Calmodulin Domain Protein Kinase PiCDPK1 Regulates Pollen Tube Growth Polarity through Interaction with RhoGDI
Abstract
:1. Introduction
2. Results
2.1. Yeast 2-Hybrid Library Screening
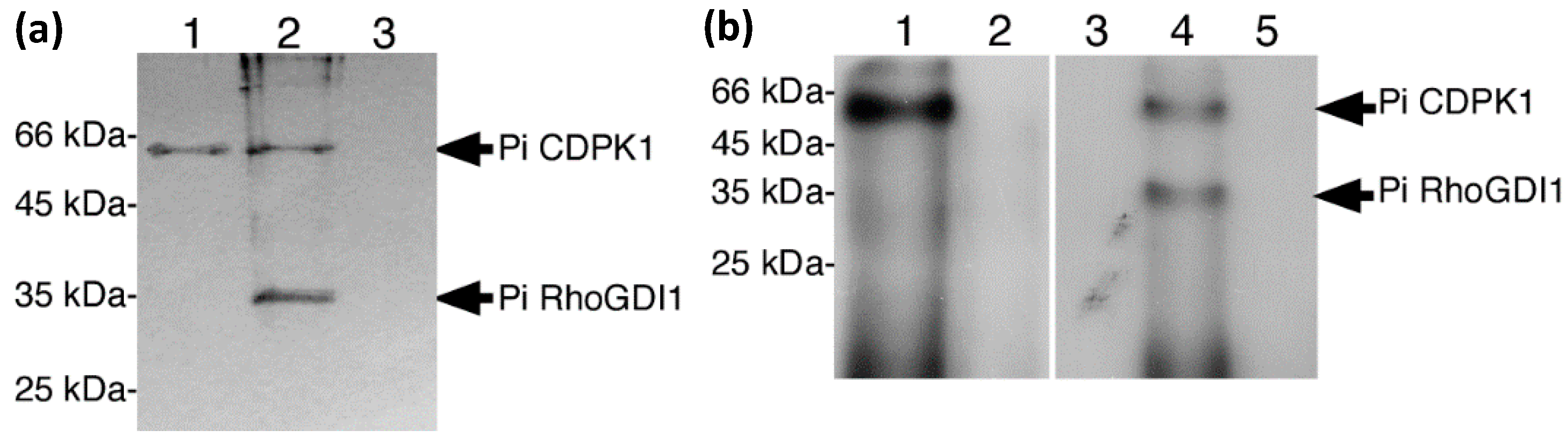
2.2. PiRhoGDI1 Interacts with and Is Phosphorylated by PiCDPK1 In Vitro
2.3. Confirmation of the Interaction of PiRhoGDI1 with ROP GTPase
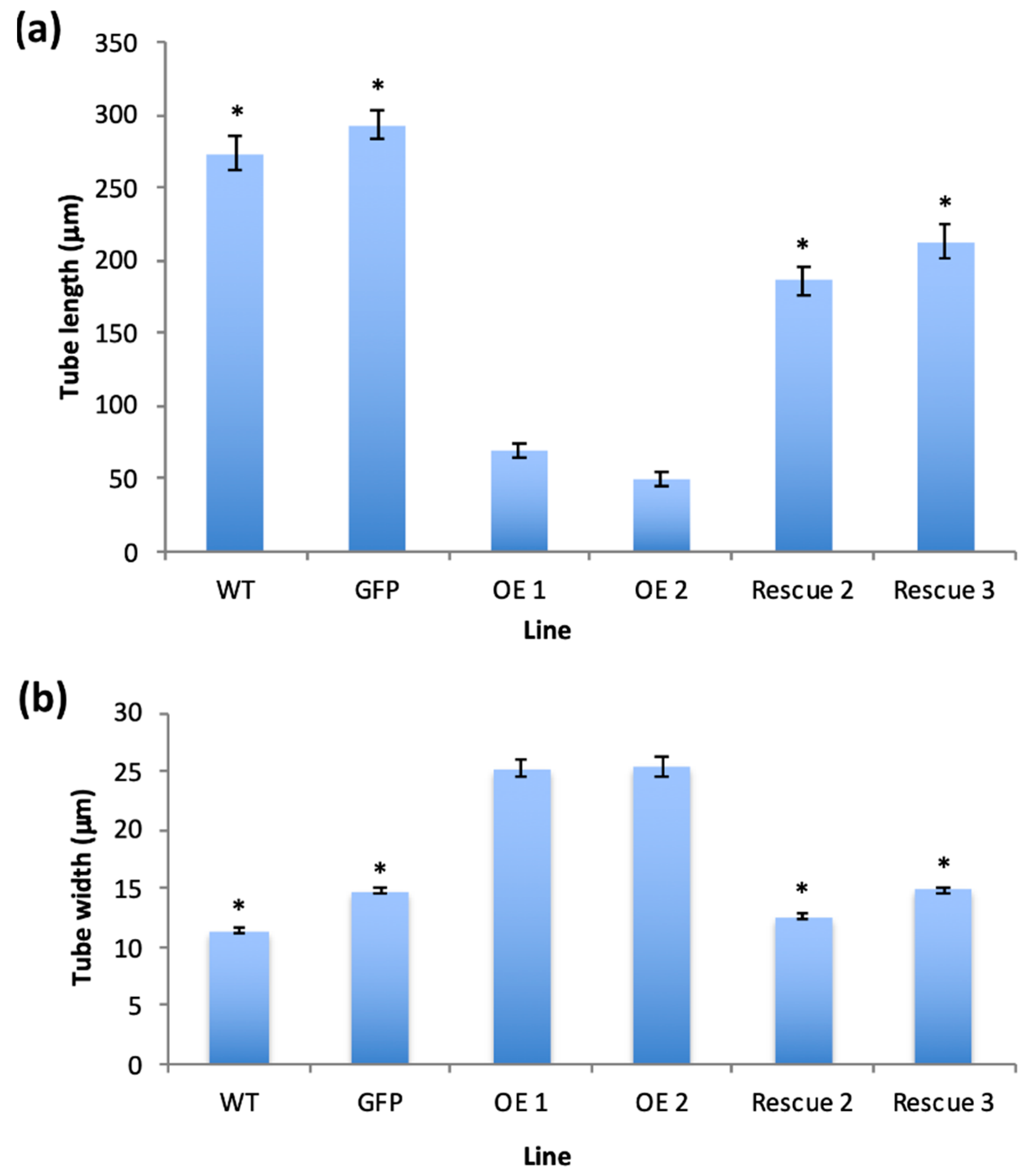
2.4. PiCDPK1 Suppresses the Effect of PiRhoGDI1 Over-Expression In Vivo
2.5. Co-Expression with PiRhoGDI1 Rescues the PiCDPK1 Overexpression Phenotype
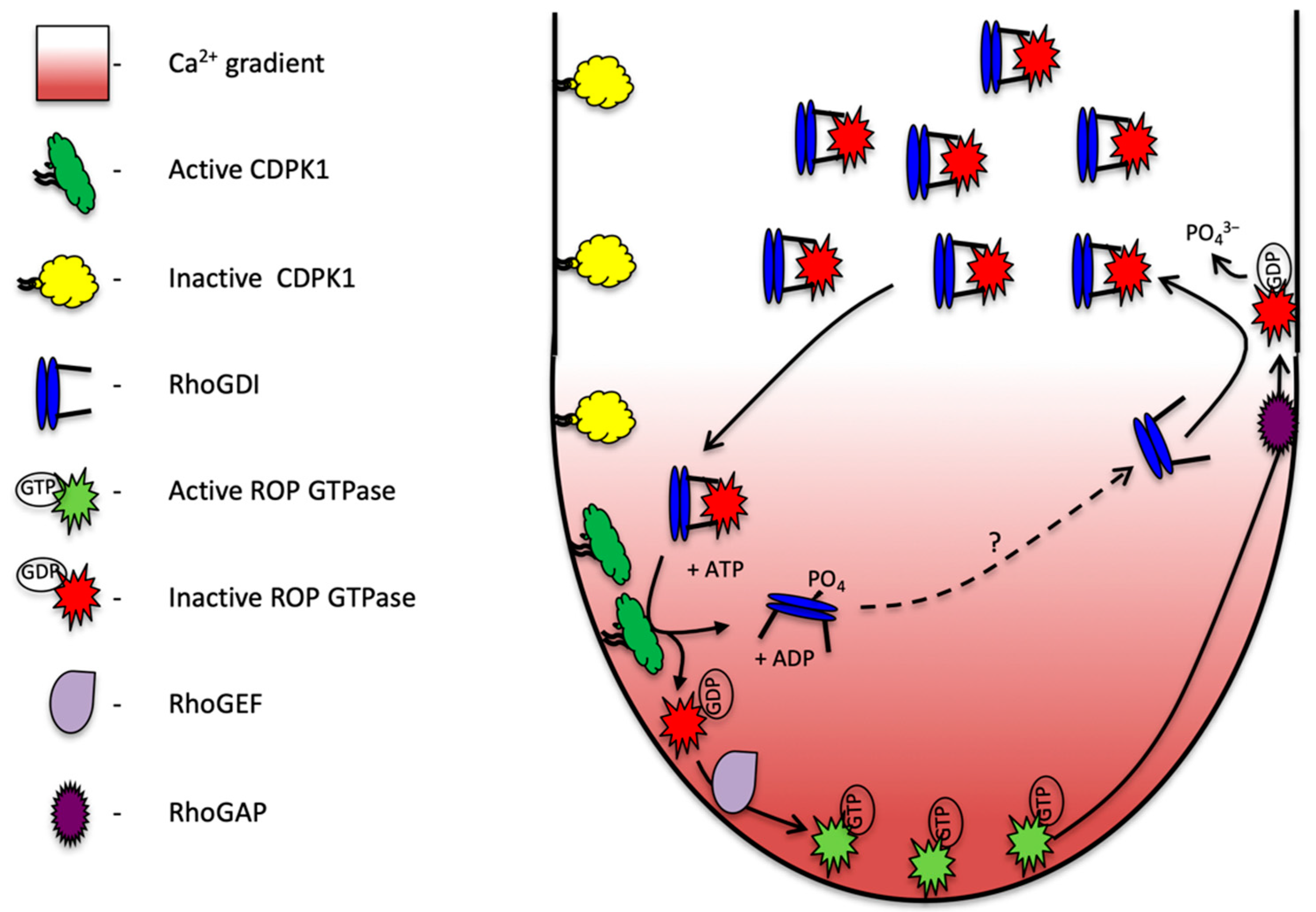
3. Discussion
4. Materials and Methods
4.1. Yeast 2-Hybrid Library Screening
4.2. Expression of Recombinant Proteins
4.3. Pull-Down Assay
4.4. Phosphorylation Assay
4.5. Stable Expression in Tobacco
4.6. Analysis of Transformed Pollen Tubes
4.7. Quantitative RT-PCR
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gilroy, S.; Swanson, S.J. Tip Growth. eLS 2017, 1–4. [Google Scholar] [CrossRef]
- Miller, D.D.; Callaham, D.A.; Gross, D.J.; Hepler, P.K. Free Ca2+ gradient in growing pollen tubes of Lilium. J. Cell Sci. 1992, 101, 7–12. [Google Scholar] [CrossRef]
- Pierson, E.S.; Miller, D.D.; Callaham, D.A.; Shipley, A.M.; Rivers, B.A.; Cresti, M.; Hepler, P.K. Pollen tube growth 1 s coupled to the extracellular calcium ion flux and the intracellular calcium gradient: Effect of BAPTA-type buffers and hypertonic media. Plant Cell 1994, 6, 1815–1828. [Google Scholar]
- Malho, R.; Trewavas, A.J. Localized apical increases of cytosolic free calcium control pollen tube orientation. Plant Cell 1996, 8, 1935–1949. [Google Scholar] [CrossRef]
- Hepler, P.K. Tip growth in pollen tubes: Calcium leads the way. Trend Plant Sci. 1997, 2, 79–80. [Google Scholar] [CrossRef]
- Messerli, M.; Robinson, K.R. Tip localized Ca2+ pulses are coincident with peak pulsatile growth rates in pollen tubes of Lilium longiflorum. J. Cell Sci. 1997, 110, 1269–1278. [Google Scholar] [CrossRef]
- Rathore, K.S.; Cork, R.J.; Robinson, K.R. A cytoplasmic gradient of Ca2+ is correlated with the growth of lily pollen tubes. Dev. Biol. 1991, 148, 612–619. [Google Scholar] [CrossRef]
- Li, Y.Q.; Zhang, H.Q.; Pierson, E.S.; Huang, F.Y.; Linskens, H.F.; Hepler, P.K.; Cresti, M. Enforced growth-rate fluctuation causes pectin ring formation in the cell wall of Lilium longiflorum pollen tubes. Planta 1996, 200, 41–49. [Google Scholar] [CrossRef]
