Growth and Competitiveness of ALS-Inhibiting Herbicide-Resistant Amaranthus retroflexus L.
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Germination Assay
2.3. Noncompetitive Study
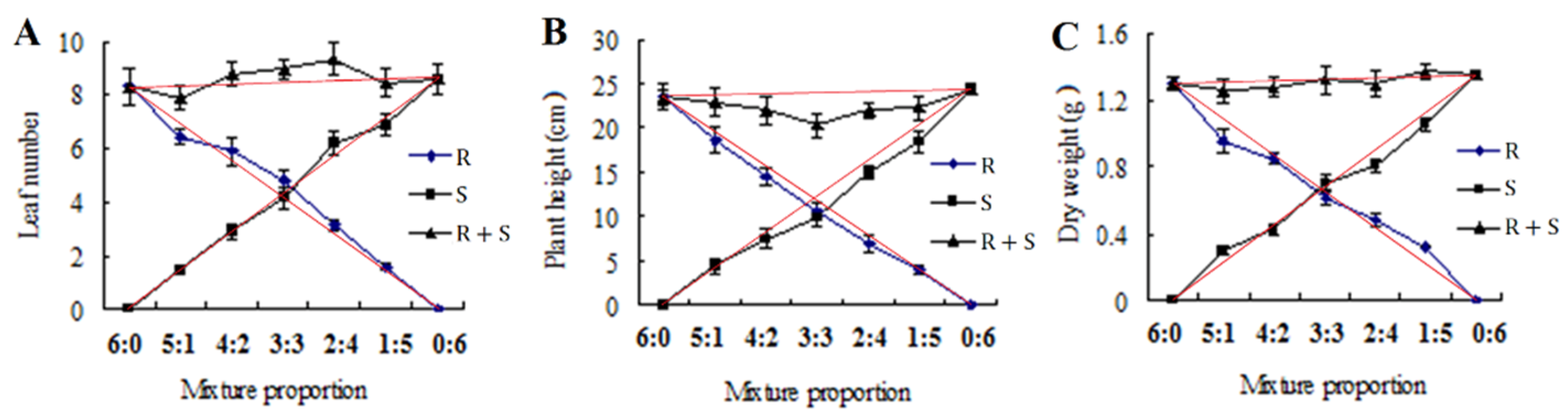
2.4. Competitive Study
2.5. Statistical Analysis
3. Results
3.1. Germination Experiment
3.2. Noncompetitive Study
3.3. Competitive Study
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cao, Y.; Zhou, X.X.; Huang, Z.F. Amino acid substitution (Gly-654-Tyr) in acetolactate synthase (ALS) confers broad spectrum resistance to ALS-inhibiting herbicides. Pest Manag. Sci. 2022, 78, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Heap, I.M. The International Survey of Herbicide Resistant Weeds. Available online: http://www.weedscience.com (accessed on 30 August 2022).
- Mazur, B.J.; Falco, S.C. The development of herbicide resistant crops. Annu. Rev. Plant Biol. 1989, 40, 441–470. [Google Scholar] [CrossRef]
- Chen, J.Y.; Huang, Z.F.; Zhang, C.X.; Huang, H.J.; Wei, S.H.; Chen, J.C.; Wang, X. Molecular basis of resistance to imazethapyr in redroot pigweed (Amaranthus retroflexus L.) populations from China. Pestic. Biochem. Phys. 2015, 124, 43–47. [Google Scholar] [CrossRef]
- Tranel, P.J.; Wright, T.R. Resistance of weeds to ALS-inhibiting herbicides: What have we learned? Weed Sci. 2002, 50, 700–712. [Google Scholar] [CrossRef]
- Maxwell, B.D.; Roush, M.L.; Radosevich, S.R. Predicting the evolution and dynamics of herbicide resistance in weed populations. Weed Technol. 1990, 4, 2–13. [Google Scholar] [CrossRef]
- Vila-Aiub, M.M.; Neve, P.; Steadman, K.J.; Powles, S.B. Ecological fitness of a multiple herbicide-resistant Lolium rigidum population: Dynamics of seed germination and seedling emergence of resistant and susceptible phenotypes. J. Appl. Ecol. 2005, 42, 288–298. [Google Scholar] [CrossRef]
- Yu, Q.; Han, H.P.; Vila-aiub, M.M.; Powles, S.B. AHAS herbicide resistance endowing mutations: Effect on AHAS functionality and plant growth. J. Exp. Bot. 2010, 61, 3925–3934. [Google Scholar] [CrossRef]
- Westhoven, A.M.; Kruger, G.R.; Gerber, C.K.; Stachler, J.M.; Loux, M.M.; Johnson, W.G. Characterization of selected common lambsquarters (Chenopodium album) populations with tolerance to glyphosate. Weed Sci. 2008, 56, 685–691. [Google Scholar] [CrossRef]
- Li, M.; Yu, Q.; Han, H.P.; Vila-aiub, M.M.; Powles, S.B. ALS herbicide resistance mutations in Raphanus raphanistrum: Evaluation of pleiotropic effects on vegetative growth and ALS activity. Pest Manag. Sci. 2013, 69, 689–695. [Google Scholar] [CrossRef] [PubMed]
- Travlos, I.S.; Chachalis, D. Relative competitiveness of glyphosate-resistant and glyphosate susceptible populations of hairy fleabane, Conyza bonariensis. J. Pest. Sci. 2013, 86, 345–351. [Google Scholar] [CrossRef]
- Alcocer-Ruthling, M.; Thill, D.C.; Shafii, B. Differential competitiveness of sulfonylurea resistant and susceptible prickly lettuce (Lactuca serriola). Weed Technol. 1992, 6, 303–309. [Google Scholar] [CrossRef]
- Vaughn, K.C. Herbicide resistance work in the United States Department of Agriculture—Agricultural research service. Pest Manag. Sci. 2003, 59, 764–769. [Google Scholar] [CrossRef] [PubMed]
