Simple Summary
This review is a summary of published studies that contain drug residue depletion data for edible tissues and milk following treatment of sheep and goats. The information is separated by antibiotic class for ease of comparison between studies. This summary is useful for understanding medication residue depletion following extra-label drug use and can be used to help estimate withdrawal intervals in order to help protect the human food chain.
Abstract
This review provides a summary of extracted data from the published literature that contains drug residue depletion data for edible tissues and milk following treatment of sheep and goats. Out of 20,234 records obtained during the initial search, data from 177 records were included in this review. The data is separated by antibiotic class for ease of comparison between studies. Extracted data includes the active ingredient, dosing information, animal health status, analytical method and limits of detection, tolerance and maximum residue limit information, and time frames relative to residue absence or detection. This information is useful for understanding drug residue depletion profiles following extra-label use and for estimating withdrawal intervals, in order to protect the human food chain.
1. Introduction
Drinking water and availability of food for both humans and animals are affected by climate change that lowers rainfall and an increasing world population, especially in semi-arid climates [1]. Small ruminants present a unique opportunity for developing nations, specifically in developing nations that are in semi-arid climates, due to their multi-purpose use (meat, milk and fibers), lower production cost compared to large ruminants, and tolerance to low rainfall and hot climates [1].
According to data from the Food and Agriculture Organization of the United Nations (FAO), the number of sheep and goats worldwide has increased from approximately 1.4 billion head combined (1 billion sheep, ~400 million goats) in 1961 to approximately 2.3 billion head combined (~1.2 billion sheep, ~1.1 billion goats) in 2019 [2]. Between 2014 and 2019, the largest producers of sheep meat worldwide were China, Australia, New Zealand, Turkey and Algeria, whereas during this same time period, the largest producers of goat meat worldwide were China, India, Pakistan, Nigeria and Bangladesh.
In the United States, sheep and goats are considered minor species by the Food and Drug Administration (FDA) [3]; however, sheep are considered major species while goats are considered minor species by the European Medicines Agency (EMA) Committee for Medicinal Products for Veterinary Use [4]. In the United States, there is a “severe shortage of approved new animal drugs for use in minor species” [5].
The Food Animal Residue Avoidance and Depletion Program (FARAD) is a United States Department of Agriculture (USDA)-funded program with a mission to provide veterinary practitioners with scientifically based withdrawal interval recommendations following extra-label drug use or chemical/pesticide contamination in food-producing species. FARAD call submission data for small ruminants indicates a steady increase in the number of withdrawal interval request submissions from 2015 to 2019, with a steep increase in the number of submissions in 2020 (2015 = 435 submissions for sheep, 223 for goats; 2019 = 343 submissions for sheep, 710 for goats; 2020 = 595 submissions for sheep, 1401 for goats). The most commonly requested drug categories include antibiotics, anthelmintics, and non-steroidal anti-inflammatory drugs (NSAIDs). This data reflects the increasing numbers of backyard or hobby-farm environments, where the food-products are consumed by the family keeping the sheep or goats. Given the limited FDA-approved medications for use in sheep or goats, drugs are commonly prescribed in an extra-label manner which is legalized by the Animal Medicinal Drug Use Clarification Act of 1994 (AMDUCA) [6].
Given the importance of sheep and goats as commodity groups worldwide, the purpose of this review is to summarize research studies investigating antibiotic medication use in small ruminants with respect to the potential for drug residues to be present in small ruminant meat and milk products. Due to the large volume of published literature in small ruminants, this review only includes antibacterial medications; however, a second review will be completed incorporating anthelmintics and other medication classes not included here. It is important to note that residue depletion times referenced in the text are based on data from scientific studies. Normal industry practice to withdraw feed 8 to 12 h prior to processing the animals in order to minimize fecal contamination [7] may not have occurred in scientific research studies examining a zero day withdrawal. In addition, the residue depletion times listed in this manuscript are dependent on the sensitivity of the analytical method utilized in the study. Summaries of drug residue studies, drug approvals, tolerances (in the United States), and maximum residue limits (MRLs; in the European Union) have been provided in the tables for the reader’s convenience. If available, FDA-approved medications for use in sheep and goats should be utilized according to directions and labeled withdrawal times adhered to in order to guarantee human food safety.
2. Materials and Methods
2.1. Search Strategy
A systematic literature search was conducted using various databases and compared to publications included FARAD Program’s literature database. The aim of the search was to collect milk and edible tissue residue data for antibiotics that had been administered to small ruminants. Published literature between 1926 and 2021 was searched using PubMed, Cab Direct, Scopus, and Web of Science. Search terms and key words included: “sheep”, “goat(s)”, “small ruminants”, “caprine”, “ovine”, “drug absorption”, “clearance”, “drug residue(s)”, “pharmacokinetics”, “metabolic clearance rate”, “intestinal absorption”, “bioavailability”, “biological availability”, “metabolism”.
2.2. Screening Results
For systematic screening, search results were imported into the Covidence online platform (Covidence Systematic Review Software, Veritas Health Innovation, Melbourne, Australia) and duplicate results were removed by the Covidence software. Initially, the 20,234 “Titles and Abstracts” were screened by one reviewer (EDR or CED) for relevancy and categorized as ‘yes’, ‘no’, or ‘maybe’ using predetermined inclusion and exclusion criteria. The category of ‘maybe’ was used for trials that did not explicitly state the inclusion or exclusion criteria in the abstract and thus required further review of the full text. Inclusion criteria were as follows: in vivo sheep or goat drug trial; drug or metabolite concentration data and time point in tissue and/or milk; drug dose, route of administration, and dosing frequency stated. Exclusion criteria were as follows: any animal not a sheep or goat; in vitro study; concentration or residue data for non-drug substances (pesticide, toxin, vitamins) or drugs of abuse; drug plasma or serum concentrations only; dose of drug, route of administration, and dosing frequency missing. After initial screening for exclusion criteria, 1769 ‘yes’ and ‘maybe’ results moved to a ‘Full Text’ screening by one reviewer (EDR, KLM, or CED). These records were further excluded or included based on the above criteria and a reason was assigned. Records were excluded due to: not being a study (e.g., review, short communication, corrigendum; n = 128), not being able to verify text (e.g., full text not available from lenders worldwide, abstract only from proceedings, text unable to be translated; n = 141), being the wrong patient population/study design (e.g., not in live animals, in live animals other than small ruminants, etc.; n = 84), chemical product of study was a non-drug substance (n = 10), matrices under study did not consist of tissues or milk (n = 1076), and lack of specific concentration versus time presented in the paper (n = 60). A total of 270 records met the complete inclusion criteria. Figure 1 displays a flowchart representation of the screening process completed in this literature search.
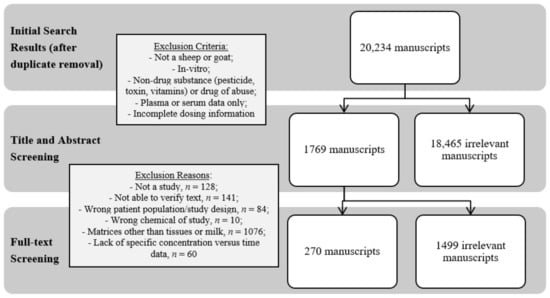
Figure 1.
Schematic diagram of the process for three independent evaluators to assess published manuscripts and the numerical outcome of each step. The ultimate goal was to curate manuscripts with tissue and milk residue data from live sheep and goat antibacterial drug studies.
For comparison, the FARAD database returned 832 records for both sheep and goats; however, 78 records were removed from the review due to incorrect matrices (i.e., plasma or serum data only). Ultimately, only 177 records met the complete inclusion criteria.
3. Data Extraction and Presentation (Antibiotic Drug Classes, Residue Detection, and Analytical Methods)
The published literature presenting tissue and milk residue data for antibiotics used in sheep and goats is presented in the Tables below and is categorized by antibiotic class. Tolerances or maximum residue limits are presented for FDA-approvals and EMA-approvals, respectively. The basic analytical method is described, with a focus on the limit of detection and limit of quantitation, alongside the dosing regimen for each study. Animal health status and additional information are also included, since variations in health- or lactation-status may affect drug residue depletion. Finally, two columns are included to indicate when residues were last detected. The column titled ‘Last sampling time point for which residues WERE detected (post-last treatment)’ refers the last sampling point when residues were detected based on the study sampling protocol. This is in contrast to the column titled ‘Sampling time point when NO residues were detected (post-last treatment)’ which refers to the last sampling point when residues were not detected based on the study sampling protocol. Instances where a greater than symbol (“>”) is utilized refers to situations where residues were still detected at the last sampling time point of the study protocol.
