Neurological Impact of Respiratory Viruses: Insights into Glial Cell Responses in the Central Nervous System
Abstract
1. Introduction
2. Glial Cells in the Central Nervous System
2.1. Microglia
2.2. Astrocytes
3. Respiratory Viruses and the Glial Cells of the CNS
3.1. Human Respiratory Syncytial Virus
3.2. Severe Acute Respiratory Syndrome Coronavirus 2
3.3. Influenza Virus
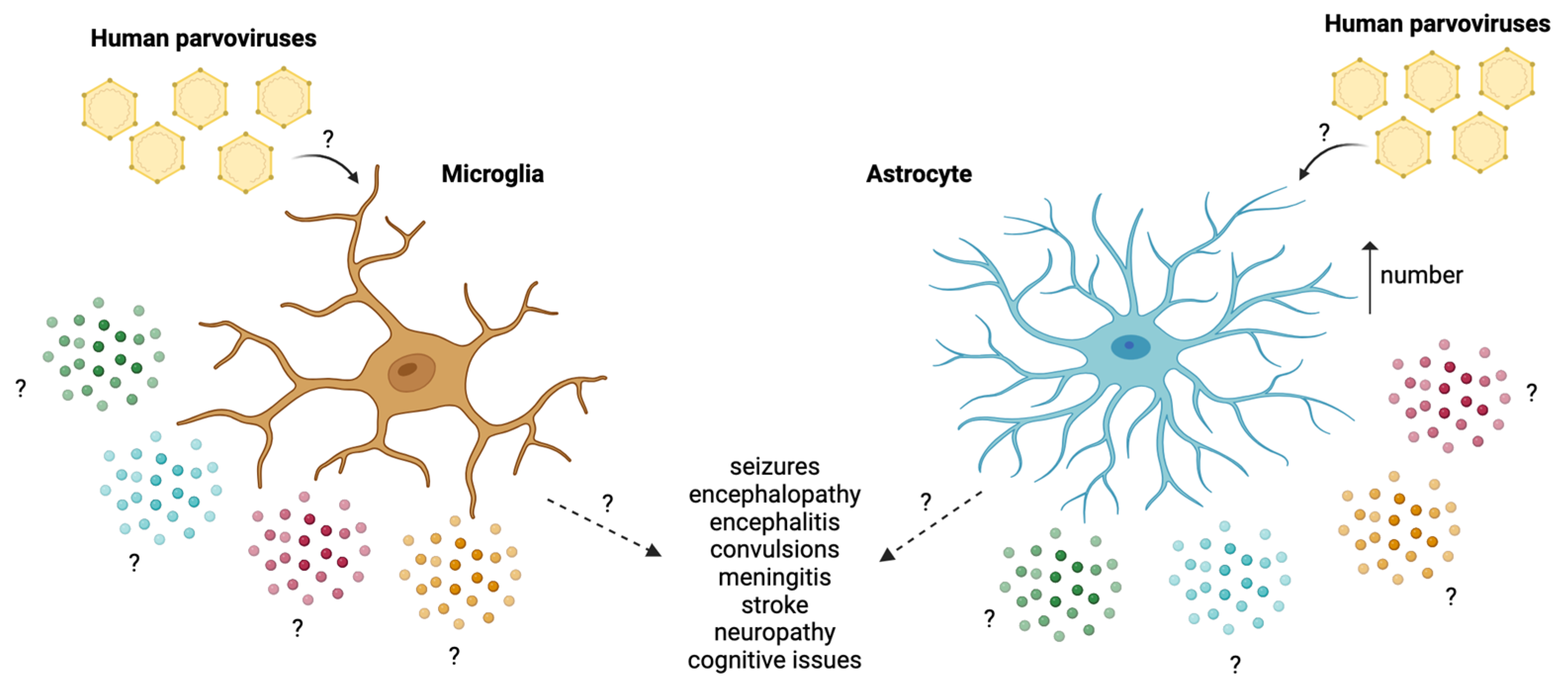
3.4. Human Parvoviruses
| Respiratory Virus | Parameter | Microglia | Astrocytes | References |
|---|---|---|---|---|
| hRSV | Receptor | CX3CR1? Nucleolin? EGFR? ICAM-1? HSPGs? | CX3CR1? Nucleolin? EGFR? ICAM-1? HSPGs? | [48,49,50,51,52,53,54,55,56] |
| Cytokine secretion | IL-1β | IL-4 IL-10 CCL2 IL-6 TNF-α | [46,58] | |
| Effect | Microglial activation M1 polarization ROS production Neuronal death | Astrocyte activation Increase BBB permeability | [46,57,58,59] | |
| SARS-CoV-2 | Receptor | ACE2? CD147? HSPGs? AXL? | ACE2? NRP-1 Eph? CD147? HSPGs? AXL? | [22,55,56,71,72,73,74,87,88,91] |
| Cytokine secretion | IL-1β IL-6 TNF-α | IL-6 TNF-α MCP1 MMP9 IL-17 CXCL10 CXCL6 CXCL1 CXCL2 | [63,78,86,88] | |
| Effect | Microgliosis M1 polarization ER stress responses Apoptosis Synapsis elimination IFN signaling upregulation TLR2 activation Neurodegeneration? | Astrogliosis BBB disruption Translocation to the piriform cortex Morphological changes Downregulation of CAMK2D, ERBB2, C1QL, SYPL1 Upregulation of IFN-1 Neuron cell death Decrease in metabolites of neural metabolism and function Increase in gene expression of apoptosis, DNA methylation, and neurodegeneration | [63,64,75,79,80,84] | |
| IVs | Receptor | SA-a 2,3-Gal SA-a 2,6-Gal EGFR? | SA-a 2,3-Gal SA-a 2,6-Gal EGFR? | [98,99,102] |
| Cytokine secretion | IFN-γ IFN-β IL-1β IL-6 MCP-1 TNF-α | IL-6 TNF-α IL-8 CCL2 IFN-β | [98,100,102,103] | |
| Effect | Microglial activation Increase in MHC I/II, F4/80, CD80 BBB impairment? Dysregulation of pruning of excessive synapses Alteration of neuronal morphology Decrease in BDNF expression Increase in NGF and OPN expression | Increase in apoptotic rate Increase in expression of caspase 3 and Bax Cytopathic effect | [98,100,101] | |
| Human Parvoviruses | Receptor | AXL? | AXL? | [74,91,125,126,127] |
| Cytokine secretion | Unknown | Unknown | ||
| Effect | Gliosis? Glial cell aggregates? | Increase in B19V-positive astrocytes Gliosis? Glial cell aggregates? | [112,128] |
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Jessen, K.R. Glial cells. Int. J. Biochem. Cell Biol. 2004, 36, 1861–1867. [Google Scholar] [CrossRef]
- Allen, N.J.; Lyons, D.A. Glia as architects of central nervous system formation and function. Science 2018, 362, 181–185. [Google Scholar] [CrossRef]
- Xu, S.; Lu, J.; Shao, A.; Zhang, J.H.; Zhang, J. Glial Cells: Role of the Immune Response in Ischemic Stroke. Front. Immunol. 2020, 11, 294. [Google Scholar] [CrossRef]
- Bohmwald, K.; Gálvez, N.M.S.; Ríos, M.; Kalergis, A.M. Neurologic alterations due to respiratory virus infections. Front. Cell. Neurosci. 2018, 12, 386. [Google Scholar] [CrossRef] [PubMed]
- Heegaard, E.D.; Brown, K.E. Human parvovirus B19. Clin. Microbiol. Rev. 2002, 15, 485–505. [Google Scholar] [CrossRef]
- Boncristiani, H.F.; Criado, M.F.; Arruda, E. Respiratory Viruses. In Encyclopedia of Microbiology, 3rd ed.; Academic Press: Cambridge, MA, USA, 2009; pp. 500–518. [Google Scholar] [CrossRef]
- Qiu, J.; Söderlund-Venermo, M.; Young, N.S. Human parvoviruses. Clin. Microbiol. Rev. 2017, 30, 43–113. [Google Scholar] [PubMed]
- Bhattacharya, S.; Agarwal, S.; Shrimali, N.M.; Guchhait, P. Interplay between hypoxia and inflammation contributes to the progression and severity of respiratory viral diseases. Mol. Aspects Med. 2021, 81, 101000. [Google Scholar] [CrossRef] [PubMed]
- Bohmwald, K.; Andrade, C.A.; Mora, V.P.; Muñoz, J.T.; Ramírez, R.; Rojas, M.F.; Kalergis, A.M. Neurotrophin Signaling Impairment by Viral Infections in the Central Nervous System. Int. J. Mol. Sci. 2022, 23, 5817. [Google Scholar] [CrossRef]
- Watanabe, T. Acute encephalitis and encephalopathy associated with human parvovirus B19 infection in children. World J. Clin. Pediatr. 2015, 4, 126. [Google Scholar] [CrossRef]
