Essential Oil and Major Non-Volatile Secondary Metabolites from the Leaves of Amazonian Piper subscutatum
Abstract
:1. Introduction
2. Results
2.1. Chemical Analysis of the EO and Hydrolate
2.2. Enantioselective Analysis of the EO
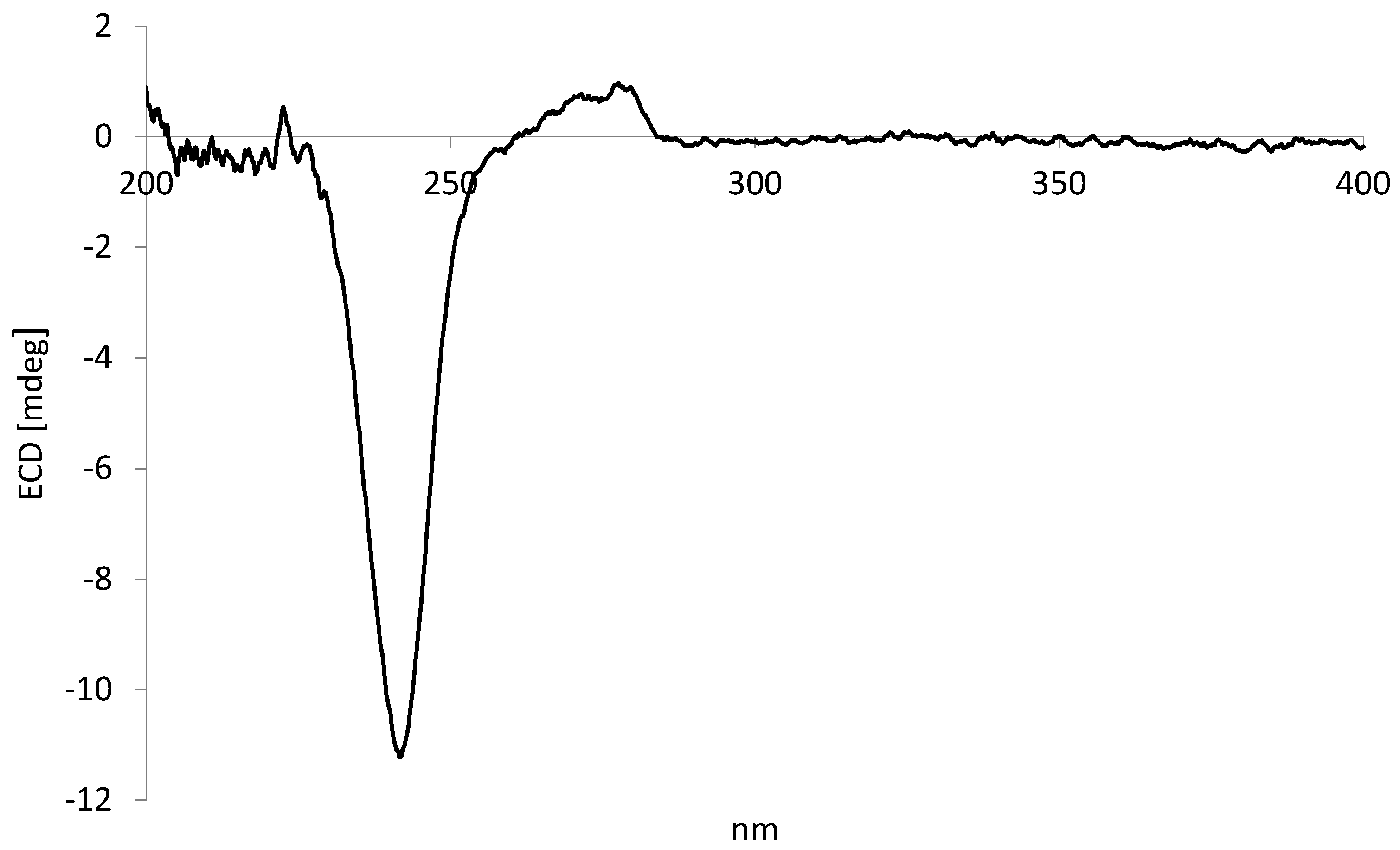
2.3. Lignans from P. Subscutatum Ethyl Acetate Extract
3. Discussion
4. Materials and Methods
4.1. General Information
4.2. Plant Material
4.3. Distillation of the EO and GC Sample Preparation
4.4. GC-MS Qualitative Analyses
4.5. GC-FID Quantitative Analyses
4.6. Enantioselective Analysis of the EO
4.7. Preparation of the Ethyl Acetate Extract
4.8. Separation of the Lignans
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tropicos.org. Missouri Botanical Garden. Available online: https://www.tropicos.org/name/25000956 (accessed on 3 May 2021).
