Repeated Fecal Microbial Transplantations and Antibiotic Pre-Treatment Are Linked to Improved Clinical Response and Remission in Inflammatory Bowel Disease: A Systematic Review and Pooled Proportion Meta-Analysis
Abstract
:1. Introduction
2. Methods
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Study Selection
2.4. Data Extraction
2.5. Data Synthesis
3. Results
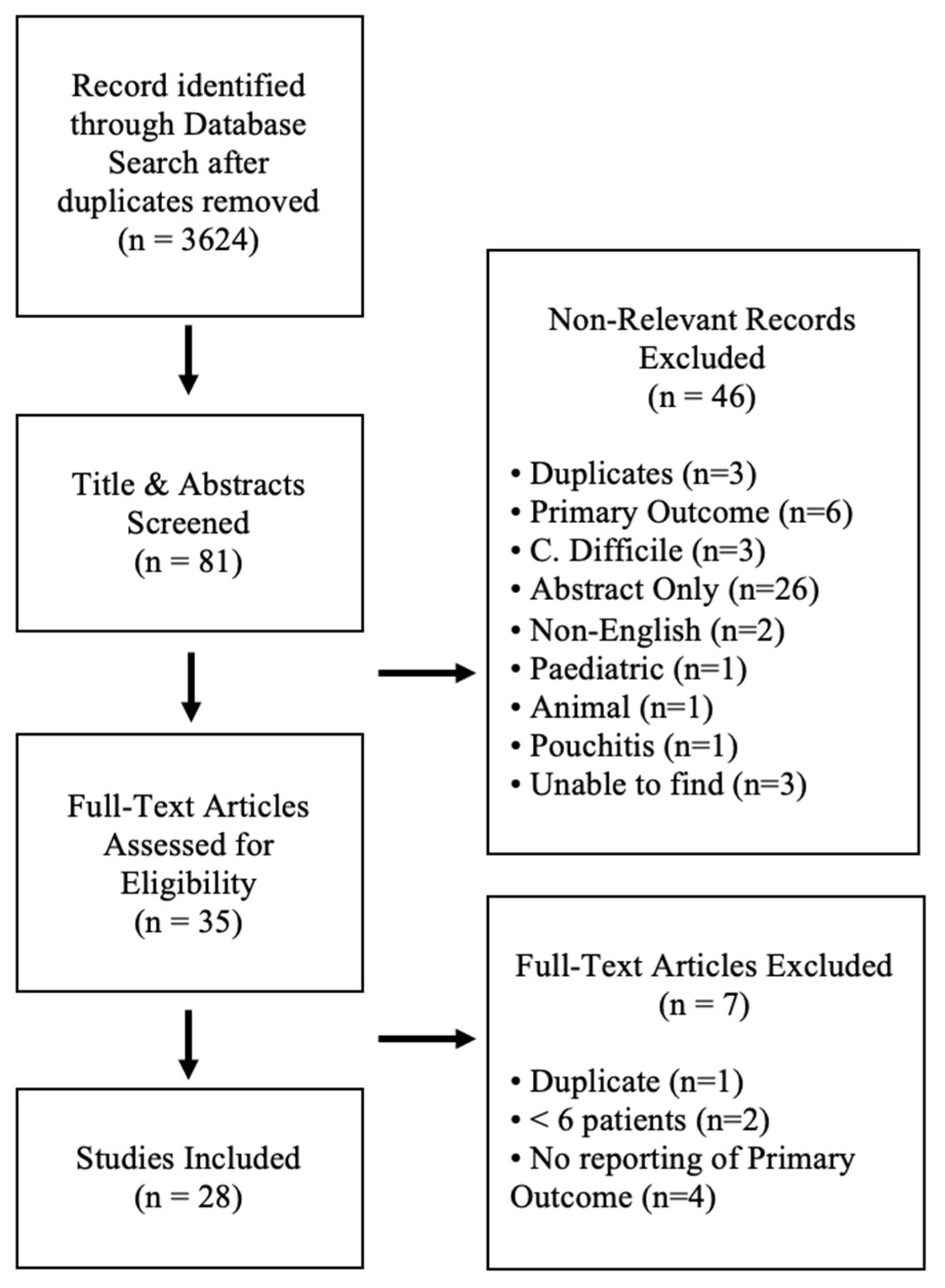
3.1. Search Results and Study Characteristics
3.2. Risk of Bias Assessment
3.3. Baseline Demographics
3.4. FMT Administration, Dosing, and Donor Characterization
3.5. Response and Remission Rates for Repeated FMT Regimens
3.6. Response and Remission Rates for Antibiotic Pre-Treatments
3.7. Fecal Microbiota Compositional Changes Following FMT
3.7.1. Overview of Microbiota Reporting of Included Studies
3.7.2. Changes in Alpha and Beta Diversity Following FMT
3.7.3. Recipient and Donor Microbial Ecology Associated with IBD Outcomes
3.8. Reported Adverse Events
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Loftus, E.V. Clinical epidemiology of inflammatory bowel disease: Incidence, prevalence, and environmental influences. Gastroenterology 2004, 126, 1504–1517. [Google Scholar] [CrossRef]
- Nikolaus, S.; Schreiber, S. Diagnostics of Inflammatory Bowel Disease. Gastroenterology 2007, 133, 1670–1689. [Google Scholar] [CrossRef] [PubMed]
- Molodecky, N.A.; Soon, I.S.; Rabi, D.M.; Ghali, W.A.; Ferris, M.; Chernoff, G.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Barkema, H.W.; et al. Increasing Incidence and Prevalence of the Inflammatory Bowel Diseases With Time, Based on Systematic Review. Gastroenterology 2012, 142, 46–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palmela, C.; Chevarin, C.; Xu, Z.; Torres, J.; Sevrin, G.; Hirten, R.; Barnich, N.; Ng, S.C.; Colombel, J.F. Adherent-invasive Escherichia coli in inflammatory bowel disease. Gut 2018, 67, 574–587. [Google Scholar] [CrossRef]
- Kim, D.H.; Cheon, J.H. Pathogenesis of Inflammatory Bowel Disease and Recent Advances in Biologic Therapies. Immune Netw. 2017, 17, 25–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, H.; Tremaroli, V.; Backhed, F. Linking Microbiota to Human Diseases: A Systems Biology Perspective. Trends Endocrinol. Metab. 2015, 26, 758–770. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.D.; Lewis, J.D. Analysis of the human gut microbiome and association with disease. Clin. Gastroenterol. Hepatol. 2013, 11, 774–777. [Google Scholar] [CrossRef] [Green Version]
- McIlroy, J.; Ianiro, G.; Mukhopadhya, I.; Hansen, R.; Hold, G.L. The gut microbiome in inflammatory bowel disease-avenues for microbial management. Aliment. Pharmacol. Ther. 2018, 47, 26–42. [Google Scholar] [CrossRef]
- Philpott, J.; Ashburn, J.; Shen, B. Efficacy of vedolizumab in patients with antibiotic and anti-tumor necrosis alpha refractory pouchitis. Inflamm. Bowel Dis. 2017, 23, E5–E6. [Google Scholar] [CrossRef] [Green Version]
- Wilhelm, S.M.; Love, B.L. Management of patients with inflammatory bowel disease: Current and future treatments. Clin. Pharm. 2017, 3, 83–92. [Google Scholar] [CrossRef]
- Furfaro, F.; Fiorino, G.; Allocca, M.; Gilardi, D.; Danese, S. Emerging therapeutic targets and strategies in Crohn’s disease. Expert Rev. Gastroenterol. Hepatol. 2016, 10, 735–744. [Google Scholar] [CrossRef] [PubMed]
- Abu-Sbeih, H.; Wang, Y. Management Considerations for Immune Checkpoint Inhibitor-Induced Enterocolitis Based on Management of Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2019. [Google Scholar] [CrossRef] [Green Version]
- Torres, J.; Danese, S.; Colombel, J. New therapeutic avenues in ulcerative colitis: Thinking out of the box. Gut 2013, 62, 1642–1652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chande, N.; Costello, S.P.; Limketkai, B.N.; Parker, C.E.; Nguyen, T.M.; Macdonald, J.K.; Feagan, B.G. Alternative and Complementary Approaches for the Treatment of Inflammatory Bowel Disease: Evidence From Cochrane Reviews. Inflamm. Bowel Dis. 2019, 26, 843–851. [Google Scholar] [CrossRef]
- Maharshak, N.; Cohen, N.A.; Reshef, L.; Tulchinsky, H.; Gophna, U.; Dotan, I. Low enteric microbial diversity in patients with ulcerative colitis after pouch surgery having a mature normal ileal pouch may be predictive of pouchitis. J. Crohn’s Colitis 2016, 10, S490. [Google Scholar] [CrossRef]
- Ishikawa, D.; Osada, T.; Sasaki, T.; Kuwahara-Arai, K.; Haga, K.; Shibuya, T.; Kodani, T.; Hiramatsu, K.; Watanabe, S. Alterations of intestinal microbiota in ulcerative colitis patients treated with sequential antibiotic combination and faecal microbiota transplantation. J. Crohn’s Colitis 2015, 9, S364–S365. [Google Scholar] [CrossRef]
- Zuo, T.; Ng, S.C. The Gut Microbiota in the Pathogenesis and Therapeutics of Inflammatory Bowel Disease. Front. Microbiol. 2018, 9, 2247. [Google Scholar] [CrossRef] [PubMed]
- Pigneur, B.; Sokol, H. Fecal microbiota transplantation in inflammatory bowel disease: The quest for the holy grail. Mucosal Immunol. 2016, 9, 1360–1365. [Google Scholar] [CrossRef] [PubMed]
- Gallo, A.; Passaro, G.; Gasbarrini, A.; Landolfi, R.; Montalto, M. Modulation of microbiota as treatment for intestinal inflammatory disorders: An uptodate. World J. Gastroenterol. 2016, 22, 7186–7202. [Google Scholar] [CrossRef]
