Abstract
Shigellosis remains one of the leading causes of morbidity and mortality worldwide and is the second leading cause of diarrheal mortality among all age groups. However, the global emergence of antimicrobial-resistant Shigella strains, limiting the choice of effective drugs for shigellosis, has become the major challenge in the treatment of Shigella infections. The aim of this systematic review and meta-analysis was to provide an updated picture of the prevalence of antimicrobial-resistant Shigella species in Asia. A comprehensive and systematic search was performed on three electronic databases (PubMed, ScienceDirect and Scopus), in which 63 eligible studies published between 2010 and 2022 were identified. From our meta-analysis of proportions using a random-effects model, the overall prevalence of Shigella spp. in Asian patients was estimated to be 8.0% (95% CI: 5.5–10.5). The pooled prevalence rates of multidrug-resistant (MDR) and extended-spectrum beta-lactamase (ESBL)-producing Shigella strains were 68.7% (95% CI: 59.9–77.5) and 23.9% (95% CI: 12.9–34.8), respectively. Concerning recommended antimicrobial drugs for Shigella, the prevalence of resistance was highest for ciprofloxacin (29.8%) and azithromycin (29.2%), followed by ceftriaxone (23.8%), in spite of their importance as first- and second-line treatments for shigellosis. In contrast, resistance to carbapenems, such as ertapenem (0.0%), imipenem (0.1%) and meropenem (0.0%), was almost non-existent among the 49 tested antibiotics. The significantly high prevalence estimation suggests that the multidrug-resistant Shigella is a pressing threat to public health worthy of careful and justified interventions. Effective antibiotic treatment strategies, which may lead to better outcomes for the control and treatment of shigellosis in Asia, are essential.
1. Introduction
The Shigella species is one the most prevalent diarrheal pathogens in Asia, particularly in South Asia [1], and is responsible for shigellosis, an intestinal infection that is also known as bacillary dysentery. Among the four Shigella spp.—Shigella dysenteriae (12 serotypes), Shigella boydii (23 serotypes), Shigella flexneri (12 serotypes) and Shigella sonnei (1 serotype)—more than 90% of global shigellosis cases are caused by S. flexneri and S. sonnei [2]. The current global epidemiology studies demonstrate that S. sonnei is increasingly overtaking S. flexneri to become the predominant species in some parts of the world, including Asia [3,4,5]. Shigellosis is characterized by the invasion and disruption of the epithelium lining the terminal ileum, colon and rectum, resulting in acute watery diarrhea and dysentery, with frequent bloody stools, fever and abdominal cramps [6]. The global burden of shigellosis is estimated to be 125 million episodes annually, with more than 200,000 deaths among all age groups [7]. The World Health Organization (WHO) recommended ciprofloxacin as the first-line antimicrobial therapy for all shigellosis patients with bloody diarrhea, irrespective of age. Pivmecillinam and ceftriaxone were recommended as the second-line treatments in all age groups, while azithromycin was recommended as an alternative for treatment in adults [8]. However, overuse or misuse of antibiotics in treating shigellosis increases antibiotic resistance, further limiting therapeutic options for Shigella infections.
Antimicrobial-resistant Shigella strains have become a major global concern and a significant threat to public health. Nowadays, the reliance on ciprofloxacin for shigellosis treatment has markedly prompted the spread of fluoroquinolone-resistant S. sonnei worldwide, which most likely evolved from a single common progenitor in South Asia around 2007 [9,10]. This single clonal expansion event was associated with classical sequential mutations in the quinolone resistance determining region; specifically, in gyrA codon 83 or 87 and parC codon 80 [9]. Back in the 1980s, a high proportion of Southeast Asian S. sonnei and S. flexneri strains were found to exhibit resistance to sulfonamides, tetracyclines and streptomycin due to the acquisition of plasmids harboring the sul2, tetra and strAB genes [10]. A surveillance study later reported that the majority of Shigella spp. isolated from Asian countries in the early 2000s were resistant to ampicillin and co-trimoxazole, among which S. flexneri and S. sonnei were found to be highly associated with ampicillin and co-trimoxazole resistance, respectively [11]. Subsequently, in the mid-2000s to early 2010s, horizontal transfer of the pKSR100 plasmid encoding for resistance to azithromycin from S. flexneri into other Shigella spp. has intensified the emergence of novel variants. For instance, the recent dominance of dual ciprofloxacin and azithromycin resistance in S. sonnei was reported in the late 2010s [12].
Apart from the overwhelming number of particular drug resistance strains, the emergence of multidrug resistance (MDR) is a burden in the treatment of shigellosis, and nearly half of Shigella strains around the world have now become resistant to multiple antibiotics [13]. Most of the MDR Shigella strains confer resistance to third-generation cephalosporins, such as ceftriaxone and cefoperazone, azithromycin and fluoroquinolones, commonly via point mutations and horizontal transfer of plasmid-mediated resistance mechanisms [3,13]. Third-generation cephalosporin resistance has also been linked to the production of extended-spectrum beta-lactamases (ESBLs), which confer resistance to the CTX-M, TEM and SHV-lactamase families [4]. The rising prevalence of ESBL-producing Shigella spp. in Asian countries [14,15] signals a wake-up call, requiring new approaches and alternative therapies in the management of shigellosis. Hence, updates on the antimicrobial resistance in Shigella spp. are necessary for effective and justified treatments to reduce the morbidity and mortality rates associated with shigellosis. The aim of this systematic review and meta-analysis (SRMA) was to provide a current and comprehensive pooled prevalence estimation of MDR- and ESBL-producing Shigella spp. in Asia from more than a decade of published empirical data.
2. Results
2.1. Study Selection and Characteristics of the Included Studies
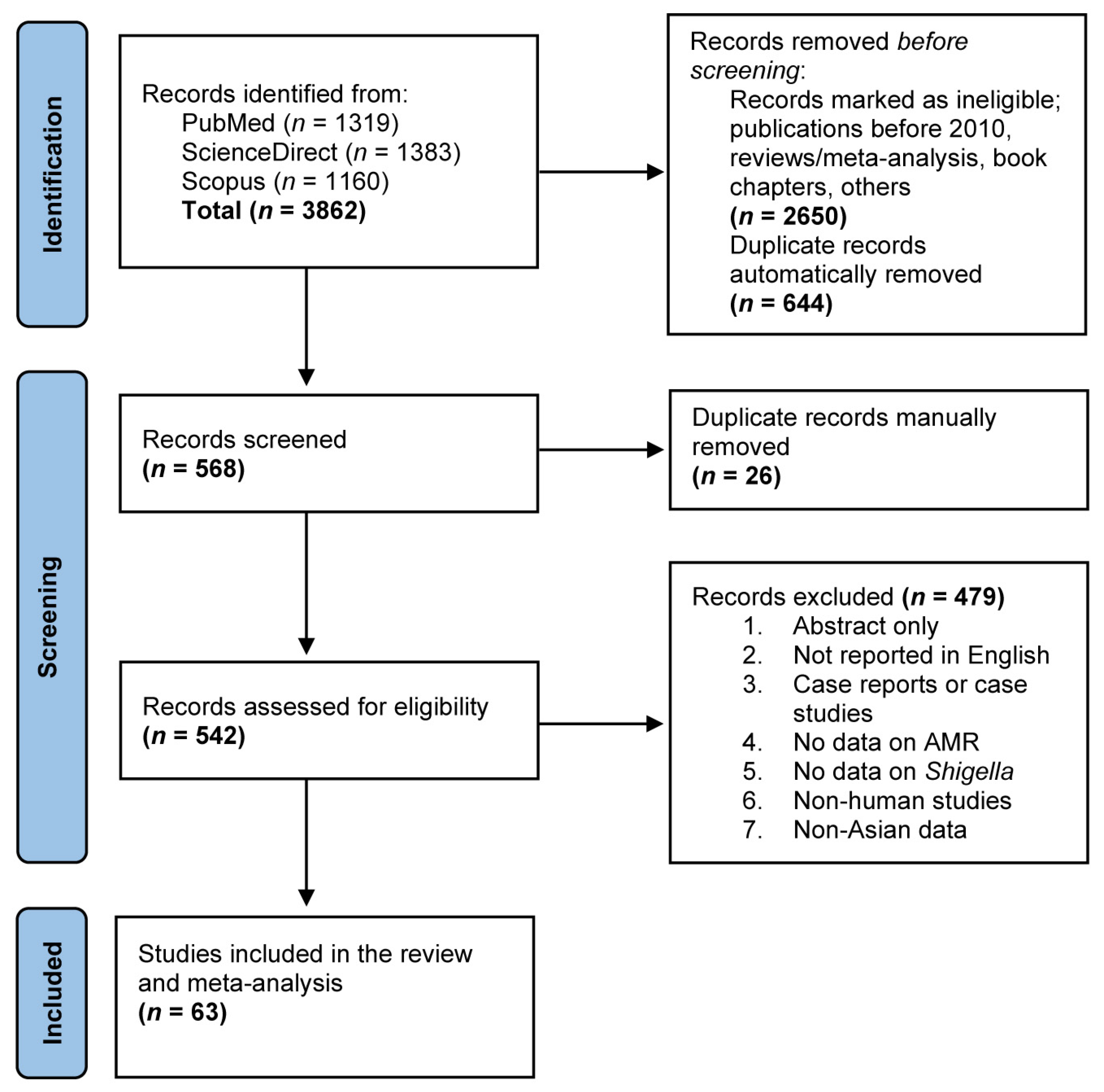
Figure 1 shows a Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) flow diagram of our study selection process, as well as results from the literature search. Our online search of three different web databases—namely, PubMed, ScienceDirect and Scopus—returned a total of 3862 records. Following the initial pre-screening eligibility check and duplicate removal, 568 records were screened for their titles and abstracts, of which, after rounds of manual assessment to exclude 26 records, a total of 542 records were retained for full-text evaluation, where they were assessed based on the predetermined exclusion criteria. A total of 479 records were removed and, subsequently, a total of 63 articles were found to be eligible for this study and, thus, were included in the qualitative study and meta-analysis.
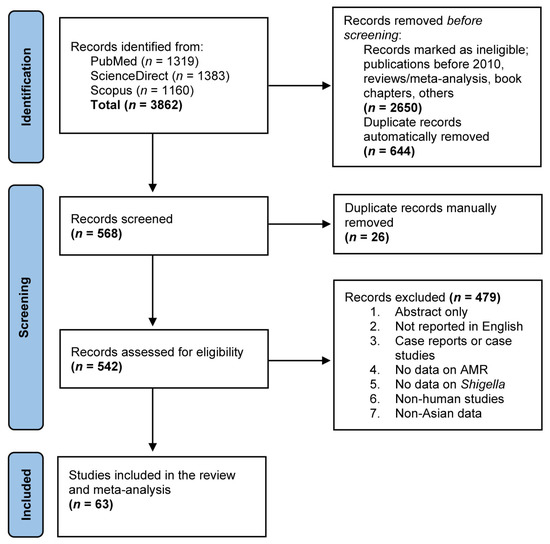
Figure 1.
A PRISMA flow diagram of the study selection process and literature search results. Three different web databases (PubMed, ScienceDirect and Scopus) were used to search eligible studies reporting antimicrobial-resistant Shigella spp. using predefined search strategies. A total of 3862 records were retrieved and duplicates were removed using EndNote 20 software, following which they were screened against predefined inclusion criteria before inclusion in the qualitative study and meta-analysis.
