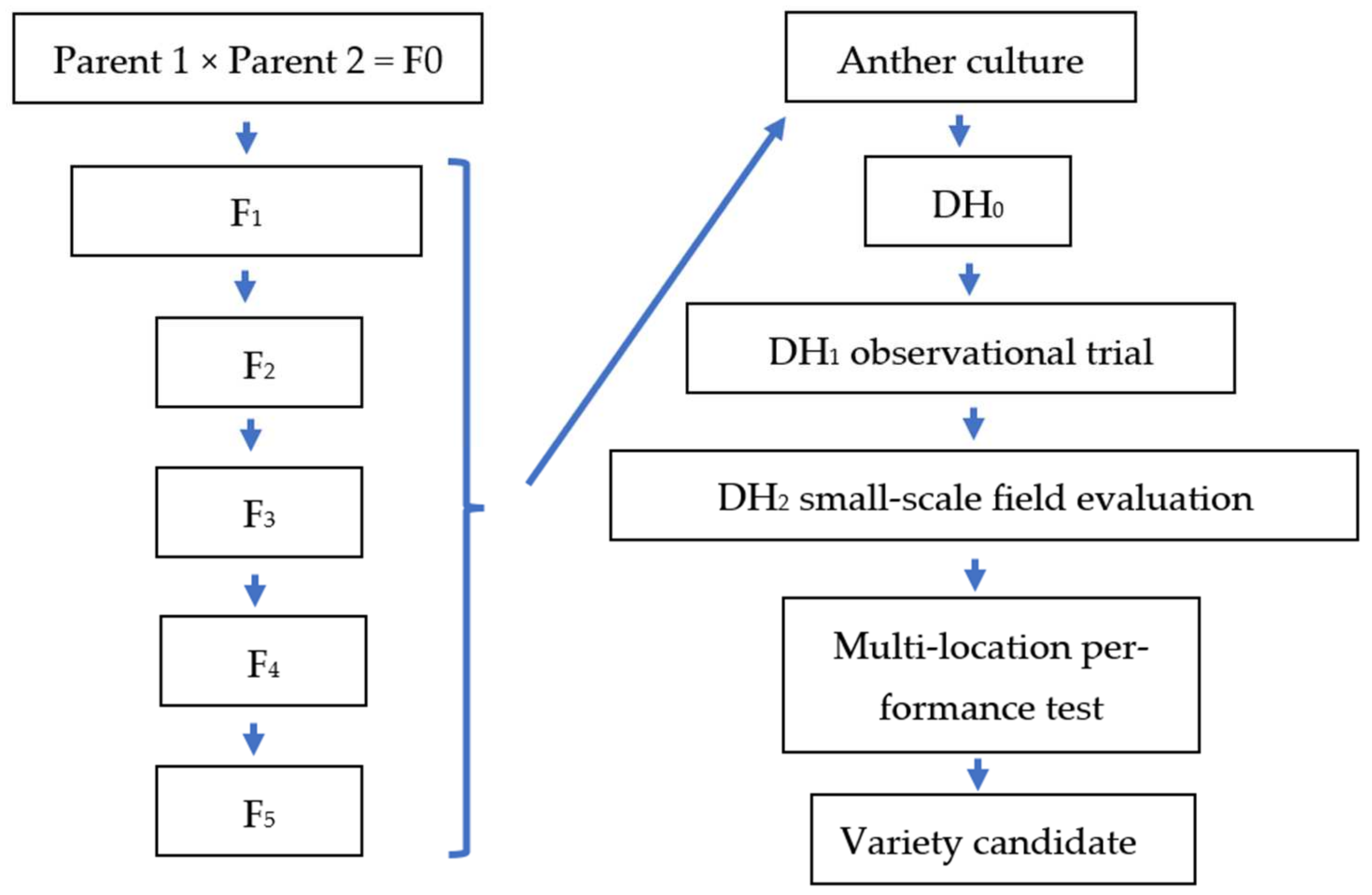
1. Introduction
Biotechnological methods support the quick and efficient production of new varieties and hybrids in crop plants. One of these techniques is anther culture (AC) which can reduce the breeding time of new varieties and hybrids due to the production of homozygous lines within one generation. In rice, AC was applied to develop new varieties in many breeding programs [
1,
2,
3,
4,
5]. Research groups have focused on the further improvement of AC in rice [
6,
7,
8,
9,
10,
11,
12,
13,
14,
15].