Data for the summarized studies includes analytical methods since it is important to consider how those methods impact the sensitivity of drug residue detection and how the analytical limits of detection compare to tolerances or MRLs. Newer analytical methods can detect drug residues at lower concentrations than historical microbiological bioassays or colorimetric testing, resulting in a greater number of days with detectable drug residues. In contrast, studies using less sensitive methods, having higher limits of detection, may have found shorter periods with detectable drug residues upon withdrawal of the drug. Readers are cautioned to keep the sensitivity of the analytical methods in mind when evaluating the data presented within this review, as well as the fact that most of the studies were completed in healthy animals. It is also important to note that US products approved for use in small ruminants should be used according to the FDA-approved label directions. The FDA-approved label withdrawal time should take precedent above any of the data summarized in this paper.
When considering antibiotic drug classes, it is important to remember that the World Health Organization (WHO) classifies antibiotics into categories based on their place in therapy for some infections in human medicine. These categories include critically important, highly important and important [8]. Some critically important antibiotics are then sub-divided by priority if they are considered sole or limited therapy for some infections in human medicine [8]. Some cephalosporins (third, fourth and fifth generations), quinolones, macrolides are classified as highest priority critically important antibiotics for human health. Aminoglycosides, some cephalosporins (first and second generations) are classified as high priority critically important antibiotics. Amphenicols, some penicillins (antipseudomonal, aminopenicillins with and without beta-lactamase inhibitors, amidinopenicillins, anti-staphylococcal, narrow spectrum), sulfonamides and tetracyclines are classified as highly important antibiotics for human health by the WHO.
3.1. Aminoglycosides
Aminoglycosides (amikacin, apramycin, dihydrostreptomycin, gentamicin, tobramycin, neomycin, streptomycin) are concentration dependent, bactericidal antibiotic agents produced from Streptomyces spp. and Micromonospora spp. Aminoglycosides act by irreversibly binding to the 30s subunit of the bacterial ribosome thereby inhibiting protein synthesis. Their spectrum of activity includes mostly Gram-negative bacteria, with some mycobacteria and staphylococci coverage. Transmission of Enterococcus spp., Enterobacteriaceae (including E. coli), and Mycobacterium spp. can occur from non-human sources and potentially result in human infection. Therefore, the appropriate use of aminoglycosides in food animal species is essential to maintain human safety.
Aminoglycosides are generally not well absorbed from the gastrointestinal tract [9], unless there is damage to the intestinal mucosa. When administered parenterally, aminoglycosides are rapidly and completely absorbed. Elimination of aminoglycosides is primarily renally, which may result in persistent residues in the kidneys. In most published studies in sheep and goats, residues in renal tissue exceeded the duration of the study [10,11,12,13,14,15,16,17]. In humans, aminoglycosides are poorly excreted into breastmilk [18]. This may also be the case for sheep and goats as a few studies have shown short duration of residue detection in milk following IV and IM administration [19,20,21,22,23,24,25,26].
In the United States, the only aminoglycoside FDA-approved for use in small ruminants is neomycin sulfate. However, the EMA has approved streptomycin/dihydrostreptomycin and kanamycin for sheep, while also extending MRLs from other species for gentamicin and neomycin. Table 1 shows the published literature that provides data for edible tissue or milk residues of aminoglycosides following treatment of sheep and goats.

Table 1.
Aminoglycoside residues in milk or edible tissue samples from sheep or goats following treatment.
3.2. Amphenicols
Amphenicols (chloramphenicol, florfenicol, thiamphenicol) are broad-spectrum antibiotics. These antibiotics are typically bacteriostatic agents that act by inhibiting microbial protein synthesis by binding to the 50s bacterial ribosomal subunit. Amphenicols are broad-spectrum against many aerobic and anaerobic Gram-positive and Gram-negative bacteria.
Little pharmacokinetic data is available following the use of amphenicols in sheep or goats. The limited data available in goats shows that florfenicol and thiamphenicol residues do enter the milk after intramuscular and intravenous administration, however tissue data was not available [35,36]. In one study, thiamphenicol concentrations were higher in the mammary gland that was frequently stripped compared to the gland that was not [35].
In the United States, there are no amphenicol products FDA-approved for use in sheep or goats. Chloramphenicol is prohibited from use in food producing animals in several countries including the United States, European Union, and Canada [6,37,38] due to the risk of blood dyscrasias, such as aplastic anemia and bone marrow suppression, in humans. Table 2 summarizes the published literature evaluating edible tissue or milk residues of amphenicols following treatment of sheep and goats.

Table 2.
Amphenicol residues in milk or edible tissue samples from sheep or goats following treatment.
3.3. Penicillin and Penicillin-Derivatives
Penicillins (penicillin G procaine, penicillin G benzathine) and penicillin-derivatives (amoxicillin, ampicillin, cloxacillin, dicloxacillin, nafcillin) are bactericidal antibiotics that act by inhibiting cell wall synthesis. These antibiotics display a broad spectrum of activity against many Gram-positive and Gram-negative bacteria, including anaerobic bacteria.
Amoxicillin and ampicillin show limited milk penetration or accumulation, even when the blood-milk barrier is altered in cases of mastitis [47,48]. However, beta-lactam products labeled for intramammary administration in cattle can result in very high antibiotic concentrations within the small ruminant udder due to the differences in both body and udder size [49,50]. Consequently, intramammary administration of cattle-labeled products to small ruminants can lead to persistent residues present in the milk and require extended withdrawal intervals beyond the labeled withdrawal times for cattle [49,51,52,53,54].In the United States, penicillin G procaine is FDA-approved for use in sheep via intramuscular administration. In the EU, MRLs have been extended from bovine species to all ruminants for nafcillin.
Due to the potential for allergic reactions to penicillin and penicillin-derivatives in humans, caution must be exhibited to ensure food-products from small ruminants do not contain traces of penicillins [55,56]. Table 3 summarizes the published literature evaluating edible tissue or milk residues of beta-lactams or penicillins following treatment of sheep and goats.

Table 3.
Penicillin and penicillin-derivative residues in milk or edible tissue samples from sheep or goats following treatment.
3.4. Cephalosporins
Cephalosporins (first-generation: cephapirin, cefacetrile, cephalothin, cephradine, cephalexin; second-generation: cefonicid; third-generation: ceftazidime, ceftiofur, ceftriaxone; fourth-generation: cefquinome, cefepime) are beta-lactam antibiotics divided into five ‘generations’ based on the spectrum of activity (first-generation cephalosporins are active against Gram-positive bacteria but not Gram-negative bacteria, while each consecutive generation has increased activity against Gram-negative bacteria with decreased Gram-positive activity). In the United States, cephalosporins are permitted to be used in an extra-label manner in minor species, such as sheep and goats, unlike major food producing species (cattle, swine, chickens & turkeys).
In general, cephalosporins have low penetration into milk [62,63,64,65,66] with variable pharmacokinetic parameters and slower milk depletion in mastitic animals [67,68]. Cephalexin exhibited a nearly double terminal serum elimination half-life in ewes compared to cattle, in addition to increased concentrations of cephalexin residues [69]. Cephapirin exhibited a longer presence of residues in goat samples compared to cattle when used for mastitis treatment [70].
Ceftiofur sodium (Naxcel®) is currently the only FDA-approved cephalosporin for use in sheep and goats with a 0 day meat and milk withdrawal time. Pharmacokinetic parameters of both intravenous and intramuscular ceftiofur sodium are found to be similar between sheep and goats when administered at the same dose [71]. Table 4 summarizes the published literature evaluating edible tissue or milk residues of cephalosporins following treatment of sheep and goats.

Table 4.
Cephalosporin residues in milk or edible tissue samples from sheep or goats following treatment.
3.5. Fluoroquinolones/Quinolones
Fluoroquinolones (ciprofloxacin, danofloxacin, difloxacin, enrofloxacin, levofloxacin, marbofloxacin, moxifloxacin, norfloxacin, orbifloxacin, pefloxacin, sarafloxacin) are broad-spectrum antibiotics that exhibit concentration-dependent bactericidal activity via inhibition of DNA gyrase in bacterial cells. As a drug class, fluoroquinolones exhibit a high lipid solubility, low protein binding, high bioavailability (especially after parenteral administration) and large volumes of distribution in most species, including small ruminants [81,82,83,84,85,86,87,88,89,90,91,92,93,94,95]. Due to the importance of fluoroquinolones to human health, fluoroquinolones are prohibited from extra-label drug use in food-producing species in the United States.
