Metal Detoxification in Land Plants: From Bryophytes to Vascular Plants. STATE of the Art and Opportunities
Abstract
1. Introduction
2. Spore-Bearing Plants
2.1. Bryophytes
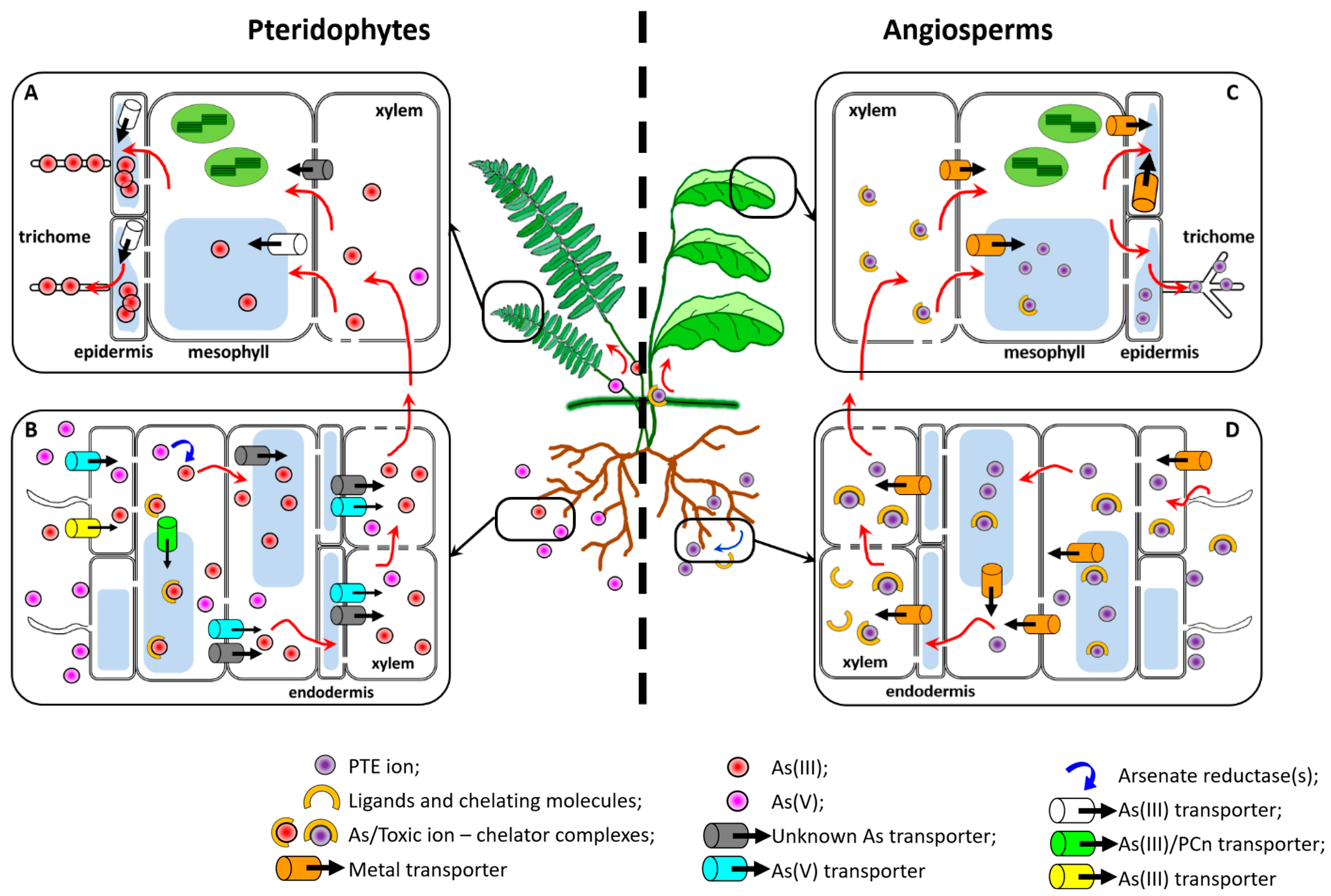
2.2. Pteridophytes
3. Seed-Bearing Plants: Gymnosperms and Angiosperms
3.1. Gymnosperms
3.2. Angiosperms and the Evolution of the Hyperaccumulation and Hypertolerance Traits
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Holleman, A.F.; Wiberg, E. Lehrbuch der Anorganischen Chemie; Walter de Gruyter: Berlin, Germany, 1985; p. 868. [Google Scholar]
- Duffus, J.H. “Heavy Metals”-a meaningless term? Pure Appl. Chem. 2002, 74, 793–807. [Google Scholar] [CrossRef]
- Pourret, O.; Hursthouse, A. It’s Time to Replace the Term “Heavy Metals” with “Potentially Toxic Elements” When Reporting Environmental Research. Int. J. Environ. Res. Public Health 2019, 16, 4446. [Google Scholar] [CrossRef] [PubMed]
- Rai, P.K. Heavy metal phytoremediation from aquatic ecosystems with special reference to macrophytes. Crit. Rev. Environ. Sci. 2009, 39, 697–753. [Google Scholar] [CrossRef]
- Fytianos, K.; Katsianis, G.; Triantafyllou, P.; Zachariadis, G. Accumulation of heavy metals in vegetables grown in an industrial area in relation to soil. Bull. Environ. Contam. Toxicol. 2001, 67, 423–430. [Google Scholar] [CrossRef]
- Wieczorek-Dąbrowska, M.; Tomza-Marciniak, A.; Pilarczyk, B.; Balicka-Ramisz, A. Roe and red deer as bioindicators of heavy metals contamination in north-western Poland. Chem. Ecol. 2013, 29, 100–110. [Google Scholar] [CrossRef]
- Jarup, L. Hazards of heavy metal contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef] [PubMed]
- Wirth, J.J.; Mijal, R.S. Adverse effects of low level heavy metal exposure on male reproductive function. Syst. Biol. Reprod. Med. 2010, 56, 147–167. [Google Scholar] [CrossRef]
- Hall, J.L. Cellular mechanisms for heavy metals detoxification and tolerance. J. Exp. Bot. 2002, 53, 1–11. [Google Scholar] [CrossRef]
- Kakkar, P.; Jaffery, F.N. Biological markers for metal toxicity. Environ. Toxicol. Pharmacol. 2005, 19, 335–349. [Google Scholar] [CrossRef] [PubMed]
- Jan, A.T.; Azam, M.; Siddiqui, K.; Ali, A.; Choi, I.; Haq, Q.M.R. Heavy Metals and Human Health: Mechanistic Insight into Toxicity and Counter Defense System of Antioxidants. Int. J. Mol. Sci. 2015, 16, 29592–29630. [Google Scholar] [CrossRef]
- Sytar, O.; Kumar, A.; Latowski, D.; Kuczynska, P.; Strzałka, K.; Prasad, M.N.V. Heavy metal-induced oxidative damage, defense reactions, and detoxification mechanisms in plants. Acta Physiol. Plant 2013, 35, 985–999. [Google Scholar] [CrossRef]
- Tennstedt, P.; Peisker, D.; Böttcher, C.; Trampczynska, A.; Clemens, S. Phytochelatin Synthesis Is Essential for the Detoxification of Excess Zinc and Contributes Significantly to the Accumulation of Zinc. Plant Physiol. 2009, 149, 938–948. [Google Scholar] [CrossRef] [PubMed]
- Salemaa, M.; Derome, J.; Helmisaari, H.S.; Nieminen, T.; Vanha-Majamaa, I. Element accumulation in boreal bryophytes, lichens and vascular plants exposed to heavy metal and sulfur deposition in Finland. Sci. Total Environ. 2004, 324, 141–160. [Google Scholar] [CrossRef] [PubMed]
- Frahm, J.P. Diversity, dispersal and biogeography of bryophytes (mosses). Biodivers. Conserv. 2008, 17, 277–284. [Google Scholar] [CrossRef]
- Puttick, M.N.; Morris, J.L.; Williams, T.A.; Cox, C.J.; Edwards, D.; Kenrick, P.; Pressel, S.; Wellman, C.H.; Schneider, H.; Pisani, D.; et al. The Interrelationships of Land Plants and the Nature of the Ancestral Embryophyte. Curr. Biol. 2018, 28, 733–745.e2. [Google Scholar] [CrossRef]
- Harris, B.J.; Harrison, C.J.; Hetherington, A.M.; Williams, T.A. Phylogenomic Evidence for the Monophyly of Bryophytes and the Reductive Evolution of Stomata. Curr. Biol. 2020, 30, 2001–2012.e2. [Google Scholar] [CrossRef] [PubMed]