- Zeng, W.; Shen, J.; Bo, T.; Peng, L.; Xu, H.; Nasser, M.I.; Zhuang, Q.; Zhao, M. Cutting Edge: Probiotics and Fecal Microbiota Transplantation in Immunomodulation. J. Immunol. Res. 2019, 2019, 1603758. [Google Scholar] [CrossRef] [Green Version]
- Weingarden, A.R.; Vaughn, B.P. Intestinal microbiota, fecal microbiota transplantation, and inflammatory bowel disease. Gut Microbes 2017, 8, 238–252. [Google Scholar] [CrossRef] [Green Version]
- Paramsothy, S.; Paramsothy, R.; Kamm, M.A.; Kaakoush, N.O.; Mitchell, H.M.; Rubin, D.T.; Castano-Rodriguez, N. Faecal microbiota transplantation for inflammatory bowel disease: A systematic review and meta-analysis. Gastroenterology 2017, 152, S1007. [Google Scholar] [CrossRef]
- Khoruts, A.; Rank, K.M.; Newman, K.M.; Viskocil, K.; Vaughn, B.P.; Hamilton, M.J.; Sadowsky, M.J. Inflammatory Bowel Disease Affects the Outcome of Fecal Microbiota Transplantation for Recurrent Clostridium difficile Infection. Clin. Gastroenterol. Hepatol. 2016, 14, 1433–1438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khanna, S.; Vazquez-Baeza, Y.; Gonazlez, A.; Weiss, S.; Schmidt, B.; Muniz-Pedrogo, D.A.; Rainey, J.F.; Kammer, P.; Nelson, H.; Sadowsky, M.; et al. Changes in microbial ecology after fecal microbiota transplantation for recurrent C-difficile infection affected by underlying inflammatory bowel disease. Microbiome 2017, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Langdon, A.; Crook, N.; Dantas, G. The effects of antibiotics on the microbiome throughout development and alternative approaches for therapeutic modulation. Genome Med. 2016, 8, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khoruts, A.; Sadowsky, M.J. Understanding the mechanisms of faecal microbiota transplantation. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 508–516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ishikawa, D.; Takahashi, M.; Ito, S.; Okahara, K.; Haga, K.; Kamei, M.; Nomura, O.; Shibuya, T.; Osada, T.; Nagahara, A. Eradication of dysbiotic indigenous species by multiple antibiotic pre-treatment contribute to effective faecal microbiota transplantation. J. Crohn’s Colitis 2018, 12, S553. [Google Scholar] [CrossRef] [Green Version]
- Ishikawa, D.; Sasaki, T.; Takahashi, M.; Okahara, K.; Ito, S.; Haga, K.; Shibuya, T.; Osada, T.; Nagahara, A. Combination therapy of fresh fecal microbial transplantation and triple-antibiotic therapy for ulcerative colitis. Am. J. Gastroenterol. 2019, 114, S14. [Google Scholar] [CrossRef]
- Shimizu, H.; Ohnishi, E.; Arai, K.; Takeuchi, I.; Kamura, H.; Hata, K. Outcome of the repetitive fecal microbiota transplantation using fecal solution prepared under the anaerobic condition following the antibiotic pretreatment in eight children with ulcerative colitis. Inflamm. Bowel Dis. 2019, 25, S21–S22. [Google Scholar] [CrossRef]
- Blesl, A.; Rainer, F.; Wurm, P.; Durdevic, M.; Petritsch, W.; Wenzl, H.; Baumann-Durchschein, F.; Posch, A.; Streit, A.; Gorkiewicz, G.; et al. Predictors of non-response to repeated faecal microbiota transplantation in patients with therapy refractory ulcerative colitis. J. Crohn’s Colitis 2019, 13, S412. [Google Scholar] [CrossRef]
- Borody, T.J.; Warren, E.F.; Leis, S.; Surace, R.; Ashman, O. Treatment of ulcerative colitis using fecal bacteriotherapy. J. Clin. Gastroenterol. 2003, 37, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, D.; Okahara, K.; Ito, S.; Takahashi, M.; Haga, K.; Nomura, K.; Shibuya, T.; Nagahara, A. Efficacy of combination of fresh fecal microbiota transplantation and triple-antibiotic therapy for ulcerative colitis. Inflamm. Bowel Dis. 2019, 25, S73. [Google Scholar] [CrossRef]
- Ishikawa, D.; Takahashi, M.; Okahara, K.; Ito, S.; Haga, K.; Shibuya, T.; Osada, T.; Nagahara, A. Efficacy of combination therapy of fresh faecal microbiota transplantation and triple-antibiotic therapy for ulcerative colitis. J. Crohn’s Colitis 2019, 13, S301. [Google Scholar] [CrossRef]
- Wei, Y.; Zhu, W.; Gong, J.; Guo, D.; Gu, L.; Li, N.; Li, J. Fecal Microbiota Transplantation Improves the Quality of Life in Patients with Inflammatory Bowel Disease. Gastroenterol. Res. Pract. 2015, 2015, 517597. [Google Scholar] [CrossRef]
- Costello, S.P.; Hughes, P.A.; Waters, O.; Bryant, R.V.; Vincent, A.D.; Blatchford, P.; Katsikeros, R.; Makanyanga, J.; Campaniello, M.A.; Mavrangelos, C.; et al. Effect of Fecal Microbiota Transplantation on 8-Week Remission in Patients With Ulcerative Colitis: A Randomized Clinical Trial. JAMA 2019, 321, 156–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sood, A.; Mahajan, R.; Singh, A.; Midha, V.; Mehta, V.; Narang, V.; Singh, T.; Singh Pannu, A. Role of Faecal Microbiota Transplantation for Maintenance of Remission in Patients With Ulcerative Colitis: A Pilot Study. J. Crohn’s. Colitis 2019, 13, 1311–1317. [Google Scholar] [CrossRef]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef] [Green Version]
- Higgins, J.P.T. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0; Cochrane Collaboration: London, UK, 2011; Available online: https://ci.nii.ac.jp/naid/20000796633/ (accessed on 19 February 2021).
