Thermodynamic Modelling of Harsh Environments on the Solid Phase Assemblage of Hydrating Cements Using PHREEQC
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Chemical and Minerological Analysis
3. Thermodynamic Modelling of Cement Hydration
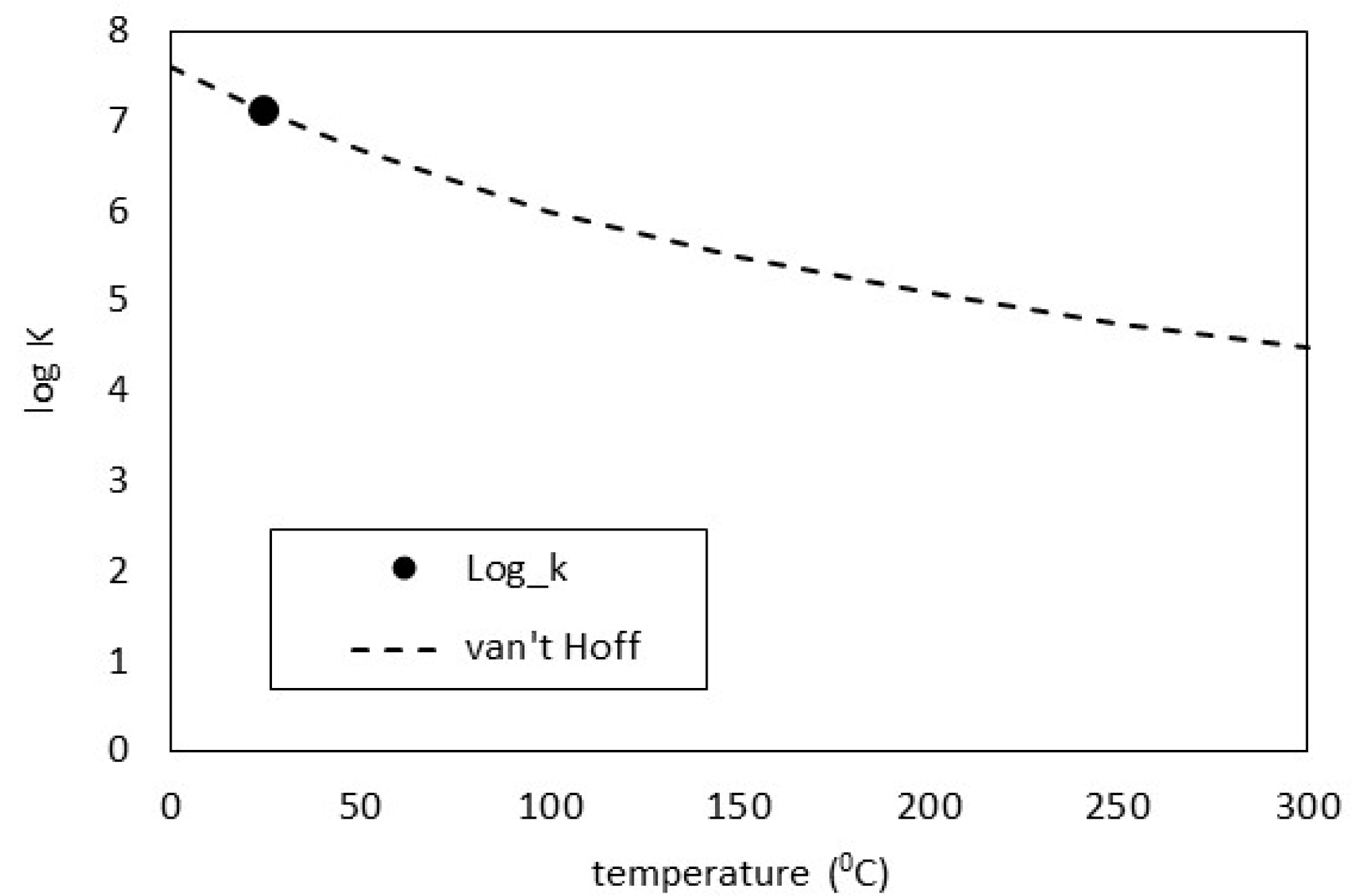
3.1. Thermodynamic Descriptions of Vaterite
3.2. Discrete Solid Phases (DSP) Derivation for M-S-H and Si-Hydrogarnet
3.3. Description of Input for Models
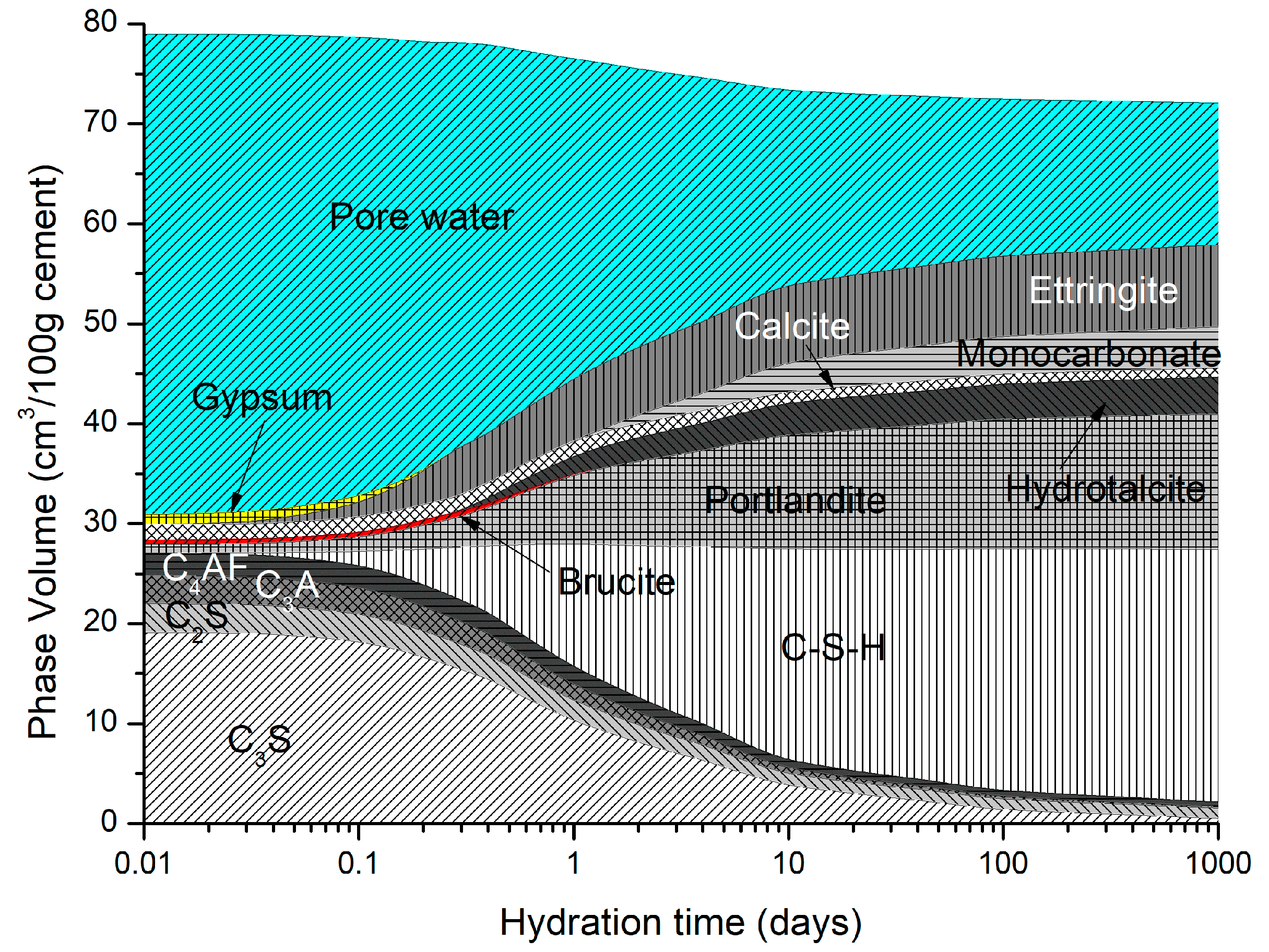
4. Analysis Output
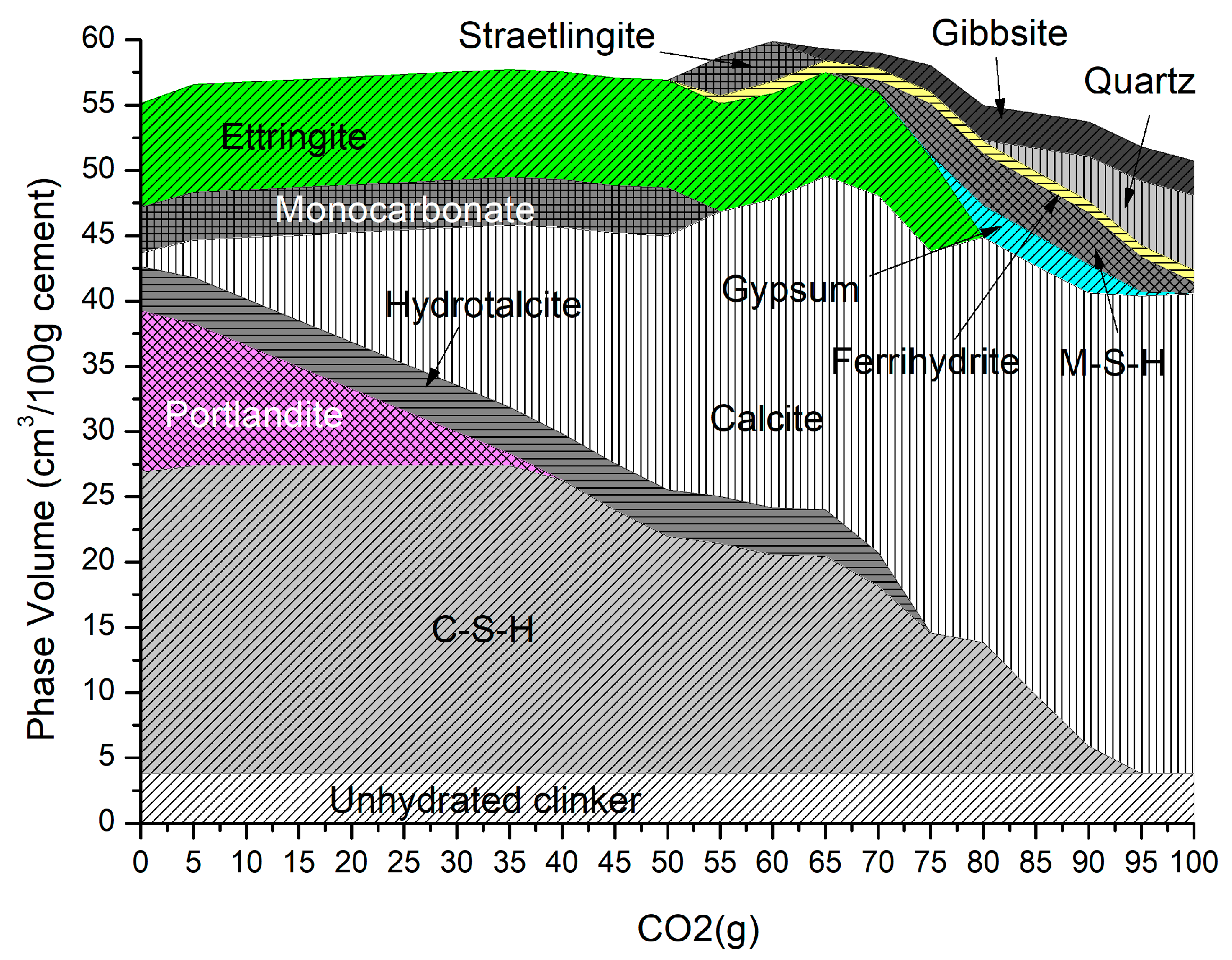
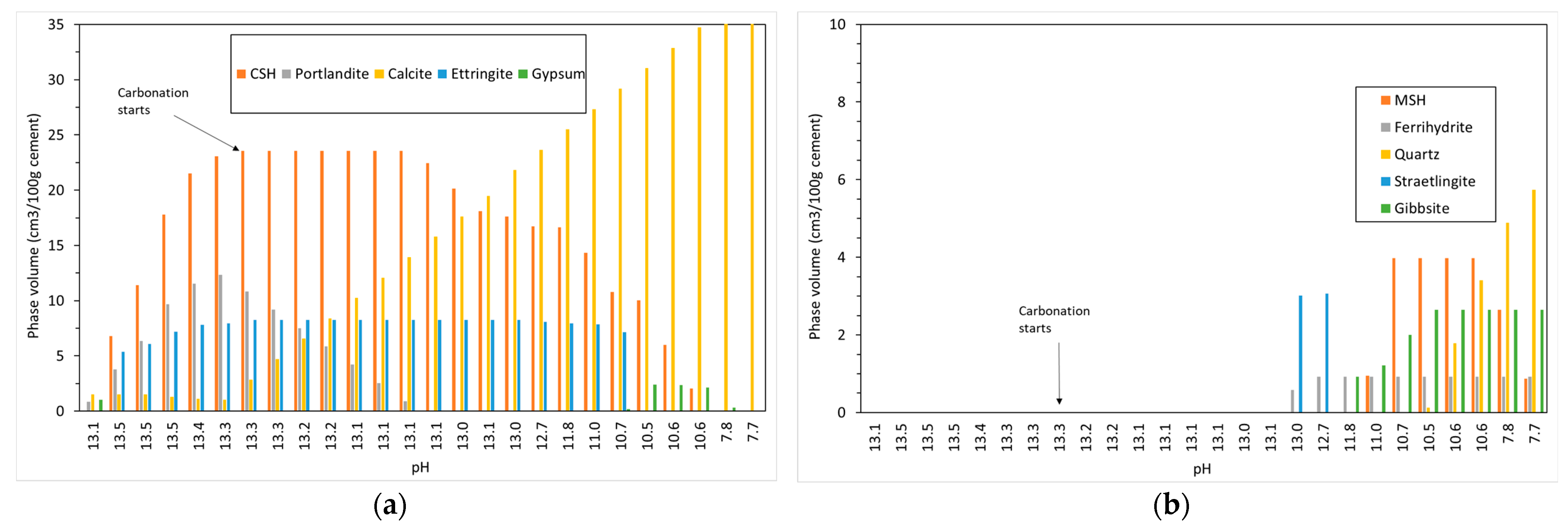
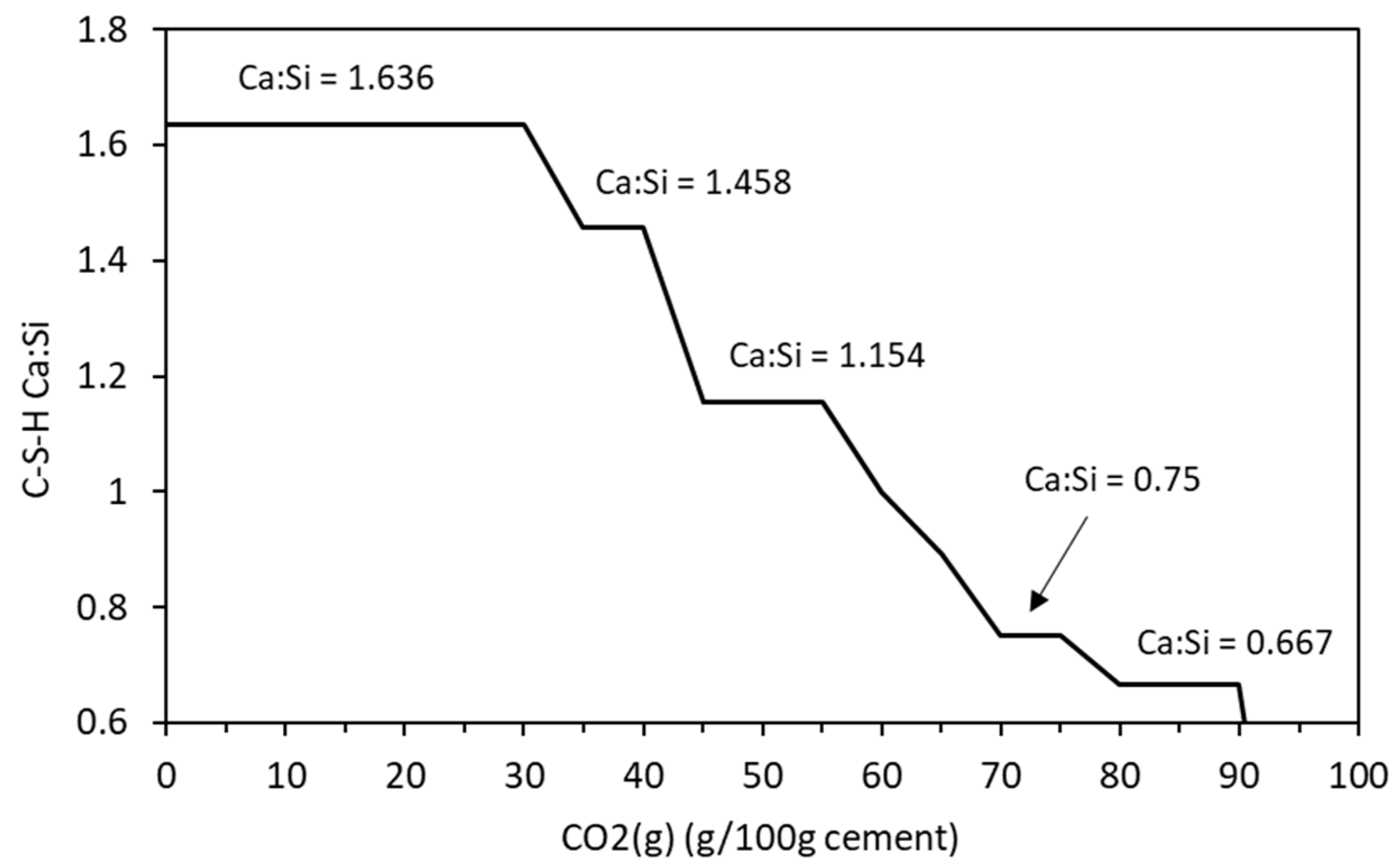
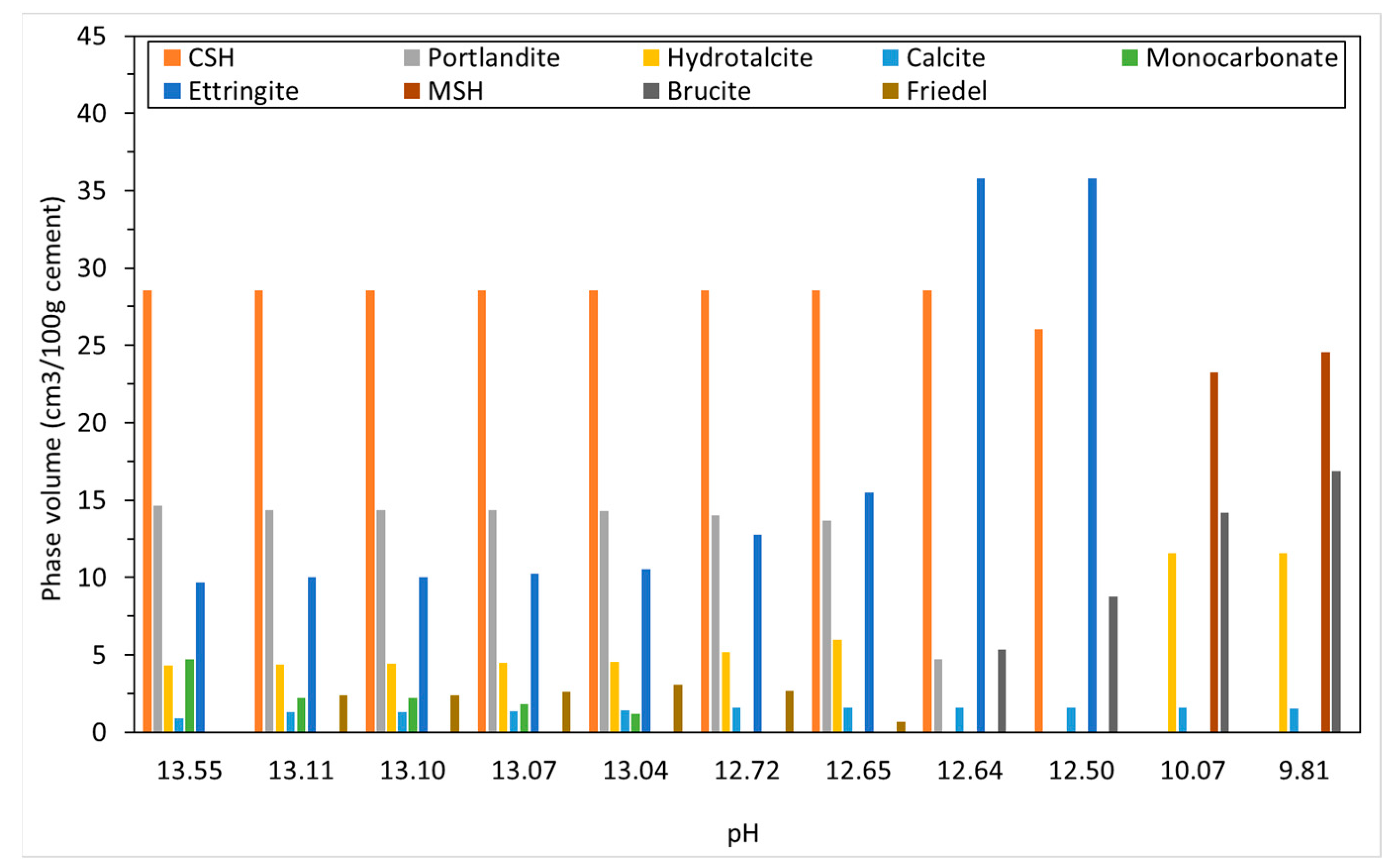
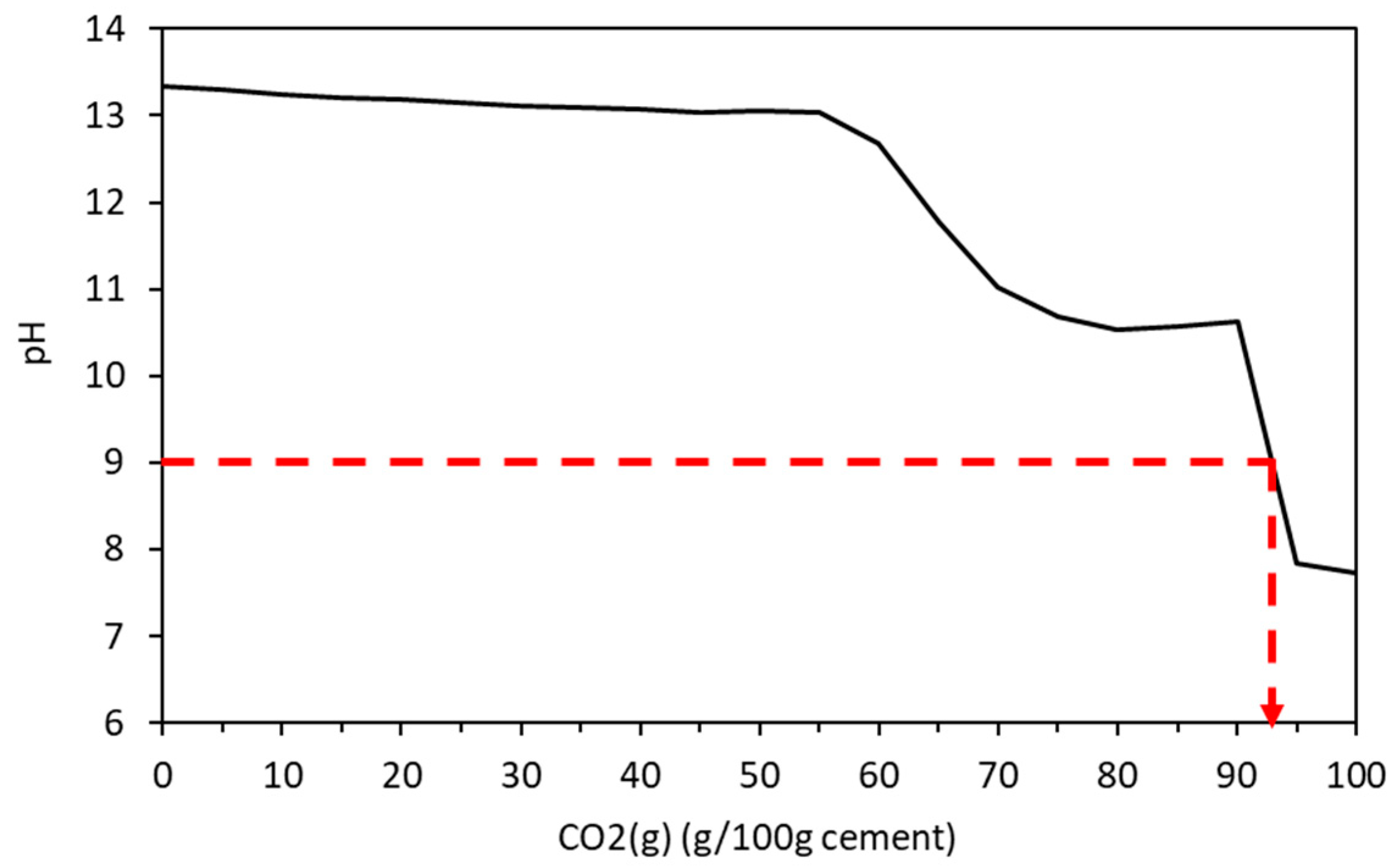
4.1. Carbonation
4.2. Sulfate Ingress
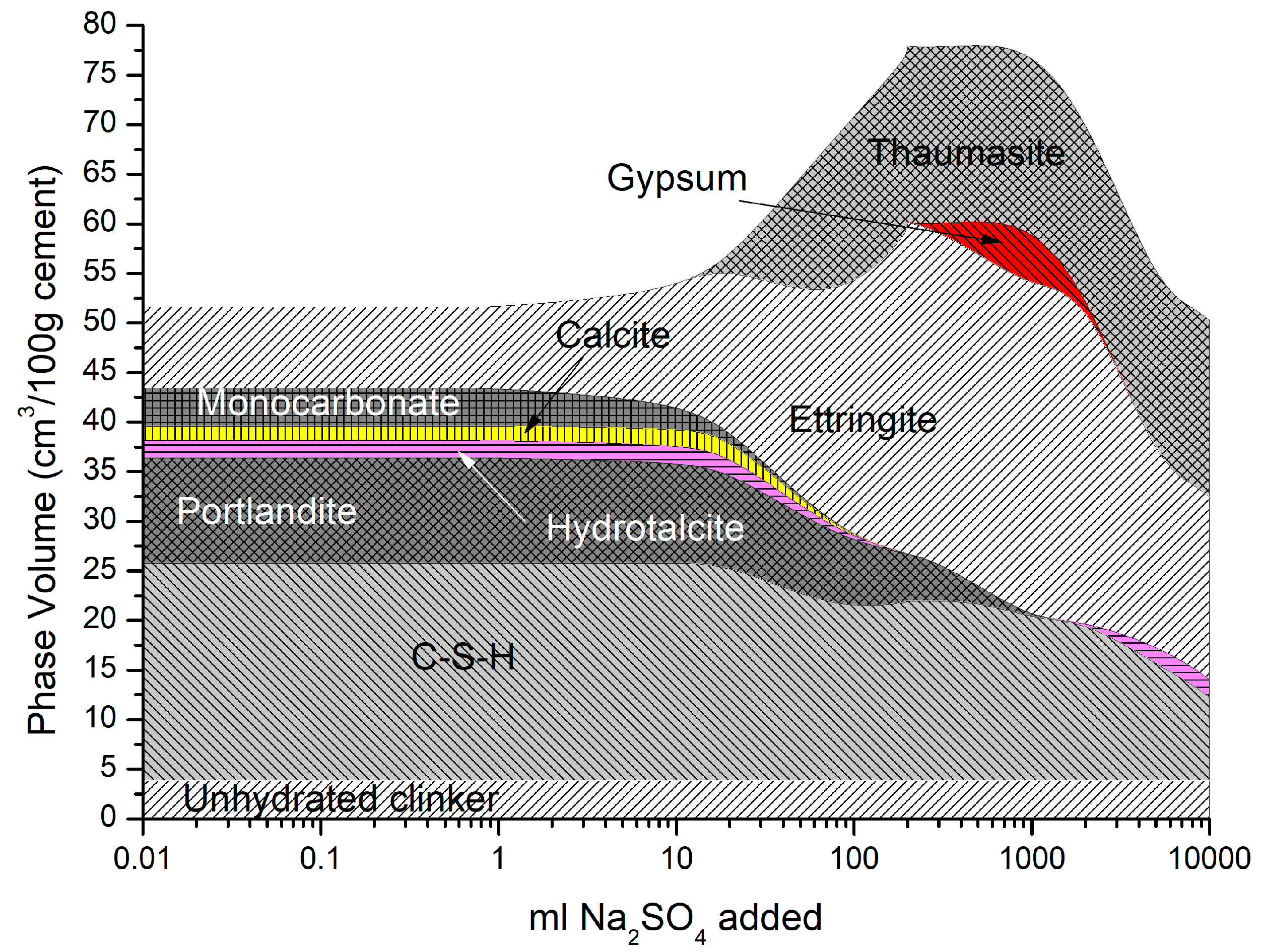
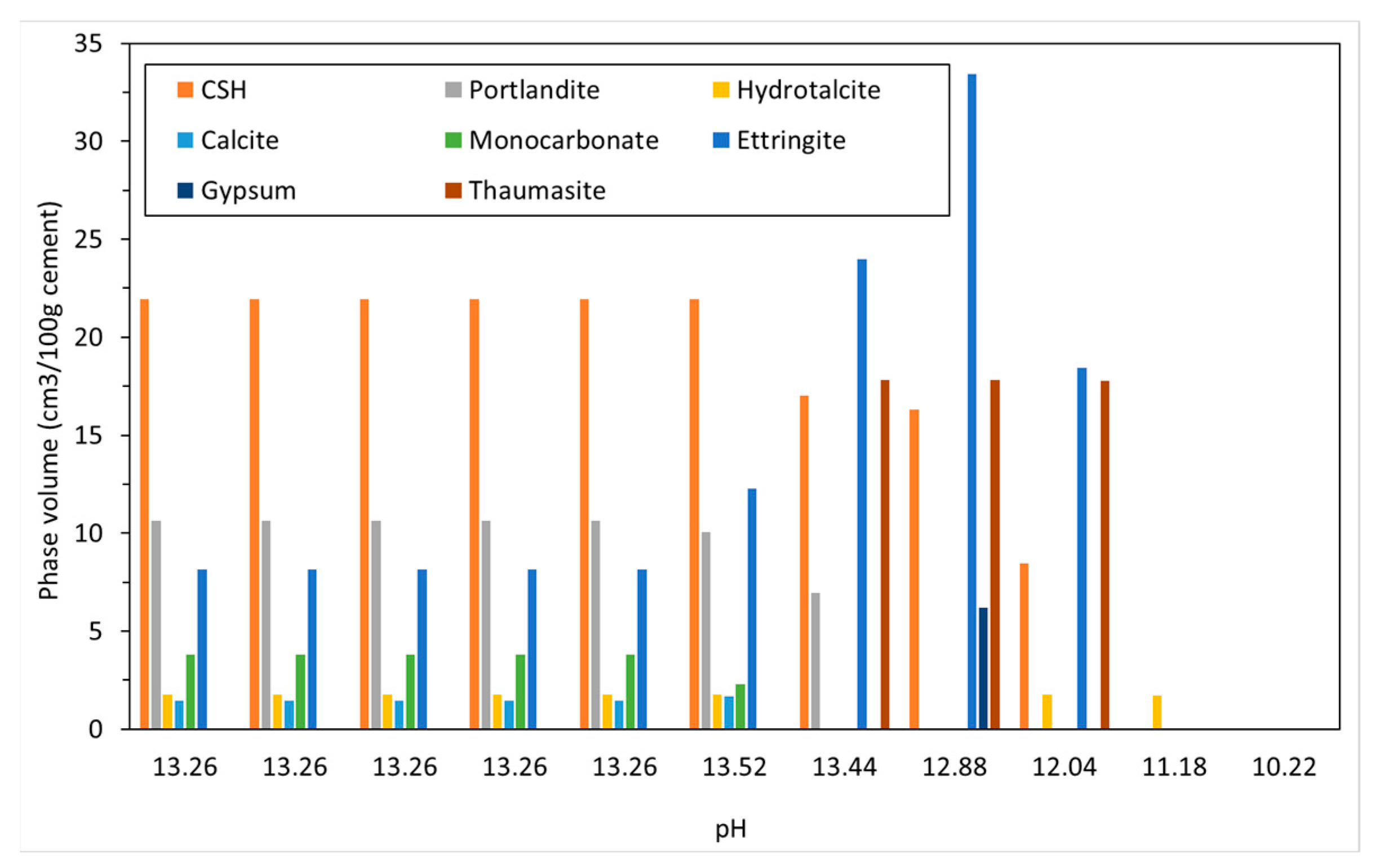
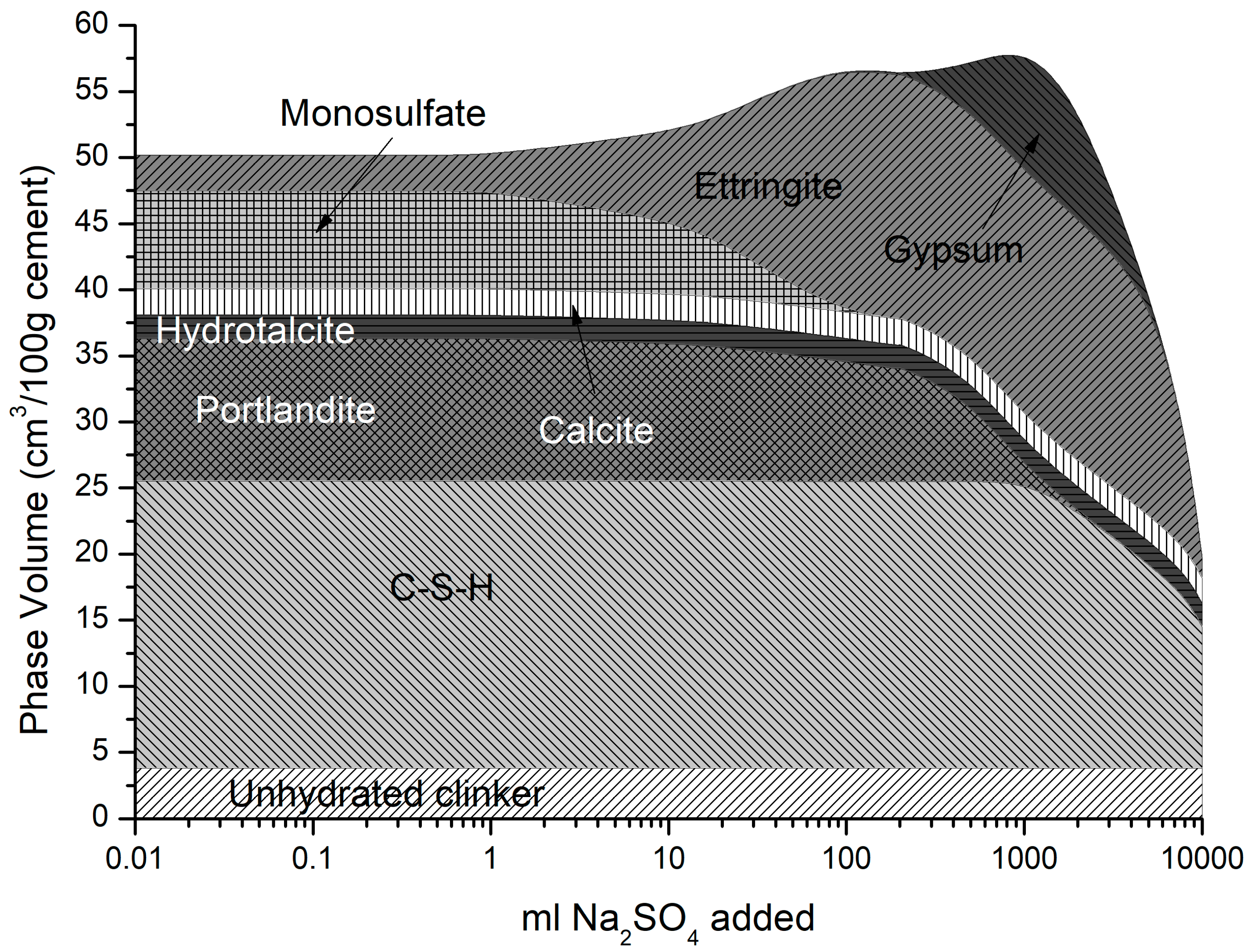
4.2.1. Sodium Sulfate
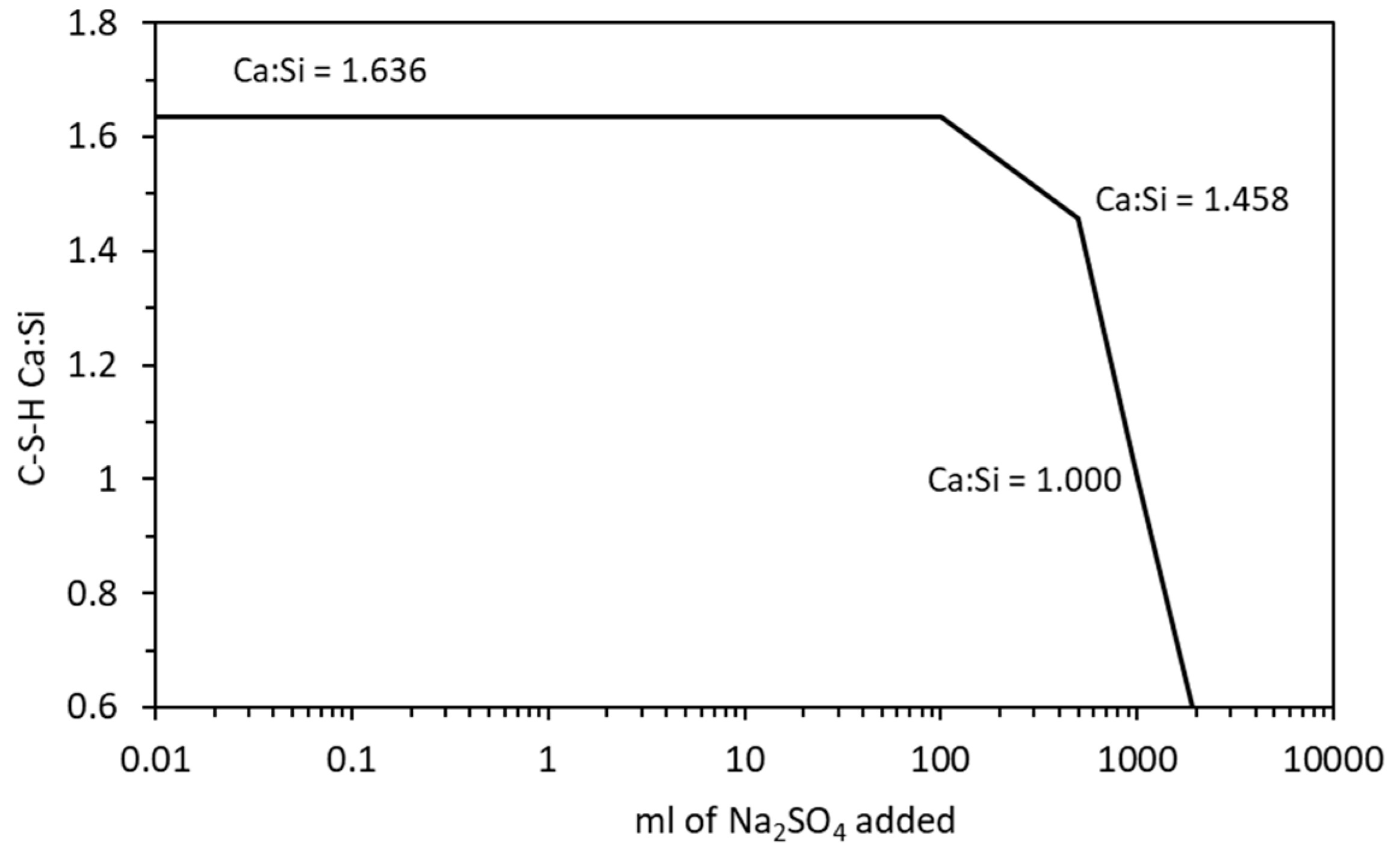
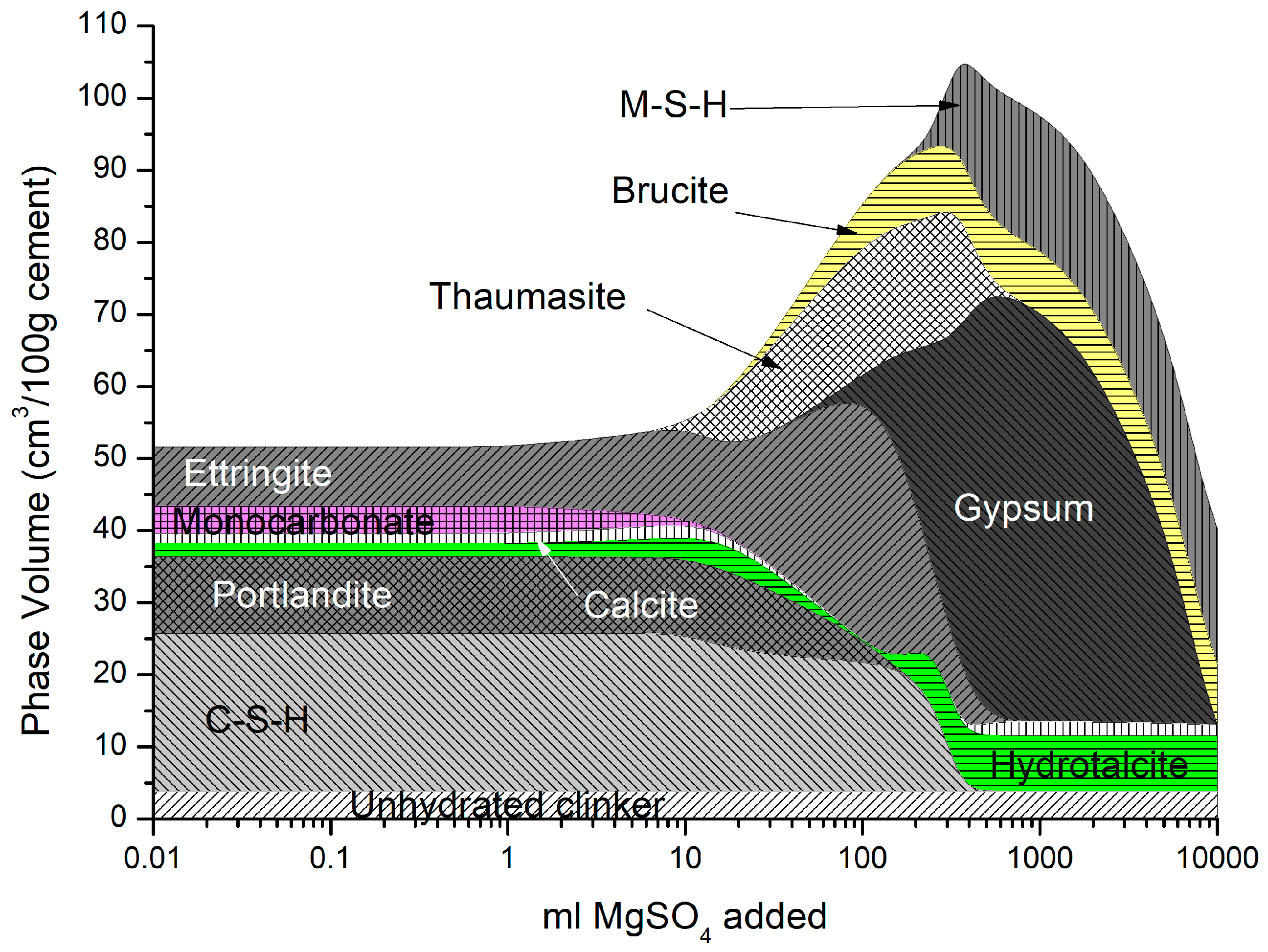
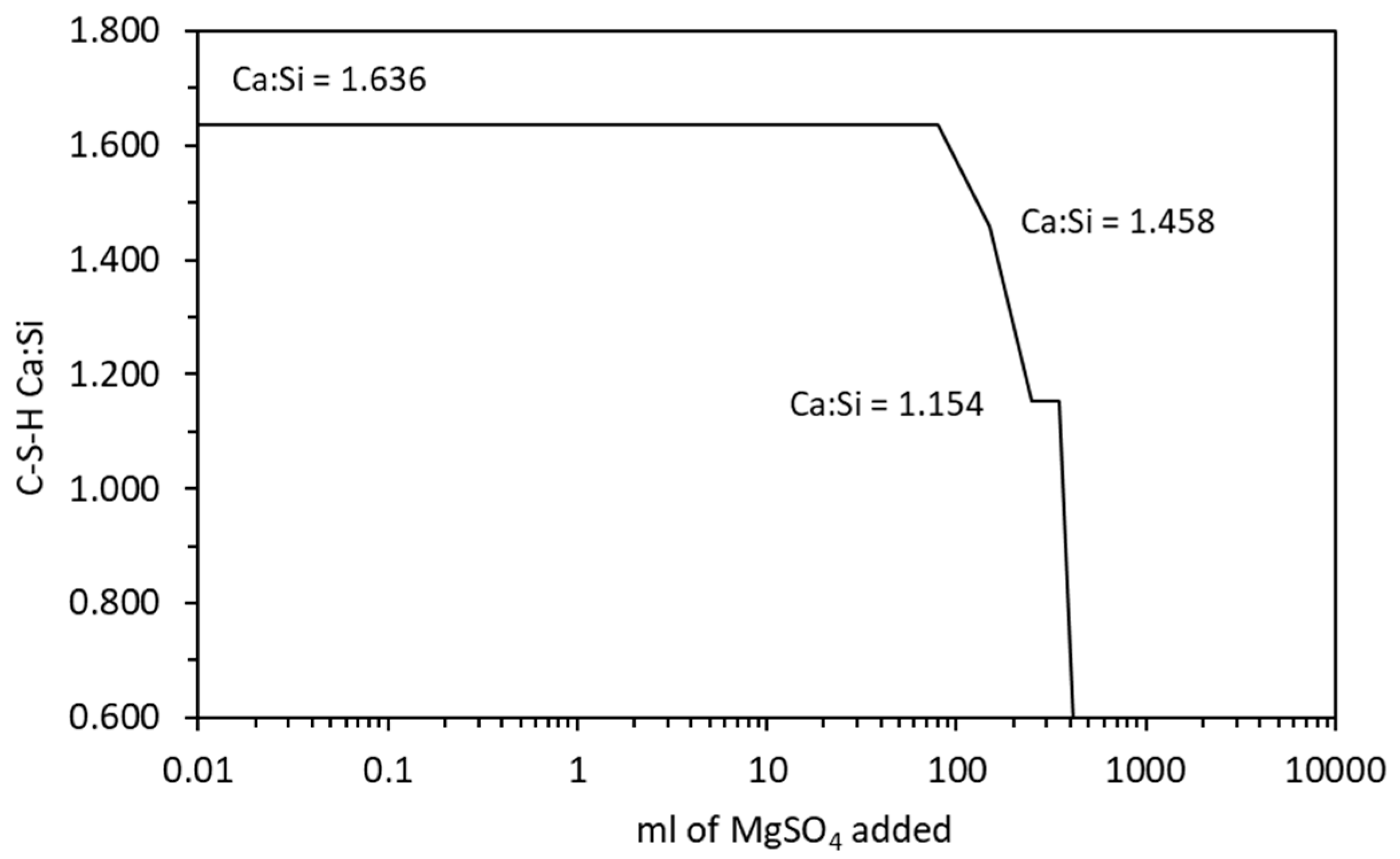
4.2.2. Magnesium Sulfate
