A Leaf Disc Assay for Evaluating the Response of Tea (Camellia sinensis) to PEG-Induced Osmotic Stress and Protective Effects of Azoxystrobin against Drought
Abstract
:1. Introduction
2. Results
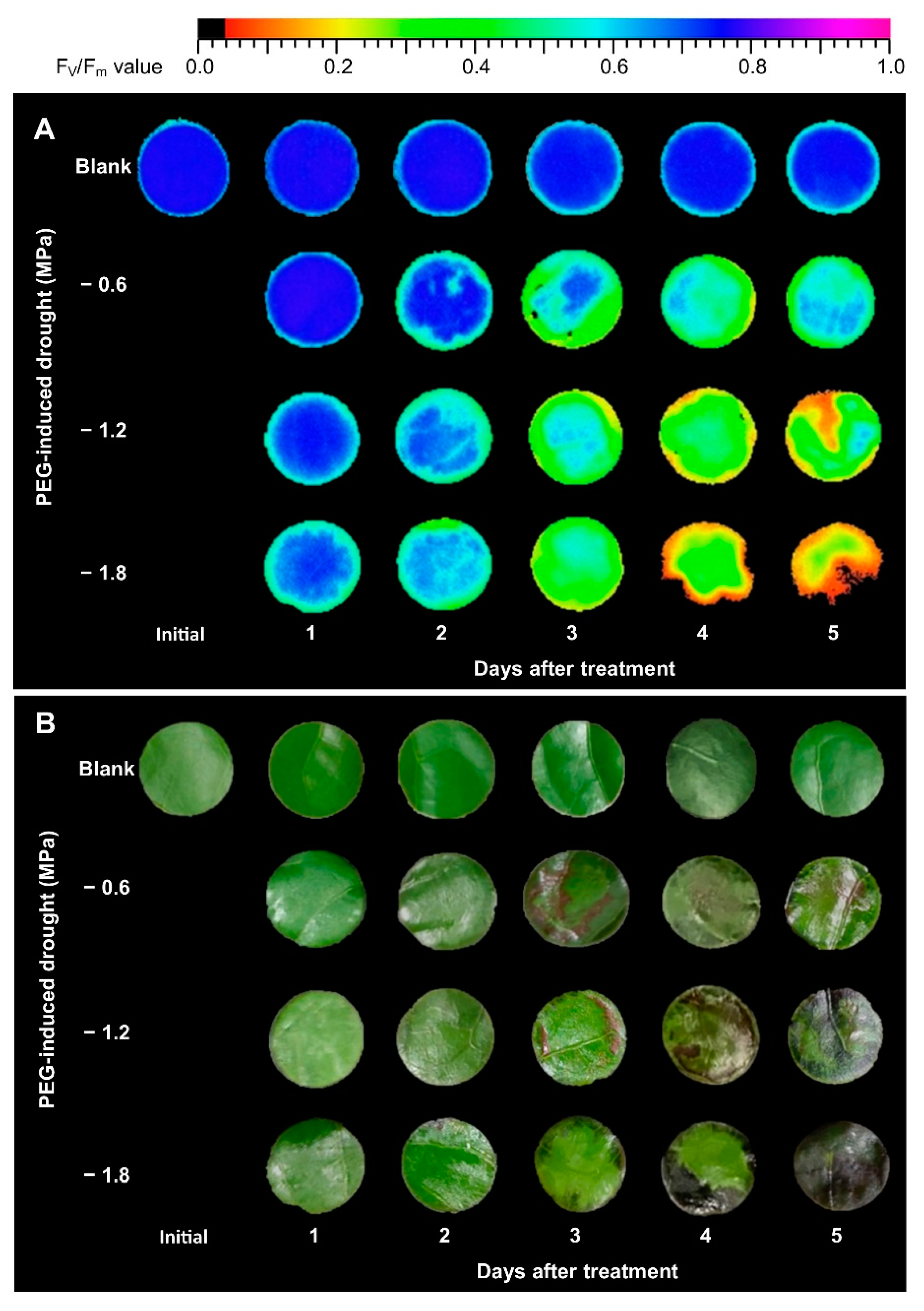
2.1. Chlorophyll Fluorescence in Leaf Discs Collected from PEG-Induced Osmotic Stress
2.2. Photosynthetic Pigments in Leaf Discs Collected from PEG-Induced Osmotic Stress
2.3. Antioxidant Status in Leaf Discs Collected from PEG-Induced Osmotic Stress
2.4. Chlorophyll Fluorescence in Leaf Discs of Exogenous AZ Experiment
2.5. Photosynthetic Pigments in Leaf Discs of Exogenous AZ Experiment
2.6. Antioxidant Status in Leaf Discs of Exogenous AZ Experiment
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Treatments
4.2. Measurements of Chlorophyll Fluorescence
4.3. Determination of Photosynthetic Pigments
4.4. Estimation of Total Phenols
4.5. Antioxidant Capacity Assays
4.6. Determination of Lipid Peroxidation
4.7. Measurement of Ascorbate Peroxidase Activity
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, Q.; Li, T.; Wang, Q.; LeCompte, J.; Harkess, R.L.; Bi, G. Screening Tea Cultivars for Novel Climates: Plant Growth and Leaf Quality of Camellia sinensis Cultivars Grown in Mississippi, United States. Front. Plant Sci. 2020, 11, 280. [Google Scholar] [CrossRef]
- Food and Agriculture Organization Corporate Statistical Database (FAOSTAT). Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 6 March 2021).
