Ecological and Biological Properties of Satureja cuneifolia Ten. and Thymus spinulosus Ten.: Two Wild Officinal Species of Conservation Concern in Apulia (Italy). A Preliminary Survey
Abstract
:1. Introduction
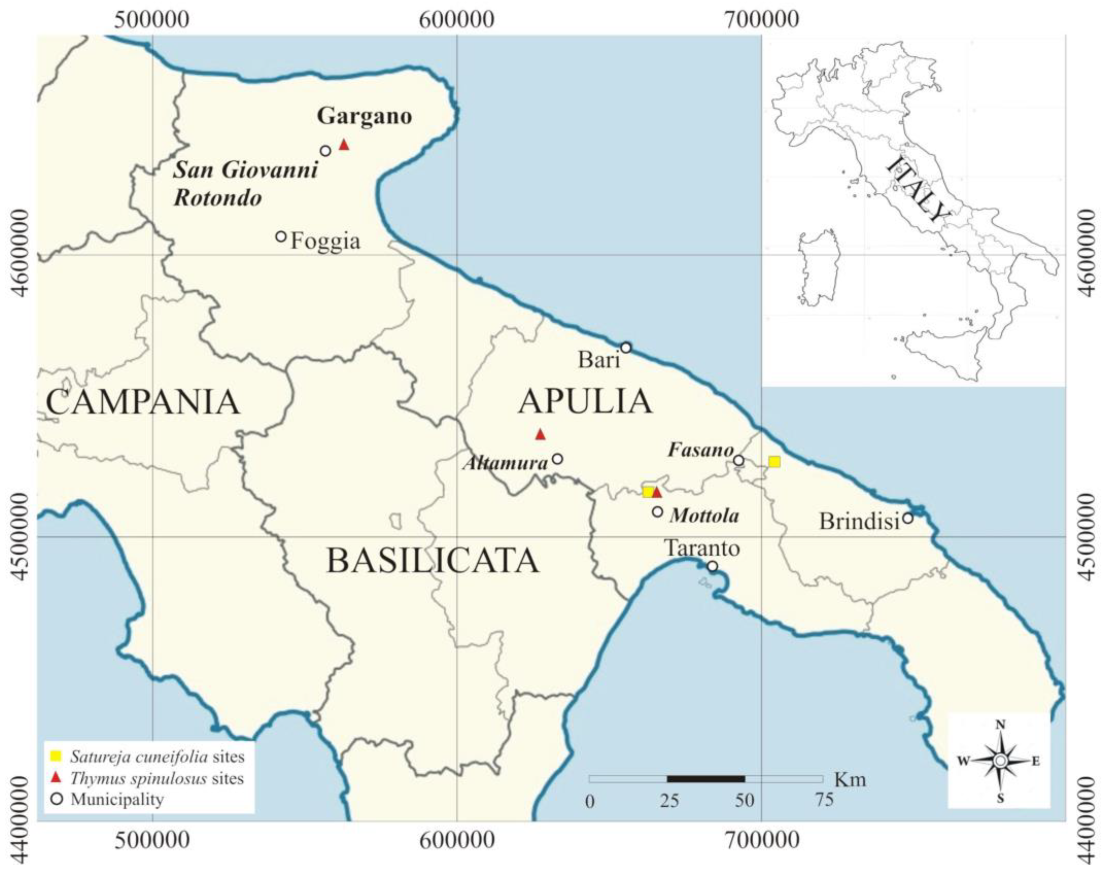
2. Study Area
3. Materials and Methods
3.1. Vegetation Analysis
3.2. Essential Oils Analysis
3.3. Soil Analysis
4. Results and Discussions
4.1. Satureja Cuneifolia Ten.
4.2. Thymus Spinulosus Ten.
4.3. Syntaxonomical Scheme of Surveyed Vegetation
- Festuco-Brometea Br.-Bl. et Tx. ex Soó 1947
- Scorzoneretalia villosae Kovačević 1959
- Hippocrepido glaucae-Stipion austroitalicae Forte et Terzi 2005 in Forte, Perrino and Terzi 2005
- Hippocrepido glaucae-Stipienion austroitalicae Biondi and Galdenzi 2012
- Acino suaveolentis-Stipetum austroitalicae Forte et Terzi in Forte, Perrino et Terzi 2005
- Sideritido syriacae-Stipetum austroitalicae Biondi and Guerra 2008
- Ononido-Rosmarinetea Br.-Bl. in A. Bolòs y Vayreda 1950
- Cisto-Micromerietalia julianae Oberd. 1954
- Cisto cretici-Ericion manipuliflorae Horvatić 1958
- Phagnalo saxatilii-Saturejetum cuneifoliae Biondi and Guerra 2008
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cock, I.E. Medicinal and Aromatic Plants Australia, In Ethnopharmacology Section, Biological, Physiological and Health Sciences, Encyclopedia of Life Support Systems (EOLSS); Developed under the Auspices of the Unesco, Eolss Publishers: Oxford, UK, 2011. [Google Scholar]
- Petrovska, B.B. Historical review of medicinal plants’ usage. Pharm. Rev. 2012, 6, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, M.; Sultana, S.; Fazl-i-Hadi, S.; Ben Hadda, T.; Rashid, S.; Zafar, M.; Khan, M.A.; Khan, M.P.Z.; Yaseen, G. An ethnobotanical study of medicinal plants in high mountainous region of Chail valley (District Swat-Pakistan). J. Ethnobiol. Ethnomed. 2014, 10, 36. [Google Scholar] [CrossRef] [Green Version]
- Sargin, S.A.; Büyükcengiz, M. Plants used in ethnomedicinal practices in Gulnar district of Mersin, Turkey. J. Herb. Med. 2019, 15, 100224. [Google Scholar] [CrossRef]
- Şimşek Sezer, E.N.; Uysal, T. Phytochemical Analysis, Antioxidant and Anticancer Potential of Sideritis niveotomentosa: Endemic Wild Species of Turkey. Molecules 2021, 26, 2420. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Mikallou, M.; Petropoulos, S.; Tzortzakis, N. Profiling of Essential Oils Components and Polyphenols for Their Antioxidant Activity of Medicinal and Aromatic Plants Grown in Different Environmental Conditions. Agronomy 2020, 10, 727. [Google Scholar] [CrossRef]
- Pichersky, E.; Gershenzon, J. The formation and function of plant volatiles: Perfumes for pollinator attraction and defense. Curr. Opin. Plant Biol. 2002, 5, 237–243. [Google Scholar] [CrossRef]
- Dhifi, W.; Bellili, S.; Jazi, S.; Bahloul, N.; Mnif, W. Essential Oils’ Chemical Characterization and Investigation of Some Biological Activities: A Critical Review. Medicines 2016, 3, 25. [Google Scholar] [CrossRef] [Green Version]
- Dini, I. Use of Essential Oils in Food Packaging. In Essential Oils in Food Preservation, Flavor and Safety; Academic Press: Cambridge, MA, USA, 2016; pp. 139–147, (Chapter 14). [Google Scholar] [CrossRef]
- Mari, M.; Bautista-Baños, A.; Sivakumar, D. Decay control in the postharvest system: Role of microbial and plant volatile organic compounds. Postharvest Biol. Technol. 2016, 122, 70–81. [Google Scholar] [CrossRef]
- Moghaddam, M.; Mehdizadeh, L. Chemistry of essential oils and factors influencing their constituents. In Soft Chemistry and Food Fermentation; Grumezescu, A.M., Holban, A.M., Eds.; Academic Press: London, UK, 2019; Volume 13, pp. 379–419. [Google Scholar] [CrossRef]
- Zuzarte, M.; Salgueiro, L. Essential Oils Chemistry. In Bioactive Essential Oils and Cancer; de Sousa, D., Ed.; Springer: Cham, Switzerland, 2015. [Google Scholar] [CrossRef]
- The Plant List, 2013 Version 1.1. Published on the Internet. Available online: http://www.theplantlist.org/ (accessed on 20 May 2021).