- Bohmwald, K.; Andrade, C.A.; Kalergis, A.M. Contribution of pro-inflammatory molecules induced by respiratory virus infections to neurological disorders. Pharmaceuticals 2021, 14, 340. [Google Scholar] [CrossRef]
- Jäkel, S.; Dimou, L. Glial Cells and Their Function in the Adult Brain: A Journey through the History of Their Ablation. Front. Cell. Neurosci. 2017, 11, 24. [Google Scholar] [CrossRef]
- Antel, J.P.; Becher, B.; Ludwin, S.K.; Prat, A.; Quintana, F.J. Glial Cells as Regulators of Neuroimmune Interactions in the Central Nervous System. J. Immunol. 2020, 204, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Nayak, D.; Roth, T.L.; McGavern, D.B. Microglia Development and function. Annu. Rev. Immunol. 2014, 32, 367. [Google Scholar] [CrossRef]
- Spiteri, A.G.; Wishart, C.L.; Pamphlett, R.; Locatelli, G.; King, N.J.C. Microglia and monocytes in inflammatory CNS disease: Integrating phenotype and function. Acta Neuropathol. 2022, 143, 179–224. [Google Scholar] [CrossRef]
- Borst, K.; Dumas, A.A.; Prinz, M. Microglia: Immune and non-immune functions. Immunity 2021, 54, 2194–2208. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Yu, Y.; Wu, P. Role of microglia in brain development after viral infection. Front. Cell Dev. Biol. 2024, 12, 1340308. [Google Scholar] [CrossRef]
- Yang, I.; Han, S.J.; Kaur, G.; Crane, C.; Parsa, A.T. The role of microglia in central nervous system immunity and glioma immunology. J. Clin. Neurosci. 2010, 17, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Fiebich, B.L.; Batista, C.R.A.; Saliba, S.W.; Yousif, N.M.; de Oliveira, A.C.P. Role of microglia TLRs in neurodegeneration. Front. Cell. Neurosci. 2018, 12, 329. [Google Scholar] [CrossRef]
- Olson, J.K.; Miller, S.D. Microglia Initiate Central Nervous System Innate and Adaptive Immune Responses through Multiple TLRs. J. Immunol. 2004, 173, 3916–3924. [Google Scholar] [CrossRef]
- Guo, S.; Wang, H.; Yin, Y. Microglia Polarization From M1 to M2 in Neurodegenerative Diseases. Front. Aging Neurosci. 2022, 14, 815347. [Google Scholar] [CrossRef]
- Tremblay, M.E.; Madore, C.; Bordeleau, M.; Tian, L.; Verkhratsky, A. Neuropathobiology of COVID-19: The Role for Glia. Front. Cell. Neurosci. 2020, 14, 592214. [Google Scholar] [CrossRef]
- Ransohoff, R.M. A polarizing question: Do M1 and M2 microglia exist. Nat. Neurosci. 2016, 19, 987–991. [Google Scholar] [CrossRef] [PubMed]
- Giovannoni, F.; Quintana, F.J. The Role of Astrocytes in CNS Inflammation. Trends Immunol. 2020, 41, 805–819. [Google Scholar] [CrossRef] [PubMed]
- Sofroniew, M.V.; Vinters, H.V. Astrocytes: Biology and pathology. Acta Neuropathol. 2009, 119, 7–35. [Google Scholar] [CrossRef] [PubMed]
- Deneen, B.; Akdemir, E.S.; Huang, A.Y.S. Astrocytogenesis: Where, when, and how. F1000Research 2020, 9, 233. [Google Scholar] [CrossRef]
- Filous, A.R.; Silver, J. Targeting astrocytes in CNS injury and disease: A translational research approach. Prog. Neurobiol. 2016, 144, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Lundgaard, I.; Osório, M.J.; Kress, B.T.; Sanggaard, S.; Nedergaard, M. White matter astrocytes in health and disease. Neuroscience 2014, 276, 161–173. [Google Scholar] [CrossRef]
- Dong, Y.; Benveniste, E.N. Immune function of astrocytes. Glia 2001, 36, 180–190. [Google Scholar] [CrossRef]
- Villablanca, C.; Vidal, R.; Gonzalez-Billault, C. Are cytoskeleton changes observed in astrocytes functionally linked to aging? Brain Res. Bull. 2023, 196, 59–67. [Google Scholar] [CrossRef]
- Lawrence, J.M.; Schardien, K.; Wigdahl, B.; Nonnemacher, M.R. Roles of neuropathology-associated reactive astrocytes: A systematic review. Acta Neuropathol. Commun. 2023, 11, 42. [Google Scholar] [CrossRef]
- Oberheim, N.A.; Goldman, S.A.; Nedergaard, M. Heterogeneity of Astrocytic Form and Function. Methods Mol. Biol. 2012, 814, 23. [Google Scholar] [CrossRef]
- Siracusa, R.; Fusco, R.; Cuzzocrea, S. Astrocytes: Role and functions in brain pathologies. Front. Pharmacol. 2019, 10, 1114. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.B.; Song, L.J.; Wang, Q.; Kumar, G.; Yan, Y.Q.; Ma, C.G. Astrocytes: A double-edged sword in neurodegenerative diseases. Neural Regen. Res. 2021, 16, 1702–1710. [Google Scholar] [PubMed]
