Abstract
In this paper, we aimed to evaluate clinical and imagistic features, and also to provide a diagnostic algorithm for patients presenting with gastrointestinal involvement from hepatocellular carcinoma (HCC). We conducted a systematic search on the PubMed, Scopus and Web of Science databases to identify and collect papers oncases of HCC with gastrointestinal involvement. This search was last updated on 29 April 2022. One hundred and twenty-three articles were included, corresponding to 197 patients. The majority of the patients were male (87.30%), with a mean age of 61.21 years old. The analysis showed large HCCs located mainly in the right hepatic lobe, and highly elevated alfa-fetoprotein (mean = 15,366.18 ng/mL). The most frequent etiological factor was hepatitis B virus (38.57%). Portal vein thrombosis was present in 27.91% of cases. HCC was previously treated in most cases by transarterial chemoembolization (32.99%) and surgical resection (28.93%). Gastrointestinal lesions, developed mainly through direct invasion and hematogenous routes, were predominantly detected in the stomach and duodenum in equal measure—27.91%. Gastrointestinal bleeding was the most common presentation (49.74%). The main diagnostic tools were esophagogastroduodenoscopy (EGD) and computed tomography. The mean survival time was 7.30 months. Gastrointestinal involvement in HCC should be included in the differential diagnosis of patients with underlying HCC and gastrointestinal manifestations or pathological findings in EGD.
1. Introduction
Hepatocellular carcinoma (HCC) is the most common primary malignant tumor of the liver and the sixth most common cancer, according to GLOBOCAN 2020 data. Although both incidence and mortality rates declined in many high-risk areas, many patients have already reached an advanced stage at diagnosis, resulting in 830,000 deaths worldwide [1].
Hepatocellular carcinoma usually disseminates to the liver [2]. Although less common, in 30–50% of the cases, HCC can have extrahepatic spread. The most frequent areas are the lungs, followed by lymph nodes and bones [3,4,5]. Involvement of the gastrointestinal (GI) tract is a rare event, with a reported incidence of 0.5–2% of all HCCs. Higher rates of 4–12% have been recorded in autopsy cases [6,7].
Due to non-specific symptomatology or endoscopic features, GI involvement by HCC is underdiagnosed premortem [8]. The available data about this condition are provided mainly by case reports and only a few literature reviews. In 2004, Fujii, K. et al. reviewed the characteristics of 29 HCC patients with GI tract invasion [9]. Later on, in 2011, Lin, T.L. et al. assessed the course of disease and survival in 44 patients reported in the English literature with direct invasion of the GI tract by HCC [10]. In the same year, Kato, Y. et al. (2011) investigated the role of surgical treatment based on 18 cases from the literature (English literature and Japanese literature with English abstract), including his reported case [11]. In 2018, Harada, J. et al. also reviewed the clinical characteristics of esophageal metastases from HCC [12]. Recently, Yu, Y.M. et al. (2020) listed and analyzed 15 patients with metastases from HCC in the small bowel and large intestine, followed in 2021 by Mu, M. et al., who provided a literature summary of 21 HCC colonic metastases [13,14]. However, these studies included a limited number of cases and were focused on a specific route of metastasis, or the involvement of a particular segment of the GI tract. Additional cases have been reported since these previous publications.
The HCC survival rate has increased over the last three decades and we expect it to further increase as a result of improvements in therapy and early diagnosis [15]. For that reason, we also expect to see a more significant number of patients with atypical complications in our clinical practice [16]. An early diagnosis of GI involvement from HCC is a challenge for clinicians, and raising awareness of this issue is a crucial step toward it. Our systematic review complements previous studies and gives a bigger picture of the main clinical and imagistic characteristics of GI involvement from HCC. We also propose an algorithm diagnosis that would serve clinicians in making a rapid diagnosis.
2. Materials and Methods
A systematic review of case studies of gastrointestinal involvement from hepatocellular carcinoma was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (Table S1) [17].
2.1. Data Sources
The systematic search was performed in the PubMed, Scopus and Web of Science databases up to November 2021, with the last search update on 29 April. A search in the Scopus and Web of Science database was also performed to avoid missing relevant articles up to 29 April. Randomly discovered searches, mainly from the manual search of references, were also included.
2.2. Inclusion and Exclusion Criteria
Our search strategy was developed on PICO (Patient/Population, Intervention, Comparison, Outcome) elements as follows: population: patients with an imagistic or histopathological diagnosis of hepatocellular carcinoma involving the gastrointestinal tract; intervention: none; comparison: none; outcomes: case studies that reported clinical presentation, diagnosis or survival of HCC with gastrointestinal involvement. All published case reports, case series or retrospective observational studies on the PubMed database concerning GI involvement from hepatocellular carcinoma were considered eligible for inclusion. We extended our search to editorial material that provided enough data for retrieval and analysis. No restriction for the time of publication was fixed. Only articles that addressed the specific clinical topic were selected for the examination. We excluded articles written in languages other than English, those not focused on the main issue, and papers not available as full text or papers with insufficient data to support the analysis.
2.3. Search Strategy
The search strategy included a combination of the following terms connected with the Boolean operators “AND” and “OR”: “hepatocellular carcinoma”, “esophagus”, “stomach”, “gastric”, “duodenum”, “jejunum”, “ileum”, “intestine”, “colon”, “rectum”, “invasion”, “metastasis”. No filters were added. The search strategy is reported as Supplementary Materials (Table S2). In the first stage, duplicate references were removed from our results using EndNote 20 (Clarivate Analytics, Chandler, AZ, USA). Further, tiles/± abstracts of the records found in the first stage were screened by two reviewers, and reports that were not on the main subject were excluded. Cohen’s Kappa coefficient was calculated, obtaining a satisfactory value of 0.94. The next step was to eliminate non-English articles. The full text of the articles was retrieved when available and further analyzed. Articles containing insufficient data and those not related to the main topic were excluded.
Further, we composed a data-extracting sheet in Microsoft Excel (Microsoft Office Professional Plus 2019), Microsoft® Excel® 2019 MSO (Version 2111 Build 16.0.14701.20204) 32-bit. One member of our team performed the initial data extraction and it was checked for reliability by a second member.
The following data were extracted: additional bibliographic information, including name of the first author, year of publication and type of study; number of patients reported in the paper; patient’s demographics, such as gender and age; etiology; location and size of HCC; previous treatment for HCC; AFP value (ng/mL); presence of portal vein thrombosis; the involved segment of the gastrointestinal tract; clinical presentation; involvement route; imagistic methods used for diagnosis of GI lesions; endoscopic features; methods used to obtain histological specimens and whether or not immunohistochemistry was used, as well as the survival time. The extracted data were further processed. Descriptive statistics (mean, standard deviation, percentages, minimum, maximum) were used to summarize the analyzed data.
Any discrepancy between reviewers was discussed, and a final consensus was reached.
In order to assess the risk of bias in our systematic review of case reports and case series, we used the Joanna Briggs Critical Appraisal Checklist for case reports and for case series (Tables S3 and S4) [18].
3. Results
3.1. Publication Characteristics
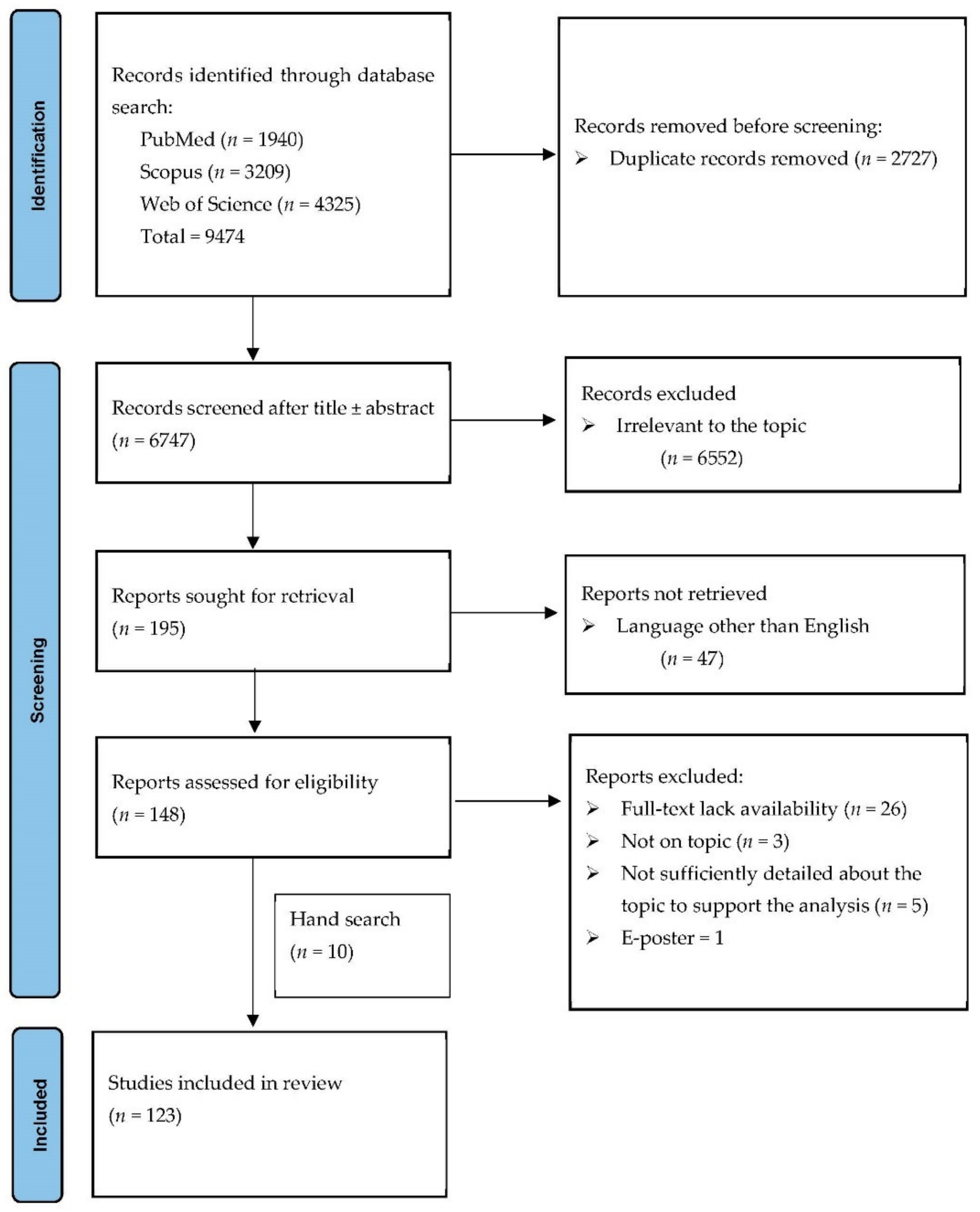
Figure 1 illustrates the PRISMA flow diagram of our search strategy. We initially identified 9474 record titles, from which we excluded 2727 duplicates. Further, from the remaining 6747 records, only 195 were sought for retrieval. Forty-seven articles were non-English publications. In the last stage, from the 148 reports considered eligible, one was an E-poster, 26 did not have full-text availability, three were not on discussed topic and five did not have sufficient detailed data to support the analysis (Table S5). We identified ten additional publications on manual search. One hundred twenty-three articles concerning 197 patients were included in the final analysis (Table 1 and Table 2).
