Advancements and Future Prospects in Molecular Targeted and siRNA Therapies for Chronic Myeloid Leukemia
Abstract
:1. Introduction
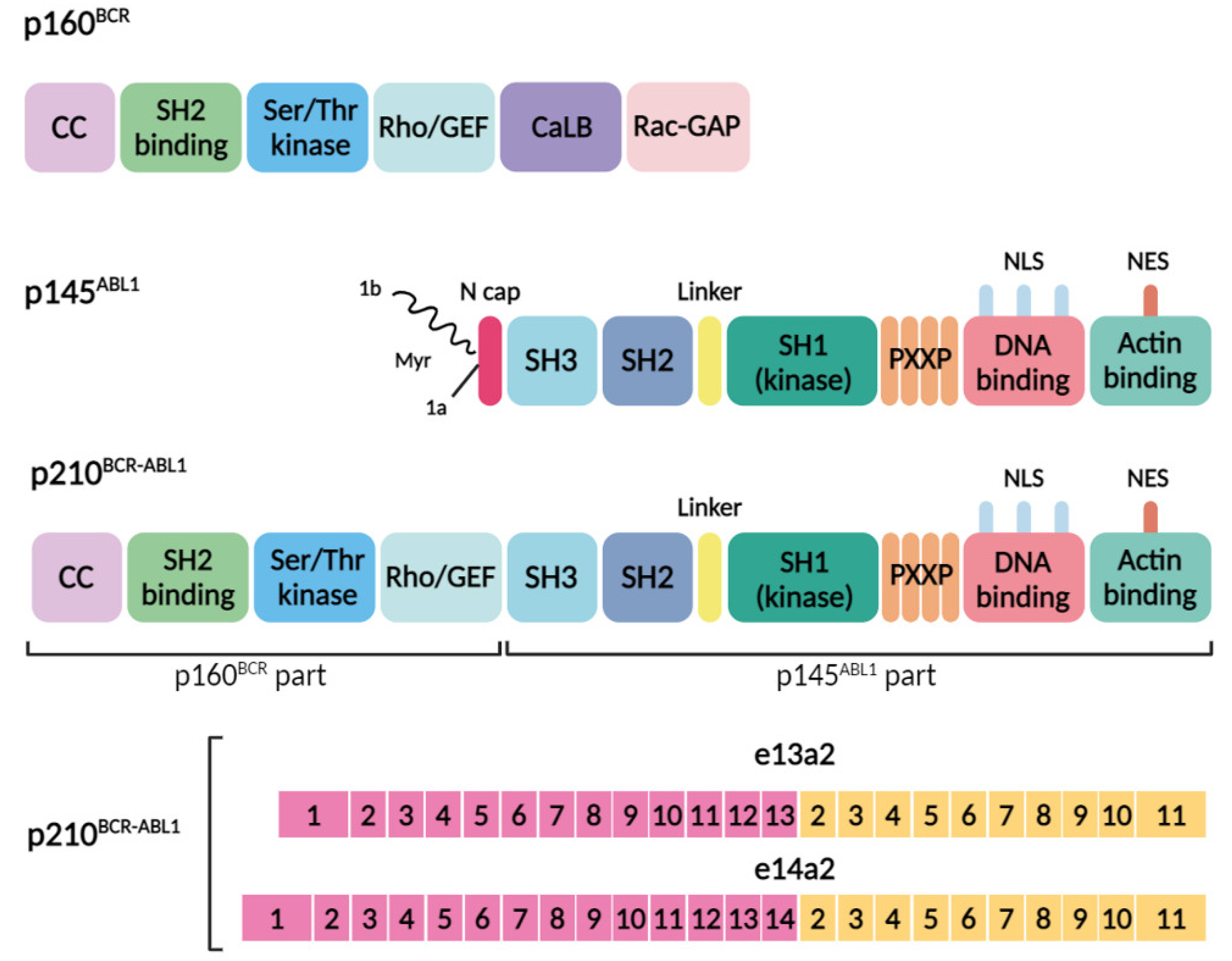
2. The Protein p210 BCR–ABL1 Is a Key Player in the Pathogenesis of CML
- -
- The N-terminal region possesses a coiled-coil oligomerization domain (CC) implicated in binding to proteins containing the Src-homology 2 domain.
- -
- A serine/threonine kinase domain capable of phosphorylating tyrosine residues, including tyrosine 117 (Y177).
- -
- A segment homologous to the Rho guanine nucleotide exchange factor (Rho-GEF).
- -
- A calcium-binding domain (CaLB).
