Simple Summary
Liriomyza trifolii is an important insect pest that infects many horticultural crops and vegetables, displaying strong interspecific competitiveness and inflicting serious harm. Here, chitin synthase 2 transcript was studied, and a prepupal immersion dsRNA delivery method was established for L. trifolii. The dsRNA can enter in the prepupal stage and play a role in the pupal stage, resulting in a decrease in the CHS2 expression level and eclosion rate. This study is an important supplement to the research on the RNAi of L. trifolii, enhances knowledge of the function of chitin synthase 2 in Liriomyza species, and may also provide a new idea for its control strategies.
Abstract
Liriomyza trifolii is an important invasive pest that infects horticultural vegetables, displaying a strong competitive advantage and showing great potential for inflicting harm. Chitin synthase is one of the key enzymes in insect chitin metabolism and plays an important role in insect growth and development. In this study, a chitin synthase (CHS) transcript of L. trifolii was cloned, and the results showed that LtCHS belongs to the CHS2 family. The expression analysis indicated the presence of the highest abundance of LtCHS2 in the pupae at different developmental stages but showed no significant difference among different tissues in the adult. Furthermore, a dsRNA immersion method was developed for RNA interference (RNAi) in L. trifolii using LtCHS2 transcript. RNAi can significantly reduce the expression of LtCHS2 in pupae, and the emergence rate of the pupae was significantly lower than that of the control. The results provide a theoretical basis for exploring the role of chitin synthase gene in L. trifolii and proposing new pest control strategies.
1. Introduction
Liriomyza trifolii (Diptera: Agromyzidae) is a pest infecting horticultural crops across the world and an invasive insect pest in both field and greenhouse settings in China [1,2]. The larvae of L. trifolii damage plants by producing tunnels in leaf tissue, whereas the adults pierce the foliage during oviposition [3]. In recent years, with the development of facility agriculture, the damage inflicted by Liriomyza spp. is becoming increasingly serious. In China, L. trifolii is mainly distributed in the southeast coastal areas, while recent investigations show that L. trifolii has invaded the northern regions of China, reaching as far as Hengshui city (37–38° N) [4,5]. Although pesticides have been an effective and convenient option for controlling L. trifolii, their irrational use has created resistance among pests. Together with other competitive factors, pesticide resistance will change the current population pattern of Liriomyza, increasing the threat of L. trifolii. In general, L. trifolii has obvious competitive advantages compared with species closely related to it [6,7,8].
RNA interference (RNAi) is a highly specific approach that suppresses gene expression at the post-transcriptional level. It can be used to investigate gene function [9]. In addition, ingested double-stranded RNA (dsRNA) may also have insecticidal properties [10,11]. However, in the RNAi-based crop protection method, the delivery of dsRNA is a major challenge. After identifying the target gene, it is important to choose a convenient strategy to deliver dsRNA to insects. Microinjection is a good strategy for functional genomic studies, but this method is not suitable for controlling insect pests in the field. Furthermore, microinjection has some limitations. For example, it is highly technical and difficult to achieve in some tiny insects [12]. Feeding methods require insects to feed and achieve RNAi goals through midgut absorption. However, midgut barriers lead to the low efficiency of RNAi in some insects [12]. It is feasible to supply dsRNA to herbivorous insects through root absorption or injection into plant tissues [13,14], and the direct immersion method is also carried out in nematode and corn borers [15,16]. Furthermore, plant-mediated RNAi and nanomaterial-mediated RNAi delivery is also being carried out, improving RNAi efficiency [12,17].
Insect chitin synthase (CHS) is one of the key enzymes in the process of insect chitin metabolism. It plays an important role in the growth and development of insects. It is also an ideal potential target gene for the development of new insecticides and has a high research value [18]. The chitin synthase gene was first obtained from Lucilia cuprin in 2000 [19], after which the chitin synthase sequences of other insects, such as Drosophila melanogaster, Tribolium castaneum, and Spodoptera frugiperda, were also cloned [20,21,22]. Chitin synthase gene sequences of different species were analyzed. It was found that chitin synthase genes can be divided into CHS1 and CHS2 [23]. CHS1 is mainly expressed in the outer embryonic layer cells, which is responsible for the synthesis of chitin in epidermis and trachea, while Chs2 is mainly expressed in the epithelial cells of midgut peritrophic membrane, which is responsible for the synthesis of chitin in midgut peritrophic membrane [18,19]. Many studies have used RNAi to inhibit the expression of CHS. The injection of small interfering RNA (siRNA) targeting CHS1 to knock down CHS1 transcripts at larval, pupal, and adult stages of Culex pipiens pallens has resulted in the appearance of different lethal phenotypes. When larval and pupal stages were injected with siCHS1, CHS1 knockdown prevented the growth and development of different insect stages and impaired the production of chitin and chitin degradation, which resulted in an ecdysis defect phenotype of mosquitoes [24]. In Henosepilachna vigintioctopunctata, dsCHS1 and dsCHS2 were used to immerse potato foliage and the treated leaves were provided to the newly molted larvae. The knockdown of HvCHS1, rather than HvCHS2, affected its growth and metamorphosis [25]. In Cnaphalocrocis medinalis, CmCHS2 was expressed throughout development and in all of the adult tissues tested, with the highest expression level in the adult and in the midgut. Silencing of CmCHS2 severely affected C. medinalis larval growth and caused larval lethality [26].
In this study, CHS2 transcript, the key to the growth and development of L. trifolii, was examined. Prepupal immersion in dsRNA was used to explore its function in the growth and development of L. trifolii. This research is of great significance to exploring the role of CHS2 in L. trifolii and developing a new control strategy based on RNAi.
2. Materials and Methods
2.1. Insects
L. trifolii populations were collected from Yangzhou (32.39° N, 119.42° E), China, and reared in the laboratory on kidney bean plants for more than five years at 26 °C with a 16:8 h (L:D) photoperiod as described [27]. Kidney bean plants were used to feed larvae and adults, and foliage with tunnels was collected for pupation.
2.2. Cloning, Sequence Alignment, and Expression of CHS2 Transcript
The RNeasy reagent (Vazyme, Nanjing, China) was used to isolate total RNA from L. trifolii pupae. RNA quality, purity and integrity were determined by spectrophotometry (Thermo NanoDrop One, Madison, WI, USA) and agarose gel electrophoresis. On the basis of previously published transcriptome data [28], we selected chitin synthase 2 transcript for further study. A partial fragment of the chitin synthase 2 transcript was amplified using specific primers (Table 1), and 5′- and 3′-Rapid amplification of cDNA ends (5′- and 3′-RACE) was used to obtain complete cDNAs as described [29].