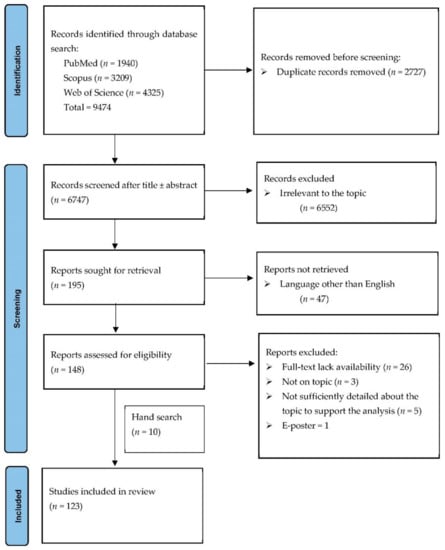
Figure 1.
PRISMA flow diagram for the selection process of the cases.

Table 1.
Literature review of cases with gastrointestinal involvement from hepatocellular carcinoma—characteristics of HCC tumors.

Table 2.
Literature review of cases with gastrointestinal involvement from hepatocellular carcinoma—features of GI metastases.
3.2. Patient Characteristics
We included 197 cases, with a mean age of 61.21 (standard deviation = 11.66), ranging from 22–86. The majority of patients were male (n = 172; 87.30%), with a ratio of man: woman of 6.88.
3.3. Etiology
Data concerning the etiology of liver disease in patients with HCC were described in 158 cases (Table 3). Hepatitis B virus (HBV) was incriminated in most patients (38.57%), followed by hepatitis C virus (HCV) (17.76%) and alcohol (7.61%). Coinfection with HBV and HCV was reported in 2.03% of cases, and only 0.5% of patients were identified with HBV, HCV, and hepatitis D virus (HVD) coinfection. Autoimmune etiology and non-alcoholic fatty liver disease (NAFLD) were suggested in one patient. In 19 cases, the underlying cause was unknown (9.64%).

Table 3.
Etiology of liver disease in patients with hepatocellular carcinoma.
3.4. Clinical Findings in the Study Population
A summary of the clinical characteristics of the study population is described in Table 4. Most HCCs were bulky masses, with an average tumor size of 8.66 cm (n = 92 hepatic nodules). Liver tumor localization was described in 158 patients. Four patients did not have any tumor recurrence at the moment of diagnosis. Hepatocellular carcinoma was located as follows: right hepatic lobe (31.47%), left hepatic lobe (21.82%), both hepatic lobes (19.28%), caudate lobe (2.03%), hepatic hilum (1.01%), peritoneum (1.01%), left hepatic lobe and caudate lobe (0.50%), right hepatic lobe and caudate lobe (0.50%) and lymph nodes (0.50%). Portal vein thrombosis was found in 27.91% (55/197) of the evaluated cases. Regarding the tumoral markers, the mean value of serum AFP was 15,366.18 ng/mL (n = 112 available data, 14 patients were reported as having a normal value).

Table 4.
Summary of clinical characteristics of study patients.
3.5. Previous Treatment for Hepatocellular Carcinoma
Information on prior therapy for HCC is supplied in detail below in Table 5. The percentage of patients who did not receive any specific therapy was 26.90% (53/197). A relatively high number of patients were treated with TACE (32.99%, 65/197) and surgical resection (28.93%; 57/197). Among locoregional therapies, TACE was followed by: transarterial embolization (TAE) (23/197; 11.67%), radiofrequency ablation (RFA) (10. 15%, 20/197), percutaneous ethanol injection (PEI) (14/197, 7.10%) and intra-arterial chemotherapy (4/197, 2.39%). Liver transplant was performed in 5.58% of included patients. Molecular targeted therapies and systemic chemotherapy were administered to 4.56% and 5.58% of the patients, respectively. Less commonly used treatment methods were yttrium-90 radioembolization, hepatic arterial ligation, cryoablation, immunotherapy, radiotherapy and ultrasound-guided percutaneous microwave ablation.

Table 5.
Previous treatment of HCC.
3.6. Involved GI Site and Presumed Mode of Involvement
The most commonly involved sites in the gastrointestinal tract were the stomach (55/197; 27.91%) and duodenum (55/197; 27.91%,), followed by colon (32/197, 16.24%), esophagus (18/197; 8.92%), jejunum and ileum (14/197; 9.13%,) and rectum (3/197; 1.52%). Synchronous localization occurred in 9.64% of patients. The sites with concomitant involvement were: stomach and esophagus, stomach and duodenum, stomach and colon, stomach and small bowel, duodenum and colon and rectum and colon. Our study suggested that in most cases, GI involvement occurred through direct invasion (87/197; 44.16%). HCC metastasized through the hematogeneous route in 31.97% of situations (63/197). Translymphatic dissemination was reported for 3.04% (6/197); meanwhile, peritoneal spreading was found in 3.55% of patients (7/197). We also detected three cases of iatrogenically induced metastases. Moreover, both direct invasion and hematogenous spread were considered in two patients with concomitant duodenal and colon involvement and esophagus and stomach involvement, respectively. Details of the segments of the GI tract involved and the routes of involvement are listed in Table 6.

Table 6.
Involved GI site and involvement route.
3.7. Clinical Presentation
GI bleeding was the most common clinical presentation (49.74%), followed by abdominal pain (26.90%), nausea and vomiting (14.72%), fecal occult blood+ (4.06%), palpable abdominal mass (3.55%) and anemia (5.07%). Among the included patients, only 2.03% of them were asymptomatic. Other less frequent clinical characteristics are summarized in Table 7.

Table 7.
Clinical features of included patients.
3.8. Diagnosis of GI Lesions
The most frequently used diagnostic tools were upper GI endoscopy (112/197; 56.86%) and CT (112/197; 56.86%), followed by colonoscopy (24/197; 12.18%), upper GI series (18/197; 9.13%) and endoscopic ultrasound (EUS) (9/197; 4.56%). Other less frequent diagnostic methods were: magnetic resonance imaging (MRI) (7/197; 3.55%),positron emission tomography-computed tomography (PET-CT) (6/197; 3.04%), double/single-balloon enteroscopy (5/197; 2.53%), superior mesenteric angiography (4/197; 2.03), lower GI series (3/197; 1.52%), angio-CT (3/197; 1.52%), capsule endoscopy (2/197; 1.01%), scintigraphy TC-99 pyridoxyl-5-methyltryptophan (PMT) (1/197; 0.50) and celiac angiography (1/97; 0.50%). In 15 patients, the diagnosis was made either intraoperatively (9/168, 4.56%) or at autopsy (6/197; 3.04%). The full palette of the combination of diagnostic tools used is summarized in Table 8.

Table 8.
Palette of diagnostic tools used for GI lesion.
Endoscopic features of GI lesions are listed in Table 2. GI endoscopic procedures were used for diagnosis in 86.78% of cases (169/197). Detailed descriptions of the endoscopic aspect of the GI lesions were related in 84.61% (143/169). In two cases there was no endoscopic evidence of GI lesions. The most common endoscopic findings were: exophytic mass (15.73%), polypoid lesions (14.72%), ulcerative lesions (14.21%), submucosal tumor (8.62%), ulcer (5.58%), fistula (2.53%) and extrinsic compression (1.52%). Non-specific aspects (ulcerations, erosions etc.) were seen in 9.64% of cases.
Specimens for pathological diagnosis (n = 159) were obtained through endoscopic biopsies (76/197; 38.57%), surgical intervention (61/197; 30.96%), endoscopic biopsies and resected specimens (10/197, 5.07%), endoscopic ultrasound-fine needle aspiration (1/197; 0.50%) and autopsy (11/197; 5.58%). The diagnosis was not confirmed through histopathology in 15.22% of cases (30/197). Immunohistochemistry techniques were used in 26.39% of patients (52/197).
