Abstract
Porifera are filter-feeding organisms known to bioaccumulate different contaminants in their tissues. The presence of mercury (Hg) has been reported in different Mediterranean species, mainly collected in the southern coast of France. In the present study, mercury concentrations in the tissue of the sponges of Montecristo and Giglio, two islands of Tuscany Archipelago National Park (TANP), are presented for the first time. Analyses of total mercury content were performed by Direct Mercury Analyzer. Statistical differences have been reported in the Hg concentrations of species collected in both islands, but they do not appear related to the anthropic impacts of the islands. Among the collected species, a high intra- and inter-variability have been recorded, with Cliona viridis showing the lowest concentration (0.0167–0.033 mg·kg−1 dry weight), and Chondrosia reniformis and Sarcotragus spinosulus the highest (0.57 ± 0.15 and 0.64 ± 0.01 mg·kg−1 dry weight, respectively). The variability of Hg measured did not allow us to identify sponges as bioindicators of toxic elements. Anyway, these results improve knowledge on the ecosystem of the TANP, underlining the species-specificity of metal concentrations for Porifera, and providing additional data to address the main input of the Marine Strategy guidelines to protect coasts, seas and oceans.
1. Introduction
Mercury (Hg) is considered to be one of the most toxic heavy metals due to its persistence in the environment, bioaccumulation in organisms and biomagnification in the trophic chain [1,2]. Atmospheric inputs of Hg have tripled during the last 150 years, being two-thirds of its actual concentration from anthropogenic sources [2,3]. Traces of mercury have been found in all the compartments of the ecosphere (atmosphere, hydrosphere, lithosphere and biosphere) [4,5,6]. In aquatic environments, it is transformed by chemical and biological reactions in organomercury compounds, as methylmercury (MeHg), the most toxic mercury species, which can be bioaccumulated more than other trace elements along the trophic chain [6]. Mercury can have many different effects on a wide range of organisms, both vertebrates, e.g., References [7,8,9], and invertebrates, e.g., References [10,11,12]. For all these reasons, Hg is listed in the European Water Framework Directive (WFD 2000/60/EC) as a priority substance representing a risk for the good chemical status for the aquatic environment, with a Maximum Allowable Concentration (MAC) of 0.07 μg.L−1.
The presence of mercury in the Mediterranean Sea has been documented since the 1970s [13,14]. The principal mercury input (94%) was recognized in rivers’ dischargements, while only 5.5% was related to direct industrial wastewater and 0.5% to domestic sewage [15]. The Mediterranean area presents a high number of natural deposits of mercury distributed along the coasts of many countries, containing about 65% of the world’s cinnabar (HgS) deposits [15,16]. Particularly, in Tuscany (Italy), the levels of Hg in different environmental matrices are derived from both a natural contribution of the mineralization and the pollution caused by the huge exploitation of the area of Mount Amiata [17]. In fact, Mount Amiata, located in the south of Tuscany, is part of the geologic anomaly of the Mediterranean basin and it is characterized by a large cinnabar deposit [16,18]. Moreover, it is well known from ARPAT (acronym for Regional Agency for Environmental Protection of Tuscany) and ISPRA (acronym for Higher Institute for Environmental Protection and Research) technical reports, and scientific papers [19], the presence of mercury in the waters and organisms of the Tuscany coast and island. From 2012 to 2017, high concentrations of mercury have been reported in water, sediment and biota (as Posidonia oceanica, Mytilus galloprovincialis and different fish species) in the Tuscan Archipelago islands, e.g., References [20,21,22,23,24].
Many studies have been conducted worldwide on the bioaccumulation and the effects of mercury and its organic compounds in edible species, such as bivalves, cephalopods, decapods crustaceans and fish [6,25,26,27]. Although, other filter-feeding organisms, such as polychaetes, tunicates, sponges and barnacles, have been proposed as bioindicators in shallow waters [28], but these taxa are still little used for this purpose. Especially, sponges satisfy all the characteristics listed in References [29,30] for a suitable bioindicator and have been recommended by many authors, e.g., References [31,32], and by the WFD as possible monitors for heavy metals. Being filter-feeders, sponges can filter a large amount of water, and they can collect and accumulate many different contaminants (such as hydrocarbons, organochlorinated compounds, heavy metals, etc.) in their tissues, which are present in the water column both in the soluble and particulate phases [10,28]. Moreover, the level of the bioaccumulation in their tissues is a function of the contaminants’ concentration in the water [33]. The presence of heavy metals in sponges can affect their physiology and survival [28,34,35]. Only a few authors have investigated the presence of Hg in sponges and most of the studies have been conducted on samples, mainly on the genus Spongia, collected in the coast of Marseille [10,36,37,38].
The current paper presents, for the first time, total mercury content in the tissue of the sponges of Montecristo and Giglio, two islands of Tuscany Archipelago National Park (TANP). The study addresses two hypotheses: (1) sponges collected show detectable total mercury contents, and (2) there are any differences in Hg concentrations between the sponge species collected in both studied islands. Our results point out that sponges have detectable Hg concentrations, showing a high inter- and intra-specific variability. This variability could also be responsible of the lack of trend in the Hg concentrations between the specimens collected from the more anthropic Giglio and the Integral Reserve of Montecristo. Anyway, the results obtained with statistical analyses give new important insights for the area on the differences among Hg concentration in sponges.