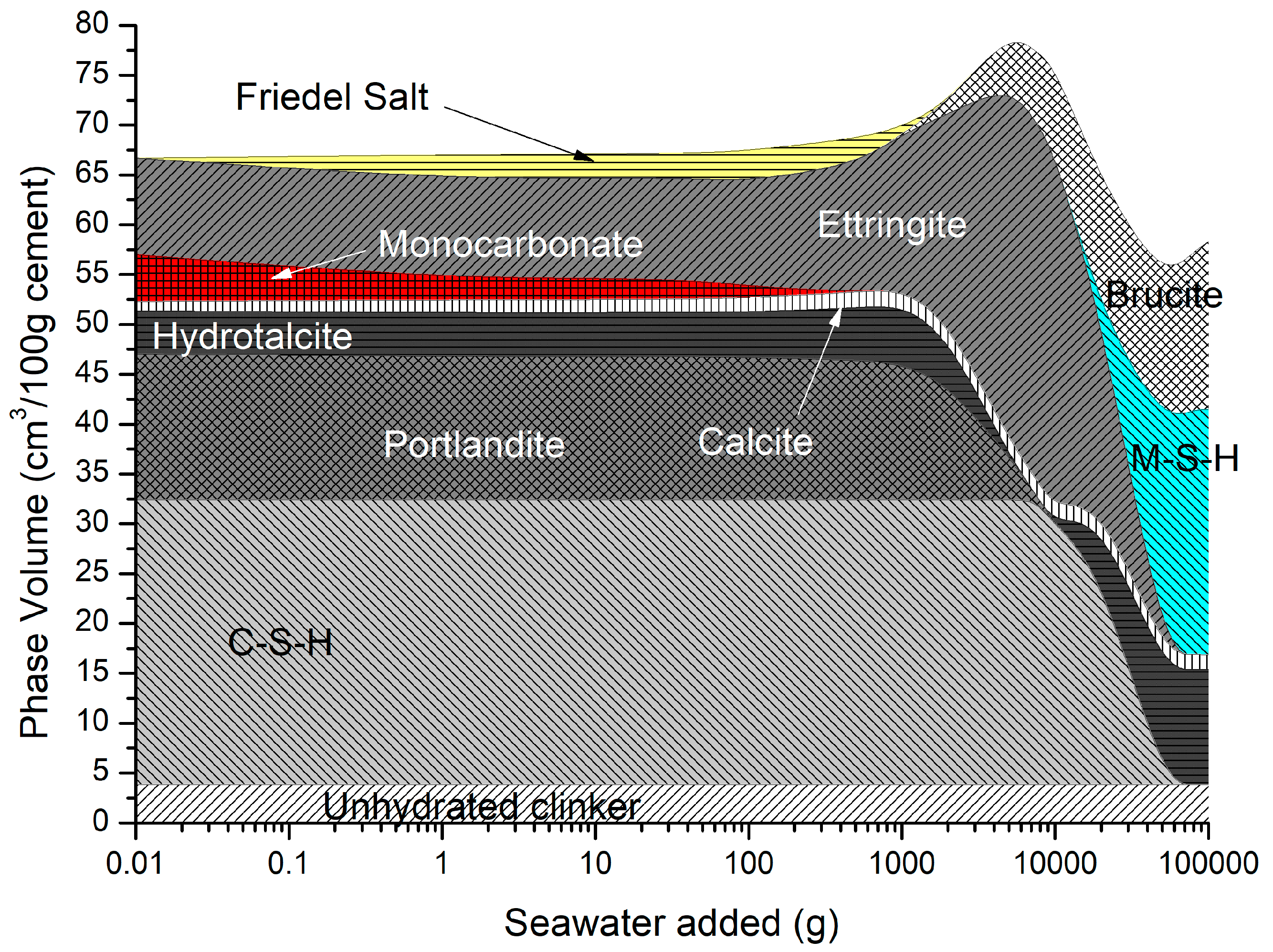
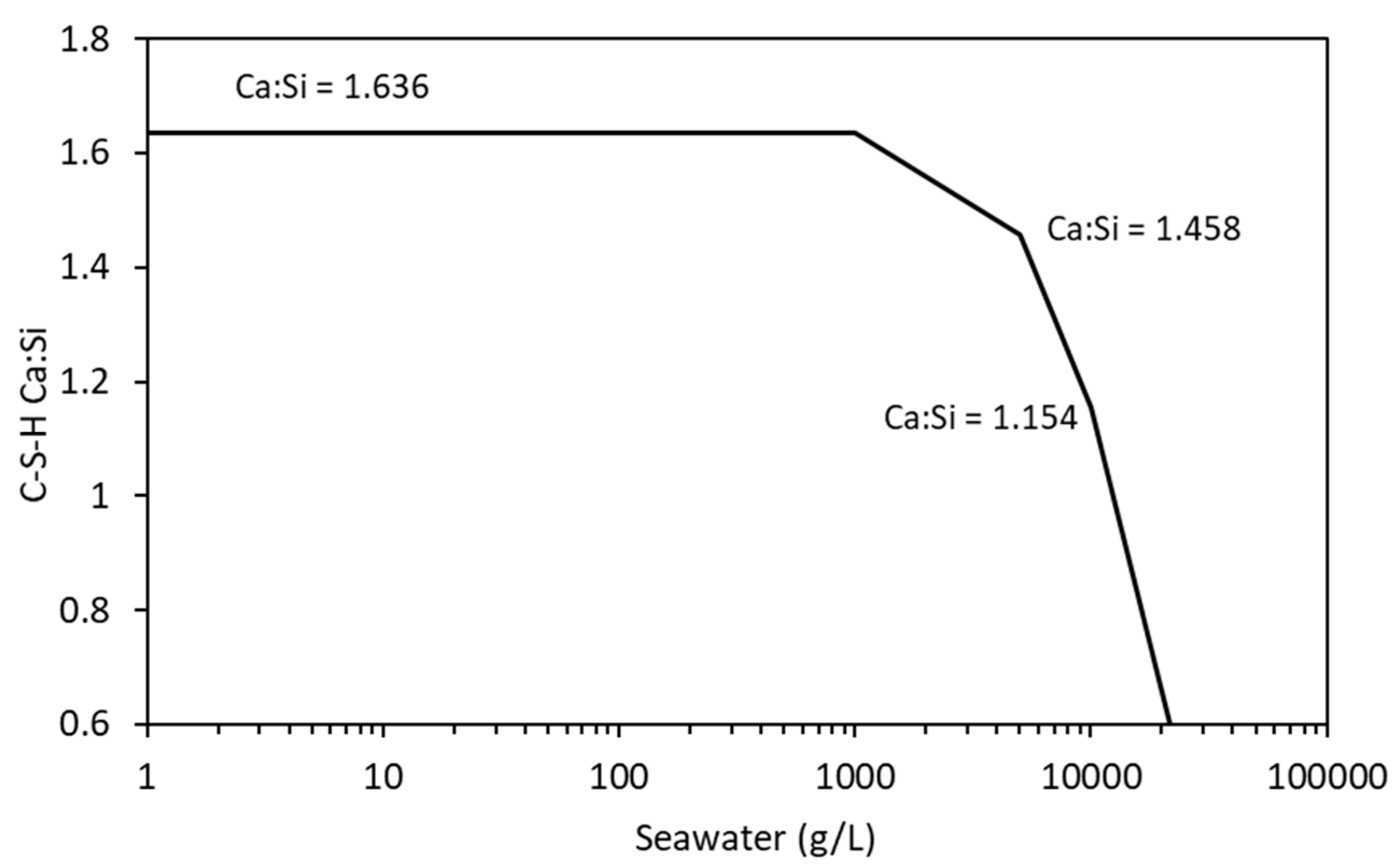
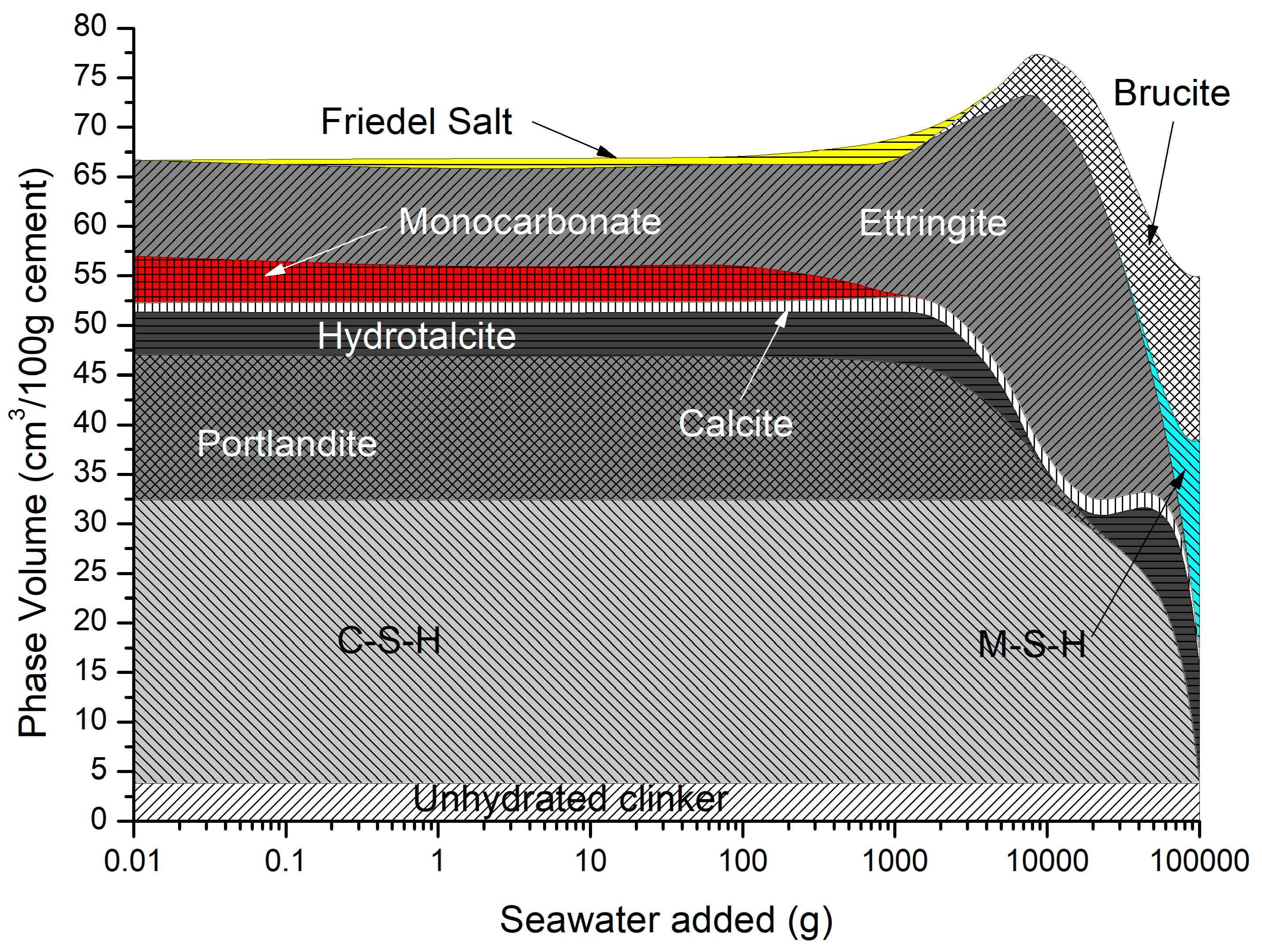
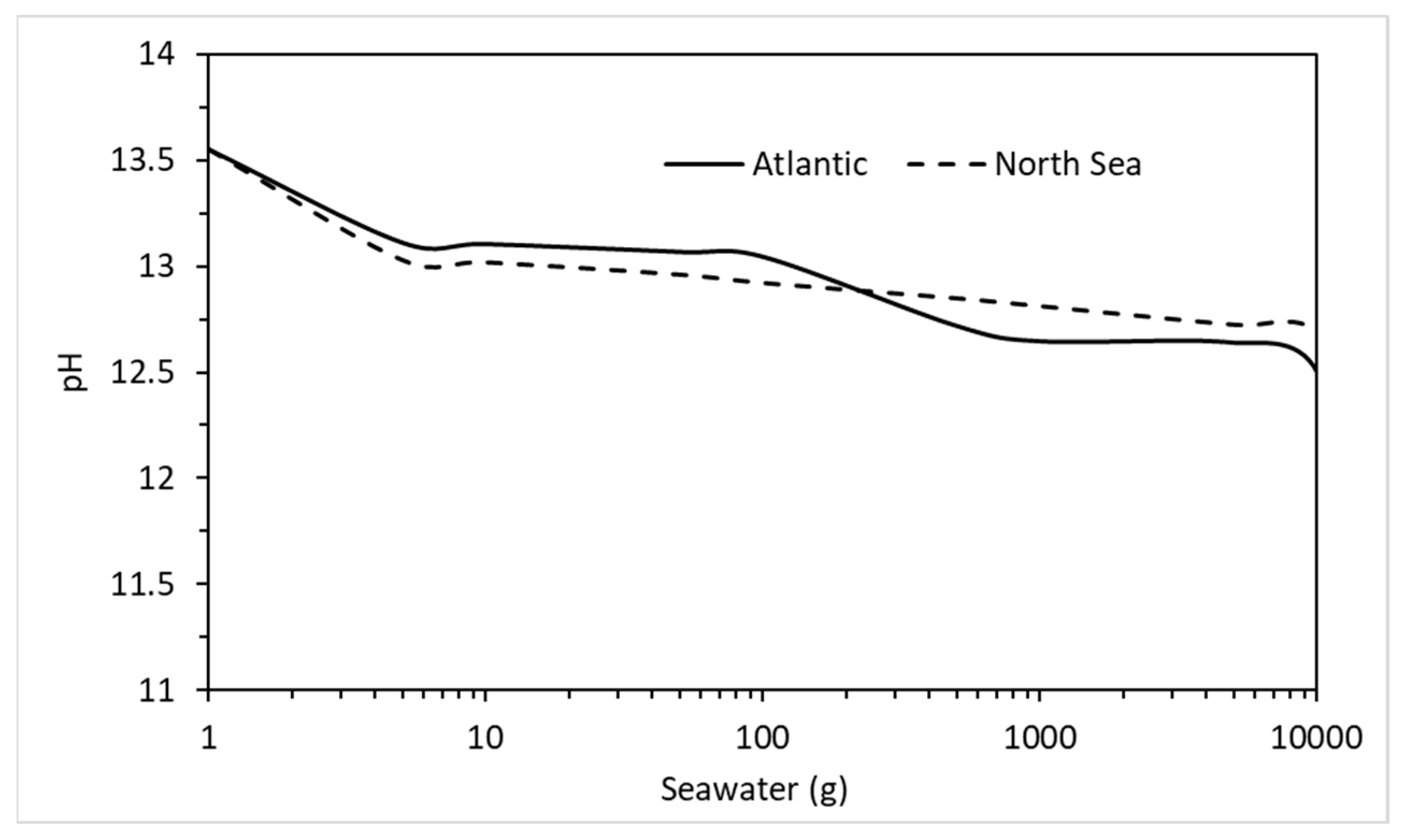
4.3. Sea Water Ingress
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Weerdt, K.; Plusquellec, G.; Revert, A.B.; Geiker, M.R.; Lothenbach, B. Effect of carbonation on the pore solution of mortar. Cem. Concr. Res. 2019, 118, 38–56. [Google Scholar] [CrossRef]
- Jeong, J.; Alemu, A.S.; Park, S.; Lee, H.K.; Liyew, G.; Ramézani, H.; Papadakis, V.G.; Kim, H.-K. Phase profiling of carbonated cement paste: Quantitative X-ray diffraction analysis and numerical modeling. Case Stud. Constr. Mater. 2022, 16, e00890. [Google Scholar] [CrossRef]
- Iwama, K.; Maekawa, K. Modeling of carbonation, de-carbonation and re-carbonation processes of structural concrete subjected to high temperature heating. Cem. Concr. Compos. 2022, 129, 104493. [Google Scholar] [CrossRef]
- Bertola, F.; Gastaldi, D.; Irico, S.; Paul, G.; Canonico, F. Influence of the amount of calcium sulfate on physical/mineralogical properties and carbonation resistance of CSA-based cements. Cem. Concr. Res. 2022, 151, 106634. [Google Scholar] [CrossRef]