3.9. Prognosis of Gastrointestinal Involvement in Patients with HCC
Prognosis of patients with hepatocellular carcinoma and GI tract involvement (n = 158 available data; from which 12 were lost to follow-up) was dismal, with an average survival of 7.30 months. In 3.04% of cases (6/197), the diagnosis was made post-mortem at autopsy, and 1.01% (2/197) survived for less than 24 h. In the present study, only 14.72% (29/197) were still alive at the moment of the last follow-up.
4. Discussion
Hepatocellular carcinoma yields high recurrence rates, even after radical resection. Liver transplantation is the best treatment method because it also cures the underlying liver disease, but it is not broadly applicable [132]. However, other available therapeutic approaches, such as locoregional therapies, have been developed with the purpose of increasing the survival rate in patients with unresectable HCC [133]. Improvements in the survival period are associated with a higher risk of developing extrahepatic metastases. Among them, gastrointestinal involvement from hepatocellular carcinoma is rare [118].
In our study, both stomach and duodenum were the areas of the GI tract most frequently affected. In reviews reported in literature, the stomach was most commonly involved, followed by duodenum [10]. On the other side, esophagus metastases were very uncommon in our analysis, accounting for less than 9% of cases. Sites of the lower part of the gastrointestinal tract, such as the colon, jejunum, ileum and rectum were also less affected. In exceptional cases, a liver tumor can simultaneously involve more than one segment of the GI tract [7,51,58,59,65,66,80].
As in most reports, direct invasion was the predominant spread pattern. Factors favoring GI involvement were growth mode, size and localization of hepatic tumors [14]. Due to the the anatomical relationship between the liver and GI tract segments, HCCs localized in the right hepatic lobe tend to invade the duodenum, and those located in the left lobe usually involve the stomach [84]. The role of TACE is controversial. TACE can induce tumor adherence to the liver capsule and GI tract through necrosis and inflammation. On the other hand, HCC was diagnosed concomitant with the GI invasion in many cases, and these patients had not received any previous treatment [14]. In our analysis, TACE was previously performed in 32.99% of cases, and 26.90% did not receive any treatment.
The hematogenous route was the second most frequent path. Although it can be detected in liver cirrhosis, reverse portal flow is more frequently observed in primary hepatocellular carcinoma due to arteriovenous communications and arterial neovascularization [134]. Tumor emboli can be disseminated from the liver to the gastrointestinal tract by the hepatofugal portal flow [131]. Portal vein thrombosis is also a significant contributing element that can exacerbate the reversal of the flow [119]. This aspect is supported by the fact that 27.91% of the assessed cases in our study presented portal vein thrombosis. It is hypothesized that endoscopic therapy of esophageal varices, in particular esophageal band ligation, can promote the development of esophageal metastasis by trapping the tumoral thrombi [31]. Hiraoka, T. et al. (1986) presented two cases of hepatocellular carcinoma with invasion of the portal vein branches, in which microscopic tumoral thrombi were found in sclerosed esophageal varices [20]. Kume, K. et al. also presented a case of HCC metastases developed at the place of variceal band ligation [21].
In our review of the literature, GI tract involvement in HCC was also reported to develop after liver transplant [25,28,29,32,41,55,121]. The risk of developing uncommon metastases of HCC, including gastrointestinal metastases, can increase after a liver transplant due to a delicate physiological state or to the administration of immunosuppressive agents [32].
In exceptional cases, dissemination of tumor cells through needle track following endoscopic ultrasound-fine needle aspiration (EUS-FNA) performed for the confirmation of HCC has also been described [61]. Moreover, periampullary metastasis from HCC has been reported after biliary interventions in a patient with HCC invaded in the biliary tract [84].
Several authors also reported GI tract involvement from peritoneal spreading or lymph node metastases [8,43,55,77,80,93,101,117].
Gastrointestinal bleeding, either frank or occult, was the most common presenting feature among the studied cases. Similarly to our results, in an analysis of 30 cases with direct GI involvement from HCC, reported in the English literature, Korkolis, D.P. et al. (2009) also concluded that gastrointestinal bleeding was the main clinical presentation [46]. Besides GI bleeding, the spectrum of clinical manifestations was vast in our study results. It included abdominal pain, palpable mass, chronic anemia, dysphagia, fatigue, weight loss, nausea, vomiting, diarrhea and gastric outlet obstruction.
Algorithm of Diagnosis
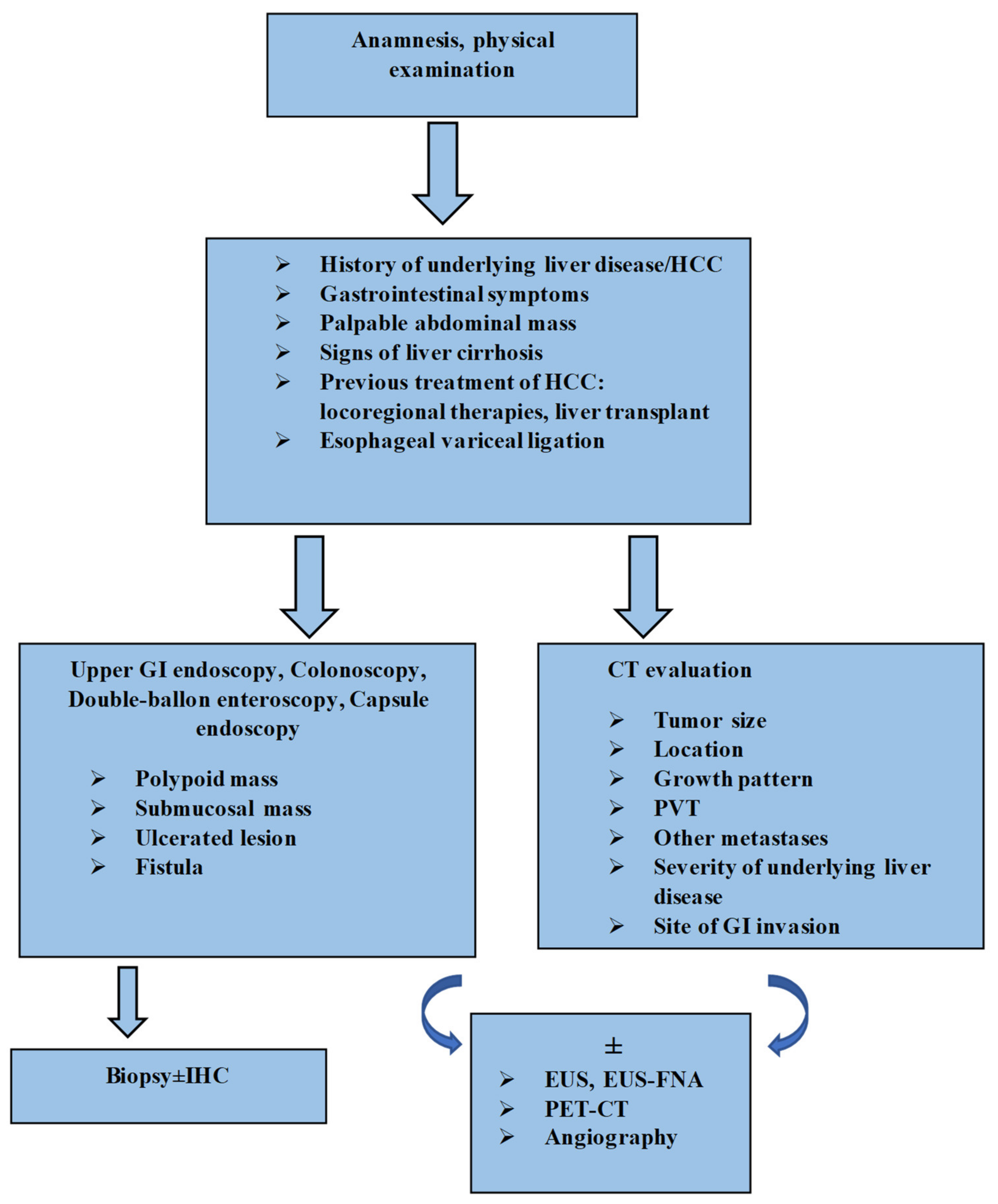
On the basis of data found in the literature and the results of our research, we propose a diagnostic algorithm (Figure 2).
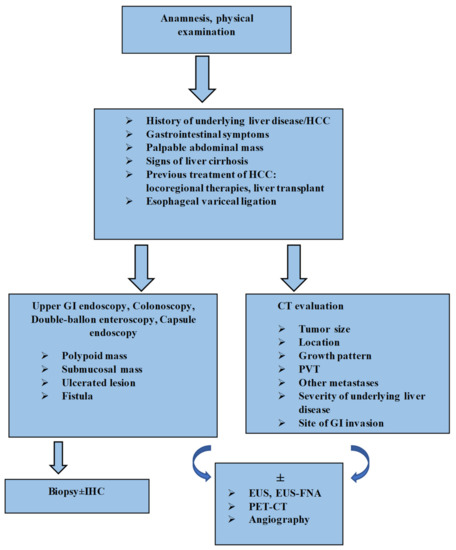
Figure 2.
Algorithm for diagnosis of GI tract involvement from HCC. HCC: hepatocellular carcinoma; GI: gastrointestinal; CT: computed tomography; PVT: portal vein thrombosis; IHC: immunohistochemistry; EUS: endoscopic ultrasound; EUS-FNA: endoscopic ultrasound-fine needle aspiration; PET-CT: positron emission tomography-computed tomography.