Studies suggest that the pharmacokinetics of fluoroquinolones change during lactation due to the increased elimination of the drug from serum [88,96]. Additionally, multiple fluoroquinolones extensively penetrate into milk, with some drugs in the class exhibiting up to a 10× higher concentration in milk compared to plasma or serum [88,96,97,98]. This variation can be useful in mastitis cases since these drugs can accumulate in the milk at concentrations above the MIC for a sustained period of time [96,97,99].
In the United States, there are no fluoroquinolones FDA-approved for use in small ruminants, and due to the stipulations outlined by AMDUCA in the CFR, fluoroquinolones are prohibited from extra-label use in food-producing species [6]. In the European Union, flumequine is the only approved fluoroquinolone for use in sheep, while MRLs have been extended from bovine species to all food-producing species for enrofloxacin. Table 5 summarizes the published literature evaluating edible tissue or milk residues of quinolones following treatment of sheep and goats.

Table 5.
Fluoroquinolone residues in milk or edible tissue samples from sheep or goats following treatment.
3.6. Macrolides
Marcolides (erythromycin, gamithromycin, spiramycin, tilmicosin, tulathromycin and tylosin) are a group of bacteriostatic compounds that bind to the 50S bacterial ribosomal subunit inhibiting bacterial protein synthesis and cell growth [110]. These antibiotics are effective against Mycoplasma spp. and Gram-positive organisms, and less effective against Gram-negative organisms.
Penetration into tissues, milk and blood are shown to be relatively quick with high systemic availability [111]. Macrolides show good penetration and distribution into the udder. In particular, tilmicosin and tulathromycin have been shown to have persistent drug residues in the milk [112,113,114,115,116,117], thus they are not recommended for use in lactating animals. Erythromycin, spiramycin and tylosin also exhibit good udder penetration, but result in shorter withdrawal intervals [29,60,111,118,119,120,121,122]. Some small ruminant macrolide pharmacokinetic parameters (absorption, volume of distribution and elimination) were found to be similar to those reported in cattle [111,112,116,123,124].
In the United States, the only FDA-approved macrolide for use in sheep is tilmicosin; however, this approval specifically excludes lactating sheep. Therefore, no tolerance has been established for milk. In the European Union, multiple macrolides are approved for use in small ruminants: gamithromycin and tilmicosin in sheep, and tulathromycin in both sheep and goats. Additionally, MRLs have been extended from other species for erythromycin, tilmicosin (in goats) and tylosin. Table 6 summarizes the published literature evaluating edible tissue or milk residues of macrolides following treatment of sheep and goats.

Table 6.
Macrolide residues in milk or edible tissue samples from sheep or goats following treatment.
3.7. Sulfonamides
Sulfonamides (sulfadiazine, sulfadimethoxine, sulfamethoxazole, sulfachlorpyrazine) are bacteriostatic antibacterial medications that complete with para-aminobenzoic acid disrupting folic acid synthesis. They are active against Gram-positive and Gram-negative bacteria and protozoa.
One study administered sulfonamides in both normal and mastitic ewes. Sulfonamide concentrations were found to be much higher in the mastitic ewe milk, which the authors attributed in part to the increase in milk pH of mastitic milk [131]. Another study found that some sulfonamides are found in the milk in higher concentrations than blood, whereas others (sulfathiazole, sulfadimidine, sulfadiazine and sulfacetamide) are found in the milk in lower concentrations than blood [132].
Due to the potential for allergic reactions to sulfonamides, caution must be exhibited to ensure food-products from small ruminants do not contain traces of sulfonamides [133,134]. In the US, extra-label use of sulfonamides is prohibited in dairy cattle 20 months of age and older, due to allergic potential of affected milk and increased violative residues.
In the United States, there are no sulfonamide products FDA-approved for use in small ruminants, whereas there are some sulfonamide active ingredients with established milk MRLs for small ruminants in the EU. Table 7 summarizes the published literature evaluating edible tissue or milk residues of sulfonamides following treatment of sheep and goats.

Table 7.
Sulfonamide residues in milk or edible tissue samples from sheep or goats following treatment.
3.8. Tetracyclines
Tetracyclines (chlortetracycline, doxycycline oxytetracycline, tetracycline) are broad-spectrum antibiotics that act by inhibiting the 30S bacterial ribosomal subunit thus inhibiting protein synthesis. They are active against Gram-positive and Gram-negative bacteria, as well as some atypical mycobacteria and protozoa [144,145,146].
In oxytetracycline- and chlortetracycline-treated animals, milk production decreased 15% [147]. Infusion of drug into one half of the udder resulted in diffusion of low concentrations into the untreated udder half [147].
Following intramammary infusion of chlortetracycline, residues were detected for a shorter time in goat milk compared to cow milk; however, parenteral chlortetracycline administration results in similar milk residue depletion between goats and cows [122].
In the United States, there are multiple tetracycline approvals for both sheep and goats: chlortetracycline (medicated feed for sheep), oxytetracycline (sheep) and tetracycline (sheep and goats; only topical administration for goats). In the EU, MRLs have been determined for chlortetracycline, oxytetracycline and tetracycline in all food-producing species. Table 8 summarizes the published literature evaluating edible tissue or milk residues of tetracyclines following treatment of sheep and goats.

Table 8.
Tetracycline residues in milk or edible tissue samples from sheep or goats following treatment.
4. Conclusions
The judicious use of medications and drug residue avoidance is an important topic in animal agriculture and for veterinarians treating animals that provide food for humans. Although there are numerous published studies that describe drug residues in sheep and goat meat and milk, they are scattered throughout the primary literature. In this review, these studies are compiled, and data extracted for easy reference to help facilitate a comprehensive overview of the scientific data, with respect to drug residues in edible tissues and milk from sheep and goats for antibiotics used in small ruminant practice.
Author Contributions
Conceptualization, L.A.T.; methodology, L.A.T.; visualization, M.O.C.; writing—original draft preparation, E.D.R. and L.A.T.; writing—review and editing, K.L.M. and C.E.D.; project administration, L.A.T. All authors have read and agreed to the published version of the manuscript.
Funding
This project was supported by United States Department of Agriculture, National Institute of Food and Agriculture, Food Animal Residue Avoidance and Depletion Program grant numbers 2020-41480-32518 and 2021-41480-35268.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this review are included in the main manuscript and included tables. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| AMDUCA | Animal Medicinal Drug Use Clarification Act of 1994 |
| CFR | Code of Federal Regulations |
| EMA | European Medicines Agency |
| FAO | Food and Agriculture Organization of the United Nations |
| FARAD | Food Animal Residue Avoidance and Depletion Program |
| FDA | Food and Drug Administration |
| MIC | Minimum inhibitory concentration |
| MRL | Maximum residue limit |
| NSAIDs | Non-steroidal anti-inflammatory drugs |
| USDA | United States Department of Agriculture |
| WHO | World Health Organization |
References
- Akinmoladun, O.F.; Muchenje, V.; Fon, F.N.; Mpendulo, C.T. Small Ruminants: Farmers’ Hope in a World Threatened by Water Scarcity. Animals 2019, 9, 456. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations. FAOSTAT. 2022. Available online: https://www.fao.org/faostat/en/#compare (accessed on 22 November 2021).
- Food and Drug Administration. Minor Use/Minor Species; Food and Drug Administration: Spring, MD, USA, 2022.
- European Medicines Agency. Guideline on Safety and Residue Data Requirements for the Establishment of Maximum Residue Limits in Minor Species; European Medicines Agency: Amsterdam, The Netherlands, 2022. [Google Scholar]
- Congress of the United States. Minor Use and Minor Species Animal Health Act of 2004; Congress of the United States: Washington, DC, USA, 2004.
- Food and Drug Administration. Title 21, Chapter I, Subchapter E, Part 530: Extralabel Drug Use in Animals; Food and Drug Administration: Silver Spring, MD, USA, 2022.