- Megadiverse Countries, UNEP-WCMC. Available online: https://www.biodiversitya-z.org/content/megadiverse-countries (accessed on 3 May 2021).
- Malagón, O.; Ramírez, J.; Andrade, J.; Morocho, V.; Armijos, C.; Gilardoni, G. Phytochemistry and ethnopharmacology of the Ecuadorian flora. A review. Nat. Prod. Commun. 2016, 11, 297–314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Espinosa, S.; Bec, N.; Larroque, C.; Ramírez, J.; Sgorbini, B.; Bicchi, C.; Cumbicus, N.; Gilardoni, G. A novel chemical profile of a selective in vitro cholinergic essential oil from Clinopodium taxifolium (Kunth) Govaerts (Lamiaceae), a native Andean species of Ecuador. Molecules 2021, 26, 45. [Google Scholar] [CrossRef] [PubMed]
- Gilardoni, G.; Montalván, M.; Ortiz, M.; Vinueza, D.; Montesinos, J.V. The flower essential oil of Dalea mutisii Kunth (Fabaceae) from Ecuador: Chemical, enantioselective, and olfactometric analyses. Plants 2020, 9, 1403. [Google Scholar] [CrossRef] [PubMed]
- Gilardoni, G.; Matute, Y.; Ramírez, J. Chemical and enantioselective analysis of the leaf essential oil from Piper coruscans Kunth (Piperaceae), a costal and Amazonian native species of Ecuador. Plants 2020, 9, 791. [Google Scholar] [CrossRef]
- García, J.; Gilardoni, G.; Cumbicus, N.; Morocho, V. Chemical analysis of the essential oil from Siparuna echinata (Kunth) A. DC. (Siparunaceae) of Ecuador and isolation of the rare terpenoid Sipaucin A. Plants 2020, 9, 187. [Google Scholar] [CrossRef] [Green Version]
- Montalván, M.; Peñafiel, M.; Ramirez, J.; Cumbicus, N.; Bec, N.; Larroque, C.; Bicchi, C.; Gilardoni, G. Chemical composition, enantiomeric distribution, and sensory evaluation of the essential oils distilled from the Ecuadorian species Myrcianthes myrsinoides (Kunth) Grifo and Myrcia mollis (Kunth) DC. (Myrtacee). Plants 2019, 8, 511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Espinosa, S.; Bec, N.; Larroque, C.; Ramirez, J.; Sgorbini, B.; Bicchi, C.; Gilardoni, G. Chemical, enantioselective, and sensory analysis of a cholinesterase inhibitor essential oil from Coreopsis triloba S.F. Blake (Asteraceae). Plants 2019, 8, 448. [Google Scholar] [CrossRef] [Green Version]
- Gilardoni, G.; Ramirez, J.; Montalvan, M.; Quinche, W.; León, J.; Benítez, L.; Morocho, V.; Cumbicus, N.; Bicchi, C. Phytochemistry of three Ecuadorian Lamiaceae: Lepechinia heteromorpha (Briq.) Epling, Lepechinia radula (Benth.) Epling and Lepechinia paniculata (Kunth) Epling. Plants 2018, 8, 1. [Google Scholar] [CrossRef] [Green Version]
- Ramirez, J.; Gilardoni, G.; Ramón, E.; Tosi, S.; Picco, A.; Bicchi, C.; Vidari, G. Phytochemical study of the Ecuadorian species Lepechinia mutica (Benth.) Epling and high antifungal activity of carnosol against Pyricularia oryzae. Pharmaceuticals 2018, 11, 33. [Google Scholar] [CrossRef] [Green Version]
