Sediment Characteristics Determine the Flowering Effort of Zostera noltei Meadows Inhabiting a Human-Dominated Lagoon
Abstract
:1. Introduction
2. Material and Methods
2.1. Study Area
2.2. Monitoring of Flowering Effort, Seagrass Biometrics and Abiotic Parameters
2.3. Data Analyses
3. Results
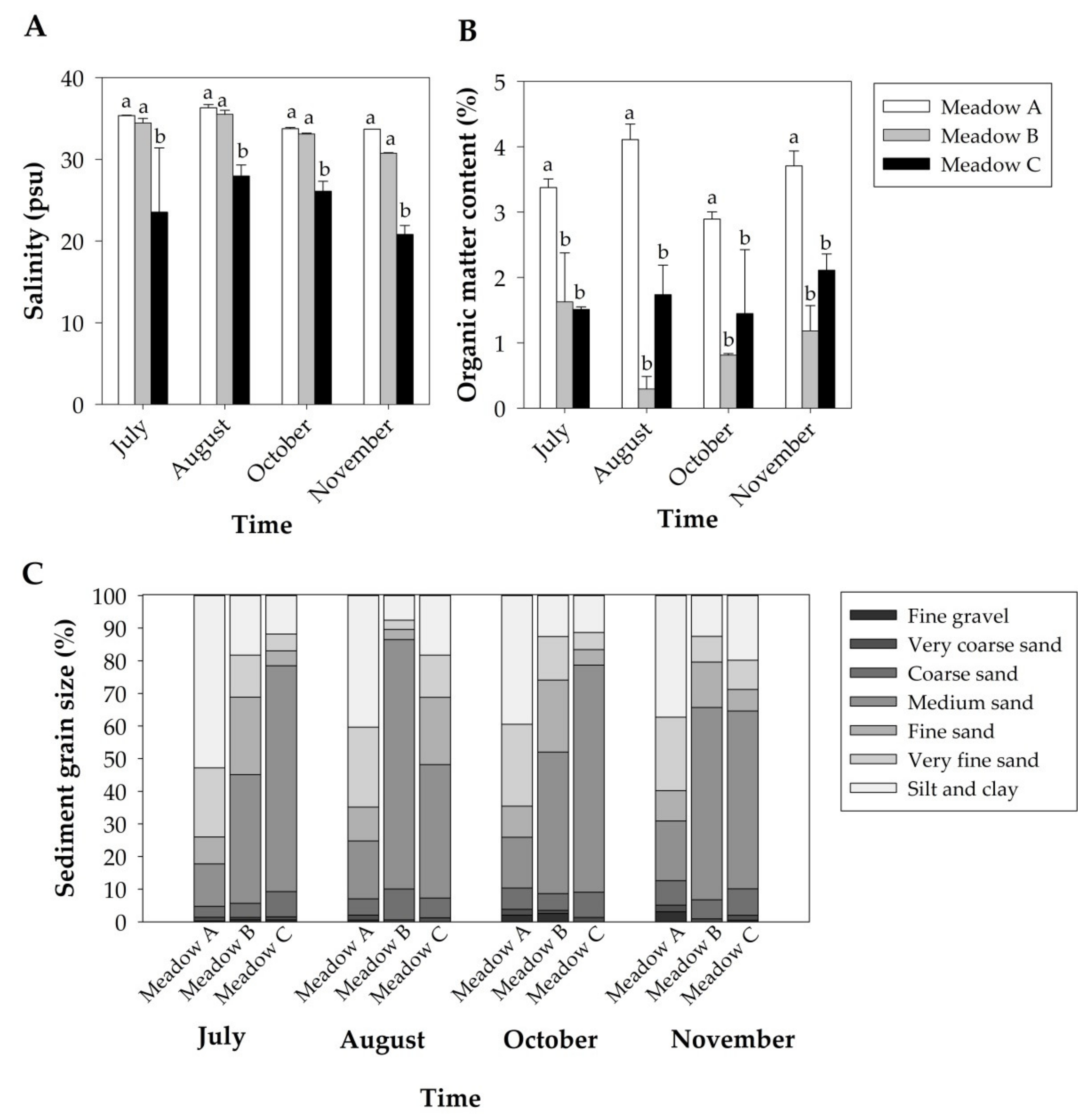
3.1. Abiotic Parameters
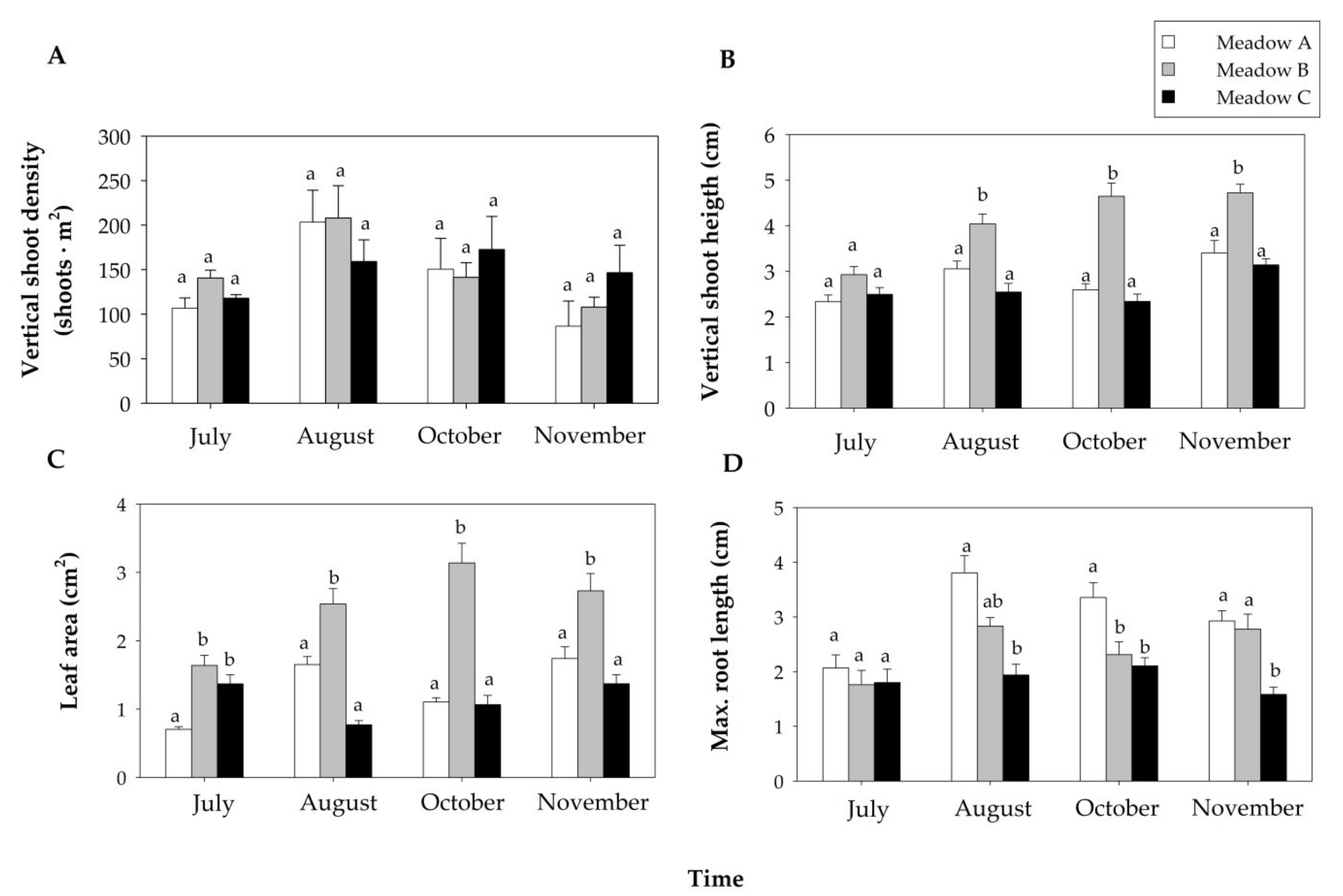
3.2. Seagrass Biometrics
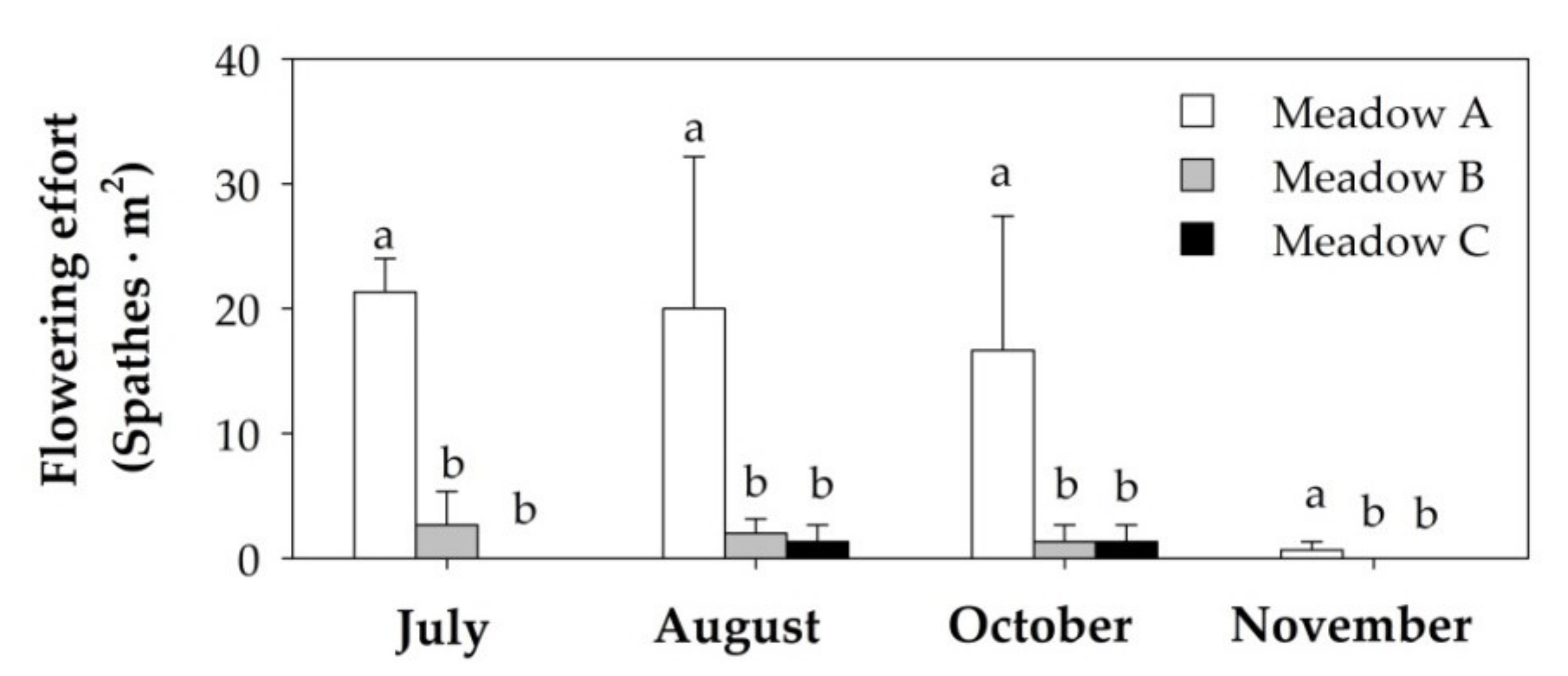
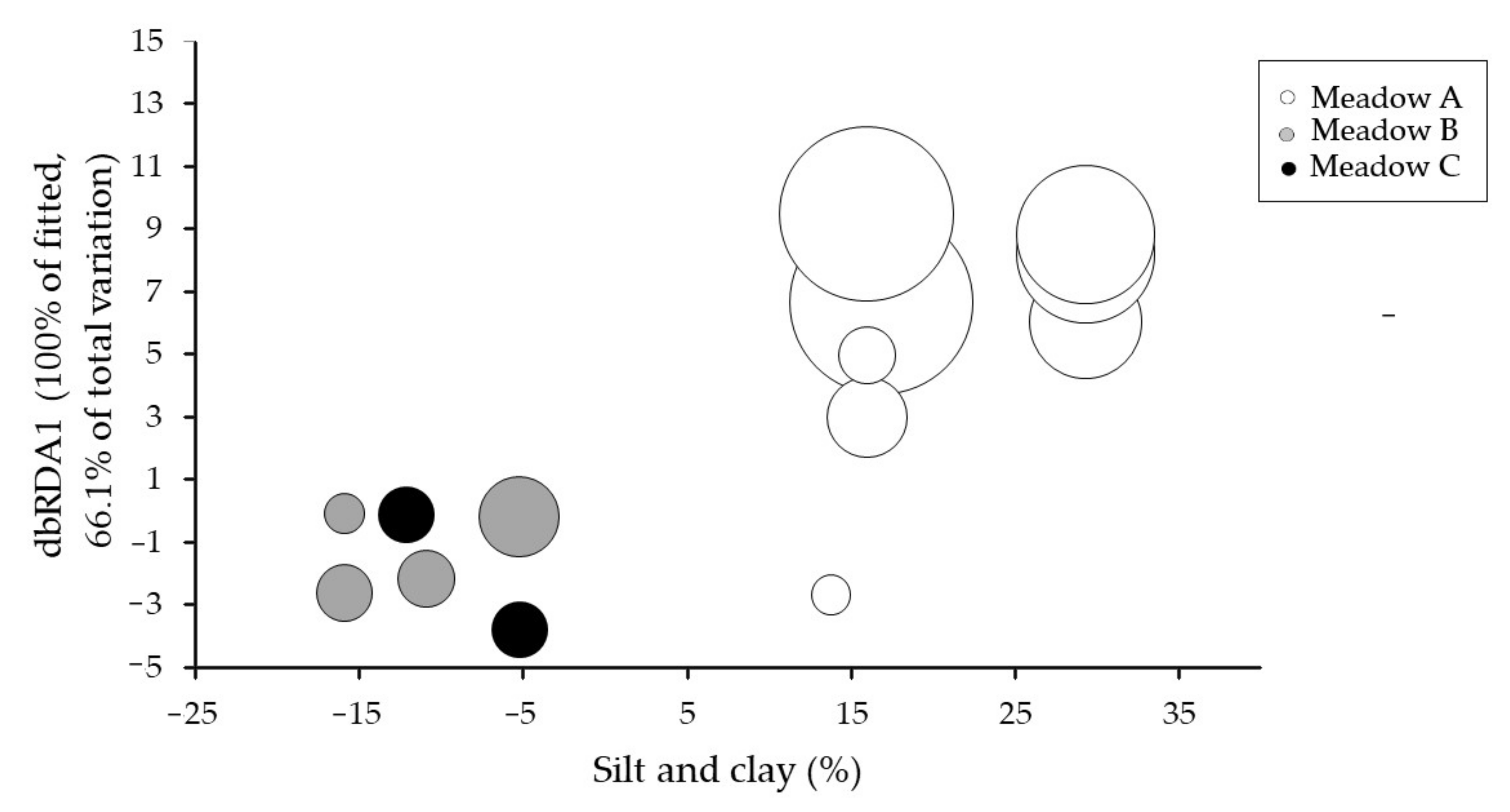
3.3. Flowering Effort and the Relationship with Seagrass Biometrics and Abiotic Parameters
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Hemminga, M.A.; Duarte, C.M. Seagrass Ecology; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Chen, S.-N.; Sanford, L.P.; Koch, E.W.; Shi, F.; North, E.W. A nearshore model to investigate the effects of seagrass bed geometry on wave attenuation and suspended sediment transport. Estuaries Coasts 2007, 30, 296–310. [Google Scholar] [CrossRef]
- Krause-Jensen, D.; Greve, T.M.; Nielsen, K. Eelgrass as a bioindicator under the European water framework directive. Water Resour. Manag. 2005, 19, 63–75. [Google Scholar] [CrossRef]
- Cullen-Unsworth, L.; Unsworth, R. Seagrass meadows, ecosystem services, and sustainability. Environ. Sci. Policy Sustain. Dev. 2013, 55, 14–28. [Google Scholar] [CrossRef]
- Smith, S.V. Marine macrophytes as a global carbon sink. Science 1981, 211, 838–840. [Google Scholar] [CrossRef] [Green Version]
- Duarte, C.; Middelburg, J.J.; Caraco, N. Major role of marine vegetation on the oceanic carbon cycle. Biogeosciences 2005, 1, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Kennedy, H.; Beggins, J.; Duarte, C.M.; Fourqurean, J.W.; Holmer, M.; Marbà, N.; Middelburg, J.J. Seagrass sediments as a global carbon sink: Isotopic constraints. Glob. Biogeochem. Cycles 2010, 24. [Google Scholar] [CrossRef] [Green Version]
- Mcleod, E.; Chmura, G.L.; Bouillon, S.; Salm, R.; Björk, M.; Duarte, C.M.; Lovelock, C.E.; Schlesinger, W.H.; Silliman, B.R. A blueprint for blue carbon: Toward an improved understanding of the role of vegetated coastal habitats in sequestering CO2. Front. Ecol. Environ. 2011, 9, 552–560. [Google Scholar] [CrossRef] [Green Version]