- -
- A region resembling the Rac GTPase-activating protein (Rac-GAP).
3. Strategies for Inhibiting the Chimeric Protein BCR–ABL1
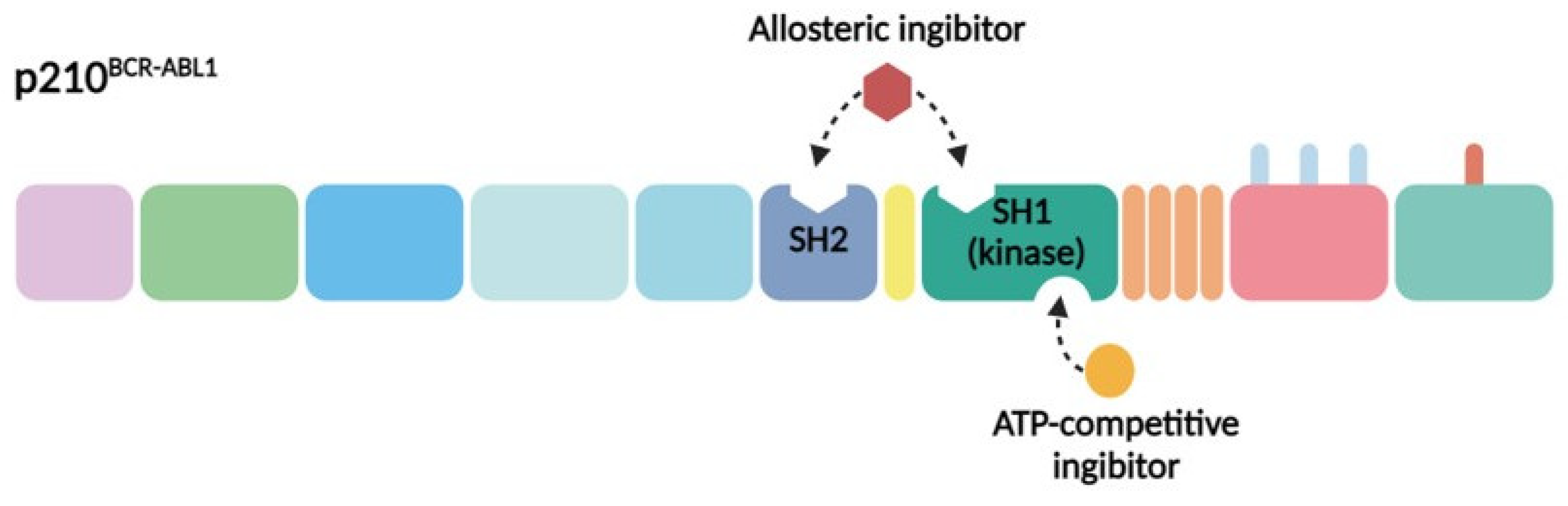
3.1. ATP-Competitive Inhibitors of BCR–ABL1
3.2. Allosteric Inhibitors of BCR–ABL1
3.3. Inhibition of the Chimeric Oncogene BCR–ABL1 Using siRNA
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nowell, P.C.; Hungerford, D.A. A Minute Chromosome in Human Chronic Granulocytic Leukemia. Science 1960, 132, 1497. [Google Scholar]
- Rowley, J.; Golomb, H.; Dougherty, C. 15/17 translocation, a consistent chromosomal change in acute promyelocytic leukaemia. Lancet 1977, 309, 549–550. [Google Scholar] [CrossRef] [PubMed]
- de Klein, A.; van Kessel, A.G.; Grosveld, G.; Bartram, C.R.; Hagemeijer, A.; Bootsma, D.; Spurr, N.K.; Heisterkamp, N.; Groffen, J.; Stephenson, J.R. A cellular oncogene is translocated to the Philadelphia chromosome in chronic myelocytic leukaemia. Nature 1982, 300, 765–767. [Google Scholar] [CrossRef] [PubMed]
- Heisterkamp, N.; Stephenson, J.R.; Groffen, J.; Hansen, P.F.; de Klein, A.; Bartram, C.R.; Grosveld, G. Localization of the c-abl oncogene adjacent to a translocation break point in chronic myelocytic leukaemia. Nature 1983, 306, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Daley, G.Q.; Van Etten, R.A.; Baltimore, D. Induction of chronic myelogenous leukemia in mice by the P210bcr/abl Gene of the Philadelphia Chromosome. Science 1990, 247, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.L.; Burthem, J.; Whetton, A.D. Molecular pathogenesis of chronic myeloid leukaemia. Expert Rev. Mol. Med. 2003, 5, 1–27. [Google Scholar] [CrossRef]
- Chereda, B.; Melo, J.V. Natural course and biology of CML. Ann. Hematol. 2015, 94 (Suppl. S2), 107–121. [Google Scholar] [CrossRef] [PubMed]
- Gunnarsson, N.; Sandin, F.; Höglund, M.; Stenke, L.; Björkholm, M.; Lambe, M.; Olsson-Strömberg, U.; Richter, J.; Själander, A. Population-based assessment of chronic myeloid leukemia in Sweden: Striking increase in survival and prevalence. Eur. J. Haematol. 2016, 97, 387–392. [Google Scholar] [CrossRef]
- Jabbour, E.; Kantarjian, H. Chronic myeloid leukemia: 2018 update on diagnosis, therapy and monitoring. Am. J. Hematol. 2018, 93, 442–459. [Google Scholar] [CrossRef]
- Singh, A.; Trivedi, P.; Jain, N.K. Advances in siRNA delivery in cancer therapy. Artif. Cells Nanomed. Biotechnol. 2018, 46, 274–283. [Google Scholar] [CrossRef]
- Charbe, N.B.; Amnerkar, N.D.; Ramesh, B.; Tambuwala, M.M.; Bakshi, H.A.; Aljabali, A.A.; Khadse, S.C.; Satheeshkumar, R.; Satija, S.; Metha, M.; et al. Small interfering RNA for cancer treatment: Overcoming hurdles in delivery. Acta Pharm. Sin. B 2020, 10, 2075–2109. [Google Scholar] [CrossRef] [PubMed]
- Peiris, M.N.; Li, F.; Donoghue, D.J. BCR: A promiscuous fusion partner in hematopoietic disorders. Oncotarget 2019, 10, 2738–2754. [Google Scholar] [CrossRef] [PubMed]
- Melo, J.V.; Deininger, M.W. Biology of chronic myelogenous leukemia—signaling pathways of initiation and transformation. Hematol. Clin. N. Am. 2004, 18, 545–568. [Google Scholar] [CrossRef] [PubMed]