- Zhao, L.-N.; Shen, L.-K.; Zhang, W.-Z.; Zhang, W.; Wang, Y.; Wu, W.-H. Ca2+-dependent protein kinase 11 and 24 modulate the activity of the inward rectifying K+ channels in Arabidopsis pollen tubes. Plant Cell 2013, 25, 649–661. [Google Scholar] [CrossRef] [Green Version]
- Yoon, G.M.; Dowd, P.E.; Gilroy, S.; McCubbin, A.G. Calcium-Dependent Protein Kinase isoforms in Petunia have distinct functions in pollen tube growth, including regulating polarity. Plant Cell 2006, 18, 867–878. [Google Scholar] [CrossRef] [Green Version]
- Myers, C.; Romanowsky, S.M.; Barron, Y.D.; Garg, S.; Azuse, C.L.; Curran, A.; Davis, R.M.; Hatton, J.; Harmon, A.C.; Harper, J.F. Calcium-dependent protein kinases regulate polarized tip growth in pollen tubes. Plant J. 2009, 59, 528–539. [Google Scholar] [CrossRef]
- Di Giorgio, J.A.; Bienert, G.P.; Ayub, N.D.; Yaneff, A.; Barberini, M.L.; Mecchia, M.A.; Amodeo, G.; Soto, G.C.; Muschietti, J.P. Pollen-specific aquaporins NIP4;1 and NIP4;2 are required for pollen development and pollination in Arabidopsis thaliana. Plant Cell 2016, 28, 1053–1077. [Google Scholar] [CrossRef] [Green Version]
- Gutermuth, T.; Lassig, R.; Portes, M.-T.; Maierhofer, T.; Romeis, T.; Borst, J.-W.; Hedrich, R.; Feijo, J.A.; Konrad, K.R. Pollen tube growth regulation by free anions depends on the interaction between the anion channel SLAH3 and calcium-dependent protein kinases CPK2 and CPK20. Plant Cell 2013, 25, 4525–4543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, Q.-F.; Fei, C.-F.; Dong, J.-Y.; Gu, L.-L.; Wang, Y.-F. Arabidopsis CNGC18 is a Ca2+-permeable channel. Mol. Plant 2014, 7, 739–743. [Google Scholar] [CrossRef] [Green Version]
- Lin, Y.; Yang, Z. Inhibition of pollen tube elongation by microinjected anti-Rop1Ps antibodies suggests a crucial role for Rho type GTPases in the control of tip-growth. Plant Cell 1997, 9, 1647–1659. [Google Scholar] [CrossRef] [PubMed]
- Kost, B.; Lemichez, E.; Spielhofer, P.; Hong, Y.; Tolias, K.; Carpenter, C.; Chua, N.H. Rac homologues and compartmentalized phosphatidylinositol 4,5-bisphosphate act in a common pathway to regulate polar pollen tube growth. J. Cell Biol. 1999, 145, 317–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.; Lin, Y.; Heath, R.M.; Zhu, M.X.; Yang, Z. Control of pollen tube tip growth by a Rop GTPase–dependent pathway that leads to tip-localized calcium influx. Plant Cell 1999, 11, 1731–1742. [Google Scholar] [PubMed] [Green Version]