The diagnosis can be extremely challenging and clinicians must be aware of this condition, as early detection and prompt treatment are crucial for a better prognosis. In patients with a known clinical context of cirrhosis and hepatocellular carcinoma, with new-onset gastrointestinal symptoms, especially GI bleeding, gastrointestinal involvement from HCC should be considered as a possible etiology if other common causes are excluded [47,71]. Clinical examination plays a key role in identifying an abdominal mass [100]. In the next stage, risk-factor assessment should be conducted: tumor size, location, growth pattern, presence of portal vein thrombosis and previous locoregional therapies for the primary liver tumor, or even liver transplant and endoscopic therapy for esophageal varices [14,31,32,119]. Endoscopic examination is the standard gold method for identifying GI lesions [7]. As evidenced by the various aspects described, endoscopic features are not specific and can pose differential diagnosis problems. However, the following aspects should draw suspicion in a patient with HCC: polypoid mass, a submucosal tumor/extrinsic compression, ulcerative lesions or even the presence of a fistula [8,48,50,63,80]. The histopathologic examination is mandatory for a certain diagnosis. In some situations where there is uncertainty, investigations should include immunohistochemical tests [7,114,126]. Hepatocyte paraffin-1 (Hep par-1), glypican-3 (GPC-3), arginase-1 and polyclonal carcinoembryonicantigen (pCEA) effectively differentiate GI metastases of HCC from other types of tumors [126]. EUS and EUS-FNA are the alternative diagnostic methods for GI submucosal lesions or for when endoscopy fails to identify the tumor [33]. Radiological investigations are an excellent guidance modality. CT can describe the localization, size and extension of primary liver tumor, the status of the portal vein and lymph nodes, the site of invasion, the contiguity of HCC with the GI tract lesions and the severity of underlying liver cirrhosis, and can also exclude other metastases [8,118]. GI metastases of HCC usually display hyperenhancement in the arterial phase on CT scans, similarly to liver tumors [8]. FDG-PET/CT and angiography can complete the diagnostic workup [6,7,29,32,33,71].
Based on our reviewed articles, the mean survival was 7.30 months. Fujii, K. et al. (2004) evaluated median survival in 29 patients with HCC invading the GI tract. The estimated median survival time was 1.2 months for patients who received supportive treatment, and three months for nonsurgical treatment; meanwhile, patients treated with curative surgery showed an average survival of 9.7 months [9].
Although it may improve the clinical approach of cases with HCC and GI involvement, our study has a number of limitations. Subjectivity in data interpretation, lack of a large control group and the fact that only English-language articles were included are just some of these limitations, and may have influenced the final results.
5. Conclusions
In conclusion, to our knowledge, we are reporting the most extensive systematic review of case reports to date on the involvement of the gastrointestinal tract in HCC. Gastrointestinal involvement in HCC could be included in the differential diagnosis of patients with underlying HCC and gastrointestinal manifestations or pathological findings in EGD.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/diagnostics12051270/s1. Table S1: PRISMA 2020 checklist. Table S2: Complete search strategy. Table S3: Methodological quality of included case reports using JBI Critical Appraisal Checklist for case reports. Table S4: Methodological quality of included case reports using JBI Critical Appraisal Checklist for case series. Table S5: Relevant studies excluded from the systematic review.
Author Contributions
Conceptualization, C.M.U., L.D.S. and S.D.; methodology, C.M.U., L.D.S. and S.D.; formal analysis, C.M.U., L.D.S. and S.M.S.; investigation, C.M.U., L.S. and S.M.S.; data curation, C.M.U., L.S. and V.F.I.; writing—original draft preparation, C.M.U., L.S., V.F.I. and S.M.S.; writing—review and editing L.D.S. and L.S.; visualization, C.M.U.; supervision, L.D.S. and S.D. All authors have read and agreed to the published version of the manuscript.
Funding
The article processing charges were funded by the Doctoral School of the University of Medicine and Pharmacy of Craiova, Romania.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data supporting the findings of this study are available within the article and its Supplementary Materials.
Acknowledgments
This work was supported by a grant from the Romanian Ministry of Education and Research, CNCS-UEFISCDI, project number PN-III-P1-1.1-TE-2019-1474, within PNCDI III.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Terada, T.; Maruo, H. Unusual extrahepatic metastatic sites from hepatocellular carcinoma. Int. J. Clin. Exp. Pathol. 2013, 6, 816–820. [Google Scholar] [PubMed]
- Sawabe, M.; Nakamura, T.; Kanno, J.; Kasuga, T. Analysis of morphological factors of hepatocellular carcinoma in 98 autopsy cases with respect to pulmonary metastasis. Pathol. Int. 1987, 37, 1389–1404. [Google Scholar] [CrossRef] [PubMed]
- Katyal, S.; Oliver, J.H., 3rd; Peterson, M.S.; Ferris, J.V.; Carr, B.S.; Baron, R.L. Extrahepatic metastases of hepatocellular carcinoma. Radiology 2000, 216, 698–703. [Google Scholar] [CrossRef]
- Natsuizaka, M.; Omura, T.; Akaike, T.; Kuwata, Y.; Yamazaki, K.; Sato, T.; Karino, Y.; Toyota, J.; Suga, T.; Asaka, M. Clinical features of hepatocellular carcinoma with extrahepatic metastases. J. Gastroenterol. Hepatol. 2005, 20, 1781–1787. [Google Scholar] [CrossRef]
- Chen, L.-T.; Chen, C.-Y.; Jan, C.-M.; Wang, W.-M.; Lan, T.-S.; Hsieh, M.-Y.; Liu, G.-C. Gastrointestinal Tract Involvement in Hepatocellular Carcinoma: Clinical, Radiological and Endoscopic Studies. Endoscopy 1990, 22, 118–123. [Google Scholar] [CrossRef]
- Lin, C.; Cheng, J.; Lai, K.; Lo, G.; Hsu, P.; Chan, H.; Hsu, J.; Wang, Y.; Pan, H.; Tseng, H. Gastrointestinal metastasis in hepatocellular carcinoma: Radiological and endoscopic studies of 11 cases. J. Gastroenterol. Hepatol. 2000, 15, 536–541. [Google Scholar] [CrossRef]
- Park, M.-S.; Kim, K.W.; Yu, J.-S.; Kim, M.-J.; Yoon, S.-W.; Chung, K.-W.; Lee, J.T.; Yoo, H.S. Radiologic Findings of Gastrointestinal Tract Involvement in Hepatocellular Carcinoma. J. Comput. Assist. Tomogr. 2002, 26, 95–101. [Google Scholar] [CrossRef]
- Fujii, K.; Nagino, M.; Kamiya, J.; Uesaka, K.; Sano, T.; Yuasa, N.; Oda, K.; Nimura, Y. Complete resection of hepatocellular carcinoma with direct invasion to the stomach remnant. J. Hepatobiliary Pancreat. Surg. 2004, 11, 441–444. [Google Scholar] [CrossRef]
- Lin, T.-L.; Yap, A.Q.; Wang, J.-H.; Chen, C.-L.; Iyer, S.G.; Low, J.-K.; Lin, C.-C.; Li, W.-F.; Chen, T.-Y.; Bora, D.; et al. Long term survival in patients with hepatocellular carcinoma directly invading the gastrointestinal tract: Case reports and literature review. Surg. Oncol. 2011, 20, e207–e214. [Google Scholar] [CrossRef]
- Kato, Y.; Matsubara, K.; Akiyama, Y.; Hattori, H.; Hirata, A.; Yamamoto, T.; Suzuki, F.; Ohtaka, H.; Sugiura, Y.; Kitajima, M. Direct biliopancreatoduodenal invasion by hepatocellular carcinoma: Report of the first resected case and review of the literature. Int. J. Clin. Oncol. 2010, 16, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Harada, J.-I.; Matsutani, T.; Hagiwara, N.; Kawano, Y.; Matsuda, A.; Taniai, N.; Nomura, T.; Uchida, E. Metastasis of Hepatocellular Carcinoma to the Esophagus: Case Report and Review. Case Rep. Surg. 2018, 2018, 8685371. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.-M.; Cao, Y.-S.; Wu, Z.; Huang, R.; Shen, Z.-L. Colon metastasis from hepatocellular carcinoma: A case report and literature review. World J. Surg. Oncol. 2020, 18, 189. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Mu, M.; Liang, P.; Yu, X.; Cheng, Z. Colonic metastasis from hepatocellular carcinoma after treated by ablation and transarterial chemoembolization manifested by intestinal obstruction: A case report and review of the literature. J. Cancer Res. Ther. 2021, 17, 814–817. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Wen, Z. Survival improvement and prognosis for hepatocellular carcinoma: Analysis of the SEER database. BMC Cancer 2021, 21, 1157. [Google Scholar] [CrossRef]
- Hashimoto, M.; Watanabe, G.; Matsuda, M.; Yamamoto, T.; Tsutsumi, K.; Tsurumaru, M. Case Report: Gastrointestinal bleeding from a hepatocellular carcinoma invading the transverse colon. J. Gastroenterol. Hepatol. 1996, 11, 765–767. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 88, 105906. [Google Scholar] [CrossRef]
- Moola, S.; Munn, Z.; Tufanaru, C.; Aromataris, E.; Sears, K.; Sfetc, R.; Currie, M.; Lisy, K.; Qureshi, R.; Mattis, P.; et al. Chapter 7: Systematic reviews of etiology and risk. In JBI Manual for Evidence Synthesis; Aromataris, E., Munn, Z., Eds.; JBI: Adelaide, Australia, 2020; Available online: https://synthesismanual.jbi.global (accessed on 22 December 2021).