- Soverini, S.; Mancini, M.; Bavaro, L.; Cavo, M.; Martinelli, G. Chronic myeloid leukemia: The paradigm of targeting oncogenic tyrosine kinase signaling and counteracting resistance for successful cancer therapy. Mol. Cancer 2018, 17, 49. [Google Scholar] [CrossRef] [PubMed]
- Radziwill, G.; Erdmann, R.A.; Margelisch, U.; Moelling, K. The Bcr kinase downregulates Ras signaling by phosphorylating AF-6 and binding to its PDZ domain. Mol. Cell. Biol. 2003, 23, 4663–4672. [Google Scholar] [CrossRef] [PubMed]
- Deininger, M.W.N.; Goldman, J.M.; Melo, J.V. The molecular biology of chronic myeloid leukemia. Blood 2000, 96, 3343–3356. [Google Scholar] [CrossRef] [PubMed]
- Schindler, T.; Bornmann, W.; Pellicena, P.; Miller, W.T.; Clarkson, B.; Kuriyan, J. Structural mechanism for STI-571 inhibition of abelson tyrosine kinase. Science 2000, 289, 1938–1942. [Google Scholar] [CrossRef] [PubMed]
- Cowan-Jacob, S.W.; Guez, V.; Fendrich, G.; Griffin, J.D.; Fabbro, D.; Furet, P.; Liebetanz, J.; Mestan, J.; Manley, P.W. Imatinib (STI571) resistance in chronic myelogenous leukemia: Molecular basis of the underlying mechanisms and potential strategies for treatment. Mini-Rev. Med. Chem. 2004, 4, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Gross, A.W.; Zhang, X.; Ren, R. Bcr-Abl with an SH3 Deletion Retains the Ability to Induce a Myeloproliferative Disease in Mice, yet c-Abl Activated by an SH3 Deletion Induces Only Lymphoid Malignancy. Mol. Cell. Biol. 1999, 19, 6918–6928. [Google Scholar] [CrossRef]
- Wang, J.Y.J. The Capable ABL: What is its biological function? Mol. Cell. Biol. 2014, 34, 1188–1197. [Google Scholar] [CrossRef]
- Zhou, T.; Medeiros, L.J.; Hu, S. Chronic Myeloid Leukemia: Beyond BCR-ABL1. Curr. Hematol. Malig. Rep. 2018, 13, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Melo, J. The diversity of BCR-ABL fusion proteins and their relationship to leukemia phenotype. Blood 1996, 88, 2375–2384. [Google Scholar] [CrossRef] [PubMed]
- Saglio, G.; Pane, F.; Martinelli, G.; Guerrasio, A. BCR/ABL transcripts and leukemia phenotype: An unsolved puzzle. Leuk. Lymphoma 1997, 26, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.-H.; Wood, L.J.; Agarwal, A.; O’Hare, T.; Elsea, C.R.; Griswold, I.J.; Deininger, M.W.; Imamoto, A.; Druker, B.J. A specific need for CRKL in p210BCR-ABL-induced transformation of mouse hematopoietic progenitors. Cancer Res. 2010, 70, 7325–7335. [Google Scholar] [CrossRef]
- Gallipoli, P.; Cook, A.; Rhodes, S.; Hopcroft, L.; Wheadon, H.; Whetton, A.D.; Jørgensen, H.G.; Bhatia, R.; Holyoake, T.L. JAK2/STAT5 inhibition by nilotinib with ruxolitinib contributes to the elimination of CML CD34+ cells in vitro and in vivo. Blood 2014, 124, 1492–1501. [Google Scholar] [CrossRef] [PubMed]
- Reckel, S.; Hamelin, R.; Georgeon, S.; Armand, F.; Jolliet, Q.; Chiappe, D.; Moniatte, M.; Hantschel, O. Differential signaling networks of Bcr–Abl p210 and p190 kinases in leukemia cells defined by functional proteomics. Leukemia 2017, 31, 1502–1512. [Google Scholar] [CrossRef] [PubMed]
- Kurzrock, R.; Kantarjian, H.M.; Druker, B.J.; Talpaz, M. Philadelphia chromosome positive leukemias: From basic mechanisms to molecular therapeutics. Ann. Intern. Med. 2003, 138, 819–830. [Google Scholar] [CrossRef] [PubMed]