Table 1.
Primers used in cDNA cloning, dsRNA synthesis, and real-time quantitative PCR.
Full-length cDNA of the chitin synthase 2 (CHS2) was queried against other Diptera chitin synthase genes using BLAST programs (http://www.ncbi.nlm.gov/BLAST/, accessed on 10 May 2022). Clustal X was used to align sequences [30], and open reading frames (ORFs) were identified with ORF Finder (https://www.ncbi.nlm.nih.gov/orffinder/, accessed on 10 May 2022). Tools available on the ExPASy Molecular Biology Server (https://prosite.expasy.org/, accessed on 10 May 2022) were deployed to scan specific motifs in the chitin synthase gene. MEGA software [31] and the neighbor-joining method were used to create phylogenetic trees of chitin synthase genes.
To explore the function of LtCHS2, qRT-PCR was used to study the expression pattern of LtCHS2 in different developmental stages and tissues. The developmental stages included 3rd instar larvae, prepupa, two-day-old pupae (new pupae), seven-day-old pupae (old pupae), female adults, and male adults (n = 10). The different adult tissues used were from head, thorax, abdomen, and gut, and the treatments contained three independent biological replicates. Total RNA (0.5 μg) was reverse-transcribed using the HiScript II Q RT SuperMix for qPCR (+gDNA wiper) (Vazyme, Nanjing, China). qRT-PCR was executed with gene-specific primers (Table 1) in 20 µL volumes, comprised 10 μL ChamQ SYBR qPCR Master Mix (2×) (Vazyme, Nanjing, China), 1 μL of each gene-specific primer (10 μM) (Table 1), 2 μL of cDNA template, and 6 μL of ddH2O as described [32]. Reactions were conducted with a CFX-96 real-time PCR system (Bio-Rad Laboratories, Berkeley, CA, USA) under the following conditions: 3 min at 95 °C, 39 cycles of denaturation at 95 °C for 30 s, and annealing at the Tm of primer pairs (Table 1) for 30 s. Each treatment contained four replicates, and each reaction was performed in triplicate.
2.3. Synthesis and Delivery of dsRNA
Full-length L. trifolii chitin synthase cDNA sequence was analyzed with siDirect v. 2.0 (http://sidirect2. rnai.jp/, accessed on 10 May 2022) to select potential small interfering RNA (siRNA) sequences that could be used to design dsRNA primers. Forward and reverse primers included a T7 promoter sequence (TAATACGACTCACTATAGGGAGA) at the 5′ ends to catalyze transcription from both cDNA strands (Table 1). As a control, dsRNA specific to the gene encoding green florescent protein (GFP) was used (Table 1). PCR products were inserted into pGEM-T easy vector (Promega, Madison, WI, USA), and resulting constructs were used as template DNA in subsequent amplifications. Purified DNA templates (1.5 µg) were used for in vitro dsRNA synthesis and purified using the MEGAscriptTM RNAi Kit (Thermo, Waltham, MA, USA) according to manufacturer’s protocol. The quality and integrity of dsRNA was evaluated by spectrophotometry and gel electrophoresis.
We selected the prepupa that had just left the leaf as the experimental insect developmental stages, placed them into the Petri dish, and used different concentrations of dsRNA and 1% RNATransMate (Sangon Biotech, Shanghai, China) to immerse the prepupa. After 10 s immersion, we removed the excess droplets of dsRNA using a soft brush, to prevent it from blocking the stomata and affecting pupation, and used the prepupa for subsequent experiments. Each treatment involved 10 prepupae, each treatment was repeated 3 times, and dsGFP was used as the control.
2.4. Analysis of Silencing Efficiency
Concentration experiments were performed using the experimental design described above to evaluate the impact of dsRNA delivery on the pupae. The experiment was performed with three replicates per treatment, and dsGFP was used as a control. Three concentrations (dsCHS: 300, 600, and 900 ng/μL; dsGFP: 600 ng/μL) were selected for further analysis, and silencing efficiency was determined 2 days after pupation. Each treatment involved 30 pupae (n = 30). The pupae were collected for RNA extraction, and silencing efficiency was analyzed by qRT-PCR. In addition, survival rates were calculated for the 600 ng/μL dsRNA group, containing 30 pupae (10 individuals representing one repetition). The number of eclosion adults was recorded.
2.5. Statistical analysis
Expression levels of CHS2 were identified using the 2−ΔΔCt method [33], and Actin and 18S served as reference genes [32]. Relative transcript abundance was calculated using the average Cq values of the two reference genes. The expression of the CHS2 transcript under different developmental stages, using tissues and different dsRNA concentrations, was analyzed with one-way ANOVA, followed by Tukey’s multiple comparison and analysis with SPSS v. 16.0. For ANOVA, data were tested for homogeneity of variances using Levene’s test and transformed to follow a normal distribution. In addition, Student’s t-test was used to compare differences in mortality with SPSS v. 16.0, and differences were considered significant at p < 0.05.