- Hozo, S.P.; Djulbegovic, B.; Hozo, I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med. Res. Methodol. 2005, 10, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Chapter 10: Analysing Data and Undertaking Meta-Analyses; Cochrane: London, UK, 2019. [Google Scholar]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 3, 557–560. [Google Scholar] [CrossRef] [Green Version]
- Chen, M.; Liu, X.L.; Zhang, Y.J.; Nie, Y.Z.; Wu, K.C.; Shi, Y.Q. Efficacy and safety of fecal microbiota transplantation by washed preparation in patients with moderate to severely active ulcerative colitis. J. Dig. Dis. 2020, 21, 621–628. [Google Scholar] [CrossRef]
- Chen, H.T.; Huang, H.L.; Xu, H.M.; Luo, Q.L.; He, J.; Li, Y.Q.; Zhou, Y.L.; Nie, Y.Q.; Zhou, Y.J. Fecal microbiota transplantation ameliorates active ulcerative colitis. Exp. Ther. Med. 2020, 19, 2650–2660. [Google Scholar] [CrossRef] [Green Version]
- Cold, F.; Browne, P.D.; Gunther, S.; Halkjaer, S.I.; Petersen, A.M.; Al-Gibouri, Z.; Hansen, L.H.; Christensen, A.H. Multidonor FMT capsules improve symptoms and decrease fecal calprotectin in ulcerative colitis patients while treated—An open-label pilot study. Scand. J. Gastroenterol. 2019, 54, 289–296. [Google Scholar] [CrossRef]
- Cui, B.; Feng, Q.; Wang, H.; Wang, M.; Peng, Z.; Li, P.; Huang, G.; Liu, Z.; Wu, P.; Fan, Z.; et al. Fecal microbiota transplantation through mid-gut for refractory Crohn’s disease: Safety, feasibility, and efficacy trial results. J. Gastroenterol. Hepatol. 2015, 30, 51–58. [Google Scholar] [CrossRef]
- Dang, X.F.; Wang, Q.X.; Yin, Z.; Sun, L.; Yang, W.H. Recurrence of moderate to severe ulcerative colitis after fecal microbiota transplantation treatment and the efficacy of re-FMT: A case series. BMC Gastroenterol. 2020, 20, 401. [Google Scholar] [CrossRef]
- Damman, C.J.; Brittnacher, M.J.; Westerhoff, M.; Hayden, H.S.; Radey, M.; Hager, K.R.; Marquis, S.R.; Miller, S.I.; Zisman, T.L. Low Level Engraftment and Improvement following a Single Colonoscopic Administration of Fecal Microbiota to Patients with Ulcerative Colitis. PLoS ONE 2015, 10, e0133925. [Google Scholar] [CrossRef] [Green Version]
- Ishikawa, D.; Sasaki, T.; Osada, T.; Kuwahara-Arai, K.; Haga, K.; Shibuya, T.; Hiramatsu, K.; Watanabe, S. Changes in Intestinal Microbiota Following Combination Therapy with Fecal Microbial Transplantation and Antibiotics for Ulcerative Colitis. Inflamm. Bowel Dis. 2017, 23, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Jacob, V.; Crawford, C.; Cohen-Mekelburg, S.; Viladomiu, M.; Putzel, G.G.; Schneider, Y.; Chabouni, F.; O’Neil, S.; Bosworth, B.; Woo, V.; et al. Single Delivery of High-Diversity Fecal Microbiota Preparation by Colonoscopy Is Safe and Effective in Increasing Microbial Diversity in Active Ulcerative Colitis. Inflamm. Bowel Dis. 2017, 23, 903–911. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kump, P.; Wurm, P.; Grochenig, H.P.; Wenzl, H.; Petritsch, W.; Halwachs, B.; Wagner, M.; Stadlbauer, V.; Eherer, A.; Hoffmann, K.M.; et al. The taxonomic composition of the donor intestinal microbiota is a major factor influencing the efficacy of faecal microbiota transplantation in therapy refractory ulcerative colitis. Aliment. Pharmacol. Ther. 2018, 47, 67–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mizuno, S.; Nanki, K.; Matsuoka, K.; Saigusa, K.; Ono, K.; Arai, M.; Sugimoto, S.; Kiyohara, H.; Nakashima, M.; Takeshita, K.; et al. Single fecal microbiota transplantation failed to change intestinal microbiota and had limited effectiveness against ulcerative colitis in Japanese patients. Intest. Res. 2017, 15, 68–74. [Google Scholar] [CrossRef] [Green Version]
- Moayyedi, P.; Surette, M.G.; Kim, P.T.; Libertucci, J.; Wolfe, M.; Onischi, C.; Armstrong, D.; Marshall, J.K.; Kassam, Z.; Reinisch, W.; et al. Fecal Microbiota Transplantation Induces Remission in Patients With Active Ulcerative Colitis in a Randomized Controlled Trial. Gastroenterology 2015, 149, 102–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishida, A.; Imaeda, H.; Ohno, M.; Inatomi, O.; Bamba, S.; Sugimoto, M.; Andoh, A. Efficacy and safety of single fecal microbiota transplantation for Japanese patients with mild to moderately active ulcerative colitis. J. Gastroenterol. 2017, 52, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Okahara, K.; Ishikawa, D.; Nomura, K.; Ito, S.; Haga, K.; Takahashi, M.; Shibuya, T.; Osada, T.; Nagahara, A. Matching between Donors and Ulcerative Colitis Patients Is Important for Long-Term Maintenance after Fecal Microbiota Transplantation. J. Clin. Med. 2020, 9, 1650. [Google Scholar] [CrossRef] [PubMed]
- Paramsothy, S.; Kamm, M.A.; Kaakoush, N.O.; Walsh, A.J.; van den Bogaerde, J.; Samuel, D.; Leong, R.W.L.; Connor, S.; Ng, W.; Paramsothy, R.; et al. Multidonor intensive faecal microbiota transplantation for active ulcerative colitis: A randomised placebo-controlled trial. Lancet 2017, 389, 1218–1228. [Google Scholar] [CrossRef]
- Rossen, N.G.; MacDonald, J.K.; de Vries, E.M.; D’Haens, G.R.; de Vos, W.M.; Zoetendal, E.G.; Ponsioen, C.Y. Fecal microbiota transplantation as novel therapy in gastroenterology: A systematic review. World J. Gastroenterol. 2015, 21, 5359–5371. [Google Scholar] [CrossRef] [PubMed]
- Schierova, D.; Brezina, J.; Mrazek, J.; Fliegerova, K.O.; Kvasnova, S.; Bajer, L.; Drastich, P. Gut Microbiome Changes in Patients with Active Left-Sided Ulcerative Colitis after Fecal Microbiome Transplantation and Topical 5-aminosalicylic Acid Therapy. Cells 2020, 9, 2283. [Google Scholar] [CrossRef]