- Calva, J.; Bec, N.; Gilardoni, G.; Larroque, C.; Cartuche, L.; Bicchi, C.; Montesinos, J. Acorenone B: AChE and BChE inhibitor as a major compound of the essential oil distilled from the Ecuadorian species Niphogeton dissecta (Benth.) JF Macbr. Pharmaceuticals 2017, 10, 84. [Google Scholar] [CrossRef] [Green Version]
- Ramírez, J.; Gilardoni, G.; Jácome, M.; Montesinos, J.; Rodolfi, M.; Guglielminetti, M.; Cagliero, C.; Bicchi, C.; Vidari, G. Chemical composition, enantiomeric analysis, AEDA sensorial evaluation and antifungal activity of the essential oil from the Ecuadorian plant Lepechinia mutica Benth (Lamiaceae). Chem. Biodivers. 2017, 14, e1700292. [Google Scholar] [CrossRef]
- Chiriboga, X.; Gilardoni, G.; Magnaghi, I.; Vita Finzi, P.; Zanoni, G.; Vidari, G. New Anthracene derivatives from Coussarea macrophylla. J. Nat. Prod. 2003, 66, 905–909. [Google Scholar] [CrossRef]
- Gilardoni, G.; Malagon, O.; Morocho, V.; Negri, R.; Tosi, S.; Guglielminetti, M.; Vidari, G.; Vita Finzi, P. Phytochemical research and antimicrobial activity of Clinopodium nubigenum Kunth (Kuntze) raw extracts. Rev. Bras. Farmacogn. 2011, 21, 850–855. [Google Scholar] [CrossRef] [Green Version]
- Gilardoni, G.; Chiriboga, X.; Finzi, P.V.; Vidari, G. New 3,4-secocycloartane and 3,4-secodammarane triterpenes from the Ecuadorian plant Coussarea macrophylla. Chem. Biodivers. 2015, 12, 946–954. [Google Scholar] [CrossRef]
- Herrera, C.; Morocho, V.; Vidari, G.; Bicchi, C.; Gilardoni, G. Phytochemical investigation of male and female Hedyosmum scabrum (Ruiz & Pav.) Solms leaves from Ecuador. Chem. Biodivers. 2018, 15, e1700423. [Google Scholar]
- Torres-Naranjo, M.; Suárez, A.I.; Gilardoni, G.; Cartuche, L.; Flores, P.; Morocho, V. Chemical constituents of Muehlenbeckia tamnifolia (Kunth) Meisn (Polygonaceae) and its in vitro α-amilase and α-glucosidase inhibitory activities. Molecules 2016, 21, 1461. [Google Scholar] [CrossRef]
- Ramírez, J.; Suarez, A.I.; Bec, N.; Armijos, C.; Gilardoni, G.; Larroque, C.; Vidari, G. Carnosol from Lepechinia mutica and tiliroside from Vallea stipularis: Two promising inhibitors of BuChE. Rev. Bras. Farmacogn. 2018, 28, 559–563. [Google Scholar] [CrossRef]
- Vidari, G.; Abdo, S.; Gilardoni, G.; Ciapessoni, A.; Gusmeroli, M.; Zanoni, G. Fungitoxic metabolites from Erigeron apiculatus. Fitoterapia 2006, 77, 318–320. [Google Scholar] [CrossRef]
- Quílez, A.; Berenguer, B.; Gilardoni, G.; Souccar, C.; De Mendonça, S.; Oliveira, L.F.S.; Martin-Calero, M.J.; Vidari, G. Anti- secretory, anti-inflammatory and anti-Helicobacter pylori activities of several fractions isolated from Piper carpunya Ruiz & Pav. J. Ethnopharmacol. 2010, 128, 583–589. [Google Scholar]
- Council of Europe European Pharmacopoeia, 8th ed.; Council of Europe: Strasbourg, France, 2013; p. 743.
