1. Introduction
Anhydrobiosis is one of the special forms of cryptobiosis, in which the yeasts survive through adverse environmental conditions, in this case, the absence of water (desiccation). To survive, cells adapt to the changed environment, temporarily suspending their metabolism [
1,
2,
3]. Understanding of the mechanism of anhydrobiosis and the main determinants important for cell viability after transfer to the state of anhydrobiosis and knowledge of events during the subsequent processes of rehydration and reactivation are important for fundamental science and could be applied in biomedicine and biotechnology. In biomedicine, an understanding of anhydrobiosis is important for research in translational medicine and the production of new vaccines. Further improvement of the quality of dry biopreparations is needed in biotechnology, including active dry yeasts for wine, beer, and ethanol production, as well as for the production of various biologically active compounds [
4,
5]. Big companies provide high-quality preparations of dry baker’s yeast with a very high level of cell viability. At the same time, such a result still cannot be achieved for other yeast species, as well as for other
S. cerevisiae strains necessary for practical goals. One of the approaches to understand the reasons for these differences is the comparison of yeasts demonstrating various resistance patterns during dehydration–rehydration treatment. Previous studies showed [
6], that osmotolerant halophilic yeasts
Debaryomyces hansenii are more resistant to dehydration compared to
S. cerevisiae. Being in the exponential phase of growth, these cells can effectively transit into anhydrobiosis, when other actively growing yeast species are very sensitive to such treatment. Further research revealed essential differences in the chemical composition of their plasma membranes, which led to a lower phase transition temperature of the membrane lipids [
7,
8]. In another study, it was revealed that thermotolerant strains of S. cerevisiae are more resistant to dehydration compared to mesophilic strains [
8]. Recently, it was shown [
9] that psychrophilic yeast species are also more resistant to dehydration than mesophilic ones. These results mean that yeast strains and species able to survive in more extreme environmental conditions also possess higher resistance to dehydration–rehydration treatment. Therefore, we expected that continuation of the studies directed at the comparison of strains with different resistances to dehydration–rehydration treatment may lead to information that is important for general biology, as well as facilitate the development of technologies for the improvement of the resistance of sensitive strains, which is important for current biotechnology.
The purpose of this study was to compare mesophilic and thermotolerant strains of S. cerevisiae to determine the sequence of events in the energy metabolism of these cells during dehydration and the regeneration of these processes during rehydration. The intensities of respiration and acidification of the medium, the amount of phenyldicarbaundecaborane (PCB−) bound to cellular membranes, and the capability of cells to accumulate K+ were assayed using an electrochemical monitoring system. The results of our experiments indicated that very resistant to desiccation strain 77 cells show respiration activity after 22 h of drying, demonstrate a stronger plasma membrane barrier to lipophilic anions, and maintain a higher content of intracellular ATP, although mesophilic strain 14 cells maintained the K+ gradient for longer during desiccation and more strongly acidified the incubation medium. It was concluded that the higher viability of rehydrated strain 77 cells could be due to the higher stability of their mitochondria. These results advance our understanding of the impact of dehydration on energy metabolism in yeasts.
4. Discussion
The main goal of this study was to determine factors important for the survival of yeast cells during their transition into the state of anhydrobiosis and out of it, with the aim to analyze the possibility of them having highly active biological systems after reactivation of the dry cells. It is important to enrich our knowledge about the main biochemical mechanisms working during the process of transition. Such findings would be key to improving methods for yeast preservation and to understanding the mechanisms of anhydrobiosis. Considering that yeasts are an optimal model of eukaryotic cells and are widely used for various studies in the fields of molecular biology, translational medicine, and pharmaceutics, they have more general importance, including the treatment of pathological states of the human body.
During desiccation, yeast cells face mechanical, structural, and oxidative challenges, including intracellular crowding, plasma membrane lysis, and permeabilization. To survive these stresses, yeasts have developed many endogenous protective mechanisms, including a unique elastic cell wall, the accumulation of intracellular glycerol and trehalose, the induction of stress proteins, and biochemical antioxidative systems [
2,
15].
