Genome-Wide Association Studies Reveal Candidate Genes Associated with Pigmentation Patterns of Single Feathers of Tianfu Nonghua Ducks
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Ethics Standards
2.2. Experimental Animals
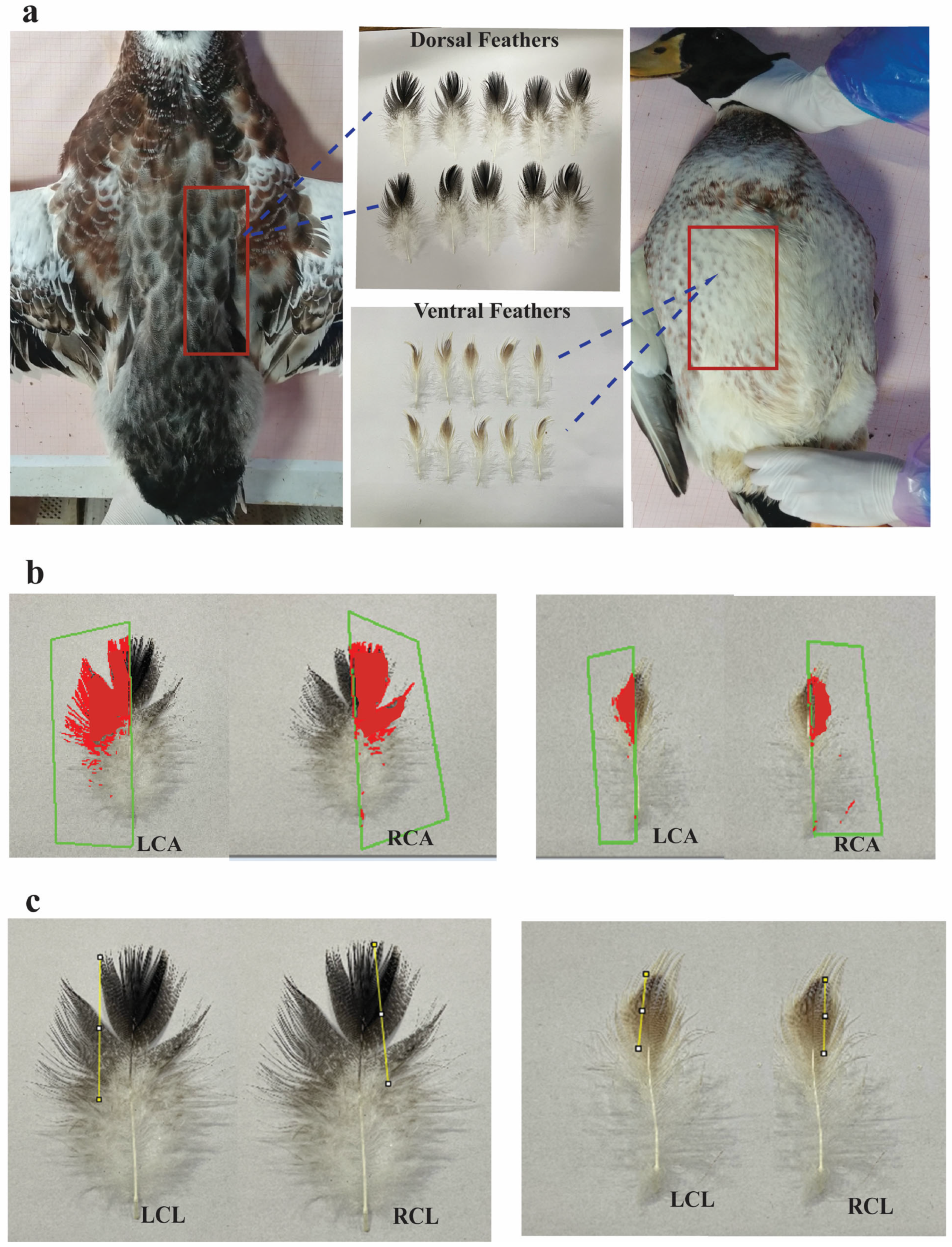
2.3. Phenotype Collection
2.4. Blood Sample Collection and DNA Extraction
2.5. Genomic Resequencing, Genotyping, and SNP Calling
2.6. Population Structure Analysis (PCA)
2.7. GWAS Analysis of the Single Feather Color Trait
2.8. Linkage Disequilibrium (LD) Analysis
2.9. Functional Annotation of Genes
2.10. Statistical Analyses
3. Results
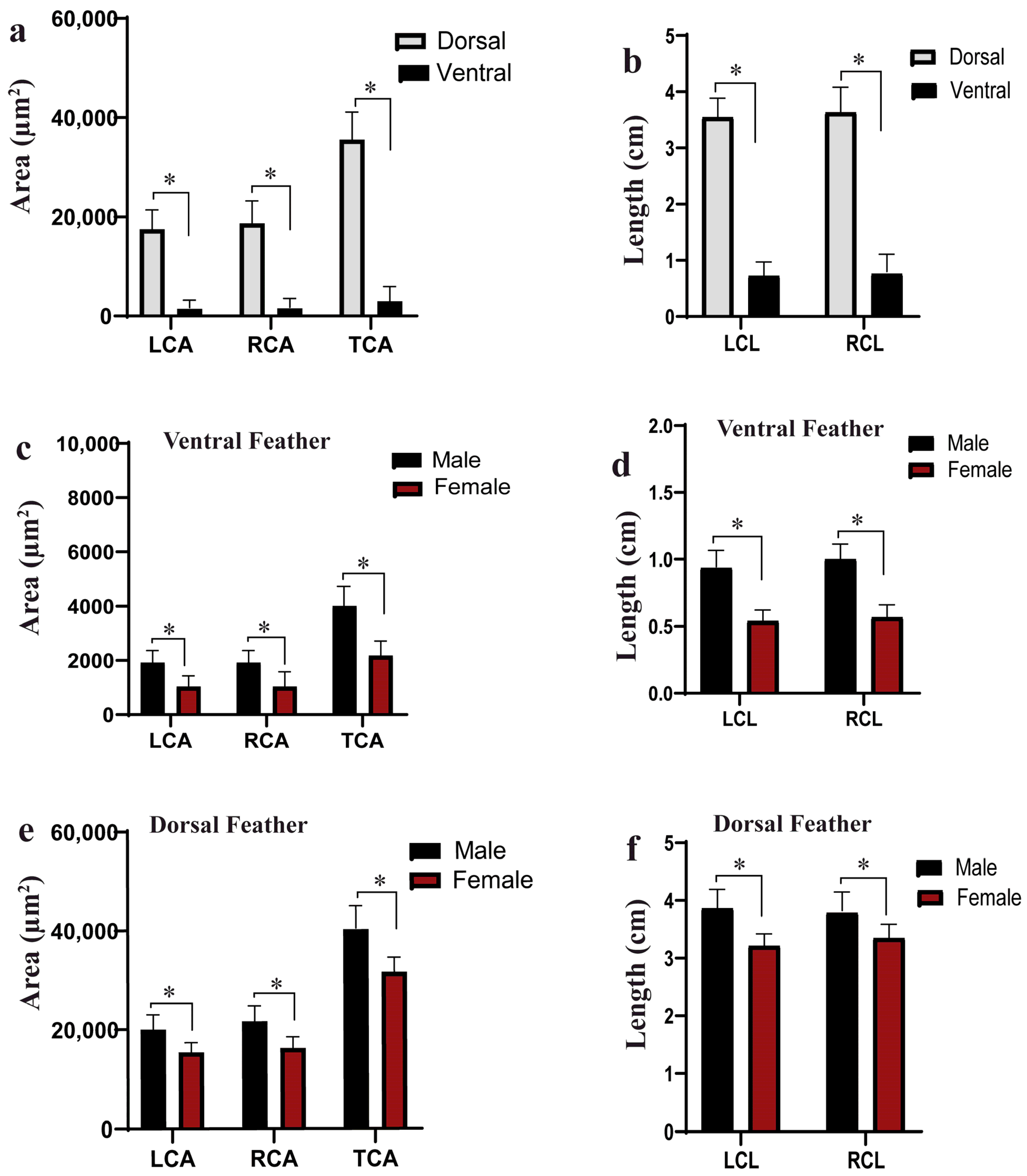
3.1. Phenotypic Parameters
3.2. Correlation and t-Test Analysis
3.3. Population Structure
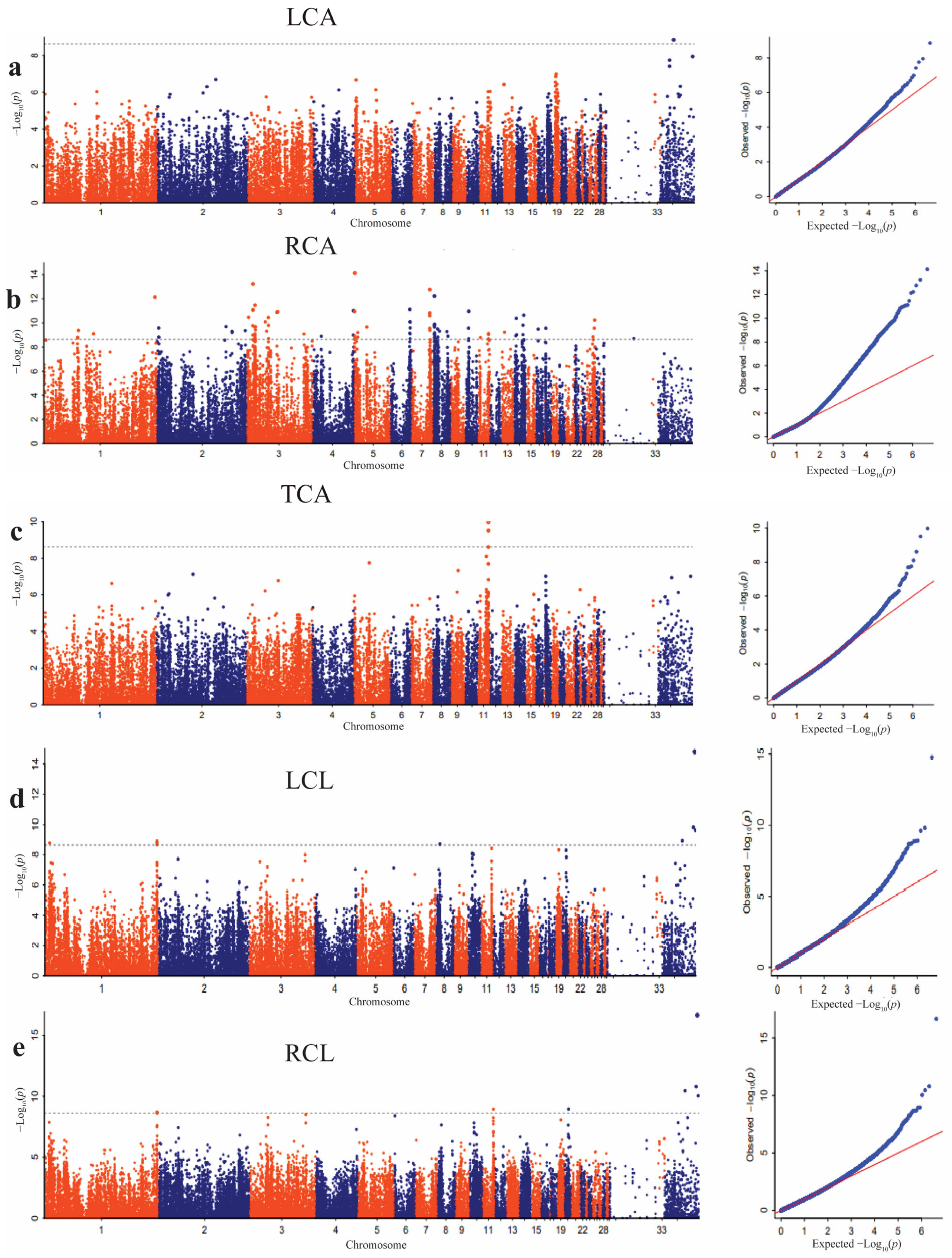
3.4. Significant SNPs Associated with Dorsal Feather Color Traits
3.5. Significant SNPs Associated with the Ventral Feather Color Traits
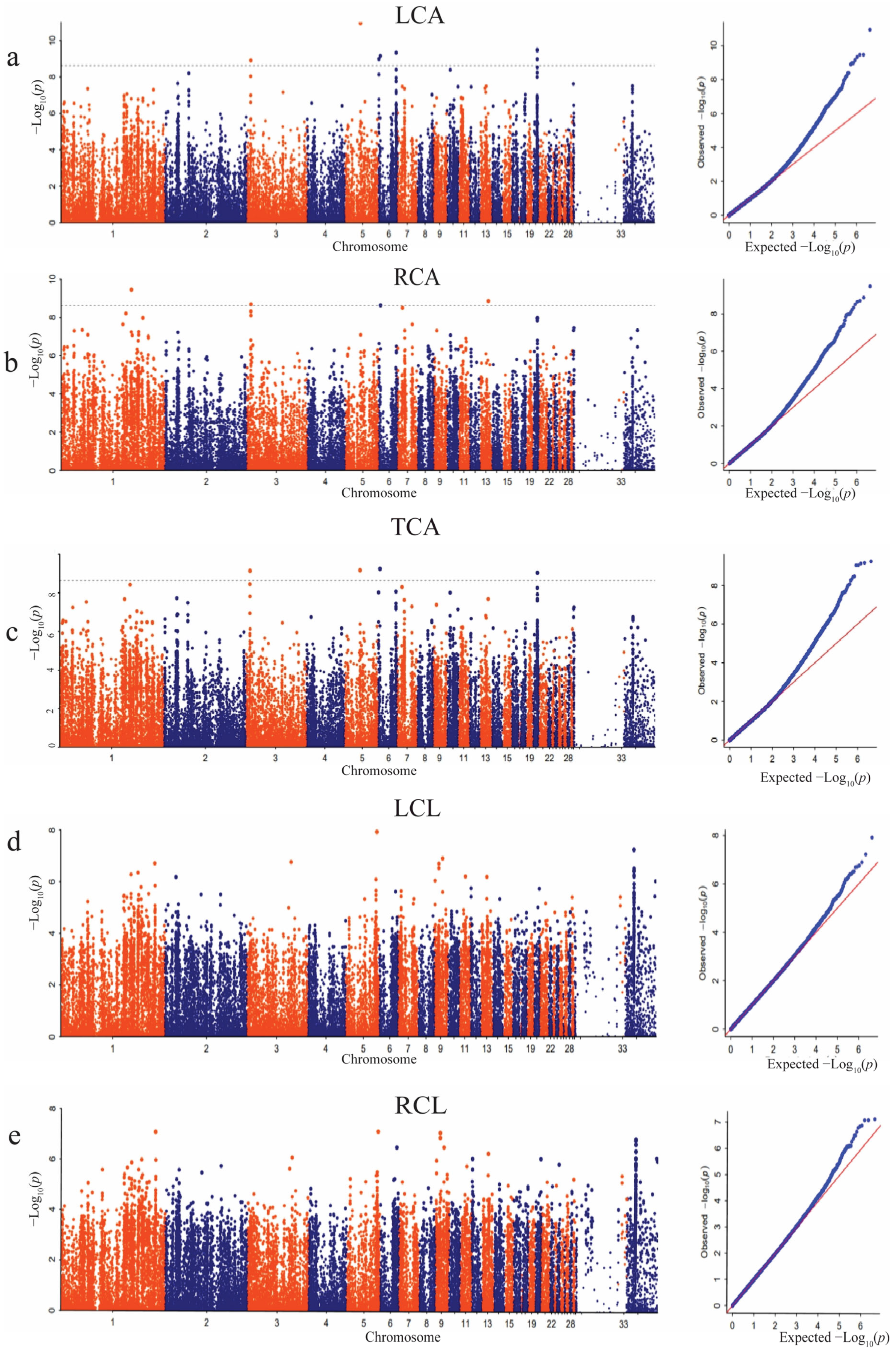
3.6. GWAS Results of Countershading in Tianfu Nonghua Duck
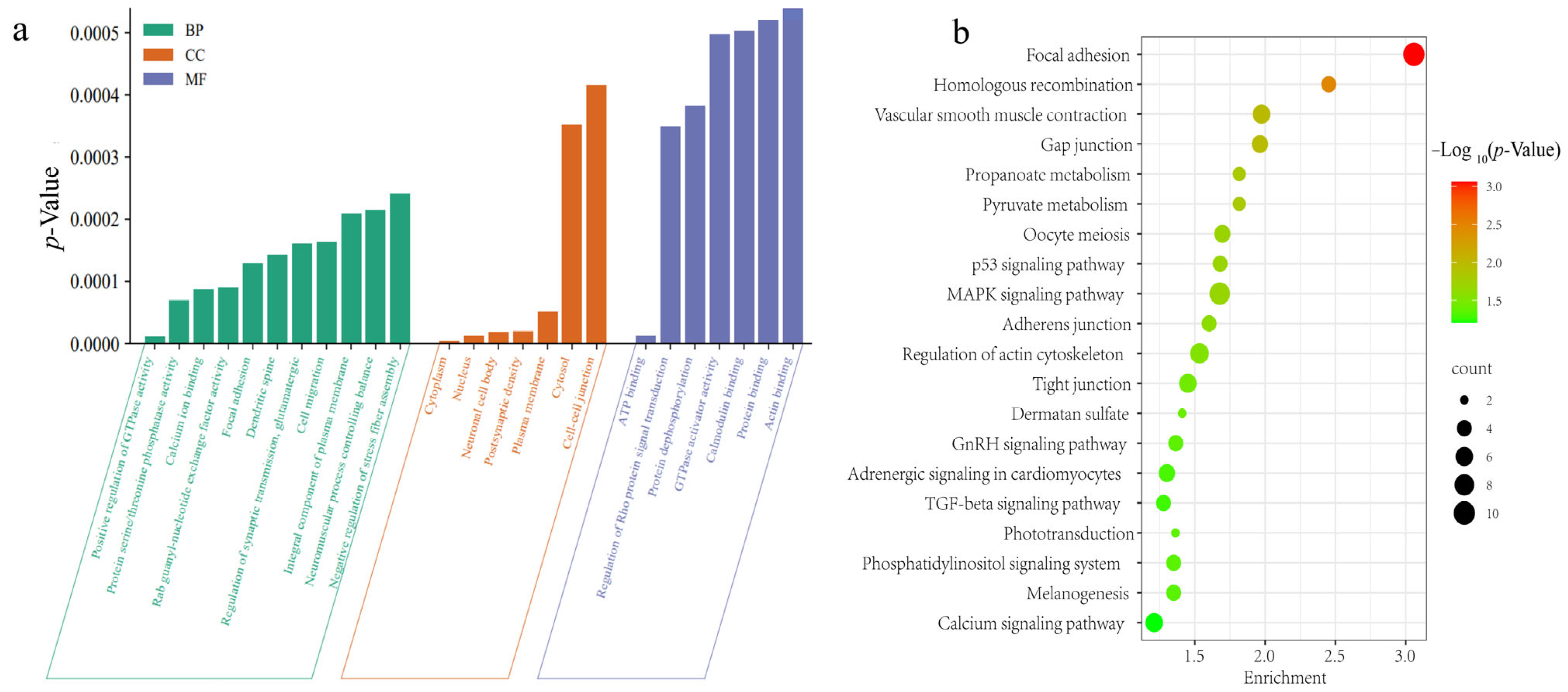
3.7. Functional Annotations of Genes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Chr | Chromosome |
| LCL | Left color length |
| RCL | Right color area |
| TCA | Total color area |
| LCL | Left color length |
| RCL | Right color length |
| BP | Biological process |
| CC | Cellular component |
| MF | Molecular function |
| IPP | Image Pro Plus |
| AOI | Area of interest |
| QTL | Quantitative trait loci |
| LD | Linkage disequilibrium |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| GO | Gene Ontology |
| DNA | Deoxyribonucleic acid |
| SNP | Single-nucleotide polymorphism |
| GWAS | Genome-wide association studies |
| VCF | Variant call format |
| PCA | Principal component analysis |
| Quantile–quantile | |
| ADP | Adenosine diphosphate |
References
- Xi, Y.; Xu, Q.; Huang, Q.; Ma, S.; Wang, Y.; Han, C.; Zhang, R.; Wang, J.; Liu, H.; Li, L. Genome-wide association analysis reveals that EDNRB2 causes a dose-dependent loss of pigmentation in ducks. BMC Genom. 2021, 22, 381. [Google Scholar] [CrossRef] [PubMed]