- Kokkini, S.; Karousou, R.; Hanlidou, E. Herbs of the Labiatae. In Encyclopedia of Food Sciences and Nutrition; Elsevier Science Ltd.: Amsterdam, The Netherlands, 2003; pp. 3082–3090. [Google Scholar]
- Ozkan, G.; Simsek, B.; Kuleasan, H. Antioxidant activities of Satureja cilicica essential oil in butter and in vitro. J. Food Eng. 2007, 79, 1391–1396. [Google Scholar] [CrossRef]
- Carović-Stanko, K.; Petek, M.; Grdiša, M.; Pintar, J.; Bedekovic, D.; HerakCustic, M.; Satovic, Z. Medicinal plants of the family Lamiaceae as functional foods—A review. Czech J. Food Sci. 2016, 34, 377–390. [Google Scholar] [CrossRef] [Green Version]
- Maksimović, M.; Vidic, D.; Miloš, M.; Šolić, M.E.; Abadžić, S.; Siljak-Yakovlev, S. Effect of the environmental conditions on essential oil profile in two Dinaric Salvia species: S. brachyodon Vandas and S. officinalis L. Biochem. Syst. Ecol. 2018, 35, 473–478. [Google Scholar] [CrossRef]
- Yeddes, W.; Aidi Wannes, W.; Hammami, M.; Smida, M.; Chebbi, A.; Marzouk, B.; Saidani Tounsi, M. Effect of environmental conditions on the chemical composition and antioxidant activity of essential oils from Rosmarinus officinalis L. growing wild in Tunisia. J. Essent. Oil Bear. Plants 2018, 21, 972–986. [Google Scholar] [CrossRef]
- Perrino, E.V.; Valerio, F.; Gannouchi, A.; Trani, A.; Mezzapesa, G. Ecological and Plant Community Implication on Essential Oils Composition in Useful Wild Officinal Species: A Pilot Case Study in Apulia (Italy). Plants 2021, 10, 574. [Google Scholar] [CrossRef] [PubMed]
- Italian Ministry for the Environment, Land and Sea (MATTM) Geoportale Nazionale. 2021. Available online: http://www.pcn.minambiente.it/viewer/ (accessed on 26 May 2021).
- Bellini, F.; Dursi, F.; Perrino, E.V.; Wagensommer, R.P. Caratterizzazione floristica di aree di proprietà pubblica per la reintroduzione delle specie vegetali in situ. Il parco naturale regionale Terra delle Gravine. In Grastepp tra Gravine e Steppe—Azioni per la Conservazione Della Biodiversità nel Parco Nazionale dell’Alta Murgia e nel Parco Naturale Regionale Terra Delle Gravine—Rapporto Finale. APQ FAS Biodiversità Delibera Cipe 19/2004; Ufficio Parchi e Tutela della Biodiversità della Regione Puglia: Bari, Italy, 2014; pp. 117–127. [Google Scholar]
- Braun−Blanquet, J.; Conard, H.S.; Fuller, G.D. Plant Sociology; McGraw−Hill: New York, NY, USA, 1932; p. 439. [Google Scholar]
- Géhu, J.M.; Rivas-Martínez, S. Notions Fondamentales de Phytosociologie; Dierschke, H., Ed.; Berichte der Internationalen Symposien der Internationalen Vereinigung fur Vegetationskunde: Cramer, Vaduz, 1981; pp. 5–33, (Syntaxonomie). [Google Scholar]
- Pignatti, S. Flora d’Italia; Edagricole: Bologna, Italy, 1982; Volume 1–3. [Google Scholar]
- Tutin, T.G.; Heywood, V.H.; Burges, N.A.; Moore, D.M.; Valentine, D.H.; Walters, S.M.; Webb, D.A. Flora Europea, 1st ed.; Cambridge University Press: Cambridge, UK, 1964; Volume 1–5. [Google Scholar]
- Bartolucci, F.; Peruzzi, L.; Galasso, G.; Albano, A.; Alessandrini, A.; Ardenghi, N.M.G.; Astuti, G.; Bacchetta, G.; Ballelli, S.; Banfi, E.; et al. An updated checklist of the vascular flora native to Italy. Plant Biosyst. 2018, 152, 179–303. [Google Scholar] [CrossRef]
- Galasso, G.; Conti, F.; Peruzzi, L.; Ardenghi, N.M.G.; Banfi, E.; Celesti-Grapow, L.; Albano, A.; Alessandrini, A.; Bacchetta, G.; Ballelli, S.; et al. An updated checklist of the vascular flora alien to Italy. Plant Biosyst. 2018, 152, 556–592. [Google Scholar] [CrossRef]
- Mucina, L.; Bültmann, H.; Dierssen, D.; Theurillat, J.P.; Raus, T.; Čarni, A.; Šumberová, K.; Willner, W.; Dengler, J.; García, R.G. Vegetation of Europe: Hierarchical floristic classification system of vascular plant, bryophyte, lichen, and algal communities. Appl. Veg. Sci. 2016, 9, 3–264. [Google Scholar] [CrossRef]
- Rivas-Martínez, S. Global Bioclimatics. Clasificación Bioclimática de la Tierra. 2004. Available online: http://www.globalbioclimatics.org/book/bioc/bioc1.pdf (accessed on 21 November 2020).
- Biondi, E.; Blasi, C. Prodromo della Vegetazione d’Italia; Check-list sintassonomica aggiornata di classi, ordini e alleanze presenti in Italia; Onlus, Ed.; Società Botanica Italiana: Firenze, Italy, 2013; Available online: http://www.prodromo-vegetazione-italia.org (accessed on 20 November 2020).