- Rodgers, K.R.; Lin, Y.; Langan, T.J.; Iwakura, Y.; Chou, R.C. Innate Immune Functions of Astrocytes are Dependent Upon Tumor Necrosis Factor-Alpha. Sci. Rep. 2020, 10, 7047. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, R.K.; Balasubramani, G.K.; D’Agostino, H.E.A.; Clarke, L.; Yassin, M.; Middleton, D.B.; Silveira, F.P.; Wheeler, N.D.; Landis, J.; Peterson, A.; et al. Population-based hospitalization burden estimates for respiratory viruses, 2015–2019. Influenza Other Respi. Viruses 2022, 16, 1133–1140. [Google Scholar] [CrossRef] [PubMed]
- Bohmwald, K.; Andrade, C.A.; Gálvez, N.M.S.; Mora, V.P.; Muñoz, J.T.; Kalergis, A.M. The Causes and Long-Term Consequences of Viral Encephalitis. Front. Cell. Neurosci. 2021, 15, 755875. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, C.; Drews, S.J.; Marchant, D.J. Respiratory Syncytial Virus: Infection, Detection, and New Options for Prevention and Treatment. Am. Soc. Microbiol. 2017, 30, 277–319. [Google Scholar]
- Andrade, C.A.; Kalergis, A.M.; Bohmwald, K. Potential Neurocognitive Symptoms Due to Respiratory Syncytial Virus Infection. Pathogens 2022, 11, 47. [Google Scholar]
- Chen, L.; Cai, J.; Zhang, J.; Tang, Y.; Chen, J.; Xiong, S.; Li, Y.; Zhang, H.; Liu, Z.; Li, M.; et al. Respiratory syncytial virus co-opts hypoxia-inducible factor-1α-mediated glycolysis to favor the production of infectious virus. mBio 2023, 14, e0211023. [Google Scholar]
- Haeberle, H.A.; Dürrstein, C.; Rosenberger, P.; Hosakote, Y.M.; Kuhlicke, J.; Kempf, V.A.J.; Garofalo, R.P.; Eitzschig, H.K. Oxygen-independent stabilization of hypoxia inducible factor (HIF)-1 during RSV infection. PLoS ONE 2008, 3, 14–16. [Google Scholar] [CrossRef]
- Zhuang, X.; Gallo, G.; Sharma, P.; Ha, J.; Magri, A.; Borrmann, H.; Harris, J.M.; Tsukuda, S.; Bentley, E.; Kirby, A.; et al. Hypoxia inducible factors inhibit respiratory syncytial virus infection by modulation of nucleolin expression. iScience 2024, 27, 108763. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Kim, J.M.; Keum, H.R.; Kim, S.W.; Baek, H.S.; Byun, J.C.; Kim, Y.K.; Kim, S.; Lee, J.M. Seasonal Trends in the Prevalence and Incidence of Viral Encephalitis in Korea (2015–2019). J. Clin. Med. 2023, 12, 2003. [Google Scholar] [CrossRef] [PubMed]
- Saravanos, G.L.; King, C.L.; Deng, L.; Dinsmore, N.; Ramos, I.; Takashima, M.; Crawford, N.; Clark, J.E.; Dale, R.C.; Jones, C.A.; et al. Respiratory Syncytial Virus–Associated Neurologic Complications in Children: A Systematic Review and Aggregated Case Series. J. Pediatr. 2021, 239, 39–49.e9. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, Z.; Wei, J.; Yang, Z.; Ren, L.; Deng, Y.; Chen, S.; Zang, N.; Liu, E. Exploring Key Genes and Mechanisms in Respiratory Syncytial Virus-Infected BALB/c Mice via Multi-Organ Expression Profiles. Front. Cell. Infect. Microbiol. 2022, 12, 858305. [Google Scholar] [CrossRef] [PubMed]
- Bohmwald, K.; Soto, J.A.; Andrade-Parra, C.; Fernández-Fierro, A.; Espinoza, J.A.; Ríos, M.; Eugenin, E.A.; González, P.A.; Opazo, M.C.; Riedel, C.A.; et al. Lung pathology due to hRSV infection impairs blood–brain barrier permeability enabling astrocyte infection and a long-lasting inflammation in the CNS. Brain. Behav. Immun. 2021, 91, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Morichi, S.; Kawashima, H.; Ioi, H.; Yamanaka, G.; Kashiwagi, Y.; Hoshika, A.; Nakayama, T.; Watanabe, Y. Classification of acute encephalopathy in respiratory syncytial virus infection. J. Infect. Chemother. 2011, 17, 776–781. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Xu, L.; Xie, Z. Receptors for Respiratory Syncytial Virus Infection and Host Factors Regulating the Life Cycle of Respiratory Syncytial Virus. Front. Cell. Infect. Microbiol. 2022, 12, 858629. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, D.; Nakamura, T.; Koike, M.; Hirano, K.; Miki, Y.; Beppu, M. Shuttling protein nucleolin is a microglia receptor for amyloid beta peptide 1-42. Biol. Pharm. Bull. 2013, 36, 1587–1593. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Müller, N. The role of intercellular adhesion molecule-1 in the pathogenesis of psychiatric disorders. Front. Pharmacol. 2019, 10, 1251. [Google Scholar] [CrossRef]