- Hutsemekers, V.; Dopagne, C.; Vanderpoorten, A. How far and how fast do bryophytes travel at the landscape scale? Divers. Distrib. 2008, 14, 483–492. [Google Scholar] [CrossRef]
- Shimamura, M. Marchantia polymorpha: Taxonomy, phylogeny and morphology of a model system. Plant Cell Physiol. 2016, 57, 230–256. [Google Scholar] [CrossRef]
- Coelho, M.C.M.; Gabriel, R.; Hespanhol, H.; Borges, P.A.V.; Ah-Peng, C. Bryophyte diversity along an elevational gradient on pico island (Azores, portugal). Diversity 2021, 13, 162. [Google Scholar] [CrossRef]
- Zechmeister, H.G.; Dirnböck, T.; Hülber, K.; Mirtl, M. Assessing airborne pollution effects on bryophytes-lessons learned through long-term integrated monitoring in Austria. Environ. Pollut. 2007, 147, 696–705. [Google Scholar] [CrossRef]
- Zvereva, E.L.; Kozlov, M.V. Impacts of industrial polluters on bryophytes: A meta-analysis of observational studies. Water Air Soil Pollut. 2011, 218, 573–586. [Google Scholar] [CrossRef]
- Chen, Y.E.; Cui, J.M.; Yang, J.C.; Zhang, Z.W.; Yuan, M.; Song, C.; Yang, H.; Liu, H.M.; Wang, C.Q.; Zhang, H.Y.; et al. Biomonitoring heavy metal contaminations by moss visible parameters. J. Hazard. Mater. 2015, 296, 201–209. [Google Scholar] [CrossRef]
- Di Palma, A.; González, A.G.; Adamo, P.; Giordano, S.; Reski, R.; Pokrovsky, O.S. Biosurface properties and lead adsorption in a clone of Sphagnum palustre (Mosses): Towards a unified protocol of biomonitoring of airborne heavy metal pollution. Chemosphere 2019, 236, 124375. [Google Scholar] [CrossRef]
- Samecka-Cymerman, A.; Marczonek, A.; Kempers, A.J. Bioindication of heavy metals in soil by liverworts. Arch. Environ. Contam. Toxicol. 1997, 33, 162–171. [Google Scholar] [CrossRef]
- Vásquez, C.; Calva, J.; Morocho, R.; Donoso, D.A.; Benítez, Á. Bryophyte communities along a tropical urban river respond to heavy metal and arsenic pollution. Water 2019, 11, 813. [Google Scholar] [CrossRef]
- Sassmann, S.; Wernitznig, S.; Lichtscheidl, I.K.; Lang, I. Comparing copper resistance in two bryophytes: Mielichhoferia elongata Hornsch. versus Physcomitrella patens Hedw. Protoplasma 2010, 246, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Papadia, P.; Barozzi, F.; Migoni, D.; Rojas, M.; Fanizzi, F.P.; Di Sansebastiano, G.P. Aquatic mosses as adaptable bio-filters for heavy metal removal from contaminated water. Int. J. Mol. Sci. 2020, 21, 4769. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.; Antonovics, J.; Anderson, L.E. Inter- and intraspecific variation of mosses in tolerance to copper and zinc. Evolution 1987, 41, 1312–1325. [Google Scholar]
- Itouga, M.; Hayatsu, M.; Sato, M.; Tsuboi, Y.; Kato, Y.; Toyooka, K.; Suzuki, S.; Nakatsuka, S.; Kawakami, S.; Kikuchi, J.; et al. Protonema of the moss Funaria hygrometrica can function as a lead (Pb) adsorbent. PLoS ONE 2017, 12, e0189726. [Google Scholar] [CrossRef]
- Sandhi, A.; Landberg, T.; Greger, M. Phytofiltration of arsenic by aquatic moss (Warnstorfia fluitans). Environ. Pollut. 2018, 237, 1098–1105. [Google Scholar] [CrossRef]
- Sharma, S. Marchantia polymorpha L.: A Bioaccumulator. Aerobiologia 2007, 23, 181–187. [Google Scholar] [CrossRef]
- Ares, Á.; Itouga, M.; Kato, Y.; Sakakibara, H. Differential Metal Tolerance and Accumulation Patterns of Cd, Cu, Pb and Zn in the Liverwort Marchantia polymorpha L. Bull. Environ. Contam. Toxicol. 2018, 100, 444–450. [Google Scholar] [CrossRef]
- Pratas, J.; Favas, P.J.C.; Varun, M.; D’Souza, R.; Paul, M.S. Distribution of rare earth elements, thorium and uranium in streams and aquatic mosses of Central Portugal. Environ. Earth Sci. 2017, 76, 156. [Google Scholar] [CrossRef]
- Krzesłowska, M.; Rabȩda, I.; Lewandowski, M.; Samardakiewicz, S.; Basińska, A.; Napieralska, A.; Mellerowicz, E.J.; Wozny, A. Pb induces plant cell wall modifications-In particular-The increase of pectins able to bind metal ions level. In Proceedings of the E3S Web of Conferences, 16th International Conference on Heavy Metals in the Environment, Rome, Italy, 23–27 September 2012; EDP Sciences: Les Ulis, France, 2013; Volume 1. [Google Scholar]