3. Results
3.1. Sequence Characteristics and Phylogenetic Analysis of CHS2 Transcript from L. trifolii
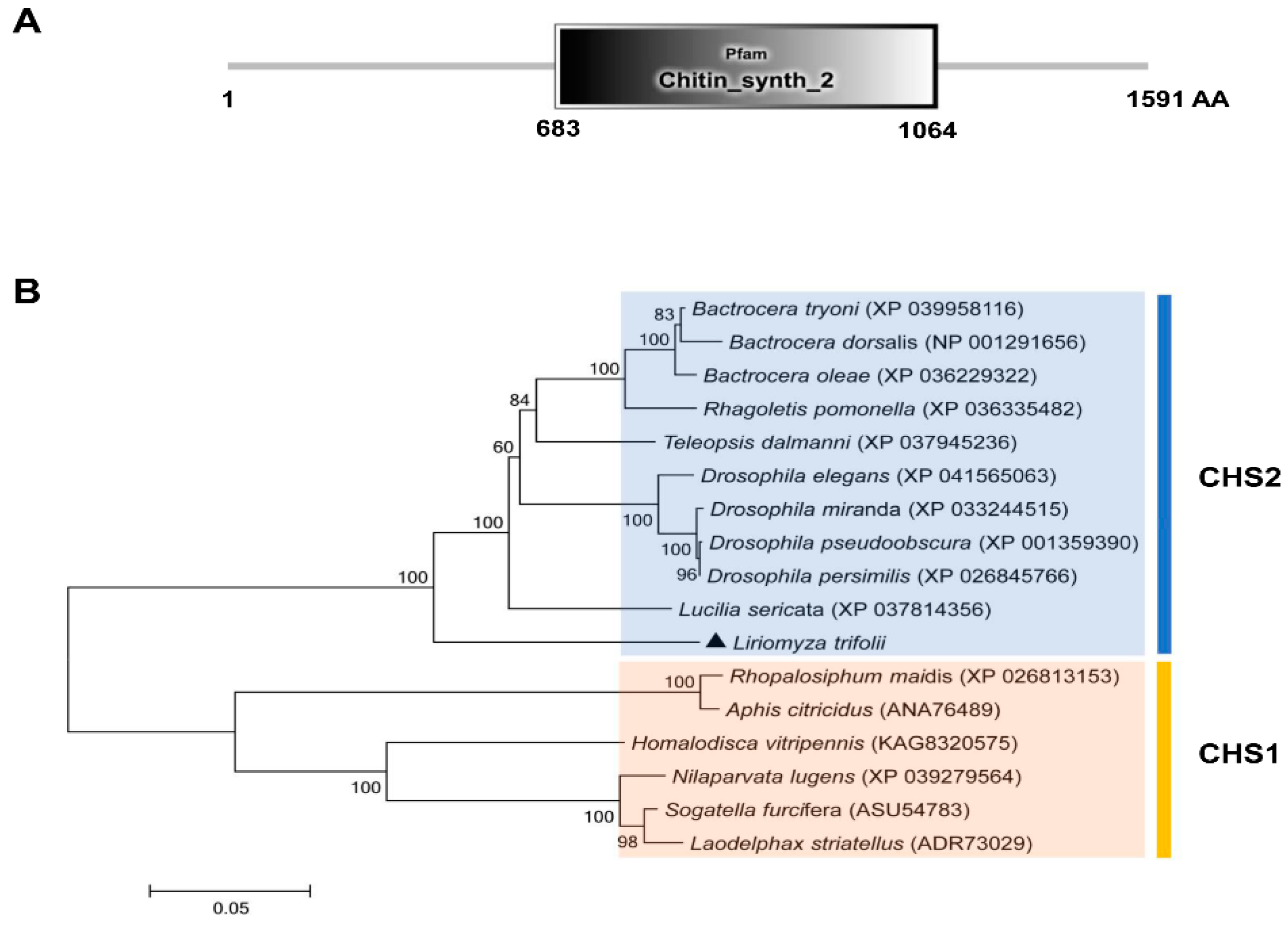
In this study, a CHS2 cDNA sequence was cloned in L. trifolii. The full length is 5650 bp and the open reading frame (ORF) is 4776 bp long. It is predicted to encode 1591 amino acids, has a molecular weight of 180.41 kDa and an isoelectric point of 6.38. The sequence was submitted to GenBank as accession no. ON453844. The characteristic sequence of the CHS2 gene family was found to be located in the amino acid sequence encoded by the CHS2 gene (chitin_synth_2: 683-1064) (Figure 1A and Figure S1). In addition, the characteristic sequences of chitin synthase (EDR and QRRRW) were also found (Figure S1). A phylogenetic tree was obtained using the amino acid sequences of 16 chitin synthases in Diptera, including 10 chitin synthase 2 and 6 chitin synthase 1 sequences. The phylogenetic tree contained two distinct clusters containing chitin synthase 1 and chitin synthase 2. The results showed that CHS2 of L. trifolii formed a branch of chitin synthase 2 and was close to Lucillia sericata (Figure 1B).
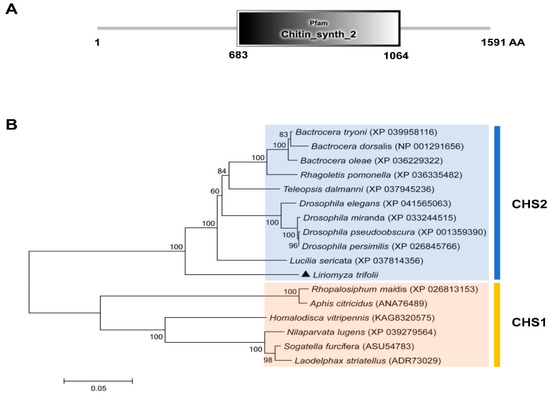
Figure 1.