- Zheng, Z.-L.; Yang, Z. The Rop GTPase: An emerging signaling switch in plants. Plant Mol. Biol. 2000, 44, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Fu, Y.; Dowd, P.; Li, S.; Vernoud, V.; Gilroy, S.; Yang, Z. A Rho family GTPase controls actin dynamics and tip growth via two counteracting downstream pathways in pollen tubes. J. Cell Biol. 2005, 169, 127–138. [Google Scholar] [CrossRef] [Green Version]
- Lee, Y.J.; Szumlanski, A.; Nielsen, E.; Yang, Z. Rho-GTPase-dependent filamentous actin dynamics coordinate vesicle targeting and exocytosis during tip growth. J. Cell Biol. 2008, 181, 1155–1168. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Z.; Shi, H.; Chen, B.; Zhang, R.; Huang, S.; Fun, Y. Arabidopsis RIC1 severs actin filaments at the apex to regulate pollen tube growth. Plant Cell 2015, 27, 1140–1161. [Google Scholar] [CrossRef] [Green Version]
- Chang, F.; Gu, Y.; Ma, H.; Yang, Z. AtPRK2 promotes ROP1 activation via RopGEFs in the control of polarized pollen tube growth. Mol. Plant 2013, 6, 1187–1201. [Google Scholar] [CrossRef] [Green Version]
- Li, E.; Cui, Y.; Ge, F.-R.; Chai, S.; Zhang, W.-T.; Feng, Q.-N.; Jiang, L.; Li, S.; Zhang, Y. AGC1.5 Kinase Phosphorylates RopGEFs to Control Pollen Tube Growth. Mol. Plant 2018, 11, 1198–1209. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Luo, N.; Wang, W.; Liu, Z.; Chen, J.; Zhao, L.; Tan, L.; Wang, C.; Qin, Y.; Li, C.; et al. The REN4 rheostat dynamically coordinates the apical and lateral domains of Arabidopsis pollen tubes. Nat. Commun. 2018, 9, 2573. [Google Scholar] [CrossRef]
- Zhang, Y.; He, J.; McCormick, S. Two Arabidopsis AGC kinases are critical for the polarized growth of pollen tubes. Plant J. 2009, 58, 474–484. [Google Scholar] [CrossRef]
- Klahre, U.; Becker, C.; Schmitt, A.C.; Kost, B. Nt-RhoGDI2 regulates Rac/Rop signaling and polar cell growth in tobacco pollen tubes. Plant J. 2006, 46, 1018–1031. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.-N.; Kang, H.; Song, S.-J.; Ge, F.-R.; Zhang, Y.-L.; Li, E.; Li, S.; Zhang, Y. Arabidopsis RhoGDIs are critical for cellular homeostasis of pollen tubes. Plant Physiol. 2016, 170, 841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fauré, J.; Vignais, P.V.; Dagher, M.C. Phosphoinositide-dependent activation of Rho A involves partial opening of the RhoA/Rho-GDI complex. Eur. J. Biochem. 1999, 262, 879–889. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Mata, R.; Boulter, E.; Burridge, K. The ‘invisible hand’: Regulation of RHO GTPases by RHOGDIs. Nat. Rev. Mol. Cell Biol. 2011, 12, 493–504. [Google Scholar] [CrossRef] [Green Version]