- Basile, A.; Sorbo, S.; Pisani, T.; Paoli, L.; Munzi, S.; Loppi, S. Bioacumulation and ultrastructural effects of Cd, Cu, Pb and Zn in the moss Scorpiurum circinatum (Brid.) Fleisch. & Loeske. Environ. Pollut. 2012, 166, 208–211. [Google Scholar] [PubMed]
- Lang, I.; Wernitznig, S. Sequestration at the cell wall and plasma membrane facilitates zinc tolerance in the moss Pohlia drummondii. Environ. Exp. Bot. 2011, 74, 186–193. [Google Scholar] [CrossRef]
- Shakya, K.; Chettri, M.K.; Sawidis, T. Impact of heavy metals (copper, zinc, and lead) on the chlorophyll content of some mosses. Arch. Environ. Contam. Toxicol. 2008, 54, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Degola, F.; De Benedictis, M.; Petraglia, A.; Massimi, A.; Fattorini, L.; Sorbo, S.; Basile, A.; Di Toppi, L.S. A Cd/Fe/Zn-responsive phytochelatin synthase is constitutively present in the ancient liverwort Lunularia cruciata (L.) dumort. Plant Cell Physiol. 2014, 55, 1884–1891. [Google Scholar] [CrossRef]
- Lane, T.S.; Rempe, C.S.; Davitt, J.; Staton, M.E.; Peng, Y.; Soltis, D.E.; Melkonian, M.; Deyholos, M.; Leebens-Mack, J.H.; Chase, M.; et al. Diversity of ABC transporter genes across the plant kingdom and their potential utility in biotechnology. BMC Biotechnol. 2016, 16, 47. [Google Scholar] [CrossRef]
- Petraglia, A.; De Benedictis, M.; Degola, F.; Pastore, G.; Calcagno, M.; Ruotolo, R.; Mengoni, A.; Di Toppi, L.S. The capability to synthesize phytochelatins and the presence of constitutive and functional phytochelatin synthases are ancestral (plesiomorphic) characters for basal land plants. J. Exp. Bot. 2014, 65, 1153–1163. [Google Scholar] [CrossRef]
- Bellini, E.; Maresca, V.; Betti, C.; Castiglione, M.R.; Fontanini, D.; Capocchi, A.; Sorce, C.; Borsò, M.; Bruno, L.; Sorbo, S.; et al. The moss Leptodictyum riparium counteracts severe cadmium stress by activation of glutathione transferase and phytochelatin synthase, but slightly by phytochelatins. Int. J. Mol. Sci. 2020, 21, 1583. [Google Scholar] [CrossRef]
- Li, M.; Barbaro, E.; Bellini, E.; Saba, A.; di Toppi, L.S.; Varotto, C. Ancestral function of the phytochelatin synthase C-terminal domain in inhibition of heavy metal-mediated enzyme overactivation. J. Exp. Bot. 2020, 71, 6655–6669. [Google Scholar] [CrossRef] [PubMed]
- Inupakutika, M.A.; Sengupta, S.; Devireddy, A.R.; Azad, R.K.; Mittler, R. The evolution of reactive oxygen species metabolism. J. Exp. Bot. 2016, 67, 5933–5943. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.Q.; He, M.; Cao, T.; Zhang, Y.C.; Han, W. Response mechanisms of antioxidants in bryophyte (Hypnum plumaeforme) under the stress of single or combined Pb and/or Ni. Environ. Monit. Assess. 2009, 149, 291–302. [Google Scholar] [CrossRef]
- Dazy, M.; Masfaraud, J.F.; Férard, J.F. Induction of oxidative stress biomarkers associated with heavy metal stress in Fontinalis antipyretica Hedw. Chemosphere 2009, 75, 297–302. [Google Scholar] [CrossRef]
- Smith, A.R.; Pryer, K.M.; Schuettpelz, E.; Korall, P.; Schneider, H.; Wolf, P.G. A classification for extant ferns. Names 2006, 55, 705–731. [Google Scholar] [CrossRef]
- Prance, G.T. Discovering the plant world. Taxon 2001, 50, 345–359. [Google Scholar] [CrossRef]
- Niklas, K.J.; Tiffney, B.H.; Knoll, A.H. Patterns in vascular land plant diversification. Nature 1983, 303, 614–616. [Google Scholar] [CrossRef]
- Schneider, H.; Schuettpelz, E.; Pryer, K.M.; Cranfill, R.; Magallón, S.; Lupia, R. Ferns diversified in the shadow of angiosperms. Nature 2004, 428, 553–557. [Google Scholar] [CrossRef]
- Kreft, H.; Jetz, W.; Mutke, J.; Barthlott, W. Contrasting environmental and regional effects on global pteridophyte and seed plant diversity. Ecography 2010, 33, 408–419. [Google Scholar] [CrossRef]