- Fourqurean, J.W.; Duarte, C.M.; Kennedy, H.; Marbà, N.; Holmer, M.; Mateo, M.A.; Apostolaki, E.T.; Kendrick, G.A.; Krause-Jensen, D.; McGlathery, K.J.; et al. Seagrass ecosystems as a globally significant carbon stock. Nat. Geosci. 2012, 5, 505–509. [Google Scholar] [CrossRef]
- Short, F.T.; Carruthers, T.; Dennison, W.; Waycott, M. Global seagrass distribution and diversity: A bioregional model. J. Exp. Mar. Biol. Ecol. 2007, 350, 3–20. [Google Scholar] [CrossRef]
- Grech, A.; Chartrand-Miller, K.; Erftemeijer, P.; Fonseca, M.; McKenzie, L.; Rasheed, M.; Taylor, H.; Coles, R. A comparison of threats, vulnerabilities and management approaches in global seagrass bioregions. Environ. Res. Lett. 2012, 7. [Google Scholar] [CrossRef]
- Orth, R.J.W.; Carruthers, J.B.T.J.B.; Dennison, W.C.; Duarte, C.M.; Fourqurean, J.W.; Heck, K.L.; Hughes, A.R.; Kendrick, G.A.; Kenworthy, W.J.; Olyarnik, S.; et al. A global crisis for seagrass ecosystems. BioScience 2006, 56, 987. [Google Scholar] [CrossRef] [Green Version]
- Cochón, G.; Sánchez, J.M. Variations of seagrass beds in Pontevedra (North- Western Spain): 1947–2001. Thalassas 2005, 21, 9–19. [Google Scholar]
- Silva, F.J.; Duck, R.W.; Catarino, J.B. Seagrasses and sediment response to changing physical forcing in a coastal lagoon. Hydrol. Earth Syst. Sci. 2004, 8, 151–159. [Google Scholar] [CrossRef] [Green Version]
- Short, F.T.; Wyllie-Echeverria, S. Natural and human-induced disturbance of seagrasses. Environ. Conserv. 1996, 23, 17–27. [Google Scholar] [CrossRef]
- Sousa, A.I.; Silva, F.J.; Azevedo, A.; Lillebø, A.I. Blue Carbon stock in Zostera noltei meadows at Ria de Aveiro coastal lagoon (Portugal) over a decade. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef] [Green Version]
- de los Santos, C.B.; Krause-Jensen, D.; Alcoverro, T.; Marbà, N.; Duarte, C.M.; van Katwijk, M.M.; Pérez, M.; Romero, J.; Sánchez-Lizaso, J.L.; Roca, G.; et al. Recent trend reversal for declining European seagrass meadows. Nat. Commun. 2019, 10, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Román, M.; Fernández, E.; Zamborain-Mason, J.; Martínez, L.; Méndez, G. Decadal changes in the spatial coverage of Zostera noltei in two seagrass meadows (Ría de Vigo; NW Spain). Reg. Stud. Mar. Sci. 2020, 36, 101264. [Google Scholar] [CrossRef]