- Northcutt, J.K. Preslaughter factors affecting poultry meat quality. In Poultry Meat Processing; Woodhead Publishing: Cambridge, UK, 2000. [Google Scholar]
- World Health Organization. Critically Important Antimicrobials for Human Medicine 6th Revision; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Riviere, J.E.; Craigmill, A.L.; Sundlof, S.F. Aminoglycosides. In Handbook of Comparative Pharmacokinetics and Residues of Veterinary Antimicrobials; CRC Press: Boca Raton, FL, USA, 1991. [Google Scholar]
- European Medicines Agency Committee for Veterinary Medicinal Products. Apramycin Summary Report (2); European Medicines Agency Committee for Veterinary Medicinal Products: Amsterdam, The Netherlands, 1999; Volume 526, pp. 1–8. [Google Scholar]
- Heitzman, R. Dihydrostreptomycin Streptomycin. Residues Some Vet. Drugs Anim. Foods 1995, 41, 17–29. [Google Scholar]
- Brown, S.; Riviere, J.; Coppoc, G.; Hinsman, E.; Carlton, W.; Steckel, R. Single intravenous and multiple intramuscular dose pharmacokinetics and tissue residue profile of gentamicin in sheep. Am. J. Vet. Res. 1985, 46, 69–74. [Google Scholar] [PubMed]
- Brown, S.; Coppoc, G.; Riviere, J. Effects of dose and duration of therapy on gentamicin tissue residues in sheep. Am. J. Vet. Res. 1986, 47, 2373–2379. [Google Scholar] [PubMed]
- Brown, S.; Baird, A. Evaluation of renal gentamicin depletion kinetic properties in sheep, using serial percutaneous biopsies. Am. J. Vet. Res. 1988, 49, 2056–2059. [Google Scholar]
- The Upjohn Company. NADA 011-315 Neomycin 325/Neomix Ag 325 Soluble Powder—Supplemental Approval (Date of Approval: 3 April 1996); FOI—Neomix- NADA 011-315; The Upjohn Company: Hastings, MI, USA, 1996; pp. 1–3. [Google Scholar]
- European Medicines Agency Committee for Veterinary Medicinal Products. Neomycin Summary Report (2); EMEA/MRL/730/00-Final; European Medicines Agency Committee for Veterinary Medicinal Products: Amsterdam, The Netherlands, 2000; Volume 730, pp. 1–8. [Google Scholar]
- Livingston, R.C. Neomycin. Residues Some Vet. Drugs Anim. Foods 1995, 41, 57–67. [Google Scholar]
- National Library of Medicine. Drugs and Lactation Database (LactMed); National Library of Medicine: Bethesda, MD, USA, 2006. [Google Scholar]
- El-Sooud, K.A. Pharmacokinetics of amikacin in lactating goats. Zent. Vet. A 1999, 46, 239–246. [Google Scholar] [CrossRef]
- Agrawal, A.; Singh, S.; Jayachandran, C. Pharmacokinetics of amikacin in goats after single intramuscular administration. Indian J. Pharmacol. 2001, 33, 374. [Google Scholar]
- Haritova, A.; Lashev, L. Pharmacokinetics of amikacin in lactating sheep. Vet. Res. Commun. 2004, 28, 429–435. [Google Scholar] [CrossRef]
- Ziv, G.; Kurtz, B.; Risenberg, R.; Glickman, A. Serum and milk concentrations of apramycin in lactating cows, ewes and goats. J. Vet. Pharmacol. Ther. 1995, 18, 346–351. [Google Scholar] [CrossRef]
- El-Gendi, A.Y.; Amer, A.M.; Azooz, H.A. Disposition kinetics and milk residues of apramycin in goats. In Proceedings of the European Association of Veterinary Pharmacology and Toxicology 8th Congress, Jerusalem, Israel, 30 July–3 August 2000. [Google Scholar]
- Ziv, G.; Bogin, E.; Shani, J.; Sulman, F.G. Distribution and blood-to-milk transfer of labeled antibiotics. Antimicrob. Agents Chemother. 1973, 3, 607–613. [Google Scholar] [CrossRef]
- European Medicines Agency Committee for Veterinary Medicinal Products. Dihydrostreptomycin (Extrapolation to All Ruminants) Summary Report (4); EMEA/CVMP/211249/2005-Final; European Medicines Agency Committee for Veterinary Medicinal Products: Amsterdam, The Netherlands, 2005; Volume 211249, pp. 1–5. [Google Scholar]
- Friedlander, L.G.; Stephany, R.W. Dihydrostreptomycin and Streptomycin Addendum. Residues Some Vet. Drugs Anim. Foods 2002, 41, 37–41. [Google Scholar]
- Lohuis, J.; Poutrel, B.; Cremoux, R.; Parez, V.; Aguer, D. Milk residues of penicillin, nafcillin and dihydrostreptomycin in dairy goats postpartum treated with Nafpenzal N8 at drying-off. In Proceedings of the Residues of Antimicrobial Drugs and Other Inhibitors in Milk, Kiel, Germany, 28–31 August 1995. [Google Scholar]
- Lohuis, J.; Berthelot, X.; Cester, C.; Parez, V.; Aguer, D. Pharmacokinetics and milk residues of penicillin, nafcillin and dihydrostreptomycin in dairy sheep treated with nafpenzal DC at drying-off. In Proceedings of the Residues of Antimicrobial Drugs and Other Inhibitors in Milk, Kiel, Germany, 28–31 August 1995. [Google Scholar]
- Ziv, G.; Bogin, E.; Shani, J.; Sulman, F. Penetration of radioactive-labeled antibiotics from blood serum into milk in normal and mastitic ewes. Ann. Rech. Vét. 1974, 5, 15–28. [Google Scholar]
- European Medicines Agency Committee for Veterinary Medicinal Products. Streptomycin and Dihydrostreptomycin Summary Report (2); EMEA/MRL/728/00-Final; European Medicines Agency Committee for Veterinary Medicinal Products: Amsterdam, The Netherlands, 2000; Volume 728, pp. 1–7. [Google Scholar]
- Heitzman, R.J. Dihydrostreptomycin/Streptomycin Addendum. Residues Some Vet. Drugs Anim. Foods 1998, 41, 39–44. [Google Scholar]
- Toutain, P.; De Pomyers, H.; Larrieu, G.; Periquet, B.; More, J. An in vivo model for pharmacokinetic studies in the kidney. J. Pharmacol. Methods 1985, 14, 1–11. [Google Scholar] [CrossRef]
- Andreini, G.; Pignattelli, P. Kanamycin blood levels and residues in domestic animals. Vet. Milano 1972, 21, 51–72. [Google Scholar]
- Pfizer, Inc. ANADA 200-046 Neomycin Sulfate Soluble Powder 325 g/lb—Supplemental Approval (3 April 1996); FOI—Neomycin ANADA 200-046; Pfizer: New York, NY, USA, 1996; pp. 1–3. [Google Scholar]
- Lavy, E.; Ziv, G.; Glickman, A. Single-dose pharmacokinetics of thiamphenicol in lactating goats. Acta Vet. Scand. Suppl. 1991, 87, 99–102. [Google Scholar]
- Lavy, E.; Ziv, G.; Soback, S.; Glickman, A.; Winkler, M. Clinical pharmacology of florfenicol in lactating goats. Acta Vet. Scand. Suppl. 1991, 87, 133–136. [Google Scholar]
- Government of Canada. Questions and Answers on Health Canada’s Policy on Extra-Label Drug Use (ELDU) in Food-Producing Animals; Government of Canada: Ottowa, ON, Canada, 2009.
- European Food Safety Authority. Scientific Opinion on Chloramphenicol in Food and Feed; European Food Safety Authority: Parma, Italy, 2014. [Google Scholar] [CrossRef]
- Ziv, G.; Bogin, E.; Sulman, F. Blood and milk levels of chloramphenicol in normal and mastitic cows and ewes after intramuscular administration of chloramphenicol and chloramphenicol sodium succinate. Zent. Vet. Reihe A 1973, 20, 801–811. [Google Scholar] [CrossRef]
- Dagorn, M.; Guillot, P.; Sanders, P. Pharmacokinetics of chloramphenicol in sheep after intravenous, intramuscular and subcutaneous administration. Vet. Q. 1990, 12, 166–174. [Google Scholar] [CrossRef]
- Ziv, G. Pharmacokinetics of antimycoplasma antibiotics in dairy cows and ewes. In Proceedings of the 8th International Meeting of Diseases in Cattle, Milan, Italy, 9–13 September 1974; pp. 80–92. [Google Scholar]
- Wetzlich, S.E.; Lane, V.M.; Craigmill, A.L. Tissue residue depletion after multiple subcutaneous administration of florfenicol to sheep. In Proceedings of the European Association of Veterinary Pharmacology and Toxicology 10th Congress, Turin, Italy, 17–22 September 2006; Volume 29, pp. 154–155. [Google Scholar]
- Lane, V.M.; Villarroel, A.; Wetzlich, S.; Clifford, A.; Taylor, I.; Craigmill, A. Tissue residues of florfenicol in sheep. J. Vet. Pharmacol. Ther. 2008, 31, 178–180. [Google Scholar] [CrossRef]
- Wells, R.J. Thiamphenicol; Addendum to the thiamphenicol residue monograph prepared by the 47th meeting of the Committee and published in FAO Food and Nutrition Paper 41/9, Rome 1996. Residues Some Vet. Drugs Anim. Foods 2000, 41, 119–128. [Google Scholar]
- Roy, B.; Banerjee, N. Distribution of chloramphenicol in goat blood and milk after intramuscular administration. Indian J. Anim. Sci. 1983, 53, 847–849. [Google Scholar]
- Wal, J.-M.; Peleran, J.-C.; Bories, G.F. High performance liquid chromatographic determination of chloramphenicol in milk. J. Assoc. Off. Anal. Chem. 1980, 63, 1044–1048. [Google Scholar] [CrossRef]
- Ziv, G.; Nouws, J. Serum and milk concentrations of ampicillin and amoxycillin in ruminants. Refuah Vet. 1979, 36, 104–110. [Google Scholar]
- Ramos, F.; Boison, J.; Friedlander, L.G. Amoxicillin. Residues Some Vet. Drugs Anim. Foods 2012, 1–35. Available online: https://www.fao.org/fileadmin/user_upload/vetdrug/docs/12-2012-amoxicillin.pdf (accessed on 7 August 2022).