Since the development of AC in rice [
16], many efforts have been made in several countries to improve the efficiency of AC [
17,
18,
19,
20]. However, several factors limit the high frequency of doubled haploid (DH) plant production in rice, such as the genotype and the growing condition of donor plants, the developmental stage of microspores, pre-treatments, and the induction and regeneration media used.
Many studies have demonstrated the effect of genotype on the efficiency of AC in rice [
2,
21,
22,
23,
24,
25]. The japonica genotypes are known as high-responding rice genotypes, while indica genotypes are known to be less responsive to AC [
6,
13,
21,
22,
24,
26,
27,
28]. A low frequency of plant regeneration and albinism are also known as limiting factors for AC in rice [
6,
8,
14,
15,
19,
21,
22,
25].
Several studies have focused on the improvement of culture conditions in rice AC. The same medium was applied for induction of androgenesis and plant regeneration in a one-step method [
3,
29], while other protocols used different media for induction and plant regeneration in rice AC. The N
6 medium [
30] has been the most frequently used medium for the induction of androgenesis over the past two decades [
5,
6,
7,
8,
10,
11,
12,
13,
14,
21,
22,
23,
24,
25,
27,
31,
32]. Maltose is considered the best carbon source for the induction of androgenesis and has a positive effect, increasing callus induction and green plantlet regeneration in AC [
12,
20,
21]. Different growth regulators, such as 1-naphtylacetic acid (NAA), phenylacetic acid, Picloram, Dicamba, 2,4-dichlorophenoxyacetic acid (2,4-D) and their combinations, were applied for the induction of androgenesis in rice AC [
23]. The combination of 2,4-D and kinetin has been frequently applied in the induction media of rice AC [
5,
7,
14,
22,
24], while other investigations have used a combination of indol-3-acetic acid (IAA) and kinetin [
10]. In indica rice, a combination of 2,4-D and 6-benzylaminopurin (BAP) was used to increase the efficiency of androgenesis induction in AC [
11,
12], while Mayakaduwa and Silva [
13] applied a combination of 2,4-D, NAA and kinetin. A combination of NAA, BAP and kinetin was also applied for induction of androgenesis in japonica and japonica × indica crossing combinations [
28,
33].
A large number of calli can be induced in AC for some rice genotypes, improving plant regeneration efficiency for DH plant production. Two basal media, namely MS [
11,
12,
13] and N
6 [
5,
7,
10,
14], were applied mainly to achieve plant regeneration of AC-derived calli. According to published protocols, different combinations of growth regulators (BAP, IAA, NAA and kinetin) were successfully applied to achieve increased plantlet regeneration [
5,
7,
8,
10,
11,
12,
13,
14,
21,
22,
23,
24,
27,
31]. Some published data are available on the effects of exogenous growth regulators during plant regeneration of rice AC-derived calli [
11,
12]. The authors of these studies reported that the combination of 1.5 mg/L BAP, 0.5 mg/L NAA and 0.5 mg/L kinetin positively affected green plantlet production of AC-derived calli [
11,
12].
The aims of the present study were the improvement of in vitro AC in rice and the integration of the method into rice breeding. In the experiments undertaken, androgenesis was induced in AC of five rice genotypes to investigate the effect of genotypes on in vitro androgenesis. Different combinations of growth regulators (2 mg/L 2,4-D and 0.5 mg/L BAP–N6DB; 2.5 mg/L NAA, 1 mg/L 2,4-D and 0.5 mg/L kinetin–N6NDK) and of induction medium were tested on the AC parameters (i.e., number of AC-derived calli, regenerated green and albino plantlets). In the plant regeneration, two basal media (MS and N6) and two combinations of growth regulators (1 mg/L NAA, 1 mg/L BAP and 1 mg/L kinetin–NBK1; 1.5 mg/L BAP, 0.5 mg/L kinetin and 0.5 mg/L NAA–NBK2) were compared to identify the most efficient plant regeneration medium. The percentage of spontaneous chromosome doubling was determined based on seed production. In addition, DH lines in the DH1 and DH2 generations were selected in micro- and small-plot field experiments examining plant height, biomass, yield, lodging, blast resistance, TKW, and quality parameters, to identify promising lines for the Hungarian temperate rice breeding program.