- Bertelli, C.M.; Robinson, M.T.; Mendzil, A.F.; Pratt, L.R.; Unsworth, R.K.F. Finding some seagrass optimism in Wales, the case of Zostera noltii. Mar. Pollut. Bull. 2018, 134, 216–222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calleja, F.; Galván, C.; Silió-Calzada, A.; Juanes, J.A.; Ondiviela, B. Long-term analysis of Zostera noltei: A retrospective approach for understanding seagrasses’ dynamics. Mar. Environ. Res. 2017, 130, 93–105. [Google Scholar] [CrossRef]
- Guerrero-Meseguer, L.; Veiga, P.; Sampaio, L.; Rubal, M. Resurgence of Zostera marina in the Ria de Aveiro lagoon, Portugal. Aquat. Bot. 2021, 169, 103338. [Google Scholar] [CrossRef]
- Barillé, L.; Robin, M.; Harin, N.; Bargain, A.; Launeau, P. Increase in seagrass distribution at Bourgneuf Bay (France) detected by spatial remote sensing. Aquat. Bot. 2010, 92, 185–194. [Google Scholar] [CrossRef]
- Kilminster, K.; Mcmahon, K.; Waycott, M.; Kendrick, G.A.; Scanes, P.; Mckenzie, L.; O’Brien, K.R.; Lyons, M.; Ferguson, A.; Maxwell, P.; et al. Unravelling complexity in seagrass systems for management: Australia as a microcosm. Sci. Total. Environ. 2015, 534, 97–109. [Google Scholar] [CrossRef]
- Den Hartog, C. The seagrasses of the world. In Verhandelingen der Koninklijke Nederlandse Akademie van Wetenschappen, Afd. Natuurkunde. Tweede Reeks; North Holland Publishing Co.: Amsterdam, The Netherlands, 1970; Volume 59, p. 273. [Google Scholar] [CrossRef]
- Wolff, W.J.; Duiven, A.G.; Duiven, P.; Esselink, P.; Gueye, A.; Meijboom, A.; Moerland, G.; Zegers, J. Biomass of macrobenthic tidal flat fauna of the Banc d’Arguin, Mauritania. In Ecological Studies in the Coastal Waters of Mauritania; Springer Netherlands: Dordrecht, The Netherlands, 1993; pp. 151–163. [Google Scholar]
- Massa, S.I.; Arnaud-Haond, S.; Pearson, G.A.; Serrão, E.A. Temperature tolerance and survival of intertidal populations of the seagrass Zostera noltii (Hornemann) in Southern Europe (Ria Formosa, Portugal). Hydrobiologia 2008, 619, 195–201. [Google Scholar] [CrossRef]
- Vermaat, J.E.; Agawin, N.S.R.; Fortes, M.D.; Uri, J.S. The capacity of seagrasses to survive increased turbidity and siltation: The significance of growth form and light use. Ambio 1997, 26, 499–504. [Google Scholar]
- Auby, I.; Labourg, P.J. Seasonal dynamics of Zostera noltii Hornem. in the Bay of Arcachon (France). J. Sea Res. 1996, 35, 269–277. [Google Scholar] [CrossRef]