- Sokol, H.; Landman, C.; Seksik, P.; Berard, L.; Montil, M.; Nion-Larmurier, I.; Bourrier, A.; Le Gall, G.; Lalande, V.; De Rougemont, A.; et al. Fecal microbiota transplantation to maintain remission in Crohn’s disease: A pilot randomized controlled study. Microbiome 2020, 8, 12. [Google Scholar] [CrossRef]
- Sood, A.; Mahajan, R.; Juyal, G.; Midha, V.; Grewal, C.S.; Mehta, V.; Singh, A.; Joshi, M.C.; Narang, V.; Kaur, K.; et al. Efficacy of fecal microbiota therapy in steroid dependent ulcerative colitis: A real world intention-to-treat analysis. Intest. Res. 2019, 17, 78–86. [Google Scholar] [CrossRef] [Green Version]
- Sood, A.; Singh, A.; Mahajan, R.; Midha, V.; Kaur, K.; Singh, D.; Bansal, N.; Dharni, K. Clinical Predictors of response to Faecal Microbiota Transplantation in patients with active ulcerative colitis. J. Crohn’s. Colitis 2020, jjaa163. [Google Scholar] [CrossRef]
- Uygun, A.; Ozturk, K.; Demirci, H.; Oger, C.; Avci, I.Y.; Turker, T.; Gulsen, M. Fecal microbiota transplantation is a rescue treatment modality for refractory ulcerative colitis. Medicine 2017, 96, e6479. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, B.P.; Vatanen, T.; Allegretti, J.R.; Bai, A.; Xavier, R.J.; Korzenik, J.; Gevers, D.; Ting, A.; Robson, S.C.; Moss, A.C. Increased Intestinal Microbial Diversity Following Fecal Microbiota Transplant for Active Crohn’s Disease. Inflamm. Bowel Dis. 2016, 22, 2182–2190. [Google Scholar] [CrossRef] [Green Version]
- Vermeire, S.; Joossens, M.; Verbeke, K.; Wang, J.; Machiels, K.; Sabino, J.; Ferrante, M.; Van Assche, G.; Rutgeerts, P.; Raes, J. Donor Species Richness Determines Faecal Microbiota Transplantation Success in Inflammatory Bowel Disease. J. Crohn’s. Colitis 2016, 10, 387–394. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Ren, R.; Sun, G.; Peng, L.; Tian, Y.; Yang, Y. Pilot study of cytokine changes evaluation after fecal microbiota transplantation in patients with ulcerative colitis. Int. Immunopharmacol. 2020, 85, 106661. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Bu, C.; Yuan, W.; Shen, Z.; Quan, Y.; Wu, S.; Zhu, C. Fecal Microbiota Transplant via Endoscopic Delivering Through Small Intestine and Colon: No Difference for Crohn’s Disease. Dig. Dis. Sci. 2020, 65, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Cui, B.; Li, P.; Zhang, F. Short-term surveillance of cytokines and CRP cannot predict efficacy of fecal microbiota transplantation for ulcerative colitis. Gastroenterology 2016, 150, S380–S381. [Google Scholar] [CrossRef]
- Yu, E.W.; Gao, L.; Stastka, P.; Cheney, M.C.; Mahabamunuge, J.; Torres Soto, M.; Ford, C.B.; Bryant, J.A.; Henn, M.R.; Hohmann, E.L. Fecal microbiota transplantation for the improvement of metabolism in obesity: The FMT-TRIM double-blind placebo-controlled pilot trial. PLOS Med. 2020, 17, e1003051. [Google Scholar] [CrossRef]
- El-Salhy, M.; Mazzawi, T. Fecal microbiota transplantation for managing irritable bowel syndrome. Expert Rev. Gastroenterol. Hepatol. 2018, 12, 439–445. [Google Scholar] [CrossRef]
- Baunwall, S.M.; Lee, M.M.; Eriksen, M.K.; Mullish, B.H.; Marchesi, J.R.; Dahlerup, J.F.; Hvas, C.L. Faecal microbiota transplantation for recurrent Clostridioides difficile infection: An updated systematic review and meta-analysis. EClinicalMedicine 2020, 29, 100642. [Google Scholar] [CrossRef]
- Kim, K.O.; Gluck, M. Fecal Microbiota Transplantation: An Update on Clinical Practice. Clin. Endosc. 2019, 52, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Freitag, T.L.; Hartikainen, A.; Jouhten, H.; Sahl, C.; Meri, S.; Anttila, V.-J.; Mattila, E.; Arkkila, P.; Jalanka, J.; Satokari, R. Minor Effect of Antibiotic Pre-treatment on the Engraftment of Donor Microbiota in Fecal Transplantation in Mice. Front. Microbiol. 2019, 10, 2685. [Google Scholar] [CrossRef]
- Oliphant, K.; Cochrane, K.; Schroeter, K.; Daigneault, M.C.; Yen, S.; Verdu, E.F.; Allen-Vercoe, E. Effects of Antibiotic Pretreatment of an Ulcerative Colitis-Derived Fecal Microbial Community on the Integration of Therapeutic Bacteria In Vitro. mSystems 2020, 5, e00404-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ji, S.K.; Yan, H.; Jiang, T.; Guo, C.Y.; Liu, J.J.; Dong, S.Z.; Yang, K.L.; Wang, Y.J.; Cao, Z.J.; Li, S.L. Preparing the Gut with Antibiotics Enhances Gut Microbiota Reprogramming Efficiency by Promoting Xenomicrobiota Colonization. Front. Microbiol. 2017, 8, 1208. [Google Scholar] [CrossRef] [PubMed]


| Study | Study Design | Patients (n) | Country | Disease | Severity | FMT Delivery | FMT Donor | FMT Dosage | FMT Frequency | Pre-Treatment Antibiotics | Antibiotic Frequency | Total Follow-up (Weeks) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Borody 2003 [31] | Case series | 6 | Australia | UC | Severe | Enema | Healthy donors chosen by patient | 200–300 g/200–300 mL saline | Daily for 5 days | Vancomycin (500 mg bid), metronidazole (400 mg bid), rifampicin (150 mg bid) | 7–10 days | 676 |
| Chen 2020 [42] | Prospective cohort | 9 | China | UC | Moderate-severe | Naso-jejunal | Healthy donor | 200–250 mL of fecal suspension | 3 doses at 1, 3 and 5 days | - | - | 12 |
| Chen 2020 [43] | Prospective cohort | 44 | China | UC | Mild- moderate | Colonoscopy | Healthy donor | 150–200 g stool/1000 mL saline | ×3 in 1 week | - | - | 12 |
| Cold 2019 [44] | Prospective cohort | 7 | Denmark | UC | Active | Oral capsules | Healthy volunteers | 12 g daily dose of 25 capsules | 25 capsules/day for 50 days | - | - | 24 |