- Vartak-Sharma, N.; Ghorpade, A. Astrocyte elevated gene-1 regulates astrocyte responses to neural injury: Implications for reactive astrogliosis and neurodegeneration. J. Neuroinflamm. 2012, 9, 195. [Google Scholar] [CrossRef]
- Meucci, O.; Fatatis, A.; Simen, A.A.; Miller, R.J. Expression of CX3CR1 chemokine receptors on neurons and their role in neuronal survival. Proc. Natl. Acad. Sci. USA 2000, 97, 8075–8080. [Google Scholar] [CrossRef] [PubMed]
- Romano, R.; Bucci, C. Role of EGFR in the Nervous System. Cells 2020, 9, 1887. [Google Scholar] [CrossRef] [PubMed]
- Qu, W.S.; Tian, D.S.; Guo, Z.B.; Fang, J.; Zhang, Q.; Yu, Z.Y.; Xie, M.J.; Zhang, H.Q.; Lü, J.G.; Wang, W. Inhibition of EGFR/MAPK signaling reduces microglial inflammatory response and the associated secondary damage in rats after spinal cord injury. J. Neuroinflamm. 2012, 9, 178. [Google Scholar] [CrossRef] [PubMed]
- Tselnicker, I.F.; Boisvert, M.M.; Allen, N.J. The role of neuronal versus astrocyte-derived heparan sulfate proteoglycans in brain development and injury. Biochem. Soc. Trans. 2014, 42, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, P.; Zhang, X.; Li, J.P. Heparan Sulfate Proteoglycans as Relays of Neuroinflammation. J. Histochem. Cytochem. 2018, 66, 305–319. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Bajinka, O.; Tang, Z.; Qiu, X.; Tan, Y. Lung–brain axis: Metabolomics and pathological changes in lungs and brain of respiratory syncytial virus-infected mice. J. Med. Virol. 2022, 94, 5885–5893. [Google Scholar] [CrossRef]
- Bajinka, O.; Tang, Z.; Mao, Y.; Qiu, X.; Darboe, A.; Tan, Y. Respiratory syncytial virus infection disrupts pulmonary microbiota to induce microglia phenotype shift. J. Med. Virol. 2023, 95, e28976. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Zhang, X.C.; Yu, H.Y.; Wang, Y.; Chen, J.; Wang, Y.; Yu, L.; Zhu, G.X.; Cao, X.J.; Huang, S.H. Respiratory syncytial virus infection of microglia exacerbates SH-SY5Y neuronal cell injury by inducing the secretion of inflammatory cytokines: A Transwell in vitro study. Iran. J. Basic Med. Sci. 2021, 24, 213–221. [Google Scholar] [CrossRef]
- Gusev, E.; Sarapultsev, A.; Solomatina, L.; Chereshnev, V. SARS-CoV-2-Specific Immune Response and the Pathogenesis of COVID-19. Int. J. Mol. Sci. 2022, 23, 1716. [Google Scholar] [CrossRef]
- Pal, M.; Berhanu, G.; Desalegn, C.; Kandi, V. Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2): An Update. Cureus 2020, 12, e7423. [Google Scholar] [CrossRef]
- Lamers, M.M.; Haagmans, B.L. SARS-CoV-2 pathogenesis. Nat. Rev. Microbiol. 2022, 20, 270–284. [Google Scholar] [CrossRef]
- Jeong, G.U.; Lyu, J.; Kim, K.-D.; Chung, Y.C.; Yoon, G.Y.; Lee, S.; Hwang, I.; Shin, W.-H.; Ko, J.; Lee, J.-Y.; et al. SARS-CoV-2 Infection of Microglia Elicits Proinflammatory Activation and Apoptotic Cell Death. Microbiol. Spectr. 2022, 10, e0109122. [Google Scholar] [CrossRef] [PubMed]
- Samudyata; Oliveira, A.O.; Malwade, S.; Rufino de Sousa, N.; Goparaju, S.K.; Gracias, J.; Orhan, F.; Steponaviciute, L.; Schalling, M.; Sheridan, S.D.; et al. SARS-CoV-2 promotes microglial synapse elimination in human brain organoids. Mol. Psychiatry 2022, 27, 3939–3950. [Google Scholar] [CrossRef]
- Jahani, M.; Dokaneheifard, S.; Mansouri, K. Hypoxia: A key feature of COVID-19 launching activation of HIF-1 and cytokine storm. J. Inflamm. 2020, 17, 33. [Google Scholar] [CrossRef]
- Luo, Z.; Tian, M.; Yang, G.; Tan, Q.; Chen, Y.; Li, G.; Zhang, Q.; Li, Y.; Wan, P.; Wu, J. Hypoxia signaling in human health and diseases: Implications and prospects for therapeutics. Signal Transduct. Target. Ther. 2022, 7, 218. [Google Scholar] [CrossRef] [PubMed]
- Rutkai, I.; Mayer, M.G.; Hellmers, L.M.; Ning, B.; Huang, Z.; Monjure, C.J.; Coyne, C.; Silvestri, R.; Golden, N.; Hensley, K.; et al. Neuropathology and virus in brain of SARS-CoV-2 infected non-human primates. Nat. Commun. 2022, 13, 1745. [Google Scholar] [CrossRef] [PubMed]
- Adingupu, D.D.; Soroush, A.; Hansen, A.; Twomey, R.; Dunn, J.F. Brain hypoxia, neurocognitive impairment, and quality of life in people post-COVID-19. J. Neurol. 2023, 270, 3303–3314. [Google Scholar] [CrossRef] [PubMed]