- Druker, B.J.; Tamura, S.; Buchdunger, E.; Ohno, S.; Segal, G.M.; Fanning, S.; Zimmermann, J.; Lydon, N.B. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr–Abl positive cells. Nat. Med. 1996, 2, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Corbin, A.S.; La Rosée, P.; Stoffregen, E.P.; Druker, B.J.; Deininger, M.W. Several Bcr-Abl kinase domain mutants associated with imatinib mesylate resistance remain sensitive to imatinib. Blood 2003, 101, 4611–4614. [Google Scholar] [CrossRef]
- Barnes, D.J.; Palaiologou, D.; Panousopoulou, E.; Schultheis, B.; Yong, A.S.; Wong, A.; Pattacini, L.; Goldman, J.M.; Melo, J.V. Bcr-Abl expression levels determine the rate of development of resistance to imatinib mesylate in chronic myeloid leukemia. Cancer Res. 2005, 65, 8912–8919. [Google Scholar] [CrossRef]
- Druker, B.J.; Sawyers, C.L.; Kantarjian, H.; Resta, D.J.; Reese, S.F.; Ford, J.M.; Capdeville, R.; Talpaz, M. Activity of a specific inhibitor of the BCR-ABL tyrosine kinase in the blast crisis of chronic myeloid leukemia and acute lympoblastic leukemia with th. N. Engl. J. Med. 2001, 344, 1038–1042. [Google Scholar] [CrossRef] [PubMed]
- Branford, S.; Rudzki, Z.; Walsh, S.; Parkinson, I.; Grigg, A.; Szer, J.; Taylor, K.; Herrmann, R.; Seymour, J.F.; Arthur, C.; et al. Detection of BCR-ABL mutations in patients with CML treated with imatinib is virtually always accompanied by clinical resistance, and mutations in the ATP phosphate-binding loop (P-loop) are associated with a poor prognosis. Blood 2003, 102, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Soverini, S.; Martinelli, G.; Rosti, G.; Iacobucci, I.; Baccarani, M. Advances in treatment of chronic myeloid leukemia with tyrosine kinase inhibitors: The evolving role of Bcr–Abl mutations and mutational analysis. Pharmacogenomics 2012, 13, 1271–1284. [Google Scholar] [CrossRef] [PubMed]
- Hochhaus, A.; Kreil, S.; Corbin, A.; La Rosée, P.; Müller, M.; Lahaye, T.; Hanfstein, B.; Schoch, C.; Cross, N.; Berger, U.; et al. Molecular and chromosomal mechanisms of resistance to imatinib (STI571) therapy. Leukemia 2002, 16, 2190–2196. [Google Scholar] [CrossRef] [PubMed]
- Mahon, F.-X.; Belloc, F.; Lagarde, V.; Chollet, C.; Moreau-Gaudry, F.; Reiffers, J.; Goldman, J.M.; Melo, J.V. MDR1 gene overexpression confers resistance to imatinib mesylate in leukemia cell line models. Blood 2003, 101, 2368–2373. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Giannoudis, A.; Lane, S.; Williamson, P.; Pirmohamed, M.; Clark, R. Expression of the uptake drug transporter hOCT1 is an important clinical determinant of the response to imatinib in chronic myeloid leukemia. Clin. Pharmacol. Ther. 2008, 83, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, M.W.; Pritchard, J.R.; Leighow, S.M.; Aminov, B.I.; Beppu, L.; Kim, D.S.; Hodgson, J.G.; Rivera, V.M.; Loeb, L.A.; Radich, J.P. Single-Molecule Sequencing Reveals Patterns of Preexisting Drug Resistance That Suggest Treatment Strategies in Philadelphia-Positive Leukemias. Clin. Cancer Res. 2018, 24, 5321–5334. [Google Scholar] [CrossRef] [PubMed]