It is still not clear why strain 77 cells are more resistant to desiccation than strain 14 ones. Most probably, there is a complex of reasons. In addition, resistance to desiccation depends on the growth phase. For the experiments, we used cells from the stationary growth phase. Such cells show better adaptation to the environment [
3,
5]. Results of the monitoring of respiration activities indicated the differences in behavior of the studied
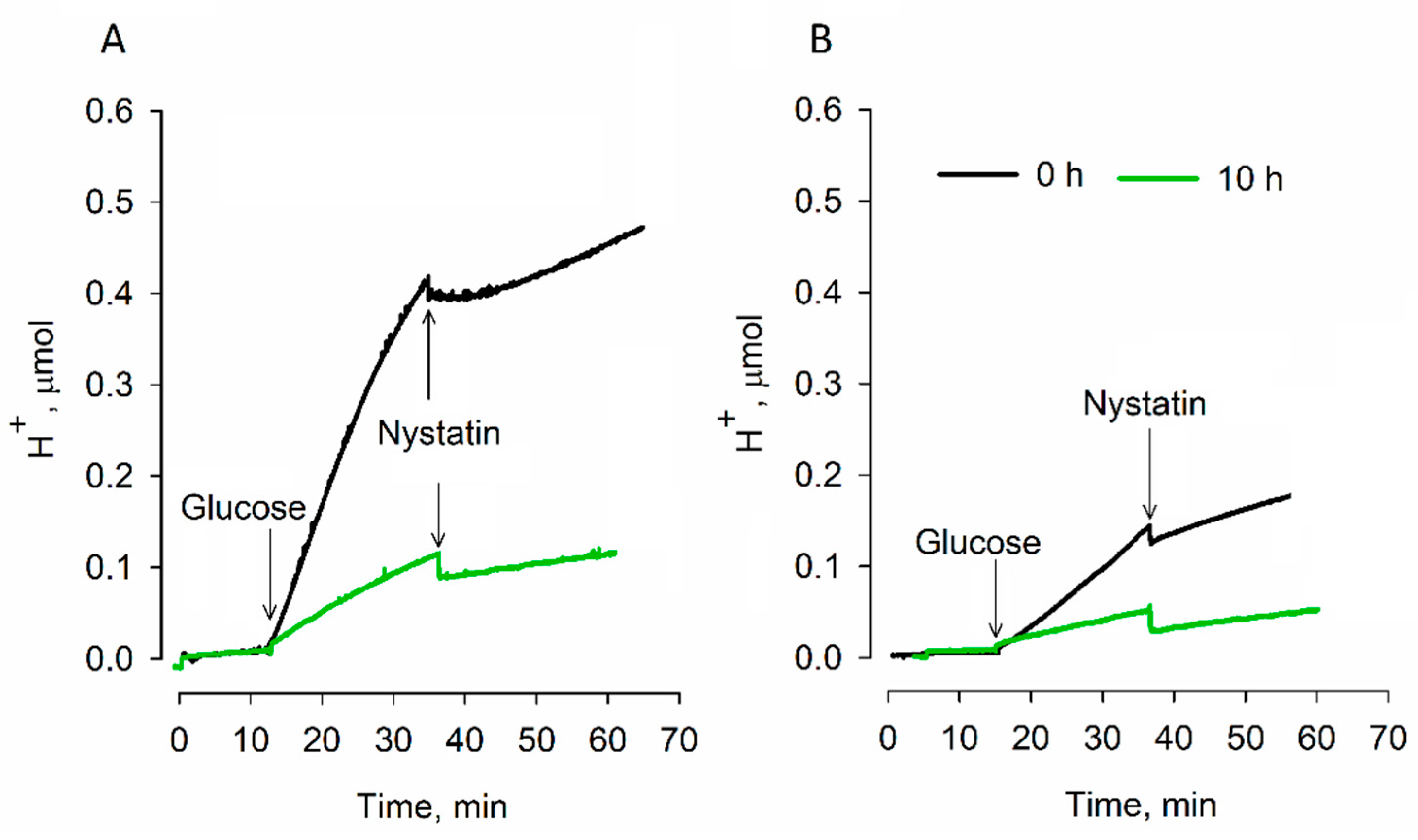
S. cerevisiae cells during dehydration: very resistant to dehydration strain 77 cells maintained higher glucose-induced respiration activity than semi-resistant strain 14 ones. Responding to the addition of glucose,
S. cerevisiae 14 cells not only demonstrated weaker respiration activity, but also carried out more intense acidification of the incubation medium.
It is known that energy-deprived yeasts acidify their cytoplasm. This feature is related to the dormancy state of cells [
16,
17]. The increase in acidity of the cytosol during dormancy causes many proteins to interact with each other and form large clumps or filament structures that result in the cytoplasm becoming stiffer. Mobility of the organelles decreases, and mechanical stability of the cells increases. The results of our experiments indicate that strain 14 cells acidify the medium almost four times more efficiently compared to strain 77 cells. After 10 h of desiccation, the medium acidification considerably decreased, but strain 14 cells still extruded acid twice as extensively. It is possible that a lower extrusion of acidic products, but more intense respiration (stronger extrusion of H
+ from mitochondria to the cytosol), helps strain 77 cells to reach the dormancy state more efficiently than strain 14 cells do.