- Meyer−Warnod, B. Natural essential oils: Extraction processes and application to some major oils. Perfum. Flavorist 1984, 9, 93–104. [Google Scholar]
- Kováts, E. Gas chromatographische charakterisierung organischer verbindungen. Teil 1: Retentions indices aliphatischer halogenide, alkohole, aldehyde und ketone. Helv. Chim. Acta 1958, 41, 1915–1932. [Google Scholar] [CrossRef]
- Babushok, V.I.; Linstrom, P.J.; Zenkevich, I.G. Retention indices for frequently reported compounds of plant essential oils. J. Phys. Chem. Ref. Data 2011, 40, 3–47. [Google Scholar] [CrossRef] [Green Version]
- Soil Survey Staff. Keys to Soil Taxonomy, 11th ed.; Department of Agriculture: Washington, WA, USA, 2010. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon, and organic matter. In Methods of Soil Analysis; Sparks, D.L., Ed.; Part 3, Chemical Methods, SSSA Book Series n. 5; Soil Science Society of America/American Society of Agronomy: Madison, WI, USA, 1996; pp. 961–1010. [Google Scholar]
- Bremner, J.M. Nitrogen—Total. In Methods of Soil Analysis; Sparks, D.L., Ed.; Part 3, Chemical Methods, SSSA Book Series n. 5; Soil Science Society of America/American Society of Agronomy: Madison, WI, USA, 1996; pp. 1085–1121. [Google Scholar]
- Olsen, S.L.; Sommers, L.E. Phosphorus. Methods of Soil Analysis, 2nd ed.; Chemical and Microbiological Properties, SSSA Book Series; American Society of Agronomy: Madison, WI, USA, 1982; Number 9; pp. 403–427. [Google Scholar]
- Bektas, T.; Cilkiz, M. A pharmacological and phytochemical overview on Satureja. Pharm. Biol. 2016, 54, 375–412. [Google Scholar] [CrossRef] [Green Version]
- Zavatti, M.; Zanoli, P.; Benelli, A.; Rivasi, M.; Baraldi, C.; Baraldi, M. Experimental study on Satureja montana as a treatment for premature ejaculation. J. Ethnopharmacol. 2011, 133, 629–633. [Google Scholar] [CrossRef]
- Momtaz, S.; Abdollahi, M. A Systematic Review of the Biological Activities of Satureja L. Species. Pharmacologyonline 2008, 2, 34–54. [Google Scholar]
- Rustaiyan, A.; Feizbakhsh, A.; Masoudi, S.; Ameri, N. Comparison of the Volatile Oils of Satureja atropatana Bung. and Satureja mutica Fisch. et C.A. Mey. from Iran. J. Essent. Oil. Res. 2004, 16, 594–596. [Google Scholar] [CrossRef]
- Epling, C.C. Studies on South American Libiatae. III. Synopsis of the Genus Satureia. Annals Miss. Bot. Gard. 1927, 14, 47–86. [Google Scholar] [CrossRef]
- Cantino, P.D.; Harley, R.M.; Wagstaff, S.J. Genera of Labiatae status and classification. In Advances in Labiatae Science; Harley, R.M., Reynolds, T., Eds.; Royal Botanic Gardens Press: Kew, UK, 1992; pp. 511–522. [Google Scholar]
- Slavkovska, V.; Jancic, R.; Bojovic, S.; Milosavljevic, S.; Djokovic, D. Variability of essential oils of Satureja montana L. and Satureja kitaibelii Wierzb. ex Heuff. from the central part of the Balkan peninsula. Phytochemistry 2001, 57, 71–76. [Google Scholar] [CrossRef]
- Greuter, W.; Burdet, H.M.; Long, G. Med-Checklist 3: A Critical Inventory of Vascular Plants of the Circum-Mediterranean Countries, Dicotyledones (Convolvulaceae-Labiatae); Conservatoire et Jardin botanique de la Ville Genève: Genève, Swiss, 1986; p. 395. [Google Scholar]
- Bezić, N.; Skočibišić, M.; Dunkić, V. Phytochemical composition and antimicrobial activity of Satureja montana L. and Satureja cuneifolia Ten. essential oils. Acta Bot. Croat. 2005, 64, 313–322. [Google Scholar]
- Bojović, D.; Šoškićb, M.; Tadić, V. Comparative study of chemical composition of the essential oils from Satureja cuneifolia Ten. and Satureja montana L., lamiaceae collected at National Park Lovćen, Montenegro. Stud. Univ. Babes-Bolyai Chem. 2018, 63, 167–180. [Google Scholar] [CrossRef]
- Perrino, E.V.; Ladisa, G.; Calabrese, G. Flora and plant genetic resources of ancient olive groves of Apulia (southern Italy). Genet. Resour. Crop Evol. 2014, 61, 23–53. [Google Scholar] [CrossRef]
- Biondi, E.; Guerra, V. Vegetazione e paesaggio vegetale delle gravine dell’arco jonico. Fitosociologia 2008, 45, 57–125. [Google Scholar]
- Wagensommer, R.P.; Di Pietro, R. Analisi fitosociologica su alcune specie rare e/o minacciate del Parco Nazionale del Gargano (Italia centro-meridionale) e considerazioni sintassonomiche sulle comunità casmofitiche della Puglia. Fitosociologia 2008, 45, 177–200. [Google Scholar]
- Bianco, P.; Brullo, S.; Pignatti, E.; Pignatti, S. La vegetazione delle rupi calcaree della Puglia. Braun Blanquetia 1988, 2, 133–151. [Google Scholar]
- Francini Corti, E. Il Pino d’Aleppo in Puglia. Annali della Facoltà di Agraria dell’Università di Bari 1953, 8, 309–416. [Google Scholar]
- Gambetta, G.; Medagli, P. Guida alla flora del Parco e del territorio materano. In Parco Regionale Della Murgia Materana; Graficom srl: Matera, Italy, 2019; p. 431. [Google Scholar]
- Di Pietro, R.; Wagensommer, R.P. A new Sesleria juncifolia association from south-eastern Italy and its position in the amphi-Adriatic biogeographical context. Acta Bot. Croat. 2014, 73, 171–207. [Google Scholar] [CrossRef] [Green Version]
- Karl, R.; Scholz, H. Med-Checklist Notulae, 28. Willdenowia 2009, 39, 335–345. [Google Scholar]
- Terlević, A.; Koopman, J.; Wieclaw, H.; Rešetnik, I.; Bogdanović, S. Carex phyllostachys (Cyperaceae), a new species in Croatia. Acta Bot. Croat. 2021, 80, 106–111. [Google Scholar] [CrossRef]
- Wagensommer, R.P.; Perrino, E.V.; Silletti, G.N. Carex phyllostachys C.A. Mey. (Cyperaceae) new for Italy and phytogeographical considerations. Phyton 2014, 54, 215–222. [Google Scholar] [CrossRef]
- Wagensommer, R.P.; Fröhlich, T.; Fröhlich, M. First record of the southeast European species Cerinthe retorta Sibth. & Sm. (Boraginaceae) in Italy and considerations on its distribution and conservation status. Acta Bot. Gall. Bot. Lett. 2014, 161, 111–115. [Google Scholar] [CrossRef]
- Wagensommer, R.P.; Bartolucci, F.; Fiorentino, M.; Licht, W.; Peccenini, S.; Perrino, E.V.; Venanzoni, R. First record for the flora of Italy and lectotypification of the name Linum elegans (Linaceae). Phytotaxa 2017, 296, 161–170. [Google Scholar] [CrossRef]
- Perrino, E.V.; Signorile, G.; Marvulli, M. A first checklist of the vascular flora of the “Polignano a Mare” coast (Apulia, southern Italy). Nat. Croat. 2013, 22, 295–318. [Google Scholar]
- Fenu, G.; Bernardo, L.; Calvo, R.; Cortis, P.; De Agostini, A.; Gangale, C.; Gargano, D.; Gargano, M.L.; Lussu, M.; Medagli, P.; et al. Global and Regional IUCN Red List Assessments: 8. Ital. Bot. 2019, 8, 17–33. [Google Scholar] [CrossRef]
- Wagensommer, R.P.; Medagli, P.; Turco, A.; Perrino, E.V. IUCN Red List Evaluation of the Orchidaceae endemic to Apulia Region (Italy) and considerations on the application of the IUCN protocol to rare species. Nat. Conserv. Res. 2020, 5, 90–101. [Google Scholar] [CrossRef]
- Di Pietro, R.; Misano, G. Shrubland and garrigue vegetation in the « Gravine » gorges (Apulia region, south—eastern Italy). Acta Bot. Gall. 2010, 157, 195–229. [Google Scholar] [CrossRef]
- Biondi, E.; Blasi, C.; Burrascano, S.; Casavecchia, S.; Copiz, R.; Del Vico, E.; Galdenzi, D.; Gigante, D.; Lasen, C.; Spampinato, G.; et al. Italian Interpretation Manual of the Habitats (92/43/EEC Directive). Ministry of Environment, Land and Sea Protection. 2010. Available online: http://vnr.unipg.it/habitat/ (accessed on 3 June 2021).