- Liu, J.; Jing, Y.; Li, L. Modeling carbonation and chloride ingress in well cements. J. Pet. Sci. Eng. 2022, 212, 110308. [Google Scholar] [CrossRef]
- Hu, L.; Jia, Y.; Chen, Z.; Yao, Y.; Sun, J.; Xie, Q.; Yang, H. An insight of carbonation-hydration kinetics and microstructure characterization of cement paste under accelerated carbonation at early age. Cem. Concr. Compos. 2022, 134, 104763. [Google Scholar] [CrossRef]
- Shi, Z.; Lothenbach, B.; Geiker, M.R.; Kaufmann, J.; Leemann, A.; Ferreiro, S.; Skibsted, J. Experimental studies and thermodynamic modeling of the carbonation of Portland cement, metakaolin and limestone mortars. Cem. Concr. Res. 2016, 88, 60–72. [Google Scholar] [CrossRef]
- Qoku, E.; Bier, T.A.; Schmidt, G.; Skibsted, J. Impact of sulphate source on the hydration of ternary pastes of Portland cement, calcium aluminate cement and calcium sulphate. Cem. Concr. Compos. 2022, 131, 104502. [Google Scholar] [CrossRef]
- Zou, D.; Zhang, M.; Qin, S.; Liu, T.; Tong, W.; Zhou, A.; Jivkov, A. Calcium leaching from cement hydrates exposed to sodium sulfate solutions. Constr. Build. Mater. 2022, 351, 128975. [Google Scholar] [CrossRef]
- Lv, X.; Yang, L.; Wang, F.; Hu, S.; Li, J. Hydration, microstructure characteristics, and mechanical properties of high-ferrite Portland cement in the presence of fly ash and phosphorus slag. Cem. Concr. Compos. 2023, 136, 104862. [Google Scholar] [CrossRef]
- Xu, B.; Winnefeld, F.; Ma, B.; Rentsch, D.; Lothenbach, B. Influence of aluminum sulfate on properties and hydration of magnesium potassium phosphate cements. Cem. Concr. Res. 2022, 156, 106788. [Google Scholar] [CrossRef]
- Georget, F.; Lothenbach, B.; Wilson, W.; Zunino, F.; Scrivener, K.L. Stability of hemicarbonate under cement paste-like conditions. Cem. Concr. Res. 2022, 153, 106692. [Google Scholar] [CrossRef]
- Gollop, R.S.; Taylor, H.F.W. Microstructural and microanalytical studies of sulfate attack. I. Ordinary portland cement paste. Cem. Concr. Res. 1992, 22, 1027–1038. [Google Scholar] [CrossRef]