| Costello 2019 [35] | RCT | 73 | Australia | UC | Mild- moderate | Colonoscopy and enema | Healthy volunteers recruited by advertisement | 50 g/200 mL saline colonoscopy, 25 g/100 mL saline enema | 1× colonoscopy then 2× enemas over 1 week | - | - | 8 |
| Cui 2015 [45] | Prospective cohort | 30 | China | CD | Moderate-severe | Gastroscopy | Related or unrelated volunteer | 60 mL/100 mL saline | ×1 | - | - | 65 |
| Dang 2020 [46] | Case series | 12 | China | UC | Moderate-severe | Colonoscopy | Healthy volunteers | 15 mL bacterial pellet in 75 mL saline | multiple, exact frequency NR | - | - | 52 |
| Damman 2015 [47] | Prospective cohort | 7 | USA | UC | Mild- moderate | Colonoscopy | Chosen by patient | Diluted with 2–3 mL saline/g of stool | ×1 | - | - | 12 |
| Ishikawa 2017 [48] | Prospective cohort | 36 | Japan | UC | Mild-severe | Colonoscopy | Spouse or relative | 150–250 g/350–500 mL saline | ×1 | Amoxicillin (1500 mg/day), Fosfomycin (3000 mg/day), metronidazole (750 mg/day) | 2 weeks until 2 days before FMT | 4 |
| Jacob 2017 [49] | Prospective cohort | 20 | USA | UC | Active | Colonoscopy | Healthy donor | 60 mL | ×1 | - | - | 4 |
| Kump 2017 [50] | Prospective cohort | 27 | Austria | UC | Mild-severe | Colonoscopy | Related or unrelated volunteer | 50 g/200–500 mL saline | 5×, 14 days apart | Vancomycin (250 mg qid), paromomycin (250 mg tid), nystatin (10 mL, 1 million IE qid) | 10 days | 13 |
| Mizuno 2017 [51] | Prospective cohort | 10 | Japan | UC | Moderate-severe | Colonoscopy | Healthy relatives | 50–300 g/50–100 mL saline | ×1 | - | - | 12 |
| Moayyedi 2015 [52] | RCT | 70 | Canada | UC | Mild-moderate | Enema | Healthy donors | 50 g/300 mL water | ×6; 0, 1, 2, 3, 4, 5, 6 weeks | - | - | 7 |
| Nishida 2017 [53] | Prospective cohort | 41 | Japan | UC | Mild-moderate | Colonoscopy | Healthy relatives | 150–200 g/500 mL saline | ×1 | - | - | 12 |
| Okahara 2020 [54] | Prospective cohort | 92 | Japan | UC | Mild-severe | Colonoscopy | Spouses and relatives | 350–500 mL filtered bacterial suspension infusion | ×1 | Amoxicillin (1500 mg/day), Fosfomycin (3000 mg/day), metronidazole (750 mg/day) | 2 weeks prior to FMT | 104 |
| Paramsothy 2017 [55] | RCT | 85 | Australia | UC | Mild-moderate | Colonoscopy and enema | Healthy volunteers recruited by advertisement | 37.5 g | ×5/week for 8 weeks | - | - | 8 |
| Rossen 2015 [56] | RCT | 50 | Finland | UC | Mild- moderate | Nasoduodenal tube | Relatives, partner, or volunteer | 120 g | ×2; 3 weeks apart | - | - | 12 |
| Schierova 2020 [57] | Prospective cohort | 16 | Czech Republic | UC | NR | Enema | Healthy donors | 50 g stool/150 mL saline | 5×/week for 1 week then weekly × 6 weeks | - | - | 12 |
| Sokol 2020 [58] | RCT | 17 | France | CD | NR | Colonoscopy | Healthy donors | 50–100 g/250–350 mL saline | ×1 | - | - | 24 |
| Sood 2019 [59] | Prospective cohort | 41 | India | UC | Mild- moderate | Colonoscopy | Two healthy unrelated volunteers | NR | ×7; 0, 2, 6, 10, 14, 18, 22 weeks | - | - | 22 |
| Sood 2020 [60] | Prospective cohort | 140 | India | UC | Moderate-severe | Colonoscopy | Healthy donors | 80 g stool/ 200 mL saline | ×7; 0, 2, 6, 10, 14, 18, 22 weeks | - | - | 30 |
| Uygun 2017 [61] | Prospective cohort | 30 | Turkey | UC | Moderate-severe | Colonoscopy | Relatives, partner, or volunteer | 120–150 g | ×1 | - | - | 12 |
| Vaughn 2016 [62] | Prospective cohort | 19 | USA | CD | Active | Colonoscopy | Healthy unrelated volunteers | 50 g/250 mL saline | ×1 | - | - | 4 |
| Vermeire 2016 [63] | Prospective cohort | 14 | Belgium | UC+ CD | Refractory | Naso-jejunal or rectal tube | Family, friend, or partner | 200 g/400 mL saline | ×2; 2 consecutive days | - | - | 8 |
| Wang 2020 [64] | Prospective cohort | 16 | China | UC | Moderate- severe | Colonoscopy | Healthy donor | 100 g stool/ 500 mL saline | ×3; 2–3 month intervals | - | - | >24 |
| Wei 2015 [34] | Prospective cohort | 14 | China | UC+ CD | NR | Colonoscopy or naso-jejunal tube | Healthy unrelated donor | 60 g/350 mL saline | ×1 | Vancomycin (500 mg) | Twice a day for 3 days before FMT | 4 |
| Yang 2019 [65] | RCT | 27 | China | CD | Mild- moderate | Gastroscopy or colonoscopy | Healthy donors | 200 g in saline | ×2; 1 week apart | - | - | 2 |
| Zhang 2016 [66] | Prospective cohort | 19 | China | UC | Moderate-severe | Gastroscopy | NR | NR | ×1 | - | - | 13 |
| Study | Disease | Intervention Arm | Patients (n) | Age | Sex (% male) | Disease Duration (Years) | Ongoing Systemic Corticosteroids (%) | Total Mayo Score | CDAI |
|---|---|---|---|---|---|---|---|---|---|
| Borody 2003 | UC | Antibiotic pre-treatment and repeated FMT | 6 | 35.8 (11.0) | 50.0 | 11.7 (5.8) | NR | NR | - |
| Chen 2020 | UC | Repeated FMT | 9 | 47.9 (10.6) | 77.8 | 5.3 (5.1) | 33.3 | 5.9 (2.0) | - |
| Chen 2020 | UC | Repeated FMT | 44 | 44.4 (15.5) | 57 | 4.6 (2.1) | 25.0 | 5.9 (2.0) | - |
| Cold 2019 | UC | Repeated FMT | 7 | 38.3 (5.8) | 71.4 | 10.8 (3.8) | NR | NR | - |
| Costello 2019 | UC | Repeated donor FMT | 38 | 38.5 (6) | 53.0 | 4.9 (4.8) | 21.0 | 7.2 (1.7) | - |
| Repeated autologous FMT | 35 | 35.0 (5.25) | 57.0 | 5.8 (2.2) | 31.0 | 7.4 (1.9) | - | ||
| Cui 2015 | CD | Single FMT | 30 | 38.0 (13.8) | 64.5 | 7.4 (5.3) | 56.7 | NR | NR |
| Damman 2015 | UC | Single FMT | 8 | 41.1 (15.5) | 25.0 | 16.6 (13.1) | NR | NR | - |
| Dang 2020 | UC | Repeated FMT | 12 | 51 (14.0) | 66.0 | NR | 41.7 | NR | - |
| Ishikawa 2017 | UC | Antibiotic pre-treatment and single FMT | 17 | 40.4 (14.2) | 76.5 | 7.8 (8.4) | 29.4 | 7.5 (1.9) | - |
| Antibiotic pre-treatment only | 19 | 44.8 (14.9) | 63.2 | 7.0 (8.0) | 47.4 | 8.2 (2.2) | - | ||