- Xi, Y.; Liu, H.; Li, L.; Xu, Q.; Liu, Y.; Wang, L.; Ma, S.; Wang, J.; Bai, L.; Zhang, R.; et al. Transcriptome Reveals Multi Pigmentation Genes Affecting Dorsoventral Pattern in Avian Body. Front. Cell Dev. Biol. 2020, 8, 560766. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Li, M.; Cheng, H.; Fan, W.; Yuan, Z.; Gao, Q.; Xu, Y.; Guo, Z.; Zhang, Y.; Hu, J.; et al. An intercross population study reveals genes associated with body size and plumage color in ducks. Nat. Commun. 2018, 9, 2648. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wu, Q.; Lin, R.; Chen, H.; Zhang, M.; Jiang, B.; Wang, Y.; Xue, P.; Gan, Q.; Shen, Y.; et al. Genome-wide association study for the primary feather color trait in a native Chinese duck. Front. Genet. 2023, 14, 1065033. [Google Scholar] [CrossRef]
- Kerje, S.; Lind, J.; Schütz, K.; Jensen, P.; Andersson, L. Melanocortin 1-receptor (MC1R) mutations are associated with plumage colour in chicken. Anim. Genet. 2003, 34, 241–248. [Google Scholar] [CrossRef]
- Hiragaki, T.; Inoue-Murayama, M.; Miwa, M.; Fujiwara, A.; Mizutani, M.; Minvielle, F.; Ito, S. Recessive black Is Allelic to the yellow Plumage Locus in Japanese Quail and Associated With a Frameshift Deletion in the ASIP Gene. Genetics 2008, 178, 771–775. [Google Scholar] [CrossRef]
- Li, S.; Wang, C.; Yu, W.; Zhao, S.; Gong, Y. Identification of Genes Related to White and Black Plumage Formation by RNA-Seq from White and Black Feather Bulbs in Ducks. PLoS ONE 2012, 7, e36592. [Google Scholar] [CrossRef]
- Ma, S.; Li, P.; Liu, H.; Xi, Y.; Xu, Q.; Qi, J.; Wang, J.; Li, L.; Wang, J.; Hu, J.; et al. Genome-wide association analysis of the primary feather growth traits of duck: Identification of potential Loci for growth regulation. Poult. Sci. 2023, 102, 102243. [Google Scholar] [CrossRef]
- Yang, J.; Lee, S.H.; Goddard, M.E.; Visscher, P.M. GCTA: A tool for genome-wide complex trait analysis. Am. J. Hum. Genet. 2011, 88, 76–82. [Google Scholar] [CrossRef]
- Yu, W.; Wang, C.; Xin, Q.; Li, S.; Feng, Y.; Peng, X.; Gong, Y. Non-synonymous SNPs in MC1R gene are associated with the extended black variant in domestic ducks (Anas platyrhynchos). Anim. Genet. 2013, 44, 214–216. [Google Scholar] [CrossRef]
- Sultana, H.; Seo, D.W.; Park, H.B.; Choi, N.R.; Hoque, R.; Bhuiyan, S.A.; Heo, K.N.; Lee, S.H.; Lee, J.H. Identification of MC1R SNPs and their Association with Plumage Colors in Asian Duck. J. Poult. Sci. 2017, 54, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, S.; Akiyama, T. Mechanisms of Feather Structural Coloration and Pattern Formation in Birds. In Pigments, Pigment Cells and Pigment Patterns; Springer: Singapore, 2021; pp. 343–366. [Google Scholar] [CrossRef]
