Gaussian Mixture Models for Detecting Sleep Apnea Events Using Single Oronasal Airflow Record
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Set
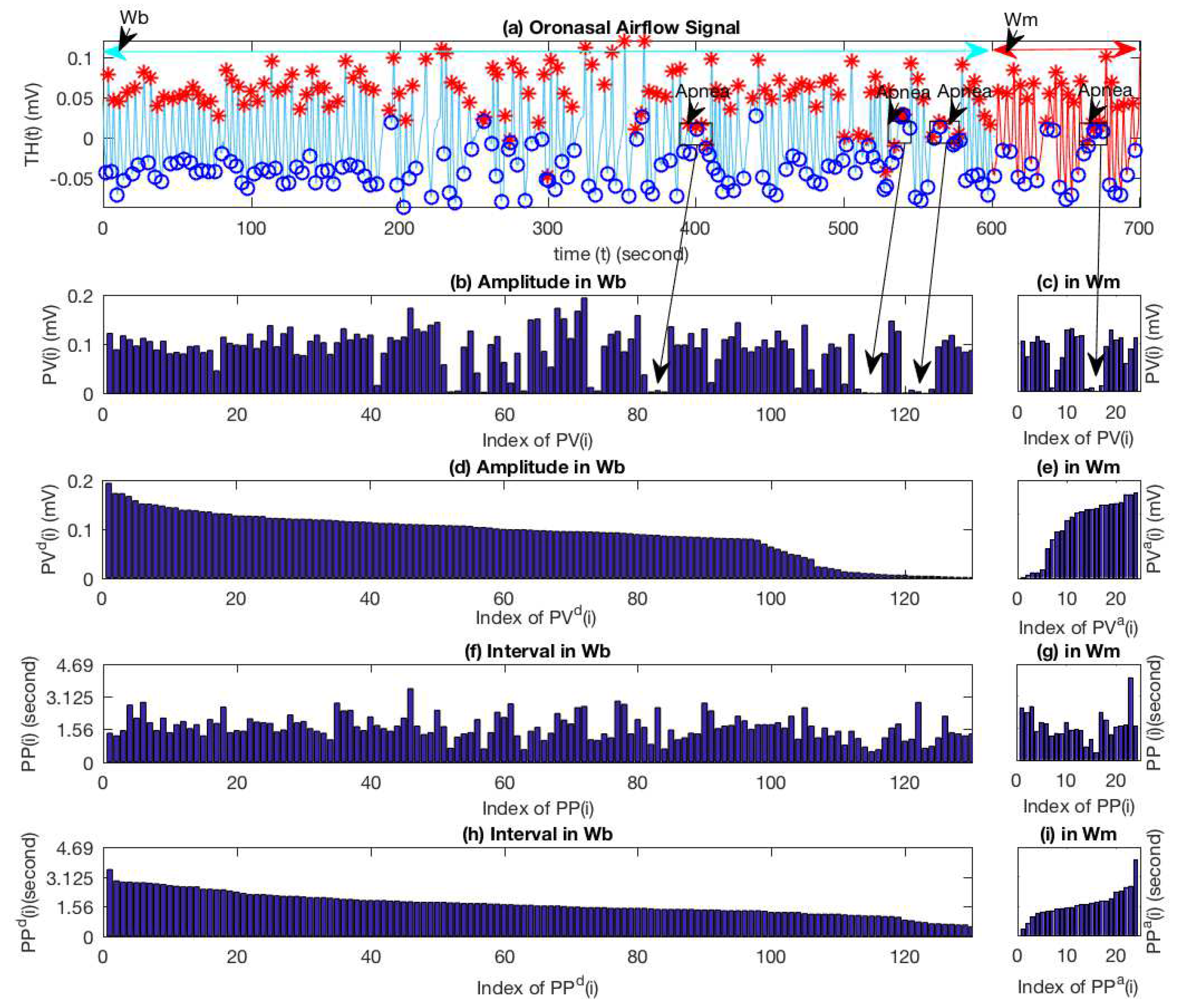
2.2. A Data-Driven Approach for Characterizing Changes in Respiratory Baseline
2.3. Detection of Apnea Events based on Relative Changes in Respiratory Baseline
2.3.1. Rule-Based Threshold Based Classification
2.3.2. Classification with Gaussian Mixture Models (GMM)
2.4. Evaluation of Apnea Detection Results
2.4.1. Classification Performance over Detection Windows
2.4.2. Receiver Operating Characteristics () Curve
3. Results
3.1. Rule-Based Threshold Classifier
3.2. GMM Classifier
3.3. Classification Performance Comparison over the Testing Data Set
4. Discussion of Results
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Somers, V.K.; White, D.P.; Amin, R.; Abraham, W.T.; Costa, F.; Culebras, A.; Daniels, S.; Floras, J.S.; Hunt, C.E.; Olson, L.J.; et al. Sleep apnea and cardiovascular disease: An American heart association/American college of cardiology foundation scientific statement from the American heart association council for high blood pressure research professional education committee, council on clinical cardiology, stroke council, and council on cardiovascular nursing in collaboration with the national heart, lung, and blood institute national center on sleep disorders research (national institutes of health). J. Am. Coll. Cardiol. 2008, 52, 686–717. [Google Scholar] [PubMed]
- Botros, N.; Concato, J.; Mohsenin, V.; Selim, B.; Doctor, K.; Yaggi, H.K. Obstructive sleep apnea as a risk factor for type 2 diabetes. Am. J. Med. 2009, 122, 1122–1127. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Kent, B.D. Obstructive sleep apnoea and coronary artery disease. J. Thorac. Dis. 2018, 10, S4212. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Culebras, A. Sleep apnoea and stroke. Stroke Vasc. Neurol. 2016, 1, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Quan, S.; Gillin, J.C.; Littner, M.; Shepard, J. Sleep-related breathing disorders in adults: Recommendations for syndrome definition and measurement techniques in clinical research. editorials. Sleep 1999, 22, 662–689. [Google Scholar] [CrossRef]
- American Academy of Sleep Medicine. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications; American Academy of Sleep Medicine: Westchester, IL, USA, 2007. [Google Scholar]
- Dempsey, J.A.; Veasey, S.C.; Morgan, B.J.; O’Donnell, C.P. Pathophysiology of sleep apnea. Physiol. Rev. 2010, 90, 47–112. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, Q.; Wang, Y.; Qiu, C. A real-time auto-adjustable smart pillow system for sleep apnea detection and treatment. In Proceedings of the 2013 ACM/IEEE International Conference on Information Processing in Sensor Networks (IPSN), Philadelphia, PA, USA, 9–11 April 2013; pp. 179–190. [Google Scholar]