- European Commission DG Environment. Interpretation Manual of European Union Habitats (Version EUR27); European Commission DG Environment: Brussels, Belgium, 2007; Available online: https://ec.europa.eu/environment/nature/legislation/habitatsdirective/docs/2007_07_im.pdf (accessed on 3 June 2021).
- Jafari, F.; Ghavidel, F.; Zarshenas, M.M. A Critical Overview on the Pharmacological and Clinical Aspects of Popular Satureja Species. J. Acupunct. Meridian Study 2016, 9, 118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bezić, N.; Samanić, I.; Dunkić, V.; Besendorfer, V.; Puizina, J. Essential Oil Composition and Internal Transcribed Spacer (ITS) Sequence Variability of Four South-Croatian Satureja Species (Lamiaceae). Molecules 2009, 14, 925–938. [Google Scholar] [CrossRef] [Green Version]
- Musarella, C.M.; Paglianiti, I.; Cano-Ortiz, A.; Spampinato, G. Indagine etnobotanica nel territorio del Poro e delle Preserre Calabresi (Vibo Valentia, S-Italia). Atti Soc. Toscana Sci. Nat. Mem. Ser. B 2019, 126, 13–28. [Google Scholar] [CrossRef]
- Milos, M.; Radonić, A.; Bezić, N.; Dunkić, V. Localities and seasonal variations in the chemical composition of essential oils of Satureja montana L. and S. cuneifolia Ten. Flavour Fragr. J. 2001, 16, 157–160. [Google Scholar] [CrossRef]
- Tommasi, L.; Negro, C.; De Bellis, L.; Miceli, A. Essential Oil Variability of Satureja cuneifolia Ten. Growing Wild in Southern Puglia (Italy). J. Essent. Oil Res. 2008, 20, 295–302. [Google Scholar] [CrossRef]
- De Oliveira, T.L.C.; De Araújo Soares, R.; Ramos, E.M.; Das Graças Cardoso, M.; Alves, E.; Piccoli, R.H. Antimicrobial activity of Satureja montana L. essential oil against Clostridium perfringens type A inoculated in mortadella-type sausages formulated with different levels of sodium nitrite. Int. J. Food Microbiol. 2011, 144, 546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oke, F.; Aslim, B.; Ozturk, S.; Altundag, S. Essential oil composition, antimicrobial and antioxidant activities of Satureja cuneifolia Ten. Food Chem. 2009, 112, 874–879. [Google Scholar] [CrossRef]
- Linnaeus, C. Species Plantarum; Laurentii Salvii: Stockholm, Sweden, 1753. [Google Scholar]
- Bartolucci, F.; Peruzzi, L.; Passalacqua, N. Typification of names and taxonomic notes within the genus Thymus L. (Lamiaceae). Taxon 2013, 62, 1308–1314. [Google Scholar] [CrossRef]
- Morales, R. The history, botany and taxonomy of the genus Thymus. In Thyme: The Genus Thymus; Stahl-Biskup, E., Sáez, F., Eds.; Taylor & Francis: London, UK; New York, NY, USA, 2002; pp. 1–43. [Google Scholar]
- Bartolucci, F.; Walter, J. Typification of names in the genus Thymus (Lamiaceae). Phytotaxa 2015, 221, 137–147. [Google Scholar] [CrossRef]
- Bartolucci, F.; Mráz, P. A new combination in the genus Thymus and clarification on the type material of T. thracicus (Lamiaceae). Phytotaxa 2016, 263, 161–164. [Google Scholar] [CrossRef]
- Morales, R. Synopsis of the genus Thymus L. in the Mediterranean area. Lagascalia 1997, 19, 249–262. [Google Scholar]
- Perrino, E.V.; Tomaselli, V.; Costa, R.; Pavone, P. Conservation status of habitats (Directive 92/43 EEC) of coastal and low hill belts in a mediterranean biodiversity hot spot (Gargano-Italy). Plant Biosyst. 2013, 147, 1006–1028. [Google Scholar] [CrossRef]
- Alekseeva, L.I.; Gruzdev, I.V. Polymorphism of Essential Oils in Thyme Species Growing in European Part of North East Russia and Ural. Russ. J. Plant Physiol. 2012, 59, 818–827. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.; Jafarikukhdan, A.; Hosseini, A.; Armand, R. The application of Medicinal Plants in Tradicional and Modern Medicine: A Review of Thymus vulgaris. Int. J. Clin. Med. 2015, 6, 635–642. [Google Scholar] [CrossRef] [Green Version]
- Jarić, S.; Mitrović, M.; Pavlović, P. Review of Ethnobotanical, Phytochemical, and Pharmacological Study of Thymus serpyllum L. Evid. Based Complement. Altern. Med. 2015, 2015, 101978. [Google Scholar]
- Bhat, M.N.; Singh, B.; Surmal, O.; Singh, B.; Shivgotra, V.; Musarella, C.M. Knowledge and practices of ethnobotany in developing countries: Validation vs traditional use of plant resources of Kashmir Himalaya, India. Biology 2021, 10, 851. [Google Scholar] [CrossRef]
- Maruca, G.; Spampinato, G.; Turiano, D.; Laghetti, G.; Musarella, C.M. Ethnobotanical notes about medicinal and useful 1110 plants of the Reventino Massif tradition (Calabria region, Southern Italy). Genet. Resour. Crop Evol. 2019, 66, 1027–1040. [Google Scholar] [CrossRef]
- Koul, O.; Walia, S.; Dhaliwal, G.S. Essential oils as green pesticides: Potential and constraints. Biopestic. Int. 2008, 4, 63–84. [Google Scholar]
- Chan, W.K.; Tan, L.T.H.; Chan, K.G.; Lee, L.H.; Goh, B.H. Nerolidol: A sesquiterpene alcohol with multi-faceted pharmacological and biological activities. Molecules 2016, 21, 529. [Google Scholar] [CrossRef] [Green Version]