- Baccarani, M.; Deininger, M.W.; Rosti, G.; Hochhaus, A.; Soverini, S.; Apperley, J.F.; Cervantes, F.; Clark, R.E.; Cortes, J.E.; Guilhot, F.; et al. European LeukemiaNet recommendations for the management of chronic myeloid leukemia: 2013. Blood 2013, 122, 872–884. [Google Scholar] [CrossRef] [PubMed]
- García-Gutiérrez, V.; Hernández-Boluda, J.C. Tyrosine Kinase Inhibitors Available for Chronic Myeloid Leukemia: Efficacy and Safety. Front. Oncol. 2019, 9, 603. [Google Scholar] [CrossRef]
- Cortes, J.E.; Kantarjian, H.; Shah, N.P.; Bixby, D.; Mauro, M.J.; Flinn, I.; O’Hare, T.; Hu, S.; Narasimhan, N.I.; Rivera, V.M.; et al. Ponatinib in refractory Philadelphia chromosome-positive leukemias. N. Engl. J. Med. 2012, 367, 2075–2088. [Google Scholar] [CrossRef]
- Chan, O.; Talati, C.; Isenalumhe, L.; Shams, S.; Nodzon, L.; Fradley, M.; Sweet, K.; Pinilla-Ibarz, J. Side-effects profile and outcomes of ponatinib in the treatment of chronic myeloid leukemia. Blood Adv. 2020, 4, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Adrián, F.J.; Jahnke, W.; Cowan-Jacob, S.W.; Li, A.G.; Iacob, R.E.; Sim, T.; Powers, J.; Dierks, C.; Sun, F.; et al. Targeting Bcr–Abl by combining allosteric with ATP-binding-site inhibitors. Nature 2010, 463, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Wylie, A.A.; Schoepfer, J.; Jahnke, W.; Cowan-Jacob, S.W.; Loo, A.; Furet, P.; Marzinzik, A.L.; Pelle, X.; Donovan, J.; Zhu, W.; et al. The allosteric inhibitor ABL001 enables dual targeting of BCR–ABL1. Nature 2017, 543, 733–737. [Google Scholar] [CrossRef] [PubMed]
- Réa, D.; Mauro, M.J.; Boquimpani, C.; Minami, Y.; Lomaia, E.; Voloshin, S.; Turkina, A.G.; Kim, D.-W.; Apperley, J.F.; Abdo, A.; et al. A phase 3, open-label, randomized study of asciminib, a STAMP inhibitor, vs bosutinib in CML after 2 or more prior TKIs. Blood 2021, 138, 2031–2041. [Google Scholar] [CrossRef]
- Deeks, E.D. Asciminib: First Approval. Drugs 2022, 82, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Grebien, F.; Hantschel, O.; Wojcik, J.; Kaupe, I.; Kovacic, B.; Wyrzucki, A.M.; Gish, G.D.; Cerny-Reiterer, S.; Koide, A.; Beug, H.; et al. Targeting the SH2-kinase interface in Bcr-Abl inhibits leukemogenesis. Cell 2011, 147, 306–319. [Google Scholar] [CrossRef] [PubMed]
- Wojcik, J.; Lamontanara, A.J.; Grabe, G.; Koide, A.; Akin, L.; Gerig, B.; Hantschel, O.; Koide, S. Allosteric Inhibition of Bcr-Abl Kinase by High Affinity Monobody Inhibitors Directed to the Src Homology 2 (SH2)-Kinase Interface. J. Biol. Chem. 2016, 291, 8836–8847. [Google Scholar] [CrossRef] [PubMed]
- Zhelev, Z.; Bakalova, R.; Ohba, H.; Ewis, A.; Ishikawa, M.; Shinohara, Y.; Baba, Y. Suppression of Bcr-Abl synthesis by siRNAs or tyrosine kinase activity by Glivec alters different oncogenes, apoptotic/antiapoptotic genes and cell proliferation factors (microarray study). FEBS Lett. 2004, 570, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Baker, B.E.; Kestler, D.P.; Ichiki, A.T. Effects of siRNAs in combination with Gleevec on K-562 cell proliferation and Bcr-Abl expression. J. Biomed. Sci. 2006, 13, 499–507. [Google Scholar] [CrossRef]
- Bakalova, R. RNA interference—About the reality to be exploited in cancer therapy. Methods Find. Exp. Clin. Pharmacol. 2007, 29, 417–421. [Google Scholar] [CrossRef]
