017, 14, e1700176. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. GENALEX 6: Genetic analysis in Excel. Population genetic software for teaching and research. Mol. Ecol. Notes 2006, 6, 288–295. [Google Scholar] [CrossRef]
- Wright, S. The genetical structure of populations. Ann. Eugen. 1951, 15, 323–354. [Google Scholar] [CrossRef]
- Goudet, J. FSTAT (Version 1.2): A Computer Program to Calculate F-Statistics. J. Hered. 1995, 86, 485–486. [Google Scholar] [CrossRef]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar] [CrossRef]
- Falush, D.; Stephens, M.; Pritchard, J.K. Inference of population structure using multilocus genotype data: Dominant markers and null alleles. Mol. Ecol. Notes 2007, 7, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Earl, D.A.; vonHoldt, B.M. STRUCTURE HARVESTER: A website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Besta, E. Bormio Antica e Medievale, e le Sue Relazioni Con le Potenze Finitime; Edizioni Giuffrè: Milano, Italy, 1945. [Google Scholar]
- Della Misericordia, M. Giustizia, identità locale ed esclusione a Bormio nel Quattrocento. Boll. Stor. Alta Valtellina 2010, 13, 79–126. [Google Scholar]
- Antonioli, G. Una relazione ottocentesca sullo stato dell’ex contado di Bormio. Boll. Stor. Dell’alta Valtellina 2001, 4, 133–149. [Google Scholar]
- Scaramellini, G. Coltura della vite, produzione e commercio del vino in Valtellina (secoli IX-XVIII). Territ. du vin 2014. Available online: http://preo.u-bourgogne.fr/territoiresduvin/index.php?id=829 (accessed on 22 December 2023). [CrossRef]
- Marques, I.; Shiposha, V.; López-Alvarez, D.; Manzaneda, A.J.; Hernandez, P.; Olonova, M.; Catálan, P. Environmental isolation explains Iberian genetic diversity in the highly homozygous model grass Brachypodium distachyon. BMC Evol. Biol. 2017, 17, 139. [Google Scholar] [CrossRef] [PubMed]
- Adamo, M.; Blandino, M.; Capo, L.; Ravetto Enri, S.; Fusconi, A.; Lonati, M.; Mucciarelli, M.A. ddRADseq Survey of the Genetic Diversity of Rye (Secale cereale L.) Landraces from the Western Alps Reveals the Progressive Reduction of the Local Gene Pool. Plants 2021, 10, 2415. [Google Scholar] [CrossRef] [PubMed]
- Lopes, M.S.; El-Basyoni, I.; Baenziger, P.S.; Singh, S.; Royo, C.; Ozbek, K.; Aktas, H.; Ozer, E.; Ozdemir, F.; Manickavelu, A.; et al. Exploiting genetic diversity from landraces in wheat breeding for adaptation to climate change. J. Exp. Bot. 2015, 66, 3477–3486. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, D.; Yu, K.; Ji, J.; Liu, N.; Zhang, Y.; Xu, M.; Zhang, Y.-J.; Ma, X.; Liu, S.; et al. Frequent germplasm exchanges drive the high genetic diversity of Chinese-cultivated common apricot germplasm. Hortic. Res. 2021, 8, 215. [Google Scholar] [CrossRef] [PubMed]
- Stetter, M.G.; Vidal-Villarejo, M.; Schmid, K.J. Parallel Seed Color Adaptation during Multiple Domestication Attempts of an Ancient New World Grain. Mol. Biol. Evol. 2020, 37, 1407–1419. [Google Scholar] [CrossRef] [PubMed]
- Giraldo, P.; Royo, C.; González, M.; Carrillo, J.M.; Ruiz, M. Genetic diversity and association mapping for agromorphological and grain quality traits of a structured collection of durum wheat landraces including subsp. durum, turgidum and diccocon. PLoS ONE 2016, 11, e0166577. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Moreno, F.; Giraldo, P.; Cátedra, M.d.M.; Ruiz, M. Evaluation of Leaf Rust Resistance in the Spanish Core Collection of Tetraploid Wheat Landraces and Association with Ecogeographical Variables. Agriculture 2021, 11, 277. [Google Scholar] [CrossRef]
- Tao, Y.; Mace, E.S.; Tai, S.; Cruickshank, A.; Campbell, B.C.; Zhao, X.; Van Oosterom, E.J.; Godwin, I.D.; Botella, J.R.; Jordan, D.R. Whole-Genome Analysis of Candidate genes Associated with Seed Size and Weight in Sorghum bicolor Reveals Signatures of Artificial Selection and Insights into Parallel Domestication in Cereal Crops. Front. Plant Sci. 2017, 8, 1237. [Google Scholar] [CrossRef]