- De Chazal, P.; Penzel, T.; Heneghan, C. Automated detection of obstructive sleep apnoea at different time scales using the electrocardiogram. Physiol. Meas. 2004, 25, 967. [Google Scholar] [CrossRef]
- Patil, S.P.; Schneider, H.; Schwartz, A.R.; Smith, P.L. Adult obstructive sleep apnea: Pathophysiology and diagnosis. Chest J. 2007, 132, 325–337. [Google Scholar] [CrossRef]
- Berry, R.B.; Brooks, R.; Gamaldo, C.E.; Harding, M.; loy, R.M.; Marcos, C.; Vaughn, B.V. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications; Version 2.6.0; American Academy of Sleep Medicine: Darien, IL, USA, 2020. [Google Scholar]
- Berry, R.B.; Budhiraja, R.; Gottlieb, D.J.; Gozal, D.; Iber, C.; Kapur, V.K.; Marcus, C.L.; Mehra, R.; Parthasarathy, S.; Quan, S.F.; et al. Rules for scoring respiratory events in sleep: Update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. J. Clin. Sleep Med. 2012, 8, 597–619. [Google Scholar] [CrossRef]
- Agarwal, R.; Gotman, J. Computer-assisted sleep staging. IEEE Trans. Biomed. Eng. 2001, 48, 1412–1423. [Google Scholar] [CrossRef]
- Flemons, W.W.; Littner, M.R.; Rowley, J.A.; Gay, P.; Anderson, W.M.; Hudgel, D.W.; McEvoy, R.D.; Loube, D.I. Home diagnosis of sleep apnea: A systematic review of the literature: An evidence review cosponsored by the American Academy of Sleep Medicine, the American College of Chest Physicians, and the American Thoracic Society. CHEST J. 2003, 124, 1543–1579. [Google Scholar] [CrossRef]
- de Almeida, F.R.; Ayas, N.T.; Otsuka, R.; Ueda, H.; Hamilton, P.; Ryan, F.C.; Lowe, A.A. Nasal pressure recordings to detect obstructive sleep apnea. Sleep Breath. 2006, 10, 62–69. [Google Scholar] [CrossRef]
- Khandoker, A.H.; Gubbi, J.; Palaniswami, M. Automated scoring of obstructive sleep apnea and hypopnea events using short-term electrocardiogram recordings. Inf. Technol. Biomed. IEEE Trans. 2009, 13, 1057–1067. [Google Scholar] [CrossRef] [PubMed]
- Whitney, C.W.; Gottlieb, D.J.; Redline, S.; Norman, R.G.; Dodge, R.R.; Shahar, E.; Surovec, S.; Nieto, F.J. Reliability of scoring respiratory disturbance indices and sleep staging. Sleep 1998, 21, 749–757. [Google Scholar] [CrossRef]
- Bennett, J.; Kinnear, W. Sleep on the Cheap: The Role of Overnight Oximetry in the Diagnosis of Sleep Apnoea Hypopnoea Syndrome; BMJ Publishing Group Ltd.: London, UK, 1999. [Google Scholar]