- Sohn, D.; Valensi, Q.; Bryk, D. Hepatoma metastasizing to the esophagus. JAMA 1965, 194, 910–912. [Google Scholar] [CrossRef]
- Hiraoka, T.; Iwai, K.; Yamashita, R.; Tada, I.; Miyauchi, Y. Metastases from hepatocellular carcinoma in sclerosed oesophageal varices in cirrhotic patients. Br. J. Surg. 1986, 73, 932. [Google Scholar] [CrossRef]
- Kume, K.; Murata, I.; Yoshikawa, I.; Kanagawa, K.; Otsuki, M. Polypoid Metastatic Hepatocellular Carcinoma of the Esophagus Occurring After Endoscopic Variceal Band Ligation. Endoscopy 2000, 32, 419–421. [Google Scholar] [CrossRef]
- Sohara, N.; Takagi, H.; Yamada, T.; Ichikawa, T.; Abe, T.; Itoh, H.; Mori, M. Esophageal metastasis of hepatocellular carcinoma. Gastrointest. Endosc. 2000, 51, 739–741. [Google Scholar] [CrossRef] [PubMed]
- Tsubouchi, E.; Hirasaki, S.; Kataoka, J.; Hidaka, S.; Kajiwara, T.; Yamauchi, Y.; Masumoto, T.; Hyodo, I. Unusual Metastasis of Hepatocellular Carcinoma to the Esophagus. Intern. Med. 2005, 44, 444–447. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.-L.; Hung, Y.-H.; Yang, T.-H. Metastatic hepatocellular carcinoma of the esophagus: An unusual cause of upper gastrointestinal bleeding. Endoscopy 2007, 39, E257–E258. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xie, L.-Y.; Fan, M.; Fan, J.; Wang, J.; Xu, X.-L.; Jiang, G.-L. Metastatic hepatocellular carcinoma in the esophagus following liver transplantation. Liver Transplant. 2008, 14, 1680–1682. [Google Scholar] [CrossRef]
- Choi, C.S.; Kim, H.C.; Kim, T.H.; Seo, G.S.; Kim, K.H.; Cho, E.Y.; Seo, S.O.; Oh, H.J.; Choi, S.C. Does the endoscopic finding of esophageal metastatic hepatocellular carcinoma progress from submucosal mass to polypoid shape? Gastrointest. Endosc. 2008, 68, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Hsu, K.-F.; Hsieh, T.-Y.; Yeh, C.-L.; Shih, M.-L.; Hsieh, C.-B. Polypoid esophageal and gastric metastases of recurrent hepatocellular carcinoma after liver transplantation. Endoscopy 2009, 41, E82–E83. [Google Scholar] [CrossRef][Green Version]
- Kahn, J.; Kniepeiss, D.; Langner, C.; Wagner, D.; Iberer, F.; Tscheliessnigg, K. Oesophageal metastases of hepatocellular carcinoma after liver transplantation. Transpl. Int. 2010, 23, 438–439. [Google Scholar] [CrossRef]
- Boonnuch, W.; Akaraviputh, T.; Nino, C.; Yiengpruksawan, A.; Christiano, A.A. Successful treatment of esophageal metastasis from hepatocellular carcinoma using the da Vinci robotic surgical system. World J. Gastrointest. Surg. 2011, 3, 82–85. [Google Scholar] [CrossRef]
- Škurla, B.; Mlinarić, A.; Nadalin, S.; Katičić, M.; Naumovski-Mihalić, S. Esophageal metastases of hepatocellular carcinoma following liver transplantation. Int. J. Case Rep. Images 2010, 1, 7–11. [Google Scholar] [CrossRef]
- Fukatsu, H.; Miura, S.; Kishida, H.; Takagi, S.; Morishita, H.; Uchino, K.; Fujisawa, M. Gastrointestinal: Esophageal metastasis from hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2012, 27, 1536. [Google Scholar] [CrossRef]
- Chen, J.-X.; Jeng, L.-B.; Lin, Y.-S.; Lu, T.-Y.; Kao, P.-Y.; Fang, H.-Y. A Mimicking Esophageal Cancer After Liver Transplant for Hepatocellular Carcinoma: A Rare Posttransplant Metastasis. Exp. Clin. Transplant. 2016, 14, 571–574. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kongkam, P. Esophageal Metastatic Sub-mucosal Lesion of Hepatocellular Carcinoma Diagnosed by EUS. Gastroenterol. Hepatol. Open Access 2018, 9, 282. [Google Scholar] [CrossRef][Green Version]
- Bonboire, R.; Miscu, C.; Yengue, P.; Maes, B.; Patris, A.; Gallez, J.-F. Gastrointestinal tract involvement in hepatocellular carcinoma: Two cases illustrating duodenal and oesophageal invasion. Acta Gastro Enterol. Belg. 2021, 84, 656–658. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, S.K.; Bhakta, D.; Thosani, N.; Ertan, A. Hepatocellular carcinoma masquerading as nodular Barrett’s esophagus. Gastrointest. Endosc. 2021, 93, 1182–1183. [Google Scholar] [CrossRef]
- Shiota, T.; Watanabe, A.; Mitani, K.; Ito, T.; Tobe, K.; Nagashima, H. Long-term survival in a case of hepatocellular carcinoma. Acta Med. Okayama 1983, 37, 73–78. [Google Scholar]
- Makino, H.; Takazakura, E.; Nakamura, S.; Kobayashi, K.; Hattori, N.; Nonomura, A.; Ohta, G. Hepatocellular carcinoma with metastatic gastric cancer simulating Borrmann type 2 and hyper-lipidemia. Acta Pathol. Jpn. 1986, 36, 577–586. [Google Scholar]
- De Nardi, P.; Braga, M.; Zerbi, A.; Sassi, I.; Mangili, F.; Cristallo, M.; Di Carlo, V. Bleeding Gastric Polyposis-Like Metastases of Hepatocellular Carcinoma. Dig. Surg. 1992, 9, 105–108. [Google Scholar] [CrossRef]
- Nicoll, A.J.; Ireton, H.J.C.; Crotty, B. Gastrointestinal bleeding from hepatocellular carcinoma invading the stomach. J. Gastroenterol. Hepatol. 1994, 9, 533–535. [Google Scholar] [CrossRef]
- Maruyama, A.; Murabayashi, K.; Hayashi, M.; Nakano, H.; Isaji, S.; Uehara, S.; Kusuda, T.; Miyahara, S.; Kondo, A.; Nakano, H.; et al. Hepatocellular carcinoma complicated by gastrointestinal hemorrhage caused by direct tumor invasion of stomach. J. Hepato-Biliary Pancreat. Surg. 1999, 6, 90–93. [Google Scholar] [CrossRef]
- Srivastava, D.N.; Gandhi, D.; Julka, P.K.; Tandon, R.K. Gastrointestinal hemorrhage in hepatocellular carcinoma: Management with transhepatic arterioembolization. Abdom. Imaging 2000, 25, 380–384. [Google Scholar] [CrossRef]
- Wang, M.-H.; Wu, M.-S.; Wang, H.-P.; Change, Y.-L.; Lin, J.-T. Endoscopic manifestations of metastatic hepatocellular carcinoma to the stomach: Report of two cases. Gastrointest. Endosc. 2000, 52, 273–275. [Google Scholar] [CrossRef] [PubMed]
- Inoue, H.; Sawada, Y.; Ochiai, K.; Honda, H.; Murayama, J.; Kudo, Y.; Nakashima, Y.; Sagihara, N.; Miyatani, H.; Nakamura, I.; et al. Hepatocellular Carcinoma with Direct Invasion to the Stomach. Intern. Med. 2007, 46, 845–848. [Google Scholar] [CrossRef] [PubMed]
- Ong, J.C.; Chow, P.K.; Chan, W.-H.; Chung, A.Y.; Thng, C.H.; Wong, W.-K. Hepatocellular carcinoma masquerading as a bleeding gastric ulcer: A case report and a review of the surgical management. World J. Gastroenterol. 2007, 13, 4523–4525. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Miura, H.; Saito, S.; Suzaki, A.; Kondo, K.; Hamada, T.; Kitamura, S.; Yamada, H. Hepatocellular Carcinoma with Direct Invasion to the Stomach Causing Gastrointestinal Hemorrhage. Intern. Med. 2008, 47, 671–674. [Google Scholar] [CrossRef] [PubMed]
- Korkolis, D.P.; Aggeli, C.; Plataniotis, G.D.; Gontikakis, E.; Zerbinis, H.; Papantoniou, N.; Xinopoulos, D.; Apostolikas, N.; Vassilopoulos, P.P. Successful en bloc resection of primary hepatocellular carcinoma directly invading the stomach and pancreas. World J. Gastroenterol. 2009, 15, 1134–1137. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.-L.; Tai, W.-C.; Chuah, S.-K.; Chiu, Y.-C.; Wu, K.-L.; Chou, Y.-P.; Kuo, C.-M.; Hu, T.-H.; Chiu, K.-W. Gastric metastasis of hepatocellular carcinoma via a possible existing retrograde hematogenous pathway. J. Gastroenterol. Hepatol. 2010, 25, 408–412. [Google Scholar] [CrossRef]
- Park, H.; Kim, S.U.; Choi, J.; Park, J.Y.; Ahn, S.H.; Han, K.-H.; Chon, C.Y.; Park, Y.N.; Kim, Y. Hepatogastric fistula caused by direct invasion of hepatocellular carcinoma after transarterial chemoembolization and radiotherapy. Korean J. Hepatol. 2010, 16, 401–404. [Google Scholar] [CrossRef]