- Alexandre, A.; Santos, R.; Serrão, E. Effects of clam harvesting on sexual reproduction of the seagrass Zostera noltii. Mar. Ecol. Prog. Ser. 2005, 298, 115–122. [Google Scholar] [CrossRef] [Green Version]
- Cabaço, S.; Santos, R. Seagrass reproductive effort as an ecological indicator of disturbance. Ecol. Indic. 2012, 23, 116–122. [Google Scholar] [CrossRef]
- Plus, M.; Deslous-Paoli, J.-M.; Dagault, F. Seagrass (Zostera marina L.) bed recolonisation after anoxia-induced full mortality. Aquat. Bot. 2003, 77, 121–134. [Google Scholar] [CrossRef]
- Lee, K.S.; Park, S.R.; Kim, Y.K. Effects of irradiance, temperature, and nutrients on growth dynamics of seagrasses: A review. J. Exp. Mar. Biol. Ecol. 2007, 350, 144–175. [Google Scholar] [CrossRef]
- Bell, S.S.; Fonseca, M.S.; Kenworthy, W.J. Dynamics of a subtropical seagrass landscape: Links between disturbance and mobile seed banks. Landsc. Ecol. 2008, 23, 67–74. [Google Scholar] [CrossRef]
- Kendrick, G.A.; Waycott, M.; Carruthers, T.J.B.; Cambridge, M.L.; Hovey, R.; Krauss, S.L.; Lavery, P.S.; Les, D.H.; Lowe, R.J.; Vidal, O.M.I.; et al. The central role of dispersal in the maintenance and persistence of seagrass populations. BioScience 2012, 62, 56–65. [Google Scholar] [CrossRef] [Green Version]
- Ehlers, A.; Worm, B.; Reusch, T. Importance of genetic diversity in eelgrass Zostera marina for its resilience to global warming. Mar. Ecol. Prog. Ser. 2008, 355, 1–7. [Google Scholar] [CrossRef]
- Loques, F.; Caye, G.; Meinesz, A. Flowering and fruiting of Zostera noltii in Golfe Juan (French Mediterranean). Aquat. Bot. 1988, 32, 341–352. [Google Scholar] [CrossRef]
- Ramage, D.L.; Schiel, D.R. Patch dynamics and response to disturbance of the seagrass Zostera novazelandica on intertidal platforms in southern New Zealand. Mar. Ecol. Prog. Ser. 1999, 189, 275–288. [Google Scholar] [CrossRef]
- von Staats, D.A.; Hanley, T.C.; Hays, C.G.; Madden, S.R.; Sotka, E.E.; Hughes, A.R. Intra-meadow variation in seagrass flowering phenology across depths. Estuaries Coasts 2021, 44, 325–338. [Google Scholar] [CrossRef]
- Hootsmans, M.J.M.; Vermaat, J.E.; Van Vierssen, W. Seed-bank development, germination and early seedling survival of two seagrass species from The Netherlands: Zostera marina L. and Zostera noltii hornem. Aquat. Bot. 1987, 28, 275–285. [Google Scholar] [CrossRef]
