Salt Stress Differentially Affects the Primary and Secondary Metabolism of Peppers (Capsicum annuum L.) According to the Genotype, Fruit Part, and Salinity Level
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Fruit Sampling
2.3. Extraction of Sugars and Organic Acids
2.4. Extraction of Phenolics and Capsaicinoids
2.5. Chemicals
2.6. Statistical Analysis
3. Results
3.1. Individual and Total Sugars
3.2. Individual and Total Organic Acids
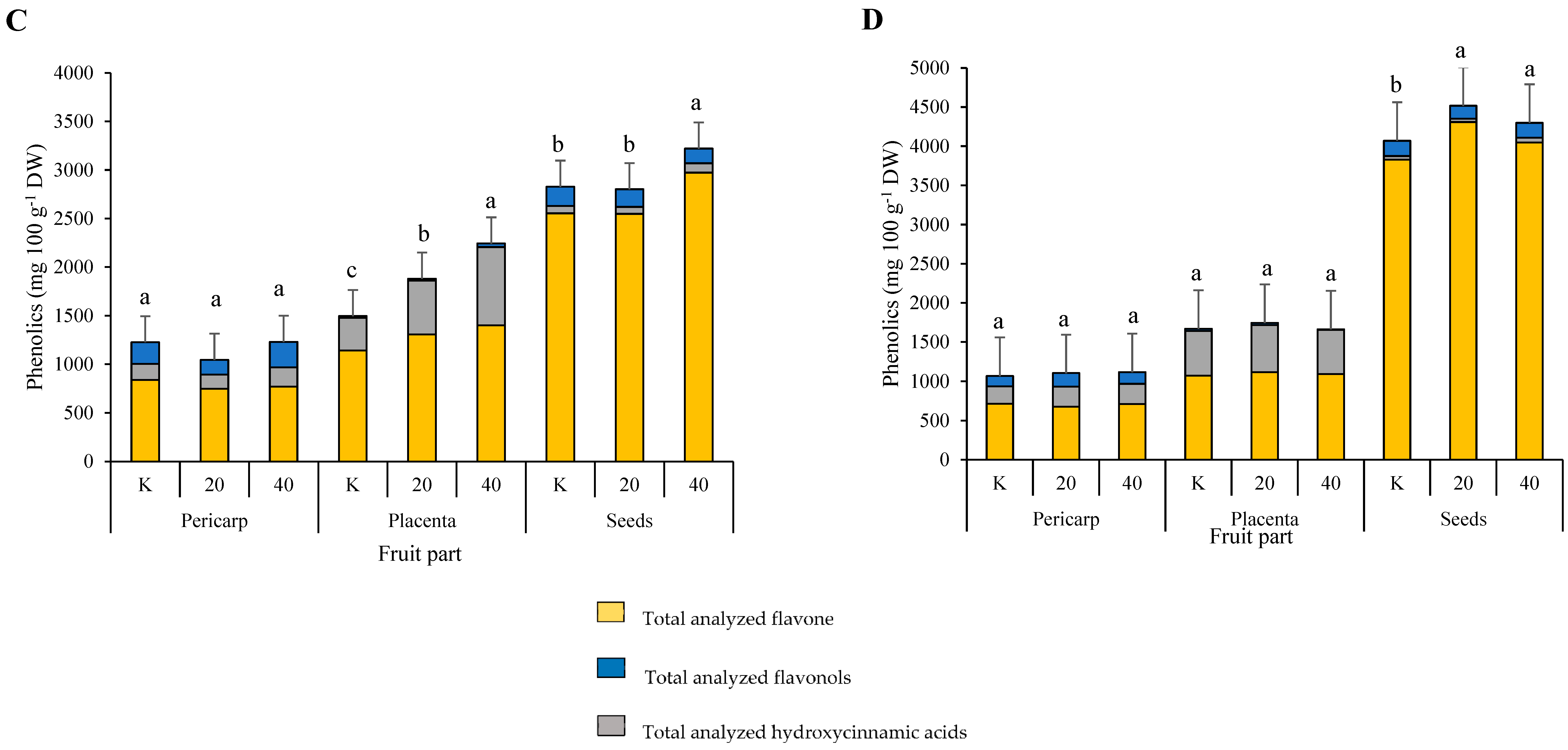
3.3. Individual and Total Analyzed Phenolics
3.3.1. Identification of Individual Phenolics
3.3.2. Quantification of Individual Phenolics
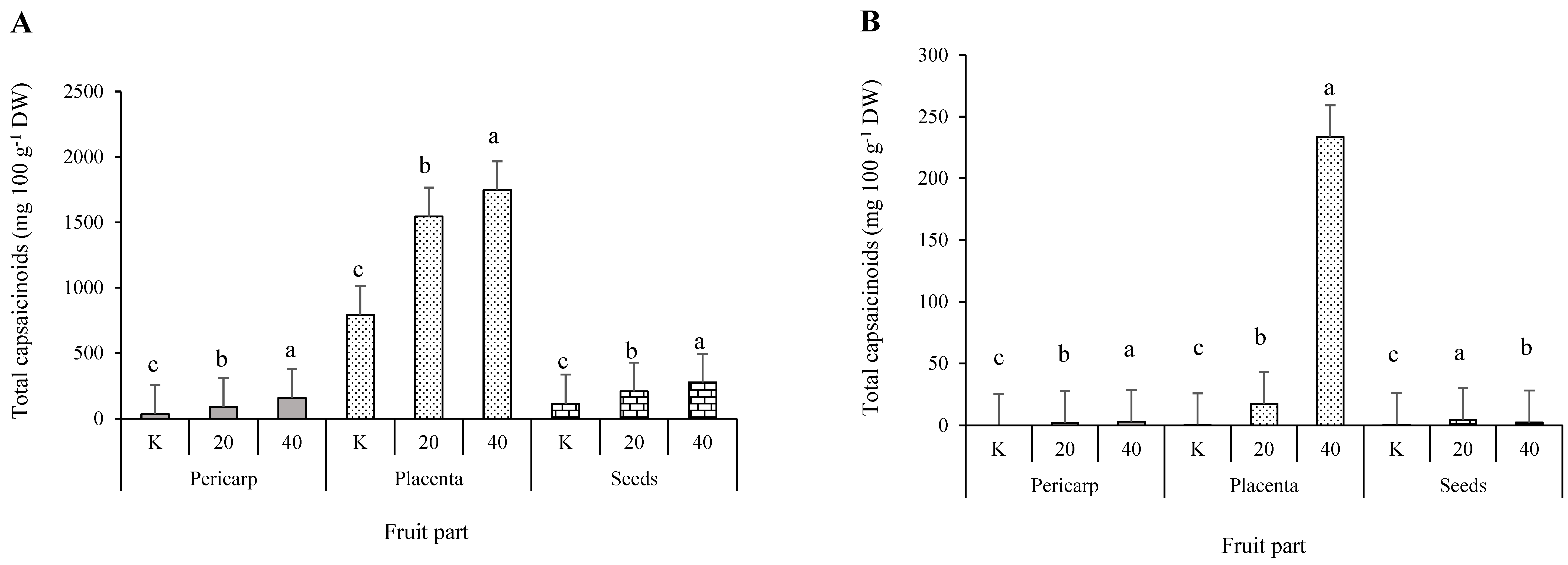
3.3.3. Individual and Total Capsaicinoids
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Robert, L.J.; Gloria, E.B.; da Costa, B.F.R.; Terry, B.; Yu-Yu, C.; Amanda, H.-K.; Neftali, O.-A.; Pasquale, T.; Aniko, V.; Carolina Carrizo, G.; et al. Capsicum—An Abbreviated Compendium. J. Am. Soc. Hortic. Sci. 2019, 144, 3–22. [Google Scholar] [CrossRef] [Green Version]
- García-Gaytán, V.; Gómez-Merino, F.C.; Trejo-Téllez, L.I.; Baca-Castillo, G.A.; García-Morales, S. The chilhuacle chili (Capsicum annuum L.) in Mexico: Description of the variety, its cultivation, and uses. Int. J. Agron. 2017, 2017, 5641680. [Google Scholar] [CrossRef] [Green Version]
- Widuri, L.I.; Lakitan, B.; Sakagami, J.; Yabuta, S.; Kartika, K.; Siaga, E. Short-term drought exposure decelerated growth and photosynthetic activities in chili pepper (Capsicum annuum L.). Ann. Agric. Sci. 2020, 65, 149–158. [Google Scholar] [CrossRef]
- Kaouther, Z.; Mariem, B.; Fardaous, M.; Cherif, H.; Mariem, C. Impact of salt stress (NaCl) on growth, chlorophyll content and fluorescence of Tunisian cultivars of chili pepper (Capsicum frutescens L.). J. Stress Physiol. Biochem. 2012, 8, 236–252. [Google Scholar]
- Saha, S.; Hossain, M.; Rahman, M.M.-E.; Kuo, C.; Abdullah, S. Effect of high temperature stress on the performance of twelve sweet pepper genotypes. Bangladesh J. Agric. Res. 2010, 35, 525–534. [Google Scholar] [CrossRef]
- Welbaum, G. Vegetable Production and Practices; Welbaum, G.E., Ed.; CAB International: Wallingforth, UK, 2015; p. 486. [Google Scholar]
- Shahid, S.A.; Zaman, M.; Heng, L. Soil Salinity: Historical Perspectives and a World Overview of the Problem. In Guideline for Salinity Assessment, Mitigation and Adaptation Using Nuclear and Related Techniques; Zaman, M., Shahid, S.A., Heng, L., Eds.; Springer: Cham, Switzerland, 2018; pp. 43–53. [Google Scholar]
- Corwin, D.L.; Scudiero, E. Chapter One-Review of soil salinity assessment for agriculture across multiple scales using proximal and/or remote sensors. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: Cambridge, MA, USA, 2019; Volume 158, pp. 1–130. [Google Scholar]