- Saino, N.; Romano, M.; Rubolini, D.; Teplitsky, C.; Ambrosini, R.; Caprioli, M.; Canova, L.; Wakamatsu, K. Sexual dimorphism in melanin pigmentation, feather coloration and its heritability in the barn swallow (Hirundo rustica). PLoS ONE 2013, 8, e58024. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.H.; Cheong, K.A.; Lee, T.R.; Lee, A.Y. PDZK1 upregulation in estrogen-related hyperpigmentation in melasma. J. Investig. Dermatol. 2012, 132, 2622–2631. [Google Scholar] [CrossRef] [PubMed]
- Crawford, K.; Oliver, P.L.; Agnew, T.; Hunn, B.H.M.; Ahel, I. Behavioural Characterisation of Macrod1 and Macrod2 Knockout Mice. Cells 2021, 10, 368. [Google Scholar] [CrossRef]
- Kim, M.Y.; Lee, H.E.; Im, M.; Lee, Y.; Kim, C.D.; Lee, J.H.; Seo, Y.J. Effect of adenosine on melanogenesis in b16 cells and zebrafish. Ann. Dermatol. 2014, 26, 209–213. [Google Scholar] [CrossRef]
- Chen, C.F.; Foley, J.; Tang, P.C.; Chuong, C. Development, Regeneration, and Evolution of Feathers. Annu. Rev. Anim. Biosci. 2015, 3, 169–195. [Google Scholar] [CrossRef] [PubMed]
- Jenni, L.; Ganz, K.; Milanesi, P.; Winkler, R. Determinants and constraints of feather growth. PLoS ONE 2020, 15, e0231925. [Google Scholar] [CrossRef]
- Prum, R.O.; Williamson, S. Reaction-diffusion models of within-feather pigmentation patterning. R. Soc. Proc. B 2002, 269, 781–792. [Google Scholar] [CrossRef]
- Guo, H.; Xing, Y.; Liu, Y.; Luo, Y.; Deng, F.; Yang, T.; Yang, K.; Li, Y. Wnt/β-catenin signaling pathway activates melanocyte stem cells in vitro and in vivo. J. Dermatol. Sci. 2016, 83, 45–51. [Google Scholar] [CrossRef]
- Guo, H.; Yang, K.; Deng, F.; Ye, J.; Xing, Y.; Li, Y.; Lian, X.; Yang, T. Wnt3a promotes melanin synthesis of mouse hair follicle melanocytes. Biochem. Biophys. Res. Commun. 2012, 420, 799–804. [Google Scholar] [CrossRef]
- Dongkyun, K.; Jinsoo, S.; Jin, E.J. Wnt-3 and Wnt-3a play different region-secific roles in neural crest development in avians. Cell Biol. Int. 2010, 34, 763–768. [Google Scholar] [CrossRef]
- Chiang, S.K.; Chang, W.C.; Chen, S.E.; Chang, L.C. DOCK1 Regulates Growth and Motility through the RRP1B-Claudin-1 Pathway in Claudin-Low Breast Cancer Cells. Cancers 2019, 11, 1762. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Ma, Y.; Yu, X.; Mort, R.L.; Lindsay, C.R.; Stevenson, D.; Strathdee, D.; Insall, R.H.; Chernoff, J.; Snapper, S.B.; et al. Rac1 drives melanoblast organization during mouse development by orchestrating pseudopod- driven motility and cell-cycle progression. Dev. Cell 2011, 21, 722–734. [Google Scholar] [CrossRef] [PubMed]