- Leng, A.; Shah, M.; Ahmad, S.A.; Premraj, L.; Wildi, K.; Li Bassi, G.; Pardo, C.A.; Choi, A.; Cho, S.M. Pathogenesis Underlying Neurological Manifestations of Long COVID Syndrome and Potential Therapeutics. Cells 2023, 12, 816. [Google Scholar] [CrossRef] [PubMed]
- Zalpoor, H.; Akbari, A.; Samei, A.; Forghaniesfidvajani, R.; Kamali, M.; Afzalnia, A.; Manshouri, S.; Heidari, F.; Pornour, M.; Khoshmirsafa, M.; et al. The roles of Eph receptors, neuropilin-1, P2X7, and CD147 in COVID-19-associated neurodegenerative diseases: Inflammasome and JaK inhibitors as potential promising therapies. Cell. Mol. Biol. Lett. 2022, 27, 10. [Google Scholar] [CrossRef]
- Steardo, L.; Steardo, L.; Scuderi, C. Astrocytes and the Psychiatric Sequelae of COVID-19: What We Learned from the Pandemic. Neurochem. Res. 2023, 48, 1015–1025. [Google Scholar] [CrossRef]
- Awogbindin, I.O.; Ben-Azu, B.; Olusola, B.A.; Akinluyi, E.T.; Adeniyi, P.A.; Di Paolo, T.; Tremblay, M.È. Microglial Implications in SARS-CoV-2 Infection and COVID-19: Lessons From Viral RNA Neurotropism and Possible Relevance to Parkinson’s Disease. Front. Cell. Neurosci. 2021, 15, 670298. [Google Scholar] [CrossRef]
- Yao, C.; Liu, X.; Tang, Y.; Wang, C.; Duan, C.; Liu, X.; Chen, M.; Zhou, Y.; Tang, E.; Xiang, Y.; et al. Lipopolysaccharide induces inflammatory microglial activation through CD147-mediated matrix metalloproteinase expression. Environ. Sci. Pollut. Res. 2023, 30, 35352–35365. [Google Scholar] [CrossRef]
- Huang, Y.; Happonen, K.E.; Burrola, P.G.; O’Connor, C.; Hah, N.; Huang, L.; Nimmerjahn, A.; Lemke, G. Microglia use TAM receptors to detect and engulf amyloid β plaques. Nat. Immunol. 2021, 22, 586–594. [Google Scholar] [CrossRef]
- Dey, R.; Bishayi, B. Microglial Inflammatory Responses to SARS-CoV-2 Infection: A Comprehensive Review. Cell. Mol. Neurobiol. 2024, 44, 2. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, X.; Jia, L.; Zhang, Y. Potential mechanism of SARS-CoV-2-associated central and peripheral nervous system impairment. Acta Neurol. Scand. 2022, 146, 225–236. [Google Scholar] [CrossRef]
- Ferraro, E.; Germanò, M.; Mollace, R.; Mollace, V.; Malara, N. HIF-1, the Warburg Effect, and Macrophage/Microglia Polarization Potential Role in COVID-19 Pathogenesis. Oxid. Med. Cell. Longev. 2021, 2021, 8841911. [Google Scholar] [CrossRef]
- Olivarria, G.M.; Cheng, Y.; Furman, S.; Pachow, C.; Hohsfield, L.A.; Smith-Geater, C.; Miramontes, R.; Wu, J.; Burns, M.S.; Tsourmas, K.I.; et al. Microglia Do Not Restrict SARS-CoV-2 Replication following Infection of the Central Nervous System of K18-Human ACE2 Transgenic Mice. J. Virol. 2022, 96, e0196921. [Google Scholar] [CrossRef]
- Savelieff, M.G.; Feldman, E.L.; Stino, A.M. Neurological sequela and disruption of neuron-glia homeostasis in SARS-CoV-2 infection. Neurobiol. Dis. 2022, 168, 293. [Google Scholar] [CrossRef]
- Albornoz, E.A.; Amarilla, A.A.; Modhiran, N.; Parker, S.; Li, X.X.; Wijesundara, D.K.; Aguado, J.; Zamora, A.P.; McMillan, C.L.D.; Liang, B.; et al. SARS-CoV-2 drives NLRP3 inflammasome activation in human microglia through spike protein. Mol. Psychiatry 2022, 28, 2878–2893. [Google Scholar] [CrossRef]
- Garber, C.; Soung, A.; Vollmer, L.L.; Kanmogne, M.; Last, A.; Brown, J.; Klein, R.S. T cells promote microglia-mediated synaptic elimination and cognitive dysfunction during recovery from neuropathogenic flaviviruses. Nat. Neurosci. 2019, 22, 1276. [Google Scholar] [CrossRef]
- Su, W.; Ju, J.; Gu, M.; Wang, X.; Liu, S.; Yu, J.; Mu, D. SARS-CoV-2 envelope protein triggers depression-like behaviors and dysosmia via TLR2-mediated neuroinflammation in mice. J. Neuroinflamm. 2023, 20, 110. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, W.; Chen, F.; Cui, L. COVID-19 and cognitive impairment: Neuroinvasive and blood–brain barrier dysfunction. J. Neuroinflamm. 2022, 19, 222. [Google Scholar] [CrossRef]
- Beckman, D.; Bonillas, A.; Diniz, G.B.; Ott, S.; Roh, J.W.; Elizaldi, S.R.; Schmidt, B.A.; Sammak, R.L.; Van Rompay, K.K.A.; Iyer, S.S.; et al. SARS-CoV-2 infects neurons and induces neuroinflammation in a non-human primate model of COVID-19. Cell Rep. 2022, 41, 111573. [Google Scholar] [CrossRef]