- Schmöckel, S.M.; Jarvis, D.E. Salt Stress. In Encyclopedia of Applied Plant Sciences, 2nd ed.; Thomas, B., Murray, B.G., Murphy, D.J., Eds.; Academic Press: Oxford, UK, 2017; pp. 40–43. [Google Scholar]
- Elkelish, A.A.; Alnusaire, T.S.; Soliman, M.H.; Gowayed, S.; Senousy, H.H.; Fahad, S. Calcium availability regulates antioxidant system, physio-biochemical activities and alleviates salinity stress mediated oxidative damage in soybean seedlings. J. Appl. Bot. Food Qual. 2019, 92, 258–266. [Google Scholar]
- Abdelaal, K.A.; EL-Maghraby, L.M.; Elansary, H.; Hafez, Y.M.; Ibrahim, E.I.; El-Banna, M.; El-Esawi, M.; Elkelish, A. Treatment of Sweet Pepper with Stress Tolerance-Inducing Compounds Alleviates Salinity Stress Oxidative Damage by Mediating the Physio-Biochemical Activities and Antioxidant Systems. Agronomy 2020, 10, 26. [Google Scholar] [CrossRef] [Green Version]
- Waskiewicz, A.; Muzolf-Panek, M.; Golinski, P. Phenolic Content Changes in Plants Under Salt Stress; Springer: Berlin/Heidelberg, Germany, 2013; pp. 283–314. [Google Scholar]
- Gupta, B.; Huang, B. Mechanism of Salinity Tolerance in Plants: Physiological, Biochemical, and Molecular Characterization. Int. J. Genom. 2014, 2014, 701596. [Google Scholar] [CrossRef]
- Fidanka, T.; Nicolas, P.; Dimitrios, S. Comparative Effects of NaCl and CaCl2 Salinity on Cucumber Grown in a Closed Hydroponic System. HortScience HortSci. 2006, 41, 437–441. [Google Scholar] [CrossRef] [Green Version]
- Villaseñor-Aguilar, M.-J.; Bravo-Sánchez, M.-G.; Padilla-Medina, J.-A.; Vázquez-Vera, J.L.; Guevara-González, R.-G.; García-Rodríguez, F.-J.; Barranco-Gutiérrez, A.-I. A Maturity Estimation of Bell Pepper (Capsicum annuum L.) by Artificial Vision System for Quality Control. Appl. Sci. 2020, 10, 5097. [Google Scholar] [CrossRef]
- Maršić, N.K.; Štolfa, P.; Vodnik, D.; Košmelj, K.; Mikulič-Petkovšek, M.; Kump, B.; Vidrih, R.; Kokalj, D.; Piskernik, S.; Ferjančič, B.; et al. Physiological and Biochemical Responses of Ungrafted and Grafted Bell Pepper Plants (Capsicum annuum L. var. grossum (L.) Sendtn.) Grown under Moderate Salt Stress. Plants 2021, 10, 314. [Google Scholar] [CrossRef]
- Zamljen, T.; Hudina, M.; Veberič, R.; Slatnar, A. Biostimulative effect of amino acids and green algae extract on capsaicinoid and other metabolite contents in fruits of Capsicum spp. Chem. Biol. Technol. Agric. 2021, 8, 63. [Google Scholar] [CrossRef]
- Medic, A.; Zamljen, T.; Slatnar, A.; Hudina, M.; Veberic, R. Is Juglone the Only Naphthoquinone in Juglans regia L. with Allelopathic Effects? Agriculture 2021, 11, 784. [Google Scholar] [CrossRef]
- Medic, A.; Zamljen, T.; Hudina, M.; Veberic, R. Identification and Quantification of Naphthoquinones and Other Phenolic Compounds in Leaves, Petioles, Bark, Roots, and Buds of Juglans regia L., Using HPLC-MS/MS. Horticulutrae 2021, 7, 326. [Google Scholar] [CrossRef]