- Steffen, A.; Ladwein, M.; Dimchev, G.A.; Hein, A.; Schwenkmezger, L.; Arens, S.; Ladwein, K.I.; Margit Holleboom, J.; Schur, F.; Victor Small, J.; et al. Rac function is crucial for cell migration but is not required for spreading and focal adhesion formation. J. Cell Sci. 2013, 126, 4572–4588. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Li, Y.; Jia, Y.; Ding, B.; Yu, J. Rab1A knockdown represses proliferation and promotes apoptosis in gastric cancer cells by inhibition of mTOR/p70S6K pathway. Arch. Biochem. Biophys. 2020, 685, 108352. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, F.; Wu, K.; Wu, W.; Wu, H.; Zhang, W. Targeting dipeptidyl peptidase 8 genes inhibits proliferation, migration and invasion by inhibition of cyclin D1 and MMP2MMP9 signal pathway in cervical cancer. J. Gene Med. 2018, 20, e3056. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Shao, X.; Xu, M.; Wang, J.; Kuai, X.; Zhang, L.; Wu, J.; Zhou, C.; Mao, J. Rab1A promotes proliferation and migration abilities via regulation of the HER2/AKT-independent mTOR/S6K1 pathway in colorectal cancer. Oncol. Rep. 2019, 41, 2717–2728. [Google Scholar] [CrossRef] [PubMed]
- Ishida, M.; Ohbayashi, N.; Fukuda, M. Rab1A regulates anterograde melanosome transport by recruiting kinesin-1 to melanosomes through interaction with SKIP. Sci. Rep. 2015, 5, 8238. [Google Scholar] [CrossRef]
- Zhang, X.; Zhu, T.; Wang, L.; Lv, X.; Yang, W.; Qu, C.; Li, H.; Wang, H.; Ning, Z.; Qu, L. Genome-Wide Association Study Reveals the Genetic Basis of Duck Plumage Colors. Genes 2023, 14, 856. [Google Scholar] [CrossRef]
- Videira, I.F.; Moura, D.F.; Magina, S. Mechanisms regulating melanogenesis. An. Bras. Dermatol. 2013, 88, 76–83. [Google Scholar] [CrossRef]
- Hume, A.N.; Ushakov, D.S.; Tarafder, A.K.; Ferenczi, M.A.; Seabra, M.C. Rab27a and MyoVa are the primary Mlph interactors regulating melanosome transport in melanocytes. J. Cell Sci. 2007, 120, 3111–3122. [Google Scholar] [CrossRef]
- Chen, D.; Guo, J.; Miki, T.; Tachibana, M.; Gahl, W.A. Molecular cloning and characterization of rab27a and rab27b, novel human rab proteins shared by melanocytes and platelets. Biochem. Mol. Med. 1997, 60, 27–37. [Google Scholar] [CrossRef]