- Kempuraj, D.; Aenlle, K.K.; Cohen, J.; Mathew, A.; Isler, D.; Pangeni, R.P.; Nathanson, L.; Theoharides, T.C.; Klimas, N.G. COVID-19 and Long COVID: Disruption of the Neurovascular Unit, Blood-Brain Barrier, and Tight Junctions. Neuroscientist 2023, 11, 10738584231194927. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, L.; Bao, L.; Liu, J.; Zhu, H.; Lv, Q.; Liu, R.; Chen, W.; Tong, W.; Wei, Q.; et al. SARS-CoV-2 crosses the blood–brain barrier accompanied with basement membrane disruption without tight junctions alteration. Signal Transduct. Target. Ther. 2021, 6, 337. [Google Scholar] [CrossRef]
- Malik, J.R.; Acharya, A.; Avedissian, S.N.; Byrareddy, S.N.; Fletcher, C.V.; Podany, A.T.; Dyavar, S.R. ACE-2, TMPRSS2, and Neuropilin-1 Receptor Expression on Human Brain Astrocytes and Pericytes and SARS-CoV-2 Infection Kinetics. Int. J. Mol. Sci. 2023, 24, 8622. [Google Scholar] [CrossRef]
- Kong, W.; Montano, M.; Corley, M.J.; Helmy, E.; Kobayashi, H.; Kinisu, M.; Suryawanshi, R.; Luo, X.; Royer, L.A.; Roan, N.R.; et al. Neuropilin-1 Mediates SARS-CoV-2 Infection of Astrocytes in Brain Organoids, Inducing Inflammation Leading to Dysfunction and Death of Neurons. MBio 2022, 13, e0230822. [Google Scholar] [CrossRef]
- Crunfli, F.; Carregari, V.C.; Veras, F.P.; Silva, L.S.; Henrique, M.; Mayara, E.; Firmino, S.; Paiva, I.M. Morphological, cellular, and molecular basis of brain infection in COVID-19 patients. Proc. Natl. Acad. Sci. USA 2022, 119, e2200960119. [Google Scholar] [CrossRef]
- Patrizz, A.; Doran, S.J.; Chauhan, A.; Ahnstedt, H.; Roy-O’Reilly, M.; Lai, Y.J.; Weston, G.; Tarabishy, S.; Patel, A.R.; Verma, R.; et al. EMMPRIN/CD147 plays a detrimental role in clinical and experimental ischemic stroke. Aging 2020, 12, 5121–5139. [Google Scholar] [CrossRef]
- Meertens, L.; Labeau, A.; Dejarnac, O.; Cipriani, S.; Sinigaglia, L.; Bonnet-Madin, L.; Le Charpentier, T.; Hafirassou, M.L.; Zamborlini, A.; Cao-Lormeau, V.M.; et al. Axl Mediates ZIKA Virus Entry in Human Glial Cells and Modulates Innate Immune Responses. Cell Rep. 2017, 18, 324–333. [Google Scholar] [CrossRef]
- Mifsud, E.J.; Kuba, M.; Barr, I.G. Innate immune responses to influenza virus infections in the upper respiratory tract. Viruses 2021, 13, 2090. [Google Scholar] [CrossRef]
- Ekstrand, J.J. Neurologic Complications of Influenza. Semin. Pediatr. Neurol. 2012, 19, 96–100. [Google Scholar] [CrossRef]
- Guo, X.; Zhu, Z.; Zhang, W.; Meng, X.; Zhu, Y.; Han, P.; Zhou, X.; Hu, Y.; Wang, R. Nuclear translocation of HIF-1α induced by influenza A (H1N1) infection is critical to the production of proinflammatory cytokines. Emerg. Microbes Infect. 2017, 6, e39. [Google Scholar] [CrossRef]
- Zhao, C.; Chen, J.; Cheng, L.; Xu, K.; Yang, Y.; Su, X. Deficiency of HIF-1α enhances influenza A virus replication by promoting autophagy in alveolar type II epithelial cells. Emerg. Microbes Infect. 2020, 9, 691–706. [Google Scholar] [CrossRef]
- Fugate, J.E.; Lam, E.M.; Rabinstein, A.A.; Wijdicks, E.F.M. Acute hemorrhagic leukoencephalitis and hypoxic brain injury associated with H1N1 influenza. Arch. Neurol. 2010, 67, 756–758. [Google Scholar] [CrossRef]
- Wang, K.Y.; Singer, H.S.; Crain, B.; Gujar, S.; Lin, D.D.M. Hypoxic-ischemic encephalopathy mimicking acute necrotizing encephalopathy. Pediatr. Neurol. 2015, 52, 110–114. [Google Scholar] [CrossRef]
- Ding, X.M.; Wang, Y.F.; Lyu, Y.; Zou, Y.; Wang, X.; Ruan, S.M.; Wu, W.H.; Liu, H.; Sun, Y.; Zhang, R.L.; et al. The effect of influenza A (H1N1) pdm09 virus infection on cytokine production and gene expression in BV2 microglial cells. Virus Res. 2022, 312, 198716. [Google Scholar] [CrossRef]
- Sieben, C.; Sezgin, E.; Eggeling, C.; Manley, S. Influenza a viruses use multivalent sialic acid clusters for cell binding and receptor activation. PLoS Pathog. 2020, 16, e1008656. [Google Scholar] [CrossRef]
- Düsedau, H.P.; Steffen, J.; Figueiredo, C.A.; Boehme, J.D.; Schultz, K.; Erck, C.; Korte, M.; Faber-Zuschratter, H.; Smalla, K.H.; Dieterich, D.; et al. Influenza A Virus (H1N1) Infection Induces Microglial Activation and Temporal Dysbalance in Glutamatergic Synaptic Transmission. mBio 2021, 12, e0177621. [Google Scholar] [CrossRef]