- Koley, B.; Dey, D. Automated detection of apnea and hypopnea events. In Proceedings of the 2012 Third International Conference on Emerging Applications of Information Technology, Kolkata, India, 30 November–1 December 2012; pp. 85–88. [Google Scholar]
- Gutiérrez-Tobal, G.C.; Álvarez, D.; Marcos, J.V.; Del Campo, F.; Hornero, R. Pattern recognition in airflow recordings to assist in the sleep apnoea–hypopnoea syndrome diagnosis. Med Biol. Eng. Comput. 2013, 51, 1367–1380. [Google Scholar] [CrossRef]
- Gutiérrez-Tobal, G.; Hornero, R.; Álvarez, D.; Marcos, J.; Del Campo, F. Linear and nonlinear analysis of airflow recordings to help in sleep apnoea–hypopnoea syndrome diagnosis. Physiol. Meas. 2012, 33, 1261. [Google Scholar] [CrossRef]
- Gutiérrez-Tobal, G.C.; Álvarez, D.; del Campo, F.; Hornero, R. Utility of adaboost to detect sleep apnea-hypopnea syndrome from single-channel airflow. IEEE Trans. Biomed. Eng. 2016, 63, 636–646. [Google Scholar] [CrossRef] [PubMed]
- Nigro, C.A.; Dibur, E.; Aimaretti, S.; González, S.; Rhodius, E. Comparison of the automatic analysis versus the manual scoring from ApneaLink™ device for the diagnosis of obstructive sleep apnoea syndrome. Sleep Breath. 2011, 15, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Nakano, H.; Tanigawa, T.; Furukawa, T.; Nishima, S. Automatic detection of sleep-disordered breathing from a single-channel airflow record. Eur. Respir. J. 2007, 29, 728–736. [Google Scholar] [CrossRef]
- BaHammam, A.; Sharif, M.; Gacuan, D.E.; George, S. Evaluation of the accuracy of manual and automatic scoring of a single airflow channel in patients with a high probability of obstructive sleep apnea. Med Sci. Monit. Int. Med J. Exp. Clin. Res. 2011, 17, MT13. [Google Scholar] [CrossRef]
- Lin, Y.Y.; Wu, H.T.; Hsu, C.A.; Huang, P.C.; Huang, Y.H.; Lo, Y.L. Sleep apnea detection based on thoracic and abdominal movement signals of wearable piezoelectric bands. IEEE J. Biomed. Health Inf. 2016, 21, 1533–1545. [Google Scholar] [CrossRef]
- Vaughn, C.M.; Clemmons, P. Piezoelectric belts as a method for measuring chest and abdominal movement for obstructive sleep apnea diagnosis. Neurodiagn. J. 2012, 52, 275–280. [Google Scholar]
- Avcı, C.; Akbaş, A. Sleep apnea classification based on respiration signals by using ensemble methods. Bio-Med Mater. Eng. 2015, 26, S1703–S1710. [Google Scholar] [CrossRef]
- Azimi, H.; Gilakjani, S.S.; Bouchard, M.; Goubran, R.A.; Knoefel, F. Automatic apnea-hypopnea events detection using an alternative sensor. In Proceedings of the 2018 IEEE Sensors Applications Symposium (SAS), Seoul, Korea, 12–14 March 2018; pp. 1–5. [Google Scholar]
- Penzel, T.; McNames, J.; De Chazal, P.; Raymond, B.; Murray, A.; Moody, G. Systematic comparison of different algorithms for apnoea detection based on electrocardiogram recordings. Med Biol. Eng. Comput. 2002, 40, 402–407. [Google Scholar] [CrossRef] [PubMed]