- Wu, C.T.; Yang, S.S. A report on the regional trials of the two new tea varieties TTES No.12 and TTES No.13 registered in 1981. Taiwan Tea Res. Bull. 1982, 1, 1–14. [Google Scholar]
- Gunathilaka, R.D.; Smart, J.C.; Fleming, C.M. The impact of changing climate on perennial crops: The case of tea production in Sri Lanka. Clim. Chang. 2017, 140, 577–592. [Google Scholar] [CrossRef]
- Qian, W.; Hu, J.; Zhang, X.; Zhao, L.; Wang, Y.; Ding, Z. Response of tea plants to drought stress. In Stress Physiology of Tea in the Face of Climate Change; Han, W.Y., Li, X., Ahammed, G., Eds.; Springer: Singapore, 2018; pp. 63–81. [Google Scholar]
- Joyce, S.M.; Cassells, A.C.; Jain, S.M. Stress and aberrant phenotypes in vitro culture. Plant Cell Tissue Organ Cult. 2003, 74, 103–121. [Google Scholar] [CrossRef]
- Carr, M.K.V. Irrigating seedling tea in Southern Tanzania: Effects on total yields, distribution of yield and water use. J. Agric. Sci. 1974, 83, 363–378. [Google Scholar] [CrossRef]
- Damayanthi, M.M.N.; Mohotti, A.J.; Nissanka, S.P. Comparison of tolerant ability of mature field grown tea (Camellia sinensis L.) cultivars exposed to a drought stress in Passara Area. Trop. Agric. Res. 2010, 22, 66–75. [Google Scholar] [CrossRef] [Green Version]
- Ho, C.H.; Lur, H.S.; Yao, M.H.; Liao, F.C.; Lin, Y.T.; Yagi, N.; Lu, H.J. The impact on food security and future adaptation under climate variation: A case study of Taiwan’s agriculture and fisheries. Mitig. Adapt. Strateg. Glob. Chang. 2018, 23, 311–347. [Google Scholar] [CrossRef]
- Berlett, B.S.; Stadtman, E.R. Protein oxidation in aging, disease, and oxidative stress. J. Biol. Chem. 1997, 272, 20313–20316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sairam, R.K.; Srivastava, G.C.; Agarwal, S.; Meena, R.C. Differences in antioxidant activity in response to salinity stress in tolerant and susceptible wheat genotypes. Biol. Plant. 2005, 49, 85. [Google Scholar] [CrossRef]
- Li, J.; Yang, Y.; Sun, K.; Chen, Y.; Chen, X.; Li, X. Exogenous melatonin enhances cold, salt and drought stress tolerance by improving antioxidant defense in tea plant (Camellia sinensis (L.) O. Kuntze). Molecules 2019, 24, 1826. [Google Scholar] [CrossRef] [Green Version]
- Zhou, L.; Xu, H.; Mischke, S.; Meinhardt, L.W.; Zhang, D.; Zhu, X.; Li, X.; Fang, W. Exogenous abscisic acid significantly affects proteome in tea plant (Camellia sinensis) exposed to drought stress. Hortic. Res. 2014, 1, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Upadhyaya, H.; Dutta, B.K.; Sahoo, L.; Panda, S.K. Comparative effect of Ca, K, Mn and B on post-drought stress recovery in tea (Camellia sinensis (L.) O Kuntze). Am. J. Plant Sci. 2012, 3, 443. [Google Scholar] [CrossRef] [Green Version]
- Debona, D.; Rodrigues, F.A. A strobilurin fungicide relieves Bipolaris oryzae-induced oxidative stress in rice. J. Plant Pathol. 2016, 164, 571–581. [Google Scholar]
- Nason, M.A.; Farrar, J.; Bartlett, D. Strobilurin fungicides induce changes in photosynthetic gas exchange that do not improve water use efficiency of plants grown under conditions of water stress. Pest Manag. Sci. 2007, 63, 1191–1200. [Google Scholar] [CrossRef] [PubMed]
- Bertelsen, J.R.; De Neergaard, E.; Smedegaard-Petersen, V. Fungicidal effects of azoxystrobin and epoxiconazole on phyllosphere fungi, senescence and yield of winter wheat. Plant Pathol. 2001, 50, 190–205. [Google Scholar] [CrossRef]