2. Materials and Methods
2.1. Study Area
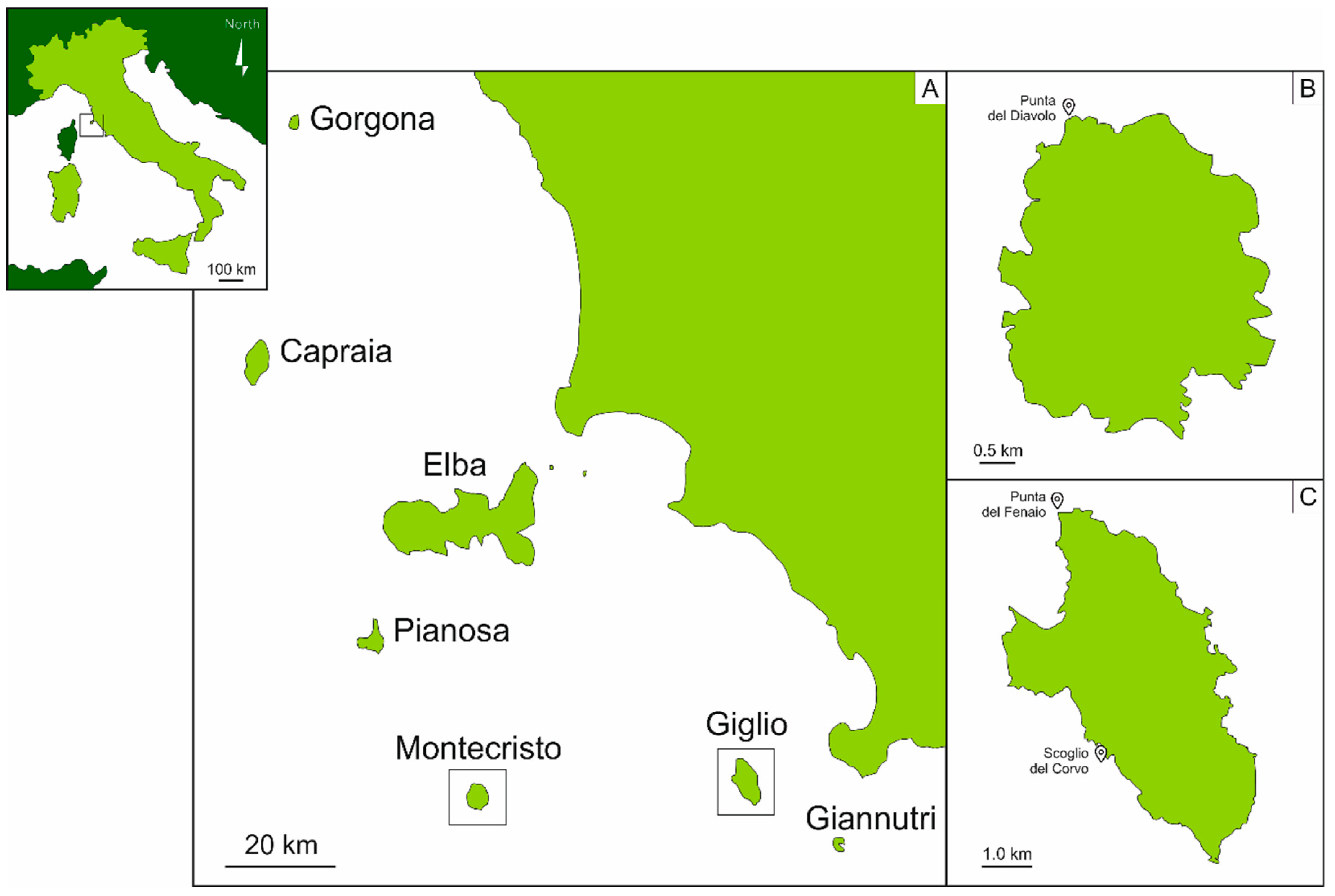
The study was carried out in June 2019 in Montecristo and Giglio islands (Tuscany, Italy) (Figure 1).
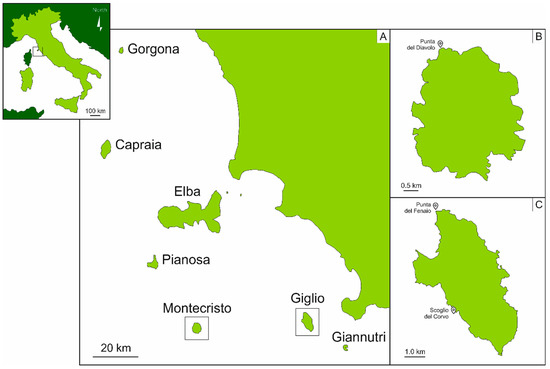
Figure 1.
Map of the Tuscan Archipelago National Park (TANP) (A), Montecristo Island (B) and Giglio Island (C). The sampling sites are indicated in the islands’ maps.
The Montecristo Island (42.3317° N, 10.3083° E) (Figure 1B) is an uninhabited and isolated island, sub-circular in shape, fourth in size after Elba, Giglio and Capraia islands, and completely mountainous, reaching a height of 645 m above sea-level with an almost constant slope of 25° [39,40,41,42]. Its history and geography distinguish the island from the others: during the Quaternary period, Montecristo remained in contact with the Tuscany’s littoral for a shorter period than the other islands, ending up located 63 km from the mainland, on the limit of the continental shelf, closer to Corsica than to the mainland [39].
In 1971, Montecristo was established as an Integral Nature Reserve, and in 1988, was declared a biogenetic nature reserve by the Council of Europe. Moreover, the island has the status of Special Protection Area (SPA, Directive 79/409/EEC). The land and the adjacent waters, up to 1 km offshore, are controlled by the Coast Guard and the Carabinieri Corps [41,43]. Bathing, diving, fishing, mooring and circumnavigating are forbidden, while landing, berthing and scientific activities are allowed, only under specific conditions and with the permission of the Territorial Office for Biodiversity of the Carabinieri Corps of Follonica [41]. Due to the high protection which Montecristo has undergone, the island is considered one of the most pristine and best-preserved sites of the Mediterranean Sea, being previously identified as a reference site for the ecological quality assessment of the western Mediterranean benthic assemblages on rocky bottoms [44,45].
The Giglio Island (42.3603° N, 10.9229° E) (Figure 1C), being the second in size after Elba, is included in the southern group of the Archipelago, only 14 km from the Italian coast [42,46]. It is an oval-shaped island, characterized by a mountain chain along the north–south axis, and it reaches a height of 496 m above the sea-level. Contrary to Montecristo, the island hosts 1500 residents, spread out among the villages of Giglio Castello, Giglio Porto and Campese [42]. Only 40% of the island is included inside the protected area, while adjacent waters and many terrestrial areas do not have any protection (DPR 22 July 1996) [47]. The areas included in the legislation of the TANP are divided into four zones: (1) zone A of strict reserve, including five rocks (Cappa, Corvo, Mezzo Franco, Pietra Bona, and Le Scole), (2) zone B of general reserve, (3) zone C of general protection and (4) zone D of socio-economic promotion [42]. In general, Giglio is considered to have undergone anthropogenic pressures [47], due to the high flow of tourists during Summer [42], and to be more exposed to potential stress deriving from the mainland.
2.2. Samples Collection and Identification
Surveys were conducted by SCUBA diving and eighteen sponge samples were collected between 5 and 40 m, nine on the hard bottom assemblages of Punta del Diavolo (42°21′02.46″ N; 10°17′55.86″ E) in the Montecristo Island (TANP permission #00068010) (Figure 1B), and nine between Punta del Fenaio (42°23′21.54″ N; 10°52′48.18″ E) and Scoglio del Corvo (42°20′17.76″ N; 10°53′21.36″ E) in the Giglio Island (Figure 1C). The low number of samples for this preliminary study is strictly subordinated to protect and respect the island ecosystem, which needs to be protected even in the case of scientific research.
Samples were immediately frozen on dry ice, and then stored at −20 °C until analysis.
Among the samples, fourteen species of Demospongiae have been identified: Agelas oroides (Schmidt, 1864), Axinella damicornis (Esper, 1794), Cliona viridis (Schmidt, 1862), Haliclona (Halichoclona) fulva (Topsent, 1893), Haliclona (Soestella) mucosa (Griessinger, 1971), Penares euastrum (Schmidt, 1868) and Sarcotragus spinosulus Schmidt, 1862 in Montecristo, and Chondrosia reniformis Nardo, 1847, C. viridis, Crambe crambe (Schmidt, 1862), H. (H.) fulva, Hemimycale columella (Bowerbank, 1874), Hymedesmia (Hymedesmia) baculifera (Topsent, 1901) and Petrosia (Petrosia) ficiformis (Poiret, 1789) in Giglio. For the species P. euastrum and C. reniformis, three samples each were collected; therefore, we denominated them with the name of the species followed by the number 1, 2 or 3 between brackets.