- Durant-Archibold, A.A.; Santana, A.I.; Gupta, M.P. Ethnomedical uses and pharmacological activities of most prevalent species of genus Piper in Panama: A review. J. Ethnopharmacol. 2018, 217, 63–82. [Google Scholar] [CrossRef] [PubMed]
- Xiang, C.P.; Shi, I.N.; Liu, F.F.; Li, H.Z.; Zhang, Y.J.; Yang, C.R.; Xua, M. A survey of the chemical compounds of Piper spp. (Piperaceae) and their biological activities. Nat. Product Commun. 2016, 11, 1403–1408. [Google Scholar]
- Da Silva, J.K.; da Trindade, R.; Alves, N.S.; Figueiredo, P.L.; Guilherme, J.; Maia, S.; Setzer, W.N. Essential oils from neotropical Piper species and their biological activities. Int. J. Mol. Sci. 2017, 18, 2571. [Google Scholar] [CrossRef] [Green Version]
- Salehi, B.; Zakaria, Z.A.; Gyawali, R.; Ibrahim, S.A.; Rajkovic, J.; Shinwari, Z.K.; Khan, T.; Sharifi-Rad, J.; Ozleyen, A.; Turkdonmez, E.; et al. Piper species: A comprehensive review on their phytochemistry, biological activities and applications. Molecules 2019, 24, 1364. [Google Scholar] [CrossRef] [Green Version]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing Corporation: Carol Stream, IL, USA, 2007; ISBN 10-1932633219. [Google Scholar]
- Kundakovic, T.; Fokialakis, N.; Kovacevi, N.; Chinou, I. Essential oil composition of Achillea lingulata and A. umbellate. Flavour Fragr. J. 2007, 22, 184–187. [Google Scholar] [CrossRef]
- Osorio, C.; Alarcon, M.; Moreno, C.; Bonilla, A.; Barrios, J.; Garzon, C.; Duque, C. Characterization of odor-active volatiles in Champa (Campomanesia lineatifolia R. & P.). J. Agric. Food Chem. 2006, 54, 509–516. [Google Scholar] [PubMed]
- Flamini, G.; Tebano, M.; Cioni, P.L.; Bagci, Y.; Dural, H.; Ertugrul, K.; Uysal, T.; Savran, A. A multivariate statistical approach to Centaurea classification using essential oil composition data of some species from Turkey. Plant Syst. Evol. 2006, 261, 217–228. [Google Scholar] [CrossRef]
- Bianchi, F.; Careri, M.; Mangia, A.; Musci, M. Retention indices in the analysis of food aroma volatile compounds in temperature-programmed gas chromatography: Database creation and evaluation of precision and robustness. J. Sep. Sci. 2007, 30, 563–572. [Google Scholar] [CrossRef] [Green Version]
- Babushok, V.I.; Linstrom, P.J.; Zenkevich, I.G. Retention indices for frequently reported compounds of plant essential oils. J. Phys. Chem. Ref. Data 2001, 40, 043101. [Google Scholar] [CrossRef] [Green Version]
- Carrer, R.P.; Vanderlinde, R.; Dutra, S.; Marcon, A.; Echeverrigaray, S. Essential oil variation among Brazilian accessions of Salvia guaranitica L. Flavour Fragr. J. 2007, 22, 430–434. [Google Scholar] [CrossRef]
- Cheng, H.Z.; Kyoung, H.K.; Tae, H.K.; Hyong, J.L. Analysis and characterization of aroma-active compounds of Schizandra chinensis (omija) leaves. J. Sci. Food Agric. 2005, 85, 161–166. [Google Scholar]
- Chanegriha, N.; Baaliouamer, A.; Rolando, C. Polarity changes during capillary gas chromatographic and gas chromatographic–mass spectrometric analysis using serially coupled columns of different natures and temperature programming application to the identification of constituents of essential oils. J. Chromatogr. A 1998, 819, 61–65. [Google Scholar] [CrossRef]
- Demirci, B.; Can Baser, K.H.; Yildiz, B.; Bahçecioglu, Z. Composition of the essential oils of six endemic Salvia spp. from Turkey. Flavour Fragr. J. 2003, 18, 116–121. [Google Scholar] [CrossRef]
- Bisio, A.; Ciarallo, G.; Romussi, G.; Fontana, N.; Mascolo, N.; Capasso, R.; Biscardi, D. Chemical composition of the essential oil from some Salvia species. Phytother. Res. 1998, 12, S117–S120. [Google Scholar] [CrossRef]
- Hachicha, S.F.; Skanji, T.; Barrek, S.; Ghrabi, Z.G.; Zarrouk, H. Composition of the essential oil of Teucrium ramosissimum Desf. (Lamiaceae) from Tunisia. Flavour Fragr. J. 2007, 22, 101–104. [Google Scholar] [CrossRef]