- Tan, W.J.; Chia, C.S.; Ong, H.S. A rare cause of gastrointestinal haemorrhage: Gastric invasion by hepatocellular carcinoma. J. Surg. Case Rep. 2013, 2013, rjs050. [Google Scholar] [CrossRef]
- Sayana, H.; Yousef, O.; Clarkston, W.K. Massive upper gastrointestinal hemorrhage due to invasive hepatocellular carcinoma and hepato-gastric fistula. World J. Gastroenterol. 2013, 19, 7472–7475. [Google Scholar] [CrossRef]
- Okay, E.; Sarı, A.; Odabasioglu, H.; Uygun, K.; Sarisoy, T.; Gürbüz, Y.; Sari, A.; Uygun, K. Hepatocellular carcinoma presenting as a huge intra-abdominal mass: A case report. Turk. J. Gastroenterol. 2014, 25, 330–332. [Google Scholar] [CrossRef]
- Inagaki, Y.; Shiraki, K.; Takei, Y. Gastric Metastasis of Hepatocellular Carcinoma Presenting as Hematemesis. Clin. Gastroenterol. Hepatol. 2014, 12, A16–A17. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.-D.; Wu, J.; Yang, H.-G.; Chen, Y.; Zhang, C.-W.; Zhao, D.-J.; Hu, Z.-M. Rare cause of upper gastrointestinal bleeding owing to hepatic cancer invasion: A case report. World J. Gastroenterol. 2014, 20, 12704–12708. [Google Scholar] [CrossRef] [PubMed]
- Grover, I.; Ahmad, N.; Googe, A.B. Hepatogastric fistula following transcatheter arterial chemoembolization of hepatocellular carcino-ma. Case Rep. Gastroenterol. 2014, 8, 286–290. [Google Scholar] [CrossRef]
- Li, L.; Zhang, W.; Meng, F.; Ma, X.; Shen, L.; Jin, B.; Li, H.; Han, J.; Zhou, G.; Liu, S. Gastric Metastasis of Hepatocellular Carcinoma with Gastrointestinal Bleeding After Liver Transplant: A Case Report. Transplant. Proc. 2015, 47, 2544–2547. [Google Scholar] [CrossRef] [PubMed]
- Hot, S.; Yeşiltaş, M.; Gökçek, B.; Eğin, S.; Şengiz, S. Massive and life-threatening upper gastrointestinal bleeding due to invasive hepatocellular carcinoma: A case report. Int. J. Surg. Case Rep. 2016, 26, 69–72. [Google Scholar] [CrossRef][Green Version]
- Haruki, K.; Misawa, T.; Gocho, T.; Saito, R.; Shiba, H.; Akiba, T.; Yanaga, K. Hepatocellular carcinoma with gastric metastasis treated by simultaneous hepatic and gastric resection: Report of a case. Clin. J. Gastroenterol. 2016, 9, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Wei, S.; Liu, B.; Wu, X.; Feng, Y.; Luo, C.; Ju, Y.; Liang, J. Effect of immune suppression on metastasis in a patient with hepatocellular carcinoma metastasized to the colon and stomach: A case report. Exp. Ther. Med. 2016, 11, 1741–1747. [Google Scholar] [CrossRef]
- Hakim, M.S.A.; Azmi, A.N.; Jayalakshmi, P.; Mahadeva, S. Gastric Metastasis from Hepatocellular Carcinoma: A Rare Manifestation. J. Gastrointest. Cancer 2017, 49, 346–348. [Google Scholar] [CrossRef]
- Xiao, W.; Peng, L.; Yu, K.; Li, Y. Gastric metastasis of recurrent hepatocellular carcinoma: A case report and literature review. J. Cancer Res. Ther. 2018, 14, S1230–S1232. [Google Scholar] [CrossRef] [PubMed]
- Kasi, M.; Rashid, S.; Wallace, S.A.J.; Sujendran, V.; Griffiths, B.; Butler, A.; Gibbs, P.; Sreedharan, L.; Zaitoun, A.M.; Venkatachalapathy, S.; et al. Seeding of hepatocellular carcinoma into the stomach wall following endoscopic ultrasound and fine-needle aspiration biopsy. Oxf. Med. Case Rep. 2018, 2018, omy039. [Google Scholar] [CrossRef]
- Sakumura, M.; Tajiri, K.; Sugiyama, T. Gastric Metastasis of Hepatocellular Carcinoma Mimicking Early Gastric Cancer. Clin. Gastroenterol. Hepatol. 2018, 16, e99–e100. [Google Scholar] [CrossRef] [PubMed]
- Bale, A.; Shetty, S.; Shetty, A.; Balaraju, G.; Pai, C.G. Upper Gastrointestinal Bleed Due to Invasive Hepatocellular Carcinoma and Hepato-Gastric Fistula. J. Clin. Exp. Hepatol. 2018, 8, 104–105. [Google Scholar] [CrossRef]
- Imai, M.; Ishikawa, T.; Okoshi, M.; Tomiyoshi, K.; Kojima, Y.; Horigome, R.; Nozawa, Y.; Sano, T.; Iwanaga, A.; Honma, T.; et al. Hemorrhagic Gastric Metastasis from Hepatocellular Carcinoma Successfully Treated Using Coil Embolization of the Left Gastric Artery. Intern. Med. 2019, 58, 2179–2183. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, P.M.; Luis, R.; Lopes, J. Hepatocellular Carcinoma Invading the Gastric Antrum. Clin. Gastroenterol. Hepatol. 2018, 17, e102–e103. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.; Song, J.; Kim, S.B. Concurrent hepatocellular carcinoma metastasis to stomach, colon, and brain: A case report. World J. Clin. Cases 2020, 8, 3534–3541. [Google Scholar] [CrossRef] [PubMed]
- Abouzied, M.M.; Fathala, A.; AlMuhaideb, A.; Almanea, H.; Al-Sugair, A.S.; AlSkaff, R.; Al-Qahtani, M.H. Gastric wall metastases from hepatocellular carcinoma: Case report and review of the literature. Radiol. Case Rep. 2020, 16, 550–554. [Google Scholar] [CrossRef]
- Eskarous, H.; Patel, D.; Geme, B.; Krishnamurthy, M. Hepatocellular Carcinoma with Gastric Metastasis. ACG Case Rep. J. 2022, 9, e00750. [Google Scholar] [CrossRef]
- Arima, K.; Suga, M.; Ikeda, N.; Takahashi, T.; Nakata, M.; Shibata, K.; Kobayashi, T.; Yabana, T.; Yachi, A.; Wakabayashi, J. Hepatocellular Carcinoma with Metastasis to the Duodenum: A Case Report. Dig. Endosc. 1992, 4, 62–67. [Google Scholar] [CrossRef]
- Moriura, S.; Ikeda, S.; Ikezawa, T.; Naiki, K.; Sakai, T.; Yokochi, K.; Kuroda, M. Hepatocellular Carcinoma Invading the Duodenum: Combined Resection of the Lateral Duodenum and Repair with a Pedicled Gastric Flap. Dig. Surg. 1995, 12, 136–139. [Google Scholar] [CrossRef]
- Okusaka, T.; Okada, S.; Ishii, H.; Nagahama, H.; Yoshimori, M.; Yamasaki, S.; Takayasu, K.; Kakizoe, T.; Ochiai, A.; Shimoda, T. Hepatocellular Carcinoma With Gastrointestional Hemorrhage Caused by Direct Tumor Invasion to the Duodenum. Jpn. J. Clin. Oncol. 1997, 27, 343–345. [Google Scholar] [CrossRef][Green Version]
- Hung, H.-C.; Huang, Y.-S.; Lin, C.-C.; Chao, Y.; Chi, K.-H.; Yen, S.-H.; Chang, F.-Y. CASE REPORT: Radiotherapy in the treatment of duodenal bleeding due to hepatocellular carcinoma invasion. J. Gastroenterol. Hepatol. 1998, 13, 1143–1145. [Google Scholar] [CrossRef] [PubMed]
- Farrell, R.; Morrin, M.; Lewis, D.; Chuttani, R.; Pleskow, D. EUS features of duodenal invasion secondary to hepatocellular carcinoma: Case report. Gastrointest. Endosc. 1999, 49, 384–387. [Google Scholar] [CrossRef]
- Del Natale, M.; Druez, P.; Brenard, R. Fistulization of a hepatocellular carcinoma into the duodenum. J. Hepatol. 2001, 34, 172. [Google Scholar] [CrossRef]
- Cho, A.; Ryu, M.; Ochiai, T. Successful resection, using pancreas-sparing duodenectomy, of extrahepatically growing hepatocellular carcinoma associated with direct duodenal invasion. J. Hepato-Biliary-Pancreat. Surg. 2002, 9, 393–396. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, S.; Hatanaka, K.; Nakanishi, M.; Hige, S.; Asaka, M. Massive Invasion of Hepatocellular Carcinoma to the Duodenum: Successful Treatment of Gastrointestinal Haemorrhage with Transcatheter Arterial Embolisation. Clin. Oncol. 2004, 16, 157. [Google Scholar] [CrossRef] [PubMed]