2.3. Samples Treatment and Mercury Analysis
A clean room laboratory ISO 14644-1 Class 6, with areas at ISO Class 5 under laminar flow, was used for all laboratory activities. After the identification, samples were weighted (laboratory analytical balance, AT261 Mettler Toledo Greifensee, Switzerland, readability 0.01 mg, repeatability standard deviation (SD) = 0.015 mg) and cleaned with ultrapure water (A10 Milli-Q system, Merk Millipore, Bedford, MA, USA). The acid-cleaning procedures, used for all the laboratory materials, were performed as described in References [48,49].
After cutting samples into small pieces, sponges were lyophilized (Edwards EF4 modulyo, Crawley, Sussex, England), minced, homogenized and divided in aliquots of about 0.02 g each. Analyses of total mercury content (THg) were performed by Direct Mercury Analyzer (DMA-1 Milestone, Sorisole (BG), Italy), as described in Reference [50]. Briefly, the total mercury content was quantified by thermal decomposition amalgamation atomic absorption spectrometry at 253.7 nm. The calibration curve method was used for the quantification of Hg content. All measurements were replicated at least 4 times.
2.4. Accuracy
Quality assurance and quality control were assessed by processing blank samples and certified reference material (dogfish muscle DORM-2, NRCC; Ottawa, ON, Canada). The experimental values obtained for Hg in blanks are negligible compared with the metal content in sponge tissue (<1%). For DORM-2 analysis (n = 8), Hg content (4.58 ± 0.10 mg·kg−1) is in agreement with the certified value (4.43 ± 0.05 mg·kg−1) and no statistically significant differences were observed (p-value > 0.05, Student’s T test, STATGRAPHICS 18 Centurion, 2018).
2.5. Data Analyses
Data are expressed as arithmetic mean ± standard deviation (SD) of the performed replications. Statistical analyses of differences within organisms were performed using the analysis of variance (one-way ANOVA) after testing the homogeneity of the variance with Levene’s test [51]. In case of heteroscedasticity, we applied the non-parametric Kruskal–Wallis analysis of variance. Depending on the resulting statistics, post-hoc comparison was eventually performed with the Bonferroni correction, always considering a significant level of 0.05. All graphs and statistical analyses were performed using STATGRAPHICS (STATGRAPHICS Centurion 2018, Statgraphics Technologies Inc., The Plains, VA, USA).
3. Results
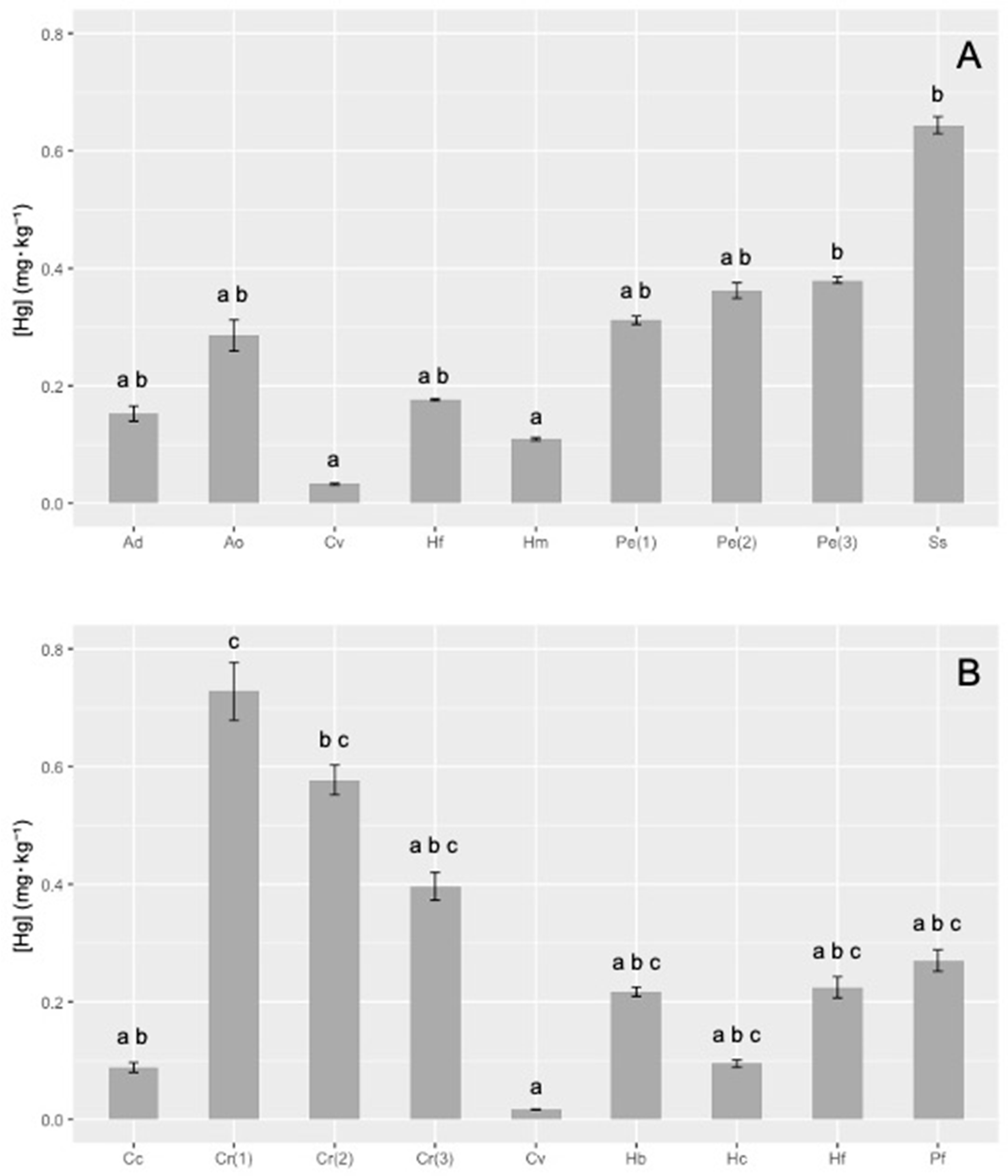
THg, expressed on a dry weight (dw) basis, in sponges collected in the area of Montecristo and Giglio islands, are reported in Figure 2A,B, respectively.
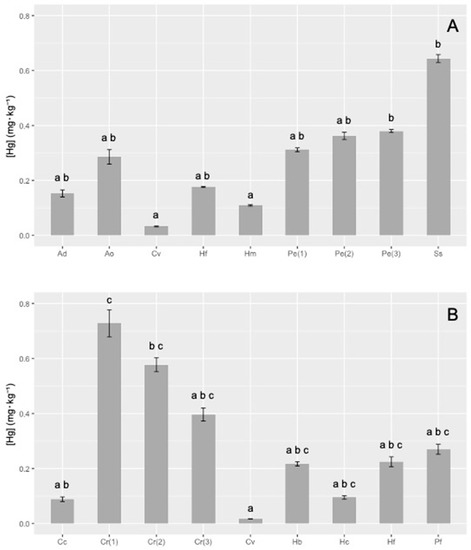
Figure 2.