- Zamljen, T.; Jakopič, J.; Hudina, M.; Veberič, R.; Slatnar, A. Influence of intra and inter species variation in chilies (Capsicum spp.) on metabolite composition of three fruit segments. Sci. Rep. 2021, 11, 4932. [Google Scholar] [CrossRef]
- Guclu, G.; Keser, D.; Kelebek, H.; Keskin, M.; Sekerli, Y.E.; Soysal, Y.; Selli, S. Impact of production and drying methods on the volatile and phenolic characteristics of fresh and powdered sweet red peppers. Food Chem. 2021, 338, 128129. [Google Scholar] [CrossRef]
- Medic, A.; Jakopic, J.; Solar, A.; Hudina, M.; Veberic, R. Walnut (J. regia) Agro-Residues as a Rich Source of Phenolic Compounds. Biology 2021, 10, 535. [Google Scholar] [CrossRef]
- Hossain, M.B.; Rai, D.K.; Brunton, N.P.; Martin-Diana, A.B.; Barry-Ryan, C. Characterization of phenolic composition in Lamiaceae spices by LC-ESI-MS/MS. J. Agric. Food Chem. 2010, 58, 10576–10581. [Google Scholar] [CrossRef]
- Mikulic-Petkovsek, M.; Schmitzer, V.; Jakopic, J.; Cunja, V.; Veberic, R.; Munda, A.; Stampar, F. Phenolic compounds as defence response of pepper fruits to Colletotrichum coccodes. Physiol. Mol. Plant Pathol. 2013, 84, 138–145. [Google Scholar] [CrossRef]
- Kang, J.; Price, W.E.; Ashton, J.; Tapsell, L.C.; Johnson, S. Identification and characterization of phenolic compounds in hydromethanolic extracts of sorghum wholegrains by LC-ESI-MSn. Food Chem. 2016, 211, 215–226. [Google Scholar] [CrossRef] [Green Version]
- Saied, A.S.; Keutgen, A.J.; Noga, G. The influence of NaCl salinity on growth, yield and fruit quality of strawberry cvs. ‘Elsanta’ and ‘Korona’. Sci. Hortic. 2005, 103, 289–303. [Google Scholar] [CrossRef]
- Lopez-Berenguer, C.; Alcaraz, C.; Carlos, M. Involvement of sugars in the response of pepper plants to salinity: Effect of calcium application. Asian J. Plant Sci. 2004, 3, 455–462. [Google Scholar] [CrossRef]
- Igamberdiev, A.U.; Eprintsev, A.T. Organic Acids: The Pools of Fixed Carbon Involved in Redox Regulation and Energy Balance in Higher Plants. Front. Plant Sci. 2016, 7, 1042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of Phenylpropanoid Pathway and the Role of Polyphenols in Plants under Abiotic Stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef] [Green Version]
- Taïbi, K.; Taïbi, F.; Abderrahim, L.A.; Ennajah, A.; Belkhodja, M.; Mulet, J.M. Effect of salt stress on growth, chlorophyll content, lipid peroxidation and antioxidant defence systems in Phaseolus vulgaris L. S. Afr. J. Bot. 2016, 105, 306–312. [Google Scholar] [CrossRef]
- Bistgani, Z.E.; Hashemi, M.; DaCosta, M.; Craker, L.; Maggi, F.; Morshedloo, M.R. Effect of salinity stress on the physiological characteristics, phenolic compounds and antioxidant activity of Thymus vulgaris L. and Thymus daenensis Celak. Ind. Crops Prod. 2019, 135, 311–320. [Google Scholar] [CrossRef]