- Buswell, J.; Knight, C.; Barber, D. Antibiotic persistence and tolerance in the lactating goat following intramammary therapy. Vet. Rec. 1989, 125, 301–303. [Google Scholar] [CrossRef]
- Roncada, P.; Tomasi, L.; Stracciari, G.; Ermini, L.; Strocchia, A. Milk depletion of dicloxacillin residues in cows and sheep following intramammary administration. J. Vet. Pharmacol. Ther. 2000, 23, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Buswell, J.; Barber, D. Antibiotic persistence and tolerance in the lactating sheep following a course of intramammary therapy. Br. Vet. J. 1989, 145, 552–557. [Google Scholar] [CrossRef]
- Pengov, A.; Kirbis, A. Risks of antibiotic residues in milk following intramammary and intramuscular treatments in dairy sheep. Anal. Chim. Acta 2009, 637, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Hill, B.; Jagusch, K.; Rajan, L.; Kidd, G. Antibiotic residues in goats milk following intramammary treatment. N. Z. Vet. J. 1984, 32, 130–131. [Google Scholar] [CrossRef]
- Ferrini, A.M.; Trenta, S.; Mannoni, V.; Rosati, R.; Coni, E. Depletion of long-acting ampicillin in goat milk following intramuscular administration. J. Agric. Food Chem. 2010, 58, 12199–12203. [Google Scholar] [CrossRef]
- Boatto, G.; Cerri, R.; Pau, A.; Palomba, M.; Pintore, G.; Denti, M.G. Monitoring of benzylpenicillin in ovine milk by HPLC. J. Pharm. Biomed. Anal. 1998, 17, 733–738. [Google Scholar] [CrossRef]
- Gee, H.-E.; Ho, K.-B.; Toothill, J. Liquid chromatographic determination of benzylpenicillin and cloxacillin in animal tissues and its application to a study of the stability at –20 °C of spiked and incurred residues of benzylpenicillin in ovine liver. J. AOAC Int. 1996, 79, 640–644. [Google Scholar] [CrossRef]
- Longo, F.; Cozzani, R.; Santis, L.d.; Boselli, C.; Rosati, R.; Fagiolo, A.; Cinquina, A. Amoxicillin detection in milk using screening tests and liquid chromatography after administration to lactating sheep. Riv. Sci. Dell’alimentazione 2002, 31, 313–319. [Google Scholar]
- Chaleva, E.; Dincheva, E. Ampicillin tolerance and content in the mammary gland of lactating cows and sheep. Vet. Med. Nauk. 1977, 14, 73–78. [Google Scholar]
- Edwards, S. Penicillin levels in the milk following intramuscular injection. Vet. Rec. 1966, 78, 583–585. [Google Scholar] [CrossRef]
- Long, P.; Heavner, J.; Ziv, G.; Geleta, J.; Nepote, K. Depletion of antibiotics from the mammary gland of goats. J. Dairy Sci. 1984, 67, 707–712. [Google Scholar] [CrossRef]
- Norbrook Laboratories, Ltd. Freedom of Information Summary, Supplemental New Animal Drug Application, NADA 065-010. Norocillin (Penecillin G Procaine Injectable Suspension) Cattle, Sheep, Swine, and Horses; To Revise the Currently Approved Formulation to Include Lecithin as a Surfactant; FOI—Norocillin NADA 065-010; Norbrook Laboratories, Ltd.: Newry, UK, 2010; pp. 1–27. [Google Scholar]
- Rule, R.; Lacchini, R.; Mordujovich, P.; Antonini, A. Evaluation of cefepime kinetic variables and milk production volume in goats. Arq. Bras. Med. Vet. Zootec. 2004, 56, 116–118. [Google Scholar] [CrossRef]
- Badillo, E.; Escudero, E.; Hernandis, V.; Galecio, J.S.; Marín, P. Pharmacokinetics of cefonicid in lactating goats after intravenous, intramuscular and subcutaneous administration, and after a long-acting formulation for subcutaneous administration. J. Vet. Pharmacol. Ther. 2020, 43, 50–56. [Google Scholar] [CrossRef]
- Yadav, K.; Jayachandran, C.; Singh, M.; Jha, H.; Sinha, S. Kinetics of intravenously administered ceftriaxone in lactating goat. Indian J. Anim. Sci. 2013, 70, 163–165. [Google Scholar]
- Ismail, M. Pharmacokinetics, urinary and mammary excretion of ceftriaxone in lactating goats. J. Vet. Med. Ser. A 2005, 52, 354–358. [Google Scholar] [CrossRef]
- Ziv, G.; Nouws, J. Clinical pharmacology of cephacetrile in ruminants. Zent. Vet. Reihe B 1977, 24, 798–811. [Google Scholar] [CrossRef]
- El Badawy, S.; Amer, A.; Kamel, G.; Eldeib, K.; Constable, P. Comparative pharmacokinetics using a microbiological assay and high performance liquid chromatography following intravenous administration of cefquinome in lactating goats with and without experimentally induced Staphylococcus aureus mastitis. Small Rumin. Res. 2015, 133, 67–76. [Google Scholar] [CrossRef]
- El Badawy, S.A.; Amer, A.M.; Kamel, G.M.; Eldeib, K.M.; Constable, P.D. Pharmacokinetics and pharmacodynamics of intramammary cefquinome in lactating goats with and without experimentally induced Staphylococcus aureus mastitis. J. Vet. Pharmacol. Ther. 2019, 42, 452–460. [Google Scholar] [CrossRef]
- Soback, S.; Ziv, G.; Bor, A.; Shapira, M. Pharmacokinetics of cephalexin glycinate in lactating cows and ewes. Zent. Vet. Reihe A 1988, 35, 755–760. [Google Scholar]
- Wetzel, R.K.; Stayer, P.A.; Wildman, E.E.; Randy, H.A. Depletion of cephapirin in goat’s milk following intramammary infusion. J. Dairy Sci. 1986, 69, 245. [Google Scholar]
- Courtin, F.; Wetzlich, S.; Gustafson, C.; Craigmill, A. Pharmacokinetics and milk residues of ceftiofur and metabolites in dairy goats. In Proceedings of the European Association of Veterinary Pharmacology and Toxicology 6th Congress, Edinburgh, UK, 7–11 August 1994; pp. 81–82. [Google Scholar]
- El-Rabbat, N.A.; Abdel-Wadood, H.M.; Sayed, M.; Mousa, H.S. High-performance liquid chromatographic determination and pharmacokinetic study of cefepime in goat plasma and milk after pre-column derivatization with Hg (I). J. Sep. Sci. 2010, 33, 2599–2609. [Google Scholar] [CrossRef]
- Rule, R.; Villagra, S.; Barrena, P.; Lacchini, R.; Reynaldni, F.J. Pharmacokinetics of ceftazidime administered to lactating and non-lactating goats. J. S. Afr. Vet. Assoc. 2011, 82, 219–223. [Google Scholar] [CrossRef][Green Version]
- European Medicines Agency Committee for Veterinary Medicinal Products. Ceftiofur (Extension to Ovine and Extrapolation to All Mammalian Species) Summary Report (4); European Medicines Agency Committee for Veterinary Medicinal Products: Amsterdam, The Netherlands, 2006; Volume 80785, pp. 1–3. [Google Scholar]
- Garrett, E.; Dirikolu, L.; Grover, G. Milk and serum concentration of ceftiofur following intramammary infusion in goats. J. Vet. Pharmacol. Ther. 2015, 38, 569–574. [Google Scholar] [CrossRef]
- Goudah, A.; Shin, H.; Shim, J.; El-Aty, A.A. Characterization of the relationship between serum and milk residue disposition of ceftriaxone in lactating ewes. J. Vet. Pharmacol. Ther. 2006, 29, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Rageh, A.H.; Abdel-Rahim, S.A.; Askal, H.F.; Saleh, G.A. Hydrophilic-interaction planar chromatography in ultra-sensitive determination of α-aminocephalosporin antibiotics. Application to analysis of cefalexin in goat milk samples using modified QuEChERS extraction technique. J. Pharm. Biomed. Anal. 2019, 166, 421–434. [Google Scholar] [CrossRef] [PubMed]