- Rzepka, Z.; Buszman, E.; Beberok, A.; Wrześniok, D. From tyrosine to melanin: Signaling pathways and factors regulating melanogenesis. Postepy Hig. Med. Dosw. (Online) 2016, 70, 695–708. [Google Scholar] [CrossRef] [PubMed]
- Jimbow, K.; Gomez, P.F.; Toyofuku, K.; Chang, D.; Miura, S.; Tsujiya, H.; Park, J.S. Biological role of tyrosinase related protein and its biosynthesis and transport from TGN to stage I melanosome, late endosome, through gene transfection study. Pigment. Cell Res. 1997, 10, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Ajami, K.; Abbott, C.A.; Obradovic, M.; Gysbers, V.; K?Hne, T.; Mccaughan, G.W.; Gorrell, M.D. Structural requirements for catalysis, expression, and dimerization in the CD26/DPIV gene family. Biochemistry 2003, 42, 694–701. [Google Scholar] [CrossRef]
- Okondo, M.C.; Johnson, D.C.; Sridharan, R.; Go, E.B.; Chui, A.J.; Wang, M.S.; Poplawski, S.E.; Wu, W.; Liu, Y.; Lai, J.H.; et al. DPP8 and DPP9 inhibition induces pro-caspase-1-dependent monocyte and macrophage pyroptosis. Nat. Chem. Biol. 2017, 13, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Cieslak, M.; Reissmann, M.; Hofreiter, M.; Ludwig, A. Colours of domestication. Biol. Rev. Camb. Philos Soc. 2011, 86, 885–899. [Google Scholar] [CrossRef]
- Boutchueng-Djidjou, M.; Belleau, P.; Bilodeau, N.; Fortier, S.; Bourassa, S.; Droit, A.; Elowe, S.; Faure, R.L. A type 2 diabetes disease module with a high collective influence for Cdk2 and PTPLAD1 is localized in endosomes. PLoS ONE 2018, 13, e0205180. [Google Scholar] [CrossRef]
- Sabath, K.; Stäubli, M.L.; Marti, S.; Leitner, A.; Moes, M.; Jonas, S. INTS10-INTS13-INTS14 form a functional module of Integrator that binds nucleic acids and the cleavage module. Nat. Commun. 2020, 11, 3422. [Google Scholar] [CrossRef]
- Pfleiderer, M.M.; Galej, W.P. Structure of the catalytic core of the Integrator complex. Mol. Cell 2021, 81, 1246–1259.e1248. [Google Scholar] [CrossRef]
- Gaudel, C.; Soysouvanh, F.; Leclerc, J.; Bille, K.; Husser, C.; Montcriol, F.; Bertolotto, C.; Ballotti, R. Regulation of Melanogenesis by the Amino Acid Transporter SLC7A5. J. Investig. Dermatol. 2020, 140, 2253–2259.e2254. [Google Scholar] [CrossRef] [PubMed]