- Mehta, D.; Rahman, A.; Malik, A.B. Protein kinase C signals Rho-guanine nucleotide dissociation inhibitor phosphorylation and Rho activation and regulates the endothelial cell barrier function. J. Biol. Chem. 2001, 276, 22614–22620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DerMardirossian, C.; Schnelzer, A.; Bokoch, G.M. Phosphorylation of RhoGDI by Pak1 mediates dissociation of Rac GTPase. Mol. Cell 2004, 15, 117–127. [Google Scholar] [CrossRef]
- Kaothien, P.; Ok, S.H.; Shuai, B.; Wengier, D.; Cotter, R.; Kelley, D.; Kiriakopolos, S.; Muschietti, J.; McCormick, S. Kinase partner protein interacts with the LePRK1 and LePRK2 receptor kinases and plays a role in polarized pollen tube growth. Plant J. 2005, 42, 492–503. [Google Scholar] [CrossRef]
- Zhang, Y.; McCormick, S. A distinct mechanism regulating a pollen-specific guanine nucleotide exchange factor for the small GTPase Rop in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2007, 104, 18830–18835. [Google Scholar] [CrossRef] [Green Version]
- Zhu, L.; Chu, L.-C.; Liang, Y.; Zhang, X.-Q.; Chen, L.-Q.; Ye, D. The Arabidopsis CrRLK1L protein kinases BUPS1 and BUPS2 are required for normal growth of pollen tubes in the pistil. Plant J. 2018, 95, 474–486. [Google Scholar] [CrossRef] [Green Version]
- Battey, N.H.; Venis, M.A. Calcium-dependent hydrophobic interaction chromatography. Meth. Mol. Biol. 1992, 11, 73–80. [Google Scholar] [CrossRef]
- Kost, B. Spatial control of Rho (Rac-Rop) signaling in tip-growing plant cells. Trends Cell Biol. 2008, 18, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Twell, D.; Klein, T.M.; Fromm, M.; McCormick, S. Transient expression of chimeric genes delivered into pollen by microprojectile bombardment. Plant Physiol. 1989, 91, 1270–1274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, G.W.; Delaney, S.K. Stable internal reference genes for normalization of real-time RT-PCR in tobacco (Nicotiana tabacum) during development and abiotic stress. Mol. Genet. Genom. 2010, 283, 233–241. [Google Scholar] [CrossRef]