- Vysochinskaya, V.; Zabrodskaya, Y.; Dovbysh, O.; Emelyanov, A.; Klimenko, V.; Knyazev, N.; Terterov, I.; Egorova, M.; Bogdanov, A.; Maslov, M.; et al. Cell-penetrating peptide and cationic liposomes mediated siRNA delivery to arrest growth of chronic myeloid leukemia cells in vitro. Biochimie 2024, 221, 1–12. [Google Scholar] [CrossRef]
- Scherr, M.; Battmer, K.; Schultheis, B.; Ganser, A.; Eder, M. Stable RNA interference (RNAi) as an option for anti-bcr-abl therapy. Gene Ther. 2005, 12, 12–21. [Google Scholar] [CrossRef]
- Merkerova, M.; Klamova, H.; Brdicka, R.; Bruchova, H. Targeting of gene expression by siRNA in CML primary cells. Mol. Biol. Rep. 2007, 34, 27–33. [Google Scholar] [CrossRef]
- Li, M.-J.; McMahon, R.; Snyder, D.S.; Yee, J.-K.; Rossi, J.J. Specific killing of Ph+ chronic myeloid leukemia cells by a lentiviral vector-delivered anti-bcr/Abl small hairpin RNA. Oligonucleotides 2003, 13, 401–409. [Google Scholar] [CrossRef]
- Arthanari, Y.; Pluen, A.; Rajendran, R.; Aojula, H.; Demonacos, C. Delivery of therapeutic shRNA and siRNA by Tat fusion peptide targeting bcr–abl fusion gene in Chronic Myeloid Leukemia cells. J. Control. Release 2010, 145, 272–280. [Google Scholar] [CrossRef]
- Freire, J.M.; de Figueiredo, I.R.; Valle, J.; Veiga, A.S.; Andreu, D.; Enguita, F.J.; Castanho, M.A. siRNA-cell-penetrating peptides complexes as a combinatorial therapy against chronic myeloid leukemia using BV173 cell line as model. J. Control. Release 2017, 245, 127–136. [Google Scholar] [CrossRef]
- Shinkai, Y.; Kashihara, S.; Minematsu, G.; Fujii, H.; Naemura, M.; Kotake, Y.; Morita, Y.; Ohnuki, K.; Fokina, A.A.; Stetsenko, D.A.; et al. Silencing of BCR/ABL Chimeric Gene in Human Chronic Myelogenous Leukemia Cell Line K562 by siRNA-Nuclear Export Signal Peptide Conjugates. Nucleic Acid Ther. 2017, 27, 168–175. [Google Scholar] [CrossRef]
- Kc, R.; Thapa, B.; Ubeda, A.; Jiang, X.; Uludağ, H. BCR-AblSilencing by siRNA: A Potent Approach to Sensitize Chronic Myeloid Leukemia Cells to Tyrosine Kinase Inhibitor Therapy. Stem Cells Dev. 2019, 28, 734–744. [Google Scholar] [CrossRef]
- Remant, K.; Thapa, B.; Valencia-Serna, J.; Domun, S.S.; Dimitroff, C.; Jiang, X.; Uludağ, H. Cholesterol grafted cationic lipopolymers: Potential siRNA carriers for selective chronic myeloid leukemia therapy. J. Biomed. Mater. Res. Part A 2020, 108, 565–580. [Google Scholar] [CrossRef]
- Valencia-Serna, J.; Aliabadi, H.M.; Manfrin, A.; Mohseni, M.; Jiang, X.; Uludag, H. siRNA/lipopolymer nanoparticles to arrest growth of chronic myeloid leukemia cells in vitro and in vivo. Eur. J. Pharm. Biopharm. 2018, 130, 66–70. [Google Scholar] [CrossRef]
- Valencia-Serna, J.; Kucharski, C.; Chen, M.; Kc, R.; Jiang, X.; Brandwein, J.; Uludağ, H. siRNA-mediated BCR-ABL silencing in primary chronic myeloid leukemia cells using lipopolymers. J. Control. Release 2019, 310, 141–154. [Google Scholar] [CrossRef]
- Koldehoff, M.; Kordelas, L.; Beelen, D.W.; Elmaagacli, A.H. Small interfering RNA against BCR-ABL transcripts sensitize mutated T315I cells to nilotinib. Haematologica 2010, 95, 388–397. [Google Scholar] [CrossRef]