- De Feo, V.; Bruno, M.; Tahiri, B.; Napolitano, F.; Senatore, F. Chemical composition and antibacterial activity of essential oils from Thymus spinulosus Ten. (Lamiaceae). J. Agric. Food Chem. 2003, 51, 3849–3853. [Google Scholar] [CrossRef]
- Desneux, N.; Decourtye, A.; Delpuech, J.M. The sublethal effects of pesticides on beneficial arthropods. Annu. Rev. Entomol. 2007, 52, 81–106. [Google Scholar] [CrossRef] [PubMed]
- Guedes, R.N.C.; Smagghe, G.; Stark, J.D.; Desneux, N. Pesticide induced stress in arthropod pests for optimized integrated pest management programs. Annu. Rev. Entomol. 2016, 61, 43–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohammed, A.A.A.; Desneux, N.; Fan, Y.; Han, P.; Ali, A.; Song, D.; Gao, X.W. Impact of imidacloprid and natural enemies on cereal aphids: Integration or ecosystem service disruption? Entomol. Gen. 2018, 37, 47–61. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.L.; Tang, Q.L.; Li, Y.D.; Campos, M.R.; Liang, P.; Gao, X.W. Widespread resistance of the aphid Myzus persicae to pirimicarb across China, and insights on ace2 mutation frequency in this species. Entomol. Gen. 2017, 36, 285–299. [Google Scholar]
- Pavela, R.; Bartolucci, F.; Desneux, N.; Lavoir, A.V.; Canale, A.; Maggi, F.; Benelli, G. Chemical profiles and insecticidal efficacy of the essential oils from four Thymus taxa growing in central-southern Italy. Ind. Crops Prod. 2019, 138, 111460. [Google Scholar] [CrossRef]
- De Natale, A.; Menale, B.; Di Lecce, S.; Pollio, A. An ethnobotanical survey of wild edible plants of Campania (Italy). Bull. Reg. Nat. Hist. 2021, 1, 1–122. [Google Scholar]
- Vernet, P.; Gouyon, P.H.; Valdeyron, G. Genetic control of the oil content in Thymus vulgaris: A case of polymorphism in a biosynthetic chain. Genetica 1986, 69, 227–231. [Google Scholar] [CrossRef]
- Vokou, D.; Margaris, N.S. Volatile oils as allelopathic agents. In Aromatic Plants: Basic and Applied Aspects; Margaris, N.S., Koldam, A., Vokou, D., Eds.; Martinus Nijhoff: The Hague, The Netherlands, 1982; pp. 59–72. [Google Scholar]
- Senatore, F. Influence of harvesting time on yield and composition of the essential oil of a Thyme (Thymus pulegioides L.) growing wild in Campania (Southern Italy). J. Agric. Food Chem. 1996, 44, 1327–1332. [Google Scholar] [CrossRef]
- Piñar Fuentes, J.C.; Leiva, F.; Cano-Ortiz, A.; Musarella, C.M.; Quinto-Canas, R.; Pinto-Gomes, C.J.; Cano, E. Impact of Grass Cover Management with Herbicides on Biodiversity, Soil Cover and Humidity in Olive Groves in the Southern Iberian. Agronomy 2021, 11, 412. [Google Scholar] [CrossRef]
- Raposo, M.A.M.; Gomes, C.J.P.; Nunes, L.J.R. Selective Shrub Management to Preserve Mediterranean Forests and Reduce the Risk of Fire: The Case of Mainland Portugal. Fire 2020, 3, 65. [Google Scholar] [CrossRef]



| Site (IC) | Macroclimate | Bioclimate | Ombrotype | Italian Distribution |
|---|---|---|---|---|
| S. Egidio (Ts3) | Temperate | Oceanic-semicontinental transition | Subhumid | Hilly areas of Apennines with Adriatic exposure |
| Scannapecora (Ts2) | Mediterranean | Coastal areas of the middle Adriatic, of the inland plains of the pre-Apennines and of Sicily | ||
| San Basilio (Sc2, Ts1) | Oceanic | Areas of the middle and lower Adriatic, the Ionian and the major islands; moderate presence also in the middle and upper Tyrrhenian regions | ||
| Difesa di Malta (Sc1) | Mediterranean oceanic | Dry |
| Site (IC) | Geo-Pedology and Geolithology | Ecopedology |
|---|---|---|
| S. Egidio (Ts3) | Colluvial, terraced alluvial, fluviolacustrine and fluvioglacial deposits (Pleistocene) | Tyrrhenian carbonate reliefs with material defined by calcareous sedimentary rocks and climate from Oceanic to Suboceanic Mediterranean partially mountainous |
| Scannapecora (Ts2) | Skeletal limestones of neritic and carbonate platform facies (Upper Cretaceous) | |
| San Basilio (Sc2, Ts1) | ||
| Difesa di Malta (Sc1) | Terrigenous-skeletal limestones such as “Panchina” (Pleistocene) | Hilly reliefs with undifferentiated tertiary sedimentary rocks and sub-continental Mediterranean to continental Mediterranean climate |
| IC | Taxon | Location | Date Retrieved | Geographic Position (WGS84) |
|---|---|---|---|---|
| Sc1 | Satureja cuneifolia Ten. | Difesa di Malta (Fasano–Brindisi) | 03.06.20 | 40.7986 N 17.4946 E |
| Sc2 | San Basilio (Mottola–Taranto) | 26.05.20 | 40.6877 N 16.9694 E | |
| Ts1 | Thymus spinulosus Ten. | San Basilio (Mottola–Taranto) | 05.06.20 | 40.6877 N 16.9693 E |
| Ts2 | Scannapecora (Altamura–Bari) | 03.06.20 | 40.9051 N 16.4422 E | |
| Ts3 | S. Egidio (S. Giovanni Rotondo–Foggia) | 12.06.20 | 41.7132 N 15.7658 E |
| Identification Code | Sc1 | Sc2 | |
|---|---|---|---|
| Altitude (m a. s. l.) | endemic | 49 | 274 |
| Aspect | NE | N | |