- Olejniczak, P.; Czarnoleski, M.; Delimat, A.; Majcher, B.M.; Szczepka, K. Seed size in mountain herbaceous plants changes with elevation in a species-specific manner. PLoS ONE 2018, 13, e0199224. [Google Scholar] [CrossRef]
- Wang, X.; Alvarez, M.; Donohue, K.; Ge, W.; Cao, Y.; Liu, K.; Du, G.; Bu, H. Elevation filters seed traits and germination strategies in the eastern Tibetan Plateau. Ecography 2021, 44, 242–254. [Google Scholar] [CrossRef]
- Laurent, A.; Pelzer, E.; Loyce, C.; Makowski, D. Ranking yields of energy crops: A meta-analysis using direct and indirect comparisons. Renew. Sustain. Energy Rev. 2015, 46, 41–50. [Google Scholar] [CrossRef]
- Brown, D.; Van den Bergh, I.; de Bruin, S.; Machida, L.; van Etten, J. Data synthesis for crop variety evaluation. A review. Agron. Sustain. Dev. 2020, 40, 25. [Google Scholar] [CrossRef] [PubMed]
- Peratoner, G.; Seling, S.; Klotz, C.; Florian, C.; Figl, U.; Schmitt, A.O. Variation of agronomic and qualitative traits and local adaptation of mountain landraces of winter rye (Secale cereale L.) from Val Venosta/Vinschgau (South Tyrol). Genet. Resour. Crop Evol. 2016, 63, 261–273. [Google Scholar] [CrossRef]
- Mattila, P.; Pihlava, J.M.; Hellström, J. Contents of phenolic acids, alkyl- and alkenylresorcinols, and avenanthramides in commercial grain products. J. Agric. Food Chem. 2005, 53, 8290–8295. [Google Scholar] [CrossRef] [PubMed]
- Gliwa, J.; Gunenc, A.; Ames, N.; Willmore, W.G.; Hosseinian, F.S. Antioxidant Activity of Alkylresorcinols from Rye Bran and Their Protective Effects on Cell Viability of PC-12 AC Cells. J. Agric. Food Chem. 2011, 59, 11473–11482. [Google Scholar] [CrossRef] [PubMed]
- Łukaszewicz, M.; Szopa, J. Pleiotropic effect of flavonoid biosynthesis manipulation in transgenic potato plants. Acta Physiol. Plant 2005, 27, 221–228. [Google Scholar] [CrossRef]
- Barcaccia, G.; Volpato, M.; Gentili, R.; Abeli, T.; Galla, G.; Orsenigo, S.; Citterio, S.; Sgorbati, S.; Rossi, G. Genetic identity of common buckwheat (Fagopyrum esculentum Moench) landraces locally cultivated in the Alps. Genet. Resour. Crop Evol. 2016, 63, 639–651. [Google Scholar] [CrossRef]
- Giupponi, L.; Pedrali, D.; Leoni, V.; Rodari, A.; Giorgi, A. The Analysis of Italian Plant Agrobiodiversity Databases Reveals That Hilly and Sub-Mountain Areas Are Hotspots of Herbaceous Landraces. Diversity 2021, 13, 70. [Google Scholar] [CrossRef]
- Akhalkatsi, M.; Otte, A.; Togonidze, N.; Bragvadze, T.; Asanidze, Z.; Arabuli, G.; Chikhelidze, N.; Mazanishvili, L. Agrobiodiversity and genetic erosion of crop varieties and plant resources in the Central Great Caucasus. Ann. Agrar. Sci. 2017, 15, 11–16. [Google Scholar] [CrossRef]
- Wan, J.; Li, R.; Wang, W.; Liu, Z.; Chen, B. Income Diversification: A Strategy for Rural Region Risk Management. Sustainability 2016, 8, 1064. [Google Scholar] [CrossRef]
- Colombo, F.; Franguelli, N.; Licheri, G.; Ghidoli, M.; Cassani, E.; Castelli, L.; Pasquali, M.; Bresciani, A.; Marti, A.; Dell’Anno, M.; et al. Agriculture in Marginal Areas: Reintroduction of Rye and Wheat Varieties for Breadmaking in the Antrona Valley. Agronomy 2022, 12, 1695. [Google Scholar] [CrossRef]
- Limpo, S.Y.; Fahmid, I.M.; Fattah, A.; Rauf, A.W.; Surmaini, E.; Muslimin; Saptana; Syahbuddin, H.; Andri, K.B. Integrating Indigenous and Scientific Knowledge for Decision Making of Rice Farming in South Sulawesi, Indonesia. Sustainability 2022, 14, 2952. [Google Scholar] [CrossRef]
- Rasul, G.; Hussain, A. Sustainable Food Security in the Mountains of Pakistan: Towards a Policy Framework. Ecol. Food Nutr. 2015, 54, 625–643. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).