- Scherr, M.; Battmer, K.; Winkler, T.; Heidenreich, O.; Ganser, A.; Eder, M. Specific inhibition of bcr-abl gene expression by small interfering RNA. Blood 2003, 101, 1566–1569. [Google Scholar] [CrossRef]
- Elmaagacli, A.H.; Koldehoff, M.; Peceny, R.; Klein-Hitpass, L.; Ottinger, H.; Beelen, D.W.; Opalka, B. WT1 and BCR-ABL specific small interfering RNA have additive effects in the induction of apoptosis in leukemic cells. Haematologica 2005, 90, 326–334. [Google Scholar]
- Koldehoff, M.; Zakrzewski, J.L.; Beelen, D.W.; Elmaagacli, A.H. Additive antileukemia effects by GFI1B- and BCR–ABL-specific siRNA in advanced phase chronic myeloid leukemic cells. Cancer Gene Ther. 2013, 20, 421–427. [Google Scholar] [CrossRef]
- Kaymaz, B.T.; Selvi, N.; Gündüz, C.; Aktan, Ç.; Dalmızrak, A.; Saydam, G.; Kosova, B. Repression of STAT3, STAT5A, and STAT5B expressions in chronic myelogenous leukemia cell line K–562 with unmodified or chemically modified siRNAs and induction of apoptosis. Ann. Hematol. 2013, 92, 151–162. [Google Scholar] [CrossRef]
- Heidel, F.H.; Bullinger, L.; Feng, Z.; Wang, Z.; Neff, T.A.; Stein, L.; Kalaitzidis, D.; Lane, S.W.; Armstrong, S.A. Genetic and pharmacologic inhibition of β-catenin targets imatinib-resistant leukemia stem cells in CML. Cell Stem Cell 2012, 10, 412–424. [Google Scholar] [CrossRef]
- Minami, Y.; Stuart, S.A.; Ikawa, T.; Jiang, Y.; Banno, A.; Hunton, I.C.; Young, D.J.; Naoe, T.; Murre, C.; Jamieson, C.H.M.; et al. BCR-ABL-transformed GMP as myeloid leukemic stem cells. Proc. Natl. Acad. Sci. USA 2008, 105, 17967–17972. [Google Scholar] [CrossRef]
- Zhang, B.; Li, M.; McDonald, T.; Holyoake, T.L.; Moon, R.T.; Campana, D.; Shultz, L.; Bhatia, R. Microenvironmental protection of CML stem and progenitor cells from tyrosine kinase inhibitors through N-cadherin and Wnt–β-catenin signaling. Blood 2013, 121, 1824–1838. [Google Scholar] [CrossRef]
- Zhou, H.; Mak, P.Y.; Mu, H.; Mak, D.H.; Zeng, Z.; Cortes, J.; Liu, Q.; Andreeff, M.; Carter, B.Z. Combined inhibition of β-catenin and Bcr–Abl synergistically targets tyrosine kinase inhibitor-resistant blast crisis chronic myeloid leukemia blasts and progenitors in vitro and in vivo. Leukemia 2017, 31, 2065–2074. [Google Scholar] [CrossRef]
- Yao, R.; Feng, W.-T.; Xu, L.-J.; Zhong, X.-M.; Liu, H.; Sun, Y.; Zhou, L.-L. DUXAP10 regulates proliferation and apoptosis of chronic myeloid leukemia via PTEN pathway. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 4934–4940. [Google Scholar] [CrossRef]
- Li, L.; Qi, Y.; Ma, X.; Xiong, G.; Wang, L.; Bao, C. TRIM22 knockdown suppresses chronic myeloid leukemia via inhibiting PI3K/Akt/mTOR signaling pathway. Cell Biol. Int. 2018, 42, 1192–1199. [Google Scholar] [CrossRef]
- Koldehoff, M.; Elmaagacli, A.H. Therapeutic targeting of gene expression by siRNAs directed against BCR-ABL transcripts in a patient with imatinib-resistant chronic myeloid leukemia. Methods Mol. Biol. 2009, 487, 451–466. [Google Scholar] [CrossRef]
- Jyotsana, N.; Sharma, A.; Chaturvedi, A.; Budida, R.; Scherr, M.; Kuchenbauer, F.; Lindner, R.; Noyan, F.; Sühs, K.-W.; Stangel, M.; et al. Lipid nanoparticle-mediated siRNA delivery for safe targeting of human CML in vivo. Ann. Hematol. 2019, 98, 1905–1918. [Google Scholar] [CrossRef]