- Golkar, P.; Taghizadeh, M. In vitro evaluation of phenolic and osmolite compounds, ionic content, and antioxidant activity in safflower (Carthamus tinctorius L.) under salinity stress. Plant Cell Tissue Organ Cult. 2018, 134, 357–368. [Google Scholar] [CrossRef]
- Yan, J.; Wang, B.; Jiang, Y.; Cheng, L.; Wu, T.J.P.; Physiology, C. GmFNSII-controlled soybean flavone metabolism responds to abiotic stresses and regulates plant salt tolerance. Plant Cell Physiol. 2014, 55, 74–86. [Google Scholar] [CrossRef] [Green Version]
- Ben-Abdallah, S.; Zorrig, W.; Amyot, L.; Renaud, J.; Hannoufa, A.; Lachâal, M.; Karray-Bouraoui, N.J.B. Potential production of polyphenols, carotenoids and glycoalkaloids in Solanum villosum Mill. under salt stress. Biologia 2019, 74, 309–324. [Google Scholar] [CrossRef]
- Scagel, C.F.; Lee, J.; Mitchell, J.N. Salinity from NaCl changes the nutrient and polyphenolic composition of basil leaves. Ind. Crops Prod. 2019, 127, 119–128. [Google Scholar] [CrossRef]
- Arrowsmith, S.; Egan, T.; Meekins, J.; Powers, D.; Metcalfe, M. Research Article: Effects of salt stress on capsaicin content, growth, and fluorescence in a Jalapeño cultivar of Capsicum annuum (Solanaceae). Bios 2012, 83, 1–7. [Google Scholar] [CrossRef]
- Julien, K.; Amoussa, A.M.; Mensah, A.; Françoise, A.; Eliane, K.; Lagnika, L.; Gandonou, C. Effect of salt stress on flowering, fructification and fruit nutrients concentration in a local cultivar of chili pepper (Capsicum frutescens L.). Int. J. Plant Physiol. Biochem. 2019, 11, 1–7. [Google Scholar] [CrossRef]
- Castro-Concha, L.A.; Baas-Espinola, F.M.; Ancona-Escalante, W.R.; Vázquez-Flota, F.A.; Miranda-Ham, M.L. Phenylalanine biosynthesis and its relationship to accumulation of capsaicinoids during Capsicum chinense fruit development. Biol. Plant. 2016, 60, 579–584. [Google Scholar] [CrossRef]
- Shams, M.; Yildirim, E. Variations in response of CaPAO and CaATG8c genes, hormone, photosynthesis and antioxidative system in pepper genotypes under salinity stress. Sci. Hortic. 2021, 282, 110041. [Google Scholar] [CrossRef]
- Aktas, H.; Abak, K.; Cakmak, I. Genotypic variation in the response of pepper to salinity. Sci. Hortic. 2006, 110, 260–266. [Google Scholar] [CrossRef] [Green Version]
- Kaur, H.; Sirhindi, G.; Bhardwaj, R.; Alyemeni, M.; Siddique, K.H.; Ahmad, P.J.S.R. 28-homobrassinolide regulates antioxidant enzyme activities and gene expression in response to salt-and temperature-induced oxidative stress in Brassica juncea. Sci. Rep. 2018, 8, 8735. [Google Scholar] [CrossRef] [PubMed]
- Ahanger, M.A.; Aziz, U.; Alsahli, A.A.; Alyemeni, M.N.; Ahmad, P.J.B. Influence of exogenous salicylic acid and nitric oxide on growth, photosynthesis, and ascorbate-glutathione cycle in salt stressed Vigna angularis. Agronomy 2020, 10, 42. [Google Scholar] [CrossRef] [Green Version]