- Curiel, D.; Bellato, A.; Rismondo, A.; Marzocchi, M. Sexual reproduction of Zostera noltii Hornemann in the lagoon of Venice (Italy, north Adriatic). Aquat. Bot. 1996, 52, 313–318. [Google Scholar] [CrossRef]
- Cabaço, S.; Santos, R.; Sprung, M. Population dynamics and production of the seagrass Zostera noltii in colonizing versus established meadows. Mar. Ecol. 2012, 33, 280–289. [Google Scholar] [CrossRef]
- Moreira, M.H.; Queiroga, H.; Machado, M.M.; Cunha, M.R. Environmental gradients in a southern Europe estuarine system: Ria de Aveiro, Portugal implications for soft bottom macrofauna colonization. Neth. J. Aquat. Ecol. 1993, 27, 465–482. [Google Scholar] [CrossRef]
- Silva, F.; Duck, R. Historical changes of bottom topography and tidal amplitude in the Ria de Aveiro, Portugal–trends for future evolution. Clim. Res. 2001, 18, 17–24. [Google Scholar] [CrossRef] [Green Version]
- Silva, F.J.; Duck, R.W.; Catarino, J.B. Changing use of the estuarine system of the Ria de Aveiro, Portugal, and resultant impact on tidal flat sediments. RMZ-Mater. Geoenvironment 2005, 111–114. [Google Scholar]
- Silva, F.J.; Duck, R.W.; Catarino, J.B. Nutrient retention in the sediments and the submerged aquatic vegetation of the coastal lagoon of the Ria de Aveiro, Portugal. J. Sea Res. 2009, 62, 276–285. [Google Scholar] [CrossRef]
- Wentworth, C.K. A scale of grade and class terms for clastic sediments. J. Geol. 1922, 30, 377–392. [Google Scholar] [CrossRef]
- Underwood, A.J. Experiments in Ecology: Their Logical Design and Interpretation Using Analysis of Variance; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- McArdle, B.H.; Anderson, M.J. Fitting multivariate models to community data: A comment on distance-based redundancy analysis. Ecology 2001, 82, 290–297. [Google Scholar] [CrossRef]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar] [CrossRef]
- Anderson, M.J. DISTLM v. 5: A FORTRAN Computer Program to Calculate a Distance-based Multivariate Analysis for a Linear Model; Department of Statistics, University of Auckland: Auckland, New Zealand, 2004; Volume 10. [Google Scholar]
- Legendre, P.; Anderson, M.J. Distance-based redundancy analysis: Testing multispecies responses in multifactorial ecological experiments. Ecol. Monogr. 1999, 69, 1e24. [Google Scholar] [CrossRef]