- De Chazal, P.; Heneghan, C.; Sheridan, E.; Reilly, R.; Nolan, P.; O’Malley, M. Automated processing of the single-lead electrocardiogram for the detection of obstructive sleep apnoea. IEEE Trans. Biomed. Eng. 2003, 50, 686–696. [Google Scholar] [CrossRef] [PubMed]
- Bsoul, M.; Minn, H.; Tamil, L. Apnea MedAssist: Real-time sleep apnea monitor using single-lead ECG. IEEE Trans. Inf. Technol. Biomed. 2010, 15, 416–427. [Google Scholar] [CrossRef] [PubMed]
- Burgos, A.; Goñi, A.; Illarramendi, A.; Bermúdez, J. Real-time detection of apneas on a PDA. IEEE Trans. Inf. Technol. Biomed. 2009, 14, 995–1002. [Google Scholar] [CrossRef]
- Magalang, U.J.; Dmochowski, J.; Veeramachaneni, S.; Draw, A.; Mador, M.J.; El-Solh, A.; Grant, B.J. Prediction of the apnea-hypopnea index from overnight pulse oximetry. Chest J. 2003, 124, 1694–1701. [Google Scholar] [CrossRef]
- Alvarez, D.; Hornero, R.; Marcos, J.V.; del Campo, F. Multivariate analysis of blood oxygen saturation recordings in obstructive sleep apnea diagnosis. IEEE Trans. Biomed. Eng. 2010, 57, 2816–2824. [Google Scholar] [CrossRef]
- Yadollahi, A.; Giannouli, E.; Moussavi, Z. Sleep apnea monitoring and diagnosis based on pulse oximetery and tracheal sound signals. Med Biol. Eng. Comput. 2010, 48, 1087–1097. [Google Scholar] [CrossRef]
- Xie, B.; Minn, H. Real-time sleep apnea detection by classifier combination. IEEE Trans. Inf. Technol. Biomed. 2012, 16, 469–477. [Google Scholar] [CrossRef]
- El Shayeb, M.; Topfer, L.A.; Stafinski, T.; Pawluk, L.; Menon, D. Diagnostic accuracy of level 3 portable sleep tests versus level 1 polysomnography for sleep-disordered breathing: A systematic review and meta-analysis. Cmaj 2014, 186, E25–E51. [Google Scholar] [CrossRef]
- Fontenla-Romero, O.; Guijarro-Berdiñas, B.; Alonso-Betanzos, A.; Moret-Bonillo, V. A new method for sleep apnea classification using wavelets and feedforward neural networks. Artif. Intell. Med. 2005, 34, 65–76. [Google Scholar] [CrossRef]
- Han, J.; Shin, H.B.; Jeong, D.U.; Park, K.S. Detection of apneic events from single channel nasal airflow using 2nd derivative method. Comput. Methods Programs Biomed. 2008, 91, 199–207. [Google Scholar] [CrossRef]
- Selvaraj, N.; Narasimhan, R. Detection of sleep apnea on a per-second basis using respiratory signals. In Proceedings of the Engineering in Medicine and Biology Society (EMBC), 2013 35th Annual International Conference of the IEEE, Osaka, Japan, 3–7 July 2013; pp. 2124–2127. [Google Scholar]