| Cultivar/Fruit Part | Sugar | Treatment | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | 20 mM NaCl | 40 mM NaCl | Significance | ||||||
| ‘Somborka’ | Pericarp | Sucrose | 5.53 ± 0.26 | a | 5.22 ± 0.25 | a | 4.96 ± 0.15 | a | NS |
| Glucose | 7.13 ± 0.16 | a | 7.36 ± 0.63 | a | 6.02 ± 0.27 | a | NS | ||
| Fructose | 6.77 ± 0.12 | a | 6.77 ± 0.51 | a | 5.88 ± 0.26 | a | NS | ||
| Total sugars | 19.44 ± 0.55 | a | 19.35 ± 1.39 | a | 16.86 ± 0.69 | b | ** | ||
| Placenta | Sucrose | 3.74 ± 0.55 | a | 4.85 ± 0.10 | a | 5.00 ± 0.17 | a | NS | |
| Glucose | 5.07 ± 0.22 | a | 5.53 ± 0.12 | a | 2.82 ± 0.19 | b | *** | ||
| Fructose | 5.69 ± 0.24 | a | 5.48 ± 0.84 | a | 3.48 ± 0.22 | b | *** | ||
| Total sugars | 14.51 ± 1.01 | a | 15.87 ± 3.15 | a | 11.31 ± 0.54 | a | NS | ||
| ‘Novosadka’ | Pericarp | Sucrose | 4.50 ± 0.67 | a | 6.35 ± 0.14 | a | 5.56 ± 0.91 | a | NS |
| Glucose | 10.44 ± 0.84 | a | 9.30 ± 0.40 | a | 8.60 ± 081 | a | NS | ||
| Fructose | 9.14 ± 0.61 | a | 8.42 ± 0.41 | ab | 7.19 ± 0.34 | b | ** | ||
| Total sugars | 24.09 ± 2.12 | a | 24.08 ± 2.26 | a | 21.37 ± 2.07 | a | NS | ||
| Placenta | Sucrose | 9.12 ± 0.64 | a | 9.45 ± 0.21 | a | 8.36 ± 0.14 | a | NS | |
| Glucose | 5.23 ± 0.22 | a | 4.51 ± 0.32 | a | 3.39 ± 0.19 | b | *** | ||
| Fructose | 4.92 ± 0.17 | a | 4.44 ± 0.35 | a | 3.17 ± 0.08 | b | ** | ||
| Total sugars | 19.28 ± 1.04 | a | 18.42 ± 2.79 | a | 14.92 ± 0.42 | b | *** | ||
| ‘Berenyi F1’ | Pericarp | Sucrose | 3.93 ± 0.28 | a | 3.73 ± 0.24 | a | 3.60 ± 0.24 | a | NS |
| Glucose | 9.96 ± 0.21 | a | 7.57 ± 1.23 | b | 7.69 ± 1.53 | b | *** | ||
| Fructose | 9.08 ± 0.16 | a | 9.65 ± 1.61 | a | 7.22 ± 1.41 | a | NS | ||
| Total sugars | 22.97 ± 0.68 | a | 20.96 ± 3.09 | a | 18.52 ± 3.18 | a | NS | ||
| Placenta | Sucrose | 6.82 ± 0.86 | a | 6.92 ± 0.91 | a | 6.97 ± 0.26 | a | NS | |
| Glucose | 4.97 ± 0.48 | a | 3.87 ± 0.31 | b | 3.54 ± 0.21 | b | *** | ||
| Fructose | 6.05 ± 1.54 | a | 3.77 ± 0.24 | b | 3.56 ± 0.24 | b | *** | ||
| Total sugars | 17.85 ± 2.90 | a | 14.56 ± 1.47 | b | 14.07 ± 0.72 | b | *** | ||
| ‘Caro F1’ | Pericarp | Sucrose | 3.98 ± 0.69 | b | 6.07 ± 0.76 | b | 9.95 ± 0.77 | a | *** |
| Glucose | 9.42 ± 0.77 | a | 10.48 ± 0.11 | a | 9.36 ± 0.43 | a | NS | ||
| Fructose | 8.35 ± 0.71 | a | 9.44 ± 0.21 | a | 7.64 ± 0.49 | a | NS | ||
| Total sugars | 21.75 ± 2.18 | a | 25.99 ± 1.08 | a | 26.95 ± 1.70 | a | NS | ||
| Placenta | Sucrose | 1.13 ± 0.21 | a | 1.09 ± 0.09 | a | 1.21 ± 0.19 | a | NS | |
| Glucose | 5.32 ± 0.31 | a | 4.41 ± 0.40 | a | 4.62 ± 0.14 | a | NS | ||
| Fructose | 4.56 ± 0.33 | a | 3.76 ± 0.23 | b | 3.63 ± 0.04 | b | *** | ||
| Total sugars | 11.01 ± 1.64 | a | 9.26 ± 1.73 | a | 9.46 ± 1.28 | a | NS | ||