- Santhanam, M.; Cohen, M.D.; Olek, J. Effects of gypsum formation on the performance of cement mortars during external sulfate attack. Cem. Concr. Res. 2003, 33, 325–332. [Google Scholar] [CrossRef]
- De Weerdt, K.; Lothenbach, B.; Geiker, M.R. Comparing chloride ingress from seawater and NaCl solution in Portland cement mortar. Cem. Concr. Res. 2019, 115, 80–89. [Google Scholar] [CrossRef]
- Qu, F.; Li, W.; Guo, Y.; Zhang, S.; Zhou, J.L.; Wang, K. Chloride-binding capacity of cement-GGBFS-nanosilica composites under seawater chloride-rich environment. Constr. Build. Mater. 2022, 342, 127890. [Google Scholar] [CrossRef]
- Lin, C.; Wang, S.; Ma, W.; Yan, Y.; Lu, Y. Effects of sodium aluminate on the performance of seawater sea-sand engineered cementitious composites. Constr. Build. Mater. 2022, 345, 128422. [Google Scholar] [CrossRef]
- Xu, B.; Lothenbach, B.; Li, Z. Properties and hydrates of seawater-mixed magnesium potassium phosphate cements with high magnesium-to-phosphate ratio. Cem. Concr. Compos. 2022, 134, 104807. [Google Scholar] [CrossRef]
- Yang, Z.; Zhu, H.; Zhang, B.; Dong, Z.; Wu, P. Short-term creep behaviors of seawater sea-sand coral aggregate concrete: An experimental study with Rheological model and neural network. Constr. Build. Mater. 2023, 363, 129786. [Google Scholar] [CrossRef]
- Holmes, N.; Tyrer, M.; West, R.P.; Lowe, A.; Kelliher, D. Using PHREEQC to model cement hydration. Constr. Build. Mater. 2022, 319, 126–129. [Google Scholar] [CrossRef]
- Holmes, N.; Walker, C.; Tyrer, M.; Kelliher, D. Deriving discrete solid phases from CSH-3T and CSHQ end-members to model cement hydration in PHREEQC. In Proceedings of the Civil Engineering Research in Ireland (CERI) Conference, Dublin, Ireland, 25–26 August 2022; pp. 28–33. [Google Scholar]
- Parkhurst, D.J.; Appelo, C.A.J. Description of Input and Examples for PHREEQC Version 3–A Computer Program for Speciation, Batch-reaction, One-Dimensional Transport and Inverse Geochemical Calculations; U.S. Geological Survey: Denver, CO, USA, 2013.