- Drăghiceanu, O.A.; Dobrescu, C.M.; Soare, L.C. Applications of pteridophytes in phytoremediation. Curr. Trends Nat. Sci. 2014, 3, 68–73. [Google Scholar]
- Praveen, A.; Pandey, V.C. Pteridophytes in phytoremediation. Environ. Geochem. Health 2020, 42, 2399–2411. [Google Scholar] [CrossRef]
- Prabhu, S.G.; Srinikethan, G.; Hegde, S. Potential of Pteridophytes in Heavy Metal Phytoremediation. Int. J. Res. Eng. Technol. 2016, 5, 1–9. [Google Scholar]
- Tiwari, S.; Sarangi, B.K.; Anusha, P.; Pandey, R.A. Metal Hyperaccumulating Ferns: Progress and Future Prospects. In Recent Advances towards Improved Phytoremediation of Heavy Metal Pollution; Leung, D.W.M., Ed.; Bentham Science Publishers: Sharjah, United Arab Emirates, 2013; ISBN 978-1-60805-787-0. [Google Scholar]
- Sukumaran, D. Phytoremediation of heavy metals from industrial effluent using constructed wetland technology. Appl. Ecol. Environ. Sci. 2013, 1, 92–97. [Google Scholar] [CrossRef]
- Dhir, B.; Sharmila, P.; Saradhi, P.P.; Sharma, S.; Kumar, R.; Mehta, D. Heavy metal induced physiological alterations in Salvinia natans. Ecotoxicol. Environ. Saf. 2011, 74, 1678–1684. [Google Scholar] [CrossRef] [PubMed]
- Bennicelli, R.; St\kepniewska, Z.; Banach, A.; Szajnocha, K.; Ostrowski, J. The ability of Azolla caroliniana to remove heavy metals (Hg (II), Cr (III), Cr (VI)) from municipal waste water. Chemosphere 2004, 55, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Shoel, N.; Barkay, Z.; Ilzycer, D.; Gilath, I.; Tel-Or, E. Biofiltration of toxic elements by Azolla biomass. Water Air Soil Pollut. 2002, 135, 93–104. [Google Scholar] [CrossRef]
- Stepniewska, Z.; Bennicelli, R.P.; Balakhina, T.I.; Szajnocha, K.; Banach, A.M.; Wolinska, A. Potential of Azolla caroliniana for the removal of Pb and Cd from wastewaters. Int. Agrophys. 2005, 19, 251–255. [Google Scholar]
- Rakhshaee, R.; Khosravi, M.; Ganji, M.T. Kinetic modeling and thermodynamic study to remove Pb (II), Cd (II), Ni (II) and Zn (II) from aqueous solution using dead and living Azolla filiculoides. J. Hazard. Mater. 2006, 134, 120–129. [Google Scholar] [CrossRef]
- Rai, P.K. Phytoremediation of Hg and Cd from industrial effluents using an aquatic free floating macrophyte Azolla pinnata. Int. J. Phytoremediat. 2008, 10, 430–439. [Google Scholar] [CrossRef]
- Mashkani, S.G.; Ghazvini, P.T.M. Biotechnological potential of Azolla filiculoides for biosorption of Cs and Sr: Application of micro-PIXE for measurement of biosorption. Bioresour. Technol. 2009, 100, 1915–1921. [Google Scholar] [CrossRef]
- Dhir, B. Role of Ferns in Environmental Cleanup. In Current Advances in Fern Research; Fernández, H., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 517–531. ISBN 978-3-319-75103-0. [Google Scholar]
- Nichols, P.B.; Couch, J.D.; Al-Hamdani, S.H. Selected physiological responses of Salvinia minima to different chromium concentrations. Aquat. Bot. 2000, 68, 313–319. [Google Scholar] [CrossRef]
- Hoffmann, T.; Kutter, C.; Santamaria, J. Capacity of Salvinia minima Baker to tolerate and accumulate As and Pb. Eng. Life Sci. 2004, 4, 61–65. [Google Scholar] [CrossRef]
- Olguin, E.J.; Hernández, E.; Ramos, I. The effect of both different light conditions and the pH value on the capacity of Salvinia minima Baker for removing cadmium, lead and chromium. Acta Biotechnol. 2002, 22, 121–131. [Google Scholar] [CrossRef]
- Olguin, E.J.; Sánchez-Galván, G.; Pérez-Pérez, T.; Pérez-Orozco, A. Surface adsorption, intracellular accumulation and compartmentalization of Pb (II) in batch-operated lagoons with Salvinia minima as affected by environmental conditions, EDTA and nutrients. J. Ind. Microbiol. Biotechnol. 2005, 32, 577–586. [Google Scholar] [CrossRef]