- Cromey, M.G.; Butler, R.C.; Mace, M.A.; Cole, A.L.J. Effects of the fungicides azoxystrobin and tebuconazole on Didymella exitialis, leaf senescence and grain yield in wheat. Crop Prot. 2004, 23, 1019–1030. [Google Scholar] [CrossRef]
- Wu, Y.X.; von Tiedemann, A. Physiological effects of azoxystrobin and epoxiconazole on senescence and the oxidative status of wheat. Pestic. Biochem. Physiol. 2001, 71, 1–10. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Zhang, X.; Chen, C.J.; Zhou, M.G.; Wang, H.C. Effects of fungicides JS399-19, azoxystrobin, tebuconazloe, and carbendazim on the physiological and biochemical indices and grain yield of winter wheat. Pestic. Biochem. Physiol. 2010, 98, 151–157. [Google Scholar] [CrossRef]
- Barányiová, I.; Klem, K. Effect of application of growth regulators on the physiological and yield parameters of winter wheat under water deficit. Plant Soil Environ. 2016, 62, 114–120. [Google Scholar] [CrossRef] [Green Version]
- Ruske, R.E.; Gooding, M.J.; Jones, S.A. The effects of triazole and strobilurin fungicide programmes on nitrogen uptake, partitioning, remobilization and grain N accumulation in winter wheat cultivars. J. Agric. Sci. 2003, 140, 395–407. [Google Scholar] [CrossRef]
- Bonasia, A.; Conversa, G.; Lazzizera, C.; Elia, A. Pre-harvest nitrogen and azoxystrobin application enhances postharvest shelf-life in butterhead lettuce. Postharvest Biol. Technol. 2013, 85, 67–76. [Google Scholar] [CrossRef]
- Ali, A.A.; Desoky, E.S.M.; Rady, M.M. Application of azoxystrobin fungicide improves drought tolerance in tomato, via enhancing physio-biochemical and anatomical feature. Nat. Sci. 2019, 76, 35. [Google Scholar] [CrossRef]
- Giuliani, M.M.; Nardella, E.; Gatta, G.; De Caro, A.; Quitadamo, M. Processing tomato cultivated under water deficit conditions: The effect of azoxystrobin. Acta Hortic. 2011, 914, 287–294. [Google Scholar] [CrossRef]
- Joseph, R.S.I. Metabolism of azoxystrobin in plants and animals. In Pesticide Chemistry and Bioscience: The Food-Environment Challenge, 1st ed.; Brooks, G.T., Roberts, T., Eds.; Royal Society of Chemistry: Cambridge, UK, 1999; pp. 265–278. [Google Scholar]
- Woo, N.S.; Badger, M.R.; Pogson, B.J. A rapid, non-invasive procedure for quantitative assessment of drought survival using chlorophyll fluorescence. Plant Methods 2008, 4, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, C.; Sun, Y.; Tang, Y.; Zhang, M. Effect of water stress on chlorophyll fluorescence in leaves of tea plant (Camellia sinensis). Chin. J. Eco-Agric. 2009, 17, 560–564. (In Chinese) [Google Scholar] [CrossRef]
- Li, R.H.; Guo, P.G.; Michael, B.; Stefania, G.; Salvatore, C. Evaluation of chlorophyll content and fluorescence parameters as indicators of drought tolerance in barley. Agric. Sci. China 2006, 5, 751–757. [Google Scholar] [CrossRef]
- Mishra, K.B.; Lannacone, R.; Petrozza, A.; Mishra, A.; Armentano, N.; La Vecchia, G.; Trtílekd, M.; Cellini, F.; Nedbal, L. Engineered drought tolerance in tomato plants is reflected in chlorophyll fluorescence emission. Plant Sci. 2012, 182, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowski, P.; Pawluśkiewicz, B.; Baczewska, A.H.; Oglecki, P.; Kalaji, H. Chlorophyll a fluorescence of perennial ryegrass (Lolium perenne L.) varieties under long term exposure to shade. Zemdirbyste 2015, 102, 305–312. [Google Scholar] [CrossRef] [Green Version]
