Hydrogen Sulfide Alleviates Cadmium Stress by Enhancing Photosynthetic Efficiency and Regulating Sugar Metabolism in Wheat Seedlings
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials and Growth Conditions
2.2. Plant Biomass and Chlorophyll Content
2.3. Lipid Peroxidation and ROS
2.4. Photosynthetic Gas Exchange Parameters
2.5. Soluble Sugar Content
2.6. Chlorophyll Fluorescence Parameters
2.7. Glucose, Fructose, and Sucrose Contents
2.8. Real-Time Quantitative PCR Assay
2.9. Statistical Analysis
3. Results
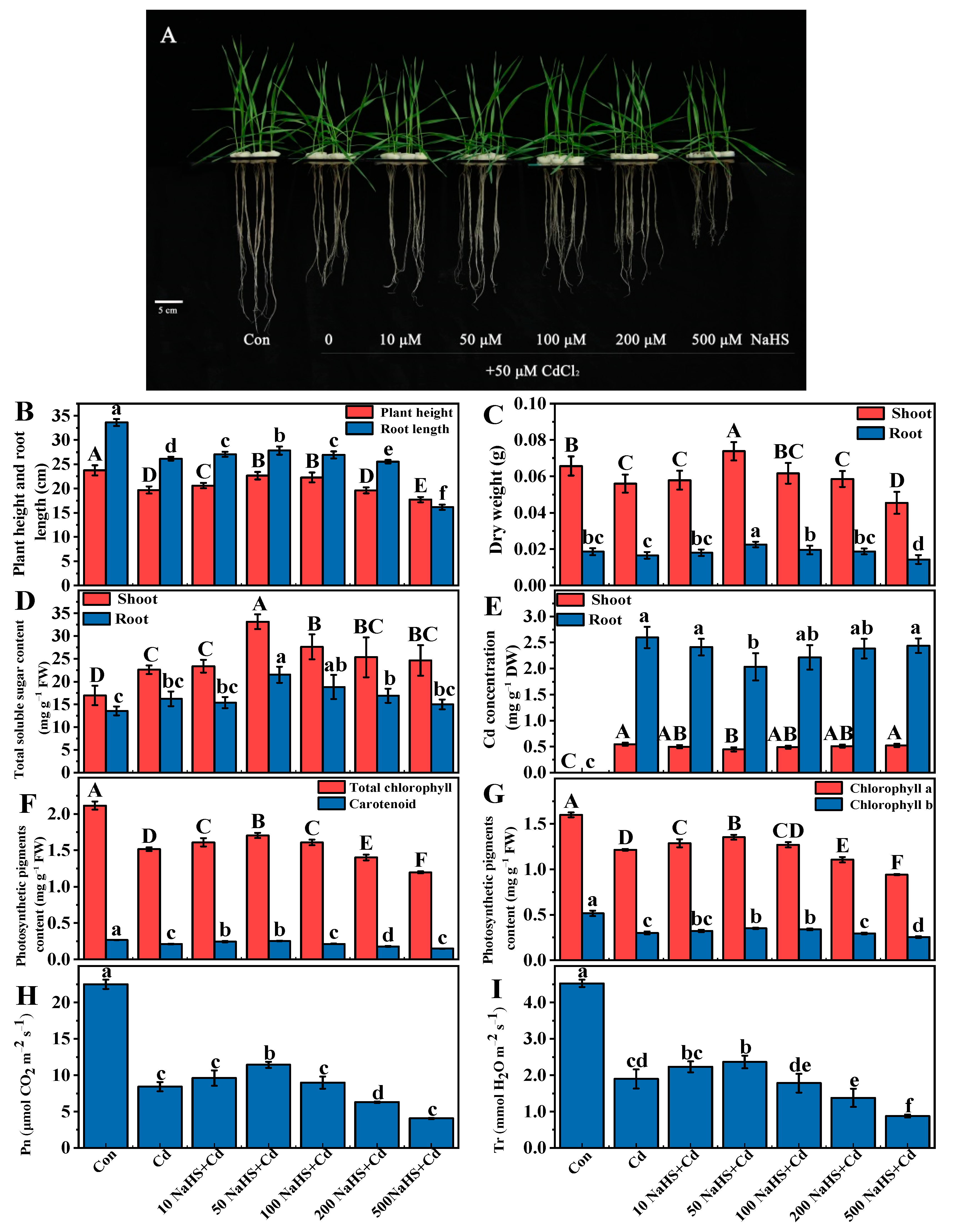
3.1. NaHS Pretreatment Alleviated Cd Toxicity in Wheat Seedlings
3.2. Effects of NaHS Pretreatment on Light-Response and Intercellular CO2-Response of Wheat Seedlings
3.3. Effect of NaHS Pretreatment on Chlorophyll Fluorescence Parameters of Wheat Seedlings
3.4. Effects of NaHS Pretreatment on the Soluble Sugar Content of Wheat Seedlings
3.5. Effects of NaHS Pretreatment on Genes Expression Related to Carbon Assimilation and Sucrose Metabolism
3.6. Effects of NaHS Pretreatment Reactive Oxygen Species of Wheat Seedlings
4. Discussions
4.1. NaHS Pretreatment Alleviated the Cd Stress to Wheat
4.2. NaHS Pretreatment Improved the Light Use Efficiency of Wheat Seedlings under Cd Stress
4.3. NaHS Pretreatment Enhanced Photosynthetic Efficiency of Wheat Seedlings under Cd Stress
4.4. NaHS Pretreatment Increased the Accumulation of Soluble Sugar in Wheat
4.5. NaHS Pretreatment Regulates the Related Gene Expression in Wheat Seedling under Cd Stress
 fructose + uridine diphosphate glucose (UDPG). SuSy is the only enzyme among the enzymes involved in sucrose metabolism that can catalyze the reversible reaction of sucrose metabolism. Its activity affects both the synthesis and catabolism of sucrose [68]. In the process of sucrose synthesis, the function of SPS is to catalyze the conversion of fructose-6-phosphate and UDPG into sucrose. The