| Slope (°) | 3 | 4 | |
| Relevé area (m2) | 20 | 25 | |
| Stoniness (%) | 20 | 10 | |
| Rockiness (%) | 60 | 50 | |
| Cover total (%) | 80 | 70 | |
| Number of species | 23 | 41 | |
| Number of individuals collected for laboratory analysis | 15 | 50 | |
| Geolithology | § | §§ | |
| Ecopedology | † | †† | |
| Macroclimate | ≠ | ≠ | |
| Bioclimate | ◊ | ◊◊ | |
| Ombrotype | # | ## | |
| Other species | 10 | 10 | |
| Characteristics association Phagnalo saxatilii-Saturejetum cuneifoliae | |||
| Satureja cuneifolia Ten. | 3 | 3 | |
| Phagnalon saxatile (L.) Cass. | + | - | |
| Characteristics Alliance Cisto cretici-Ericion manipuliflorae, Order Cisto-Micromerietalia julianae, Class Ononido-Rosmarinetea | |||
| Helianthemum jonium Lacaita & Grosser | 1 | 1 | |
| Micromeria graeca (L.) Benth. ex Rchb. | 1 | + | |
| Asphodeline lutea (L.) Rchb. | - | 1 | |
| Characteristics Alliance Hippocrepido glaucae-Stipion austroitalicae, Order Scorzoneretalia villosae | |||
| Convolvulus cantabrica L. | - | 2 | |
| Euphorbia nicaeensis All. subsp. japygica (Ten.) Arcang. | - | 2 | |
| Teucrium capitatum L. subsp. capitatum | - | 2 | |
| Petrorhagia saxifraga (L.) Link subsp. gasparrinii (Guss.) Greuter & Burdet | - | 1 | |
| Eryngium amethystinum L. | - | + | |
| Linum tommasinii (Rchb.) Nyman | - | + | |
| Phleum hirsutum Honck. subsp. ambiguum (Ten.) Cif. & Giacom. | - | + | |
| Scorzonera villosa Scop. subsp. columnae (Guss.) Nyman | E | - | + |
| Thesium humile Vahl | - | + | |
| Thymus spinulosus Ten. | E | - | + |
| Characteristics Class Festuco-Brometea | |||
| Centaurea deusta Ten. | - | 2 | |
| Anacamptis morio (L.) R.M. Bateman, Pridgeon & M.W.Chase | - | + | |
| Galium corrudifolium Vill. | - | + | |
| Poterium sanguisorba L. | - | + | |
| Transgressive Class Helianthemetea guttati | |||
| Stachys romana (L.) E.H.L. Krause | 1 | 1 | |
| Trifolium scabrum L. | 1 | 1 | |
| Lagurus ovatus L. subsp. ovatus | + | 1 | |
| Hypochaeris achyrophorus L. | + | + | |
| Alkanna tinctoria Tausch subsp. tinctoria | - | 2 | |
| Catapodium rigidum (L.) C.E. Hubb. | + | - | |
| Aegilops ovata auct. | - | + | |
| Festuca danthonii Asch. & Graebn. subsp. danthonii | - | + | |
| Medicago truncatula Gaertn. | - | + | |
| Plantago coronopus L. | - | + | |
| Stipellula capensis (Thunb.) Röser & H.R. Hamasha | - | + | |
| Transgressive Class Lygeo sparti-Stipetea tenacissimae | |||
| Daucus carota L. subsp. carota | 1 | + | |
| Asphodelus ramosus L. subsp. ramosus | + | - | |
| Convolvulus elegantissimus Mill. | + | - | |
| Pallenis spinosa (L.) Cass. subsp. spinosa | + | - | |
| Ferula communis L. subsp. communis | - | + | |
| Reichardia picroides (L.) Roth | - | + | |
| Legend E = Endemic species | |||
| Geopedology and Geolithology | |||
| §—Terrigenus-skeletal limestones like “Panchina” (Pleistocene) | |||
| §§—Skeletal limestones of neritic and carbonate platform facies (Upper Cretaceous) | |||
| §§§—Colluvial, terraced alluvial, fluviolacustrine and fluvioglacial deposits (Pleistocene) | |||
| Ecopedology | |||
| †—Hilly reliefs with undifferentiated tertiary sedimentary rocks and sub-continental Mediterranean to continental Mediterranean climate | |||
| ††—Tyrrhenian carbonate reliefs with material defined by calcareous sedimentary rocks and climate from Oceanic to Suboceanic Mediterranean, partially mountainous | |||
| Macroclimate | |||
| ≠—Mediterranean | |||
| ≠≠—Temperate | |||
| Bioclimate | |||
| ◊—Mediterranean Oceanic | |||
| ◊◊—Oceanic | |||
| ◊◊◊—Oceanic-semicontinental transition | |||
| Ombrotype | |||
| #—Dry ##—Subhumid | |||
| Soil Parameters | Unit | Sc1 | Sc2 | Ts1 | Ts2 | Ts3 |
|---|---|---|---|---|---|---|
| Sand (2–0.05 mm) | g∙kg−1 | 307 | 576 | 783 | 200 | 161 |
| Silt (0.05–0.002 mm) | g∙kg−1 | 649 | 383 | 193 | 759 | 787 |
| Clay (<0.002 mm) | g∙kg−1 | 44 | 41 | 23 | 41 | 51 |
| Texture Class (USDA) | Silt loam | Sandy loam | Loam sand | Silt loam | Silt loam | |
| Total Carbonate | g∙kg−1 | 306 | 698 | 965 | 19 | <0.1 |
| pH H2O | - | 8.1 | 8.2 | 7.9 | 7.9 | 6.9 |
| pH CaCl2 | - | 7.5 | 7.4 | 7.5 | 7.4 | 6.4 |
| Electrical Conductivity (1:2) | dS∙m−1 | 0.2 | 0.2 | 0.3 | 0.2 | 0.2 |
| Total Nitrogen | g∙kg−1 | 6.0 | 3.5 | 3.7 | 6.8 | 6.7 |
| Available P2O5 | mg∙kg−1 | 103 | 43 | 88 | 27 | 24 |
| Organic Carbon | g∙kg−1 | 63 | 34 | 42 | 62 | 69 |
| Organic Matter | g∙kg−1 | 109 | 59 | 72 | 107 | 119 |
| C/N | - | 10.5 | 10 | 11.2 | 9.2 | 10.3 |
| Ca exchangeable | mg∙kg−1 | 9772 | 4842 | 4146 | 10,450 | 7711 |
| K exchangeable | mg∙kg−1 | 253 | 93 | 87 | 515 | 855 |
| Mg exchangeable | mg∙kg−1 | 240 | 131 | 105 | 231 | 368 |
| Na exchangeable | mg∙kg−1 | 68 | 40 | 15 | 85 | 81 |
| Available Fe | mg∙kg−1 | 13.1 | 7.8 | 8.9 | 20.3 | 31.9 |
| Available Mn | mg∙kg−1 | 7.9 | 15.1 | 7.6 | 8.1 | 19.9 |
| Available Zn | mg∙kg−1 | 2.5 | 20.5 | 8.9 | 1.6 | 3.0 |