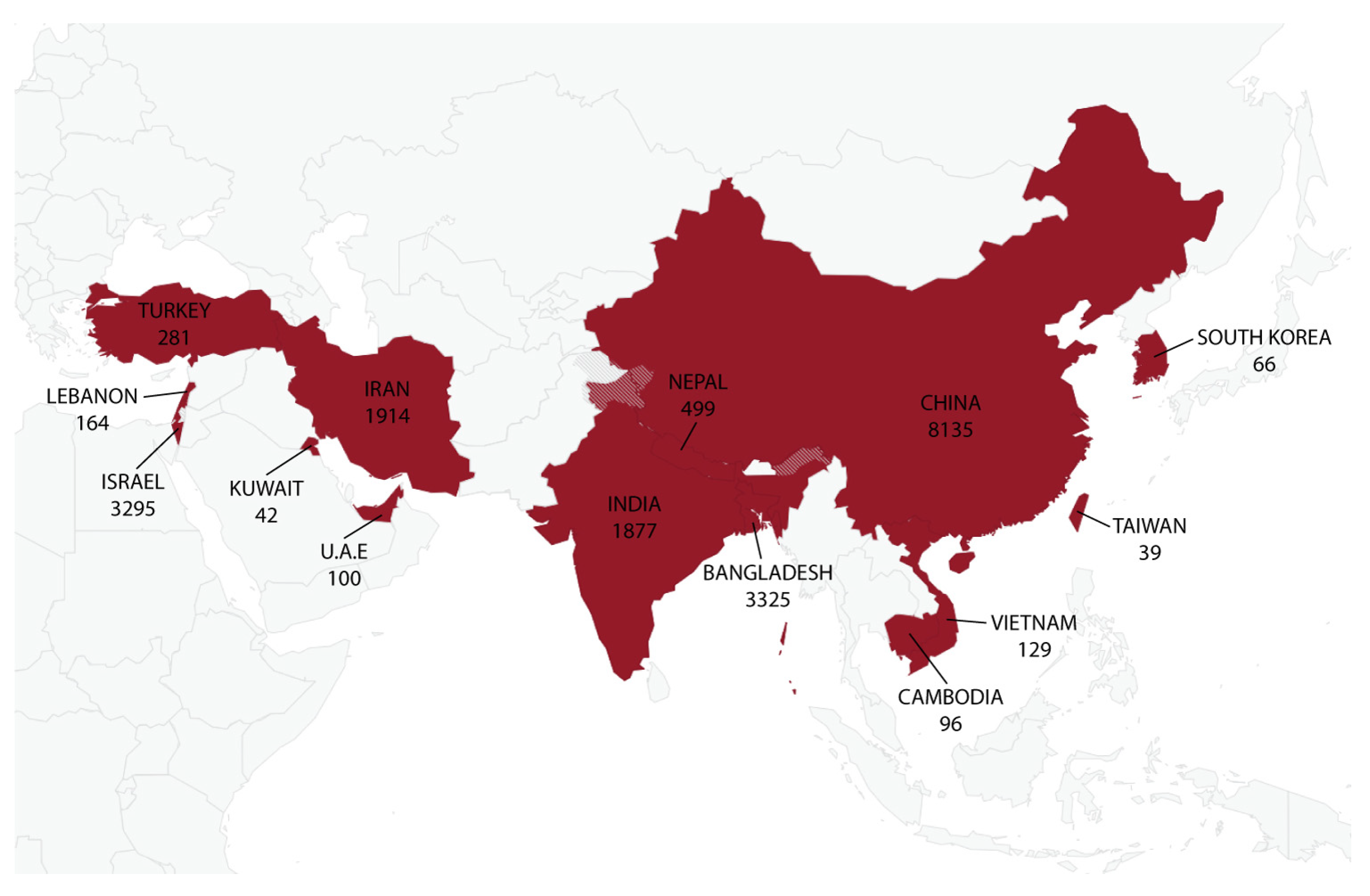
The summarized characteristics of the 63 included articles are shown in Table 1. Studies were conducted in 14 Asian countries, and China represented the country with the most cases of Shigella infections (n = 8135), as reported in 15 different studies (Figure 2). The country with the second highest number of cases involving Shigella spp. was Bangladesh, which recorded 3325 cases in only three studies. Although Iran was represented by 15 studies, the country recorded only 1914 cases involving Shigella spp., whereas Israel recorded 3295 cases of Shigella infections in just a single study. Overall, a total of 19,962 cases from the Asian region involving Shigella spp. and employing antimicrobial susceptibility testing using 49 different antibiotics were identified (Table 1). All 63 included studies were cross-sectional, and they were carried out between 2000 and 2020 and published between 2010 and 2022. Shigella isolates were collected mostly from pediatric groups and characterized using polymerase chain reactions (PCRs) or slide agglutination tests (SATs), whereas double-disk synergy tests (DDSTs) were utilized to detect the presence of ESBL genes. Biochemical tests, such as the Kirby–Bauer method (disk diffusion method), broth microdilution, agar dilution and concentration gradient method (E-test), were used in the antibiotic susceptibility tests against the Shigella spp. Out of the 63 selected studies, 39 provided data on MDR, whereas only 16 studies provided data on ESBL-producing Shigella spp.

Table 1.
Major characteristics of the 63 studies included in this systematic review and meta-analysis (SRMA).
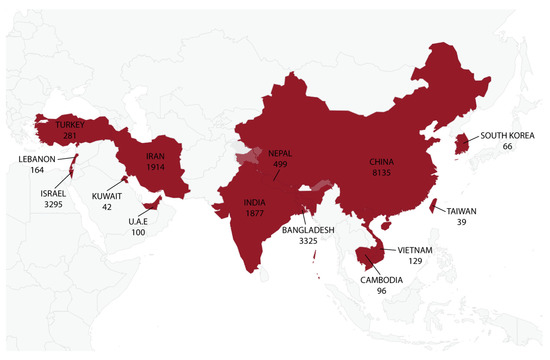
Figure 2.
Distribution of Shigella spp. across 14 Asian regions from 63 studies. A total of 19,962 cases from the continent were identified involving Shigella spp., and China represented the country with the most cases of Shigella infections (n = 8135), as reported in 15 different studies, whereas the lowest number of cases involving Shigella spp. was recorded for Kuwait (n = 42) in a single study.
2.2. Prevalence of Shigella species in Asia
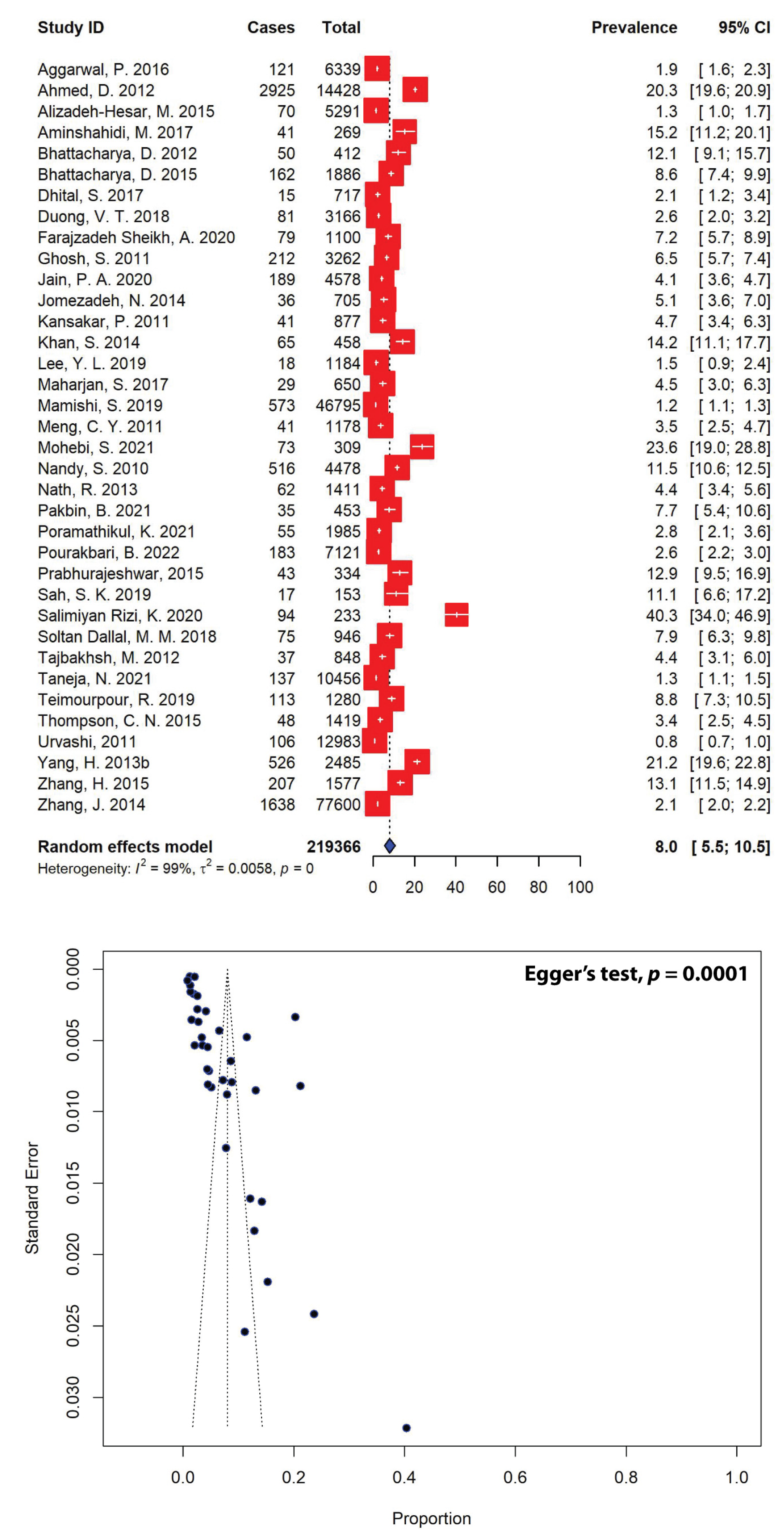
The pooled prevalence of Shigella spp. in Asian patients from 36 studies was estimated to be 8.0% (95% CI: 5.5–10.5) (Figure 3), and the lowest prevalence of Shigella spp. in Asia was 0.8%, whereas the highest prevalence of Shigella spp. was 40.3%. Of the 63 selected studies, 27 were excluded due to the absence of data on the sample population (Table 1). The asymmetrical distribution of effect estimates, which is shown by a funnel plot of the study distribution (Figure 3), prompted us to examine further the data according to subgroups. When stratified according to the regions where the studies were conducted, the highest estimated Shigella spp. prevalence was 10.2% (95% CI: 4.0–16.3), calculated from 12 different studies in the Middle East, followed by the estimates for East Asia (9.5%, 95% CI: 0.2–18.7) and South Asia (7.4%, 95% CI: 4.7–10.2). In contrast, the lowest Shigella spp. prevalence of 2.9% (95% CI: 2.5–3.3) was recorded in Southeast Asia. When stratified according to countries, Bangladesh (20.3%, 95% CI: 19.6–20.9), China (12.1%, 95% CI: 1.3–23.0) and Iran (10.2%, 95% CI: 4.0–16.3) represented the three highest Shigella spp. prevalence estimates, while Taiwan recorded the lowest Shigella spp. prevalence in Asia (1.5%, 95% CI: 0.9–2.4) (Table 2).
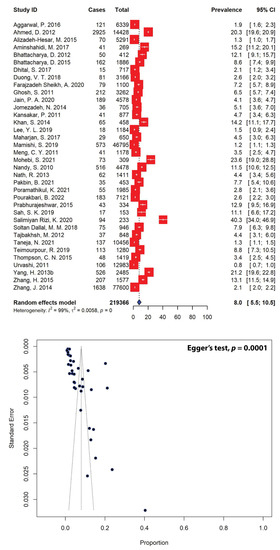
Figure 3.
Forest and funnel plots representing the pooled Asian prevalence of Shigella spp. from 36 studies. The estimates of pooled prevalence were calculated using the random-effects model (top panel). The distribution of effect estimates is represented by a funnel plot (bottom panel). Figures were generated using RStudio software.

Table 2.
Pooled prevalence of Shigella in different Asian regions.
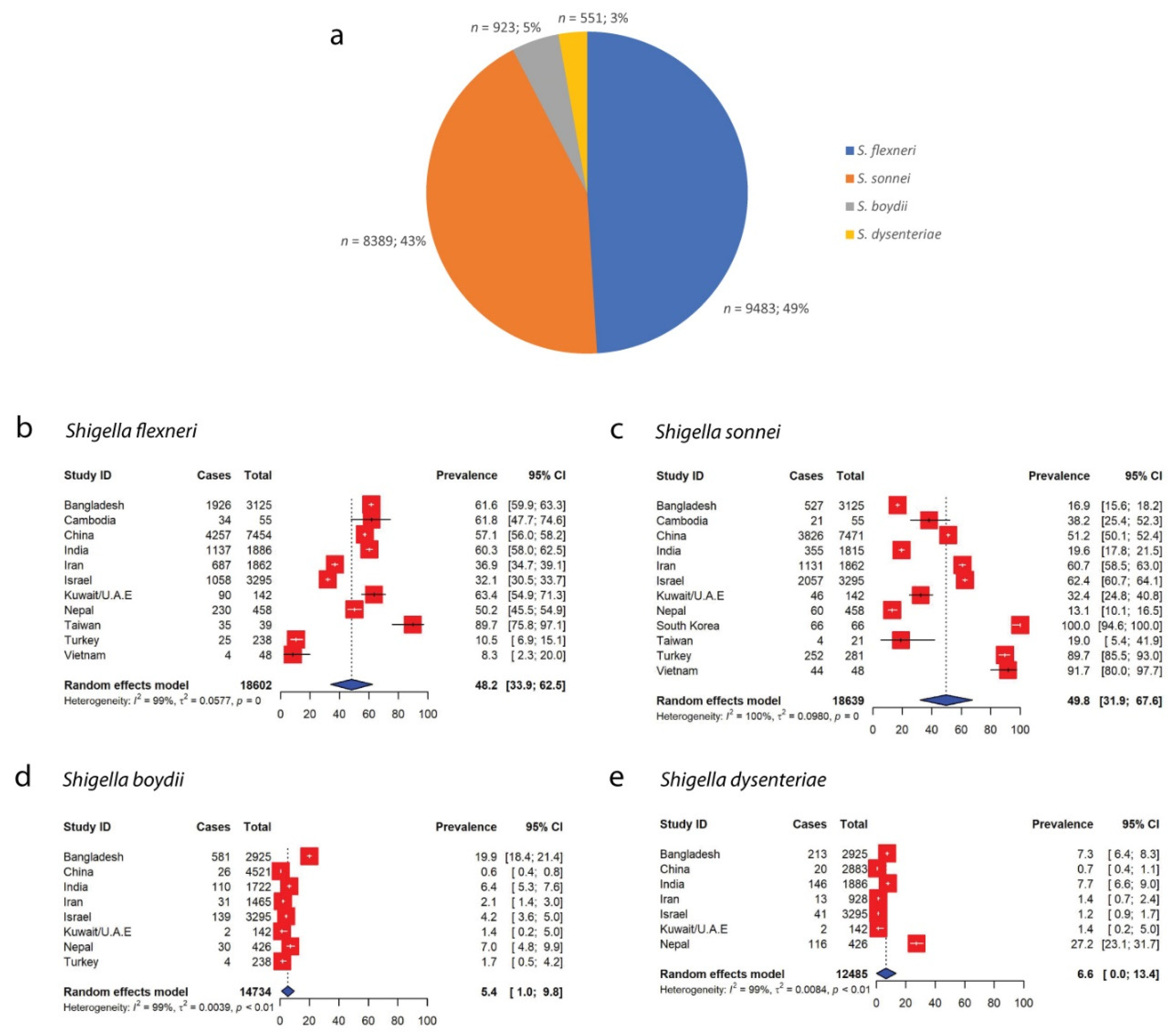
When grouped according to the species of Shigella, the highest numbers of isolates recorded were for S. flexneri, with this species accounting for 49% (n = 9483) of the 19,346 total cases of Shigella infections that were reported in Asia, followed by S. sonnei (43%, n = 8389), S. boydii (5%, n = 923) and S. dysenteriae (3%, n = 551) (Figure 4a). Overall, 19,346 out of 19,962 total Shigella isolates were characterized according to their species, whereas five studies [22,23,27,39,45] did not report the species of Shigella isolates (Table 1). Although S. flexneri accounted for the largest proportion of Shigella isolates reported in the included studies, the pooled prevalence of S. sonnei was the highest in Asia at 49.8% (95% CI: 31.9–67.6), followed by those of S. flexneri (48.2%, 95% CI: 33.9–62.5), S. dysenteriae (6.6%, 95% CI: 0.0–13.4) and S. boydii (5.4%, 95% CI: 1.0–9.8) (Figure 4). China reported the highest number of cases with S. flexneri (n = 4257) and S. sonnei (n = 3826), while the highest cases of S. boydii (n = 581) and S. dysenteriae (n = 213) infections were recorded in Bangladesh between 2005 and 2008 [16].