Mercury levels (± standard deviation (SD)) in the sponges collected in Montecristo (A) and Giglio (B) islands. In each graph, different letters (a, b, c) indicate statistically significant differences among sponges (Kruskal–Wallis test, p < 0.05). Ad = Axinella damicornis; Ao = Agelas oroides; Cv = Cliona viridis; Hf = Haliclona (Halichoclona) fulva; Hm = Haliclona (Soestella) mucosa; Pe = Penares euastrum; Ss = Sarcotragus spinosulus; Cc = Crambe crambe; Cr = Chondrosia reniformis; Hb = Hymedesmia (Hymedesmia) baculifera; Hc = Hemimycale columella; Pf = Petrosia (Petrosia) ficiformis.
Total mercury showed high variability in sponges coming from the marine area of Montecristo island, ranging from 0.033 to 0.64 mg·kg−1, with the minimum content in Cliona viridis (0.033 ± 0.001 mg·kg−1) and the maximum (20-fold higher) in Sarcotragus spinosulus (0.64 ± 0.01 mg·kg−1). Values ranging from about 0.11 to 0.29 mg·kg−1 were recorded in Agelas oroides, Axinella damicornis, Haliclona (Halichoclona) fulva and H. (Soestella) mucosa, whereas slightly higher values were found for the three samples of Penares euastrum (1), (2) and (3) (0.35 ± 0.03 mg·kg−1), that showed similar concentration among them. Statistical analysis of the data showed a significant difference (Kruskal–Wallis test, p < 0.05) between C. viridis vs. P. euastrum (3) and S. spinosulus, H. (S.) mucosa vs. P. euastrum (3) and S. spinosulus (Figure 2A).
A high variability of THg content was also found in sponges collected in the marine area of Giglio Island: total Hg ranged from 0.017 to 0.73 mg·kg−1 dw. Even in this case, the minimum content was measured in C. viridis (0.0167 ± 0.0003 mg·kg−1), which showed values from 10 to 70 times lower than other species. On the other hand, the three samples of Chondrosia reniformis (1), (2) and (3) showed the highest concentrations (ranging from 0.39 to 0.73 mg·kg−1). In the other sponges, the THg content ranged from ≈0.1 (Crambe crambe, Hemimycale columella) to about 0.22 mg·kg−1 in H. (H.) fulva, Hymedesmia baculifera and Petrosia (P.) ficiformis. The Kruskal–Wallis test highlighted significant differences (p < 0.05) between C. reniformis (1) vs. C. crambe, C. viridis vs. C. reniformis (1) and C. reniformis (3) (Figure 2B).
Since samples of C. viridis and H. (H.) fulva were found in both islands, differences in the THg content between the two islands have been investigated, to compare the possible influence of different sites. C. viridis showed a higher THg in Montecristo island (Kruskal–Wallis test, p < 0.05), while the concentration of mercury in H. (H.) fulva was higher in Giglio (Kruskal–Wallis test, p < 0.05).
4. Discussion
Samples show the presence of mercury both in Montecristo and Giglio. Although statistical differences in the Hg concentrations have been detected in the sponge species (Cliona viridis and Haliclona (H.) fulva) collected in both islands of the Tuscan Archipelago, the lack of a trend excludes a possible influence of the site. In fact, even though since 1971 Montecristo has been an Integral Nature Reserve considered a pristine area not interested by mechanic impacts, such as anchoring and diving disturbances, fishing, etc., its water can still be affected by other different impacts, such as climate change and alien species, e.g., References [52,53], and the presence of different pollutants [20,21,22,23] due to water circulation [54].
Currently, no law limit is defined for Hg in sponges and few data are available on Hg concentration in this phylum, most of which regard species and genera different from those collected in this study, e.g., References [28,55,56]. THg values of Sarcotragus spinosulus, measured by us, are comparable with the concentrations found in other species belonging to the subclass Keratosa, characterized by a skeleton of spongin fibers [57] such as Scalarispongia scalaris (Schmidt, 1862) (cited as Cacospongia scalaris) [37], Spongia (Spongia) lamella (Schulze, 1879) (cited as S. agaricina) [36,37], S. (S.) nitens (Schmidt, 1862) (cited as S. nitens) [36] and S. (S.) officinalis (Linnaeus, 1759) (cited as S. officinalis) [10,36,37,38] (Table 1). THg values of Chondrosia reniformis recorded in specimens from Montecristo and Giglio are similar to the ones recorded in Reference [37] for the same species collected in different localities near Marseille (Table 1). On the other hand, results obtained for Agelas oroides and Cliona viridis in Reference [37] are higher than our THg values for both species (Table 1). Perez et al. [37] collected samples of Cliona viridis in polluted and non-polluted areas, and he did not find any differences in the Hg concentrations. Therefore, also the difference reported between our specimen and the one of Reference [37] could be related not only to the species-specificity but also to the individual specificity of sponges, with a high intra- and inter-specific variability [58,59].

Table 1.
Selection of literature data for mercury concentrations in sponges.
Although it is known that some heavy metals (e.g., copper, lead and vanadium) have effects on sponges, increasing their fission frequency, inducing changes in cellular aggregation and reducing growth and filtration rates [34,35], no information is available for Hg. However, some studies showed, from laboratory experiments, that mercury can cause death, inhibition of gemmule formation and malformation in gemmoscleres in the freshwater sponge Ephydatia fluviatilis (Linnaeus, 1759) (0.001 to 1.000 mg·kg−1) [60], and it can arrest movement of single sponge cells of Scopalina lophyropoda (Schmidt, 1862) (1 and 5 µg·kg−1), which tended to be rounded without pseudopodia [61], while a high concentration of MeHg (0.6 mg·kg−1) induces apoptosis in tissue of Geodia cydonium (Linnaeus, 1767) [62].
The highest THg values were found in Chondrosia reniformis and Sarcotragus spinosulus, collected in Giglio and Montecristo islands, respectively. These two species share the absence of a mineral skeleton, showing C. reniformis, a dispersal fibrillary collagen, and S. spinosulus, an organic skeleton made of spongin [57]. Three papers on Antarctic and Mediterranean species [59,63,64] showed that spicules accumulate only a small part of heavy metals compared to the sponge tissues; while in Spongia spp., the skeletal spongin fibers can trap and consequently concentrate metals, such as Fe, Pb, Cr, Zn and V [37,65]. These studies suggest that a collagenous skeleton instead of a mineral one can bioaccumulate a higher concentration of heavy metals. On the other hand, the mercury concentrations recorded for C. viridis, which is the only boring species and symbiotic with zooxanthellae [66], were the lowest among all the species collected. It is possible that the association with zooxanthellae influences the accumulation capacity of the sponge, as also pointed out in Reference [37]. It has been demonstrated that C. viridis can vary its filtration rates depending on the photosynthetic activities of its zooxanthellae [67], and this could partially explain why our samples present low concentrations of THg in their tissues.