| Available Cu | mg∙kg−1 | 0.7 | 1.0 | 0.9 | 1.9 | 1.9 |
| Available Ni | mg∙kg−1 | 0.27 | 0.27 | 0.14 | 0.16 | 0.21 |
| Available Co | mg∙kg−1 | 0.03 | 0.06 | 0.03 | 0.03 | 0.11 |
| Compound Name | RT | RI | RI ref. | Sc1 | Sc2 | Ts1 | Ts2 | Ts3 |
|---|---|---|---|---|---|---|---|---|
| α-Thujene | 6.54 | 925 | 927 | - | - | 1.84 | 1.59 | 1.84 |
| α-Pinene | 6.74 | 932 | 936 | 36.8 | 38.82 | 0.95 | 0.73 | 0.83 |
| Camphene | 7.16 | 949 | 950 | 1.66 | 0.49 | 0.52 | 0.28 | 0.28 |
| 2,4(10)-Thujadiene | 7.25 | 952 | 943 | 1.28 | 0.36 | - | - | - |
| Sabinene | 7.75 | 971 | 973 | 0.74 | 1.71 | 0.13 | 0.09 | 0.08 |
| β-Pinene | 7.89 | 977 | 977 | 0.36 | 0.38 | 0.87 | 0.72 | 0.94 |
| α-Myrcene | 8.16 | 987 | 989 | 0.84 | 1.43 | 1.33 | 1.1 | 0.83 |
| 2,3-Dehydro-1,8-cineole | 8.19 | 989 | 989 | 1.78 | 0.99 | - | - | - |
| 3-Octanol | 8.34 | 994 | 993 | 0.09 | 0.07 | - | - | - |
| α-Phellandrene | 8.63 | 1005 | 1004 | 0.81 | 0.71 | 0.18 | 0.14 | 0.18 |
| α-Terpinene | 8.94 | 1016 | 1017 | 0.14 | 0.11 | 2.24 | 1.84 | 2.9 |
| p-Cymene | 9.15 | 1023 | 1024 | 1.55 | 0.63 | 17.87 | 17.51 | 17.5 |
| Limonene | 9.29 | 1028 | 1029 | 5.08 | 6.39 | 0.51 | 0.41 | 1.43 |
| β-Ocimene. (Z) | 9.41 | 1033 | 1037 | 2.8 | 3.62 | 2.48 | 1.14 | 0.9 |
| β-Ocimene. (E) | 9.74 | 1044 | 1047 | 1.17 | 1.52 | 15.4 | 11.68 | 10.97 |
| γ-Terpinene | 10.10 | 1057 | 1060 | 0.25 | 0.19 | - | - | - |
| Sabinene hydrate, cis- | 10.43 | 1069 | 1066 | 0.12 | 0.1 | 0.45 | 0.49 | 0.52 |
| Terpinolene | 10.88 | 1085 | 1087 | 0.61 | 0.3 | 0.14 | 0.14 | 0.14 |
| Linalool | 11.25 | 1098 | 1099 | 6.35 | 6.36 | 1.83 | 2.41 | 2.9 |
| Hotrienol | 11.34 | 1101 | 1106 | 0.12 | 0.13 | 0.11 | - | - |
| Thujone, cis | 11.45 | 1104 | 1105 | 0.1 | 0.05 | 0.06 | 0.49 | 0.05 |
| Chrysanthenone | 11.89 | 1120 | 1124 | 0.31 | 0.1 | - | - | - |
| α-Campholenal | 12.00 | 1124 | 1124 | 1.05 | 0.33 | 0.05 | 0.05 | 0.19 |
| Pinocarveol, trans- | 12.47 | 1141 | 1140 | 1.78 | 0.78 | - | - | - |
| trans-3-caren-2-ol | 12.58 | 1146 | 4.1 | 1.9 | - | - | - | |
| Pinocarvone | 13.05 | 1160 | 1160 | 0.33 | 0.12 | - | - | - |
| Borneol | 13.31 | 1170 | 1166 | 6.9 | 1.4 | 1 | 0.5 | 0.5 |
| α-Terpineol | 13.97 | 1193 | 1190 | 11.03 | 17.11 | 0.29 | 0.22 | 0.19 |
| Carveol, trans- | 14.60 | 1217 | 1217 | 0.92 | 0.35 | - | - | - |
| Carvacrol, methyl ether | 15.16 | 1238 | 1243 | - | - | 2.05 | 3.19 | 0.54 |
| Bornyl acetate | 16.42 | 1284 | 1283 | 0.21 | 0.07 | - | - | - |
| Thymol | 16.58 | 1289 | 1290 | - | - | 42.87 | 48.77 | 45.88 |
| Carvacrol | 16.77 | 1297 | 1300 | - | - | 0.94 | 0.62 | 2 |
| Caryophyllene, (E) | 20.01 | 1422 | 1420 | 0.07 | 0.08 | 2.07 | 1.22 | 2.22 |
| Aromadendrene | 20.48 | 1441 | 1440 | 0.21 | 0.35 | - | - | - |
| γ-Curcumene | 21.39 | 1478 | 1480 | 0.16 | 0.18 | - | - | - |
| α-Bisabolene, (Z) | 21.88 | 1497 | 1503 | 1.52 | 2.6 | - | - | - |
| γ-Bisabolene | 22.14 | 1509 | 1508 | 0.2 | 0.39 | 0.96 | 1.05 | 0.73 |
| Caryophyllene oxide | 23.75 | 1581 | 1580 | 3.25 | 2.44 | - | 0.14 | - |
| Guaia-1(10),11-diene | 24.81 | 1637 | - | 0.31 | 0.25 | 0.38 | 0.37 | 0.51 |
| Other components | 5 | 7.19 | 2.48 | 3.11 | 4.95 |
| Identification Code | Ts1 | Ts2 | Ts3 | |
|---|---|---|---|---|
| Altitude (m a. s.l.) | e n d e m i c | 274 | 561 | 626 |
| Aspect | N-NE | S-SW | SE | |
| Slope (°) | 3 | 10 | 12 | |
| Relevé area (m2) | 50 | 50 | 50 | |
| Stoniness (%) | 5 | 20 | 10 | |
| Rockiness (%) | 60 | 40 | 45 | |
| Cover total (%) | 70 | 70 | 85 | |
| Number of species | 38 | 44 | 64 | |
| Number of individuals collected for laboratory analysis | 70 | 80 | 60 | |
| Geolithology | §§ | §§ | §§§ | |
| Ecopedology | †† | †† | †† | |
| Macroclimate | ≠ | ≠ | ≠≠ | |
| Bioclimate | ◊◊ | ◊◊◊ | ◊◊◊ | |
| Ombrotype | ## | ## | ## | |
| Other species | 12 | 9 | 22 | |
| Characteristics Association Sideritido syriacae-Stipetum austroitalicae | ||||
| Sideritis italica Mill. | - | - | 2 | |
| Characteristics Association Acino suaveolentis-Stipetum austroitalicae | ||||
| Thymus spinulosus Ten. | E | 2 | 2 | 1 |
| Euphorbia nicaeensis All. subsp. japygica (Ten.) Arcang. | E | 1 | + | - |
| Clinopodium suaveolens (Sm.) Kuntze | - | 1 | - | |
| Characteristics Alliance Hippocrepido glaucae-Stipion austroitalicae | ||||
| Stipa austroitalica Martinovský subsp. austroitalica | E | + | 3 | 2 |
| Petrorhagia saxifraga (L.) Link subsp. gasparrinii (Guss.) Greuter & Burdet | 1 | 1 | + | |
| Linum tommasinii (Rchb.) Nyman | + | - | - | |
| Hippocrepis glauca Ten. | - | - | + | |
| Characteristics Order Scorzoneretalia villosae | ||||
| Teucrium capitatum L. subsp. capitatum | 1 | 1 | 1 | |
| Thesium humile Vahl | + | 1 | - | |
| Phleum hirsutum Honck. subsp. ambiguum (Ten.) Cif. & Giacom. | 1 | - | 3 | |
| Convolvulus cantabrica L. | 1 | - | + | |
| Scorzonera villosa Scop. subsp. columnae (Guss.) Nyman | E | - | + | 2 |