- Uehara, K.; Hasegawa, H.; Ogiso, S.; Sakamoto, E.; Ohira, S.; Igami, T.; Mori, T. Skip lymph node metastases from a small hepatocellular carcinoma with difficulty in preoperative diagnosis. J. Gastroenterol. Hepatol. 2003, 18, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.; Al Ali, J.; Owen, D.A.; Weiss, A.A.; Yoshida, E.M.; Tai, I.T. A rare case of isolated duodenal metastases from hepatocellular carcinoma associated with p53 and ki-67 expression: A case report. Cases J. 2009, 2, 9344. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, L.E.; Miah, A.R. Infiltrating hepatocellular carcinoma into the duodenum presenting with upper gastrointestinal bleeding. Endoscopy 2009, 41, E308. [Google Scholar] [CrossRef]
- Liang, J.-D.; Chen, C.-H.; Hsu, S.-J.; Sheu, J.-C.; Yang, P.-M.; Lee, H.-S.; Lee, C.-Z.; Huang, G.-T. Hepatocellular carcinoma with duodenal invasion and metastasis. J. Gastroenterol. Hepatol. 2011, 27, 677–683. [Google Scholar] [CrossRef]
- Kim, J.N.; Lee, H.S.; Kim, S.Y.; Kim, J.H.; Jung, S.W.; Koo, J.S.; Yim, H.J.; Lee, S.W.; Choi, J.H.; Kim, C.D.; et al. Endoscopic Treatment of Duodenal Bleeding Caused by Direct Hepatocellular Carcinoma Invasion with an Ethanol Injection. Gut Liver 2012, 6, 122–125. [Google Scholar] [CrossRef]
- Sauer, B.G.; Dustin, S.M.; Caldwell, S.H. Liver Invasion of the Duodenum Due to Hepatocellular Carcinoma. Clin. Gastroenterol. Hepatol. 2012, 10, A25. [Google Scholar] [CrossRef] [PubMed]
- Arima, K.; Chikamoto, A.; Hashimoto, D.; Kaida, T.; Higashi, T.; Taki, K.; Takeyama, H.; Okabe, H.; Nitta, H.; Hayashi, H.; et al. Hepatology: Hepatocellular carcinoma with duodenal metastasis. J. Gastroenterol. Hepatol. 2015, 30, 1115. [Google Scholar] [CrossRef] [PubMed]
- Kashani, A.; Nissen, N.N.; Guindi, M.; Jamil, L.H. Metastatic Periampullary Tumor from Hepatocellular Carcinoma Presenting as Gastrointestinal Bleeding. Case Rep. Gastrointest. Med. 2015, 2015, 732140. [Google Scholar] [CrossRef] [PubMed]
- Lin, I.-C.; Tai, C.-M.; Tseng, C.-M. Duodenal invasion of hepatocellular carcinoma following transarterial chemoembolization. Dig. Liver Dis. 2018, 50, 618. [Google Scholar] [CrossRef]
- Ito, T.; Hirose, T.; Matsumoto, A.; Yogo, A.; Okuno, T.; Doi, R. Hepatocellular carcinoma with duodenal invasion resected subsequent to multimodal therapies: A case report. Int. J. Surg. Case Rep. 2019, 60, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.H.; Lo, E.Y.J.; Lee, K.F.; Chong, C.C.N.; Lai, P.B.-S. An unusual cause of upper gastrointestinal bleeding due to recurrent hepatocellular carcinoma: A case report. Int. J. Surg. Case Rep. 2020, 70, 83–86. [Google Scholar] [CrossRef]
- Wu, Y.-H.; Hsu, W.-F.; Yang, C.-Y.; Chang, K.-P. Gastrointestinal bleeding caused by hepatocellular carcinoma in a rare case of direct invasion to the duodenum. Dig. Liver Dis. 2020, 53, 1201–1202. [Google Scholar] [CrossRef]
- Sawada, K.; Fujiya, M.; Nakajima, S.; Okumura, T. Endoscopic findings of hepatocellular carcinoma invading the duodenum. Jpn. J. Clin. Oncol. 2021, 51, 1689–1690. [Google Scholar] [CrossRef]
- Lee, Y.J.; Kim, J.H.; Song, H.-Y.; Park, J.-H.; Na, H.K.; Kim, P.H.; Fan, Y. Hepatocellular Carcinoma Complicated by Gastroduodenal Obstruction: Palliative Treatment with Metallic Stent Placement. Cardiovasc. Interv. Radiol. 2011, 35, 1129–1135. [Google Scholar] [CrossRef]
- Tsujimoto, M.; Aozasa, K.; Nakajima, Y.; Kariya, M. Hepatocellular carcinoma with sarcomatous proliferation showing an unusual and wide-spread metastasis. Pathol. Int. 1984, 34, 839–845. [Google Scholar] [CrossRef]
- Narita, T.; Nakazawa, H.; Hizawa, Y.; Ishidoya, T.; Kudo, H. Hepatocellular carcinoma with unusual metastasis to the small intestine. Pathol. Int. 1993, 43, 779–782. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Takeda, R.; Yamamoto, H.; Utsunomiya, H.; Okamura, R.; Kataoka, M.; Mukaihara, S.; Yamaoka, Y. Extrahepatic large hepatocellular carcinoma with peritoneal dissemination: Multimodal treatment, including four surgical operations. J. Hepato-Biliary-Pancreatic Surg. 2000, 7, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Byun, J.R.; Cho, S.H.; Yang, D.H.; Kim, Y.K.; Ju, J.K.; Choi, S.K.; Chung, I.J. Ileovesical Fistula Caused by Hepatocellular Carcinoma. Korean J. Intern. Med. 2005, 20, 76–79. [Google Scholar] [CrossRef]
- Kim, H.S.; Shin, J.W.; Kim, G.Y.; Kim, Y.M.; Cha, H.J.; Jeong, Y.K.; Du Jeong, I.; Bang, S.-J.; Kim, D.H.; Park, N.H. Metastasis of hepatocellular carcinoma to the small bowel manifested by intussusception. World J. Gastroenterol. 2006, 12, 1969–1971. [Google Scholar] [CrossRef]
- Iwaki, K.; Ohta, M.; Ishio, T.; Kai, S.; Iwashita, Y.; Shibata, K.; Himeno, K.; Seike, M.; Fujioka, T.; Kitano, S. Metastasis of hepatocellular carcinoma to spleen and small intestine. J. Hepato-Biliary-Pancreatic Surg. 2008, 15, 213–219. [Google Scholar] [CrossRef]
- Choi, J.-H.; Kim, T.H.; Choi, K.H.; Oh, H.J. Small intestinal perforation caused by metastatic tumor necrosis after sorafenib (Nexavar®) therapy for advanced hepatocellular carcinoma. Int. Cancer Conf. J. 2012, 1, 155–158. [Google Scholar] [CrossRef]
- Kunizaki, M.; Hidaka, S.; Isomoto, H.; Takeshita, H.; Nanashima, A.; Sawai, T.; Yasutake, T.; Nagayasu, T. Diagnosis of small-bowel metastasis of hepatocellular carcinoma by double-balloon enteroscopy. Int. J. Surg. Case Rep. 2012, 3, 263–265. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Igawa, A.; Oka, S.; Tanaka, S.; Nakano, M.; Aoyama, T.; Watari, I.; Aikata, H.; Arihiro, K.; Chayama, K. Small Bowel Metastasis of Hepatocellular Carcinoma Detected by Capsule Endoscopy. Case Rep. Gastroenterol. 2013, 7, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, M.; Sugaya, T.; Tominaga, K.; Takahashi, F.; Takenaka, K.; Nakano, M.; Hiraishi, H. Jejunal Metastasis from Hepatocellular Carcinoma. Intern Med. 2018, 57, 947–950. [Google Scholar] [CrossRef]
- Shelat, V.G.; Giron, D.M.; Oo, A.M. Acute haemoperitoneum due to small bowel metastases from hepatocellular carcinoma in a long term survivor following previous rupture. Ann. Hepato-Biliary Pancreat. Surg. 2018, 22, 416–418. [Google Scholar] [CrossRef][Green Version]
- Sun, W.-C.; Tsai, T.-J.; Tsai, W.-L.; Cheng, J.-S.; Chen, W.-C. Metastatic hepatocellular carcinoma of small bowel presenting as GI bleeding. Gastrointest. Endosc. 2018, 88, 774–775. [Google Scholar] [CrossRef] [PubMed]
- Mashiko, T.; Masuoka, Y.; Nakano, A.; Tsuruya, K.; Hirose, S.; Hirabayashi, K.; Kagawa, T.; Nakagohri, T. Intussusception due to hematogenous metastasis of hepatocellular carcinoma to the small intestine: A case report. World J. Gastroenterol. 2020, 26, 6698–6705. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Tajiri, K.; Futsukaichi, Y.; Tanaka, S.; Murayama, A.; Entani, T.; Kobayashi, S.; Takahashi, K.; Fujii, T.; Imura, J.; et al. Perforation of the Small Intestine after Introduction of Lenvatinib in a Patient with Advanced Hepatocellular Carcinoma. Case Rep. Gastroenterol. 2020, 14, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Fukui, H.; Kashiwagi, T.; Shirai, Y.; Matsuda, Y.; Kawata, S.; Nishimura, T.; Kozuka, T. Metastasis of Hepatocellular Carcinoma to the Colon Demonstrated by Tc-99m PMT Scintigraphy. Clin. Nucl. Med. 1993, 18, 512–515. [Google Scholar] [CrossRef] [PubMed]