- Pei, X.D.; Zhai, Y.F.; Zhang, H.H. Influenza virus H1N1 induced apoptosis of mouse astrocytes and the effect on protein expression. Asian Pac. J. Trop. Med. 2014, 7, 572–575. [Google Scholar] [CrossRef]
- Ng, Y.P.; Lee, S.M.Y.; Cheung, T.K.W.; Nicholls, J.M.; Peiris, J.S.M.; Ip, N.Y. Avian influenza H5N1 virus induces cytopathy and proinflammatory cytokine responses in human astrocytic and neuronal cell lines. Neuroscience 2010, 168, 613–623. [Google Scholar] [CrossRef]
- Ng, Y.P.; Yip, T.F.; Peiris, J.S.M.; Ip, N.Y.; Lee, S.M.Y. Avian influenza A H7N9 virus infects human astrocytes and neuronal cells and induces inflammatory immune responses. J. Neurovirol. 2018, 24, 752–760. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Qiu, Q.; Zhou, Y.; Zhang, Y.; Xu, W.; Cui, A.; Li, X. The epidemiological and genetic characteristics of human parvovirus B19 in patients with febrile rash illnesses in China. Sci. Rep. 2023, 13, 15913. [Google Scholar] [CrossRef] [PubMed]
- Dittmer, F.P.; de Guimarães, C.M.; Peixoto, A.B.; Pontes, K.F.M.; Bonasoni, M.P.; Tonni, G.; Araujo Júnior, E. Parvovirus B19 Infection and Pregnancy: Review of the Current Knowledge. J. Pers. Med. 2024, 14, 139. [Google Scholar] [CrossRef]
- Zou, Q.; Chen, P.; Chen, J.; Chen, D.; Xia, H.; Chen, L.; Feng, H.; Feng, L. Multisystem Involvement Induced by Human Parvovirus B19 Infection in a Non-immunosuppressed Adult: A Case Report. Front. Med. 2022, 9, 808205. [Google Scholar] [CrossRef]
- Pichon, M.; Labois, C.; Tardy-Guidollet, V.; Mallet, D.; Casalegno, J.S.; Billaud, G.; Lina, B.; Gaucherand, P.; Mekki, Y. Optimized nested PCR enhances biological diagnosis and phylogenetic analysis of human parvovirus B19 infections. Arch. Virol. 2019, 164, 2775–2781. [Google Scholar] [CrossRef]
- Pillet, S.; Le Guyader, N.; Hofer, T.; Nguyenkhac, F.; Koken, M.; Aubin, J.T.; Fichelson, S.; Gassmann, M.; Morinet, F. Hypoxia enhances human B19 erythrovirus gene expression in primary erythroid cells. Virology 2004, 327, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Leskinen, K.; Gritti, T.; Groma, V.; Arola, J.; Lepistö, A.; Sipponen, T.; Saavalainen, P.; Söderlund-Venermo, M. Prevalence, Cell Tropism, and Clinical Impact of Human Parvovirus Persistence in Adenomatous, Cancerous, Inflamed, and Healthy Intestinal Mucosa. Front. Microbiol. 2022, 13, 914181. [Google Scholar] [CrossRef]
- Barah, F.; Whiteside, S.; Batista, S.; Morris, J. Neurological aspects of human parvovirus B19 infection: A systematic review. Rev. Med. Virol. 2014, 24, 154–168. [Google Scholar] [CrossRef]
- Pattabiraman, C.; Prasad, P.; Sudarshan, S.; George, A.K.; Sreenivas, D.; Rasheed, R.; Ghosh, A.; Pal, A.; Hameed, S.K.S.; Bandyopadhyay, B.; et al. Identification and Genomic Characterization of Parvovirus B19V Genotype 3 Viruses from Cases of Meningoencephalitis in West Bengal, India. Microbiol. Spectr. 2022, 10, e0225121. [Google Scholar] [CrossRef]
- Skuja, S.; Vilmane, A.; Svirskis, S.; Groma, V.; Murovska, M. Evidence of human parvovirus B19 infection in the post-mortem brain tissue of the elderly. Viruses 2018, 10, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Isumi, H.; Nunoue, T.; Nishida, A.; Takashima, S. Fetal brain infection with human parvovirus B19. Pediatr. Neurol. 1999, 21, 661–663. [Google Scholar] [CrossRef] [PubMed]
- Manning, A.; Willey, S.J.; Bell, J.E.; Simmonds, P. Comparison of tissue distribution, persistence, and molecular epidemiology of parvovirus B19 and novel human parvoviruses PARV4 and human bocavirus. J. Infect. Dis. 2007, 195, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, J.A. Detection of adeno-associated virus 2 and parvovirus B19 in the human dorsolateral prefrontal cortex. J. Neurovirol. 2006, 12, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Trapani, S.; Caporizzi, A.; Ricci, S.; Indolfi, G. Human Bocavirus in Childhood: A True Respiratory Pathogen or a “Passenger” Virus? A Comprehensive Review. Microorganisms 2023, 11, 1243. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.M.; Chen, Q.Q.; Hao, Y.X.; Yu, T.; Zeng, S.Z.; Wu, X.B.; Zhang, B.; Duan, Z.J. Identification of human bocaviruses in the cerebrospinal fluid of children hospitalized with encephalitis in China. J. Clin. Virol. 2013, 57, 374–377. [Google Scholar] [CrossRef] [PubMed]