- Neuillé, M.; Malaichamy, S.; Vadalà, M.; Michiels, C.; Condroyer, C.; Sachidanandam, R.; Srilekha, S.; Arokiasamy, T.; Letexier, M.; Démontant, V.; et al. Next-generation sequencing confirms the implication of SLC24A1 in autosomal-recessive congenital stationary night blindness. Clin. Genet. 2016, 89, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Carrigan, M.; Duignan, E.; Malone, C.P.; Stephenson, K.; Saad, T.; McDermott, C.; Green, A.; Keegan, D.; Humphries, P.; Kenna, P.F.; et al. Panel-Based Population Next-Generation Sequencing for Inherited Retinal Degenerations. Sci. Rep. 2016, 6, 33248. [Google Scholar] [CrossRef] [PubMed]
- Neelagandan, N.; Gonnella, G.; Dang, S.; Janiesch, P.C.; Miller, K.K.; Küchler, K.; Marques, R.F.; Indenbirken, D.; Alawi, M.; Grundhoff, A.; et al. TDP-43 enhances translation of specific mRNAs linked to neurodegenerative disease. Nucleic Acids Res. 2019, 47, 341–361. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Jiang, Y.; Wang, Z.; Bi, Y.; Chen, G.; Bai, H.; Chang, G. Genome-Wide Analysis Identifies Candidate Genes Encoding Feather Color in Ducks. Genes 2022, 13, 1249. [Google Scholar] [CrossRef] [PubMed]
- Desingu, P.A.; Nagarajan, K. Detection of beak and feather disease virus in India and its implications. Transbound Emerg Dis 2022, 69, e3469–e3478. [Google Scholar] [CrossRef] [PubMed]
- Gunnarsson, U.; Hellström, A.R.; Tixier-Boichard, M.; Minvielle, F.; Bed’hom, B.; Ito, S.; Jensen, P.; Rattink, A.; Vereijken, A.; Andersson, L. Mutations in SLC45A2 cause plumage color variation in chicken and Japanese quail. Genetics 2007, 175, 867–877. [Google Scholar] [CrossRef]
- Li, R.; Wang, X.; Wang, Y.; Liu, D.; Zhang, Y.; Liu, Y.; Niu, X.; Han, R.; Li, H.; Jiang, R.; et al. Research Note: Combined analysis of BSA-seq based mapping and RNA-seq reveals candidate genes associated with sub-Columbian plumage in H line chickens. Poult. Sci. 2023, 102, 102665. [Google Scholar] [CrossRef]
- Li, R.; Wang, Y.; Liu, Y.; Li, D.; Tian, Y.; Liu, X.; Kang, X.; Li, Z. Effects of SLC45A2 and GPNMB on Melanin Deposition Based on Transcriptome Sequencing in Chicken Feather Follicles. Animals 2023, 13, 2608. [Google Scholar] [CrossRef]
- Yang, L.; Mo, C.; Shen, W.; Du, X.; Akbar Bhuiyan, A.; Li, L.; Li, N.; Gong, Y.; Li, S. The recessive C locus in the MITF gene plays a key regulatory role in the plumage colour pattern of duck (Anas platyrhynchos). Br. Poult. Sci. 2019, 60, 105–108. [Google Scholar] [CrossRef]