The aforementioned intra- and inter-specific variability suggested by our observations and other authors [37,38] (Table 1) appear to not always be in agreement with the distance from the main source of pollutants. This can be a result of physiological and skeletal differences among sponge species. For example, the diversity of morphology of the aquiferous system could influence the size range of particles that can be filtered as well as the filtration rates [37]. Moreover, sponges are long-living organisms, with variable rate of growth, and it is very difficult to age each individual [36], therefore it is possible that low concentrations of heavy metals found in one organism could be related to its young age. In different sponge species, Clathria (Clathria) prolifera (Ellis and Solander, 1786) (cited as Microciona prolifera) [68], Suberites domuncula (Olivi, 1792) [69] and Spongia (Spongia) officinalis [10], authors found the presence of metallothioneins (MT) and metallothionein-like proteins (MTLPs), which are known to be used in the sequestration of metals in some invertebrates [70,71], and a positive correlation between MTLP concentrations and some heavy metals’ (Cu, Zn and Hg) concentrations has been established [10].
Moreover, sponges can host complex communities of microorganisms, including bacteria, cyanobacteria and fungi, in their tissues [72]. The relationships between sponges and their microbiota can be defined as a symbiosis, in which microorganism communities can take part in the metabolic cycles and exchange different metabolites with their host [73]. It has been demonstrated that many bacteria, which can contribute up to 40% of the sponge biomass, are resistant to different antibiotics and pollutants, including Persistent Organic Pollutants (POPs) and heavy metals, such as Cu, Pb, Co, Cd, Zn, Ni, Hg and their organic compounds, e.g., References [72,74,75,76]. Moreover, some studies asserted that sponges and the associated bacteria can potentially be applied in the bioremediation of aquatic environments contaminated by mercury [74,75]. Therefore, the microbiota also seems to play an important role in the bioaccumulation of heavy metals in sponge tissues, suggesting the necessity of deepening the investigation towards this aspect.
5. Conclusions
In conclusion, our results are the first data about total Hg concentration in sponges from the TANP and suggest that these metazoa could accumulate toxic elements in coastal waters. On the other hand, the high variability of concentrations in THg measured in all specimens in both islands did not allow us to identify sponges as bioindicators of toxic elements. However, further studies with a higher number of sponge samples are needed to understand in which compartment (skeleton, tissue or microbiota) they accumulate the heaviest metals, analyzing the main effects on the histology and physiology of this group. Anyway, these results improve the knowledge on the ecosystem of the TANP, pointing out the species/individual-specificity of metal concentrations for Porifera and the key role of the organic skeleton, tissue and microbiota. Moreover, these data provide additional environmental information on the Tuscany Archipelago to address the main input of international guidelines on the Marine Strategy to protect and clean up coasts, seas and oceans.
Author Contributions
Conceptualization, A.A. and S.P.; Formal analysis, A.A.; Funding acquisition, S.P.; Investigation, C.R. and F.G.; Project administration, S.P.; Resources, C.R. and D.P.; Supervision, B.C.; Writing—original draft, C.R. and A.A.; Writing—review and editing, D.P., B.C., C.T., S.I. and S.P. All authors have read and agreed to the published version of the manuscript.
Funding
This study has been conducted with the financial support of PADI FOUNDATION (grant number #32694) and Università Politecnica delle Marche.
Acknowledgments
The authors are grateful to Isla Negra diving, for the logistic assistance during samplings, and to the Tuscan Archipelago National Park, for the authorization of sampling activities.
Conflicts of Interest
The authors declare they have no conflict of interest.
References
- Ramalhosa, E.C.D.; Pereira, E.; Duarte, A. Mercury behaviour in the water column of an impacted coastal lagoon: Ria de Aveiro (Portugal) as a case study. In Focus on Water Resource Research; Heikkinen, E., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2008; pp. 41–86. ISBN 978-1-60456-093-0. [Google Scholar]
- Gomes, R.M. Mercury Effects in Natural Populations of Sea Anemone Actinia equina. Ph.D. Thesis, Department of Biology, University of Aveiro, Aveiro, Portugal, 2011. [Google Scholar]
- Morel, F.M.; Kraepiel, A.M.; Amyot, M. The chemical cycle and bioaccumulation of mercury. Annu. Rev. Ecol. Syst. 1998, 29, 543–566. [Google Scholar] [CrossRef]
- Ullrich, S.M.; Tanton, T.W.; Abdrashitova, S.A. Mercury in the Aquatic Environment: A Review of Factors Affecting Methylation. Crit. Rev. Environ. Sci. Technol. 2001, 31, 241–293. [Google Scholar] [CrossRef]
- Rajar, R.; ČETINA, M.; Horvat, M.; Žagar, D. Mass balance of mercury in the Mediterranean Sea. Mar. Chem. 2007, 107, 89–102. [Google Scholar] [CrossRef]
- Droghini, E.; Annibaldi, A.; Prezioso, E.; Tramontana, M.; Frapiccini, E.; De Marco, R.; Illuminati, S.; Truzzi, C.; Spagnoli, F. Mercury Content in Central and Southern Adriatic Sea Sediments in Relation to Seafloor Geochemistry and Sedimentology. Molecules 2019, 24, 4467. [Google Scholar] [CrossRef] [PubMed]
- Cardellicchio, N.; DeCataldo, A.; Di Leo, A.; Misino, A. Accumulation and tissue distribution of mercury and selenium in striped dolphins (Stenella coeruleoalba) from the Mediterranean Sea (southern Italy). Environ. Pollut. 2002, 116, 265–271. [Google Scholar] [CrossRef]