- Mitui, M.T.; Shahnawaz Bin Tabib, S.M.; Matsumoto, T.; Khanam, W.; Ahmed, S.; Mori, D.; Akhter, N.; Yamada, K.; Kabir, L.; Nishizono, A.; et al. Detection of human bocavirus in the cerebrospinal fluid of children with encephalitis. Clin. Infect. Dis. 2012, 54, 964–967. [Google Scholar] [CrossRef] [PubMed]
- Ergul, A.B.; Altug, U.; Aydin, K.; Guven, A.S.; Altuner Torun, Y. Acute necrotizing encephalopathy causing human bocavirus. Neuroradiol. J. 2017, 30, 164–167. [Google Scholar] [CrossRef]
- Akturk, H.; Sidotlessk, G.; Salman, N.; Sutcu, M.; Tatli, B.; Akcay Ciblak, M.; Bulent Erol, O.; Hancerli Torun, S.; Citak, A.; Somer, A. Atypical presentation of human bocavirus: Severe respiratory tract infection complicated with encephalopathy. J. Med. Virol. 2015, 87, 1831–1838. [Google Scholar] [CrossRef]
- Vilmane, A.; Terentjeva, A.; Tamosiunas, P.L.; Suna, N.; Suna, I.; Petraityte-burneikiene, R.; Murovska, M.; Rasa-dzelzkaleja, S.; Nora- Krukle, Z. Human parvoviruses may affect the development and clinical course of meningitis and meningoencephalitis. Brain Sci. 2020, 10, 339. [Google Scholar] [CrossRef]
- Thapa, R.R.; Plentz, A.; Edinger, M.; Wolff, D.; Angstwurm, K.; Söderlund-Venermo, M. Human bocavirus 1 respiratory tract reactivations or reinfections in two adults, contributing to neurological deficits and death. Access Microbiol. 2021, 3, 237. [Google Scholar] [CrossRef]
- Mohammadi, M. HBoV-1: Virus structure, genomic features, life cycle, pathogenesis, epidemiology, diagnosis and clinical manifestations. Front. Cell. Infect. Microbiol. 2023, 13, 1198127. [Google Scholar] [CrossRef]
- Agungpriyono, D.R.; Uchida, K.; Tabaru, H.; Yamaguchi, R.; Tateyama, S. Subacute massive necrotizing myocarditis by canine parvovirus type 2 infection with diffuse leukoencephalomalacia in a puppy. Vet. Pathol. 1999, 36, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Shen, W.; Wang, S.; Qiu, J. Recent Advances in Molecular Biology of Human Bocavirus 1 and Its Applications. Front. Microbiol. 2021, 12, 696604. [Google Scholar] [CrossRef]
- Luo, Y.; Qiu, J. Human parvovirus B19: A mechanistic overview of infection and DNA replication. Future Virol. 2015, 10, 155–167. [Google Scholar] [CrossRef]
- Ning, K.; Zou, W.; Xu, P.; Cheng, F.; Zhang, E.Y.; Zhang-Chen, A.; Kleiboeker, S.; Qiu, J. Identification of AXL as a co-receptor for human parvovirus B19 infection of human erythroid progenitors. Sci. Adv. 2023, 9, ade0869. [Google Scholar] [CrossRef] [PubMed]
- Piewbang, C.; Wardhani, S.W.; Dankaona, W.; Lacharoje, S.; Chai-In, P.; Yostawonkul, J.; Chanseanroj, J.; Boonrungsiman, S.; Kasantikul, T.; Poovorawan, Y.; et al. Canine bocavirus-2 infection and its possible association with encephalopathy in domestic dogs. PLoS ONE 2021, 16, 0255425. [Google Scholar] [CrossRef]
- Schildgen, O.; Schildgen, V. The Role of the Human Bocavirus (HBoV) in Respiratory Infections. Adv. Tech. Diagn. Microbiol. 2018, 2, 281. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mora, V.P.; Kalergis, A.M.; Bohmwald, K. Neurological Impact of Respiratory Viruses: Insights into Glial Cell Responses in the Central Nervous System. Microorganisms 2024, 12, 1713. https://doi.org/10.3390/microorganisms12081713
Mora VP, Kalergis AM, Bohmwald K. Neurological Impact of Respiratory Viruses: Insights into Glial Cell Responses in the Central Nervous System. Microorganisms. 2024; 12(8):1713. https://doi.org/10.3390/microorganisms12081713
Chicago/Turabian StyleMora, Valentina P., Alexis M. Kalergis, and Karen Bohmwald. 2024. "Neurological Impact of Respiratory Viruses: Insights into Glial Cell Responses in the Central Nervous System" Microorganisms 12, no. 8: 1713. https://doi.org/10.3390/microorganisms12081713
APA StyleMora, V. P., Kalergis, A. M., & Bohmwald, K. (2024). Neurological Impact of Respiratory Viruses: Insights into Glial Cell Responses in the Central Nervous System. Microorganisms, 12(8), 1713. https://doi.org/10.3390/microorganisms12081713