- Hehlmann, R.; Berger, U.; Pfirrmann, M.; Hochhaus, A.; Metzgeroth, G.; Maywald, O.; Hasford, J.; Reiter, A.; Hossfeld, D.K.; Kolb, H.-J.; et al. Randomized comparison of interferon alpha and hydroxyurea with hydroxyurea monotherapy in chronic myeloid leukemia (CML-study II): Prolongation of survival by the combination of interferon alpha and hydroxyurea. Leukemia 2003, 17, 1529–1537. [Google Scholar] [CrossRef]
- Hughes, T.P.; Ross, D.M. Moving treatment-free remission into mainstream clinical practice in CML. Blood 2016, 128, 17–23. [Google Scholar] [CrossRef]
- Mahon, F.-X. Treatment-free remission in CML: Who, how, and why? Hematol. Am. Soc. Hematol. Educ. Program 2017, 2017, 102–109. [Google Scholar] [CrossRef]
- Harrington, P.; Radia, D.; de Lavallade, H. What are the considerations for tyrosine kinase inhibitor discontinuation in chronic-phase chronic myeloid leukemia? Expert Rev. Hematol. 2020, 13, 213–222. [Google Scholar] [CrossRef]

| Drug | Pharmacological Strategy for Inhibiting the BCR–ABL1 | Mechanism of Action | Activity against T315I Mutations | Clinical Status |
|---|---|---|---|---|
| First-line treatment | ||||
| Imatinib (Gleevec; Novartis) | ATP-competitive inhibitor | first generation TKI | no | FDA approved |
| Dasatinib (Sprycel; Bristol Myers Squibb) | ATP-competitive inhibitor | second-generation TKI | no | FDA approved |
| Nilotinib (Tasigna; Novartis) | ATP-competitive inhibitor | second-generation TKI | no | FDA approved |
| Bosutinib (Bosulif; Pfizer) | ATP-competitive inhibitor | second-generation TKI | no | FDA approved |
| Ponatinib (Iclusig; Takeda) | ATP-competitive inhibitors | third-generation TKI | yes | FDA approved |
| Asciminib (Scemblix; Novartis) | Allosteric inhibitor | targeting the ABL myristoyl pocket (STAMP) | yes | FDA approved |
| Emerging Therapies | ||||
| Radotinib | ATP-competitive inhibitors | second-generation TKI | no | NCT03722420 |
| Flumatinib | ATP-competitive inhibitors | second-generation TKI | no | NCT02204644 |
| Olverembatinib (HQP1351) | ATP-competitive inhibitors | third-generation TKI | yes | NCT05594758 |
| Vodobatinib (K0706) | ATP-competitive inhibitors | third-generation TKI | yes | NCT02629692 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vysochinskaya, V.; Dovbysh, O.; Gorshkov, A.; Brodskaia, A.; Dubina, M.; Vasin, A.; Zabrodskaya, Y. Advancements and Future Prospects in Molecular Targeted and siRNA Therapies for Chronic Myeloid Leukemia. Biomolecules 2024, 14, 644. https://doi.org/10.3390/biom14060644
Vysochinskaya V, Dovbysh O, Gorshkov A, Brodskaia A, Dubina M, Vasin A, Zabrodskaya Y. Advancements and Future Prospects in Molecular Targeted and siRNA Therapies for Chronic Myeloid Leukemia. Biomolecules. 2024; 14(6):644. https://doi.org/10.3390/biom14060644
Chicago/Turabian StyleVysochinskaya, Vera, Olesya Dovbysh, Andrey Gorshkov, Alexandra Brodskaia, Michael Dubina, Andrey Vasin, and Yana Zabrodskaya. 2024. "Advancements and Future Prospects in Molecular Targeted and siRNA Therapies for Chronic Myeloid Leukemia" Biomolecules 14, no. 6: 644. https://doi.org/10.3390/biom14060644