| Cultivar/Fruit Part | Organic Acid | Treatment | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | 20 mM NaCl | 40 mM NaCl | Significance | ||||||
| ‘Somborka’ | Pericarp | Oxalic a. | 0.22 ± 0.01 | a | 0.17 ± 0.02 | a | 0.18 ± 0.01 | a | NS |
| Citric a. | 0.52 ± 0.02 | b | 0.77 ± 0.29 | a | 1.28 ± 0.10 | a | *** | ||
| Malic a. | 1.75 ± 0.09 | a | 1.84 ± 0.30 | a | 1.55 ± 0.08 | a | NS | ||
| Qunic a. | 0.43 ± 0.05 | a | 0.84 ± 0.17 | a | 0.53 ± 0.10 | a | NS | ||
| Succinic a. | 10.31 ± 0.14 | a | 10.51 ± 0.48 | a | 10.43 ± 0.01 | a | NS | ||
| Fumaric a. | 0.021 ± 0.001 | a | 0.022 ± 0.002 | a | 0.017 ± 0.001 | a | NS | ||
| Ascorbic a. | 3.06 ± 0.24 | b | 4.08 ± 0.29 | a | 4.72 ± 0.31 | a | *** | ||
| Total acids | 16.33 ± 0.57 | b | 18.25 ± 0.15 | a | 18.72 ± 0.63 | a | *** | ||
| Placenta | Oxalic a. | 0.35 ± 0.02 | a | 0.06 ± 0.06 | b | 0.17 ± 0.07 | b | ** | |
| Citric a. | 1.11 ± 0.26 | a | 0.62 ± 0.07 | b | 0.68 ± 0.13 | b | *** | ||
| Malic a. | 2.12 ± 0.14 | a | 2.25 ± 0.21 | a | 1.61 ± 0.28 | a | NS | ||
| Qunic a. | 0.08 ± 0.13 | a | 0.37 ± 0.26 | a | 0.06 ± 0.26 | a | NS | ||
| Succinic a. | 14.79 ± 0.26 | a | 12.98 ± 0.78 | b | 10.68 ± 1.07 | b | *** | ||
| Fumaric a. | 0.023 ± 0.005 | a | 0.014 ± 0.003 | a | 0.012 ± 0.005 | a | NS | ||
| Ascorbic a. | 0.11 ± 0.01 | a | 0.06 ± 0.01 | b | 0.04 ± 0.01 | b | *** | ||
| Total acids | 18.39 ± 0.88 | a | 16.33 ± 1.45 | b | 13.28 ± 1.84 | b | *** | ||
| ‘Novosadka’ | Pericarp | Oxalic a. | 0.24 ± 0.02 | a | 0.20 ± 0.04 | a | 0.18 ± 0.01 | a | NS |
| Citric a. | 0.89 ± 0.23 | a | 0.76 ± 0.21 | a | 1.05 ± 0.17 | a | NS | ||
| Malic a. | 1.53 ± 0.29 | a | 1.68 ± 0.14 | a | 1.70 ± 0.12 | a | NS | ||
| Qunic a. | 2.02 ± 1.31 | a | 0.84 ± 0.15 | a | 0.86 ± 0.15 | a | NS | ||
| Succinic a. | 11.90 ± 0.48 | a | 10.92 ± 0.11 | b | 10.22 ± 0.20 | b | *** | ||
| Fumaric a. | 0.028 ± 0.005 | a | 0.025 ± 0.005 | a | 0.023 ± 0.001 | a | NS | ||
| Ascorbic a. | 6.34 ± 0.92 | a | 5.29 ± 0.80 | a | 5.61 ± 0.19 | a | NS | ||
| Total acids | 22.97 ± 0.32 | a | 19.75 ± 1.47 | b | 19.66 ± 0.87 | b | *** | ||
| Placenta | Oxalic a. | 0.40 ± 0.07 | a | 0.35 ± 0.01 | a | 0.23 ± 0.01 | a | NS | |
| Citric a. | 1.33 ± 0.08 | a | 1.20 ± 0.27 | a | 1.06 ± 0.09 | a | NS | ||
| Malic a. | 1.40 ± 0.17 | b | 1.71 ± 0.21 | a | 2.07 ± 0.14 | a | ** | ||
| Qunic a. | N/D | N/D | N/D | ||||||
| Succinic a. | 10.93 ± 0.59 | a | 11.48 ± 0.66 | a | 10.46 ± 0.35 | a | NS | ||
| Fumaric a. | 0.023 ± 0.005 | a | 0.030 ± 0.011 | a | 0.022 ± 0.003 | a | NS | ||
| Ascorbic a. | 0.41 ± 0.06 | a | 0.35 ± 0.04 | a | 0.54 ± 0.12 | a | NS | ||
| Total acids | 14.49 ± 1.19 | a | 14.92 ± 1.38 | a | 14.41 ± 0.89 | a | NS | ||
| ‘Berenyi F1’ | Pericarp | Oxalic a. | 0.16 ± 0.02 | a | 0.10 ± 0.01 | b | 0.09 ± 0.01 | b | *** |
| Citric a. | 1.62 ± 0.08 | a | 1.01 ± 0.04 | a | 1.56 ± 0.20 | a | NS | ||