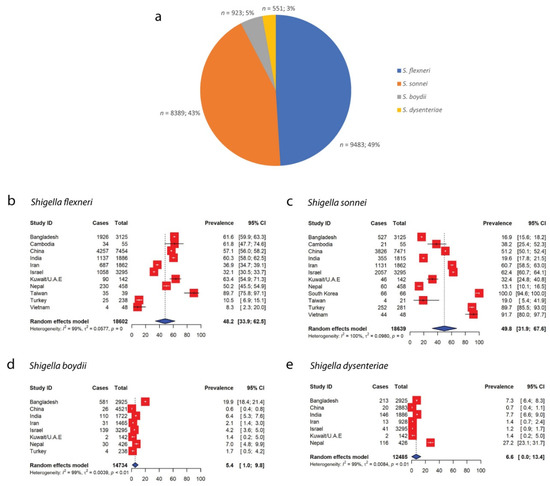
Figure 4.
Distribution of different species of Shigella across Asian countries. (a) Proportions of different species of Shigella. S. flexneri constituted the highest proportion of Shigella isolates in Asia (49%, n = 9483), followed by S. sonnei (43%), S. boydii (5%) and S. dysenteriae (3%). (b) Forest plot representing the pooled Asian prevalence of S. flexneri. (c) Forest plot representing the pooled Asian prevalence of S. sonnei. (d) Forest plot representing the pooled Asian prevalence of S. boydii. (e) Forest plot representing the pooled Asian prevalence of S. dysenteriae.
2.3. Antimicrobial Resistance Patterns of Shigella spp.
The antimicrobial susceptibility levels of the Shigella isolates from the 63 included studies were tested against various antibiotics (Table 1). The pooled prevalence estimates of the resistant Shigella isolates tested against 49 different antibiotics are presented in Table 3. The antibiotics were classified into 13 groups, with the penicillin group of antibiotics being the most commonly used; in particular, ampicillin was identified as the most commonly used antibiotic (57 studies), while ceftiofur, oxacillin and fosfomycin were the least frequently tested against Shigella spp. (one study each) [54,55,64]. Our meta-analysis revealed that resistant Shigella strains exist for the majority of the antibiotics tested, although they are varied (Table 3). The antimicrobial drug resistance patterns for Shigella spp. in Asia revealed that 98.4% (95% CI: 96.9–100.0) of the isolates were resistant to streptomycin, followed by trimethoprim (95.5%, 95% CI:92.3–98.8) and ticarcillin (90.5%, 95% CI: 84.7–96.3), whereas antimicrobials belonging to the carbapenem class of antibiotics, such as ertapenem (0.0%, 95% CI: 0.0–1.1), meropenem (0.0%, 95% CI: 0.0–0.7) and imipenem (0.1%, 95% CI: 0.0–0.2), had the lowest Shigella resistance rate, in addition to nitrofurantoin (0.0%, 95% CI: 0.0–0.2).

Table 3.
Pooled prevalence estimates of antimicrobial drug-resistant Shigella spp. tested against 49 antibiotics in Asia.
2.4. Prevalence of Multidrug-Resistant Shigella spp. in Asia
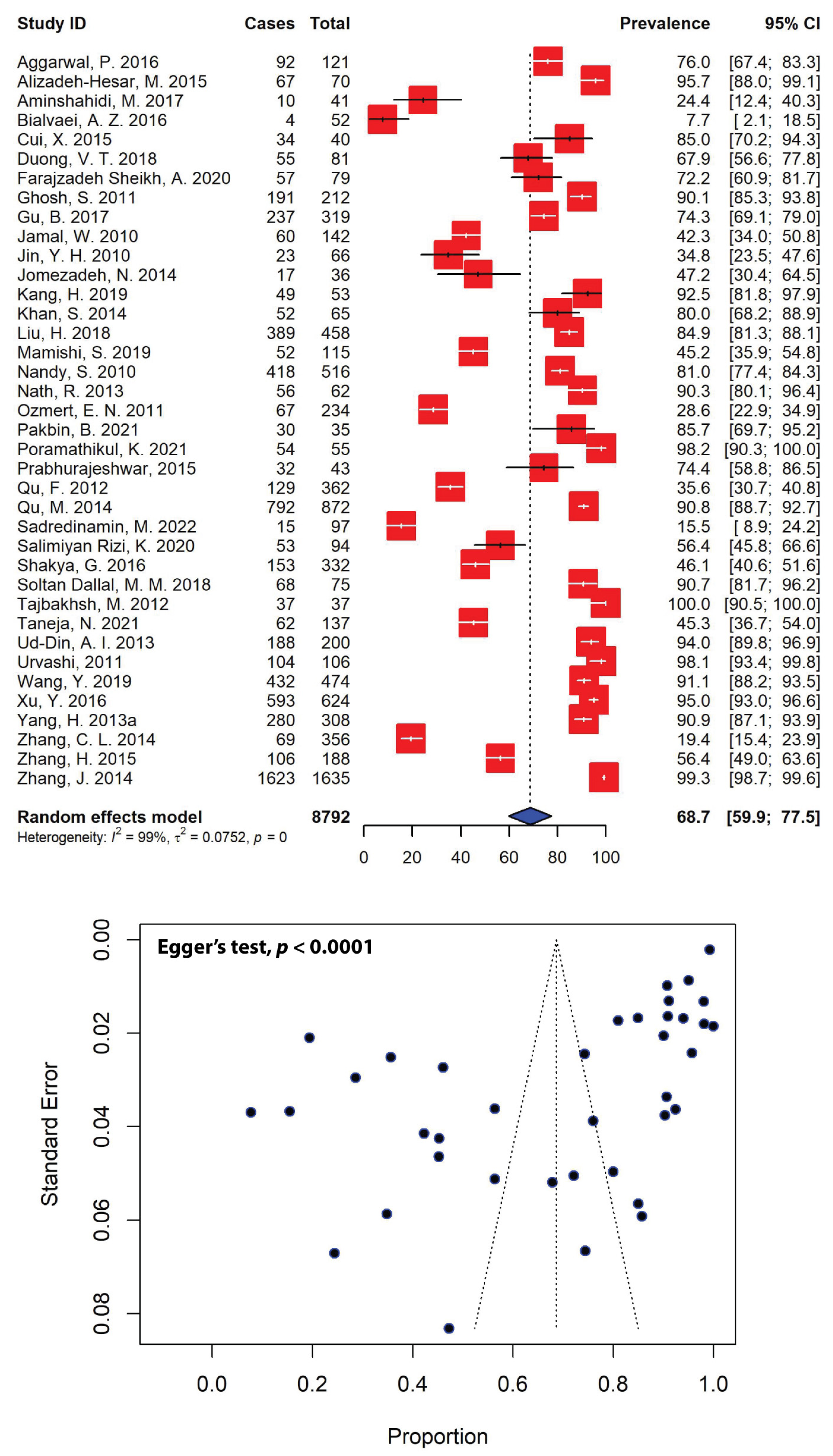
The estimated prevalence of MDR Shigella spp. was significantly high across different studies, with the majority (26 out of the 38 included studies) reporting multiple resistance rates higher than 50.0% for total Shigella isolates (Figure 5). Our meta-analysis revealed that the pooled prevalence of MDR Shigella spp. in Asia was estimated to be 68.7% (95% CI: 59.9–77.5), with evidence of significant heterogeneity (I2 = 99%, τ2 = 0.0752, p = 0). The highest and lowest prevalence rates of MDR Shigella spp. were estimated to be 100.0% and 7.7%, respectively. The presence of publication bias, represented by an asymmetrical funnel plot (Figure 5), was statistically confirmed with Egger’s test (p < 0.0001).

Figure 5.
Forest and funnel plots representing the pooled prevalence of multidrug-resistant Shigella spp. in Asia. The estimate of prevalence was calculated by pooling 38 selected studies using the random-effects model (top panel). The distribution of effect estimates is shown by a funnel plot (bottom panel). Figures were generated using RStudio software.
When stratified according to different Asian regions, the highest prevalence of MDR Shigella spp. was estimated to be 83.4% (95% CI: 53.8–100.0), which was calculated for Southeast Asia, followed by the estimates for South Asia (77.7%, 95% CI: 66.1–89.3) and East Asia (73.2%, 95% CI: 58.5–87.9). Conversely, the lowest MDR Shigella spp. prevalence of 54.3% (95% CI: 38.1–70.4) was reported in the Middle East (Table 4). Overall, a total of 6750 MDR Shigella isolates were recorded over a 20-year period (2000–2020). Our meta-analysis revealed that the highest MDR Shigella spp. prevalence levels were recorded in Cambodia (98.2%, 95% CI: 90.3–100.0), followed by Bangladesh (94.0%, 95% CI: 89.8–96.9) and India (79.6%, 95% CI: 66.8–92.4), while the lowest prevalence was recorded in Turkey (28.6%, 95% CI: 22.9–34.9). As well as having a Shigella spp. prevalence of 76.3% (95% CI: 61.8–90.8), China recorded the highest number of MDR Shigella isolates in Asia (n = 4733) (Table 4).

Table 4.
Pooled prevalence of multidrug-resistant Shigella in different Asian regions.