The domain organization of CHS2 (A) and the neighbor-joining phylogenetic tree of CHS2 (B). L. trifolii CHS2 is labeled with triangles. Numbers on the branches represent bootstrap values obtained from 1000 replicates (only bootstrap values >50 are shown).
3.2. Expression Pattern of CHS2 Transcript in L. trifolii
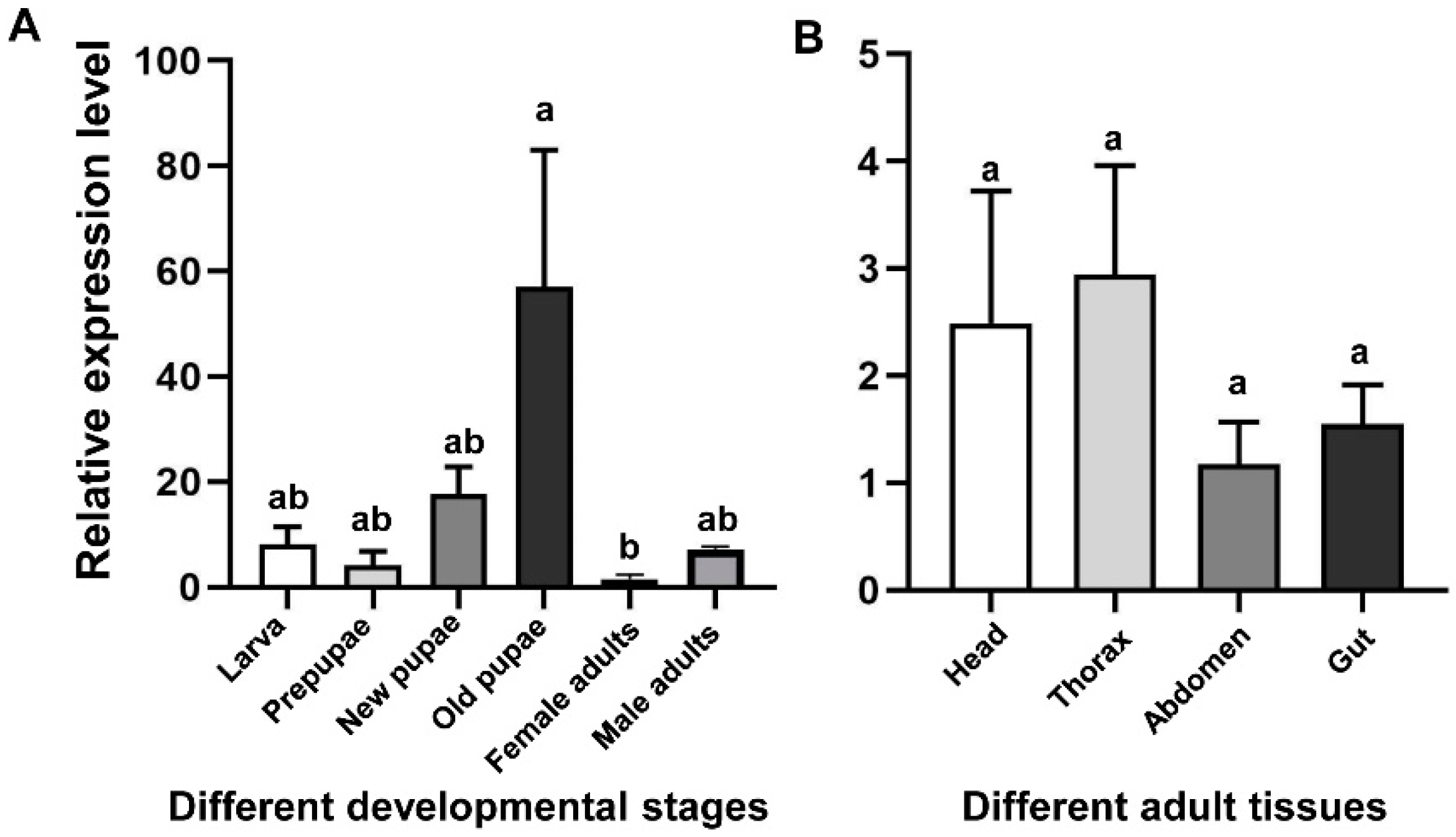
qRT-PCR was used to detect the mRNA levels of CHS2 transcript in different developmental stages (larvae, prepupae, new pupae, old pupae, and female and male adults) and in different tissues from the adult (head, thorax, abdomen, and gut) (Figure 2). The results showed that the expression levels of CHS2 transcript in L. trifolii were significantly different at different developmental stages. The expression level in the old pupae was the highest, 16.17-fold higher than that in the control group (female adults) (F5,12 = 4.230; p < 0.05) (Figure 2A). However, in different tissues, there was no significant difference among adult tissues (F3,8 = 0.945; p = 0.463) (Figure 2B).
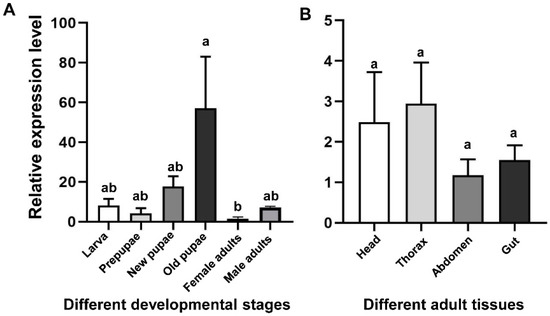
Figure 2.
Transcript relative expression analysis of CHS2 in L. trifolii at different developmental stages (A) and adult tissues (B). Different lowercase letters in panels indicate significant differences between treatments. Tukey’s multiple range test was used for pairwise comparison of means (p < 0.05).
3.3. Functional Verification of CHS2 Transcript of L. trifolii