- Ciołek, M.; Niedźwiecki, M.; Sieklicki, S.; Drozdowski, J.; Siebert, J. Automated detection of sleep apnea and hypopnea events based on robust airflow envelope tracking in the presence of breathing artifacts. IEEE J. Biomed. Health Inf. 2015, 19, 418–429. [Google Scholar] [CrossRef]
- Koley, B.L.; Dey, D. Automatic detection of sleep apnea and hypopnea events from single channel measurement of respiration signal employing ensemble binary SVM classifiers. Measurement 2013, 46, 2082–2092. [Google Scholar] [CrossRef]
- Koley, B.L.; Dey, D. Real-time adaptive apnea and hypopnea event detection methodology for portable sleep apnea monitoring devices. IEEE Trans. Biomed. Eng. 2013, 60, 3354–3363. [Google Scholar] [CrossRef] [PubMed]
- Várady, P.; Micsik, T.; Benedek, S.; Benyó, Z. A novel method for the detection of apnea and hypopnea events in respiration signals. Biomed. Eng. IEEE Trans. 2002, 49, 936–942. [Google Scholar] [CrossRef]
- Tian, J.; Liu, J. Apnea detection based on time delay neural network. In Proceedings of the 27th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Shanghai, China, 17–18 Januray 2005; pp. 2571–2574. [Google Scholar]
- Norman, R.G.; Rapoport, D.M.; Ayappa, I. Detection of flow limitation in obstructive sleep apnea with an artificial neural network. Physiol. Meas. 2007, 28, 1089. [Google Scholar] [CrossRef]
- Ozerov, A.; Lagrange, M.; Vincent, E. GMM-based classification from noisy features. In Proceedings of the International Workshop on Machine Listening in Multisource Environments (CHiME), Florence, Italy, 1 September 2011; pp. 30–35. [Google Scholar]
- Kim, J.; ElMoaqet, H.; Tilbury, D.M.; Ramachandran, S.K.; Penzel, T. Time domain characterization for sleep apnea in oronasal airflow signal: A dynamic threshold classification approach. Physiol. Meas. 2019, 40, 054007. [Google Scholar] [CrossRef]
- Bishop, C.M. Pattern Recognition and Machine Learning; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- Thomas, E.; Temko, A.; Lightbody, G.; Marnane, W.; Boylan, G. Gaussian mixture models for classification of neonatal seizures using EEG. Physiol. Meas. 2010, 31, 1047. [Google Scholar] [CrossRef] [PubMed]
- ElMoaqet, H.; Tilbury, D.M.; Ramachandran, S.K. Predicting oxygen saturation levels in blood using autoregressive models: A threshold metric for evaluating predictive models. In Proceedings of the 2013 American Control Conference, Washington, DC, USA, 17–19 June 2013; pp. 734–739. [Google Scholar]
- ElMoaqet, H.; Tilbury, D.M.; Ramachandran, S.K. Evaluating predictions of critical oxygen desaturation events. Physiol. Meas. 2014, 35, 639. [Google Scholar] [CrossRef]
- ElMoaqet, H.; Tilbury, D.M.; Ramachandran, S.K. Multi-step ahead predictions for critical levels in physiological time series. IEEE Trans. Cybern. 2016, 46, 1704–1714. [Google Scholar] [CrossRef]