- Klahre, U.; Kost, B. Tobacco RhoGTPase ACTIVATING PROTEIN1 spatially restricts signaling of RAC/Rop to the apex of pollen tubes. Plant Cell 2006, 18, 3033–3046. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Gong, P.; Guo, J.; Li, H.; Li, R.; Xing, W.; Yang, Z.; Guan, Y. Glycolysis regulates pollen tube polarity via Rho GTPase signaling. PLoS Genet. 2018, 14, e1007373. [Google Scholar] [CrossRef] [Green Version]
- Gu, Y.; Li, S.; Lord, E.M.; Yang, Z. Members of a novel class of Arabidopsis Rho guanine nucleotide exchange factors control Rho GTPase-dependent polar growth. Plant Cell 2006, 18, 366–381. [Google Scholar] [CrossRef] [Green Version]
- Hwang, J.U.; Gu, Y.; Lee, Y.J.; Yang, Z. Oscillatory ROP GTPase activation leads the oscillatory polarized growth of pollen tubes. Mol. Biol. Cell 2005, 16, 5385–5399. [Google Scholar] [CrossRef]
- Cole, R.A.; Fowler, J.E. Polarized growth: Maintaining focus on the tip. Curr. Opin. Plant Biol. 2006, 9, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Malhó, R.; Read, N.D.; Pais, M.S.; Trewavas, A.J. Role of cytosolic free calcium in the reorientation of pollen tube growth. Plant J. 1994, 5, 331–341. [Google Scholar] [CrossRef]
- Cho, H.J.; Kim, J.T.; Lee, S.J.; Hwang, Y.S.; Park, S.Y.; Kim, B.Y.; Yoo, J.; Hong, K.S.; Min, J.K.; Lee, C.H.; et al. Protein phosphatase 1B dephosphorylates Rho guanine nucleotide dissociation inhibitor 1 and suppresses cancer cell migration and invasion. Cancer Lett. 2018, 417, 141–151. [Google Scholar] [CrossRef]
- Foissner, I.; Grolig, F.; Obermeyer, G. Reversible protein phosphorylation regulates the dynamic organization of the pollen tube cytoskeleton: Effects of calyculin A and okadaic acid. Protoplasma 2002, 220, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Malhó, R.; Read, N.D.; Trewavas, A.J.; Pais, M.S. Calcium channel activity during pollen tube growth and reorientation. Plant Cell 1995, 7, 1173–1184. [Google Scholar] [CrossRef] [Green Version]
- Dutta, R.; Robinson, K.R. Identification and characterization of stretch-activated ion channels in pollen protoplasts. Plant Physiol. 2004, 135, 1398–1406. [Google Scholar] [CrossRef] [Green Version]
- Hamilton, E.S.; Jensen, G.S.; Maksaev, G.; Katims, A.; Sherp, A.M.; Haswell, E.S. Mechano-sensitive channel MSL8 regulates osmotic forces during pollen hydration and germination. Science 2015, 350, 438–441. [Google Scholar] [CrossRef] [Green Version]