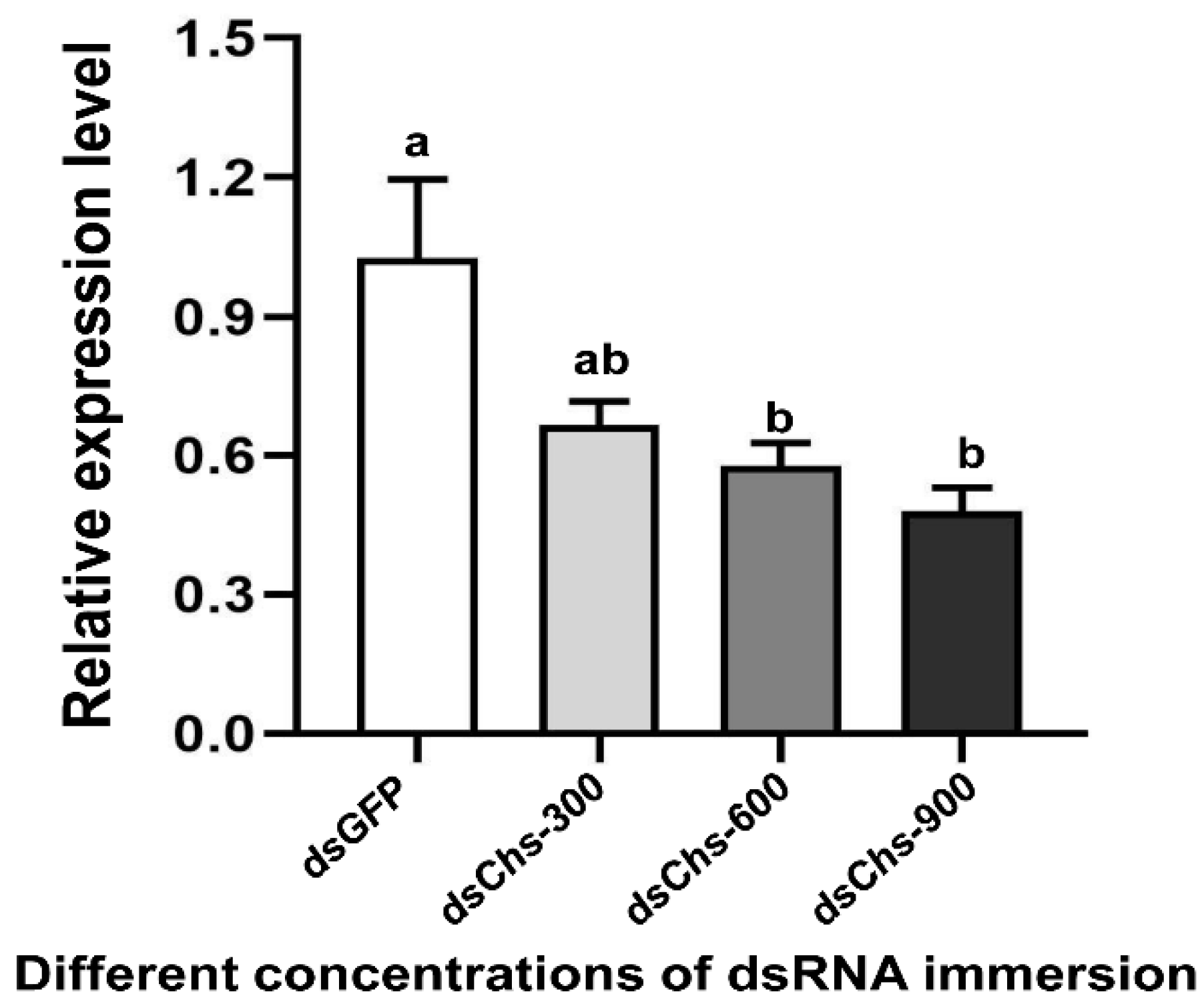
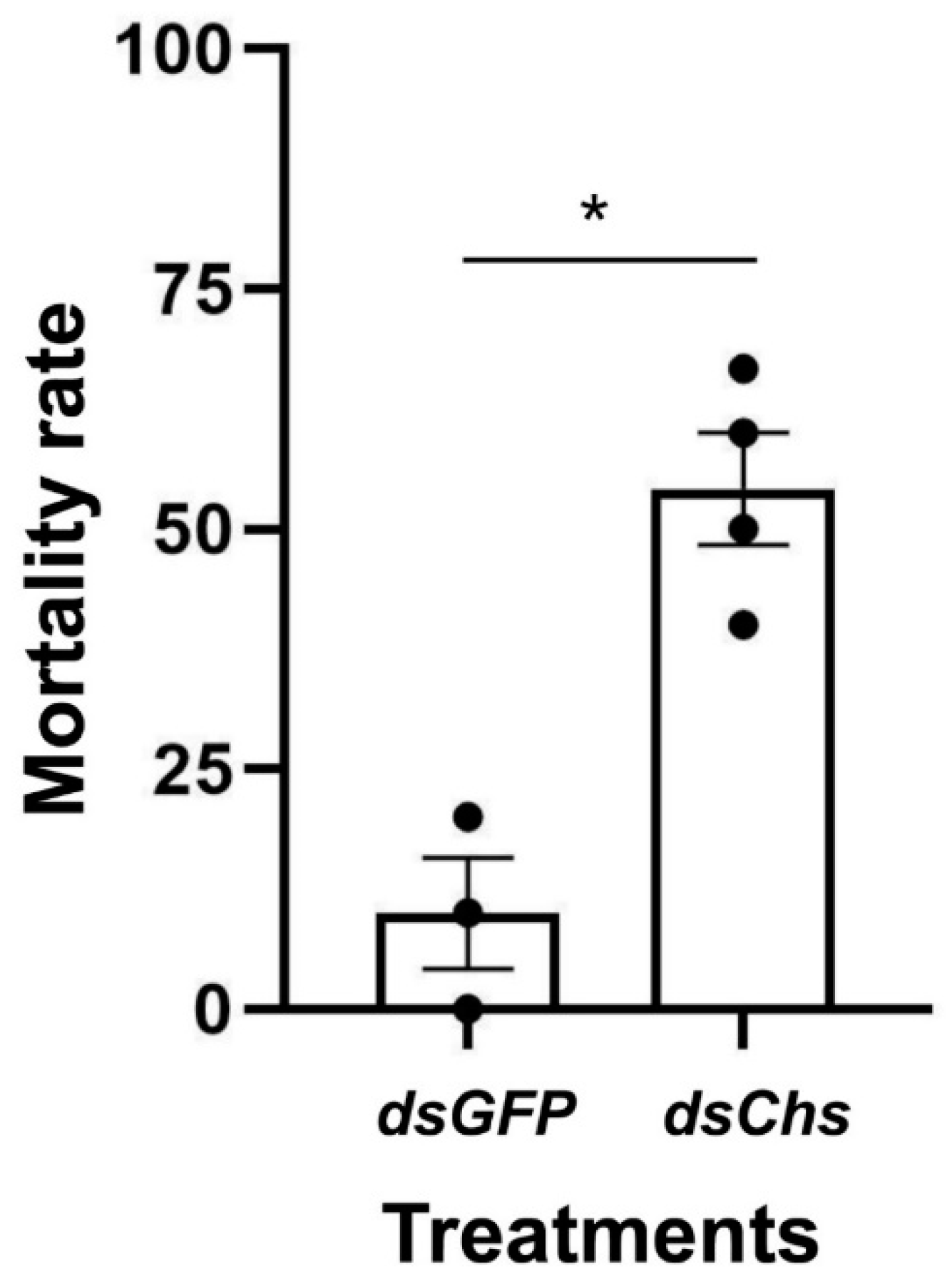
RNA interference studies were conducted by immersing L. trifolii prepupae in the dsRNA of CHS2. There was a significant difference in the CHS2 expression levels in L. trifolii with 600 and 900 ng/μL of dsCHS2, which were 56.86 and 56.04% of the dsGFP control group, respectively (F3,8 = 8.207; p < 0.05). However, CHS2 expression was not significantly different with 300 ng/μL of dsCHS2 compared with the dsGFP control group (Figure 3). In terms of mortality, compared with the dsGFP control group, the mortality rate of L. trifolii, when immersed in dsCHS2 at the prepupal stage, was significantly higher than that of the dsGFP control group, which were 54.17% and 10%, respectively (t = 5.242; p < 0.05) (Figure 4).
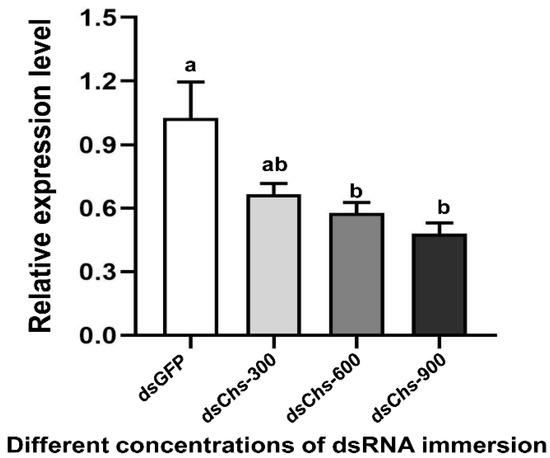
Figure 3.
Relative expression levels of CHS2 under immersion treatments with different concentrations of dsRNA. The data are denoted as the mean ± the SE. One-way analysis of variance (ANOVA) was used to analyze the relative expression levels of CHS2 under different treatments. Different lowercase letters indicate significant differences among different temperature treatments. Tukey’s multiple range test was used for pairwise comparison for mean separation (p < 0.05).