- Schaedler, C.E.; Borgos, N.R.; Noldin, J.A.; Alcober, E.A.; Salas, R.A.; Agostinetto, D. Competitive ability of ALS-inhibitor herbicide-resistant Fimbristylis miliacea. Weed Res. 2015, 55, 482–492. [Google Scholar] [CrossRef]
- Marisa, A.; Matthew, W.F.; Kerri, L.S.; Anil, S. Competitive effects of glyphosate-resistant and glyphosate-susceptible horseweed (Conyza canadensis) on young grapevines (Vitis vinifera). Weed Sci. 2011, 59, 489–494. [Google Scholar]
- Huang, Z.F.; Chen, J.Y.; Zhang, C.X.; Huang, H.J.; Wei, S.H.; Zhou, X.X.; Chen, J.; Wang, X. Target-site basis for resistance to imazethapyr in redroot amaranth (Amaranthus retroflexus L.). Pestic. Biochem. Phys. 2016, 128, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Novak, M.G.; Higley, L.G.; Christianssen, C.A.; Rowley, W.A. Evaluating larval competition between Aedes albopictus and A. triseriatus (Diptera: Culicidae) through replacement series experiments. Environ. Entomol. 1993, 22, 311–318. [Google Scholar] [CrossRef]
- O’Donovan, J.T.; Newman, J.C.; Blackshaw, R.E.; Harker, K.N.; Derksen, D.A.; Thomas, A.G. Growth, competitiveness, and seed germination of triallate/difenzoquat-susceptible and -resistant wild oat populations. Can. J. Plant Sci. 1999, 79, 303–312. [Google Scholar] [CrossRef]
- Abdul-Baki, A.A.; Anderson, J.D. Vigour determination in soybean seed by multiple criteria. Crop Sci. 1973, 13, 630–633. [Google Scholar] [CrossRef]
- Cousens, R.; O’Neill, M. Density dependence of replacement series experiments. Oikos 1993, 66, 347–352. [Google Scholar] [CrossRef]
- Hoffman, M.L.; Buhler, D.D. Utilizing sorghum as a functional model of crop-weed competition. I. Establishing a competitive hierarchy. Weed Sci. 2002, 50, 466–472. [Google Scholar] [CrossRef]
- Dal, M.T.; Schaedler, C.E.; Fontana, L.C.; Agostinetto, D.; Vargas, L. Competitive ability between biotypes of Cyperus difformis L. resistant or susceptible to ALS-inhibitor herbicide and those with flooded rice. Bragantia 2011, 70, 294–301. [Google Scholar]
- Marshall, M.W.; Al-Khatib, K.; Loughin, T. Gene flow, growth, and competitiveness of imazethapyr-resistant common sunflower. Weed Sci. 2001, 49, 14–21. [Google Scholar] [CrossRef]
- Shrestha, A.; Hanson, B.D.; Fidelibus, M.W.; Alcorta, M. Growth, phenology, and intraspecific competition between glyphosate-resistant and glyphosate-susceptible horseweeds (Conyza canadensis) in the San Joaquin valley of California. Weed Sci. 2010, 58, 147–153. [Google Scholar] [CrossRef]
- Wu, H.; Walker, S.; Rollin, M.J.; Tan, D.K.Y.; Robinson, G.; Werth, J. Germination, persistence, and emergence of flaxleaf fleabane (Conyza bonariensis [L.] Cronquist). Weed Biol. Manag. 2007, 7, 192–199. [Google Scholar] [CrossRef]
- Karlsson, L.M.; Milberg, P. Comparing after-ripening response and germination requirements of Conyza canadensis and C. bonariensis (Asteraceae) through logistic functions. Weed Res. 2007, 47, 433–441. [Google Scholar] [CrossRef]
- Travlos, I.S.; Economou, G.; Kotoulas, V.E.; Kanatas, P.J.; Kontogeorgos, A.N.; Karamanos, A.I. Potential effects of diurnally alternating temperatures on purple nutsedge (Cyperus rotundus) tuber sprouting. J. Arid Environ. 2009, 73, 22–25. [Google Scholar] [CrossRef]