- Espinoza-Quinones, F.R.; Zacarkim, C.E.; Palacio, S.M.; Obregon, C.L.; Zenatti, D.C.; Galante, R.M.; Rossi, N.; Rossi, F.L.; Pereira, I.R.A.; Welter, R.A.; et al. Removal of heavy metal from polluted river water using aquatic macrophytes Salvinia sp. Braz. J. Phys. 2005, 35, 744–746. [Google Scholar] [CrossRef]
- Molisani, M.M.; Rocha, R.; Machado, W.; Barreto, R.C.; Lacerda, L.D. Mercury contents in aquatic macrophytes from two reservoirs in the Paraba do Sul: Guandú river system, SE Brazil. Braz. J. Biol. 2006, 66, 101–107. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Suñe, N.; Sánchez, G.; Caffaratti, S.; Maine, M.A. Cadmium and chromium removal kinetics from solution by two aquatic macrophytes. Environ. Pollut. 2007, 145, 467–473. [Google Scholar] [CrossRef]
- Sánchez-Galván, G.; Monroy, O.; Gómez, J.; Olguin, E.J. Assessment of the hyperaccumulating lead capacity of Salvinia minima using bioadsorption and intracellular accumulation factors. Water Air Soil Pollut. 2008, 194, 77–90. [Google Scholar] [CrossRef]
- Fuentes, I.I.; Espadas-Gil, F.; Talavera-May, C.; Fuentes, G.; Santamaria, J.M. Capacity of the aquatic fern (Salvinia minima Baker) to accumulate high concentrations of nickel in its tissues, and its effect on plant physiological processes. Aquat. Toxicol. 2014, 155, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Kumari, S.; Kumar, B.; Sheel, R. Bioremediation of Heavy Metals by Serious Aquatic Weed, Salvinia. Int. J. Curr. Microbiol. Appl. Sci. 2016, 5, 355–368. [Google Scholar] [CrossRef]
- Emiliani, J.; Oyarce, W.G.L.; Salvatierra, L.M.; Novo, L.A.B.; Pérez, L.M. Evaluation of Cadmium Bioaccumulation-Related Physiological Effects in Salvinia biloba: An Insight towards Its Use as Pollutant Bioindicator in Water Reservoirs. Plants 2021, 10, 2679. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, G.; Sarker, S. The role of Salvinia rotundifolia in scavenging aquatic Pb (II) pollution: A case study. Bioprocess Eng. 1997, 17, 295–300. [Google Scholar] [CrossRef]
- Dhir, B.; Sharmila, P.; Saradhi, P.P. Potential of aquatic macrophytes for removing contaminants from the environment. Crit. Rev. Environ. Sci. Technol. 2009, 39, 754–781. [Google Scholar] [CrossRef]
- Chang, J.S.; Yoon, I.H.; Kim, K.W. Heavy metal and arsenic accumulating fern species as potential ecological indicators in As-contaminated abandoned mines. Ecol. Indic. 2009, 9, 1275–1279. [Google Scholar] [CrossRef]
- Claveria, R.J.R.; Perez, T.R.; Perez, R.E.C.; Algo, J.L.C.; Robles, P.Q. The identification of indigenous Cu and As metallophytes in the Lepanto Cu-Au Mine, Luzon, Philippines. Environ. Monit. Assess. 2019, 191, 185. [Google Scholar] [CrossRef] [PubMed]
- Ha, N.T.H.; Nga, T.T.H.; Minh, N.N.; Anh, B.T.K.; Hang, N.T.A.; Duc, N.A.; Nhuan, M.T.; Kim, K.W. Uptake of arsenic and heavy metals by native plants growing near Nui Phao multi-metal mine, northern Vietnam. Appl. Geochem. 2019, 108, 104368. [Google Scholar] [CrossRef]
- Prasetia, H.; Sakakibara, M.; Takehara, A.; Sueoka, Y. Heavy metals accumulation by Athyrium yokoscence in a mine area, Southwestern Japan. IOP Conf. Ser. Earth Environ. Sci. 2017, 71, 12025. [Google Scholar] [CrossRef]
- Christenhusz, M.J.M.; Chase, M.W. Trends and concepts in fern classification. Ann. Bot. 2014, 113, 571–594. [Google Scholar] [CrossRef]
- Morishita, T.; Boratynski, J.K. Accumulation of cadmium and other metals in organs of plants growing around metal smelters in Japan. Soil Sci. Plant Nutr. 1992, 38, 781–785. [Google Scholar] [CrossRef]
- Kamachi, H.; Komori, I.; Tamura, H.; Sawa, Y.; Karahara, I.; Honma, Y.; Wada, N.; Kawabata, T.; Matsuda, K.; Ikeno, S.; et al. Lead tolerance and accumulation in the gametophytes of the fern Athyrium yokoscense. J. Plant Res. 2005, 118, 137–145. [Google Scholar] [CrossRef]
- Nishizono, H.; Suzuki, S.; Ishii, F. Accumulation of heavy metals in the metal-tolerant fern, Athyrium yokoscense, growing on various environments. Plant Soil 1987, 102, 65–70. [Google Scholar] [CrossRef]