- Rule, R.; Cordiviola, C.; Vita, M.; Lacchini, R. Correlations between milk production and kinetic variables in milk of cephalothin administered to lactating goats. Vet. Med. 2004, 49, 370. [Google Scholar] [CrossRef]
- Rule, R.; Lacchini, R.; Román, A.G.; Antonini, A.; de Buschiazzo, P. Influence of feed type on the pharmacokinetics of cephalothin administered to lactating goats. Arch. Zootec. 2007, 56, 807–815. [Google Scholar]
- El-Sayed, M.; Atef, M.; El-Komy, A. Disposition kinetics of cephradine in normal and Escherichia coli infected goats. Dtsch. Tierarztl. Wochenschr. 1994, 101, 56–60. [Google Scholar]
- Singh, C.S.; Singh, S.D.; Singh, M.K.; Jayachandran, C. Disposition kinetics and distribution of ciprofloxacin in biological fluids of goats after intravenous administration. Indian J. Anim. Sci. 2001, 71, 635–637. [Google Scholar]
- El-Banna, H.; El-Sooud, K.A. Disposition kinetics of ciprofloxacin in lactating goats. Dtsch. Tierarztl. Wochenschr. 1998, 105, 35–38. [Google Scholar]
- Escudero, E.; Cárceles, C.; Fernandez-Varon, E.; Marin, P.; Benchaoui, H. Pharmacokinetics of danofloxacin 18% in lactating sheep and goats. J. Vet. Pharmacol. Ther. 2007, 30, 572–577. [Google Scholar] [CrossRef]
- Escudero, E.; Marín, P.; Cárceles, C.M.; Ramírez, M.J.; Fernández-Varón, E. Pharmacokinetic and milk penetration of a difloxacin long-acting poloxamer gel formulation with carboxy-methylcellulose in lactating goats. Vet. J. 2011, 188, 92–95. [Google Scholar] [CrossRef]
- El-Sooud, K.A. Influence of albendazole on the disposition kinetics and milk antimicrobial equivalent activity of enrofloxacin in lactating goats. Pharmacol. Res. 2003, 48, 389–395. [Google Scholar] [CrossRef]
- Marin, P.; Escudero, E.; Fernandez-Varon, E.; Espuny, A.; Titos, J.; Hernandis, V.; Carceles, C. Pharmacokinetics and milk penetration of an enrofloxacin long-acting poloxamer 407 gel formulation in lactating goats. J. Vet. Pharamcol. Ther. 2009, 32, 195–196. [Google Scholar]
- Narayan, J.P.; Kumar, N.; Jha, H.; Jayachandran, C. Effect of probenecid on kinetics of enrofloxacin in lactating goats after subcutaneous administration. Indian J. Exp. Biol. 2009, 47, 53–56. [Google Scholar]
- Haritova, A.; Lashev, L.; Pashov, D. Pharmacokinetics of enrofloxacin in lactating sheep. Res. Vet. Sci. 2003, 74, 241–245. [Google Scholar] [CrossRef]
- Marín, P.; Cárceles, C.M.; Escudero, E.; Fernández-Varón, E. Pharmacokinetics and milk penetration of ibafloxacin after intravenous administration to lactating goats. Can. J. Vet. Res. 2007, 71, 74. [Google Scholar]
- Goudah, A.; Abo-El-Sooud, K. Pharmacokinetics, urinary excretion and milk penetration of levofloxacin in lactating goats. J. Vet. Pharmacol. Ther. 2009, 32, 101–104. [Google Scholar] [CrossRef]
- Lorenzutti, A.; Litterio, N.; Himelfarb, M.; Zarazaga, M.D.P.; Andrés, M.S.; De Lucas, J. Pharmacokinetics, milk penetration and PK/PD analysis by Monte Carlo simulation of marbofloxacin, after intravenous and intramuscular administration to lactating goats. J. Vet. Pharmacol. Ther. 2017, 40, 629–640. [Google Scholar] [CrossRef]
- Shem-Tov, M.; Ziv, G.; Glickman, A.; Saran, A. Pharmacokinetics and penetration of marbofloxacin from blood into the milk of cows and ewes. J. Vet. Med. Ser. A 1997, 44, 511–519. [Google Scholar] [CrossRef]
- Fernández-Varón, E.; Villamayor, L.; Escudero, E.; Espuny, A.; Cárceles, C.M. Pharmacokinetics and milk penetration of moxifloxacin after intravenous and subcutaneous administration to lactating goats. Vet. J. 2006, 172, 302–307. [Google Scholar] [CrossRef]
- Goudah, A.; Cho, H.J.; Shin, H.C.; Shim, J.H.; Regmi, N.L.; Shimoda, M.; Abd El-Aty, A.M. Pharmacokinetics and milk distribution characteristics of orbifloxacin following intravenous and intramuscular injection in lactating ewes. J. Vet. Pharmacol. Ther. 2009, 32, 338–344. [Google Scholar] [CrossRef]
- El-Aty, A.M.A.; Goudah, A. Some pharmacokinetic parameters of pefloxacin in lactating goats. Vet. Res. Commun. 2002, 26, 553–561. [Google Scholar] [CrossRef]
- Real, R.; Egido, E.; Pérez, M.; Gonzalez-Lobato, L.; Barrera, B.; Prieto, J.; Alvarez, A.; Merino, G. Involvement of breast cancer resistance protein (BCRP/ABCG2) in the secretion of danofloxacin into milk: Interaction with ivermectin. J. Vet. Pharmacol. Ther. 2011, 34, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Perez, M.; Otero, J.A.; Barrera, B.; Prieto, J.G.; Merino, G.; Alvarez, A.I. Inhibition of ABCG2/BCRP transporter by soy isoflavones genistein and daidzein: Effect on plasma and milk levels of danofloxacin in sheep. Vet. J. 2013, 196, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Pulido, M.M.; Molina, A.J.; Merino, G.; Mendoza, G.; Prieto, J.G.; Alvarez, A.I. Interaction of enrofloxacin with breast cancer resistance protein (BCRP/ABCG2): Influence of flavonoids and role in milk secretion in sheep. J. Vet. Pharmacol. Ther. 2006, 29, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Shem-Tov, M.; Ziv, G.; Glickman, A.; Saran, A. Pharmacokinetics and penetration of danofloxacin from the blood into the milk of ewes. Vet. Res. 1997, 28, 571–579. [Google Scholar]
- Barrera, B.; González-Lobato, L.; Otero, J.A.; Real, R.; Prieto, J.G.; Álvarez, A.I.; Merino, G. Effects of triclabendazole on secretion of danofloxacin and moxidectin into the milk of sheep: Role of triclabendazole metabolites as inhibitors of the ruminant ABCG2 transporter. Vet. J. 2013, 198, 429–436. [Google Scholar] [CrossRef]
- Otero, J.A.; García-Mateos, D.; Alvarez-Fernández, I.; García-Villalba, R.; Espín, J.C.; Álvarez, A.I.; Merino, G. Flaxseed-enriched diets change milk concentration of the antimicrobial danofloxacin in sheep. BMC Vet. Res. 2018, 14, 14. [Google Scholar] [CrossRef]
- Marín, P.; Escudero, E.; Fernández-Varón, E.; Ramírez, M.; Cárceles, C. Pharmacokinetics and milk penetration of difloxacin after a long-acting formulation for subcutaneous administration to lactating goats. J. Dairy Sci. 2010, 93, 3056–3064. [Google Scholar] [CrossRef]
- Fernandez-Varon, E.; Escudero, E.; Marin, P.; Titos, J.; Espuny, A.; Carceles, C. Pharmacokinetics and milk penetration of enrofloxacin and its metabolite ciprofloxacin after subcutaneous administration to lactating goats. J. Vet. Pharmacol. Ther. 2009, 32, 196–197. [Google Scholar]
- European Medicines Agency Committee for Veterinary Medicinal Products. Enrofloxacin (Extension to Sheep, Rabbits and Lactating Cows) Summary Report (3); EMEA/MRL/389/98-Final; European Medicines Agency Committee for Veterinary Medicinal Products: Amsterdam, The Netherlands, 1998; Volume 389, pp. 1–5. [Google Scholar]