2.5. Patterns of Extended-Spectrum β-Lactamase-Producing Shigella spp.
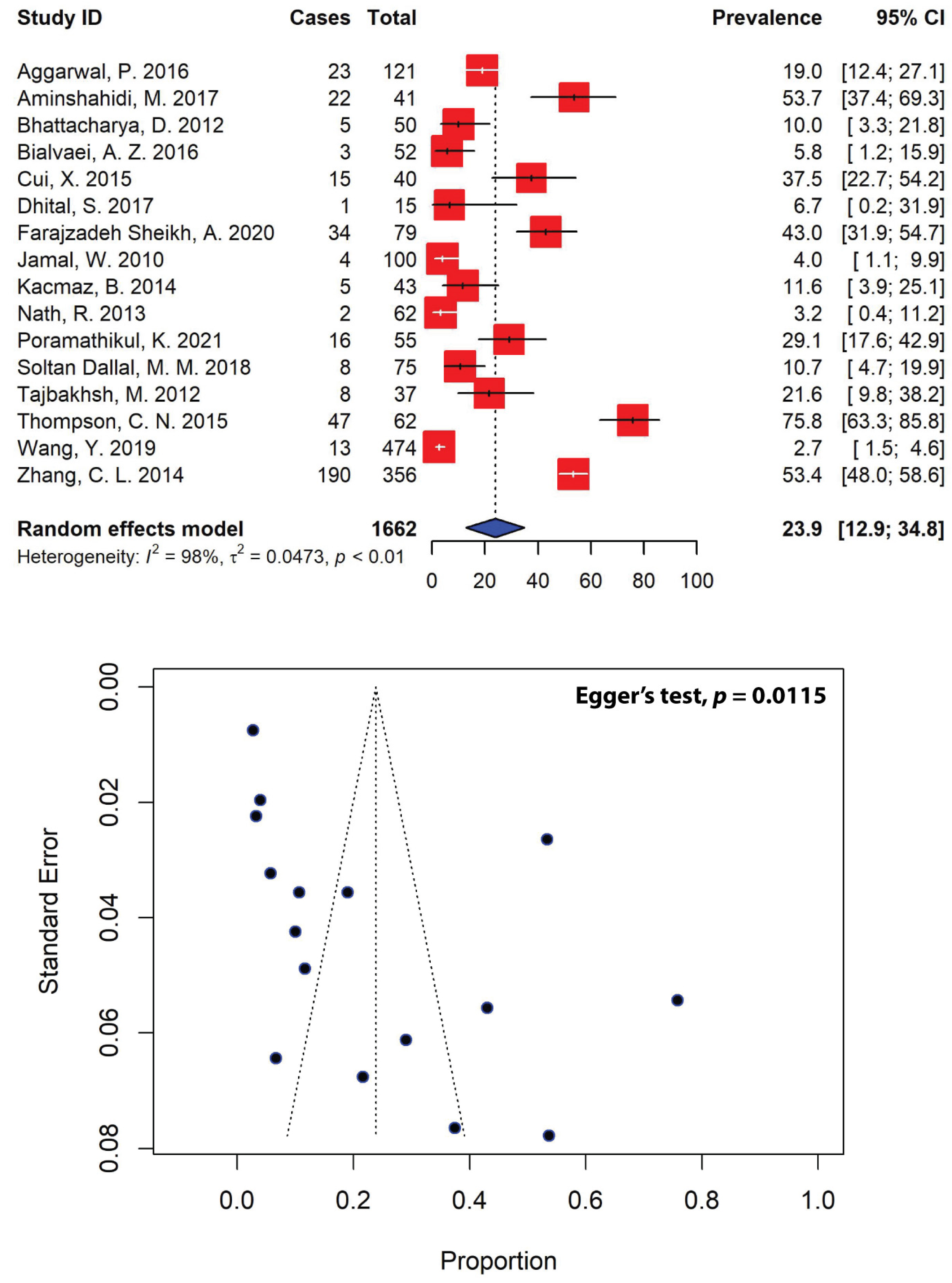
Of the 63 eligible studies analyzed in our meta-analysis, only 16 studies, with a total sample size of 396, reported ESBL-producing Shigella spp. in Asian patients (Figure 6). The overall pooled prevalence of ESBL-producing Shigella spp. in Asia was estimated to be 23.9% (95% CI: 12.9–34.8), with substantial heterogeneity between studies (I2 = 98%, τ2 = 0.0473, p < 0.01). Our meta-analysis revealed that the highest prevalence of ESBL-producing Shigella spp. was recorded in a study conducted in Vietnam (75.8%, 95% CI: 63.3–85.8) [67], whereas the lowest prevalence of ESBL-producing Shigella spp. was recorded in the U.A.E (4.0%, 95% CI: 1.1–9.9) in a single study [33]. Regionally, Southeast Asia represented the highest prevalence of ESBL-producing Shigella spp., with a prevalence of 52.5% (95% CI: 6.8–98.3), followed by East Asia (31.0%, 95% CI: 0.9–61.0), the Middle East (20.7%, 95% CI: 6.7–34.7) and South Asia (9.7%, 95% CI: 2.3–17.1) (Table 5).
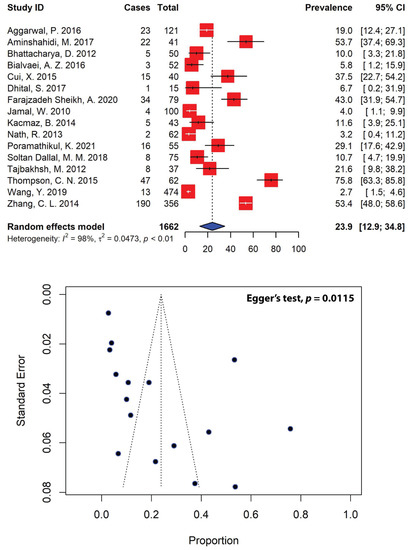
Figure 6.
Forest and funnel plots representing the pooled prevalence of extended-spectrum β-lactamase (ESBL)-producing Shigella spp. in Asia. The estimate of prevalence was calculated by pooling 16 studies using the random-effects model (top panel). The distribution of effect estimates is shown by a funnel plot (bottom panel). Figures were generated using RStudio software.

Table 5.
Pooled prevalence of ESBL-producing Shigella spp. in different Asian regions.
3. Discussion
Shigella continues to represent a significant cause of mortality and morbidity worldwide and has been found to be the second-leading cause of diarrheal mortality among all age groups, accounting for more than 200,000 deaths, including over 63,000 children under the age of 5 years [7]. In a recent global disease burden report, diarrhea was ranked third among the top ten causes of mortality in children younger than 9 years old [77]. Although diarrhea mortality has significantly decreased since 1990, morbidity rates from diarrheal diseases remain high, predominantly in low- and middle-income countries [7,78]. Similarly, shigellosis is a major cause of illness among children, travelers, expatriates and military personnel in low- and middle-income countries, where it is associated with persistent diarrhea of more than 14 days in these populations [7]. However, the global emergence of antimicrobial-resistant Shigella strains, which limit the choice of effective drugs for shigellosis, has become the main challenge in the treatment of Shigella infections. Moreover, changes in the prevalent Shigella serogroups/types and resistance patterns are creating a major burden for the choice of an appropriate therapy for shigellosis treatment. The distribution of Shigella serogroups/types has varied over different periods of time and in different geographical regions. For instance, S. flexneri serotypes such as 2a, 3a and 1a, are most prevalent in Asian countries such as China, India and Pakistan [79]. Even though S. sonnei is traditionally most commonly found in developed countries, the species is currently dominant and undergoing an unprecedented expansion across developing countries in Asia, Latin America and the Middle East [3]. A previous study reported that Shigella infections increase during the monsoon and summer seasons, when the humidity and temperature are high [65]. S. flexneri, together with S. sonnei, are responsible for more than 90% of the global shigellosis cases [2]. Although shigellosis is mostly a self-limiting disease, antibiotics are recommended to reduce deaths and disease progressions. However, in recent years, Shigella has been reported to be becoming resistant to many antimicrobial drugs, such as ampicillin, tetracyclines, chloramphenicol, ciprofloxacin, nalidixic acid and trimethoprim/sulfamethoxazole [79,80]. This phenomenon can be seen in many regions across the globe [2,28,29,30,81,82,83]. Therefore, determining the Shigella burden in a population is vital for the design of targeted therapeutic strategies to reduce the incidence of mortality and morbidity from shigellosis. This requires comprehensive data on the prevalence and patterns of drug-resistant Shigella spp. but, to the best of our knowledge, such data are unavailable to date in Asia.
Here, we present the prevalence of multidrug-resistant and extended-spectrum beta-lactamase-producing Shigella spp. in Asia. Our findings in this SRMA were calculated by combining all eligible data on the prevalence of antimicrobial-resistant Shigella spp. from community- and hospital-based studies, as reported in the 63 selected studies from Asia. Nevertheless, as expected from various studies with different backgrounds and settings, our findings were mostly heterogeneous. Significant heterogeneity was also observed in our previous study on the prevalence of multidrug-resistant diarrheagenic Escherichia coli (DEC) [84]. The heterogeneity in our findings was most likely due to the fact that different sample sizes, methodologies and research settings—such as study regions, time periods and population ages—were employed in different studies. This is expected, as our SRMA utilized a random-effects model, which presumes heterogeneity, as opposed to being a meta-analysis using a fixed-effects model [85]. Nonetheless, to the best of our knowledge, our SRMA is the first to evaluate the prevalence of antimicrobial-resistant Shigella spp. in Asia and, thus, it will hopefully be useful for designing targeted strategies in the treatment of shigellosis.
The pooled prevalence estimates revealed that 8.0% (95% CI: 5.5–10.5) of all Asian diarrheal cases in the last couple of decades were caused by Shigella (Figure 3), and the highest and lowest prevalence estimates for Shigella were 40.3% [60] and 0.8% [68], respectively. Iran had the highest number of studies reporting the prevalence of Shigella among those analyzed in our SRMA, accounting for 12 different studies (Table 2). The pooled prevalence of Shigella in Iran was estimated at 10.2% (95% CI: 4.0–16.3), slightly higher than the overall pooled estimate for Shigella in Asia. Nevertheless, the pooled prevalence of Shigella calculated in our SRMA was higher than the results of a similar comprehensive prevalence estimate from Iran, for which an estimate of 6.2% was calculated [86]. Differences in the pooled prevalence estimates between our study and the recently published study might be attributed to differences in the numbers of included studies, sample sizes, study settings and methods, publication years and many other factors. It is likely that the prevalence of Shigella calculated in this SRMA was higher due to the smaller number of included studies (n = 12), whereas there were 34 included studies, published in English and Persian from 2000 to 2020, in the previous comprehensive review [86]. The pooled prevalence estimate for Shigella in India was calculated at 6.2% (95% CI: 3.4–9.0)—lower than the overall prevalence of Shigella in Asia—from 10 included studies (Table 2), whereas a prevalence of 12.1% (95% CI: 1.3–23.0) was estimated for China from three included studies. It is important to note that, although China was represented by only three studies, the country had the largest sample population of 81,662, which constituted 37.2% of the total diarrheal Asian population analyzed in this meta-analysis. Taiwan had the smallest sample population of 1184, with the prevalence estimate for Shigella in the country being calculated at 1.5% (95% CI: 0.9–2.4) [41]—the lowest in Asia. In contrast, Bangladesh had the highest prevalence of Shigella in Asia, which as estimated at 20.3% (95% CI: 19.6–20.9) from a single study conducted in Dhaka [16].
Historically, S. flexneri is known to be commonly found in developing countries, while S. sonnei is traditionally most commonly isolated in developed countries. However, in recent years, S. sonnei has become increasingly dominant and can be found in many developing countries across the globe. Our pooled prevalence estimates revealed that S. sonnei had the highest prevalence of 49.8% (95% CI: 31.9–67.6) in Asia, followed by S. flexneri (48.2%, 95% CI: 33.9–62.5), S. dysenteriae (6.6%, 95% CI: 0.0–13.4) and S. boydii (5.4%, 95% CI: 1.0–9.8) (Figure 4). While South Korea had the highest prevalence of S. sonnei in Asia, the species has now become dominant in developing countries, such as Iran, Israel, Turkey and Vietnam, where it was estimated at 60.7%, 62.4%, 89.7% and 91.7% of the total Shigella spp. isolated from the respective countries (Figure 4c). S. flexneri, conversely, was found to still be dominant in many developing countries, such as Bangladesh, Cambodia, China, India, Kuwait/U.A.E and Nepal (Figure 4b). Although Taiwan is regarded as a highly developed country, S. flexneri was found to have the highest prevalence (89.7%, 95% CI: 75.8–97.1) in the country, followed by S. sonnei (19.0%, 95% CI: 5.4–41.9). The prevalence estimates of S. boydii and S. dysenteriae, on the other hand, were the highest in Bangladesh (19.9%, 95% CI: 18.4–21.4) and Nepal (27.2%, 95% CI: 23.1–31.7), respectively. Our estimated prevalence for S. sonnei in Iran was slightly higher than previously reported, where it was estimated to be 54.1%, while our pooled prevalence for S. flexneri was slightly lower (36.9% vs. 40.1%) [86]. In an SRMA published in 2012, it was reported that the prevalence of S. sonnei in China was estimated to be 21.3% [87]. However, here we report a significant increase in the prevalence of S. sonnei in that country, estimated to be 51.2%. While it was previously reported that the prevalence of S. flexneri in China was estimated to be 76.2% [87], our analysis showed a substantial decrease in the prevalence of the species in that country (Figure 4b). Increases in the prevalence of S. sonnei in both countries could be attributable to the unprecedented global spread of the species across developing countries in Asia, Latin America and the Middle East [3].