- Thompson, C.R.; Thill, D.C.; Shafii, B. Germination characteristics of sulfonylurea-resistant and -susceptible kochia (Kochia scoparia). Weed Sci. 1994, 42, 50–56. [Google Scholar] [CrossRef]
- Goulart, I.C.G.R.; Matzenbacher, F.O.; Merotto, A.J.R. Differential germination pattern of rice cultivars resistant to imidazolinone herbicides carrying different acetolactate synthase gene mutations. Weed Res. 2012, 52, 224–232. [Google Scholar] [CrossRef]
- François, J.T.; Irena, R.; Mihai, C. A mutation in the herbicide target site acetohydroxyacid synthase produces morphological and structural alterations and reduces fitness in Amaranthus powellii. New Phytol. 2006, 169, 251–264. [Google Scholar]
- Sibony, M.; Rubin, B. The ecological fitness of ALS-resistant Amaranthus retroflexus and multiple resistant Amaranthus blitoides. Weed Res. 2003, 43, 40–47. [Google Scholar] [CrossRef]
- Park, K.W.; Mallory-Smith, C.A.; Ball, D.A.; Mueller-Warrant, G.W. Ecological fitness of acetolactate synthase inhibitor-resistant and-susceptible downy brome (Bromus tectorum) biotypes. Weed Sci. 2004, 52, 768–773. [Google Scholar] [CrossRef]
- Duff, M.G.; Al-Khatib, K.; Peterson, D.E. Relative competitiveness of protoporphyrinogen oxidase-resistant common waterhemp (Amaranthus rudis). Weed Sci. 2009, 57, 169–174. [Google Scholar] [CrossRef]


| Temperature Regime (°C) | Germination Percentage (%) | Vigor Index | ||
|---|---|---|---|---|
| R | S | R | S | |
| 10/20 | 9.3 ± 0.9 | 9.8 ± 0.7 | 38 ± 3.9 | 42 ± 3.2 |
| 15/25 | 72 ± 5.3 | 77.3 ± 6.1 | 1369 ± 64.5 | 1282 ± 78.5 |
| 20/30 | 76.6 ± 6.1 | 79.3 ± 5.5 | 3541 ± 178.1 | 3857 ± 146.3 |
| 25/35 | 81.3 ± 7.7 | 86.6 ± 7.1 | 3387 ± 161.9 | 3345 ± 114.7 |
| 30/40 | 77.6 ± 3.2 | 81.2 ± 1.2 | 1796 ± 181.9 | 1821 ± 109.3 |
| Biotype | Seed Number/Plant | Seed Weight (g/plant) |
|---|---|---|
| R | 1389 ± 257 a | 0.58 ± 0.02 a |
| S | 1317 ± 206 a | 0.57 ± 0.03 a |
| DAT | Relative Crowding Coefficient (±SE) | ||
|---|---|---|---|
| Leaf Number | Plant Height | Dry Weight | |
| 14 | 0.91 ± 0.08 | 1.19 ± 0.1 | 1.03 ± 0.06 |
| 28 | 0.95 ± 0.06 | 0.91 ± 0.05 | 0.95 ± 0.11 |
| 42 | 0.98 ± 0.13 | 0.94 ± 0.18 | 0.96 ± 0.17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, R.; Han, Y.; Sun, Y.; Huang, H.; Wei, S.; Huang, Z. Growth and Competitiveness of ALS-Inhibiting Herbicide-Resistant Amaranthus retroflexus L. Plants 2022, 11, 2639. https://doi.org/10.3390/plants11192639
Wang R, Han Y, Sun Y, Huang H, Wei S, Huang Z. Growth and Competitiveness of ALS-Inhibiting Herbicide-Resistant Amaranthus retroflexus L. Plants. 2022; 11(19):2639. https://doi.org/10.3390/plants11192639
Chicago/Turabian StyleWang, Ruolin, Yujun Han, Ying Sun, Hongjuan Huang, Shouhui Wei, and Zhaofeng Huang. 2022. "Growth and Competitiveness of ALS-Inhibiting Herbicide-Resistant Amaranthus retroflexus L." Plants 11, no. 19: 2639. https://doi.org/10.3390/plants11192639
APA StyleWang, R., Han, Y., Sun, Y., Huang, H., Wei, S., & Huang, Z. (2022). Growth and Competitiveness of ALS-Inhibiting Herbicide-Resistant Amaranthus retroflexus L. Plants, 11(19), 2639. https://doi.org/10.3390/plants11192639