- Kulik, D.A.; Wagner, T.; Dmytrieva, S.V.; Kosakowski, G.; Hingerl, F.F.; Chudnenko, K.V.; Berner, U.R. GEM-Selektor geochemical modeling package: Revised algorithm and GEMS3K numerical kernel for coupled simulation codes. Comput. Geosci. 2013, 17, 24. [Google Scholar] [CrossRef]
- Thoenen, T.; Hummel, W.; Berner, U.; Curti, E. The PSI/Nagra Chemical Thermodynamic Database 12/07; Villigen PSI: Würenlingen, Switzerland, 2014. [Google Scholar]
- Lothenbach, B.; Kulik, D.A.; Matschei, T.; Balonis, M.; Baquerizo, L.; Dilnesa, B.; Miron, G.D.; Myers, R.J. Cemdata18: A chemical thermodynamic database for hydrated Portland cements and alkali-activated materials. Cem. Concr. Res. 2019, 115, 472–506. [Google Scholar] [CrossRef]
- European Committee for Standardization (CEN). BS EN 197-1 Cement: Composition, Specification and Conformity Criteria for Common Cements; European Committee for Standardization (CEN): Brussels, Belgium, 2011; Volume 2000. [Google Scholar]
- Schmidt, T.; Lothenbach, B.; Romer, M.; Neuenschwander, J.; Scrivener, K. Physical and microstructural aspects of sulfate attack on ordinary and limestone blended Portland cements. Cem. Concr. Res. 2009, 39, 1111–1121. [Google Scholar] [CrossRef]
- Walker, C.; Sutou, S.; Oda, C.; Mihara, M.; Honda, A. Calcium silicate hydrate (C-S-H) gel solubility data and a discrete solid phase model at 25 °C based on two binary non-ideal solid solutions. Cem. Concr. Res. 2016, 79, 1–30. [Google Scholar] [CrossRef]
- Holmes, N.; Tyrer, M. Employing Discrete Solid Phases to represent C-S-H solid solutions in the cemdata07 thermodynamic database to model cement hydration using PHREEQC. J. Adv. Concr. Technol. Rev. 2022, 12, 10039. [Google Scholar] [CrossRef]
- CEN. EN 196-6 Methods of Testing Cement—Part 6: Determination of Fineness; European Committee for Standardization (CEN): Brussels, Belgium, 2018. [Google Scholar]
- Borges, P.H.R.; Costa, J.O.; Milestone, N.B.; Lynsdale, C.J.; Streatfield, R.E. Carbonation of CH and C–S–H in composite cement pastes containing high amounts of BFS. Cem. Concr. Res. 2010, 40, 284–292. [Google Scholar] [CrossRef]
- Slegers, P.A.; Rouxhet, P.G. Carbonation of the hydration products of tricalcium silicate. Cem. Concr. Res. 1976, 6, 381–388. [Google Scholar] [CrossRef]
- Šavija, B.; Luković, M. Carbonation of cement paste: Understanding, challenges, and opportunities. Constr. Build. Mater. 2016, 117, 285–301. [Google Scholar] [CrossRef]
- Groves, G.W.; Rodway, D.I.; Richardson, I.G. The carbonation of hardened cement pastes. Adv. Cem. Res. 1990, 3, 117–125. [Google Scholar] [CrossRef]
- Hong, S.Y.; Glasser, F.P. Alkali binding in cement pastes: Part I. The C-S-H phase. Cem. Concr. Res. 1999, 29, 1893–1903. [Google Scholar] [CrossRef]
- Holmes, N.; Basheer, L.; Nanukuttan, S.; Srinivasan, S.; Basheer, P.A.M. Development of a new marine exposure site on the Atlantic North-West coast of Ireland. In Proceedings of the Bridge and Concrete Research in Ireland (BCRI), Cork, Ireland, 2–3 September 2010; pp. 627–636. [Google Scholar]
- Lothenbach, B.; Bary, B.; Le Bescop, P.; Schmidt, T.; Leterrier, N. Sulfate ingress in Portland cement. Cem. Concr. Res. 2010, 40, 1211–1225. [Google Scholar] [CrossRef]
- Thompson, D.; Holmes, N.; Reddy, J. Evaluating a new CEM III/A cement for concretes exposed to harsh acid rich environments. In Proceedings of the Civil Engineering Research in Ireland (CERI) Conference, Galway, Ireland, 29–30 August 2016; pp. 371–375. [Google Scholar]
- Cohen, M.D.; Mather, B. Sulfate attack on concrete: Research needs. Mater. J. 1991, 88, 62–69. [Google Scholar]
- Bonen, D.; Cohen, M.D. Magnesium sulfate attack on portland cement paste-I. Microstructural analysis. Cem. Concr. Res. 1992, 22, 169–180. [Google Scholar] [CrossRef]
- Bonen, D. A microstructural study of the effect produced by magnesium sulfate on plain and silica fume-bearing Portland cement mortars. Cem. Concr. Res. 1993, 23, 541–553. [Google Scholar] [CrossRef]
- Zelić, J.; Rušić, D.; Krstulović, R. A mathematical model for prediction of compressive strength in cement–silica fume blends. Cem. Concr. Res. 2004, 34, 2319–2328. [Google Scholar] [CrossRef]
- Liu, Z.; Deng, D.; De Schutter, G.; Yu, Z. The effect of MgSO4 on thaumasite formation. Cem. Concr. Compos. 2013, 35, 102–108. [Google Scholar] [CrossRef]
- Türker, F.; Aköz, F.; Koral, S.; Yüzer, N. Effects of magnesium sulfate concentration on the sulfate resistance of mortars with and without silica fume. Cem. Concr. Res. 1997, 27, 205–214. [Google Scholar] [CrossRef]
- Yang, Z.; Jiang, J.; Jiang, X.; Mu, S.; Wu, M.; Sui, S.; Wang, L.; Wang, F. The influence of sodium sulfate and magnesium sulfate on the stability of bound chlorides in cement paste. Constr. Build. Mater. 2019, 228, 116775. [Google Scholar] [CrossRef]
- Lee, S.T.; Moon, H.Y.; Hooton, R.D.; Kim, J.P. Effect of solution concentrations and replacement levels of metakaolin on the resistance of mortars exposed to magnesium sulfate solutions. Cem. Concr. Res. 2005, 35, 1314–1323. [Google Scholar] [CrossRef]
- Vuk, T.; Gabrovšek, R.; Kaučič, V. The influence of mineral admixtures on sulfate resistance of limestone cement pastes aged in cold MgSO4 solution. Cem. Concr. Res. 2002, 32, 943–948. [Google Scholar] [CrossRef]
- Li, X.; Qiu, R.; Xue, F.; Fang, L.; Cheng, F. Effects of unreactive MgO and impurities in light burned MgO on the hydration process and performance of base magnesium sulfate cement. Constr. Build. Mater. 2020, 240, 117854. [Google Scholar] [CrossRef]
- Pinto, S.R.; da Luz, C.A.; Munhoz, G.S.; Medeiros-Junior, R.A. Durability of phosphogypsum-based supersulfated cement mortar against external attack by sodium and magnesium sulfate. Cem. Concr. Res. 2020, 136, 106172. [Google Scholar] [CrossRef]
- Torres, S.M.; Sharp, J.H.; Swamy, R.N.; Lynsdale, C.J.; Huntley, S.A. Long term durability of Portland-limestone cement mortars exposed to magnesium sulfate attack. Cem. Concr. Compos. 2003, 25, 947–954. [Google Scholar] [CrossRef]
- Tang, S.; Yuan, J.; Cai, R.; Wei, C.; Chen, J.; Chen, E. In situ monitoring of hydration of magnesium oxysulfate cement paste: Effect of MgO/MgSO4 ratio. Constr. Build. Mater. 2020, 251, 119003. [Google Scholar] [CrossRef]
- Hartshorn, S.A.; Sharp, J.H.; Swamy, R.N. The thaumasite form of sulfate attack in Portland-limestone cement mortars stored in magnesium sulfate solution. Cem. Concr. Compos. 2002, 24, 351–359. [Google Scholar] [CrossRef]
- Tian, B.; Cohen, M.D. Expansion of alite paste caused by gypsum formation during sulfate attack. J. Mater. Civ. Eng. 2000, 12, 24–25. [Google Scholar] [CrossRef]
- Hansen, W.C. Attack on portland cement concrete by alkali soils and waters-A critical review. Highw. Res. Rec. 1966, 113, 1–32. [Google Scholar]
- Mather, B. Discussion of ‘The Process of Sulfate Attack on Cement Mortars’ by Shen Yang, Xu Zhongzi, and Tang Mingshu. Adv. Cem. Based Mater. 1997, 5, 109–110. [Google Scholar] [CrossRef]
- Al-Amoudi, O.S.B.; Maslehuddin, M.; Saadi, M.M. Effect of magnesium sulfate and sodium sulfate on the durability performance of plain and blended cements. ACI Mater. J. 1995, 92, 15–24. [Google Scholar]
- Bonen, D.; Sarkar, S.L. Replacement of portlandite by gypsum in the interfacial zone and cracking related to crystallisation pressure. Ceram. Trans. 1993, 37, 49–59. [Google Scholar]
- Yang, S.; Zhongzi, X.; Mingshu, T. The process of sulfate attack on cement mortars. Adv. Cem. Based Mater. 1996, 4, 1–5. [Google Scholar] [CrossRef]
- Neville, A. Properties of Concrete; Longman London: London, UK, 1995; Volume 4. [Google Scholar]
- Taylor, H.F.W. Cement Chemistry; Thomas Telford Publishing: London, UK, 1997. [Google Scholar]
- McCarter, W.; Chrisp, T.; Starrs, G.; Holmes, N.; Basheer, L.; Basheer, M.; Nanukuttan, S.V. Developments in monitoring techniques for durability assessment of cover-zone concrete. Comput. Aided Des. 2010, 17, 294–303. [Google Scholar]
- EN 206, B.S.; Concrete-Specification, Performance, Production and Conformity. British Standards Institution, Her Majesty Stationery Office: London, UK, 2013.