Concerning the antimicrobial drugs used to treat shigellosis, the WHO published guidelines in 2005 for shigellosis treatment, with a recommendation of ciprofloxacin as the first-line antimicrobial therapy for all patients with bloody diarrhea, irrespective of age. In cases of multi-resistant strains of Shigella with resistance against ciprofloxacin, pivmecillinam and ceftriaxone were recommended as the second-line treatment in all age groups, while azithromycin was also recommended as an alternative for treatment of adults [8,88]. Antibiotics such as nalidixic acid, ampicillin, amoxicillin, chloramphenicol, sulfamethoxazole/trimethoprim and tetracyclines were highlighted as ineffective against Shigella due to increasing antimicrobial resistance [8]. These antibiotics were commonly used in the past to treat shigellosis. In this meta-analysis, resistance against all classes of antibiotics was common among Shigella isolates, except for carbapenems, against which resistance was almost non-existent (Table 3). The pooled prevalence of Shigella resistant against streptomycin was the highest, estimated at 98.4% (95% CI: 96.9–100.0), followed by trimethoprim (95.5%, 95% CI: 92.3–98.8) and ticarcillin (90.5%, 95% CI: 84.7–96.3). High resistance rates were observed against the penicillin class of antibiotics, with more than 70% of Shigella isolates being resistant to amoxicillin (73.2%, 95% CI: 52.4–94.1), ampicillin (72.6%, 95% CI: 66.4–78.7) and oxacillin (71.9%, 95% CI: 53.3–86.3). While the prevalence of Shigella isolates resistant to penicillins was relatively high, combinations with other antimicrobial agents somehow decreased the resistance to amoxicillin, ampicillin, piperacillin and ticarcillin. For instance, the combination of amoxicillin with clavulanic acid rendered Shigella more susceptible, with the pooled prevalence of resistant Shigella decreasing from 73.2% to 23.4% (95% CI: 12.4–34.4). Similarly, the combination of ampicillin with sulbactam decreased the resistance of Shigella from 72.6% to 27.5% (95% CI: 15.1–39.9). It is possible that utilizing more than one β-lactam antibiotic in the treatment of shigellosis decreases the resistance of the pathogen significantly.
Relatively high proportions of Shigella isolates were resistant to ciprofloxacin (29.8%, 95% CI: 22.4–37.1), mecillinam (37.8%, 95% CI: 8.8–66.7), ceftriaxone (23.8%, 95% CI: 16.1–31.6) and azithromycin (29.2%, 95% CI: 20.8–37.6), in spite of their importance as first- and second-line treatments for shigellosis. High rates of resistance to these antibiotics are unfortunate developments in the majority of Asian countries and could reflect the excessive and unjustified use of antibiotics in general care to treat shigellosis. In India, 67.9% of the total Shigella isolates were reported to be resistant to ciprofloxacin, and 60.2% of S. flexneri and 57.6% of S. dysenteriae were found to be resistant to the antibiotic between 2004 and 2008 [68]. In a more recent study published in 2021, resistance to ciprofloxacin was recorded in 61.5% of the total Shigella isolates in the country, and resistance rates in S. flexneri and S. sonnei isolates were reported at 63.9% and 58.3%, respectively [65]. A study in China found that the rate of resistance against ciprofloxacin was comparatively low, recorded at 11.6% of the total Shigella isolates in the past decade [75], while another study reported a higher rate of resistance of 27.9% against the antibiotic within the same period [71]. A total of 25.4% of S. flexneri and 35.0% of S. sonnei were found to be resistant to ciprofloxacin in China between 2005 and 2011 [71]. Additionally, findings of high resistance against first-generation cephalosporins, such as cephalothin (33.6%, 95% CI: 10.7–56.5) and cefalexin (56.4%, 95% CI: 0.0–100.0); kanamycin (51.9%, 95% CI: 0.8–100.0); and erythromycin (57.4%, 95% CI: 51.6–63.2) are alarming, as we are now left with a limited number of effective antibiotics for the treatment of shigellosis.
Resistance in Shigella spp. against a wide array of antimicrobial drugs was also reported by a comprehensive study in Ethiopia, where high resistance rates against ampicillin (83.1%), amoxicillin (84.1%) and erythromycin (86.5%) were recorded, while comparably low resistance was reported for ciprofloxacin (8.9%), ceftriaxone (9.3%), norfloxacin (8.2%) and gentamycin (17.3%) [82]. In another report, rates of resistance to nalidixic acid and ciprofloxacin in Asia–Africa were 33.6% and 5.0%, respectively, but progressively increased each year, reaching 64.5% and 29.1%, respectively, in 2007–2009. In contrast, rates of resistance to nalidixic acid and ciprofloxacin in Europe–America remained low, estimated at 2.1% and 0.6%, respectively [2]. As resistance to ciprofloxacin and ceftriaxone continues to increase each year, the use of both antibiotics empirically as the first-line antimicrobial therapy in the treatment of shigellosis should be reconsidered. Due to the distribution of different Shigella spp. and variability in their antibiotic resistance profiles from one geographical location to another, which may also change with time, continuous assessment of resistance patterns is necessary for appropriate treatment of shigellosis. Nevertheless, our results showed that resistance to carbapenems, such as ertapenem (0.0%, 95% CI: 0.0–1.1), imipenem (0.1%, 95% CI: 0.0–0.2) and meropenem (0.0%, 95% CI: 0.0–0.7), was the lowest in Asia among the 49 tested antibiotics. Although the resistance rates against the carbapenem antibiotics were almost non-existent, careful and justified use of such antibiotics is essential to limit the emergence of new drug-resistant strains.
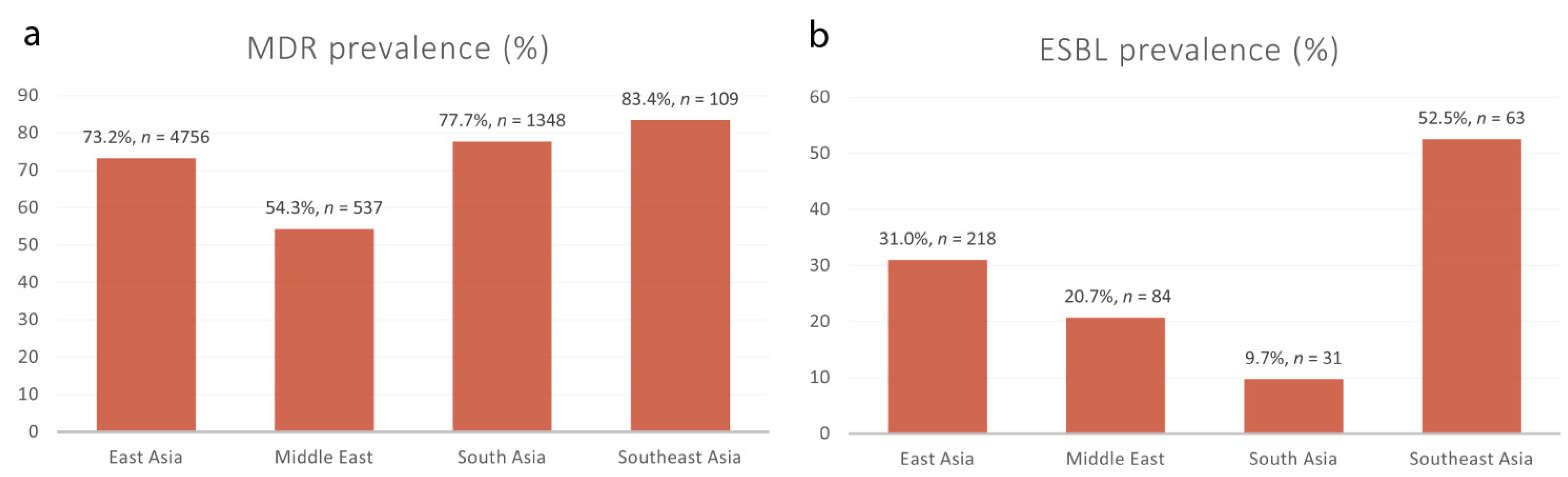
Furthermore, the high resistance burden of many antimicrobial drugs has resulted in relatively high rates of MDR in Shigella isolates. Evidence suggests that MDR is an alarming challenge in the treatment of shigellosis, with about half of Shigella strains around the globe now being resistant to multiple antibiotics [13]. For instance, S. sonnei isolates carrying the plasmid-mediated polymyxin resistance gene mcr-1 have been shown to be resistant to azithromycin and third-generation cephalosporins, including ceftriaxone and cefoperazone [89]. Resistance to third-generation cephalosporins has also been linked to the production of ESBLs, conferring resistance to β-lactamases [90]. Our pooled estimate of MDR Shigella in Asia was 68.7% (95% CI: 59.9–77.5) (Figure 5)—substantially higher than that in a report from Brazil (43.8%) [91], but much lower than reports from Egypt (88.0%) [92] and Ethiopia (83.2%) [82]. The highest prevalence of MDR Shigella spp. was recorded in Southeast Asia, which was estimated to be 83.4%, followed by South Asia (77.7%), East Asia (73.2%) and the Middle East (54.3%) (Figure 7). This is alarming, as Shigella resistance to multiple antibiotics in the Asian population has been steadily increasing in the past few decades, especially resistance against quinolones, penicillins and third-generation cephalosporins. Multiple drug-resistant pathogens are currently considered to be significant global public health threats. For example, the pooled prevalence of MDR Pseudomonas aeruginosa in Spain was reported at 5.0%, while MDR Acinetobacter baumanii, Klebsiella pneumoniae and E. coli were reported at 10.0%, 32.5% and 40.0%, respectively [93]. Among diarrheagenic pathogens in Ethiopia, the highest prevalence of 80.8% was recorded for MDR Campylobacter, followed by MDR E. coli and Salmonella, recorded at 78.2% and 59.5%, respectively [94]. In Asia, the pooled prevalence of MDR-DEC was estimated to be 66.3% (95% CI: 58.9–73.7) [84]. The acquisition of ESBL genes is one of many mechanisms that could lead to an increase in MDR Shigella. In our analysis, the prevalence of ESBL-producing Shigella spp. in Asia was 23.9% (95% CI: 12.9–34.8) (Figure 6). The highest prevalence of ESBL-producing Shigella spp. was recorded in Southeast Asia (52.5%), followed by East Asia (31.0%), the Middle East (20.7%) and South Asia (9.7%) (Figure 7). In Australia, there was a significant increase in cases of ESBL-producing S. sonnei in the recent years, with 65% of S. sonnei isolates carrying the blaCTX-M-27 gene, which confers resistance to extended-spectrum cephalosporins. These isolates were multidrug-resistant, including resistance to azithromycin and co-trimoxazole, and had reduced susceptibility to ciprofloxacin [95]. In a study of the Dutch population, the prevalence of ESBL-producing Enterobacteriaceae was estimated to be 10.1%, and the most common isolate was CTX-M-15, accounting for 47% of the ESBL-producing organisms in the country. This is worrying; even though the rate of antibiotic consumption in humans is low in the Netherlands, due to the spread of CTX-M ESBLs, particularly CTX-M-15, resistance is emerging [96].
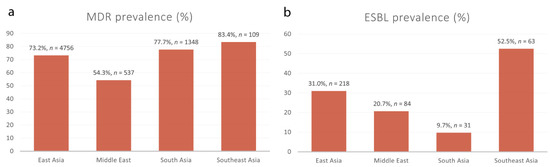
Figure 7.
The prevalence rates of MDR and ESBL-producing Shigella isolates grouped according to different Asian regions. (a) The prevalence rates of MDR Shigella spp. in East Asia (73.2%), the Middle East (54.3%), South Asia (77.7%) and Southeast Asia (83.4%). (b) The prevalence rates of ESBL-producing Shigella spp. in East Asia (31.0%), the Middle East (20.7%), South Asia (9.7%) and Southeast Asia (52.5%).
Our SRMA is the first to provide a comprehensive analysis of antimicrobial-resistant Shigella spp. in the Asian population. However, it has several notable limitations. First, even though a significant number of studies were included (n = 63) in this SRMA, they did not incorporate all of the countries in Asia; thus, the estimated prevalence might not wholly represent the true scale of antimicrobial-resistant Shigella spp. in Asia. Nevertheless, data were collected from 14 Asian countries—a large number of shigellosis cases (n = 19,962) were analyzed. Second, significant heterogeneity was observed in our SRMA, although this is common in meta-analyses of prevalence estimation [82,84,87,94,97]. It was expected, as we utilized a random-effects model in our meta-analysis, which presumes heterogeneity [85]. Third, the potential effects of gender and age distribution on the prevalence of Shigella spp. could not be accounted for in this study due to the nature of the data reporting in most of the included studies; while some studies reported antimicrobial resistance from pediatrics, others reported data from various age groups, and many did not disclose the patient age groups (Table 1). The last and most important limitation is that the patterns of multidrug resistance of different Shigella spp. could not be obtained owing to the different types of data presented in various studies. Many of the included studies did not report the prevalence of antimicrobial resistance of different Shigella spp. but instead reported the prevalence of drug-resistant Shigella collectively. Thus, we were also unable to account for the prevalence of antimicrobial-resistant Shigella on a yearly basis. Nevertheless, we believe that such crucial information is needed and would be helpful to clinicians, researchers and governments.
4. Methodology
4.1. Literature Search Strategy and Selection
This SRMA was conducted based on the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) [98]. The protocol of this study was registered in the International Prospective Register of Systematic Reviews (PROSPERO) database (registration number: CRD42022352131). From July 2022 to August 2022, a comprehensive literature search was performed to find eligible studies on the prevalence of antimicrobial-resistant Shigella spp. in Asia that were available in the PubMed, ScienceDirect and Scopus databases (Figure 1). All eligible articles were retrieved from the three databases using relevant search terms and keywords, which included “Shigella sonnei AND drug resistance AND Asia”, “Shigella flexneri AND drug resistance AND Asia”, “Shigella boydii AND drug resistance AND Asia”, “Shigella dysenteriae AND drug resistance AND Asia” and “Shigella AND drug resistance AND Asia”. In addition, the reference lists of the selected articles were checked for more related eligible articles.