- Van, T.K.; Kang, Y.; Fukui, T.; Sakurai, K.; Iwasaki, K.; Aikawa, Y.; Phuong, N.M. Arsenic and heavy metal accumulation by Athyrium yokoscense from contaminated soils. Soil Sci. Plant Nutr. 2006, 52, 701–710. [Google Scholar] [CrossRef]
- Francesconi, K.; Visoottiviseth, P.; Sridokchan, W.; Goessler, W. Arsenic species in an arsenic hyperaccumulating fern, Pityrogramma calomelanos: A potential phytoremediator of arsenic-contaminated soils. Sci. Total Environ. 2002, 284, 27–35. [Google Scholar] [CrossRef]
- Corzo Remigio, A.; Edraki, M.; Baker, A.J.M.; van der Ent, A. Root responses to localised soil arsenic enrichment in the fern Pityrogramma calomelanos var. austroamericana grown in rhizoboxes. Plant Physiol. Biochem. 2021, 164, 147–159. [Google Scholar] [CrossRef]
- Ma, L.Q.; Komar, K.M.; Tu, C.; Zhang, W.; Cai, Y.; Kennelley, E.D. A fern that hyperaccumulates arsenic. Nature 2001, 409, 579. [Google Scholar] [CrossRef] [PubMed]
- Danh, L.T.; Truong, P.; Mammucari, R.; Foster, N. A Critical Review of the Arsenic Uptake Mechanisms and Phytoremediation Potential of Pteris Vittata. Int. J. Phytoremediation 2014, 16, 429–453. [Google Scholar] [CrossRef]
- Tu, C.; Ma, L.Q. Effects of Arsenic Concentrations and Forms on Arsenic Uptake by the Hyperaccumulator Ladder Brake. J. Environ. Qual. 2002, 31, 641–647. [Google Scholar] [CrossRef]
- Zheng, J.; Niu, T.; Wu, G.; Chen, W. One magic pteridophyte (Pteris vittata L.): Application in remediating arsenic contaminated soils and mechanism of arsenic hyperaccumulation. Front. Agric. China 2010, 4, 293–298. [Google Scholar] [CrossRef]
- Vandana, U.K.; Gulzar, A.B.M.; Singha, L.P.; Bhattacharjee, A.; Mazumder, P.B.; Pandey, P. Hyperaccumulation of arsenic by Pteris vittata, a potential strategy for phytoremediation of arsenic-contaminated soil. Environ. Sustain. 2020, 3, 169–178. [Google Scholar] [CrossRef]
- Cai, C.; Lanman, N.A.; Withers, K.A.; DeLeon, A.M.; Wu, Q.; Gribskov, M.; Salt, D.E.; Banks, J.A. Three Genes Define a Bacterial-Like Arsenic Tolerance Mechanism in the Arsenic Hyperaccumulating Fern Pteris vittata. Curr. Biol. 2019, 29, 1625–1633.e3. [Google Scholar] [CrossRef]
- Ditusa, S.F.; Fontenot, E.B.; Wallace, R.W.; Silvers, M.A.; Steele, T.N.; Elnagar, A.H.; Dearman, K.M.; Smith, A.P. A member of the Phosphate transporter 1 (Pht1) family from the arsenic-hyperaccumulating fern Pteris vittata is a high-affinity arsenate transporter. New Phytol. 2016, 209, 762–772. [Google Scholar] [CrossRef]
- He, Z.; Yan, H.; Chen, Y.; Shen, H.; Xu, W.; Zhang, H.; Shi, L.; Zhu, Y.G.; Ma, M. An aquaporin PvTIP4;1 from Pteris vittata may mediate arsenite uptake. New Phytol. 2016, 209, 746–761. [Google Scholar] [CrossRef] [PubMed]
- Indriolo, E.; Na, G.N.; Ellis, D.; Salt, D.E.; Banks, J.A. A vacuolar arsenite transporter necessary for arsenic tolerance in the arsenic hyperaccumulating fern Pteris vittata is missing in flowering plants. Plant Cell 2010, 22, 2045–2057. [Google Scholar] [CrossRef] [PubMed]
- Lombi, E.; Zhao, F.J.; Fuhrmann, M.; Ma, L.Q.; McGrath, S.P. Arsenic distribution and speciation in the fronds of the hyperaccumulator Pteris vittata. New Phytol. 2002, 156, 195–203. [Google Scholar] [CrossRef]
- Ellis, D.R.; Gumaelius, L.; Indriolo, E.; Pickering, I.J.; Banks, J.A.; Salt, D.E. A novel arsenate reductase from the arsenic hyperaccumulating fern Pteris vittata. Plant Physiol. 2006, 141, 1544–1554. [Google Scholar] [CrossRef]
- Condamine, F.L.; Silvestro, D.; Koppelhus, E.B.; Antonelli, A. The rise of angiosperms pushed conifers to decline during global cooling. Proc. Natl. Acad. Sci. USA 2020, 117, 28867–28875. [Google Scholar] [CrossRef]
- Kandziora-Ciupa, M.; Ciepał, R.; Nadgórska-Socha, A.; Barczyk, G. Accumulation of heavy metals and antioxidant responses in Pinus sylvestris L. needles in polluted and non-polluted sites. Ecotoxicology 2016, 25, 970–981. [Google Scholar] [CrossRef]