- Malmer, A.; Grip, H. Converting tropical rainforest to forest plantation in Sabah, Malaysia. Part II. Effects on nutrient dynamics and net losses in streamwater. Hydrol. Process. 1994, 8, 195–209. [Google Scholar] [CrossRef]
- Duarte, C.; Terrados, J.; Agawin, N.; Fortes, M.; Bach, S.; Kenworthy, W. Response of a mixed Philippine seagrass meadow to experimental burial. Mar. Ecol. Prog. Ser. 1997, 147, 285–294. [Google Scholar] [CrossRef] [Green Version]
- Terrados, J.; Duarte, C.M.; Fortes, M.D.; Borum, J.; Agawin, N.S.R.; Bach, S.; Thampanya, U.; Kamp-Nielsen, L.; Kenworthy, W.J.; Geertz-Hansen, O.; et al. Changes in community structure and biomass of seagrass communities along gradients of siltation in SE Asia. Estuar. Coast. Shelf Sci. 1998, 46, 757–768. [Google Scholar] [CrossRef]
- Holmer, M.; Frederiksen, M.S. Stimulation of sulfate reduction rates in Mediterranean fish farm sediments inhabited by the seagrass Posidonia oceanica. Biogeochemistry 2007, 85, 169–184. [Google Scholar] [CrossRef]
- Smith, T.M.; York, P.H.; Macreadie, P.I.; Keough, M.J.; Ross, D.J.; Sherman, C.D.H. Spatial variation in reproductive effort of a southern Australian seagrass. Mar. Environ. Res. 2016, 120, 214–224. [Google Scholar] [CrossRef]
- Cabaço, S.; Machás, R.; Santos, R. Individual and population plasticity of the seagrass Zostera noltii along a vertical intertidal gradient. Estuar. Coast. Shelf Sci. 2009, 82, 301–308. [Google Scholar] [CrossRef]
- Harrison, P.G. Reproductive strategies in intertidal populations of two co-occurring seagrasses (Zostera spp.). Can. J. Bot. 1979, 57, 2635–2638. [Google Scholar] [CrossRef]
- Kautsky, L. Life strategies of aquatic soft bottom macrophytes. Oikos 1988, 53, 126. [Google Scholar] [CrossRef]
- van Lent, F.; Verschuure, J.M. Intraspecific variability of Zostera marina L. (eelgrass) in the estuaries and lagoons of the southwestern Netherlands. I. Population dynamics. Aquat. Bot. 1994, 48, 31–58. [Google Scholar] [CrossRef]
- Middelburg, J.J.; Soetaert, K.; Herman, P.M.J. Empirical relationships for use in global diagenetic models. Deep Sea Res. Part I Oceanogr. Res. Pap. 1997, 44, 327–344. [Google Scholar] [CrossRef]