4.2. Inclusion and Exclusion Criteria
Only studies that reported sufficient data for determining the prevalence of antimicrobial-resistant Shigella spp. in patients from countries in the Asian region, regardless of gender and age, were considered eligible for inclusion in this SRMA. Additionally, articles that reported the prevalence of ESBL-producing Shigella spp. were also included in this study. Among such articles, only full-length original research articles written in the English language were considered for the analysis. Furthermore, only articles that were published from 2010 to July 2022 were selected in order to obtain up-to-date information on the topic. Studies that did not report any data on Shigella spp. and their antimicrobial susceptibility, case reports and case studies, review articles and short communications, as well as studies with abstracts only, were excluded from the analysis. Moreover, unpublished articles or articles with incomplete information, as well as all articles published before 2010, were not included in the study. Only data from human-related studies were included in the analysis.
4.3. Data Extraction and Quality Control
All eligible studies were collected and managed using EndNote 20, in which duplicate articles were removed, and the remaining articles were assessed systematically based on the titles and abstracts. Two authors, M.Z.S. and N.M.N.N.Z., independently evaluated the eligibility of all articles by examining the full texts using predetermined inclusion criteria. Only 63 eligible articles were selected and coded, and data from each article were extracted to a table in Microsoft Excel that contained necessary information, such as the author names, title, year of publication, study period, study region, study design and methods used, sample population, sample size, sample type, age group, gender, the isolated Shigella spp., resistance patterns of the Shigella isolates and the prevalence of MDR and ESBL-producing Shigella spp. In this study, MDR was defined as resistance to three or more antimicrobial drugs.
4.4. Data Analysis
All 63 selected studies were included in the meta-analysis, and the analysis was carried out using metaprop codes in the meta (version 5.2-0) and metafor (version 3.4-0) packages of R (version 4.2.1), as implemented in RStudio (Posit, PBC, Boston, USA) (version 2022.02.2+485) [99]. The pooled prevalence of Shigella spp. resistant to each antibiotic, the pooled prevalence of MDR and ESBL-producing Shigella isolates and 95% confidence intervals (CIs) were calculated using the REML method for the random-effects model. In addition, statistical heterogeneity between the studies was measured using Cochran’s Q test for the heterogeneity significance and inconsistency index (I2) [100], for which I2 of >75% and a significance level of <0.05 (p-value) were interpreted as evidence of significant heterogeneity. The presence of publication bias was determined by evaluating a funnel plot, with significance tested using Egger’s test only for the included studies with publication bias greater than 10.
5. Conclusions
Our SRMA presents significant evidence of antimicrobial-resistant Shigella spp. distribution in the Asian population. The pooled prevalence estimates showed a substantially high proportion of multidrug-resistant Shigella in Asia, posing a major burden to public health. Our meta-analysis revealed that the pooled prevalence of MDR Shigella in Asia was estimated to be 68.7%, substantially higher than in many other countries, while the pooled prevalence of ESBL-producing Shigella strains was 23.9%. Even though the prevalence was estimated to vary across different Asian countries, the evidence suggests that multidrug resistance is a pressing threat to public health worthy of careful and justified interventions. It is therefore vital to continuously monitor antimicrobial-resistant Shigella spp. by conducting vigorous drug susceptibility tests so that reliable and effective antibiotic resistance mitigation strategies can be implemented, which may lead to better outcomes for the treatment and control of shigellosis in Asia, as well as in different parts of the world.
Author Contributions
M.Z.S.: conceptualization, methodology, data collection, curation and analysis, visualization, validation, draft writing, manuscript review. N.M.N.N.Z.: data collection, validation, draft writing, manuscript review. K.H.: curation and analysis, manuscript review. M.I.I.: supervision, manuscript review. K.K.B.S.: supervision, validation, manuscript review. Z.Z.D.: supervision, manuscript review. All authors have read and agreed to the published version of the manuscript.
Funding
This study received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data relevant to this review are included in the text and references.
Acknowledgments
We thank Universiti Sains Malaysia for providing postdoctoral fellowships to M.Z.S and N.M.N.N.Z.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chung The, H.; Rabaa, M.A.; Pham Thanh, D.; De Lappe, N.; Cormican, M.; Valcanis, M.; Howden, B.P.; Wangchuk, S.; Bodhidatta, L.; Mason, C.J.; et al. South Asia as a Reservoir for the Global Spread of Ciprofloxacin-Resistant Shigella sonnei: A Cross-Sectional Study. PLoS Med. 2016, 13, e1002055. [Google Scholar] [CrossRef] [PubMed]
- Gu, B.; Cao, Y.; Pan, S.; Zhuang, L.; Yu, R.; Peng, Z.; Qian, H.; Wei, Y.; Zhao, L.; Liu, G.; et al. Comparison of the prevalence and changing resistance to nalidixic acid and ciprofloxacin of Shigella between Europe-America and Asia-Africa from 1998 to 2009. Int. J. Antimicrob. Agents 2012, 40, 9–17. [Google Scholar] [CrossRef]
- Thompson, C.N.; Duy, P.T.; Baker, S. The Rising Dominance of Shigella sonnei: An Intercontinental Shift in the Etiology of Bacillary Dysentery. PLoS Negl. Trop. Dis. 2015, 9, e0003708. [Google Scholar] [CrossRef] [PubMed]
- Ud-Din, A.I.M.S.; Wahid, S.U.H.; Latif, H.A.; Shahnaij, M.; Akter, M.; Azmi, I.J.; Hasan, T.N.; Ahmed, D.; Hossain, M.A.; Faruque, A.S.G.; et al. Changing Trends in the Prevalence of Shigella species: Emergence of Multi-Drug Resistant Shigella sonnei Biotype g in Bangladesh. PLoS ONE 2013, 8, 82601. [Google Scholar] [CrossRef] [PubMed]
- The, H.C.; Baker, S. Out of Asia: The independent rise and global spread of fluoroquinolone-resistant Shigella. Microb. Genomics 2018, 4, e000171. [Google Scholar] [CrossRef]
- Kotloff, K.L.; Riddle, M.S.; Platts-Mills, J.A.; Pavlinac, P.; Zaidi, A.K.M. Shigellosis. Lancet 2018, 391, 801–812. [Google Scholar] [CrossRef]
- Khalil, I.A.; Troeger, C.; Blacker, B.F.; Rao, P.C.; Brown, A.; Atherly, D.E.; Brewer, T.G.; Engmann, C.M.; Houpt, E.R.; Kang, G.; et al. Morbidity and mortality due to shigella and enterotoxigenic Escherichia coli diarrhoea: The Global Burden of Disease Study 1990–2016. Lancet Infect. Dis. 2018, 18, 1229–1240. [Google Scholar] [CrossRef]
- WHO Guidelines for the Control of Shigellosis, Including Epidemics Due to Shigella Dysenteriae Type 1. Available online: https://apps.who.int/iris/handle/10665/43252 (accessed on 15 September 2022).
- Chung The, H.; Boinett, C.; Pham Thanh, D.; Jenkins, C.; Weill, F.X.; Howden, B.P.; Valcanis, M.; De Lappe, N.; Cormican, M.; Wangchuk, S.; et al. Dissecting the molecular evolution of fluoroquinolone-resistant Shigella sonnei. Nat. Commun. 2019, 10, 4828. [Google Scholar] [CrossRef]
- Chung The, H.; Bodhidatta, L.; Pham, D.T.; Mason, C.J.; Ha Thanh, T.; Voong Vinh, P.; Turner, P.; Hem, S.; Dance, D.A.B.; Newton, P.N.; et al. Evolutionary histories and antimicrobial resistance in Shigella flexneri and Shigella sonnei in Southeast Asia. Commun. Biol. 2021, 4, 353. [Google Scholar] [CrossRef]
- Von Seidlein, L.; Deok, R.K.; Ali, M.; Lee, H.; Wang, X.Y.; Vu, D.T.; Do, G.C.; Chaicumpa, W.; Agtini, M.D.; Hossain, A.; et al. A multicentre study of Shigella diarrhoea in six Asian countries: Disease burden, clinical manifestations, and microbiology. PLoS Med. 2006, 3, 1556–1569. [Google Scholar] [CrossRef]
- Baker, K.S.; Dallman, T.J.; Field, N.; Childs, T.; Mitchell, H.; Day, M.; Weill, F.X.; Lefèvre, S.; Tourdjman, M.; Hughes, G.; et al. Horizontal antimicrobial resistance transfer drives epidemics of multiple Shigella species. Nat. Commun. 2018, 9, 1462. [Google Scholar] [CrossRef] [PubMed]
- Ranjbar, R.; Farahani, A. Shigella: Antibiotic-Resistance Mechanisms and New Horizons for Treatment. Infect. Drug Resist. 2019, 12, 3137. [Google Scholar] [CrossRef] [PubMed]
- Nhu, N.T.K.; Vinh, H.; Nga, T.V.T.; Stabler, R.; Duy, P.T.; Vien, L.T.M.; Van Doorn, H.R.; Cerdeño-Tárraga, A.M.; Thomson, N.; Campbell, J.; et al. The Sudden Dominance of blaCTX–M Harbouring Plasmids in Shigella spp. Circulating in Southern Vietnam. PLoS Negl. Trop. Dis. 2010, 4, e702. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, P.; Uppal, B.; Ghosh, R.; Krishna Prakash, S.; Chakravarti, A.; Jha, A.K.; Rajeshwari, K. Multi drug resistance and Extended Spectrum Beta Lactamases in clinical isolates of Shigella: A study from New Delhi, India. Travel Med. Infect. Dis. 2016, 14, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, D.; Hoque, A.; Elahi, M.S.B.; Endtz, H.P.; Hossain, M.A. Bacterial aetiology of diarrhoeal diseases and antimicrobial resistance in Dhaka, Bangladesh, 2005–2008. Epidemiol. Infect. 2012, 140, 1678–1684. [Google Scholar] [CrossRef]
- Alizadeh-Hesar, M.; Bakhshi, B.; Najar-Peerayeh, S. Clonal dissemination of a single Shigella sonnei strain among Iranian children during Fall 2012 in Tehran, I.R. Iran. Infect. Genet. Evol. 2015, 34, 260–266. [Google Scholar] [CrossRef]
- Aminshahidi, M.; Arastehfar, A.; Pouladfar, G.; Arman, E.; Fani, F. Diarrheagenic Escherichia coli and Shigella with High Rate of Extended-Spectrum Beta-Lactamase Production: Two Predominant Etiological Agents of Acute Diarrhea in Shiraz, Iran. Microb. Drug Resist. 2017, 23, 1037–1044. [Google Scholar] [CrossRef]
- Behruznia, P.; Sadredinamin, M.; Hashemi, A.; Hajikhani, B.; Yousefi Nojookambari, N.; Behruznia, M.; Ghalavand, Z. Decreased Susceptibility of Shigella isolates to Azithromycin in Children in Tehran, Iran. Can. J. Infect. Dis. Med. Microbiol. 2022, 2022, 4503964. [Google Scholar] [CrossRef]
- Bhattacharya, D.; Bhattacharya, H.; Sayi, D.S.; Bharadwaj, A.P.; Singhania, M.; Sugunan, A.P.; Roy, S. Changing patterns and widening of antibiotic resistance in Shigella spp. over a decade (2000-2011), Andaman Islands, India. Epidemiol. Infect. 2015, 143, 470–477. [Google Scholar] [CrossRef]
- Bhattacharya, D.; Sugunan, A.P.; Bhattacharjee, H.; Thamizhmani, R.; Sayi, D.S.; Thanasekaran, K.; Manimunda, S.P.; Ghosh, A.R.; Bharadwaj, A.P.; Singhania, M.; et al. Antimicrobial resistance in Shigella—Rapid increase & widening of spectrum in Andaman Islands, India. Indian J. Med. Res. 2012, 135, 365. [Google Scholar]