4. Materials and Methods
4.1. Plant Materials and Growing Conditions
In the experiments, one Hungarian variety (Dáma), one advanced line (Karola), one F2 (1009) and 2 F1 (Mirko × Karola, Ábel × Dular) combinations of japonica rice, were tested in AC. The donor plants were grown at the Rice Research Station of the Hungarian University of Agriculture and Life Sciences (MATE), Institute of Environmental Sciences (IES), Research Center for Irrigation and Water Management (ÖVKI), located at Szarvas, Hungary. Standard Hungarian rice cultivation technology was used to grow healthy plants in the nursery. Direct seeding was applied on the 28th of April (0 days after sowing; 0 DAS) followed by pre-emergent herbicide spraying (active ingredient: pendimethalin). Permanent flooding was performed from 38 DAS; the irrigation level was set to the development of the plants (5–20 cm). Split nitrogen fertilizer (60–60 kg N/ha) applications (38 and 70 DAS) were used to optimize nutrient management at a total amount of 120 kg N/ha. During the plant growing season, manual weeding was carried out multiple times. The end of the booting stage in the reproductive phase was observed from 75 DAS to 108 DAS, determined by the duration of different varieties and advanced lines.
4.2. Collection and Pre-Treatment of Donor Tillers
The donor tillers were collected at the booting stage when the flowers in panicles contained early and mid uninucleated microspores (
Figure 1a). The developmental stages of microspores were confirmed with an Olympus CK-2 inverted microscope (Olympus Ltd., Southend-on-Sea, UK). The donor tillers were put into an Erlenmeyer flask containing tap water and were covered with PVC bags to protect the tillers from intensive evaporation under the pre-treatment. The donor tillers were cold pre-treated at 10 °C for three to seven days [
25,
42,
43].
4.3. Sterilization and Isolation of Anthers
After pre-treatment, the developmental stages of microspores were rechecked with an Olympus CK-2 inverted microscope (Olympus Ltd., Southend-on-Sea, UK). Panicles suitable for AC were surface sterilized for 20 min in 2% commercial sodium hypochlorite solution with two drops of Tween-80 solution (Duchea Biochemie B.V., Haarlem, The Netherlands). After sterilization, the panicles were rinsed three times with sterile distilled water (Millipore Elix 5). The anthers (100 anthers/Petri dish) were isolated aseptically from the donor panicles and placed in 60 mm diameter plastic Petri dishes (Sarstedt Ltd., Newton, MA, USA) containing different induction media.
4.4. In Vitro AC
The N
6 basal medium [
30] was supplemented with growth regulators, 40 g maltose, 500 mg/L L-proline and 500 mg/L L-glutamine, and pH was adjusted to 5.8. The effect of growth regulators (2 mg/L 2,4-D and 0.5 mg/L BAP; 2.5 mg/L NAA, 1 mg/L 2,4-D and 0.5 mg/L kinetin) was tested based on the induction (number of calli) and plant regeneration (green and albino plantlets) by androgenesis (
Table 9). The Petri dishes were kept at 28 °C using a thermostat.
4.5. Flow Cytometric Analyses of AC-Derived Calli
The ploidy level of 10 AC-derived calli was determined by flow cytometric analysis (CytoFLEX Flow Cytometer, Beckman Coulter International S.A., Nyon, Switzerland). Samples of friable calli with 3 mm diameter were collected from the plant regeneration medium and callus produced in somatic tissue culture (matured embryo culture) was applied as a diploid control. The samples were chopped with a razor blade with 1 mL Galbraith buffer for 1 min to isolate the nuclei from the samples [
44]. The suspensions were purified using 20 µm sieves. DNA content was painted with 40 µL propidium iodide (PI) solution (1 mg/L) for 30 min at 4 °C. After preparation, the DNA content of samples was measured by flow cytometer; the ploidy levels of samples were determined based on histograms.