- Storelli, M.M.; Storelli, A.; D’Addabbo, R.; Marano, C.; Bruno, R.; Marcotrigiano, G.O. Trace elements in loggerhead turtles (Caretta caretta) from the eastern Mediterranean Sea: Overview and evaluation. Environ. Pollut. 2005, 135, 163–170. [Google Scholar] [CrossRef] [PubMed]
- DePew, D.; Basu, N.; Burgess, N.; Campbell, L.M.; Devlin, E.W.; Drevnick, P.; Hammerschmidt, C.R.; Murphy, C.A.; Sandheinrich, M.B.; Wiener, J.G. Toxicity of dietary methylmercury to fish: Derivation of ecologically meaningful threshold concentrations. Environ. Toxicol. Chem. 2012, 31, 1536–1547. [Google Scholar] [CrossRef]
- Berthet, B.; Mouneyrac, C.; Perez, T.; Amiard-Triquet, C. Metallothionein concentration in sponges (Spongia officinalis) as a biomarker of metal contamination. Comp. Biochem. Physiol. Part C: Toxicol. Pharmacol. 2005, 141, 306–313. [Google Scholar] [CrossRef]
- Carrasco, L.; Díez, S.; Soto, D.X.; Catalan, J.; Bayona, J. Assessment of mercury and methylmercury pollution with zebra mussel (Dreissena polymorpha) in the Ebro River (NE Spain) impacted by industrial hazardous dumps. Sci. Total. Environ. 2008, 407, 178–184. [Google Scholar] [CrossRef]
- Roveta, C.; Annibaldi, A.; Vagnoni, F.; Mantas, T.P.; Domenichelli, F.; Gridelli, S.; Puce, S. Short-term effects of environmental factors on the asexual reproduction of Aurelia sp. polyps. Chem. Ecol. 2020, 36, 486–492. [Google Scholar] [CrossRef]
- Thibaud, Y. Teneur en mercure dans quelques poissons de consommation courante. Sci. Peche, Bull. Inst. Peches Marit. 1971, 209, 253–3131. [Google Scholar]
- Cumont, G.; Gilles, G.; Bernhard, F.; Briand, M.B.; Stephan, G.; Ramonda, G.; Guillen, G. Bilan de la contamination des poissons de mer par la rnercure al’occasion d’un controle port ant sur 3 annees. Ann. Hyg. Lang. Fr. Med. Nutr. 1975, 11, 17–25. [Google Scholar]
- Cossa, D.; Martin, J.-M. Mercury in the Rhône delta and adjacent marine areas. Mar. Chem. 1991, 36, 291–302. [Google Scholar] [CrossRef]
- Ferrara, R.; Mazzolai, B.; Edner, H.; Svanberg, S.; Wallinder, E. Atmospheric mercury sources in the Mt. Amiata area, Italy. Sci. Total. Environ. 1998, 213, 13–23. [Google Scholar] [CrossRef]
- Ferrara, R.; Maserti, B.; Breder, R. Mercury in abiotic and biotic compartments of an area affected by a geochemical anomaly (Mt. Amiata, Italy). Water, Air, Soil Pollut. 1991, 56, 219–233. [Google Scholar] [CrossRef]
- Cossa, D.; Coquery, M. The Mediterranean Mercury Anomaly, a Geochemical or a Biologocal Issue. In The Mediterranean Sea. Handbook of Environmental Chemistry Vol. 5K; Saliot, A., Ed.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 177–208. [Google Scholar]
- Ferrara, R.; Maserti, B.E.; Morelli, M.; Morelli, L.; Nannicini, L.; Scarano, G.; Seritti, A.; Torti, M. Metalli pesanti nelle acque dell’Arcipelago Toscanoe nella Posidonia oceanica dell’Arcipelago Toscano. In Progetto mare Ricerca sullo Stato Biologico, Chimico e Fisico dell’Alto Tirreno Toscano; University of Florence: Florence, Italy, 1993; p. 18. [Google Scholar]
- Verniani, D. Monitoraggio Acque Marino Costiere Della Toscana Anno 2012. Proposta Di Classificazione Triennio 2010–2012 (D.Lgs. 152/06); Technical report; Agenzia Regionale per la Protezione Ambientale della Toscana (ARPAT): Florence, Italy, 2013; p. 73. [Google Scholar]
- Verniani, D. Monitoraggio Acque Marino Costiere Della Toscana Anno 2012. Proposta Di Classificazione Triennio Anno 2013 (D.Lgs. 152/06); Technical report; Agenzia Regionale per la Protezione Ambientale della Toscana (ARPAT): Florence, Italy, 2014; p. 53. [Google Scholar]
- Ceccanti, M.; Verniani, D. Monitoraggio Acque Marino Costiere Della Toscana. Attività Di Monitoraggio 2014. Classificazione Provvisoria II Anno Del Triennio 2013–2015; Technical report; Agenzia Regionale per la Protezione Ambientale della Toscana (ARPAT): Florence, Italy, 2015; p. 42. [Google Scholar]
- Verniani, D.; Mancusi, C. Monitoraggio Acque Marino Costiere Della Toscana. Attività Di Monitoraggio 2016 e Proposta Di Classificazione; Technical report; Agenzia Regionale per la Protezione Ambientale della Toscana (ARPAT): Florence, Italy, 2017. [Google Scholar]
- Megaletti, E.; Tunesi, L. Il Report MSFD 2018: Aggiornamento Della Valutazione Ambientale (Art. 8 Del D.Lgs. 190/2010); Technical report; Istituto Superiore per laProtezione e la Ricerca Ambientale (ISPRA): Rome, Italy, 2019; p. 44. [Google Scholar]
- Denton, G.R.W.; Breck, W.G. Mercury in tropical marine organisms from north Queensland. Mar. Pollut. Bull. 1981, 12, 116–121. [Google Scholar] [CrossRef]
- Schuhmacher, M.; Batiste, J.; Bosque, M.A.; Domingo, J.L.; Corbella, J. Mercury concentrations in marine species from the coastal area of Tarragona Province, Spain. Dietary intake of mercury through fish and seafood consumption. Sci. Total. Environ. 1994, 156, 269–273. [Google Scholar] [CrossRef]
- Perugini, M.; Visciano, P.; Manera, M.; Zaccaroni, A.; Olivieri, V.; Amorena, M. Levels of Total Mercury in Marine Organisms from Adriatic Sea, Italy. Bull. Environ. Contam. Toxicol. 2009, 83, 244–248. [Google Scholar] [CrossRef]
- Batista, D.; Muricy, G.; Rocha, R.C.; Miekeley, N.F. Marine sponges with contrasting life histories can be complementary biomonitors of heavy metal pollution in coastal ecosystems. Environ. Sci. Pollut. Res. 2014, 21, 5785–5794. [Google Scholar] [CrossRef]
- Butler, P.A.; Andren, L.; Bonde, G.J.; Jernelov, A.; Reisch, D.J. Monitoring organisms. In Food and Agricultural Organization Technical Conference on Marine Pollution and its Effects on Living Resources and Fishing, Rome, 1970. Supplement 1: Methods of Detection, Measurement and Monitoring of Pollutants in the Marine Environment; Ruivo, M., Ed.; Fishing News Ltd.: London, UK, 1971; pp. 101–112. [Google Scholar]
- Haug, A.; Melsom, S.; Omang, S. Estimation of heavy metal pollution in two Norwegian fjord areas by analysis of the brown alga Ascophyllum nodosum. Environ. Pollut. 1974, 7, 179–192. [Google Scholar] [CrossRef]