- Vallejo-Marín, M.; Dorken, M.E.; Barrett, S.C.H. The ecological and evolutionary consequences of clonality for plant mating. Annu. Rev. Ecol. Evol. Syst. 2010, 41, 193–213. [Google Scholar] [CrossRef] [Green Version]
- Salisbury, E. The Reproductive Capacity of Plants; Bell & Sons: London, UK, 1942. [Google Scholar]
- Waller, D.M. Plant morphology and reproduction. In Plant Reproductive Ecology: Patterns and Strategies; Oxford University Press on Demand: Oxford, UK, 1988; pp. 203–227. [Google Scholar]
- Collier, C.J.; Villacorta-Rath, C.; Van Dijk, K.J.; Takahashi, M.; Waycott, M. Seagrass proliferation precedes mortality during hypo-salinity events: A stress-induced morphometric response. PLoS ONE 2014, 9, e94014. [Google Scholar] [CrossRef] [Green Version]
- Dias, J.M.; Lopes, J.F.; Dekeyser, I. Hydrological characterisation of Ria de Aveiro, Portugal, in early summer. Oceanol. Acta 1999, 22, 473–485. [Google Scholar] [CrossRef] [Green Version]
- Dias, J.M.; Lopes, J.F.; Dekeyser, I. A numerical system to study the transport properties in the Ria de Aveiro lagoon. Ocean. Dyn. 2003, 53, 220–231. [Google Scholar] [CrossRef]
- Hammerstrom, K.K.; Kenworthy, W.J.; Fonseca, M.S.; Whitfield, P.E. Seed bank, biomass, and productivity of Halophila decipiens, a deep water seagrass on the west Florida continental shelf. Aquat. Bot. 2006, 84, 110–120. [Google Scholar] [CrossRef]
- Jarvis, J.C.; Moore, K.A. The role of seedlings and seed bank viability in the recovery of Chesapeake Bay, USA, Zostera marina populations following a large-scale decline. Hydrobiologia 2010, 649, 55–68. [Google Scholar] [CrossRef]
- Zipperle, A.M.; Coyer, J.A.; Reise, K.; Stam, W.T.; Olsen, J.L. Evidence for persistent seed banks in dwarf eelgrass Zostera noltii in the German Wadden Sea. Mar. Ecol. Prog. Ser. 2009, 380, 73–80. [Google Scholar] [CrossRef] [Green Version]

| Variable | Meadow | Time | Meadow × Time |
|---|---|---|---|
| Abiotic parameters | |||
| Temperature * | 0.784 (0.479) | 4455.1 (<0.001) | 0.468 (0.820) |
| Salinity * | 21.87 (<0.001) | 2.094 (0.154) | 0.337 (0.904) |
| Redox Potential | 0.035 (0.966) | 0.931 (0.455) | 0.378 (0.879) |
| Organic matter | 39.07 (<0.001) | 1.165 (0.364) | 1.339 (0.313) |
| Fine gravel | 2.565 (0.118) | 2.044 (0.161) | 1.831 (0.175) |
| Very coarse sand * | 6.873 (0.010) | 1.689 (0.222) | 0.288 (0.931) |