Our results revealed a decrease in the ATP content in cells of both strains during dehydration. This probably indicates the usage of ATP for metabolic processes in the cells for intracellular protection of the cell structure and the maintenance of viability. While losing water content, yeasts synthesize endogenous compounds, such as glycerol and trehalose [
2]. A rapid change in the cellular energy metabolism reflects the higher energy demands required for surviving solute synthesis against osmotic shock [
18,
19]. After ATP depletion, Marini et al. [
16] observed significant cytosolic compaction and extensive cytoplasmic reorganization, as well as the emergence of distinct membrane-bound and membraneless organelles. The levels of ATP in both cells decreased with similar kinetics, but very resistant to dehydration 77 cells contained 10–20% more ATP than semi-resistant strain 14 ones. The larger amount of ATP in strain 77 cells before dehydration can also support the higher resistance of these cells to desiccation.
During reactivation, strain 77 cells more efficiently recovered intracellular ATP than strain 14 ones; the ATP level in strain 77 cells slowly increased, while the ATP content of strain 14 cells decreased. These results support the idea about the higher stability and activity of mitochondria in strain 77 cells, giving them the ability of more efficient repair of their structure and functions. However, before decreasing, the amount of ATP increased by 35% in the case of strain 77 and by ~15% in strain 14 cells during the first 2.5 h of desiccation (
Figure 5). The increase in the ATP level could be a result of a temporal stimulation of synthesis or a consequence of an abrupt ceasing of ATP-consuming processes inside the cells. In our mind, the latter process would be more probable. If desiccation initially decreases the ATP consumption, in conditions of good aeration, the yeast cells in thin pellets formed after centrifugation continue oxidative phosphorylation at a high rate.
The plasma membrane barrier to PCB
− (
Figure 4) and ATP content of strain 77 cells (
Figure 5B) grew rather slowly. This could be due to very active consumption of ATP by the processes of cell recovery. The efficiency of ATP hydrolysis depends on Mg
2+ ions, and these cations from the YPD medium could make ATP bioavailable [
18] for many antioxidative systems to abrogate the resulting damage [
2]. This could be one of the reasons why the cell reactivation in the YPD medium is more efficient than rehydration in water.
Erkut and colleagues [
19] studied entry into the state of anhydrobiosis of two eukaryotic organisms—
S. cerevisiae and the roundworm
Caenorhabditis elegans. Both organisms entered a state of anhydrobiosis during cellular survival under conditions of starvation. They discovered that a glyoxylate shunt is the metabolic switch in both organisms, which helps them to survive desiccation by enabling or promoting gluconeogenesis for the biosynthesis of trehalose. They compared the energetic/metabolic states and suggested that hypoaerobic lifestyle could be a feature of desiccation tolerance. In our case, mid-stationary phase
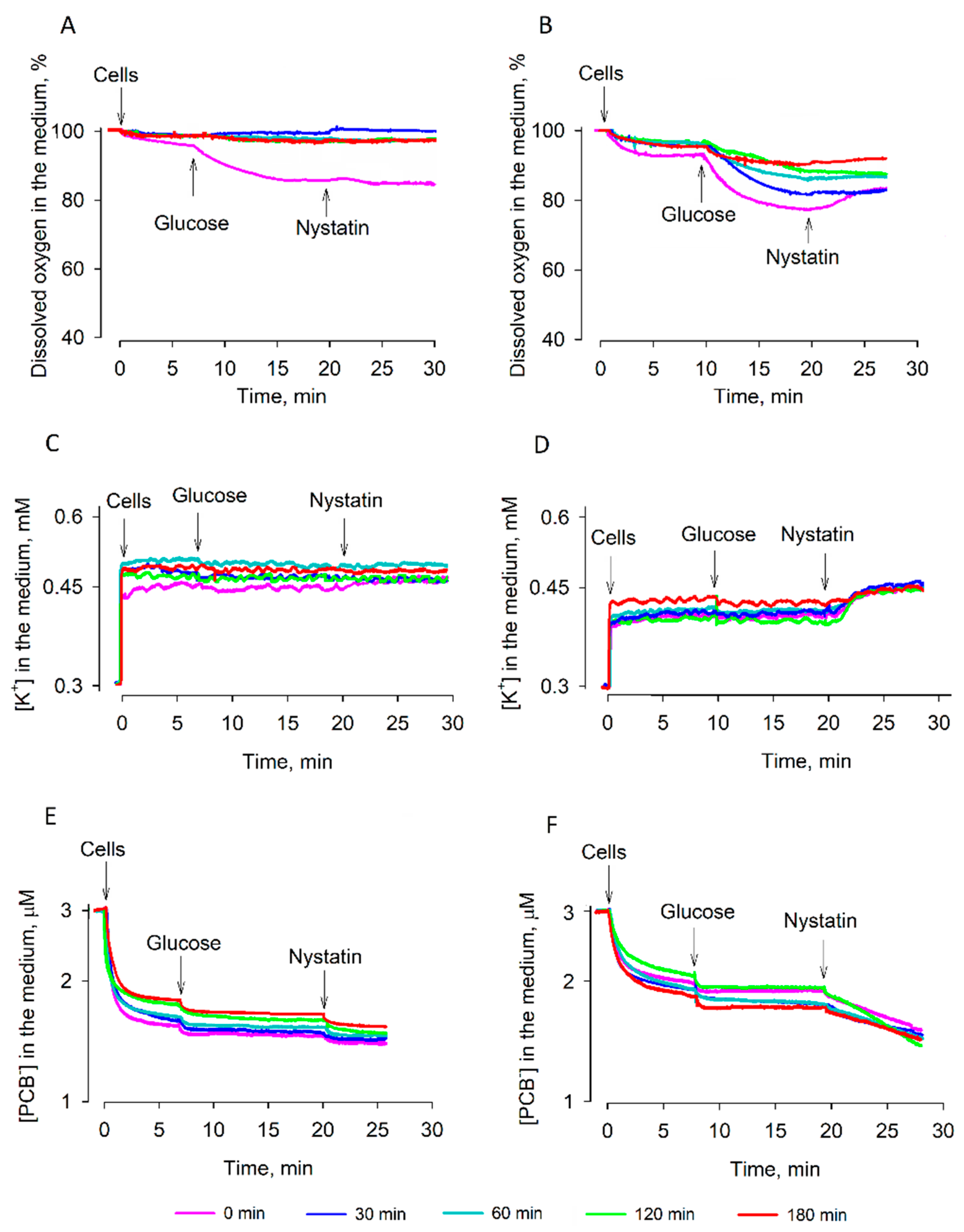
S. cerevisiae cells were not starving and were studied in aerobic conditions. Strain 14 cells started to demonstrate damaged respiration at rather early stages of dehydration; after 14 h of drying, these cells ceased the consumption of oxygen and did not demonstrate any reaction to the addition of glucose. However, this substrate was able to energize strain 77 cells even after 22 h of dehydration (