- Lan, T.; Li, Y.; Wang, Y.; Wang, Z.C.; Mu, C.Y.; Tao, A.B.; Gong, J.L.; Zhou, Y.; Xu, H.; Li, S.B.; et al. Increased endogenous PKG I activity attenuates EGF-induced proliferation and migration of epithelial ovarian cancer via the MAPK/ERK pathway. Cell Death Dis. 2023, 14, 39. [Google Scholar] [CrossRef]
- Wang, X.; Nudds, R.L.; Palmer, C.; Dyke, G.J. Size scaling and stiffness of avian primary feathers: Implications for the flight of Mesozoic birds. J. Evol. Biol. 2012, 25, 547–555. [Google Scholar] [CrossRef]
- Luo, J.; Li, H.; Xiu, J.; Zeng, J.; Feng, Z.; Zhao, H.; Li, Y.; Wei, W. Elevated ZNF704 expression is associated with poor prognosis of uveal melanoma and promotes cancer cell growth by regulating AKT/mTOR signaling. Biomark. Res. 2023, 11, 38. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, H.; Goda, M.; Futahashi, R.; Kelsh, R.; Akiyama, T. (Eds.) Pigments, Pigment Cells and Pigment Patterns; Springer Science and Business Media LLC: Dordrecht, The Netherlands, 2021. [Google Scholar]
- Cal, L.; Suarez-Bregua, P.; Comesaña, P.; Owen, J.; Braasch, I.; Kelsh, R.; Cerdá-Reverter, J.M.; Rotllant, J. Countershading in zebrafish results from an Asip1 controlled dorsoventral gradient of pigment cell differentiation. Sci. Rep. 2019, 9, 3449. [Google Scholar] [CrossRef] [PubMed]
- Cal, L.; Suarez-Bregua, P.; Braasch, I.; Irion, U.; Kelsh, R.; Cerdá-Reverter, J.M.; Rotllant, J. Loss-of-function mutations in the melanocortin 1 receptor cause disruption of dorso-ventral countershading in teleost fish. Pigment. Cell Melanoma Res. 2019, 32, 817–828. [Google Scholar] [CrossRef] [PubMed]

| Trait | Chr | Position | −Log10(p) | REF | ALT | Gene |
|---|---|---|---|---|---|---|
| RCA | Chr3 | 11106947 | 16.928888 | C | A | CEP68 |
| Chr1 | 203570330 | 12.13432 | C | A | TENM4 | |
| chr3 | 39330988 | 10.44989 | T | C | LOC101794856 | |
| chr3 | 39517020 | 9.794113 | A | G | LOC101795266 | |
| chr3 | 54977399 | 10.88282 | C | T | ARID1B | |
| chr3 | 56297389 | 10.91372 | C | T | AIG1 | |
| chr4 | 16056736 | 8.885082 | T | C | PURG | |
| TCA | chr11 | 19801194 | 9.981866 | G | A | INTS14 |
| chr11 | 19843385 | 9.511955 | C | T | HACD3 | |
| chr11 | 19791413 | 8.619472 | G | A | DENND4A | |
| LCL | chr1 | 204456821 | 8.877868 | G | A | GUCY2F |
| chr1 | 204456829 | 8.858735 | A | G | GUCY2F | |
| chr1 | 204456818 | 8.714282 | G | A | GUCY2F | |
| chr8 | 7523520 | 8.704662 | T | A | KCNT2 | |
| RCL | chr20 | 7466942 | 8.96404 | T | C | MYL10 |
| chr11 | 19969233 | 8.951823 | T | C | MEGF11 | |