- Cárceles, C.M.; Villamayor, L.; Escudero, E.; Marín, P.; Fernández-Varón, E. Pharmacokinetics and milk penetration of moxifloxacin after intramuscular administration to lactating goats. Vet. J. 2007, 173, 452–455. [Google Scholar] [CrossRef]
- Soback, S.; Gips, M.; Bialer, M.; Bor, A. Effect of lactation on single-dose pharmacokinetics of norfloxacin nicotinate in ewes. Antimicrob. Agents Chemother. 1994, 38, 2336–2339. [Google Scholar] [CrossRef]
- Marín, P.; Escudero, E.; Fernández-Varón, E.; Cárceles, C. Pharmacokinetics and milk penetration of orbifloxacin after intravenous, subcutaneous, and intramuscular administration to lactating goats. J. Dairy Sci. 2007, 90, 4219–4225. [Google Scholar] [CrossRef]
- Delmas, J.; Chapel, A.; Gaudin, V.; Sanders, P. Pharmacokinetics of flumequine in sheep after intravenous and intramuscular administration: Bioavailability and tissue residue studies. J. Vet. Pharmacol. Ther. 1997, 20, 249–257. [Google Scholar] [CrossRef]
- Francis, P.G.; Wells, R.J. Flumequine. Residues Some Vet. Drugs Anim. Foods 1998, 41, 59–70. [Google Scholar]
- European Medicines Agency Committee for Veterinary Medicinal Products. Erythromycin Summary Report (2); EMEA/MRL/720/99-Final; European Medicines Agency Committee for Veterinary Medicinal Products: Amsterdam, The Netherlands, 2000; Volume 720, pp. 1–8. [Google Scholar]
- Goudah, A.; Sher Shah, S.; Shin, H.; Shim, J.; Abd El-Aty, A. Pharmacokinetics and mammary residual depletion of erythromycin in healthy lactating ewes. J. Vet. Med. Ser. A 2007, 54, 607–611. [Google Scholar] [CrossRef]
- Ramadan, A. Pharmacokinetics of tilmicosin in serum and milk of goats. Res. Vet. Sci. 1997, 62, 48–50. [Google Scholar] [CrossRef]
- MacNeil, J.D. Tilmicosin. Residues Some Vet. Drugs Anim. Foods 1997, 41, 106–118. [Google Scholar]
- Xu, S.; Arnold, D. Tilmicosin: Addendum to tho monographs prepared by the 47th meeting of the Committee and published in the FAO Food and Nutrition paper 41/9. Residues Some Vet. Drugs Anim. Foods 2008, 41, 1–37. [Google Scholar]
- Grismer, B.; Rowe, J.D.; Carlson, J.; Wetzlich, S.; Tell, L.A. Pharmacokinetics of tulathromycin in plasma and milk samples after a single subcutaneous injection in lactating goats (C apra hircus). J. Vet. Pharmacol. Ther. 2014, 37, 205–208. [Google Scholar] [CrossRef]
- Amer, A.; Constable, P.; Goudah, A.; El Badawy, S. Pharmacokinetics of tulathromycin in lactating goats. Small Rumin. Res. 2012, 108, 137–143. [Google Scholar] [CrossRef]
- Lin, Z.; Cuneo, M.; Rowe, J.D.; Li, M.; Tell, L.A.; Allison, S.; Carlson, J.; Riviere, J.E.; Gehring, R. Estimation of tulathromycin depletion in plasma and milk after subcutaneous injection in lactating goats using a nonlinear mixed-effects pharmacokinetic modeling approach. BMC Vet. Res. 2016, 12, 258. [Google Scholar] [CrossRef]
- Atef, M.; Youssef, S.; Atta, A.; El-Maaz, A. Disposition of tylosin in goats. Dtsch. Tierarztl. Wochenschr. 1991, 98, 451–453. [Google Scholar] [CrossRef]
- Ziv, G.; Sulman, F.G. Serum and milk concentrations of spectinomycin and tylosin in cows and ewes. Am. J. Vet. Res. 1973, 34, 329–333. [Google Scholar]
- Nagy, J.; Popelka, P.; Sokol, J.; Turek, P.; Neuschl, J. The excretion of tylosin residues in ewes milk after its experimental administration. Folia Vet. 2001, 45, 196–198. [Google Scholar]
- Ambros, L.; Montoya, L.; Kreil, V.; Waxman, S.; Albarellos, G.; Rebuelto, M.; Hallu, R.; Andres, M.S. Pharmacokinetics of erythromycin in nonlactating and lactating goats after intravenous and intramuscular administration. J. Vet. Pharmacol. Ther. 2007, 30, 80–85. [Google Scholar] [CrossRef]
- Ziv, G. Concentrations and residues of antibiotics in the milk of goats after parenteral and intramammary administration. In Proceedings of the Symposium Internacional de Ordeno Mecanico de Pequenos Rumiantes, Valladolid, Spain; 1984; pp. 513–528. [Google Scholar]
- Romanet, J.; Smith, G.W.; Leavens, T.L.; Baynes, R.E.; Wetzlich, S.E.; Riviere, J.E.; Tell, L.A. Pharmacokinetics and tissue elimination of tulathromycin following subcutaneous administration in meat goats. Am. J. Vet. Res. 2012, 73, 1634–1640. [Google Scholar] [CrossRef]
- Clothier, K.A.; Leavens, T.; Griffith, R.W.; Wetzlich, S.E.; Baynes, R.E.; Riviere, J.E.; Tell, L.A. Tulathromycin assay validation and tissue residues after single and multiple subcutaneous injections in domestic goats (Capra aegagrus hircus). J. Vet. Pharmacol. Ther. 2012, 35, 113–120. [Google Scholar] [CrossRef]
- Kellermann, M.; Huang, R.A.; Forbes, A.B.; Rehbein, S. Gamithromycin plasma and skin pharmacokinetics in sheep. Res. Vet. Sci. 2014, 97, 199–203. [Google Scholar] [CrossRef]
- European Medicines Agency Committee for Veterinary Medicinal Products. European Public MRL Assessment Report (EPMAR) Gamithromycin (All Ruminants Except Bovine Species.)12 December 2016; EMA/CVMP/454092/2016; European Medicines Agency Committee for Veterinary Medicinal Products EPMAR: Amsterdam, The Netherlands, 2016; Volume 454092, pp. 1–11. [Google Scholar]
- Atef, M.; El-Sooud, K.A.; Nahed, E.; Tawfik, M. Elimination of tilmicosin in lactating ewes. Dtsch. Tierarztl. Wochenschr. 1999, 106, 291–294. [Google Scholar]
- Parker, R.; Patel, R.; Mclaren, I.; Francis, P. Residues of tilmicosin in milk of sheep after subcutaneous administration. In Proceedings of the European Association for Veterinary Pharmacology and Toxicology, Edinburgh, UK, 7–11 August 1994; pp. 226–227. [Google Scholar]
- Elanco Animal Health. Freedom of Information Summary; Micotil 300 Injection (tilmicosin phosphate); Supplement to NADA 140-929; Date of Approval: 4 September 2002; FOI—Micotil NADA 140-929; Elanco Animal Health: Greenfield, IN, USA, 2002; pp. 1–14. [Google Scholar]
- European Medicines Agency Committee for Veterinary Medicinal Products. European Public MRL Assessment Report (EPMAR) Tulathromycin (Ovine and Caprine Species) 23 February 2015; EMA/CVMP/131462/2014; European Medicines Agency Committee for Veterinary Medicinal Products EPMAR: Amsterdam, The Netherlands, 2015; Volume 131462, pp. 1–11. [Google Scholar]
- Tunnicliff, E.; Swingle, K. Sulfonamide concentrations in milk and plasma from normal and mastitic ewes treated with sulfamethazine. Am. J. Vet. Res. 1965, 26, 920–927. [Google Scholar]
- Rasmussen, F. Mammary excretion of sulphonamides. Acta Pharmacol. Toxicol. 1958, 15, 139–148. [Google Scholar] [CrossRef]
- Akogwu, E.; Saganuwan, S.; Onyeyili, P. Effects of piroxicam on tissue distribution of sulfadimidine in west African dwarf male and female goats. Hum. Exp. Toxicol. 2018, 37, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Bevill, R.; Sharma, R.; Meachum, S.; Wozniak, S.; Bourne, D.; Dittert, L. Disposition of sulfonamides in food-producing animals: Concentrations of sulfamethazine and its metabolites in plasma, urine, and tissues of lambs following intravenous administration. Am. J. Vet. Res. 1977, 38, 973–977. [Google Scholar] [PubMed]