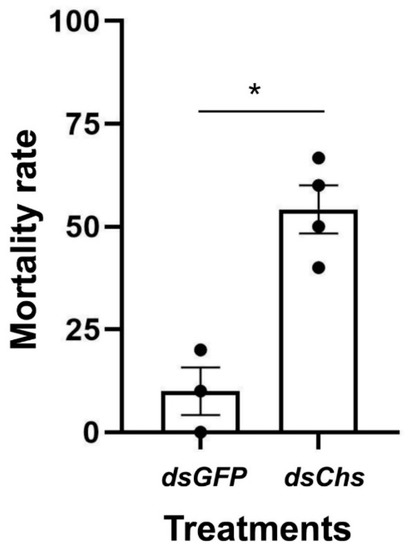
Figure 4.
RNAi-mediated knockdown of CHS2 decreases L. trifolii survival. Percentage of mortality in insects immersed in dsCHS2 and dsGFP (control). The data are denoted as the mean ± the SE. Dots represent individual repetitions. Data were analyzed by Student’s t-test, p < 0.05. Asterisks represent significant differences in mortality data.
4. Discussion
Chitin synthase (CHS) plays a critical role in the synthesis of insect cuticle [18,34]. However, the current research on CHS is still focused on some model insects, and there is far more research on CHS1 than on CHS2 in terms of gene types [20,21,24,25]. Therefore, the experimental object of this study was L. trifolii, and the CHS2 cDNA sequence and the function of CHS2 in L. trifolii were extensively studied. The CHS2 transcript of L. trifolii was originally obtained from previous transcriptome data of L. trifolii [28] and full-length cloning and sequencing in this study. CHS contains characteristic sequences of the CHS2 gene family, including the characteristic sequences EDR and QRRRW unique to chitin synthase, which exist in all types of chitin synthase [19]. CHS2 obtained in this study is distributed in the CHS2 family region in the phylogenetic tree, which lays a solid foundation for further study of this gene in L. trifolii.
Chitin synthase has different expression patterns in different developmental stages and tissues, so its functions are also different. The expression pattern results of CHS2 of L. trifolii show that CHS2 transcript is expressed in each growth and development stage of L. trifolii, indicating that the CHS2 gene plays a role in each development stage of L. trifolii. However, the expression levels vary in different development stages. The expression levels of CHS2 were the highest in old pupae and lower in larvae and new pupae, but the expression levels at the prepupae and adult stages were the lowest. This expression pattern is different from the results of previous studies on Grapholita molesta. GmCHS2 is less expressed in the larval stage, and the expression will suddenly increase in prepupae, while the expression in the pupal stage is low but the expression in the adult stage is significantly higher than that in the larval stage [35]. In addition, although the expression of D. melanogaster in the larval stage is relatively low, its expression increases significantly in prepupae [20]. This difference may be related to the feeding habits of different insect species and the habit of the L. trifolii larvae to feed inside leaves, which is significantly different from the habits of other insects. At the same time, L. trifolii needs to leave the leaf to pupate in the prepupal stage and changes in environmental conditions may also lead to changes in the expression of CHS2, but the mechanism is still unknown. The expression of CHS2 increased significantly in old pupae. L. trifolii is about to emerge in this developmental stage, and the CHS2 gene may be involved in the process of metamorphosis and development of L. trifolii and the imminent life activities, such as feeding and mating, after emergence, which is similar to the pattern of high expression of CHS2 in the pupal stage in Culex pipiens quinquefasciatus and Anopheles gambiae [36]. In general, as mentioned above, the expression patterns of CHS2 in different insects at different developmental stages are significantly different. Therefore, it is speculated that the development duration criteria of different insects may be different and may also be related to the specific living habits of different insects and other functions of the CHS2 gene at certain developmental stages, which need to be further studied. In addition, in different tissues of L. trifolii, CHS2 was expressed in the head, thorax, abdomen, and gut. The analysis of variance showed that there was no significant difference among different tissues. Relevant studies have shown that CHS2 participates in the synthesis of the peritrophic membrane of gut epithelial tissue, so it is mainly expressed in the insect gut [37]. In this study, the expression of CHS2 in the gut was also higher than that in other abdominal tissues (noted after intestinal dissection), although there was no significant difference in the results. The high expression of CHS2 in other tissues suggests that CHS2 may have other functions in different tissues.
RNA interference is a phenomenon of gene silencing at the post transcriptional level, and it has been widely used in the study of gene function [12]. The object of this study was L. trifolii, and the CHS2 transcript function was studied extensively. RNAi technology in L. trifolii adults has been reported previously. However, due to the tiny size of L. trifolii adults, the mortality caused by microinjection is relatively high. The limitations of microinjection make exploring the function of genes specifically expressed in other insect states challenging [38]. The immersion method is applicable to the RNAi of some pests, and relevant research on RNAi mediated by nanomaterials may help improve the efficiency of RNAi [10,12,39]. In this study, RNAi delivery was carried out by the direct immersion method. Through the biological habit of L. trifolii, the larvae left the leaves to pupate and were immersed in the prepupal stage of the key period. The prepupal stage can turn into pupae within a few hours [40], and dsRNA can be incorporated into the body to achieve dsRNA delivery. In this experiment, after dsCHS2 immersion in the prepupal stage, qPCR results showed that the CHS2 was significantly reduced after interference and the mortality rate of the adults was significantly increased. In this study, we chose only dsGFP with a concentration of 600 ng/μL for immersion treatment, based on the fact that dsGFP is a control and its synthetic reagents are the same as the target dsRNA. Therefore, we have reason to believe that the concentration of dsGFP in this study does not affect the survival of L. trifolii and the expression of the target LtCHS2. However, it is worth noting that the length of dsRNA and concentration of the controls still need to be carefully considered in each RNAi experiment. In other studies, when the larvae of T. castaneum were injected with dsTcCHS2, the larvae shrunk due to hunger and the peritrophic membrane was basically no longer generated [21]. Injecting dsCHS2 in the third instar silkworm larvae, most of which do not molt or do not molt normally, interferes with the expression of BmCHS2 [41]. When the second and fourth instar potato beetle larvae were fed dsCHS2, the larvae ate less and grew slowly, and the content of chitin in the midgut decreased [42]. Feeding dsCHS2 had no effect on the larval stage of Spodoptera exigua but had a certain effect on prepupae and adults, and 20% and 25% could not pupate and eclose, respectively [43]. Similarly, in a study on locusts, the intake of locusts injected with dsCHS2 was significantly reduced, and the length of the midgut was significantly shorter [44]. In addition, in a study of Anopheles gambiae, chitosan-coated dsRNA was used to interfere with AgCHS2. The results showed that the content of chitin in its peritrophic membrane decreased, affecting its growth and development [45]. Therefore, chitin synthase 2 plays an important role in insect growth and development. However, in this study, the average life span is shorter and feeding is limited for Liriomyza adults, which makes some physiological observations impractical.