- Huang, Y.T.; Chang, H.S.; Wang, G.J.; Lin, C.H.; Chen, I.S. Secondary metabolites from the roots of Beilschmiedia tsangii and their anti-inflammatory activities. Int. J. Mol. Sci. 2012, 13, 16430–16443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barbosa-Filho, J.M.; Leitao da Cunha, E.V.; Sobral da Silva, M. Complete assignment of the 1H and 13C NMR spectra of some lignoids from Lauraceae. Magn. Reson. Chem. 1998, 36, 929–935. [Google Scholar] [CrossRef]
- Lopes, N.P.; de Almeida Blumenthal, E.E.; Cavalheiro, A.J.; Kato, M.J.; Yoshida, M. Lignans, γ-lactones and propiophenones of Virola surinamensis. Phytochemistry 1996, 43, 1089–1092. [Google Scholar] [CrossRef]
- Martins, R.C.C.; Lago, J.H.G.; Albuquerque, S.; Kato, M.J. Trypanocidal tetrahydrofuran lignans from inflorescences of Piper solmsianum. Phytochemistry 2003, 64, 667–670. [Google Scholar] [CrossRef]
- Ramos, C.S.; Linnert, H.V.; de Moraes, M.M.; do Amaral, J.H.; Yamaguchi, L.F.; Kato, M.J. Configuration and stability of naturally occurring all-cis-tetrahydrofuran lignans from Piper solmsianum. RSC Adv. 2017, 7, 46932–46937. [Google Scholar] [CrossRef] [Green Version]
- Holloway, D.; Scheinmann, F. Two lignans from Litsea grandis and L. gracilipes. Phytochemistry 1974, 13, 1233–1236. [Google Scholar] [CrossRef]
- Da Silva Filho, A.A.; Albuquerque, S.; Silva, M.L.A.; Eberlin, M.N.E.; Tomazela, D.M.; Bastos, J.K. Tetrahydrofuran lignans from Nectandra megapotamica with trypanocidal activity. J. Nat. Prod. 2004, 67, 42–45. [Google Scholar] [CrossRef] [PubMed]
- Soorukram, D.; Pohmakotr, M.; Kuhakarn, C.; Reutrakul, V. Stereoselective synthesis of tetrahydrofuran lignans. Synthesis 2018, 50, 4746–4764. [Google Scholar] [CrossRef]
- Greger, H.; Pacher, T.; Vajrodaya, S.; Bacher, M.; Hofer, O. Infraspecific variation of sulfur-containing bisamides from Aglaia leptantha. J. Nat. Prod. 2000, 63, 616–620. [Google Scholar] [CrossRef]
- Parmar, V.S.; Jain, S.C.; Bisht, K.S.; Jain, R.; Taneja, P.; Jha, A.; Tyagi, O.D.; Prasad, A.K.; Wengel, J.; Olsen, C.E.; et al. Phytochemistry of the genus Piper. Phytochemistry 1997, 46, 597–673. [Google Scholar] [CrossRef]
- Thin, D.B.; Chinh, H.V.; Luong, N.X.; Hoi, T.M.; Dai, D.N.; Ogunwande, I.A. Chemical analysis of essential oils of Piper laosanum and Piper acre (Piperaceae) from Vietnam. J. Essent. Oil Bear. Plants 2018, 21, 181–188. [Google Scholar] [CrossRef]
- Bos, R.; Woerdenbag, H.J.; Kayser, O.; Quax, W.J.; Ruslan, K.; Elfami. Essential oil constituents of Piper cubeba L. fils. from Indonesia. J. Essent. Oil Res. 2007, 19, 14–17. [Google Scholar] [CrossRef]
- McBride, C.S.; Baier, F.; Omondi, A.B.; Spitzer, S.A.; Lutomiah, J.; Sang, R.; Ignell, R.; Vosshall, L.B. Evolution of mosquito preference for humans linked to an odorant receptor. Nature 2014, 515, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Barth, T.; Habenschus, M.D.; Lima Moreira, F.; Ferreira Lde, S.; Lopes, N.P.; Moraes de Oliveira, A.R. In vitro metabolism of the lignan (–)-grandisin, an anticancer drug candidate, by human liver microsomes. Drug Test Anal. 2015, 7, 780–786. [Google Scholar] [CrossRef]
- Lopes, N.P.; Chicaro, P.; Kato, M.J.; Albuquerque, S.; Yoshida, M. Flavonoids and lignans from Virola surinamensis twigs and their in vitro activity against Trypanosoma cruzi. Planta Med. 1998, 64, 667–669. [Google Scholar] [CrossRef] [PubMed]
- De Santis Ferreira, L.; Callejon, D.R.; Engemann, A.; Cramer, B.; Humpf, H.U.; de Barros, V.P.; Assis, M.D.; da Silva, D.B.; de Albuquerque, S.; Okano, L.T.; et al. In vitro metabolism of grandisin, a lignan with anti-chagasic activity. Planta Med. 2012, 78, 1939–1941. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Available online: https://www.who.int/news-room/fact-sheets/detail/chagas-disease-(american-trypanosomiasis) (accessed on 3 May 2021).