- Cosenza, C.A.; Sher, L.S.; Poletti, B.J.; Tschirhart, D.; Noguchi, H.; Hoffman, A.L.; Lopez, R.R.; Lugo, D.; Rojter, S.E.; Makowka, L. Metastasis of hepatocellular carcinoma to the right colon manifested by gastrointestinal bleeding. Am. Surg. 1999, 65, 218–221. [Google Scholar] [PubMed]
- Kurachi, K.; Suzuki, S.; Yokoi, Y.; Okumura, T.; Inaba, K.; Igarashi, T.; Takehara, Y.; Konno, H.; Baba, S.; Nakamura, S. A 5-year survivor after resection of peritoneal metastases from pedunculated-type hepatocellular carcinoma. J. Gastroenterol. 2002, 37, 571–574. [Google Scholar] [CrossRef]
- Zech, C.J.; Bilzer, M.; Mueller-Lisse, U.G.; Steitz, H.O.; Haraida, S.; Reiser, M.F.; Zech, M.C.J. Perforation of the colon: A rare complication of hepatocellular carcinoma. Acta Radiol. 2006, 47, 538–542. [Google Scholar] [CrossRef]
- Tapuria, N.; Sinha, C.K.; Michael, N.; Fisher, P.W. Haematogenous metastasis to ascending colon in a patient with hepatocellular carcinoma and autoimmune hepatitis. Eur. J. Gastroenterol. Hepatol. 2007, 19, 607–609. [Google Scholar] [CrossRef]
- Kaibori, M.; Morita, M.; Tagami, S.; Uchida, Y.; Tanaka, H.; Yoshioka, K.; Takahashi, Y.; Sakaida, N.; Uemura, Y.; Seki, T.; et al. Cutaneous and Colonic Metastases After Resection of Hepatocellular Carcinoma. Am. J. Dig. Dis. 2007, 52, 1114–1117. [Google Scholar] [CrossRef]
- Ng, D.S.C.; Chok, K.S.H.; Law, W.-L.; Collins, R.J.; Fan, S.-T. Long-term survival after resection of extrahepatic recurrence of hepatocellular carcinoma at the right colon. Int. J. Color. Dis. 2007, 22, 1411–1412. [Google Scholar] [CrossRef]
- Hirashita, T.; Ohta, M.; Iwaki, K.; Kai, S.; Shibata, K.; Sasaki, A.; Nakashima, K.; Kitano, S. Direct invasion to the colon by hepatocellular carcinoma: Report of two cases. World J Gastroenterol. 2008, 14, 4583–4585. [Google Scholar] [CrossRef] [PubMed]
- Nozaki, Y.; Kobayashi, N.; Shimamura, T.; Akiyama, T.; Inamori, M.; Iida, H.; Endo, H.; Fujita, K.; Yoneda, M.; Takahashi, H.; et al. Colonic Metastasis from Hepatocellular Carcinoma: Manifested by Gastrointestinal Bleeding. Am. J. Dig. Dis. 2008, 53, 3265–3266. [Google Scholar] [CrossRef] [PubMed]
- Yoo, D.-J.; Chung, Y.-H.; Lee, Y.-S.; Kim, S.E.; Jin, Y.-J.; Lee, Y.M.; Kim, M.-J. Sigmoid colon metastasis from hepatocellular carcinoma. Korean J. Hepatol. 2010, 16, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.-F.; Chou, J.-W.; Lai, H.-C. A Rare Cause of Bloody Stools in a 57-Year-Old Woman With Hepatocellular Carcinoma. Gastroenterology 2011, 140, e5–e6. [Google Scholar] [CrossRef]
- Shih, Y.J.; Hsu, K.F.; Yu, J.C.; Chan, D.C.; Hsieh, C.B. Synchronous hepatocellular carcinoma and sigmoid colon metastasis present-ing as liver and intra-abdominal abscesses. Acta Gastroenterol. Belg. 2012, 75, 278–279. [Google Scholar]
- Haga, Y.; Chiba, T.; Ohira, G.; Kanai, F.; Yokota, H.; Motoyama, T.; Ogasawara, S.; Suzuki, E.; Ooka, Y.; Tawada, A.; et al. Simultaneous Resection of Disseminated Hepatocellular Carcinoma and Colon Cancer. Case Rep. Gastroenterol. 2013, 7, 37–43. [Google Scholar] [CrossRef]
- Sun, L.-H.; Han, H.-Q.; Wang, P.-Z.; Tian, W.-J. Emergency caudate lobectomy for ruptured hepatocellular carcinoma with multiple primary cancers. World J. Gastroenterol. 2013, 19, 418–421. [Google Scholar] [CrossRef]
- Ou, T.-M.; Tsai, W.-C.; Hsieh, T.-Y.; Shih, Y.-L. Hepatocellular carcinoma with colonic metastasis. Singap. Med. J. 2014, 55, e93–e95. [Google Scholar] [CrossRef]
- Imada, S.; Noura, S.; Ohue, M.; Shingai, T.; Sueda, T.; Gotoh, K.; Yamada, T.; Tomita, Y.; Yano, M.; Ishikawa, O. Recurrence of hepatocellular carcinoma presenting as an asymptomatic appendiceal tumor: Report of a case. Surg. Today 2012, 43, 685–689. [Google Scholar] [CrossRef]
- Kohli, R.; Purysko, A.S.; John, B.V. Hematochezia From Metastasis of Hepatocellular Carcinoma to Colon in a Patient Who Underwent Liver Transplantation. Clin. Gastroenterol. Hepatol. 2014, 12, A23–A24. [Google Scholar] [CrossRef]
- Zhu, X.; Lou, Z.; He, J.; Sui, J.; Ma, J.; Gong, H.; Zhang, W. Hematogenous transverse colon metastasis from primary hepatocellular carcinoma. Int. J. Color. Dis. 2015, 31, 1241–1242. [Google Scholar] [CrossRef] [PubMed]
- Mitsialis, V.; Lee, L.S. Metastasis of Hepatocellular carcinoma to Distal colon Associated With Inferior Mesenteric Arteriovenous Fistula and Tumor Thrombus: A case Report. Am. J. Gastroenterol. 2018, 113, 916–918. [Google Scholar] [CrossRef] [PubMed]
- Repullo, D.; Diaz, M.; Holbrechts, S.; Gomez-Galdon, M.; Van Gestel, D.; Bohlok, A.; Liberale, G.; Donckier, V. Unusual presentation of a hepatocellular carcinoma as a potential late side effect of radiotherapy in a patient treated for Wilms tumor in childhood. World J. Surg. Oncol. 2018, 16, 48. [Google Scholar] [CrossRef] [PubMed]
- Tagliabue, F.; Burati, M.; Chiarelli, M.; Marando, A.; De Simone, M.; Cioffi, U. Left colonic metastasis from primary hepatocellular carcinoma: A case report. World J. Clin. Cases 2019, 7, 2044–2048. [Google Scholar] [CrossRef] [PubMed]
- Van Pham, B.; Phan, H.H.; Le Ngo, L.; Nguyen, H.T.T.; Van Le, K.; Dinh, T.C.; Bac, N.D.; Toi, C.D. A Rare Colonic Metastasis Case from Hepatocellular Carcinoma. Open Access Maced. J. Med Sci. 2019, 7, 4368–4371. [Google Scholar] [CrossRef] [PubMed]
- Soni, A.; Malhi, N.S. Lower Gastrointestinal Bleeding: Liver Rams into Gut! GE-Port. J. Gastroenterol. 2018, 26, 218–220. [Google Scholar] [CrossRef]
- Miyauchi, W.; Yamamoto, M.; Masahiro, M.; Shishido, Y.; Miyatani, K.; Matsunaga, T.; Sakamoto, T.; Fujiwara, Y. Colonic metastasis of hepatocellular carcinoma with repeated retroperitoneal bleeding: A case report. Surg. Case Rep. 2021, 7, 261. [Google Scholar] [CrossRef]
- Liu, K.-W.; Chen, H.-P.; Yang, C.-H. Rectal metastasis following radiofrequency ablation for hepatocellular carcinoma. Formos. J. Surg. 2013, 46, 140–143. [Google Scholar] [CrossRef]
- Nielsen, J.A.; Putcha, R.V.; Roberts, C.A. The increasing incidence of remote metastasis: A case report of metastatic hepatocellular carcinoma to the rectosigmoid. Tumori J. 2014, 100, e31–e34. [Google Scholar] [CrossRef]
- Ikeda, A.; Nagayama, S.; Yamamoto, N.; Akiyoshi, T.; Konishi, T.; Fujimoto, Y.; Fukunaga, Y.; Ishikawa, Y.; Ueno, M. A rare case of hepatocellular carcinoma metastasizing hematogenously to the rectum. Int. Cancer Conf. J. 2016, 5, 168–173. [Google Scholar] [CrossRef]
- Pasini, F.; Serenari, M.; Cucchetti, A.; Ercolani, G. Treatment options for recurrence of hepatocellular carcinoma after surgical resection: Review of the literature and current recommendations for management. Hepatoma Res. 2020, 6, 26. [Google Scholar] [CrossRef]
- Cardarelli-Leite, L.; Hadjivassiliou, A.; Klass, D.; Chung, J.; Ho, S.G.F.; Lim, H.J.; Kim, P.T.W.; Mujoomdar, A.; Liu, D.M. Current locore-gional therapies and treatment strategies in hepatocellular carcinoma. Curr. Oncol. 2020, 27 (Suppl. S3), S144–S151. [Google Scholar] [CrossRef] [PubMed]
- Okuda, K.; Moriyama, M.; Yasumoto, M.; Jinnouchi, S.; Shimokawa, Y.; Nakayama, T.; Kanda, Y.; Fukyuama, Y.; Musha, H.; Kuratomi, S.; et al. Roentgenologic demonstration of spontaneous reversal of portal blood flow in cirrhosis and primary carcinoma of the liver. Am. J. Roentgenol. 1973, 119, 419–428. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).