| Jacob 2017 | UC | Single FMT | 20 | 38.4 (12.6) | 60.0 | NR | 30.0 | 8.1 (2.4) | - |
| Kump 2017 | UC | Antibiotic pre-treatment and repeated FMT | 17 | 44.0 (18.0) | 82.0 | 8.0 (8.0) | 59.0 | 8.9 (1.6) | - |
| Antibiotic pre-treatment only | 10 | 36.0 (13.0) | 30.0 | 7.0 (6.0) | 30.0 | 8.1 (3.1) | - | ||
| Mizuno 2017 | UC | Single FMT | 10 | 31.8 (7.8) | 70.0 | 6.25 (3.5) | NR | 6.1 (1.0) | - |
| Moayyedi 2015 | UC | Repeated FMT | 38 | 42.2 (15.0) | 47.0 | 7.9 (5.6) | 39 | 8.2 (2.6) | - |
| Placebo | 37 | 35.8 (12.1) | 70.0 | 7.0 (6.8) | 35 | 7.9 (2.3) | - | ||
| Nishida 2017 | UC | Single FMT | 41 | 39.6 (16.9) | 68.3 | 7.6 (8.6) | 26.8 | 5.6 (2.4) | - |
| Okahara 2020 | UC | Antibiotic pre-treatment Single FMT | 55 | 40.1 (13.3) | 69.1 | 8.6 (7.4) | 43.2 | 6.3 (4.1) | - |
| Paramsothy 2017 | UC | Repeated FMT | 41 | 35.6 (5.3) | 54.0 | 5.8 (1.4) | 22.0 | 8 (0.8) | - |
| Placebo | 40 | 35.4 (4.5) | 63.0 | 5.8 (1.4) | 28.0 | 8 (0.8) | - | ||
| Rossen 2015 | UC | Single donor FMT | 23 | 42.3 (5.8) | 47.8 | 7 (NR) | 21.7 | NR | - |
| Single autologous FMT | 25 | 41 (4.5) | 44.0 | 9 (NR) | 20.0 | NR | - | ||
| Schierova 2020 | UC | Repeated FMT | 8 | 41.3 (10.1) | 50.0 | NR | 0 | 5.8 (1.7) | - |
| Medical therapy | 8 | 44.3 (10.4) | 50.0 | NR | 25.0 | 6.0 (1.5) | - | ||
| Sokol 2020 | CD | Single FMT | 8 | 31.8 (6.8) | 62.5 | 8.5 (8.1) | 100 | NR | 89 (30.5) |
| Placebo | 9 | 38.3 (6.0) | 44.4 | 11.3 (2.0) | 100 | NR | 61.5 (20.1) | ||
| Sood 2019 | UC | Repeated FMT | 41 | 36.5 (10.7) | 58.5 | 4.6 (4.2) | 100 | 8.8 (2.6) | - |
| Sood 2020 | UC | Repeated FMT | 93 | 35 (11) | 62.4 | 5.2 (4.6) | 78.5 | 8.1 (2.0) | - |
| Uygun 2017 | UC | Single FMT | 30 | 34.6 (10.3) | 46.7 | 5.3 (3.3) | NR | 11.1 (1.1) | - |
| Vaughn 2016 | CD | Single FMT | 19 | 36 (12.3) | 63.0 | 12.5 (10.6) | 42.0 | NR | NR |
| Vermeire 2016 | UC and CD | Repeated FMT | 14 | 38.6 (8.2) | 50.0 | 10.2 (7.5) | 21.4 | 8.4 (0.6) | 290 (29) |
| Wang 2020 | UC | Repeated FMT | 16 | 39.5 (4) | 62.5 | 7.5 (5.8) | NR | 9.9 (2.2) | - |
| Wei 2015 | UC and CD | Antibiotic pre-treatment and single FMT | 14 | 43.5 (16.4) | 42.9 | 4.1 (3.2) | 7.1 | 5.8 (1.9) | 345 (77.8) |
| Yang 2019 | CD | Repeated FMT | 30 | 72.2 (10.8)) | 57.9 | 1.3 (0.4) | NR | NR | 283 (131) |
| Zhang 2016 | UC | Single FMT | 19 | 39.2 (14.1) | 36.8 | 8.0 (5.8) | NR | 10.5 (1.7) | - |
| Study | Intervention Arm | Follow-Up at Response/Remission (Weeks) | Patients (n) | Response (%) | Remission (%) |
|---|---|---|---|---|---|
| Borody 2003 | Antibiotic pre-treatment and repeated FMT | 676 | 6 | 6 (100%) | 6 (100%) |
| Chen 2020 | Repeated FMT | 2 weeks for response 12 weeks for remission | 9 | 7 (77.8%) | 5 (55.6%) |
| Chen 2020 | Repeated FMT | 12 | 44 | 34 (77.3%) | 30 (68.2%) |
| Cold 2019 | Repeated FMT | 24 | 7 | 7 (100%) | 4 (57.1%) |
| Costello 2019 | Repeated donor FMT | 8 | 38 | 21 (55%) | 18 (47%) |
| Repeated autologous FMT | 8 | 35 | 8 (23%) | 6 (17%) | |
| Cui 2015 | Single FMT | 12–72 | 15 | 8 (53.3%) | 4 (26.7%) |
| Dang 2020 | Repeated FMT | 52 | 12 | 11 (91.7%) | 5 (41.7%) |
| Damman 2015 | Single FMT | 4 | 7 | 1 (14.3%) | 1 (14.3%) |
| Ishikawa 2017 | Antibiotic pre-treatment and single FMT | 4 | 17 | 14 (82.3%) | 9 (52.9%) |
| Antibiotic pre-treatment only | 4 | 19 | 13 (68.4%) | 3 (15.8%) | |
| Jacob 2017 | Single FMT | 4 | 20 | 7 (35%) | 3 (15%) |
| Kump 2017 | Antibiotic pre-treatment and repeated FMT | 13 | 17 | 10 (59%) | 4 (24%) |
| Antibiotic pre-treatment only | 13 | 10 | 1 (10%) | 0 (0%) | |
| Mizuno 2017 | Single FMT | 12 | 10 | 1 (10%) | 0 (0%) |
| Moayyedi 2015 | Repeated FMT | 7 | 38 | 15 (39%) | 9 (24%) |
| Placebo | 7 | 37 | 9 (24%) | 2 (5%) | |
| Nishida 2017 | Single FMT | 8 | 41 | 11 (26.8%) | 0 (0%) |
| Okahara 2020 | Single FMT | 4 | 55 | 31 (56.3%) | 19 (34.5%) |
| Paramsothy 2017 | Repeated FMT | 8 | 41 | 22 (54%) | 18 (44%) |
| Placebo | 8 | 40 | 9 (23%) | 8 (20%) | |
| Rossen 2015 | Repeated donor FMT | 12 | 23 | 11 (47.8%) | 7 (30.4%) |
| Repeated autologous FMT | 12 | 25 | 13 (52.0%) | 8 (32.0%) | |
| Schierova 2020 | Repeated FMT | 12 | 8 | 5 (62.5%) | 1 (12.5%) |
| Sokol 2020 | Single FMT | 24 | 8 | NR | 4 (50%) |
| Sood 2019 | Repeated FMT | 22 | 41 | 31 (75.6%) | 19 (46.3%) |
| Sood 2020 | Repeated FMT | 30 | 93 | NR | 57 (61.3%) |
| Uygun 2017 | Single FMT | 12 | 30 | 21 (70%) | 13 (43.3%) |
| Vaughn 2016 | Single FMT | 4 | 19 | 11 (58%) | 10 (53%) |
| Vermeire 2016 | Repeated FMT | 6 weeks for response 8 weeks for remission | 14 | 4 (50%) | 2 (14.3%) |
| Wang 2020 | Repeated FMT | >6 mo | 16 | 14 (87.5%) | 0 (0%) |
| Wei 2015 | Antibiotic pre-treatment and single FMT | 4 | 14 | 14 (100%) | 14 (100%) |
| Yang 2019 | Repeated FMT | 2 | 27 | 21 (77.8%) | 18 (66.7%) |
| Zhang 2016 | Single FMT | 13 | 19 | 11 (57.9%) | 2 (10.5%) |
| Study | Methods | Donor Microbiota Differences vs. Recipient | Recipient Microbiota Changes Following FMT | Recipient Microbiota Changes Associated with Response/Remission |
|---|---|---|---|---|
| Borody 2003 | NR | NR | NR | NR |
| Chen 2020 | NR | NR | NR | NR |
| Chen 2020 | 16 s rRNA | ↑ α—diversity (Shannon, Chao1) | ↑ α—diversity (Shannon, Chao1) ↑ F. Prausnitzii | NR |
| Cold 2019 | 16 s rRNA | NR | No change in α—diversity (Shannon, Simpson) | NR |
| Costello 2019 | 16 s rRNA | NR | ↑ α—diversity (operational taxonomic units—OTUs) ↑ Peptococcus niger, ↑ Faecalicoccus pleomorphus, ↑ Olsenella sp., ↑ Acidaminococcus intestini, ↑ Prevotella copri, ↑ Clostridium methylpentosum, ↑ Allistipes indistinctus, ↑ Odoribacter splanchnicus ↓ Anaerostipescaccae, ↓ Clostridium aldenense | NR |