| Malic a. | 1.04 ± 0.01 | a | 1.27 ± 0.16 | a | 1.34 ± 0.23 | a | NS | ||
| Qunic a. | 0.87 ± 0.04 | a | 0.73 ± 0.16 | a | 0.67 ± 0.17 | a | NS | ||
| Succinic a. | 11.79 ± 0.28 | a | 10.45 ± 0.27 | a | 9.98 ± 0.81 | a | NS | ||
| Fumaric a. | 0.046 ± 0.008 | a | 0.022 ± 0.001 | b | 0.014 ± 0.001 | b | *** | ||
| Ascorbic a. | 5.93 ± 0.55 | a | 5.13 ± 0.89 | a | 4.48 ± 1.43 | a | NS | ||
| Total acids | 21.47 ± 1.00 | a | 18.73 ± 1.55 | a | 18.17 ± 2.87 | a | NS | ||
| Placenta | Oxalic a. | 0.26 ± 0.01 | a | 0.22 ± 0.02 | a | 0.48 ± 0.22 | a | NS | |
| Citric a. | 1.58 ± 0.11 | a | 1.08 ± 0.10 | a | 1.53 ± 0.18 | a | NS | ||
| Malic a. | 1.80 ± 0.04 | a | 1.64 ± 0.14 | a | 1.58 ± 0.20 | a | NS | ||
| Qunic a. | 0.03 ± 0.00 | a | 0.15 ± 0.07 | a | 0.18 ± 0.10 | a | NS | ||
| Succinic a. | 11.62 ± 0.08 | a | 11.61 ± 0.32 | a | 12.15 ± 0.32 | a | NS | ||
| Fumaric a. | 0.022 ± 0.002 | a | 0.028 ± 0.005 | a | 0.022 ± 0.002 | a | NS | ||
| Ascorbic a. | 0.39 ± 0.11 | a | 0.29 ± 0.06 | a | 0.30 ± 0.04 | a | NS | ||
| Total acids | 15.73 ± 0.52 | a | 15.04 ± 0.74 | a | 16.26 ± 1.09 | a | NS | ||
| ‘Caro F1’ | Pericarp | Oxalic a. | 0.16 ± 0.02 | a | 0.09 ± 0.01 | b | 0.04 ± 0.01 | b | *** |
| Citric a. | 1.54 ± 0.23 | a | 1.52 ± 0.15 | a | 2.08 ± 0.10 | a | NS | ||
| Malic a. | 1.62 ± 0.52 | a | 1.31 ± 0.21 | a | 0.78 ± 0.12 | a | NS | ||
| Qunic a. | 1.76 ± 0.61 | a | 0.66 ± 0.17 | a | 0.99 ± 0.30 | a | NS | ||
| Succinic a. | 11.51 ± 0.17 | a | 9.71 ± 0.01 | b | 10.45 ± 0.04 | b | *** | ||
| Fumaric a. | 0.033 ± 0.007 | a | 0.013 ± 0.001 | b | 0.012 ± 0.003 | b | ** | ||
| Ascorbic a. | 5.66 ± 0.97 | a | 6.12 ± 0.79 | a | 4.40 ± 0.77 | a | NS | ||
| Total acids | 22.30 ± 2.55 | a | 19.44 ± 1.37 | a | 18.77 ± 1.36 | a | NS | ||
| Placenta | Oxalic a. | 0.34 ± 0.01 | a | 0.15 ± 0.08 | b | 0.15 ± 0.01 | b | *** | |
| Citric a. | 1.41 ± 0.03 | a | 1.24 ± 0.36 | a | 2.22 ± 0.48 | a | NS | ||
| Malic a. | 3.96 ± 0.09 | a | 2.17 ± 0.83 | b | 2.00 ± 0.23 | b | *** | ||
| Qunic a. | 0.04 ± 0.02 | a | 0.28 ± 0.08 | a | 0.95 ± 0.29 | a | NS | ||
| Succinic a. | 14.38 ± 0.37 | a | 10.34 ± 0.175 | b | 11.89 ± 0.53 | b | ** | ||
| Fumaric a. | 0.063 ± 0.01 | a | 0.025 ± 0.012 | b | 0.034 ± 0.004 | b | *** | ||
| Ascorbic a. | 0.54 ± 0.20 | a | 0.53 ± 0.14 | a | 0.42 ± 0.17 | a | NS | ||
| Total acids | 20.75 ± 0.73 | a | 14.77 ± 3.27 | b | 17.70 ± 1.74 | b | *** | ||
| Compound | Rt | [M-H]− | MS2 | MS3 | MS4 | Plant Tissue | ||
|---|---|---|---|---|---|---|---|---|
| (min) | (m/z) | (m/z) | (m/z) | (m/z) | Pericarp | Placenta | Seeds | |
| Coumaroylquinic acid derivative 1 | 7.39 | 391 | 216 (100), 173 (45), 111 (44), 191 (30), 129 (3) | × | × | |||
| Tricin | 9.27 | 329 | 314 (100), 311 (43), 285 (17) | × | ||||
| Caffeic acid hexoside 1 | 10.12 | 341 | 179 (100), 161 (26), 135 (4) | × | ||||