- Bialvaei, A.Z.; Kafil, H.S.; Asgharzadeh, M.; Aghazadeh, M.; Yousefi, M. CTX-M extended-spectrum β-lactamase-producing Klebsiella spp, Salmonella spp, Shigella spp and Escherichia coli isolates in Iranian hospitals. Braz. J. Microbiol. 2016, 47, 706–711. [Google Scholar] [CrossRef] [PubMed]
- Chamoun, K.; Farah, M.; Araj, G.; Daoud, Z.; Moghnieh, R.; Salameh, P.; Saade, D.; Mokhbat, J.; Abboud, E.; Hamze, M.; et al. Surveillance of antimicrobial resistance in Lebanese hospitals: Retrospective nationwide compiled data. Int. J. Infect. Dis. 2016, 46, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Yang, C.; Wang, J.; Liang, B.; Yi, S.; Li, H.; Liu, H.; Li, P.; Wu, Z.; Xie, J.; et al. Antimicrobial Resistance of Shigella flexneri Serotype 1b Isolates in China. PLoS ONE 2015, 10, e0129009. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Natarajan, M.; Mandal, J. The Emergence of Quinolone Resistant Shigella sonnei, Pondicherry, India. PLoS ONE 2016, 11, e0160290. [Google Scholar] [CrossRef]
- Dhital, S.; Sherchand, J.B.; Pokharel, B.M.; Parajuli, K.; Mishra, S.K.; Sharma, S.; Kattel, H.P.; Khadka, S.; Khatiwada, S.; Rijal, B. Antimicrobial susceptibility pattern of Shigella spp. isolated from children under 5 years of age attending tertiary care hospitals, Nepal along with first finding of ESBL-production. BMC Res. Notes 2017, 10, 192. [Google Scholar] [CrossRef]
- Duong, V.T.; Tuyen, H.T.; Van Minh, P.; Campbell, J.I.; Le Phuc, H.; Nhu, T.D.H.; Tu, L.T.P.; Chau, T.T.H.; Nhi, L.T.Q.; Hung, N.T.; et al. No Clinical Benefit of Empirical Antimicrobial Therapy for Pediatric Diarrhea in a High-Usage, High-Resistance Setting. Clin. Infect. Dis. 2018, 66, 504. [Google Scholar] [CrossRef]
- Farajzadeh Sheikh, A.; Moradi Bandbal, M.; Saki, M. Emergence of multidrug-resistant Shigella species harboring extended-spectrum beta-lactamase genes in pediatric patients with diarrhea from southwest of Iran. Mol. Biol. Rep. 2020, 47, 7097–7106. [Google Scholar] [CrossRef]
- Ghosh, S.; Pazhani, G.P.; Chowdhury, G.; Guin, S.; Dutta, S.; Rajendran, K.; Bhattacharya, M.K.; Takeda, Y.; Niyogi, S.K.; Balakrish Nair, G.; et al. Genetic characteristics and changing antimicrobial resistance among Shigella spp. isolated from hospitalized diarrhoeal patients in Kolkata, India. J. Med. Microbiol. 2011, 60, 1460–1466. [Google Scholar] [CrossRef]
- Gu, B.; Xu, T.; Kang, H.; Xu, Y.; Liu, G.; Pan, S.; Qian, H.; Ma, P. A 10-year surveillance of antimicrobial susceptibility patterns in Shigella sonnei isolates circulating in Jiangsu Province, China. J. Glob. Antimicrob. Resist. 2017, 10, 29–34. [Google Scholar] [CrossRef]
- Iqbal, M.S.; Rahman, M.; Islam, R.; Banik, A.; Amin, M.B.; Akter, F.; Talukder, K.A. Plasmid-Mediated Sulfamethoxazole Resistance Encoded by the sul2 Gene in the Multidrug-Resistant Shigella flexneri 2a Isolated from Patients with Acute Diarrhea in Dhaka, Bangladesh. PLoS ONE 2014, 9, 85338. [Google Scholar] [CrossRef]
- Jain, P.; Kulkarni, R.; Dutta, S.; Ganavali, A.; Kalabhavi, A.; Shetty, P.; Shubhada, C.; Hosamani, M.; Appannanavar, S.; Hanamaraddi, D. Prevalence and antimicrobial profile of Shigella isolates in a tertiary care hospital of North Karnataka: A 12-year study. Indian J. Med. Microbiol. 2020, 38, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Jamal, W.; Rotimi, V.O.; Pal, T.; Sonnevend, A.; Dimitrov, T.S. Comparative in vitro activity of tigecycline and other antimicrobial agents against Shigella species from Kuwait and the United Arab of Emirates. J. Infect. Public Health 2010, 3, 35–42. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jean, S.S.; Lu, M.C.; Shi, Z.Y.; Tseng, S.H.; Wu, T.S.; Lu, P.L.; Shao, P.L.; Ko, W.C.; Wang, F.-D.; Hsueh, P.R. In vitro activity of ceftazidime–avibactam, ceftolozane–tazobactam, and other comparable agents against clinically important Gram-negative bacilli: Results from the 2017 Surveillance of Multicenter Antimicrobial Resistance in Taiwan (SMART). Infect. Drug Resist. 2018, 11, 1983. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Oh, Y.; Jung, J.; Kim, S.; Kim, J.; Han, K.; Kim, M.; Park, S.; Lee, Y. Antimicrobial resistance patterns and characterization of integrons of Shigella sonnei isolates in Seoul, 1999–2008. J. Microbiol. 2010, 48, 236–242. [Google Scholar] [CrossRef]
- Jomezadeh, N.; Babamoradi, S.; Kalantar, E.; Javaherizadeh, H. Isolation and antibiotic susceptibility of Shigella species from stool samples among hospitalized children in Abadan, Iran. Gastroenterol. Hepatol. Bed Bench 2014, 7, 218. [Google Scholar]
- Kacmaz, B.; Unaldi, O.; Sultan, N.; Durmaz, R. Drug resistance profiles and clonality of sporadic Shigella sonnei isolates in Ankara, Turkey. Braz. J. Microbiol. 2014, 45, 845. [Google Scholar] [CrossRef]
- Kang, H.; Wang, L.; Li, Y.; Lu, Y.; Fan, W.; Bi, R.; Qian, H.; Gu, B. Dissemination of Multidrug-Resistant Shigella flexneri and Shigella sonnei with Class 1, Class 2, and Atypical Class 1 Integrons in China. Microb. Drug Resist. 2019, 25, 1465–1474. [Google Scholar] [CrossRef]
- Kansakar, P.; Baral, P.; Malla, S.; Ghimire, G.R. Antimicrobial susceptibilities of enteric bacterial pathogens isolated in Kathmandu, Nepal, during 2002–2004. J. Infect. Dev. Ctries. 2011, 5, 163–168. [Google Scholar] [CrossRef][Green Version]
- Khan, S.; Singh, P.; Ansari, M.; Asthana, A. Isolation of Shigella species and their resistance patterns to a panel of fifteen antibiotics in mid and far western region of Nepal. Asian Pac. J. Trop. Dis. 2014, 4, 30. [Google Scholar] [CrossRef]
- Lee, Y.L.; Lu, M.C.; Shao, P.L.; Lu, P.L.; Chen, Y.H.; Cheng, S.H.; Ko, W.C.; Lin, C.Y.; Wu, T.S.; Yen, M.Y.; et al. Nationwide surveillance of antimicrobial resistance among clinically important Gram-negative bacteria, with an emphasis on carbapenems and colistin: Results from the Surveillance of Multicenter Antimicrobial Resistance in Taiwan (SMART) in 2018. Int. J. Antimicrob. Agents 2019, 54, 318–328. [Google Scholar] [CrossRef]
- Liu, H.; Zhu, B.; Qiu, S.; Xia, Y.; Liang, B.; Yang, C.; Dong, N.; Li, Y.; Xiang, Y.; Wang, S.; et al. Dominant serotype distribution and antimicrobial resistance profile of Shigella spp. in Xinjiang, China. PLoS ONE 2018, 13, e0195259. [Google Scholar] [CrossRef]
- Maharjan, S.; Rayamajhee, B.; Shreshtha, A.; Acharya, J. Serotyping and antibiotic susceptibility patterns of Vibrio and Shigella isolates from diarrheal patients visiting a Tropical and Infectious Diseases Hospital in central Nepal. BMC Res. Notes 2017, 10, 626. [Google Scholar] [CrossRef] [PubMed]
- Mamishi, S.; Arab Yazdi, Z.; Mahmoudi, S.; Moradzadeh, M.; Taghi Haghi Ashtiani, M.; Pourakbari, B. Antimicrobial-resistance pattern of Shigella species in children: A six-year study in an Iranian referral Hospital. Ann. Ig. 2019, 31, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Meng, C.Y.; Smith, B.L.; Bodhidatta, L.; Richard, S.A.; Vansith, K.; Thy, B.; Srijan, A.; Serichantalergs, O.; Mason, C.J. Etiology of diarrhea in young children and patterns of antibiotic resistance in Cambodia. Pediatr. Infect. Dis. J. 2011, 30, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Mohebi, S.; Nave, H.H.; Javadi, K.; Amanati, A.; Kholdi, S.; Hadadi, M.; Hashemizadeh, Z.; Motamedifar, M. Evaluate the distribution of virulence genes and to investigate antibiotic resistance pattern among Shigella species isolated from children with shigellosis in Iran. Gene Rep. 2021, 23, 101189. [Google Scholar] [CrossRef]
- Nandy, S.; Mitra, U.; Rajendran, K.; Dutta, P.; Dutta, S. Subtype prevalence, plasmid profiles and growing fluoroquinolone resistance in Shigella from Kolkata, India (2001–2007): A hospital-based study. Trop. Med. Int. Health 2010, 15, 1499–1507. [Google Scholar] [CrossRef]
- Nath, R.; Saikia, L.; Choudhury, G.; Sharma, D. Drug resistant Shigella flexneri in & around Dibrugarh, north-east India. Indian J. Med. Res. 2013, 137, 183. [Google Scholar]
- Özmert, E.N.; Ince, O.T.; Örün, E.; Yalçin, S.; Yurdakök, K.; Gür, D. Clinical characteristics and antibiotic resistance of Shigella gastroenteritis in Ankara, Turkey between 2003 and 2009, and comparison with previous reports. Int. J. Infect. Dis. 2011, 15, e849–e853. [Google Scholar] [CrossRef]
- Pakbin, B.; Didban, A.; Monfared, Y.K.; Mahmoudi, R.; Peymani, A.; Modabber, M.R. Antibiotic susceptibility and genetic relatedness of Shigella species isolated from food and human stool samples in Qazvin, Iran. BMC Res. Notes 2021, 14, 144. [Google Scholar] [CrossRef]
- Peleg, I.; Givon-Lavi, N.; Leibovitz, E.; Broides, A. Epidemiological trends and patterns of antimicrobial resistance of Shigella spp. isolated from stool cultures in two different populations in Southern Israel. Diagn. Microbiol. Infect. Dis. 2014, 78, 287–291. [Google Scholar] [CrossRef]
- Poramathikul, K.; Wojnarski, M.; Sok, S.; Sokh, V.; Chiek, S.; Seng, H.; Krang, S.; Ly, S.; Nou, S.; Chann, S.; et al. Update on Shigella and Nontyphoidal Salmonella Antimicrobial Drug Resistance: Implications on Empirical Treatment of Acute Infectious Diarrhea in Cambodia. Antimicrob. Agents Chemother. 2021, 65, e00671-21. [Google Scholar] [CrossRef] [PubMed]
- Pourakbari, B.; Charati, M.G.; Mahmoudi, S.; Abdolsalehi, M.R.; Sadeghi, R.H.; Mamishi, S. High frequency of antimicrobial resistance and virulence gene in Shigella species isolated from pediatric patients in an Iranian Referral Hospital. Acta Bio Med. Atenei Parm. 2022, 93, 2022027. [Google Scholar] [CrossRef]
- Prabhurajeshwar; Oli, A.K.; Ashajyothi, C.; Chandrakanth, R.K. Prevalence and antibiotic susceptibility pattern of fluoroquinolone resistant Shigella species isolated from infants stool in Gulbarga district, Karnataka, India. Asian Pac. J. Trop. Dis. 2015, 5, S116–S120. [Google Scholar] [CrossRef]
- Qu, F.; Bao, C.; Chen, S.; Cui, E.; Guo, T.; Wang, H.; Zhang, J.; Wang, H.; Tang, Y.W.; Mao, Y. Genotypes and antimicrobial profiles of Shigella sonnei isolates from diarrheal patients circulating in Beijing between 2002 and 2007. Diagn. Microbiol. Infect. Dis. 2012, 74, 166. [Google Scholar] [CrossRef] [PubMed]
- Qu, M.; Zhang, X.; Liu, G.; Huang, Y.; Jia, L.; Liang, W.; Li, X.; Wu, X.; Li, J.; Yan, H.; et al. An eight-year study of Shigella species in Beijing, China: Serodiversity, virulence genes, and antimicrobial resistance. J. Infect. Dev. Ctries. 2014, 8, 904–908. [Google Scholar] [CrossRef][Green Version]
- Rajpara, N.; Nair, M.; Chowdhury, G.; Mukhopadhyay, A.K.; Ramamurthy, T.; Niyogi, S.K.; Bhardwaj, A.K. Molecular analysis of multidrug resistance in clinical isolates of Shigella spp. from 2001-2010 in Kolkata, India: Role of integrons, plasmids, and topoisomerase mutations. Infect. Drug Resist. 2018, 11, 87–102. [Google Scholar] [CrossRef]
- Sadredinamin, M.; Shabani, M.; Karimi, A.; Sohrabi, M.R.; Karimi-Yazdi, M.; Ghalavand, Z.; Alebouyeh, M. Virulence genes expression profiling of different Shigella flexneri serotypes in response to sub-inhibitory concentrations of azithromycin and ciprofloxacin. Gut Pathog. 2022, 14, 10. [Google Scholar] [CrossRef]