5. Conclusions
RNAi-based gene function research and pest control strategies have been tried in some species, but most of the current research is still in its infancy. In this study, CHS2, an important growth and development gene, was selected as the target gene and the prepupal immersion method was used to verify the function of the CHS2 gene. In general, dsRNA can enter in the prepupal stage and play a role in the pupal stage, resulting in a decrease in the CHS2 transcript expression level and eclosion rate. The results provide a theoretical basis for exploring the role of the chitin synthase gene in L. trifolii and may propose new pest control strategies.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/insects13090832/s1, Figure S1: Nucleotide sequences for L. trifolii CHS2 cDNA and its predicted amino acid sequence. Nucleotide numbering starts with the adenine in the first methionine codon of the putative open reading frame. The highly conserved region, chitin_synth_2 domain, is highlighted. The asterisk indicates the translational termination codon. The EDR and QRRRW motifs are boxed.
Author Contributions
Data curation, Y.-W.C., Y.-C.W. and Y.-Q.Y.; Funding acquisition, D.-R.Y. and Y.-Z.D.; Software, Y.-W.C., Y.-Q.Y. and H.-F.X.; Writing—original draft, Y.-W.C. and Y.-Z.D. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the start-up project of high-level talent of Yangzhou University (137012465), the earmarked fund for Jiangsu agricultural industry technology system (JATS [2021] 346), the Jiangsu science & technology support program (BE2014410), the postgraduate Research and Practice Innovation Program of Jiangsu Province (KYCX22_3524), and the special fund for detection and identification of sudden major agricultural pests in Nanjing Area.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data is contained within the article and Supplementary Material.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Spencer, K.A. Agromyzidae (Diptera) of Economic Importance. 9: Series Entomologica; The Hague Publishers: Bath, UK, 1973; pp. 19–28. [Google Scholar]
- Wan, F.H.; Yang, N.W. Invasion and management of agricultural alien insects in China. Annu. Rev. Entomol. 2016, 6, 77–98. [Google Scholar] [CrossRef] [PubMed]
- Kang, L.; Chen, B.; Wei, J.N.; Liu, T.X. Roles of thermal adaptation and chemical ecology in Liriomyza distribution and control. Annu. Rev. Entomol. 2009, 54, 127–145. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.L.; Reitz, S.; Xing, Z.L.; Ferguson, S.; Lei, Z.R. A decade of a leafminer invasion in China: Lessons learned. Pest Manag. Sci. 2017, 73, 1775–1779. [Google Scholar] [CrossRef]
- Chen, J.Y.; Chang, Y.W.; Tang, X.T.; Zheng, S.Z.; Du, Y.Z. Population genetics of Liriomyza trifolii (Diptera: Agromyzidae) and comparison with four Liriomyza species in China based on COI, EF-1a and microsatellites loci. Sci. Rep. 2019, 9, 17856. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.L.; Reitz, S.R.; Wei, Q.B.; Yu, W.Y.; Lei, Z.R. Insecticide-mediated apparent displacement between two invasive species of leafminer fly. PLoS ONE 2012, 7, e36622. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.H.; Reitz, S.R.; Xiang, J.C.; Smagghe, G.; Lei, Z.R. Does Temperature-Mediated Reproductive Success Drive the Direction of Species Displacement in Two Invasive Species of Leafminer Fly? PLoS ONE 2014, 9, e98761. [Google Scholar] [CrossRef]
- Chang, Y.W.; Wang, Y.C.; Zhang, X.X.; Iqbal, J.; Lu, M.X.; Gong, H.X.; Du, Y.Z. Comparative transcriptome analysis of three invasive leafminer flies provides insights into interspecific competition. Int. J. Biol. Macromol. 2020, 165, 1664–1674. [Google Scholar] [CrossRef]
- Fire, A.; Xu, S.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E.; Mello, C.C. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 1998, 391, 806–811. [Google Scholar] [CrossRef]
- Burand, J.P.; Hunter, W.B. RNAi: Future in insect management. J. Invertebr. Pathol. 2013, 112, S68–S74. [Google Scholar] [CrossRef]
- Palli, S.R. RNA interference in Colorado potato beetle: Steps toward development of dsRNA as a commercial insecticide. Curr. Opin. Insect Sci. 2014, 6, 1–8. [Google Scholar] [CrossRef]
- Joga, M.R.; Zotti, M.J.; Smagghe, G.; Christiaens, O. RNAi Efficiency, systemic properties, and novel delivery methods for pest insect control: What we know so far. Front. Physiol. 2016, 7, 553. [Google Scholar] [CrossRef] [PubMed]
- Hunter, W.B.; Glick, E.; Paldi, N.; Bextine, B.R. Advances in RNA interference: dsRNA treatment in trees and grapevines for insect pest suppression. Southwest Entomol. 2012, 37, 85–87. [Google Scholar] [CrossRef]
- Li, H.; Guan, R.; Guo, H.; Miao, X. New insights into an RNAi approach for plant defence against piercing-sucking and stem-borer insect pests. Plant Cell Environ. 2015, 38, 2277–2285. [Google Scholar] [CrossRef] [PubMed]
- Tabara, H.; Grishok, A.; Mello, C.C. RNAi in C. elegans: Soaking in the genome sequence. Science 1998, 282, 430–431. [Google Scholar] [CrossRef]
- Zhang, H.; Li, H.; Guan, R.; Miao, X. Lepidopteran insect species-specific, broad-spectrum, and systemic RNA interference by spraying dsRNA on larvae. Entomol. Exp. Appl. 2015, 155, 218–228. [Google Scholar] [CrossRef]
- Zheng, Y.; Hu, Y.; Yan, S.; Zhou, H.; Song, D.; Yin, M.; Shen, J. A polymer/detergent formulation improves dsRNA penetration through the body wall and RNAi-induced mortality in the soybean aphid Aphis glycines. Pest Manag. Sci. 2019, 75, 1993–1999. [Google Scholar] [CrossRef]
- Cohen, E. Chitin synthesis and inhibition, a revisit. Pest Manag. Sci. 2001, 57, 946–950. [Google Scholar] [CrossRef]
- Tellam, R.; Vuocolo, T.; Johnson, S.E.; Jarmey, J.; Pearson, R.D. Insect chitin synthase cDNA sequence, gene organization and expression. Eur. J. Biochem. 2000, 267, 6025–6043. [Google Scholar] [CrossRef]