| Caffeic acid hexoside 2 | 11.95 | 341 | 179 (100), 135 (4) | × | × | |||
| p-Coumaroylquinic acid | 12.18 | 371 | 325 (100), 307 (80), 191 (61), 163 (46) | 163 (100) | × | |||
| Caffeic acid hexoside derivative | 13.79 | 387 | 179 (100), 341 (69) | × | × | |||
| Ferulic acid hexoside 1 | 14.07 | 355 | 193 (100), 217 (55), 175 (30) | 134 (100), 149 (52), 178 (22) | × | × | × | |
| Ferulic acid hexoside 2 | 14.45 | 551 | 389 (100), 193 (43), 341 (15) | 341 (100), 193 (43) | 149 (100), 178 (57), 134 (30) | × | × | |
| Chlorogenic acid | 15.35 | 353 | 293 (100) | 191 (100), 131 (55) | × | |||
| Apigenin pentosyl hexoside 1 | 15.60 | 696 | 469 (100), 353 (40), 243 (30) | 325 (100), 353 (93), 243 (30) | × | × | ||
| Caffeic acid hexoside 3 | 16.37 | 583 | 241 (100), 341 (40), 179 (1) | 179 (100), 161 (23) | × | |||
| Apigenin pentosyl hexoside 2 | 17.03 | 563 | 443 (100), 473 (86), 383 (20), 353 (19) | 353 (100), 383 (14) | × | |||
| Apigenin pentosyl hexoside 3 | 18.16 | 563 | 443 (100), 473 (53), 353 (13) | 353 (100), 383 (35) | 325 (100), 297 (30) | × | ||
| Luteolin-8-C-hexoside 1 | 18.79 | 567 | 357 (100) | 209 (100) | × | × | × | |
| Luteolin-8-C-hexoside 2 | 20.77 | 567 | 477 (100), 447 (96), 387 (88), 357 (79) | 357 (100), 387 (74) | 209 (100) | × | × | |
| Kaempferol dihexoside | 21.16 | 427 | 397 (100), 257 (48), 241 (4) | 257 (100), 241 (8) | 97 (100), 231 (41), 151 (20) | × | ||
| Quercetin rutinoside | 22.19 | 609 | 301 (100), 300 (11), 179 (2) | × | ||||
| Quercetin rhamnoside | 22.88 | 447 | 301 (100), 300 (26), 179 (1) | × | × | |||
| Kaempferol hexoside 1 | 22.99 | 349 | 241 (100), 151 (10) | × | ||||
| Luteolin-7-O- hexoside | 23.05 | 665 | 621 (100) | 489 (100), 285 (56) | × | |||
| Isorhamnetin rhamnoside | 24.76 | 461 | 314 (100), 315 (58) | 285 (100), 286 (67), 271 (66), 243 (20) | × | |||
| Kaempferol hexoside 2 | 25.43 | 393 | 241 (100), 349 (61), 257 (49) | × | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zamljen, T.; Medic, A.; Hudina, M.; Veberic, R.; Slatnar, A. Salt Stress Differentially Affects the Primary and Secondary Metabolism of Peppers (Capsicum annuum L.) According to the Genotype, Fruit Part, and Salinity Level. Plants 2022, 11, 853. https://doi.org/10.3390/plants11070853
Zamljen T, Medic A, Hudina M, Veberic R, Slatnar A. Salt Stress Differentially Affects the Primary and Secondary Metabolism of Peppers (Capsicum annuum L.) According to the Genotype, Fruit Part, and Salinity Level. Plants. 2022; 11(7):853. https://doi.org/10.3390/plants11070853
Chicago/Turabian StyleZamljen, Tilen, Aljaz Medic, Metka Hudina, Robert Veberic, and Ana Slatnar. 2022. "Salt Stress Differentially Affects the Primary and Secondary Metabolism of Peppers (Capsicum annuum L.) According to the Genotype, Fruit Part, and Salinity Level" Plants 11, no. 7: 853. https://doi.org/10.3390/plants11070853