Figure 1B).
Intracellular K
+ helps to keep the osmotic balance across the plasma membrane and stabilizes the turgor pressure. During the dehydration of strain 14 cells, the reduction of the K
+ gradient on the plasma membrane proceeded more slowly and after 8 h of desiccation, these cells contained more K
+ than strain 77 ones. On the other hand, the addition of nystatin revealed that during 8–20 h of desiccation, strain 77 cells were in a “low, but stable K
+” state. Strain 14 cells were accumulating K
+ till around the fourteenth hour of desiccation, but they did not get to such a state; starting from the 16th h, nystatin did not induce any leakage of accumulated K
+. It is possible that nystatin released K
+ accumulated in the mitochondria of strain 77 cells. The activity of K
+ uptake systems in
Saccharomyces (i.e., Trk uniporters) and/or the corresponding gene expression can vary among the strains, and the cells can have different membrane potential, which is a driving force for K
+ uptake. After the reduction of the driving force, the release of accumulated K
+ can occur via transporters of this ion in the plasma membrane [
20,
21]. Our results suggest that the different modes of accumulation or the ability to maintain the amount of intracellular K
+ in
S. cerevisiae cells could affect the osmotolerance of the cells and be one of the factors determining the higher viability of strain 77 cells after rehydration.
PCB
−-binding measurements demonstrated an efficient barrier of the plasma membrane to lipophilic anions in strain 77 cells. Binding of PCB
− to desiccated strain 77 cells decreased gradually, but after 30 min of reactivation in the YPD medium, these cells bound considerably smaller amounts of this compound. Restoration of the barrier properties of semi-resistant strain 14 cells was less efficient, but the reactivation worked much better than rehydration in the deionized water. These results indicate that nutrients are needed for the active recovery of cells. As already mentioned, the remaining K
+ in the cells could help strain 77 yeasts to resist hyperosmotic stress. K
+ transporters in the plasma membrane depend on the stability of the electrical potential difference and integrity of this membrane [
22]. The accumulation of ergosterol in the plasma membrane leads to a higher stability of S. cerevisiae thermotolerant strains during dehydration compared to mesophilic strains [
8,
23]. Our results support the idea that higher stability of the cell membranes in the presence of ergosterol could help yeasts to resist desiccation and it could be related to better maintenance of K
+ in mitochondria.
Summarizing the results of our study, we suppose that at least two interconnected factors are responsible for the resistance of S. cerevisiae strain 17 cells to dehydration–rehydration: stability of mitochondria and intracellular content of ATP. The higher viability of S. cerevisiae 77 cells after reactivation could be linked to better maintenance of mitochondria in the dehydrated strain 77 cells and, correspondingly, to more efficient ATP synthesis by oxidative phosphorylation during reactivation. This means that the special stability of the mitochondria, improving biotechnological approaches or pretreatments of yeasts, could improve the resistance of cells to dehydration-rehydration. This hypothesis, of course, should be verified in future experiments.