| Cui 2015 | NR | NR | NR | NR |
| Damman 2015 | Metagenomic Shotgun Sequencing | NR | No significant difference in α diversity (Shannon) ↑ Actinobacteria, ↑ Bacteroidetes (Prevotella copri) | NR |
| Dang 2020 | NR | NR | NR | NR |
| Ishikawa 2017 | 16 s rDNA | NR | ↑ Bacteroidetes | NR |
| Jacob 2017 | 16 s rRNA | NR | ↑ α—diversity (OTUs, Shannon) Change in β—diversity (Bray-Curtis) towards donor | NR |
| Kump 2017 | 16 s rRNA | ↑ unclassified Ruminococcus sp., ↑ Akkermansia muciniphila | No change in α—diversity (Chao1) Change in β—diversity (Bray-Curtis) towards donor | ↑ Akkermansia, ↓ Dialister sp. Change in β—diversity (Bray-Curtis) towards donor in responders |
| Mizuno 2017 | 16 s rRNA | NR | No significant difference in diversity or composition | NR |
| Moayyedi 2015 | 16 s rRNA | ↑ Lachnospiraceae, ↑ Ruminococcus | Change in β—diversity (Bray-Curtis) towards donor | Change in β—diversity (Bray-Curtis) towards donor |
| Nishida 2017 | 16 s rRNA | ↑ Bifidobacterium | No significant difference in α—diversity (Shannon) or β—diversity (Bray-Curtis) between responders and non-responders | NR |
| Okahara 2020 | HSP60 Bacteroidetes Sequencing | NR | Increase in similarity of Bacteroidetes species to donor | ↑ Bacteroides uniformis, ↑ Parabacteroides distasonis, ↑ Bacteroides dorei |
| Paramsothy 2017 | 16 s rRNA shotgun sequencing | NR | ↑ α—diversity (OTUs, Shannon) Shift towards donor at OTU level ↑ Prevotella spp., ↓ Bacteroides spp. | ↑Barnesiella spp., ↑ Parabacteroides spp., ↑ Clostridium cluster IV, ↑ Ruminococcus spp. |
| Rossen 2015 | 16 s rRNA | NR | ↑ α—diversity (OTUs, Shannon) ↑ Clostridium clusters IV, XIVa, XVIII ↓ Bacteroidetes | NR |
| Schierova 2020 | 16 sRNA | NR | No difference in α—diversity (Shannon, Chao1, Faith’s phylogenetic diversity) or β—diversity | ↑ Lachnospiraceae, ↑ Ruminococcaeae, ↑ Clostridaceae, ↑ Bifidobacteriaceae, ↑ Coriobacteriaceace ↑Faecalibacterium ↑ Blautia, ↑ Coriobacteria, ↑ Collinsella, ↑ Slackia, ↑ Bifidobacterium |
| Sokol 2020 | 16 s rRNA | NR | Transient ↑ α—diversity (Shannon, Chao1) Trend towards change in β—diversity (Bray-Curtis, Sorensen similarity index) between donor/recipient correlated | Sorensen index similarity showing improved engraftment; ↑ Ruminococcaecea, ↑ Coprococcus, ↑ Desulfovibrio |
| Sood 2019 | NR | NR | NR | NR |
| Sood 2020 | NR | NR | NR | NR |
| Uygun 2017 | NR | NR | NR | NR |
| Vaughn 2016 | Whole-genome shotgun sequencing | NR | ↑ α—diversity (Shannon) ↑ Bacteroides cellulosilyticus, ↑ Bilophila unclassified, ↑ Desulfovibrio piger, ↑ Bilophila wadsorthia, ↑ Clostridium leptum, ↑ Odoribacter splanchnicus, ↑ Bacteroides dorei, ↑ Parasutterella excrementihominis, ↑ Lachnospiraceae bacterium 7 1 58FAA, ↑ Eubacterium ventriosum, ↑ Burkholderiales bacterium 1 1 47, ↑ Dorea longicatena, ↑ Alistipes finegoldii ↓ Coprobacillus unclassified, ↓ Bacteroides massiliensis, ↓ Ruminococcus lactaris, ↓ Veillonella dispar, ↓ Lachnospiraceae bacterium 5 1 57FAA, ↓ Bifidobacterium adolescentis, ↓ Bacteroides vulgatus, ↓ Bacteroides ovatus, ↓ Streptococcus parasanguinis, ↓ Streptococcus salivarius, ↓ Clostridium scindens | Change in β—diversity (Bray-Curtis) towards donor in responders |
| Vermeire 2016 | 16 s DNA | ↑ α—diversity (OTUs) | ↑ α—diversity (OTUs), ↑ Roseburia, Oscillibacter, ↑ unclassified Lachnospiraceae, ↑ unclassified Ruminococcaceae | NR |
| Wang 2020 | NR | NR | NR | NR |
| Wei 2015 | NR | NR | NR | NR |
| Yang 2019 | 16 s RNA | NR | ↑ α—diversity (OTUs, Shannon) | NR |
| Zhang 2016 | NR | NR | NR | NR |
| Study | FMT or Antibiotic Treatment Delivery and Frequency | Patients (n) | Adverse Events Per Patient | Action |
|---|---|---|---|---|
| Borody 2003 | Daily enema for 5 days | 6 | NR | NR |
| Chen 2020 | Naso-jejunal 3 doses at 1, 3 and 5 days | 9 | Mild bloating (n = 3) Treatment failure (n = 1) | Colectomy (n = 1) |
| Chen 2020 | Colonoscopy ×3 in 1 week | 44 | NR | NR |
| Cold 2019 | 25 oral capsules per day for 50 days | 7 | No adverse events | No adverse events |
| Costello 2019 | Single donor FMT (colonoscopy and 2 enemas over a week) | 38 | After 8 weeks: Worsening colitis (n = 1) C. difficile infection (n = 1) Pneumonia (n = 1) New anemia (n = 1) Mild elevation of alkaline phosphatase (n = 2) and alanine aminotransferase (n = 1) | Colectomy (n = 1) |
| Single autologous FMT (colonoscopy and 2 enemas over a week) | 35 | After 8 weeks: Worsening colitis (n = 2) New anemia (n = 2) Mild elevation of alanine aminotransferase (n = 3) | NR | |
| 61 | After 12 months: Worsening colitis (n = 13) Infections (n = 8) New psoriatic arthritis (n = 2) Entero-pathic arthritis (n = 1) Crohn’s disease (n = 1) Allergy to infliximab (n = 1) Weight gain (n = 13) Weight loss (n = 8) | Colectomy (n = 9) | ||
| Cui 2015 | Single gastroscopy | 30 | Fever (n = 2)—1–6 h after FMT Increased diarrhea (n = 7)—1–6 h after FMT | NR |
| Damman 2015 | Single colonoscopy | 7 | Abdominal cramping, increase in stool output (NR)—immediately after FMT Abdominal pain (n = 1)—after 5 days | None |
| Ishikawa 2017 | Single colonoscopy | 21 | Transient borborygmus (n = 10)—during or soon after FMT | Resolved after end of treatment (n = 10) |
| Antibiotic pre-treatment only | 20 | Nausea and watery diarrhea—after antibiotic treatment (n = 8) | Discontinued antibiotic treatment (n = 3) | |