- Saladin, G. Phytoextraction of Heavy Metals: The Potential Efficiency of Conifers. In Heavy Metal Contamination of Soils; Springer International Publishing: Cham, Switzerland, 2015; pp. 333–353. [Google Scholar]
- Rodríguez Martin, J.A.C.; Gutiérrez, M.; Torrijos, N. Nanos Wood and bark of Pinus halepensis as archives of heavy metal pollution in the Mediterranean Region. Environ. Pollut. 2018, 239, 438–447. [Google Scholar] [CrossRef]
- Zeiner, M.; Juranović Cindrić, I. Accumulation of Major, Minor and Trace Elements in Pine Needles (Pinus nigra) in Vienna (Austria). Molecules 2021, 26, 3318. [Google Scholar] [CrossRef] [PubMed]
- Kukkola, E.; Rautio, P.; Huttunen, S. Stress indications in copper- and nickel-exposed Scots pine seedlings. Environ. Exp. Bot. 2000, 43, 197–210. [Google Scholar] [CrossRef]
- Curguz, V.G.; Raicevic, V.; Veselinovic, M.; Tabakovic-Tosic, M.; Vilotic, D. Influence of heavy metals on seed germination and growth of Picea abies L. Karst. Polish J. Environ. Stud. 2012, 21, 355–361. [Google Scholar]
- Ivanov, Y.V.; Savochkin, Y.V.; Kuznetsov, V.V. Scots pine as a model plant for studying the mechanisms of conifers adaptation to heavy metal action: 2. Functioning of antioxidant enzymes in pine seedlings under chronic zinc action. Russ. J. Plant Physiol. 2012, 59, 50–58. [Google Scholar] [CrossRef]
- Radotic, K.; Ducic, T.; Mutavdic, D. Changes in peroxidase activity and isoenzymes in spruce needles after exposure to different concentrations of cadmium. Environ. Exp. Bot. 2000, 44, 105–113. [Google Scholar] [CrossRef]
- Schröder, P.; Fisher, C.; Debus, R.; Wenzel, A. Reaction of detoxification mechanisms in suspension cultured spruce cells (Picea abies L. Karst.) to heavy metals in pure mixture and in soil eluates. Environ. Sci. Pollut. Res. 2003, 10, 225–234. [Google Scholar] [CrossRef]
- DalCorso, G.; Manara, A.; Furini, A. An overview of heavy metal challenge in plants: From roots to shoots. Metallomics 2013, 5, 1117–1132. [Google Scholar] [CrossRef]
- Gekeler, W.; Grill, E.; Winnacker, E.L.; Zenk, M.H. Survey of the Plant Kingdom for the Ability to Bind Heavy Metals through Phytochelatins. Z. Für Nat. C 1989, 44, 361–369. [Google Scholar] [CrossRef]
- Thangavel, P.; Long, S.; Minocha, R. Changes in phytochelatins and their biosynthetic intermediates in red spruce (Picea rubens Sarg.) cell suspension cultures under cadmium and zinc stress. Plant Cell Tissue Organ Cult. 2007, 88, 201–216. [Google Scholar] [CrossRef]
- Hachani, C.; Lamhamedi, M.S.; Cameselle, C.; Gouveia, S.; Zine El Abidine, A.; Khasa, D.P.; Béjaoui, Z. Effects of Ectomycorrhizal Fungi and Heavy Metals (Pb, Zn, and Cd) on Growth and Mineral Nutrition of Pinus halepensis Seedlings in North Africa. Microorganisms 2020, 8, 2033. [Google Scholar] [CrossRef] [PubMed]
- Chudzińska, E.; Diatta, J.B.; Wojnicka-Półtorak, A. Adaptation strategies and referencing trial of Scots and black pine populations subjected to heavy metal pollution. Environ. Sci. Pollut. Res. Int. 2014, 21, 2165–2177. [Google Scholar] [CrossRef]
- Wegiel, A.; Bielinis, E.; Polowy, K. Heavy metals accumulation in Scots pine stands of different densities growing on not contaminated forest area (northwestern Poland). Austrian J. For. Sci. 2018, 35, 259–281. [Google Scholar]
- Moussavou Moudouma, C.F.; Riou, C.; Gloaguen, V.; Saladin, G. Hybrid larch (Larix x eurolepis Henry): A good candidate for cadmium phytoremediation? Environ. Sci. Pollut. Res. 2013, 20, 1889–1894. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, Y.V.; Kartashov, A.V.; Ivanova, A.I.; Savochkin, Y.V.; Kuznetsov, V.V. Effects of zinc on Scots pine (Pinus sylvestris L.) seedlings grown in hydroculture. Plant Physiol. Biochem. 2016, 102, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pulford, I.D.; Watson, C. Phytoremediation of heavy metal-contaminated land by trees-A review. Environ. Int. 2003, 29, 529–540. [Google Scholar] [CrossRef]