4.6. Plant Regeneration
The AC-derived calli were transferred weekly to 90 mm diameter plastic Petri dishes (Sarstedt Ltd., Newton, MA, USA) containing different plant regeneration media. The collection of AC-derived calli was finished on the 10th week of AC. During the regeneration period, the effects of two basal media (MS and N
6) and combinations of different growth regulators (1 mg/L NAA, 1 mg/L BAP and 1 mg/L kinetin; 1.5 mg/L BAP, 0.5 mg/L kinetin and 0.5 mg/L NAA) were tested based on the regeneration of green and albino plantlets [
30,
45]. The MS [
45] and N
6 [
30] media were supplemented with the above-mentioned growth regulators and 30 g/L sucrose and pH was adjusted to 5.8 (
Table 9).
Well-shooted plantlets were transferred into glass tubes containing half MS medium supplemented with 30 g/L sucrose and pH was adjusted to 5.8 [
45]. The green plantlets were cultured in glass tubes until rooting. Well-rooted and shooted green plantlets were transplanted into 720 mL glass jars containing the same half MS medium until transplantation of plantlets to the greenhouse. During the plant regeneration and rooting period, 16 h artificial light (led) and a temperature of 24 °C were maintained in the tissue culture chamber.
4.7. Acclimatization of Plantlets
In the first half of March, the well-rooted green plantlets were transferred to the greenhouse and covered with PVC bags to retain adequate humidity. After a five-day acclimatization period, the PVC bags were removed. The green plants were transferred to a paddy field for additional testing. The individual plants were transplanted after permanent flooding was applied. The hill spacing was set to 0.5 m in the nursery. Developmental and morphological characters were observed to identify differences between single plants. The ploidy levels of the regenerated plants were checked by seed set production. The seeds of DH plants were collected after the ripening stage and were integrated into the breeding program.
4.8. Field Experiments
The field experiments were carried out at the Rice Research Station of the MATE ÖVKI (Szarvas, Hungary). Genotypes were direct dry-seeded and standard paddy conditions were maintained during the experiments (temporary and permanent flooding until 38–40 DAS and 105–109 DAS, respectively; split nitrogen fertilizer (120 kg/ha); broad-spectrum herbicide (Penoxsulam) at 38–40 DAS; no fungicide). In the DH1 generation, the DH lines were tested in a micro-plot experiment (1.5 m rows) with three replications. A total of 58 DH lines were involved in the basic agronomic (observational) trial. Three Hungarian rice varieties (‘Janka’, ‘Dáma’ and ‘M 488’) were used as controls. The DH lines of the 1009 genotype were generated in the tissue culture experiments described here, while the DH lines of Köröstáj and Tünde were produced in an earlier DH plant production program.
The best DH lines were advanced to replicated yield trials (four replications) of DH lines for two years in 2019 and 2020. The yield trials were planted in a randomized complete block design with a 3.6 m
2 plot size. The evaluation of the lines was performed according to standard breeding criteria (i.e., early vegetative vigor, cold tolerance, blast resistance, earliness, plant height, panicle form, yield and biomass) and completed following the procedures of IRRI [
46] and Roumen et al. [
47]. Plots were manually harvested; the threshing was performed using a Wintersteiger LD350 laboratory threshing machine (Wintersteiger Co., Ried im Innkreis, Austria). Sartorius BP221S and Sartorius PMA7500 laboratory scales (Sartorius Co., Göttingen, Germany) were used to weigh the samples. Seeds were counted with a Pfeuffer Contador automatic seed counter (Pfeuffer Ltd., Kitzingen, Germany). A Satake THU laboratory husker and a Satake TM05 testing mill (Satake Co., Hiroshima, Japan) were used for the basic milling quality analysis.
4.9. Statistical Analyses
The laboratory experiments were carried out in five replications. Data for AC response (i.e., number of calli, albino and green plantlets) were collected and analyzed by two-way ANOVA. The statistical analyses were carried out using Microsoft Excel 2019 statistical software developed by Microsoft (Redmond, WA, USA). Data from the field experiments were statistically analyzed using IBM SPSS ver. 22 software. Three and four plots were used for the DH1 and DH2 experiments, respectively. Plant phenology and agronomic traits were collected using four replicates. One-way ANOVA with Tukey post hoc test was used to test differences among treatments at the 5% level of probability.