- Dąbrowski, P.; Baczewska, A.; Pawluśkiewicz, B.; Paunov, M.; Alexantrov, V.; Goltsev, V.; Kalaji, M. Prompt chlorophyll a fluorescence as a rapid tool for diagnostic changes in PSII structure inhibited by salt stress in Perennial ryegrass. J. Photochem. Photobiol. B Biol. 2016, 157, 22–31. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Račková, L.; Paganová, V.; Swoczyna, T.; Rusinowski, S.; Sitko, K. Can chlorophyll-a fluorescence parameters be used as bio-indicators to distinguish between drought and salinity stress in Tilia cordata Mill? Environ. Exp. Bot. 2018, 152, 149–157. [Google Scholar] [CrossRef]
- Faseela, P.; Sinisha, A.; Brestic, M.; Puthur, J. Special issue in honour of Prof. Reto, J. Strasser–Chlorophyll a fluorescence parameters as indicators of a particular abiotic stress in rice. Photosynthetica 2019, 57, 108–115. [Google Scholar]
- Dąbrowski, P.; Kalaji, M.; Baczewska, A.; Pawluśkiewicz, B.; Mastalerczuk, G.; Borawska-Jarmułowicz, B.; Paunov, M.; Goltsev, V. Delayed chlorophyll a fluorescence, MR 820, and gas exchange changes in perennial ryegrass under salt stress. J. Lumin. 2017, 183, 322–333. [Google Scholar] [CrossRef]
- Kalaji, H.; Rastogi, A.; Živčák, M.; Brestic, M.; Daszkowska-Golec, A.; Sitko, K.; Alsharafa, K.; Lotfi, R.; Stypiński, P.; Samborska, I. Prompt chlorophyll fluorescence as a tool for crop phenotyping: An example of barley landraces exposed to various abiotic stress factors. Photosynthetica 2018, 56, 953–961. [Google Scholar] [CrossRef] [Green Version]
- Dąbrowski, P.; Baczewska-Dąbrowska, A.H.; Kalaji, H.M.; Goltsev, V.; Paunov, M.; Rapacz, M.; Wójcik-Jagła, M.; Pawluśkiewicz, B.; Bąba, W.; Brestic, M. Exploration of chlorophyll a fluorescence and plant gas exchange parameters as indicators of drought tolerance in perennial ryegrass. Sensors 2019, 19, 2736. [Google Scholar] [CrossRef] [Green Version]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Lan, C.Y.; Lin, K.H.; Huang, W.D.; Chen, C.C. Physiological effects of the fungicide azoxystrobin on wheat seedlings under extreme heat. Not. Bot. Horti Agrobot. 2019, 47, 683–690. [Google Scholar] [CrossRef] [Green Version]
- Sayar, R.; Bchini, H.; Mosbahi, M.; Khemira, H. Response of durum wheat (Triticum durum Desf.) growth to salt and drought stresses. Czech J. Genet. Plant Breed. 2010, 46, 54–63. [Google Scholar] [CrossRef] [Green Version]
- Reza Yousefi, A.; Rashidi, S.; Moradi, P.; Mastinu, A. Germination and seedling growth responses of Zygophyllum fabago, Salsola kali L. and Atriplex canescens to PEG-induced drought stress. Environments 2020, 7, 107. [Google Scholar] [CrossRef]
- Gibon, Y.; Sulpice, R.; Larher, F. Proline accumulation in canola leaf discs subjected to osmotic stress is related to the loss of chlorophylls and to the decrease of mitochondrial activity. Physiol. Plant. 2008, 110, 469–476. [Google Scholar] [CrossRef]
- Trotel, P.; Bouchereau, A.; Niogret, M.F.; Larher, F. The fate of osmo-accumulated proline in leaf discs of rape (Brassica napus L.) incubated in a medium of low osmolarity. Plant Sci. 1996, 118, 31–45. [Google Scholar] [CrossRef]
- Sandanam, S.; Gee, G.W.; Mapa, R.B. Leaf water diffusion resistance in clonal tea (Camellia sinensis L.): Effects of water stress, leaf age and clones. Ann. Bot. 1981, 47, 339–349. [Google Scholar] [CrossRef]