- Patel, B.; Balani, M.; Patel, S. Sponge ‘sentinel’ of heavy metals. Sci. Total. Environ. 1985, 41, 143–152. [Google Scholar] [CrossRef]
- Hansen, I.V.; Weeks, J.M.; Depledge, M.H. Accumulation of copper, zinc, cadmium and chromium by the marine sponge Halichondria panicea Pallas and the implications for biomonitoring. Mar. Pollut. Bull. 1995, 31, 133–138. [Google Scholar] [CrossRef]
- Capón, R.J.; Elsbury, K.; Butler, M.S.; Lu, C.C.; Hooper, J.N.A.; Rostas, J.A.P.; O’Brien, K.J.; Mudge, L.M.; Sim, A.T.R. Extraordinary levels of cadmium and zinc in a marine sponge, Tedania charcoti Topsent: Inorganic chemical defense agents. Experientia 1993, 49, 263–264. [Google Scholar] [CrossRef]
- Cebrian, E.; Martí, R.; Uriz, J.; Turon, X. Sublethal effects of contamination on the Mediterranean sponge Crambe crambe: Metal accumulation and biological responses. Mar. Pollut. Bull. 2003, 46, 1273–1284. [Google Scholar] [CrossRef]
- Cebrian, E.; Agell, G.; Marti, R.; Uriz, M.J. Response of the Mediterranean sponge Chondrosia reniformis Nardo to copper pollution. Environ. Pollut. 2006, 141, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Verdenal, B.; Diana, C.; Arnoux, A.; Vacelet, J. Pollutant levels in Mediterranean commercial sponges. In New Perspectives in Sponge Biology, Proceedings of the Third International Conference on Biology of Sponges, Woods Hole, Massachusetts, USA, 17–23 November 1985; Rützler, K., Ed.; Smithsonian Inst. Press: Washington, DC, USA, 1990; pp. 516–524. [Google Scholar]
- Perez, T.; Vacelet, J.; Rebouillon, P. In situ comparative study of several Mediterranean sponges as potential biomonitors of heavy metals. In Sponge Science in the New Millennium; Pansini, M., Pronzato, R., Bavestrello, G., Manconi, R., Eds.; Officine Grafiche Canessa Rapallo: Genova, Italy, 2004; pp. 517–525. [Google Scholar]
- Perez, T.; Longet, D.; Schembri, T.; Rebouillon, P.; Vacelet, J. Effects of 12 years’ operation of a sewage treatment plant on trace metal occurrence within a Mediterranean commercial sponge (Spongia officinalis, Demospongiae). Mar. Pollut. Bull. 2005, 50, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Balsamo, M.; Fregni, E.; Tongiorgi, P. Marine and freshwater Gastrotricha from the Island of Montecristo (Tuscan Archipelago, Italy), with the description of new species. Boll. di Zool. 1994, 61, 217–227. [Google Scholar] [CrossRef]
- Innocenti, F.; Westerman, D.S.; Rocchi, S.; Tonarini, S. The Montecristo monzogranite (Northern Tyrrhenian Sea, Italy): A collisional pluton in an extensional setting. Geol. J. 1997, 32, 131–151. [Google Scholar] [CrossRef]
- Angeletti, L.; Ceregato, A.; Ghirelli, M.; Gualandi, B.; Lipparini, E.; Malatesta, D.; Sperotti, A.; Taviani, M. ROV-SCUBA integrated survey of the Montecristo Island Nature Reserve (Tuscan Archipelago National Park, Mediterranean Sea). Underw. Technol. 2010, 29, 151–154. [Google Scholar] [CrossRef]
- IslePark. Available online: http://www.islepark.it (accessed on 14 February 2020).
- Bo, M.; Canese, S.; Bavestrello, G. Discovering Mediterranean black coral forests: Parantipathes larix (Anthozoa: Hexacorallia) in the Tuscan Archipelago, Italy. Ital. J. Zool. 2013, 81, 112–125. [Google Scholar] [CrossRef]
- Cecchi, E.; Gennaro, P.; Piazzi, L.; Ricevuto, E.; Serena, F. Development of a new biotic index for ecological status assessment of Italian coastal waters based on coralligenous macroalgal assemblages. Eur. J. Phycol. 2014, 49, 298–312. [Google Scholar] [CrossRef]
- Turicchia, E.; Abbiati, M.; Sweet, M.; Ponti, M. Mass mortality hits gorgonian forests at Montecristo Island. Dis. Aquat. Org. 2018, 131, 79–85. [Google Scholar] [CrossRef]
- Bavestrello, G.; Bianchi, C.N.; Calcinai, B.; Cattaneo-Vietti, R.; Cerrano, C.; Morri, C.; Puce, S.; Sára, M. Bio-mineralogy as a structuring factor for marine epibenthic communities. Mar. Ecol. Prog. Ser. 2000, 193, 241–249. [Google Scholar] [CrossRef]
- Casoli, E.; Modica, M.V.; Belluscio, A.; Capello, M.; Oliverio, M.; Ardizzone, G.; Ventura, D. A massive ingression of the alien species Mytilus edulis L. (Bivalvia: Mollusca) into the Mediterranean Sea following the Costa Concordia cruise-ship disaster. Mediterr. Mar. Sci. 2016, 17, 404. [Google Scholar] [CrossRef]
- Illuminati, S.; Annibaldi, A.; Truzzi, C.; Scarponi, G. Recent temporal variations of trace metal content in an Italian white wine. Food Chem. 2014, 159, 493–497. [Google Scholar] [CrossRef]
- Illuminati, S.; Annibaldi, A.; Truzzi, C.; Libani, G.; Mantini, C.; Scarponi, G. Determination of water-soluble, acid-extractable and inert fractions of Cd, Pb and Cu in Antarctic aerosol by square wave anodic stripping voltammetry after sequential extraction and microwave digestion. J. Electroanal. Chem. 2015, 755, 182–196. [Google Scholar] [CrossRef]
- Truzzi, C.; Illuminati, S.; Girolametti, F.; Antonucci, M.; Scarponi, G.; Ruschioni, S.; Riolo, P.; Annibaldi, A. Influence of Feeding Substrates on the Presence of Toxic Metals (Cd, Pb, Ni, As, Hg) in Larvae of Tenebrio molitor: Risk Assessment for Human Consumption. Int. J. Environ. Res. Public Health 2019, 16, 4815. [Google Scholar] [CrossRef]
- Wayne, W.D. Analysis of variance. In Biostatistics, 8th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2005; pp. 303–320. [Google Scholar]
- Piazzi, L.; Balata, D.; Cecchi, E.; Cinelli, F.; Sartoni, G. Species composition and patterns of diversity of macroalgal coralligenous assemblages in the north-western Mediterranean Sea. J. Nat. Hist. 2009, 44, 1–22. [Google Scholar] [CrossRef]
- Stasolla, G.; Innocenti, G. New records of the invasive crabs Callinectes sapidus Rathbun, 1896 and Percnon gibbesi (H. Milne Edwards, 1853) along the Italian coasts. BioInvasions Rec. 2014, 3, 39–43. [Google Scholar] [CrossRef]