| Oxide Proportions (g/100 g Cement) | Clinker Compositions (g/100 g Cement) | ||
|---|---|---|---|
| SiO2 | 19.47 | C3S | 59.64 |
| Al2O3 | 5.05 | C2S | 9.67 |
| Fe2O3 | 2.81 | C3A | 8.45 |
| CaO | 63.71 | C4AF | 8.38 |
| MgO | 2.27 | Lime | 1.63 |
| Na2O | 0.29 | calcite | 4.14 |
| K2O | 0.55 | gypsum | 2.17 |
| CaO free | 1.66 | Periclase | 0.71 |
| CO2 | 1.86 | K2SO4 | 0.90 |
| SO3 | 1.62 | Na2SO4 | 0.29 |
| Periclase | 0.72 | ||
| Blaine fineness (m2/kg) | |||
| 400 | |||
| Mineral Name [Composition] | Dissolution Reaction | |||||||
|---|---|---|---|---|---|---|---|---|
| vaterite [CaCO3] | vi | Component | ∆fG°m (J/mol) | ∆fH°m (J/mol) | S°m (J/K/mol) | Cp°m (J/K/mol) | RMM (g/mol) | Z (−) |
| −1 | CaCO3 | −1,099,180 | −1,201,920 | 10.46 | −123.86 | 100.0869 | 0 | |
| −1 | H+ | 0 | 0 | 0 | 0 | 1.00794 | 1 | |
| 1 | Ca+2 | −552,790 | −543,069 | −56.484 | −30.9225 | 40.078 | 2 | |
| 1 | HCO3− | −586,940 | −690,010 | 98.450001 | −34.848837 | 61.01684 | −1 | |
| ∆rΞ°m | −40,550 | −31,159 | 31.506001 | 58.088663 | 0 | |||
| Phase description for PHREEQC PHASE data block | ||||||||
| CaCO3 + 1 H+ = + 1 Ca+2 + 1 HCO3− | ||||||||
| log_k 7.1040 | ||||||||
| delta_h −31.159 kJ/mol | ||||||||
| analytical_expression | −18.67594 | 0 | 2532.174 | 6.98642 | 0 | |||
| Component | G i | H i | S ii | Cp ii | V iii |
|---|---|---|---|---|---|
| M075SH | −3,218,427 | −3,505,729 | 270.26099 | 318 | 95.00 |
| M15SH | −2,355,660 | −2,594,220 | 216 | 250 | 74.00 |
| Mg+2 | −453,990 | −465,930 | −138.07 | −21.66 | |
| SiO2 | −833,411 | −887,856.2 | 41.338 | 44.4654 | |
| H2O | −237,183 | −285,881 | 69.923 | 75.3605 | |
| OH− | −157,270 | −230,010 | −10.71 | −136.34 |
| M075SH |
| (MgO)1.5 (SiO2)2 (H2O)2.5 --> 1.5 Mg+2 + 2 SiO2 + 3 OH− + 1 H2O log K −28.80 |
| analytical_expression parameters i |
| a = 189.588043, c = −12,153.394331, d = −71.58795 |
| M15SH |
| (MgO)1.5 SiO2 (H2O)2.5 --> 1.5 Mg+2 + SiO2 + 3 OH− + 1 H2O log K −23.57 |
| analytical_expression parameters i |
| a = 182.029762, c = −10,551.415516, d = −68.75740 |
| Phase | Mole Fractions | DSP Dissolution Reaction | Log K (25 °C) | Analytical Expression Parameters | Vol. cm3/mol | |||
|---|---|---|---|---|---|---|---|---|
| i | j | a | c | d | ||||
| M075SH | 1 | 0 | (MgO)1.5(SiO2)2(H2O)2.5 = 1.5 Mg+2 + 3 OH− + 1 H2O + 2 SiO2 | −28.31435 | 189.588043 | −12,153.394331 | −71.58795 | 95.000 |
| MSHSS_1 | 0.8 | 0.2 | (MgO)1.5(SiO2)1.8(H2O)2.5 = 1.5 Mg+2 + 3 OH− + 1 H2O + 1.8 SiO2 | −27.56791 | 187.859065 | −11,832.998568 | −71.02184 | 90.800 |
| MSHSS_2 | 0.6 | 0.4 | (MgO)1.5(SiO2)1.6(H2O)2.5 = 1.5 Mg+2 + 3 OH− + 1 H2O + 1.6 SiO2 | −26.67912 | 186.272445 | −11,512.602805 | −70.45573 | 86.600 |
| MSHSS_3 | 0.4 | 0.6 | (MgO)1.5(SiO2)1.4(H2O)2.5 = 1.5 Mg+2 + 3 OH− + 1 H2O + 1.4 SiO2 | −25.71536 | 184.760789 | −11,192.207042 | −69.88962 | 82.400 |
| MSHSS_4 | 0.2 | 0.8 | (MgO)1.5(SiO2)1.2(H2O)2.5 = 1.5 Mg+2 + 3 OH− + 1 H2O + 1.2 SiO2 | −24.67664 | 183.324096 | −10,871.811279 | −69.32351 | 78.200 |
| M15SH | 0 | 0 | (MgO)1.5(SiO2)1(H2O)2.5 = 1.5 Mg+2 + 3 OH− + 1 H2O + 1 SiO2 | −23.49557 | 182.029762 | −10,551.415516 | −68.75740 | 74.000 |
| Component | G i | H i | S ii | Cp ii | V iii |
|---|---|---|---|---|---|
| C3FS1.34H3.32 | −4,681,100 | −4,994,000 | 820 | 395 | 148.523 |
| C3FS0.84H4.32 | −4,479,900 | −4,823,000 | 840 | 371 | 142.492 |
| Ca+2 | −552,790 | −543,070 | −56.48 | −30.92 | |
| H2O | −237,180 | −285,880 | 69.92 | 75.36 | |
| SiO2 | −833,410 | −887,860 | 41.34 | 44.47 | |
| FeO | −368,260 | −443,820 | 44.35 | −234.93 |
| C3FS1.34H3.32 |
| Ca3Fe2O6(SiO2)1.34(H2O)3.32 + 4H+ = 2FeO2− + 3Ca+2 + 5.32H2O + 1.34SiO2 log K −16.185620 |
| analytical_expression parameters i |
| a = 189.588043, c = −12153.394331, d = −71.58795 |
| C3FS0.84H4.32 |
| (FeFeO3)(Ca3O3(SiO2)0.84(H2O)4.32) + 4H+ = 2FeO2− + 3Ca+2 + 6.32H2O + 0.84SiO2 log K −19.980634 |
| analytical_expression parameters i |
| a = 189.588043, c = −12153.394331, d = −71.58795 |
| Phase | Mole Fractions | DSP Dissolution Reaction | Log K (25 °C) | Analytical Expression Parameters | Vol. cm3/mol | |||
|---|---|---|---|---|---|---|---|---|
| i | j | a | c | d | ||||
| C3FS1.34H3.32 | 1.0 | 0 | (CaO)3(Fe2O3)1(SiO2)1.34(H2O)3.32 + 4 H+ = 3 Ca+2 + 5.32 H2O + 1.34 SiO2 + 2 FeO2− | 16.1856 | 149.183895 | 4452.847007 | −59.78885 | 148.523 |
| SiHydroSS_1 | 0.8 | 0.2 | (CaO)3(Fe2O3)1(SiO2)1.24(H2O)3.52 + 4 H+ = 3 Ca+2 + 5.52 H2O + 1.24 SiO2 + 2 FeO2− | 16.7273 | 143.875891 | 4828.350469 | −57.93366 | 147.317 |
| SiHydroSS_2 | 0.6 | 0.4 | (CaO)3(Fe2O3)1(SiO2)1.14(H2O)3.72 + 4 H+ = 3 Ca+2 + 5.72 H2O + 1.14 SiO2 + 2 FeO2 | 17.4113 | 138.710245 | 5203.853931 | −56.07847 | 146.111 |
| SiHydroSS_3 | 0.4 | 0.6 | (CaO)3(Fe2O3)1(SiO2)1.04(H2O)3.92 + 4 H+ = 3 Ca+2 + 5.92 H2O + 1.04 SiO2 + 2 FeO2− | 18.1703 | 133.619563 | 5579.357392 | −54.22328 | 144.904 |
| SiHydroSS_4 | 0.2 | 0.8 | (CaO)3(Fe2O3)1(SiO2)0.94(H2O)4.12 + 4 H+ = 3 Ca+2 + 6.12 H2O + 0.94 SiO2 + 2 FeO2− | 19.0043 | 128.603845 | 5954.860854 | −52.36809 | 143.698 |
| C3FS0.84H4.32 | 0 | 1.0 | (CaO)3(Fe2O3)1(SiO2)0.84(H2O)4.32 + 4 H+ = 3 Ca+2 + 6.32 H2O + 0.84 SiO2 + 2 FeO2− | 19.9806 | 123.730485 | 6330.364316 | −50.51290 | 142.492 |
| Element | Cl | SO4 | Na | K | Mg | Ca |
|---|---|---|---|---|---|---|
| Concentration (ppm) | 15,704 | 2254 | 9300 | 360 | 1800 | 490 |
| Element | Cl | SO4 | Na | K | Mg | Ca |
|---|---|---|---|---|---|---|
| Concentration (ppm) | 8826 | 1384 | 5600 | 290 | 900 | 310 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Holmes, N.; Tyrer, M.; Kelliher, D. Thermodynamic Modelling of Harsh Environments on the Solid Phase Assemblage of Hydrating Cements Using PHREEQC. Appl. Sci. 2023, 13, 135. https://doi.org/10.3390/app13010135
Holmes N, Tyrer M, Kelliher D. Thermodynamic Modelling of Harsh Environments on the Solid Phase Assemblage of Hydrating Cements Using PHREEQC. Applied Sciences. 2023; 13(1):135. https://doi.org/10.3390/app13010135
Chicago/Turabian StyleHolmes, Niall, Mark Tyrer, and Denis Kelliher. 2023. "Thermodynamic Modelling of Harsh Environments on the Solid Phase Assemblage of Hydrating Cements Using PHREEQC" Applied Sciences 13, no. 1: 135. https://doi.org/10.3390/app13010135
APA StyleHolmes, N., Tyrer, M., & Kelliher, D. (2023). Thermodynamic Modelling of Harsh Environments on the Solid Phase Assemblage of Hydrating Cements Using PHREEQC. Applied Sciences, 13(1), 135. https://doi.org/10.3390/app13010135