- Zweig, M.H.; Campbell, G. Receiver-Operating Characteristic (ROC) Plots: A Fundamental Evaluation Tool in Clinical Medicine. Clin. Chem. 1993, 39, 561–577. [Google Scholar] [CrossRef]
- Koley, B.; Dey, D. Adaptive classification system for real-time detection of apnea and hypopnea events. In Proceedings of the 2013 IEEE Point-of-Care Healthcare Technologies (PHT), Bangalore, India, 16–18 January 2013; pp. 42–45. [Google Scholar]
- Canadian Agency for Drugs and Technologies in Health (CADTH). Portable monitoring devices for diagnosis of obstructive sleep apnea at home: Review of Accuracy, cost-effectiveness, guidelines, and coverage in Canada. CADTH Technol. Overviews 2010, 1. [Google Scholar]
- Collop, N.A.; Anderson, W.M.; Boehlecke, B.; Claman, D.; Goldberg, R.; Gottlieb, D.J.; Hudgel, D.; Sateia, M.; Schwab, R. Clinical guidelines for the use of unattended portable monitors in the diagnosis of obstructive sleep apnea in adult patients. J. Clin. Sleep. Med. 2007, 3, 737–747. [Google Scholar]
- Baron, K.G.; Duffecy, J.; Berendsen, M.A.; Mason, I.C.; Lattie, E.G.; Manalo, N.C. Feeling validated yet? A scoping review of the use of consumer-targeted wearable and mobile technology to measure and improve sleep. Sleep Med. Rev. 2018, 40, 151–159. [Google Scholar] [CrossRef]
- Ruehland, W.R.; Rochford, P.D.; O’Donoghue, F.J.; Pierce, R.J.; Singh, P.; Thornton, A.T. The new AASM criteria for scoring hypopneas: Impact on the apnea hypopnea index. Sleep 2009, 32, 150. [Google Scholar] [CrossRef]
| Severity Level | OSA | CSA | MSA | Total |
|---|---|---|---|---|
| None/Minimal | 20 | 312 | 0 | 332 |
| Mild | 1302 | 1887 | 78 | 3267 |
| Moderate | 3056 | 4240 | 400 | 7696 |
| Severe | 10,922 | 23,960 | 2143 | 37,025 |
| Total | 15,300 | 30,399 | 2621 | 48,320 |
| Labels/ Segments | Training Set | Testing Set | Total |
|---|---|---|---|
| Normal | 208,456 | 20,789 | 229,245 |
| Apnea | 81,055 | 8048 | 89,103 |
| Total | 289,511 | 28,837 | 318,348 |
| Algorithm | (%) | (%) | (%) | (%) | (%) | (%) |
|---|---|---|---|---|---|---|
| AICPVwTH | 88.3 | 79.4 | 28.2 | 80.2 | 42.7 | 83.9 |
| AICPVwGMM | 88.5 | 82.5 | 46.6 | 83.4 | 61.1 | 86.7 |
| [45] | 62.0 | 76.9 | 19.3 | 77.6 | 29.4 | 75.7 |
| [39] | 60.3 | 79.5 | 9.2 | 78.9 | 16.0 | 69.9 |
| AICPVwGMM | (%) | (%) | (%) | (%) | (%) | (%) | |
|---|---|---|---|---|---|---|---|
| Type of Apnea | MSA | 97.2 | 80.6 | 9.9 | 80.9 | 18.0 | 88.9 |
| CSA | 88.7 | 80.6 | 41.8 | 81.7 | 56.8 | 84.6 | |
| OSA | 92.7 | 80.6 | 25.3 | 81.4 | 39.8 | 86.7 | |
| AHI Severity | Severe | 96.4 | 77.3 | 68.7 | 83.8 | 80.3 | 86.7 |
| Moderate | 98.1 | 77.5 | 32.6 | 79.5 | 48.9 | 87.8 | |