- Iacono, R.; Napolitano, E.; Marullo, S.; Artale, V.; Vetrano, A. Seasonal Variability of the Tyrrhenian Sea Surface Geostrophic Circulation as Assessed by Altimeter Data. J. Phys. Oceanogr. 2013, 43, 1710–1732. [Google Scholar] [CrossRef]
- Negri, A.P.; Burns, K.; Boyle, S.; Brinkman, D.; Webster, N.S. Contamination in sediments, bivalves and sponges of McMurdo Sound, Antarctica. Environ. Pollut. 2006, 143, 456–467. [Google Scholar] [CrossRef] [PubMed]
- De Mestre, C.; Maher, W.A.; Roberts, D.; Broad, A.; Krikowa, F.; Davis, A.R. Sponges as sentinels: Patterns of spatial and intra-individual variation in trace metal concentration. Mar. Pollut. Bull. 2012, 64, 80–89. [Google Scholar] [CrossRef]
- Pansini, M.; Manconi, R.; Pronzato, R. Porifera I. Calcarea, Demospongiae (partim), Hexactinellida, Homoscleromorpha. Fauna d’Italia, Vol. 46; Calderini-Il Sole 24 Ore: Bologna, Italy, 2011; p. 554. ISBN 978-88-506-5395-9. [Google Scholar]
- Cebrian, E.; Uriz, M.J.; Turon, X. Sponges as biomonitors of heavy metals in spatial and temporal surveys in northwestern Mediterranean: Multispecies comparison. Environ. Toxicol. Chem. 2007, 26, 2430–2439. [Google Scholar] [CrossRef] [PubMed]
- Illuminati, S.; Annibaldi, A.; Truzzi, C.; Scarponi, G. Heavy metal distribution in organic and siliceous marine sponge tissues measured by square wave anodic stripping voltammetry. Mar. Pollut. Bull. 2016, 111, 476–482. [Google Scholar] [CrossRef]
- Mysing-Gubala, M.; Poirrier, M.A. The effects of cadmium and mercury on gemmule formation and gemmosclere morphology in Ephydatia fluviatilis (Porifera: Spongillidae). Hydrobiologia 1981, 76, 145–148. [Google Scholar] [CrossRef]
- Cebrian, E.; Uriz, M.J. Contrasting Effects of Heavy Metals on Sponge Cell Behavior. Arch. Environ. Contam. Toxicol. 2007, 53, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Batel, R.; Bihari, N.; Rinkevich, B.; Dapper, J.; Schäcke, H.; Schröder, H.; Müller, W. Modulation of organotin-induced apoptosis by the water pollutant methyl mercury in a human lymphoblastoid tumor cell line and a marine sponge. Mar. Ecol. Prog. Ser. 1993, 93, 245–251. [Google Scholar] [CrossRef]
- Truzzi, C.; Annibaldi, A.; Illuminati, S.; Bassotti, E.; Scarponi, G. Square-wave anodic-stripping voltammetric determination of Cd, Pb, and Cu in a hydrofluoric acid solution of siliceous spicules of marine sponges (from the Ligurian Sea, Italy, and the Ross Sea, Antarctica). Anal. Bioanal. Chem. 2008, 392, 247–262. [Google Scholar] [CrossRef]
- Annibaldi, A.; Truzzi, C.; Illuminati, S.; Bassotti, E.; Finale, C.; Scarponi, G. First Systematic Voltammetric Measurements of Cd, Pb, and Cu in Hydrofluoric Acid-Dissolved Siliceous Spicules of Marine Sponges: Application to Antarctic Specimens. Anal. Lett. 2011, 44, 2792–2807. [Google Scholar] [CrossRef]
- Vacelet, J.; Verdenal, B.; Perinet, G. The iron mineralization of Spongia officinalis L. (Porifera, Dictyoceratida) and its relationships with the collagen skeleton. Biol. Cell 1988, 62, 189–198. [Google Scholar] [CrossRef]
- Rosell, D.; Uriz, M.J. Do associated zooxanthellae and the nature of the substratum affect survival, attachment and growth of Cliona viridis (Porifera: Hadromerida)? An experimental approach. Mar. Biol. 1992, 114, 503–507. [Google Scholar] [CrossRef]
- Hanna, C.; Schönberg, L.; De Beer, D.; Lawton, A. oxygen microsensor studies on zooxanthellate clionaid sponges from the Costa Brava, Mediterranean Sea 1. J. Phycol. 2005, 41, 774–779. [Google Scholar] [CrossRef]
- Philp, R.B. Cadmium content of the marine sponge Microciona prolifera, other sponges, water and sediment from the eastern Florida panhandle: Possible effects on Microciona cell aggregation and potential roles of low pH and low salinity. Comp. Biochem. Physiol. Part C: Pharmacol. Toxicol. Endocrinol. 1999, 124, 41–49. [Google Scholar] [CrossRef]
- Schröder, H.; Shostak, K.; Gamulin, V.; Lacorn, M.; Skorokhod, A.; Kavsan, V.; Müller, W. Purification, cDNA cloning and expression of a cadmium-inducible cysteine-rich metallothionein-like protein from the marine sponge Suberites domuncula. Mar. Ecol. Prog. Ser. 2000, 200, 149–157. [Google Scholar] [CrossRef]
- Geffard, A.; Amiard-Triquet, C.; Amiard-Triquet, C. Use of metallothionein in gills from oysters (Crassostrea gigas) as a biomarker: Seasonal and intersite fluctuations. Biomarkers 2002, 7, 123–137. [Google Scholar] [CrossRef] [PubMed]
- Wallace, W.G.; Lee, B.; Luoma, S. Subcellular compartmentalization of Cd and Zn in two bivalves. I. Significance of metal-sensitive fractions (MSF) and biologically detoxified metal (BDM). Mar. Ecol. Prog. Ser. 2003, 249, 183–197. [Google Scholar] [CrossRef]
- Selvin, J.; Priya, S.S.; Kiran, G.S.; Thangavelu, T.; Bai, N.S. Sponge-associated marine bacteria as indicators of heavy metal pollution. Microbiol. Res. 2009, 164, 352–363. [Google Scholar] [CrossRef]
- Thomas, T.; Moitinho-Silva, L.; Lurgi, M.; Björk, J.R.; Easson, C.; Astudillo-Garcia, C.; Olson, J.B.; Erwin, P.M.; Lopez-Legentil, S.; Luter, H.; et al. Diversity, structure and convergent evolution of the global sponge microbiome. Nat. Commun. 2016, 7, 11870. [Google Scholar] [CrossRef]
- Santos-Gandelman, J.F.; Cruz, K.; Crane, S.; Muricy, G.; Giambiagi-Demarval, M.; Barkay, T.; Laport, M.S. Potential Application in Mercury Bioremediation of a Marine Sponge-Isolated Bacillus cereus strain Pj1. Curr. Microbiol. 2014, 69, 374–380. [Google Scholar] [CrossRef]
- Santos-Gandelman, J.F.; Giambiagi-Demarval, M.; Muricy, G.; Barkay, T.; Laport, M.S. Mercury and methylmercury detoxification potential by sponge-associated bacteria. Antonie van Leeuwenhoek 2014, 106, 585–590. [Google Scholar] [CrossRef]
- Stabili, L.; Pizzolante, G.; Morgante, A.; Nonnis Marzano, C.; Longo, C.; Aresta, A.M.; Zambonin, C.; Corriero, G.; Alifano, P. Lindane Bioremediation Capability of Bacteria Associated with the Demosponge Hymeniacidon perlevis. Mar. Drugs 2017, 15, 108. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).