- Sah, S.K.; Basnet, S.; Shrestha, S.; Ghale, K.; Tamang, S.; Mandal, D.K.; Pun, S.B. Burden of Shigella spp and Vibrio spp, and their antibiotic sensitivity pattern in the patients with acute gastroenteritis in tertiary care hospital in Nepal. BMC Res. Notes 2019, 12, 699. [Google Scholar] [CrossRef]
- Rizi, K.S.; Farsiani, H.; Sasan, M.S. High rate of resistance to ceftriaxone and azithromycin among Shigella spp. isolates at three children’s referral hospitals in Northeast Iran. J. Infect. Chemother. 2020, 26, 955–958. [Google Scholar] [CrossRef]
- Shakya, G.; Acharya, J.; Adhikari, S.; Rijal, N. Shigellosis in Nepal: 13 years review of nationwide surveillance. J. Health Popul. Nutr. 2016, 35, 36. [Google Scholar] [CrossRef]
- Shen, Y.; Qian, H.; Gong, J.; Deng, F.; Dong, C.; Zhou, L.; Guo, H. High Prevalence of Antibiotic Resistance and Molecular Characterization of Integrons among Shigella isolates in Eastern China. Antimicrob. Agents Chemother. 2013, 57, 1549. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Soltan Dallal, M.M.; Ranjbar, R.; Yaghoubi, S.; Rajabi, Z.; Aminharati, F.; Adeli Behrooz, H. Molecular epidemiology and genetic characterization of Shigella in pediatric patients in Iran. Le Infez. Med. 2018, 26, 321–328. [Google Scholar]
- Tajbakhsh, M.; García Migura, L.; Rahbar, M.; Svendsen, C.A.; Mohammadzadeh, M.; Zali, M.R.; Aarestrup, F.M.; Hendriksen, R.S. Antimicrobial-resistant Shigella infections from Iran: An overlooked problem? J. Antimicrob. Chemother. 2012, 67, 1128–1133. [Google Scholar] [CrossRef]
- Taneja, N.; Tiewsoh, J.B.A.; Gupta, S.; Mohan, B.; Verma, R.; Shankar, P.; Narayan, C.; Yadav, V.K.; Jayashree, M.; Singh, S. Antimicrobial resistance in Shigella species: Our five years (2015–2019) experience in a tertiary care center in north India. Indian J. Med. Microbiol. 2021, 39, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Teimourpour, R.; Babapour, B.; Esmaelizad, M.; Arzanlou, M.; Peeri-Doghaheh, H. Molecular characterization of quinolone resistant Shigella spp. isolates from patients in Ardabil, Iran. Iran. J. Microbiol. 2019, 11, 496. [Google Scholar] [CrossRef]
- Thompson, C.N.; Phan, M.V.T.; Van Minh Hoang, N.; Van Minh, P.; Vinh, N.T.; Thuy, C.T.; Nga, T.T.T.; Rabaa, M.A.; Duy, P.T.; Dung, T.T.N.; et al. A Prospective Multi-Center Observational Study of Children Hospitalized with Diarrhea in Ho Chi Minh City, Vietnam. Am. J. Trop. Med. Hyg. 2015, 92, 1045. [Google Scholar] [CrossRef] [PubMed]
- Urvashi; Saxena, S.; Dutta, R. Antimicrobial Resistance Pattern of Shigella species Over Five Years at a Tertiary-care Teaching Hospital in North India. J. Health Popul. Nutr. 2011, 29, 292. [Google Scholar] [CrossRef]
- Wang, Y.; Ma, Q.; Hao, R.; Zhang, Q.; Yao, S.; Han, J.; Ren, B.; Fan, T.; Chen, L.; Xu, X.; et al. Antimicrobial resistance and genetic characterization of Shigella spp. in Shanxi Province, China, during 2006–2016. BMC Microbiol. 2019, 19, 116. [Google Scholar] [CrossRef]
- Xu, Y.; Zhuang, L.; Kang, H.; Ma, P.; Xu, T.; Pan, S.; Gu, B. Prevalence, resistance patterns, and characterization of integrons of Shigella flexneri isolated from Jiangsu Province in China, 2001–2011. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 1347–1353. [Google Scholar] [CrossRef]
- Yang, H.; Chen, G.; Zhu, Y.; Liu, Y.; Cheng, J.; Hu, L.; Ye, Y.; Li, J. Surveillance of antimicrobial susceptibility patterns among Shigella species isolated in China during the 7-year period of 2005–2011. Ann. Lab. Med. 2013, 33, 111–115. [Google Scholar] [CrossRef][Green Version]
- Yang, H.; Sun, W.; Duan, G.; Zhu, J.; Zhang, W.; Xi, Y.; Fan, Q. Serotype distribution and characteristics of antimicrobial resistance in Shigella isolated from Henan province, China, 2001–2008. Epidemiol. Infect. 2013, 141, 1946–1952. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhang, C.L.; Liu, Q.Z.; Wang, J.; Chu, X.; Shen, L.M.; Guo, Y.Y. Epidemic and virulence characteristic of Shigella spp. with extended-spectrum cephalosporin resistance in Xiaoshan District, Hangzhou, China. BMC Infect. Dis. 2014, 14, 260. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Pan, F.; Zhao, X.; Wang, G.; Tu, Y.; Fu, S.; Wang, J.; Pan, J.; Song, J.; Wang, W.; et al. Distribution and antimicrobial resistance of enteric pathogens in Chinese paediatric diarrhoea: A multicentre retrospective study, 2008–2013. Epidemiol. Infect. 2015, 143, 2512–2519. [Google Scholar] [CrossRef]
- Zhang, J.; Jin, H.; Hu, J.; Yuan, Z.; Shi, W.; Yang, X.; Xu, X.; Meng, J. Antimicrobial resistance of Shigella spp. from humans in Shanghai, China, 2004–2011. Diagn. Microbiol. Infect. Dis. 2014, 78, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.X.; Chen, H.Y.; Tu, L.H.; Xi, M.F.; Chen, M.; Zhang, J. Fluoroquinolone Resistance Mechanisms in Shigella isolates in Shanghai, China, Between 2010 and 2015. Microb. Drug Resist. 2019, 25, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Abbafati, C.; Abbas, K.M.; Abbasi-Kangevari, M.; Abd-Allah, F.; Abdelalim, A.; Abdollahi, M.; Abdollahpour, I.; Abegaz, K.H.; Abolhassani, H.; Aboyans, V.; et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Troeger, C.; Forouzanfar, M.; Rao, P.C.; Khalil, I.; Brown, A.; Reiner, R.C.; Fullman, N.; Thompson, R.L.; Abajobir, A.; Ahmed, M.; et al. Estimates of global, regional, and national morbidity, mortality, and aetiologies of diarrhoeal diseases: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Infect. Dis. 2017, 17, 909. [Google Scholar] [CrossRef]
- Muthuirulandi Sethuvel, D.P.; Devanga Ragupathi, N.K.; Anandan, S.; Veeraraghavan, B. Update on: Shigella new serogroups/serotypes and their antimicrobial resistance. Lett. Appl. Microbiol. 2017, 64, 8–18. [Google Scholar] [CrossRef]
- Williams, E.; Lew, T.E.; Fuller, A.; Spelman, D.W.; Jenney, A.W. A case of multi-drug resistant ESBL-producing Shigella sonnei acute acalculous cholecystitis and gastroenteritis in a returned traveller. J. Travel Med. 2018, 25, tay029. [Google Scholar] [CrossRef]
- Barrantes, K.; Achí, R. The importance of integrons for development and propagation of resistance in Shigella: The case of Latin America. Braz. J. Microbiol. 2016, 47, 800–806. [Google Scholar] [CrossRef]
- Hussen, S.; Mulatu, G.; Yohannes Kassa, Z. Prevalence of Shigella species and its drug resistance pattern in Ethiopia: A systematic review and meta-analysis. Ann. Clin. Microbiol. Antimicrob. 2019, 18, 22. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.R.; Reddy, V.; Hanson, H.; Johnson, K.M.; Tsoi, B.; Cokes, C.; Gallagher, L.; Lee, L.; Plentsova, A.; Dang, T.; et al. Antimicrobial resistance trends of Shigella serotypes in New York City, 2006–2009. Microb. Drug Resist. 2010, 16, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Salleh, M.Z.; Zuraina, N.M.N.N.; Hajissa, K.; Ilias, M.I.; Deris, Z.Z. Prevalence of Multidrug-Resistant Diarrheagenic Escherichia coli in Asia: A Systematic Review and Meta-Analysis. Antibiotics 2022, 11, 1333. [Google Scholar] [CrossRef] [PubMed]
- Imrey, P.B. Limitations of Meta-analyses of Studies with High Heterogeneity. JAMA Netw. Open 2020, 3, e1919325. [Google Scholar] [CrossRef] [PubMed]
- Moradi, F.; Hadi, N.; Akbari, M.; Hashemizadeh, Z.; Jahromi, R.R. Frequency and Antimicrobial Resistance of Shigella species in Iran During 2000–2020. Jundishapur J. Health Sci. 2021, 13, 114902. [Google Scholar] [CrossRef]
- Chang, Z.; Lu, S.; Chen, L.; Jin, Q.; Yang, J. Causative Species and Serotypes of Shigellosis in Mainland China: Systematic Review and Meta-Analysis. PLoS ONE 2012, 7, e52515. [Google Scholar] [CrossRef]
- Salleh, M.Z.; Banga Singh, K.K.; Deris, Z.Z. Structural Insights into Substrate Binding and Antibiotic Inhibition of Enterobacterial Penicillin-Binding Protein 6. Life 2022, 12, 1022. [Google Scholar] [CrossRef]
- Ma, Q.; Huang, Y.; Wang, J.; Xu, X.; Hawkey, J.; Yang, C.; Liang, B.; Hu, X.; Wu, F.; Yang, X.; et al. Multidrug-resistant Shigella sonnei carrying the plasmid-mediated mcr-1 gene in China. Int. J. Antimicrob. Agents 2018, 52, 14–21. [Google Scholar] [CrossRef]
- Li, J.; Li, B.; Ni, Y.; Sun, J. Molecular characterization of the extended-spectrum beta-lactamase (ESBL)-producing Shigella spp. in Shanghai. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 447–451. [Google Scholar] [CrossRef]
- Gonzales, J.C.; Seribelli, A.A.; Gomes, C.N.; dos Prazeres Rodrigues, D.; Campioni, F.; Passaglia, J.; da Silva, P.; Falcão, J.P. A high number of multidrug-resistant and predominant genetically related cluster of Shigella flexneri strains isolated over 34 years in Brazil. Braz. J. Microbiol. 2020, 51, 1563–1571. [Google Scholar] [CrossRef]
- Abd-Elmeged, G.M.; Khairy, R.M.; Abo-Eloyoon, S.M.; Abdelwahab, S.F. Changing patterns of drug-resistant Shigella isolates in egypt. Microb. Drug Resist. 2015, 21, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Izquierdo, M.; Benavente-Fernández, A.; López-Gómez, J.; Láinez-Ramos-bossini, A.J.; Rodríguez-Camacho, M.; Valero-Ubierna, M.D.C.; Martín-Delosreyes, L.M.; Jiménez-Mejías, E.; Moreno-Roldán, E.; Lardelli-Claret, P.; et al. Prevalence of Multi-Resistant Microorganisms and Antibiotic Stewardship among Hospitalized Patients Living in Residential Care Homes in Spain: A Cross-Sectional Study. Antibiotics 2020, 9, 324. [Google Scholar] [CrossRef] [PubMed]
- Beyene, A.M.; Gezachew, M.; Mengesha, D.; Yousef, A.; Gelaw, B. Prevalence and drug resistance patterns of Gram-negative enteric bacterial pathogens from diarrheic patients in Ethiopia: A systematic review and meta-analysis. PLoS ONE 2022, 17, e0265271. [Google Scholar] [CrossRef] [PubMed]
- Ingle, D.J.; Andersson, P.; Valcanis, M.; Barnden, J.; da Silva, A.G.; Horan, K.A.; Seemann, T.; Easton, M.; Williamson, D.A.; Sherry, N.L.; et al. Prolonged Outbreak of Multidrug-Resistant Shigella sonnei Harboring blaCTX-M-27 in Victoria, Australia. Antimicrob. Agents Chemother. 2020, 64, e01518-20. [Google Scholar] [CrossRef] [PubMed]
- Reuland, E.A.; Overdevest, I.T.M.A.; al Naiemi, N.; Kalpoe, J.S.; Rijnsburger, M.C.; Raadsen, S.A.; Ligtenberg-Burgman, I.; van der Zwaluw, K.W.; Heck, M.; Savelkoul, P.H.M.; et al. High prevalence of ESBL-producing Enterobacteriaceae carriage in Dutch community patients with gastrointestinal complaints. Clin. Microbiol. Infect. 2013, 19, 542–549. [Google Scholar] [CrossRef][Green Version]
- Hajissa, K.; Marzan, M.; Idriss, M.I.; Islam, M.A. Prevalence of drug-resistant tuberculosis in Sudan: A systematic review and meta-analysis. Antibiotics 2021, 10, 932. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Viechtbauer, W. Conducting Meta-Analyses in R with the metafor Package. J. Stat. Softw. 2010, 36, 1–48. [Google Scholar] [CrossRef]
- Huedo-Medina, T.B.; Sánchez-Meca, J.; Marín-Martínez, F.; Botella, J. Assessing heterogeneity in meta-analysis: Q statistic or I2 index? Psychol. Methods 2006, 11, 193–206. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).