| Mild | 100.0 | 84.8 | 12.2 | 85.2 | 21.7 | 92.4 | |
| None/Minimal | 27.3 | 71.8 | 0.0 | 83.4 | 0.0 | 86.7 | |
| All | 88.5 | 82.5 | 46.6 | 83.4 | 42.7 | 86.7 | |
| AICPVwTH | (%) | (%) | (%) | (%) | (%) | (%) | |
|---|---|---|---|---|---|---|---|
| Type of Apnea | MSA | 97.5 | 81.1 | 7.8 | 81.4 | 14.4 | 89.3 |
| CSA | 86.8 | 81.1 | 17.1 | 81.4 | 28.6 | 83.4 | |
| OSA | 88.4 | 81.1 | 20.6 | 81.6 | 33.4 | 85.6 | |
| AHI Severity | Severe | 92.4 | 90.3 | 82.5 | 91.0 | 87.2 | 91.4 |
| Moderate | 79.5 | 80.7 | 32.8 | 80.6 | 46.4 | 80.1 | |
| Mild | 76.6 | 76.1 | 3.7 | 76.1 | 7.1 | 76.3 | |
| None/Minimal | 6.3 | 97.0 | 2.0 | 96.1 | 3.0 | 51.7 | |
| All | 88.4 | 79.4 | 28.2 | 80.2 | 61.1 | 83.4 | |
| Type | AHI | (%) | (%) | (%) | (%) | (%) | (%) |
|---|---|---|---|---|---|---|---|
| OSA | All | 92.7 | 80.6 | 25.3 | 81.4 | 39.8 | 86.7 |
| Severe | 97.2 | 77.5 | 59.8 | 82.5 | 74.0 | 87.4 | |
| Moderate | 92.8 | 84.8 | 31.3 | 85.4 | 46.8 | 88.8 | |
| Mild | 87.6 | 71.8 | 1.9 | 71.9 | 3.7 | 79.7 | |
| Normal | 0.0 | 93.3 | 0.0 | 93.2 | NA | 46.6 | |
| CSA | All | 88.6 | 80.6 | 41.8 | 81.7 | 56.8 | 84.6 |
| Severe | 95.6 | 77.5 | 52.8 | 81.3 | 68.0 | 86.6 | |
| Moderate | 85.9 | 84.8 | 23.9 | 84.9 | 37.4 | 85.4 | |
| Mild | 90.1 | 71.8 | 1.7 | 71.9 | 3.3 | 80.9 | |
| Normal | 7.6 | 93.3 | 1.1 | 92.4 | 1.9 | 50.4 | |
| MSA | All | 97.2 | 80.6 | 9.9 | 80.9 | 18.0 | 88.8 |
| Severe | 98.1 | 77.5 | 32.6 | 79.5 | 48.9 | 87.8 | |
| Moderate | 100 | 84.8 | 12.2 | 85.2 | 21.7 | 92.4 | |
| Mild | 27.3 | 71.8 | 0.1 | 71.8 | 0.0 | 49.5 | |
| Normal | - | - | - | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
ElMoaqet, H.; Kim, J.; Tilbury, D.; Ramachandran, S.K.; Ryalat, M.; Chu, C.-H. Gaussian Mixture Models for Detecting Sleep Apnea Events Using Single Oronasal Airflow Record. Appl. Sci. 2020, 10, 7889. https://doi.org/10.3390/app10217889
ElMoaqet H, Kim J, Tilbury D, Ramachandran SK, Ryalat M, Chu C-H. Gaussian Mixture Models for Detecting Sleep Apnea Events Using Single Oronasal Airflow Record. Applied Sciences. 2020; 10(21):7889. https://doi.org/10.3390/app10217889
Chicago/Turabian StyleElMoaqet, Hisham, Jungyoon Kim, Dawn Tilbury, Satya Krishna Ramachandran, Mutaz Ryalat, and Chao-Hsien Chu. 2020. "Gaussian Mixture Models for Detecting Sleep Apnea Events Using Single Oronasal Airflow Record" Applied Sciences 10, no. 21: 7889. https://doi.org/10.3390/app10217889
APA StyleElMoaqet, H., Kim, J., Tilbury, D., Ramachandran, S. K., Ryalat, M., & Chu, C.-H. (2020). Gaussian Mixture Models for Detecting Sleep Apnea Events Using Single Oronasal Airflow Record. Applied Sciences, 10(21), 7889. https://doi.org/10.3390/app10217889