- Van Den Dool, H.; Kratz, P.D. A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J. Chromatogr. A 1963, 11, 463–471. [Google Scholar] [CrossRef]
- De Saint Laumer, J.Y.; Cicchetti, E.; Merle, P.; Egger, J.; Chaintreau, A. Quantification in gas chromatography: Prediction of Martinsflame ionization detector response factors from combustion enthalpies and molecular structures. Anal. Chem. 2010, 82, 6457–6462. [Google Scholar] [CrossRef] [PubMed]
- Tissot, E.; Rochat, S.; Debonneville, C.; Chaintreau, A. Rapid GC-FID quantification technique without authentic samples using predicted response factors. Flavour Fragr. J. 2012, 27, 290–296. [Google Scholar] [CrossRef]


| Compounds | DB-5ms | HP-INNOWax | EO (%) 2 | Hydrolate (mg/100 mL) | ||||
|---|---|---|---|---|---|---|---|---|
| LRI 1 | LRI [27] | LRI 1 | LRI | DB-5ms | HP-INNOWax | DB-5ms | HP-INNOWax | |
| α-pinene | 925 | 932 | 1014 | 1025 [28] | 1.6 | 2.1 | -- | -- |
| camphene | 939 | 946 | 1071 | 1075 [28] | trace | trace | -- | -- |
| β-pinene | 968 | 974 | 1103 | 1118 [28] | 2.6 | 3.4 | -- | -- |
| 6-methyl-5-hepten-2-one (sulcatone) | 983 | 981 | 1339 | 1323 [29] | 2.1 | 2.9 | 63.7 | 64.4 |
| myrcene | 986 | 988 | 1163 | 1166 [30] | 0.2 | 0.6 | -- | -- |
| α-phellandrene | 1001 | 1002 | 1159 | 1162 [29] | 0.3 | 0.3 | -- | -- |
| δ-3-carene | 1003 | 1008 | 1142 | 1135 [28] | 0.2 | trace | -- | -- |
| α-terpinene | 1011 | 1014 | 1174 | 1175 [31] | trace | trace | -- | -- |
| ƿ-cymene | 1019 | 1020 | 1268 | 1281 [28] | trace | trace | -- | -- |
| limonene | 1023 | 1024 | 1194 | 1196 [29] | 0.6 | 0.7 | -- | -- |
| 1,8-cineol | 1026 | 1026 | 1201 | 1212 [29] | 0.1 | 0.2 | -- | -- |
| 1,4-cineol | -- | -- | -- | -- | -- | -- | 0.4 | 0.5 |
| γ-terpinene | 1052 | 1054 | 1242 | 1254 [28] | trace | trace | -- | -- |
| p-mentha-2,4(8)-diene(isoterpinolene) | 1078 | 1085 | 1279 | -- | 1.8 | 2.1 | -- | -- |
| trans-linalool oxide (furanoid) | -- | -- | -- | -- | -- | -- | 1.1 | 1.1 |
| linalool | 1100 | 1095 | 1555 | 1554 [31] | 1.1 | 1.4 | 6.5 | 6.0 |
| β-ylangene | -- | -- | 1563 | 1576 [32] | -- | trace | -- | -- |
| 1,3,8-p-menthatriene | 1126 | 1108 | 1421 | 1411 [32] | trace | 0.3 | -- | -- |
| borneol | -- | -- | -- | -- | -- | -- | 0.3 | 0.7 |
| terpinen-4-ol | -- | -- | -- | -- | -- | -- | 0.5 | 0.5 |
| α -terpineol | -- | -- | -- | -- | -- | -- | 0.8 | -- |
| linalool formate | -- | -- | -- | -- | -- | -- | 0.2 | 0.4 |
| δ-elemene | 1326 | 1335 | 1465 | 1468 [32] | 0.2 | 0.2 | -- | -- |
| α-cubebene | 1337 | 1348 | 1451 | 1460 [32] | 0.3 | 0.3 | -- | -- |