- Singh, K.; Kumar, S.; Ahuja, P.S. Differential expression of Histone H3 gene in tea (Camellia sinensis (L.) O. Kuntze) suggests its role in growing tissue. Mol. Biol. Rep. 2009, 36, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Varanasi, V.; Perez-Jones, A. A non-destructive leaf disc assay for rapid diagnosis of weed resistance to multiple herbicides. Weed Sci. 2021, 1–32. [Google Scholar] [CrossRef]
- Moustakas, M.; Sperdouli, I.; Kouna, T.; Antonopoulou, C.I.; Therios, I. Exogenous proline induces soluble sugar accumulation and alleviates drought stress effects on photosystem II functioning of Arabidopsis thaliana leaves. Plant Growth Regul. 2011, 65, 315. [Google Scholar] [CrossRef]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant drought stress: Effects, mechanisms and management. Agron. Sustain. Dev. 2009, 29, 185–212. [Google Scholar] [CrossRef] [Green Version]
- Basal, O.; Szabó, A.; Veres, S. Physiology of soybean as affected by PEG-induced drought stress. Curr. Plant Biol. 2020, 100135. [Google Scholar] [CrossRef]
- Li, Q.M.; Liu, B.B.; Wu, Y.; Zou, Z.R. Interactive effects of drought stresses and elevated CO2 concentration on photochemistry efficiency of cucumber seedlings. J. Integr. Plant Biol. 2008, 50, 1307–1317. [Google Scholar] [CrossRef]
- Kura-Hotta, M.; Satoh, K.; Katoh, S. Relationship between photosynthesis and chlorophyll content during leaf senescence of rice seedlings. Plant Cell Physiol. 1987, 28, 1321–1329. [Google Scholar]
- Netto, L.A.; Jayaram, K.M.; Puthur, J.T. Clonal variation of tea (Camellia sinensis (L.) O. Kuntze) in countering water deficiency. Physiol. Mol. Biol. Plants 2010, 16, 359–367. [Google Scholar] [CrossRef] [Green Version]
- Dinç, E.; Ceppi, M.G.; Tóth, S.Z.; Bottka, S.; Schansker, G. The Chl a fluorescence intensity is remarkably insensitive to changes in the chlorophyll content of the leaf as long as the Chl a/b ratio remains unaffected. Biochim. Biophys. Acta Biomembr. 2012, 1817, 770–779. [Google Scholar] [CrossRef] [Green Version]
- Cheruiyot, E.K.; Mumera, L.M.; NG’ETICH, W.K.; Hassanali, A.; Wachira, F. Polyphenols as potential indicators for drought tolerance in tea (Camellia sinensis L.). Biosci. Biotechnol. Biochem. 2007, 71, 2190–2197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lan, C.Y.; Lin, K.H.; Huang, W.D.; Chen, C.C. Protective effects of selenium on wheat seedlings under salt stress. Agronomy 2019, 9, 272. [Google Scholar] [CrossRef] [Green Version]
- Debona, D.; Nascimento, K.J.T.; Gomes, J.G.O.; Aucique-Perez, C.E.; Rodrigues, F.A. Physiological changes promoted by a strobilurin fungicide in the rice-Bipolaris oryzae interaction. Pestic. Biochem. Physiol. 2016, 130, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, I.; Islam, M.S.; Hossen, M.S.; Jahangir, M. The effect of different groups of fungicides in controlling black rot (corticium spp.) disease and yield of tea. J. Glob. Biosci. 2016, 5, 4062–4070. [Google Scholar]
- Michel, B.E.; Kaufmann, M.R. The osmotic potential of polyethylene glycol 6000. Plant Physiol. 1973, 51, 914–916. [Google Scholar] [CrossRef]
- Van Kooten, O.; Snel, J.F. The use of chlorophyll fluorescence nomenclature in plant stress physiology. Photosynth. Res. 1990, 25, 147–150. [Google Scholar] [CrossRef]