- Jha, H.; Banerjee, N. A note on distribution of sulphadimethoxine in blood, milk and urine of goats. Indian J. Anim. Sci. 1977, 47, 496–497. [Google Scholar]
- Yndestad, M.; Underdal, B. Residues of Sulfadimidine/Sulfanilamide and Sulfamethoxypyridazine in Sheep Tissue. Acta Vet. Scand. 1977, 18, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Bevill, R.F.; Koritz, G.D.; Dittert, L.W.; Bobrne, D.W. Disposition of sulfonamides in food-producing animals V: Disposition of sulfathiazole in tissue, urine, and plasma of sheep following intravenous administration. J. Pharm. Sci. 1977, 66, 1297–1300. [Google Scholar] [CrossRef]
- Righter, H.F.; Worthington, J.M.; Mercer, H.D. Tissue residue depletion of sulfamerazine in sheep. J. Agric. Food Chem. 1972, 20, 876–878. [Google Scholar] [CrossRef]
- Atef, M. Half-life, volume of distribution and acetylation of sulphamerazine in sheep. Zent. Vet. Reihe A 1978, 25, 585–591. [Google Scholar] [CrossRef]
- Bulgin, M.; Lane, V.M.; Archer, T.; Baggot, J.; Craigmill, A. Pharmacokinetics, safety and tissue residues of sustained-release sulfamethazine in sheep. J. Vet. Pharmacol. Ther. 1991, 14, 36–45. [Google Scholar] [CrossRef]
- Bevill, R.F. Application of Pharamcokinetics to the Study of Sulfonamide Behavior in Cattle, Sheep, and Swine. In Proceedings of the Symposium of Veterinary Pharmacology and Therapeutics, Baton Rouge, LA, USA, 13–15 March 1978; pp. 75–101. [Google Scholar]
- Paulson, G.; Struble, C.; Mitchell, A. Comparative metabolism of sulfamethazine [4-amino-N-(4,6-dimethyl-2-pyrimidinyl) benzenesulfonamide] in the rat, chicken, pig and sheep. In Mode of Action, Metabolism and Toxicology; Elsevier: Amsterdam, The Netherlands, 1983; pp. 375–380. [Google Scholar]
- Hashem, M.; Tayeb, F.; El-Mekkawi, T. The level of some sulphonamide preparations in tissues and blood of cocks and sheep. J. Egypt. Vet. Med. Assoc. 1980, 40, 5–11. [Google Scholar]
- Attaie, R.; Bsharat, M.; Mora-Gutierrez, A.; Woldesenbet, S. Short communication: Determination of withdrawal time for oxytetracycline in different types of goats for milk consumption. J. Dairy Sci. 2015, 98, 4370–4376. [Google Scholar] [CrossRef]
- Payne, M.; Babish, J.; Bulgin, M.; Lane, M.; Wetzlich, S.; Craigmill, A. Serum pharmacokinetics and tissue and milk residues of oxytetracycline in goats following a single intramuscular injection of a long-acting preparation and milk residues following a single subcutaneous injection. J. Vet. Pharmacol. Ther. 2002, 25, 25–32. [Google Scholar] [CrossRef]
- Fletouris, D.; Papapanagiotou, E. Tissue residue depletion of oxytetracycline after repeated intramuscular administration of Oxysentin 100 in sheep. J. Vet. Pharmacol. Ther. 2009, 32, 56–61. [Google Scholar] [CrossRef]
- Anifantakis, E.M. Excretion rates of antibiotics in milk of sheep and their effect on yogurt production. J. Dairy Sci. 1982, 65, 426–429. [Google Scholar] [CrossRef]
- Wells, R.J. Chlortetracycline. Residues Some Vet. Drugs Anim. Foods 1996, 41, 31–66. [Google Scholar]
- Jha, V.; Jayachandran, C.; Singh, M.; Singh, S. Pharmacokinetic data on doxycycline and its distribution in different biological fluids in female goats. Vet. Res. Commun. 1989, 13, 11–16. [Google Scholar] [CrossRef]
- Jayachandran, C.; Singh, M.; Singh, S.; Jha, H. Pharmacokinetics and distribution of minocydine in different biological fluids of goats after intravenous administration. Indian J. Anim. Sci. 2012, 69, 304–306. [Google Scholar]
- Immelman, A.; Ziv, G. Serum and milk concentrations of oxytetracycline after the administration of a long-acting formulation to sheep. J. S. Afr. Vet. Assoc. 1982, 53, 199–200. [Google Scholar]
- Arndt, T.; Robinson, D.; Holland, R.; Wetzlich, S.; Craigmill, A. Oxytetracycline residues in sheep following im dosing with a long-acting formulation. J. Vet. Pharmacol. Ther. 1997, 20, 314–315. [Google Scholar]
- Boatto, G.; Pau, A.; Palomba, M.; Arenare, L.; Cerri, R. Monitoring of oxytetracycline in ovine milk by high-performance liquid chromatography. J. Pharm. Biomed. Anal. 1999, 20, 321–326. [Google Scholar] [CrossRef]
- Craigmill, A.; Holland, R.; Robinson, D.; Wetzlich, S.; Arndt, T. Serum pharmacokinetics of oxytetracycline in sheep and calves and tissue residues in sheep following a single intramuscular injection of a long-acting preparation. J. Vet. Pharmacol. Ther. 2000, 23, 345–352. [Google Scholar] [CrossRef]
- Fletouris, D.J.; Papapanagiotou, E.P. A new liquid chromatographic method for routine determination of oxytetracycline marker residue in the edible tissues of farm animals. Anal. Bioanal. Chem. 2008, 391, 1189–1198. [Google Scholar] [CrossRef] [PubMed]
- Fletouris, D.J.; Papapanagiotou, E.P.; Nakos, D.S. Liquid chromatographic determination and depletion profile of oxytetracycline in milk after repeated intramuscular administration in sheep. J. Chromatogr. B 2008, 876, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Cinquina, A.L.; Longo, F.; Barchi, D.; Fagiolo, A.; Rosati, R.; Cozzani, R. Comparative pharmacokinetics of oxytetracycline in goat and sheep milk. Residues of Veterinary Drugs in Food. In Proceedings of the European Residue Conference IV, Veldhoven, The Netherlands, 8–10 May 2000; pp. 285–290. [Google Scholar]
- Hassan, S.A.A.; Shaddad, S.A.I.; El-Tayeb, L.B.; Omer, M.A.; Al-Nazawi, M.H.; Homeida, A.M. Detection of long-acting oxytetracycline residue levels in tissue of desert sheep following intramuscular injection. Int. J. Pharmacol. 2007, 3, 299–301. [Google Scholar]
- Reja, A.; Gonzalez, R.; Serrano, J.M.; Santiago, D.; Guimera, M.E.; Cano, M. Oxytetracycline and tetracycline residues in goat milk. In Proceedings of the European Associations of Veterinary Pharmacology and Toxicology 6th Congress, Edinburgh, UK, 7–11 August 1994. [Google Scholar]
- Reja Sanchez, A.; Gonzalez Pedrajas, R.; Serrano Caballero, J.M.; Santiago Laguna, D. Experimental determination of occurrence of tetracycline and oxytetracycline residues in goat milk. Blood/milk transference rates. Rev. Toxicol. 1995, 12, 29–34. [Google Scholar]
- Cinquina, A.L.; Gianetti, L.; Barchi, D.; Lanzi, S.; Longo, F.; Coresi, A.; Fagiolo, A.; Cozzani, R. Oxytetracycline residues in goat milk. Obiettivi Doc. Vet. 2002, 23, 37–40. [Google Scholar]
- Rule, R.; Moreno, L.; Serrano, J.M.; Garcia Roman, A.; Moyano, R.; Garcia, J. Pharmacokinetics and residues in milk of oxytetracyclines administered parenterally to dairy goats. Aust. Vet. J. 2001, 79, 492–496. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).