- Schvartzman, M.S.; Corso, M.; Fataftah, N.; Scheepers, M.; Nouet, C.; Bosman, B.; Carnol, M.; Motte, P.; Verbruggen, N.; Hanikenne, M. Adaptation to high zinc depends on distinct mechanisms in metallicolous populations of Arabidopsis halleri. New Phytol. 2018, 218, 269–282. [Google Scholar] [CrossRef]
- Manara, A.; Fasani, E.; Furini, A.; DalCorso, G. Evolution of the metal hyperaccumulation and hypertolerance traits. Plant Cell Environ. 2020, 43, 2969–2986. [Google Scholar] [CrossRef]
- Verbruggen, N.; Hanikenne, M.; Clemens, S. A more complete picture of metal hyperaccumulation through next-generation sequencing technologies. Front. Plant Sci. 2013, 4, 388. [Google Scholar] [CrossRef]
- Ingle, R.A.; Mugford, S.T.; Rees, J.D.; Campbell, M.M.; and Smith, J.A.C. Constitutively high expression of the histidine biosynthetic pathway contributes to nickel tolerance in hyperaccumulator plants. Plant Cell 2005, 17, 2089–2106. [Google Scholar] [CrossRef]
- Deinlein, U.; Weber, M.; Schmidt, H.; Rensch, S.; Trampczynska, A.; Hansen, T.H.; Husted, S.; Schjoerring, J.K.; Talke, I.N.; Krämer, U.; et al. Elevated nicotianamine levels in Arabidopsis halleri roots play a key role in zinc hyperaccumulation. Plant Cell 2012, 24, 708–723. [Google Scholar] [CrossRef] [PubMed]
- Corso, M.; An, X.; Jones, C.Y.; Gonzalez-Doblas, V.; Schvartzman, M.S.; Malkowski, E.; Willats, W.G.T.; Hanikenne, M.; Verbruggen, N. Adaptation of Arabidopsis halleri to extreme metal pollution through limited metal accumulation involves changes in cell wall composition and metal homeostasis. New Phytol. 2021, 230, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Hanikenne, M.; Talke, I.N.; Haydon, M.J.; Lanz, C.; Nolte, A.; Motte, P.; Kroymann, J.; Weigel, D.; Kraemer, U. Evolution of metal hyperaccumulation required cisregulatory changes and triplication of HMA4. Nature 2008, 453, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Rai, K.K.; Pandey, N.; Meena, R.P.; Rai, S.P. Biotechnological strategies for enhancing heavy metal tolerance in neglected and underutilized legume crops: A comprehensive review. Ecotoxicol. Environ. Saf. 2021, 208, 111750. [Google Scholar] [CrossRef] [PubMed]
- Koźmińska, A.; Wiszniewska, A.; Hanus-Fajerska, E.; Muszyńska, E. Recent strategies of increasing metal tolerance and phytoremediation potential using genetic transformation of plants. Plant Biotechnol. Rep. 2018, 12, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xu, W.; Shen, H.; Yan, H.; Xu, W.; He, Z.; Ma, M. Engineering arsenic tolerance and hyperaccumulation in plants for phytoremediation by a PvACR3 transgenic approach. Environ. Sci. Technol. 2013, 47, 9355–9362. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fasani, E.; Li, M.; Varotto, C.; Furini, A.; DalCorso, G. Metal Detoxification in Land Plants: From Bryophytes to Vascular Plants. STATE of the Art and Opportunities. Plants 2022, 11, 237. https://doi.org/10.3390/plants11030237
Fasani E, Li M, Varotto C, Furini A, DalCorso G. Metal Detoxification in Land Plants: From Bryophytes to Vascular Plants. STATE of the Art and Opportunities. Plants. 2022; 11(3):237. https://doi.org/10.3390/plants11030237
Chicago/Turabian StyleFasani, Elisa, Mingai Li, Claudio Varotto, Antonella Furini, and Giovanni DalCorso. 2022. "Metal Detoxification in Land Plants: From Bryophytes to Vascular Plants. STATE of the Art and Opportunities" Plants 11, no. 3: 237. https://doi.org/10.3390/plants11030237
APA StyleFasani, E., Li, M., Varotto, C., Furini, A., & DalCorso, G. (2022). Metal Detoxification in Land Plants: From Bryophytes to Vascular Plants. STATE of the Art and Opportunities. Plants, 11(3), 237. https://doi.org/10.3390/plants11030237