- Yang, C.M.; Chang, K.W.; Yin, M.H.; Huang, H.M. Methods for the determination of the chlorophylls and their derivatives. Taiwania 1998, 43, 116–122. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Oyaizu, M. Studies on products of browning reaction. Jpn. J. Nutr. Diet. 1986, 44, 307–315. [Google Scholar] [CrossRef] [Green Version]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar]






| Effect | Regression Coefficient | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Fv/Fm | Y(II) | Y(NO) | Y(NPQ) | Total Chl | Chl a/b | Car | DPPH Scavenging | Reducing Power | Total Phenols | MDA | |
| Intercept | 0.740 *** | 0.223 *** | 0.229 *** | 0.538 *** | 11.513 *** | 2.009 *** | 2.008 *** | 69.911 *** | 281.493 *** | 112.184 *** | 0.285 *** |
| d | 0.059 ** | 0.051 *** | 0.137 *** | −0.204 *** | 4.468 *** | 0.097 *** | 0.494 *** | 50.697 *** | 163.475 *** | 42.046 *** | −0.054 *** |
| D2 | 0.033 ** | - | 0.053 *** | −0.069 *** | 1.848 *** | 0.077*** | 0.229 *** | 20.964 *** | 66.495 *** | 18.553 *** | - |
| t | −0.008 * | −0.023 *** | 0.067 *** | −0.033 * | −0.452 *** | 0.005 NS | 0.098 ** | 5.409 * | 8.513 NS | 7.081 ** | 0.044 *** |
| t2 | - | - | −0.009 *** | 0.007 ** | - | - | −0.023 *** | −1.173 ** | −2.238 ** | −1.241 ** | −0.006 *** |
| dt | 0.051 *** | - | −0.069 *** | 0.063 *** | 0.456 *** | 0.047 *** | 0.108 *** | 4.390 *** | 8.642 *** | 5.547 *** | - |
| Root MSE | 0.037 | 0.033 | 0.051 | 0.052 | 0.613 | 0.034 | 0.119 | 6.815 | 14.879 | 7.774 | 0.028 |
| R2 | 0.936 | 0.715 | 0.914 | 0.828 | 0.940 | 0.902 | 0.909 | 0.915 | 0.942 | 0.883 | 0.743 |
| Adj-R2 | 0.933 | 0.710 | 0.910 | 0.819 | 0.937 | 0.897 | 0.904 | 0.909 | 0.938 | 0.876 | 0.733 |
| AZ Conc. (g L−1) | Treatment | Total Chl (mg g−1 DW) | Chl a/b (mg mg−1) | Car (mg g−1 DW) |
|---|---|---|---|---|
| No AZ | Pre-treat | 9.87 ± 0.43 a | 1.883 ± 0.005 cd | 1.71 ± 0.07 cd |
| Blank | 9.90 ± 0.38 a | 1.912 ± 0.011 ab | 1.95 ± 0.07 a | |
| PEG | 8.90 ± 0.35 cd | 1.807 ± 0.016 f | 1.97 ± 0.07 a | |
| 0.125 | Pre-treat | 9.45 ± 0.41 ab | 1.880 ± 0.012 de | 1.63 ± 0.05 d |
| Blank | 9.80 ± 0.31 a | 1.926 ± 0.007 a | 1.85 ± 0.08 b | |
| PEG | 8.43 ± 0.25 d | 1.790 ± 0.020 f | 1.68 ± 0.03 cd | |
| 1.25 | Pre-treat | 8.84 ± 0.38 cd | 1.861 ± 0.004 e | 1.54 ± 0.07 e |
| Blank | 9.16 ± 0.47 bc | 1.900 ± 0.011 bc | 1.74 ± 0.08 c | |
| PEG | 7.60 ± 0.21 e | 1.761 ± 0.021 g | 1.51 ± 0.05 e |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiu, Y.-C.; Chen, B.-J.; Su, Y.-S.; Huang, W.-D.; Chen, C.-C. A Leaf Disc Assay for Evaluating the Response of Tea (Camellia sinensis) to PEG-Induced Osmotic Stress and Protective Effects of Azoxystrobin against Drought. Plants 2021, 10, 546. https://doi.org/10.3390/plants10030546
Chiu Y-C, Chen B-J, Su Y-S, Huang W-D, Chen C-C. A Leaf Disc Assay for Evaluating the Response of Tea (Camellia sinensis) to PEG-Induced Osmotic Stress and Protective Effects of Azoxystrobin against Drought. Plants. 2021; 10(3):546. https://doi.org/10.3390/plants10030546
Chicago/Turabian StyleChiu, Yu-Chieh, Bo-Jen Chen, Yen-Shuo Su, Wen-Dar Huang, and Chang-Chang Chen. 2021. "A Leaf Disc Assay for Evaluating the Response of Tea (Camellia sinensis) to PEG-Induced Osmotic Stress and Protective Effects of Azoxystrobin against Drought" Plants 10, no. 3: 546. https://doi.org/10.3390/plants10030546