- Hamilton, E.S.; Haswell, E.S. The tension-sensitive ion transport activity of MSL8 is critical for its function in pollen hydration and germination. Plant Cell Physiol. 2017, 58, 1222–1237. [Google Scholar] [CrossRef] [Green Version]
- Lush, W.M.; Grieser, F.; Wolters-Arts, M. Directional guidance of Nicotiana alata pollen tubes in vitro and on the stigma. Plant Physiol. 1998, 118, 733–741. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Song, J.; Tian, X.; Zhang, H.; Li, L.; Zhu, H. Arabidopsis PRK6 interacts specifically with AtRopGEF8/12 and induces depolarized growth of pollen tubes when overexpressed. Sci. China Life Sci. 2018, 61, 100–112. [Google Scholar] [CrossRef]
- Ménesi, D.; Klement, É.; Ferenc, G.; Fehér, A. The Arabidopsis Rho of Plants GTPase ROP1 Is a Potential Calcium-Dependent Protein Kinase (CDPK) Substrate. Plants 2021, 10, 2053. [Google Scholar] [CrossRef]
- Li, H.; Shen, J.J.; Zheng, Z.L.; Lin, Y.; Yang, Z. The Rop GTPase switch controls multiple developmental processes in Arabidopsis. Plant Physiol. 2001, 126, 670–684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carol, R.J.; Takeda, S.; Linstead, P.; Durrant, M.C.; Kakesova, H.; Derbyshire, P.; Drea, S.; Zarsky, V.; Dolan, L. A RhoGDP dissociation inhibitor spatially regulates growth in root hair cells. Nature 2005, 438, 1013–1016. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Claes, A.R.; Grebe, T.; Hermkes, R.; Viotti, C.; Ikeda, Y.; Grebe, M. Auxin and ROP GTPase signaling of polar nuclear migration in root epidermal hair cells. Plant Physiol. 2018, 176, 378–391. [Google Scholar] [CrossRef] [Green Version]
- Ge, F.R.; Chai, S.; Li, S.; Zhang, Y. Targeting and signaling of Rho of plants guanosine triphosphatases require synergistic interaction between guanine nucleotide inhibitor and vesicular trafficking. J. Integr. Plant. Biol. 2020, 62, 1484–1499. [Google Scholar] [CrossRef]
- Harper, J.F.; Harmon, A. Plants, symbiosis and parasites: A calcium signaling connection. Nat. Rev. Mol. Cell. Biol. 2005, 6, 556–566. [Google Scholar] [CrossRef]
- Hetherington, A.M.; Brownlee, C. The generation of Ca2+ signals in plants. Ann. Rev. Plant Biol. 2004, 55, 401–427. [Google Scholar] [CrossRef] [Green Version]
- Costa, A.; Navazio, L.; Szabo, I. The contribution of organelles to plant intracellular calcium signaling. J. Exp. Bot. 2018, 69, 4175–4193. [Google Scholar] [CrossRef] [Green Version]