| β-copaene | -- | -- | 1579 | 1579 [31] | -- | 4.7 | -- | -- |
| α-copaene | 1362 | 1374 | 1481 | 1493 [33] | 0.9 | 1,0 | -- | -- |
| β-cubebene | 1376 | 1387 | 1530 | 1541 [32] | trace | 0.6 | -- | -- |
| β-elemene | 1379 | 1389 | 1574 | 1580 [34] | 1.6 | 0.2 | -- | -- |
| cyperene | 1385 | 1398 | 1511 | 1528 [32] | 0.1 | 0.1 | -- | -- |
| sibirene | 1392 | 1400 | 1528 | -- | 1.7 | 0.3 | -- | -- |
| α-gurjunene | 1397 | 1409 | 1518 | 1529 [32] | 0.2 | 1.7 | -- | -- |
| (E)-β-caryophyllene | 1404 | 1417 | 1587 | 1598 [32] | 25.3 | 25.2 | -- | -- |
| β-gurjunene | 1414 | 1431 | 1601 | 1596 [32] | 0.2 | trace | -- | -- |
| α-guaiene | 1423 | 1437 | 1632 | 1652 [32] | 1.9 | 0.2 | -- | -- |
| α-cedrene | 1428 | 1410 | 1594 | 1600 [35] | 0.2 | 0.6 | -- | -- |
| 6,9-guaiadiene | 1433 | 1442 | 1621 | 1617 [36] | 0.3 | 0.3 | -- | -- |
| α-humulene | 1438 | 1452 | 1657 | 1666 [32] | 1.8 | 0.4 | -- | -- |
| myltayl-4(12)-ene | 1442 | 1445 | 1622 | -- | 0.4 | 0.2 | -- | -- |
| ishwarene | 1447 | 1466 | 1677 | -- | 1.2 | 0.3 | -- | -- |
| 4,5-di-epi-aristolochene | 1454 | 1471 | 1669 | -- | 0.3 | 1.4 | -- | -- |
| α-neo-clovene | 1459 | 1452 | 1667 | -- | 1.7 | 1.7 | -- | -- |
| cis-cadina- 1(6),4-diene | 1462 | 1461 | 1680 | -- | 0.6 | 0.6 | -- | -- |
| cis-muurola-4(14),5-diene | 1465 | 1465 | 1651 | 1643 [32] | 1.4 | trace | -- | -- |
| selina-3,7(11)-diene | -- | -- | 1762 | 1783 [32] | -- | trace | -- | -- |
| β-selinene | 1472 | 1489 | 1706 | 1716 [32] | 7.2 | 7.7 | -- | -- |
| viridiflorene | 1476 | 1496 | 1685 | 1696 [32] | 1.6 | 1.7 | -- | -- |
| γ-himachalene | -- | -- | 1716 | 1708 [32] | -- | 0.6 | -- | -- |
| β-chamigrene | 1480 | 1476 | 1712 | 1723 [32] | 10.3 | 7.8 | -- | -- |
| trans-cadina-1(6),4-diene | 1483 | 1475 | 1697 | -- | 0.2 | 1.4 | -- | -- |
| α-bulnesene | 1488 | 1509 | 1627 | 1629 [32] | 1.4 | 1.7 | -- | -- |
| bicyclogermacrene | 1490 | 1500 | 1722 | 1734 [32] | 3.7 | 2.4 | -- | -- |
| trans-muurola-4(14),5-diene | 1498 | 1493 | 1700 | -- | 0.3 | 0.6 | -- | -- |
| γ-cadinene | 1502 | 1513 | 1783 | 1763 [32] | 0.7 | 0.1 | -- | -- |
| δ-cadinene | 1506 | 1521 | 1750 | 1755 [32] | 2.7 | 3.9 | -- | -- |
| trans-calamenene | 1508 | 1522 | 1825 | 1823 [32] | 1.1 | 0.7 | -- | -- |
| trans-cadina-1,4-diene | 1518 | 1533 | -- | -- | 0.2 | -- | -- | -- |
| 7-epi-α-selinene | 1524 | 1520 | 1766 | 1764 [32] | 0.2 | 0.4 | -- | -- |
| cis-muurol-5-en-4-β-ol | -- | -- | 1182 | -- | -- | 0.2 | -- | -- |
| α-calacorene | 1525 | 1544 | 1907 | 1921 [32] | 0.7 | 0.5 | -- | -- |
| β-germacrene | 1539 | 1559 | 1815 | 1823 [32] | 1.8 | 1.4 | -- | -- |