| Koeleria splendens C. Presl | E | - | 1 | + |
| Eryngium amethystinum L. | - | + | 1 | |
| Satureja montana L. subsp. montana | - | 4 | - | |
| Anthyllis vulneraria L. subsp. rubriflora (DC.) Arcang. | - | 1 | - | |
| Teucrium capitatum L. subsp. capitatum | 1 | 1 | 1 | |
| Characteristic Class Festuco-Brometea | ||||
| Eryngium campestre L. | + | 2 | - | |
| Centaurea deusta Ten. | + | - | + | |
| Festuca circummediterranea Patzke | - | 2 | + | |
| Poterium sanguisorba L. | - | + | 1 | |
| Filipendula vulgaris Moench | - | - | + | |
| Euphorbia spinosa L. | - | - | 1 | |
| Helianthemum oelandicum (L.) Dum. Cours. subsp. incanum (Willk.) G. López | - | - | 1 | |
| Melica ciliata L. subsp. ciliata | - | - | + | |
| Potentilla calabra Ten. | E | - | - | + |
| Trifolium arvense L. | - | - | + | |
| Trifolium campestre Schreb. | - | - | + | |
| Transgressive Class Helianthemetea guttati | ||||
| Lagurus ovatus L. | 1 | 1 | 2 | |
| Aegilops ovata auct. | + | 1 | + | |
| Hypochaeris achyrophorus L. | + | 1 | + | |
| Stachys romana (L.) E.H.L. Krause | + | 1 | + | |
| Trifolium scabrum L. | 1 | 1 | - | |
| Alkanna tinctoria Tausch subsp. tinctoria | 1 | + | - | |
| Ononis reclinata L. | + | + | - | |
| Stipellula capensis (Thunb.) Röser & H.R. Hamasha | + | + | - | |
| Avena barbata Pott ex Link | + | - | + | |
| Trifolium stellatum L. | + | - | + | |
| Linum strictum L. | - | + | 1 | |
| Brachypodium distachyon (L.) P. Beauv. | - | + | + | |
| Helianthemum salicifolium (L.) Mill. | 1 | - | - | |
| Onobrychis aequidentata (Sm.) d’Urv. | - | + | - | |
| Briza maxima L. | - | - | 1 | |
| Hippocrepis ciliata Willd. | - | - | 1 | |
| Transgressive Class Lygeo sparti-Stipetea tenacissimae | ||||
| Asphodelus ramosus L. subsp. ramosus | + | 2 | 2 | |
| Reichardia picroides (L.) Roth | 1 | - | + | |
| Daucus carota L. | + | - | + | |
| Carlina corymbosa L. | - | + | + | |
| Convolvulus elegantissimus Mill. | - | + | + | |
| Petrosedum ochroleucum (Chaix) Niederle | - | + | + | |
| Ferula communis L. subsp. communis | + | - | - | |
| Plantago bellardii All. subsp. bellardii | - | 1 | - | |
| Dactylis glomerata L. subsp. hispanica (Roth) Nyman | - | - | + | |
| Pallenis spinosa (L.) Cass. subsp. spinosa | - | - | + | |
| Phlomis herba-venti L. subsp. herba-venti | - | - | + | |
| Transgressive Class Ononido-Rosmarinetea | ||||
| Micromeria graeca (L.) Benth. ex Rchb. | - | 1 | 1 | |
| Satureja cuneifolia Ten. | - | 3 | - | |
| Helianthemum jonium Lacaita & Grosser | - | 2 | - | |
| Rhamnus saxatilis Jacq. | - | + | - | |
| Legend E = Endemic species | ||||
| Geopedology and Geolithology | ||||
| §—Terrigenus-skeletal limestones like “Panchina” (Pleistocene) | ||||
| §§—Skeletal limestones of neritic and carbonate platform facies (Upper Cretaceous) | ||||
| §§§—Colluvial, terraced alluvial, fluviolacustrine and fluvioglacial deposits (Pleistocene) | ||||
| Ecopedology | ||||
| †—Hilly reliefs with undifferentiated tertiary sedimentary rocks and sub-continental Mediterranean to continental Mediterranean climate | ||||
| ††—Tyrrhenian carbonate reliefs with material defined by calcareous sedimentary rocks and climate from Oceanic to Suboceanic Mediterranean, partially mountainous | ||||
| Macroclimate | ||||
| ≠—Mediterranean | ||||
| ≠≠—Temperate | ||||
| Bioclimate | ||||
| ◊—Mediterranean Oceanic | ||||
| ◊◊—Oceanic | ||||
| ◊◊◊—Oceanic-semicontinental transition | ||||
| Ombrotype | ||||
| #—Dry ##—Subhumid | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perrino, E.V.; Valerio, F.; Jallali, S.; Trani, A.; Mezzapesa, G.N. Ecological and Biological Properties of Satureja cuneifolia Ten. and Thymus spinulosus Ten.: Two Wild Officinal Species of Conservation Concern in Apulia (Italy). A Preliminary Survey. Plants 2021, 10, 1952. https://doi.org/10.3390/plants10091952
Perrino EV, Valerio F, Jallali S, Trani A, Mezzapesa GN. Ecological and Biological Properties of Satureja cuneifolia Ten. and Thymus spinulosus Ten.: Two Wild Officinal Species of Conservation Concern in Apulia (Italy). A Preliminary Survey. Plants. 2021; 10(9):1952. https://doi.org/10.3390/plants10091952
Chicago/Turabian StylePerrino, Enrico V., Francesca Valerio, Shaima Jallali, Antonio Trani, and Giuseppe N. Mezzapesa. 2021. "Ecological and Biological Properties of Satureja cuneifolia Ten. and Thymus spinulosus Ten.: Two Wild Officinal Species of Conservation Concern in Apulia (Italy). A Preliminary Survey" Plants 10, no. 9: 1952. https://doi.org/10.3390/plants10091952
APA StylePerrino, E. V., Valerio, F., Jallali, S., Trani, A., & Mezzapesa, G. N. (2021). Ecological and Biological Properties of Satureja cuneifolia Ten. and Thymus spinulosus Ten.: Two Wild Officinal Species of Conservation Concern in Apulia (Italy). A Preliminary Survey. Plants, 10(9), 1952. https://doi.org/10.3390/plants10091952