| chr1 | 204456818 | 8.719696 | G | A | GUCY2F |
| Chr | Position | p-Value | REF | ALT | Gene |
|---|---|---|---|---|---|
| chr11 | 19796049 | 0.000434 | G | A | DPP8 |
| chr11 | 19797796 | 0.023138 | T | A | HACD3 |
| chr11 | 19822622 | 0.000182 | G | T | INTS14 |
| chr11 | 19843073 | 0.000000 | G | A | SLC24A1 |
| chr11 | 19877499 | 0.001007 | G | A | DENND4A |
| Trait | Chr | Pos | −Log10(p) | REF | ALT | Gene |
|---|---|---|---|---|---|---|
| LCA | chr5 | 30633861 | 10.94515 | G | A | CCDC86 |
| chr11 | 9151176 | 10.9137 | G | A | LOC119718058 | |
| chr3 | 7298398 | 10.51952 | G | A | MACROD2 | |
| chr6 | 817341 | 8.982077 | T | G | PRKG1 | |
| RCA | chr1 | 140523618 | 9.463266 | A | G | - |
| chr13 | 15682406 | 8.862459 | T | C | LOC113845051 | |
| chr3 | 7616580 | 8.693525 | T | C | MACROD2 | |
| chr6 | 3619856 | 8.638454 | C | G | LOC101796401 | |
| TCA | chr6 | 3619856 | 9.240001 | C | G | LOC101796401 |
| chr5 | 30633861 | 9.162349 | G | A | CCDC86 | |
| chr3 | 7616580 | 9.131946 | T | C | MACROD2 |
| Parameters | Male, n = 156 | Female, n = 171 | t | 95% CI | p | |||
|---|---|---|---|---|---|---|---|---|
| M | SD | M | SD | Lower | Upper | |||
| DLCA: VLCA | 16.89 | 52.34 | 40.49 | 117.30 | −2.04 | −46.3263 | −0.87246 | 0.04 |
| DRCA: VRCA | 19.21 | 93.69 | 49.58 | 181.71 | −1.66 | −66.4726 | 5.732883 | 0.09 |
| DTCA: VTCA | 12.93 | 19.10 | 48.66 | 181.08 | −2.14 | −68.5645 | −2.90038 | 0.03 |
| DTCA:TFA/VTCA: TFA | 7.39 | 16.45 | 37.22 | 148.99 | −2.17 | −56.86 | −2.81 | 0.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Twumasi, G.; Wang, H.; Xi, Y.; Qi, J.; Li, L.; Bai, L.; Liu, H. Genome-Wide Association Studies Reveal Candidate Genes Associated with Pigmentation Patterns of Single Feathers of Tianfu Nonghua Ducks. Animals 2024, 14, 85. https://doi.org/10.3390/ani14010085
Twumasi G, Wang H, Xi Y, Qi J, Li L, Bai L, Liu H. Genome-Wide Association Studies Reveal Candidate Genes Associated with Pigmentation Patterns of Single Feathers of Tianfu Nonghua Ducks. Animals. 2024; 14(1):85. https://doi.org/10.3390/ani14010085
Chicago/Turabian StyleTwumasi, Grace, Huazhen Wang, Yang Xi, Jingjing Qi, Liang Li, Lili Bai, and Hehe Liu. 2024. "Genome-Wide Association Studies Reveal Candidate Genes Associated with Pigmentation Patterns of Single Feathers of Tianfu Nonghua Ducks" Animals 14, no. 1: 85. https://doi.org/10.3390/ani14010085