| Coarse sand | 3.373 (0.069) | 2.731 (0.122) | 3.428 (0.033) |
| Medium sand | 116.0 (<0.001) | 0.577 (0.641) | 12.33 (<0.001) |
| Fine sand | 14.71 (<0.001) | 0.877 (0.480) | 19.26 (<0.001) |
| Very fine sand | 146.0 (<0.001) | 0.725 (0.556) | 10.17 (<0.001) |
| Silt/Clay | 30.77 (<0.001) | 0.502 (0.688) | 1.499 (0.259) |
| Seagrass biometrics | |||
| Vertical shoot density | 0.309 (0.737) | 5.413 (0.005) | 0.985 (0.457) |
| Vertical shoot height a | 60.54 (<0.001) | 17.83 (<0.001) | 3.770 (0.001) |
| Leaf area b | 90.00 (<0.001) | 12.62 (<0.001) | 12.53 (<0.001) |
| Maximum root length a | 26.25 (<0.001) | 12.37 (<0.001) | 2.918 (0.009) |
| Flowering effort * | 10.41 (0.001) | 1.693 (0.195) | 1.112 (0.384) |
| Variable | Variance % | F | p | Cum. % |
|---|---|---|---|---|
| Silt/Clay/Organic matter | 40.77 | 23.40 | 0.001 | 40.77 |
| Vertical shoot density | 13.64 | 9.873 | 0.007 | 54.41 |
| Very coarse sand | 3.535 | 2.689 | 0.091 | 57.94 |
| Vertical shoot height | 0.634 | 0.474 | 0.488 | 58.58 |
| Leaf area | 1.933 | 1.469 | 0.230 | 60.51 |
| Temperature | 1.410 | 1.074 | 0.301 | 61.92 |
| Maximum root length | 1.113 | 0.843 | 0.359 | 63.03 |
| Salinity | 0.847 | 0.633 | 0.433 | 63.88 |
| Very fine sand | 1.116 | 0.829 | 0.378 | 65.00 |
| Medium sand | 0.635 | 0.462 | 0.517 | 65.63 |
| Redox potential | 0.258 | 0.182 | 0.672 | 65.89 |
| Coarse sand | 0.061 | 0.041 | 0.855 | 65.95 |
| Fine sand | 0.123 | 0.080 | 0.778 | 66.07 |
| Fine gravel | 0.000 | 0.000 | 1.000 | 66.07 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guerrero-Meseguer, L.; Veiga, P.; Sampaio, L.; Rubal, M. Sediment Characteristics Determine the Flowering Effort of Zostera noltei Meadows Inhabiting a Human-Dominated Lagoon. Plants 2021, 10, 1387. https://doi.org/10.3390/plants10071387
Guerrero-Meseguer L, Veiga P, Sampaio L, Rubal M. Sediment Characteristics Determine the Flowering Effort of Zostera noltei Meadows Inhabiting a Human-Dominated Lagoon. Plants. 2021; 10(7):1387. https://doi.org/10.3390/plants10071387
Chicago/Turabian StyleGuerrero-Meseguer, Laura, Puri Veiga, Leandro Sampaio, and Marcos Rubal. 2021. "Sediment Characteristics Determine the Flowering Effort of Zostera noltei Meadows Inhabiting a Human-Dominated Lagoon" Plants 10, no. 7: 1387. https://doi.org/10.3390/plants10071387