- Kudla, J.; Becker, D.; Grill, E.; Hedrich, R.; Hippler, M.; Kummer, U.; Parniske, M.; Romeis, T.; Schumacher, K. Advances and current challenges in calcium signaling. New Phytol. 2018, 218, 414–431. [Google Scholar] [CrossRef]
- Satterlee, J.S.; Sussman, M.R. Unusual membrane-associated protein kinases in higher plants. J. Membr. Biol. 1998, 164, 205–213. [Google Scholar] [CrossRef]
- Cheng, S.H.; Willmann, M.R.; Chen, H.C.; Sheen, J. Calcium signaling through protein kinases. The Arabidopsis calcium-dependent protein kinase gene family. Plant Physiol. 2002, 129, 469–485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ludwig, A.A.; Romeis, T.; Jones, J.D. CDPK-mediated signaling pathways: Specificity and cross-talk. J. Exp. Bot. 2004, 55, 181–188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harper, J.F.; Breton, G.; Harmon, A. Decoding Ca2+ signals through plant protein kinases. Ann. Rev. Plant Biol. 2004, 55, 263–288. [Google Scholar] [CrossRef]
- Shi, S.; Li, S.; Asim, M.; Mao, J.; Xu, D.; Ullah, Z.; Liu, G.; Wang, Q.; Liu, H. The Arabidopsis calcium-dependent protein kinases (CDPKs) and their roles in plant growth regulation and abiotic stress responses. Int. J. Mol. Sci. 2018, 19, 1900. [Google Scholar] [CrossRef] [Green Version]
- Singh, A.; Sagar, S.; Biswas, D.K. Calcium dependent protein kinase, a versatile player in plant stress management and development. Crit. Rev. Plant Sci. 2017, 36, 336–352. [Google Scholar] [CrossRef]
- James, P.; Halladay, J.; Craig, E.A. Genomic libraries and a host strain designed for highly efficient two-hybrid selection in yeast. Genetics 1996, 144, 1425–1436. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scheible, N.; Yoon, G.M.; McCubbin, A.G. Calmodulin Domain Protein Kinase PiCDPK1 Regulates Pollen Tube Growth Polarity through Interaction with RhoGDI. Plants 2022, 11, 254. https://doi.org/10.3390/plants11030254
Scheible N, Yoon GM, McCubbin AG. Calmodulin Domain Protein Kinase PiCDPK1 Regulates Pollen Tube Growth Polarity through Interaction with RhoGDI. Plants. 2022; 11(3):254. https://doi.org/10.3390/plants11030254
Chicago/Turabian StyleScheible, Nolan, Gyeong Mee Yoon, and Andrew G. McCubbin. 2022. "Calmodulin Domain Protein Kinase PiCDPK1 Regulates Pollen Tube Growth Polarity through Interaction with RhoGDI" Plants 11, no. 3: 254. https://doi.org/10.3390/plants11030254