- Gagou, M.E.; Kapsetaki, M.; Turberg, A.; Kafetzopoulos, D. Stage-specific expression of the chitin synthase DmeChSA and DmeChSB genes during the onset of Drosophila metamorphosis. Insect Biochem. Mol. Biol. 2002, 32, 141–146. [Google Scholar] [CrossRef]
- Arakane, Y.; Zhu, Y.C.; Kramer, K.J.; Charles, A.; Michael, R. Characterization of two chitin synthase genes of the red flour beetle, Tribolium castaneum, and alternate exon usage in one of the genes during development. Insect Biochem. Mol. Biol. 2004, 34, 291–304. [Google Scholar] [CrossRef]
- Hogenkamp, D.G.; Arakane, Y.; Zimoch, L.; Merzendorfer, H.; Kramer, K.J.; Beeman, R.W.; Kanost, M.R.; Specht, C.A.; Muthukrishnan, S. Chitin synthase genes in Manduca sexta, characterization of a gut-specific transcript and differential tissue expression of alternately spliced mRNAs during development. Insect Biochem. Mol. Biol. 2005, 35, 529–540. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.Q.; Chen, X.F.; Tang, B.; Tian, H.G.; Chen, J.; Yao, Q. Insect chitin biosynthesis and its regulation. Chinese J. Appl. Entomol. 2011, 48, 475–479. [Google Scholar]
- Yang, X.S.; Xu, Y.; Yin, Q.; Zhang, H.B.; Yin, H.T.; Sun, Y.; Ma, L.; Zhou, D.; Shen, B. Physiological characterization of chitin synthase A responsible for the biosynthesis of cuticle chitin in Culex pipiens pallens (Diptera: Culicidae). Parasit. Vectors 2021, 14, 234. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.H.; Mu, L.L.; Jin, L.; Anjum, A.A.; Li, G.Q. RNAi for chitin synthase 1 rather than 2 causes growth delay and molting defect in Henosepilachna vigintioctopunctata. Pestic. Biochem. Physiol. 2021, 178, 104934. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.J.; Xia, L.; Du, J.; Li, S.W.; Zhao, F. Cloning, characterization, and RNAi effect of the chitin synthase B gene in Cnaphalocrocis medinalis. J. Asia Pac. Entomol. 2021, 24, 486–492. [Google Scholar] [CrossRef]
- Chen, B.; Kang, L. Cold hardiness and supercooling capacity in the pea leafminer Liriomyza huidobrensis. Cryo. Lett. 2002, 23, 173–182. [Google Scholar]
- Chang, Y.W.; Zhang, X.X.; Lu, M.X.; Gong, W.R.; Du, Y.Z. Transcriptome analysis of Liriomyza trifolii (Diptera: Agromyzidae) in response to temperature stress. Comp. Biochem. Physiol. Part D Genom. Proteom. 2020, 34, 100677. [Google Scholar] [CrossRef]
- Chang, Y.W.; Chen, J.Y.; Lu, M.X.; Gao, Y.; Tian, Z.H.; Gong, W.R.; Dong, C.S.; Du, Y.Z. Cloning and expression of genes encoding heat shock proteins in Liriomyza trifolii and comparison with two congener leafminer species. PLoS ONE 2017, 12, e0181355. [Google Scholar] [CrossRef]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The Clustal-X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA 6: Molecular Evolutionary Genetics Analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Chang, Y.W.; Chen, J.Y.; Lu, M.X.; Gao, Y.; Tian, Z.H.; Gong, W.R.; Zhu, W.; Du, Y.Z. Selection and validation of reference genes for quantitative real time PCR analysis under different experimental conditions in the leafminer Liriomyza trifolii (Diptera: Agromyzidae). PLoS ONE 2017, 12, e0181862. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−ΔΔC(T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Merzendorfer, H. Insect chitin synthases: A review. J. Comp. Physiol. B Biochem. 2006, 176, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yu, Q.; Gao, Y.; Liu, Z.F.; Shi, G.C.; Zhang, P.J.; Fan, R.J. Molecular cloning, sequence analysis and expression of chitin synthase 2gene in the oriental fruit moth Graphlitha molesta (Busck). Chinese J. Appl. Entomol. 2017, 54, 407–416. [Google Scholar]
- Zhao, W.J.; Zhang, C.L.; Zhai, S.Z.; Sun, Q.; Zhang, J. Expression and Analysis of Chitin Synthase Gene CqCHS1 and CqCHS2 in Culex quinquefasciatus. Genom. Appl. Biol. 2016, 35, 2317–2323. [Google Scholar]
- Merzendorfer, H.; Zimoch, L. Chitin metabolism in insects: Structure, function and regulation of chitin synthases and chitinases. J. Exp. Biol. 2005, 206, 4393–4412. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.W.; Wang, Y.C.; Zhang, X.X.; Iqbal, J.; Du, Y.Z. RNA interference of genes encoding the vacuolar-ATPase in Liriomyza trifolii. Insects 2021, 12, 41. [Google Scholar] [CrossRef]
- Baum, J.A.; Roberts, J.K. Progress towards RNAi-mediated insect pest management. Adv. Insect Physiol. 2014, 47, 249–295. [Google Scholar]
- Parrella, M.P. Biology of Liriomyza. Annu. Rev. Entomol. 1987, 32, 201–224. [Google Scholar] [CrossRef]
- Zhuo, W.W.; Chu, F.; Kong, L.F.; Tao, H.; Sima, Y.H.; Xu, S.Q. Chitin synthase B: A midgut-specific gene induced by insect hormones and involved in food intake in Bombyx mori larvae. Arch. Insect Biochem. Physiol. 2014, 85, 36–47. [Google Scholar] [CrossRef]
- Shi, J.F.; Mu, L.L.; Chen, X.; Guo, W.C.; Li, G.Q. RNA interference of chitin synthase genes inhibits chitin biosynthesis and affects larval performance in Leptinotarsa decemlineata (Say). Int. J. Biol. Sci. 2016, 12, 1319–1331. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.S.; Tang, B.; Chen, X.F.; Tin, H.G.; Zhang, W.Q. Molecular cloning, expression pattern and comparative analysis of chitin synthase gene B in Spodoptera exigua. Comp. Biochem. Physiol. B. Biochem. Mol. Biol. 2008, 149, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.J.; Cui, M.; Li, D.Q.; Zhang, H.H.; Yang, M.L.; Zhang, J.Z. Expression, function and regulation of Chitin synthase 2 gene in Locusta migratoria. Sci. Agric. Sin. 2014, 47, 1330–1340. [Google Scholar]
- Zhang, X.; Zhang, J.; Zhu, K.Y. Chitosan/double-stranded RNA nanoparticle-mediated RNA interference to silence chitin synthase genes through larval feeding in the African malaria mosquito (Anopheles gambiae). Insect Mol. Biol. 2010, 19, 683–693. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).