| Jacob 2017 | Single colonoscopy | 20 | Fever (n = 1) Chills (n = 1) Fatigue/malaise (n = 4) Abdominal pain (n = 3) Anorexia (n = 1) Diarrhea (n = 2) Constipation (n = 1) Transient febrile response (n = 1) Increase in Mayo score (n = 2)—at week 4 | Conservative care Anti-TNF alpha blockade therapy or colectomy |
| Kump 2017 | Colonoscopy (5 times, 14 days apart) | 17 | Worsening colitis (n = 1)—after day 3 | Required additional therapy (n = 1) |
| Antibiotic pre-treatment only | 10 | C. difficile infection (n = 3)—after 14 days Antibiotic-associated diarrhea (n = 1) Worsening colitis (n = 1) | Required additional therapy (n = 5) | |
| Mizuno 2017 | Single colonoscopy | 10 | Worsening colitis (n = 6) | |
| Moayyedi 2015 | Enema (once per week for 6 weeks) | 38 | Patchy inflammation and rectal abscess (n = 2) Abdominal discomfort (n = 1) C. difficile infection (n = 1)—after end of study | Antibiotic therapy (n = 2) |
| Placebo | 37 | Worsening colitis (n = 1) Patchy inflammation and rectal abscess (n = 1) | Colectomy (n = 1) Antibiotic therapy (n = 1) | |
| Nishida 2017 | Single colonoscopy | 41 | No adverse events | |
| Okahara 2020 | Single colonoscopy | 55 | Nausea (n = 20) | None |
| Paramsothy 2017 | Colonoscopy and enema (×5 per week for 8 weeks) | 41 | Infection-related adverse event (n = 10) Serious adverse event (n = 2) Abdominal pain (n = 12) Colitis (n = 10) Flatulence (n = 10) Bloating (n = 8) Upper respiratory tract infection (n = 7) Headache (n = 4) Dizziness (n = 3) Fever (n = 3) Rash (n = 3) | Colectomy (n = 1), intravenous corticosteroid therapy (n = 1) |
| Placebo | 40 | Infection-related adverse event (n = 14) Serious adverse event (n = 1) Abdominal pain (n = 11) Colitis (n = 9) Flatulence (n = 8) Bloating (n = 11) Upper respiratory tract infection (n = 6) Headache (n = 2) Dizziness (n = 3) Fever (n = 2) | Hospitalization (n = 1) | |
| Rossen 2015 | Donor FMT by nasoduodenal tube (twice, 3 weeks apart) | 23 | Discomfort with tube placement (n = 1) Fever (n = 2) Nausea (n = 2) Diarhea (n = 5) Headache (n = 1) Vomited fecal infusion (n = 2) Vomiting (n = 1) Abdominal pain (n = 1) Transient borborygmus (n = 4) Mild constipation (n = 1) | |
| Autologous FMT by nasoduodenal tube (twice, 3 weeks apart) | 25 | Discomfort with tube placement (n = 1) Nausea (n = 1) Malaise (n = 1) Diarrhea (n = 1) Headache (n = 1) Abdominal cramps (n = 6) Abdominal pain (n = 4) Transient borborygmus (n = 8) Dizziness (n = 1) Cytomegalovirus infection (n = 1)—7 weeks after the first FMT; unrelated to treatment | Ganciclovir (n = 1) | |
| 50 | Severe small bowel Crohn’s disease (n = 1) Abdominal pain (n = 1)—after 11 weeks Cervix carcinoma (n = 1)—after 6 weeks; unrelated to treatment | Antibiotics (n = 1) | ||
| Schierova 2020 | Enema 5× for first week then weekly × 6 weeks | 8 | No adverse events | None |
| Sokol 2020 | Single colonoscopy | 8 | Gastroenteritis (n = 2) Transient asthenia (n = 1) Cutaneous abscess (n = 1) | Self-limiting |
| Sood 2019 | Colonoscopy at 0, 2, 6, 10, 14, 18, 22 weeks | 41 | After FMT, at 0 weeks: Abdominal discomfort (n = 26) Abdominal distension (n = 14) Fever (n = 4) Worsening diarrhea (n = 4) Flatulence (n = 2) Fatigue (n = 2) | Symptoms were self-limiting Oral rehydration solution (n = 4) |
| Sood 2020 | Colonoscopy at 0, 2, 6, 10, 14, 18, 22 weeks | 93 | Abdominal discomfort (n = 28) Flatulence (n = 12) Borborygmi (n = 10) Low grade fever (n = 8) Diarrhea (n = 7) | Self-limiting |
| Uygun 2017 | Single colonoscopy | 30 | Nausea, vomiting, abdominal pain, diarrhea (n = 7) | NR |
| Vaughn 2016 | Single colonoscopy | 19 | Hives (n = 1) | Oral steroids (n = 1) |
| Vermeire 2016 | Naso-jejunal or rectal tube (twice one day, then the following day) | 14 | High fever (n = 4)—few hours after FMT Vomited and pneumonia (n = 1)—after FMT | Paracetamol (n = 4) Broad-spectrum antibiotics (n = 1) |
| Wang 2020 | Colonoscopy ×3; 2–3 month intervals | 16 | None | None |
| Wei 2015 | Single colonoscopy or naso-jejunal tube | 14 | Intolerance with FMT (n = 1) Moderate fever (n = 2)—after FMT | Self-limiting |
| Yang 2019 | Gastroscopy or colonoscopy (twice, one week apart) | 31 | Nausea (n = 1) Reflux (n = 4) Belching (n = 2) Diarrhea (n = 10) Constipation (n = 1) Fever (n = 2) Aggravation of abdominal pain (n = 5) Abdominal distension (n = 3) | NR |
| Zhang 2016 | Single endoscopy | 19 | Transient increased diarrhea (n = 7) Mild skin pruritus (n = 1) Borborygmus (n = 2) | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mocanu, V.; Rajaruban, S.; Dang, J.; Kung, J.Y.; Deehan, E.C.; Madsen, K.L. Repeated Fecal Microbial Transplantations and Antibiotic Pre-Treatment Are Linked to Improved Clinical Response and Remission in Inflammatory Bowel Disease: A Systematic Review and Pooled Proportion Meta-Analysis. J. Clin. Med. 2021, 10, 959. https://doi.org/10.3390/jcm10050959
Mocanu V, Rajaruban S, Dang J, Kung JY, Deehan EC, Madsen KL. Repeated Fecal Microbial Transplantations and Antibiotic Pre-Treatment Are Linked to Improved Clinical Response and Remission in Inflammatory Bowel Disease: A Systematic Review and Pooled Proportion Meta-Analysis. Journal of Clinical Medicine. 2021; 10(5):959. https://doi.org/10.3390/jcm10050959
Chicago/Turabian StyleMocanu, Valentin, Sabitha Rajaruban, Jerry Dang, Janice Y. Kung, Edward C. Deehan, and Karen L. Madsen. 2021. "Repeated Fecal Microbial Transplantations and Antibiotic Pre-Treatment Are Linked to Improved Clinical Response and Remission in Inflammatory Bowel Disease: A Systematic Review and Pooled Proportion Meta-Analysis" Journal of Clinical Medicine 10, no. 5: 959. https://doi.org/10.3390/jcm10050959