| (E)-nerolidol | 1558 | 1561 | 2048 | 2036 [32] | 8.1 | 7.7 | -- | -- |
| caryophyllene oxide | 1563 | 1582 | 1967 | 1986 [32] | 0.2 | 0.5 | -- | -- |
| cubebol | -- | -- | 1934 | 1941 [32] | -- | 0.2 | -- | -- |
| trans-dauca-4(11),7-diene | 1569 | 1556 | 1726 | -- | 0.5 | 0.2 | -- | -- |
| cubenol | -- | -- | 2066 | 2067 [32] | -- | 0.4 | -- | -- |
| guaiol | 1586 | 1600 | 2075 | 2090 [37] | 0.2 | 0.2 | -- | -- |
| 1-epi-cubenol | 1612 | 1627 | 2087 | 2088 [32] | 0.4 | trace | -- | -- |
| epi-α-cadinol | 1630 | 1638 | 2166 | 2169 [32] | 0.2 | 0.2 | -- | -- |
| α-muurolol (torreyol) | 1633 | 1644 | 2171 | 2183 [32] | 0.2 | trace | -- | -- |
| 10-epi-γ-eudesmol | 1637 | 1622 | 2093 | 2105 [32] | 1.5 | 0.2 | -- | -- |
| pogostol | 1641 | 1651 | 2182 | 2196 [38] | 2.5 | 0.2 | 1.1 | 0.1 |
| α-eudesmol | -- | -- | -- | -- | -- | -- | 0.8 | 0.3 |
| 5-neo-cedranol | 1679 | 1684 | 2191 | -- | 0.1 | 0.2 | -- | -- |
| trans-pinocarveol | -- | -- | -- | -- | -- | -- | -- | 0.2 |
| neral | -- | -- | -- | -- | -- | -- | -- | 0.5 |
| nerol | -- | -- | -- | -- | -- | -- | -- | 0.2 |
| Monoterpene hydrocarbons | 7.3 | 9.5 | -- | -- | ||||
| Oxygenated monoterpenes | 3.3 | 4.5 | 73.5 | 73.6 | ||||
| Sesquiterpene hydrocarbons | 72.9 | 71.1 | -- | -- | ||||
| Oxygenated sesquiterpenes | 13.4 | 10,0 | 1.9 | 1.3 | ||||
| Total | 96.9 | 95.1 | 75.4 | 74.9 | ||||
| Enantiomers | LRI 1 | Enantiomeric Ratio | ee 2 (%) |
|---|---|---|---|
| (1R,5R)-(+)-α-pinene | 927 | 64.4 | 28.8 |
| (1S,5S)-(−)-α-pinene | 928 | 35.6 | |
| (1R,5R)-(+)-β-pinene | 953 | 11.1 | 77.8 |
| (1S,5S)-(−)-β-pinene | 960 | 88.9 | |
| (S)-(−)-limonene | 1052 | 59.2 | 18.4 |
| (R)-(+)-limonene | 1067 | 40.8 | |
| (1R,2S,6S,7S,8S)-(−)-α-copaene | 1663 | 53.0 | 6.0 |
| (1S,2R,6R,7R,8R)-(+)-α-copaene | 1666 | 47.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramírez, J.; Andrade, M.D.; Vidari, G.; Gilardoni, G. Essential Oil and Major Non-Volatile Secondary Metabolites from the Leaves of Amazonian Piper subscutatum. Plants 2021, 10, 1168. https://doi.org/10.3390/plants10061168
Ramírez J, Andrade MD, Vidari G, Gilardoni G. Essential Oil and Major Non-Volatile Secondary Metabolites from the Leaves of Amazonian Piper subscutatum. Plants. 2021; 10(6):1168. https://doi.org/10.3390/plants10061168
Chicago/Turabian StyleRamírez, Jorge, María Daniela Andrade, Giovanni Vidari, and Gianluca Gilardoni. 2021. "Essential Oil and Major Non-Volatile Secondary Metabolites from the Leaves of Amazonian Piper subscutatum" Plants 10, no. 6: 1168. https://doi.org/10.3390/plants10061168