activity of SPS in plants directly reflects the capacity for sucrose synthesis in plants [69]. NaHS pretreatment simultaneously up-regulated TaSPS and down-regulated TaSuSy expression compared to Cd treatment alone, which significantly increased sucrose synthesis. Invertase is involved in the decomposition of sucrose into glucose and fructose. This mainly occurs in the form of SAInv and A/NInv according to the optimal pH [70]. When plant cells respond to hexoses, these enzymes regulate the intracellular sucrose/hexose ratio [71]. In the present study, the genes (TaSAInv, TaA/NInv, and TaSPS) that catalyzed sucrose decomposition and synthesis were significantly up-regulated under the stress of Cd. The up-regulation of these genes was consistent with the increase in glucose, fructose, and sucrose content under the stress of Cd. Moreover, the expression of TaSPS was further up-regulated by NaHS pretreatment. The up-regulation of TaSPS may be why NaHS pretreatment further increased the soluble sugar in wheat leaves under the stress of Cd [72]. In addition, compared with the control, the expression of TaSuSy showed no significant change under Cd stress. However, NaHS pretreatment significantly down-regulated its expression, which is consistent with the variation trend of two invertase genes (TaSAInv/TaA/NInv) (Figure 6). Therefore, the level of Cd-induced hexoses accumulation was decreased. We can get a result from the above that the increase in fructose and glucose content in wheat seedling leaves under Cd stress was correlated with the up-regulation expression of TaSAInv and TaA/NInv [70]. NaHS pretreatment resulted in a significant reduction of fructose and glucose contents. An increase in sucrose content in wheat seedling leaves under Cd stress compared with Cd treatment alone, which was associated with down-regulating the expression of TaSAInv, TaA/NInv, TaSuSy, and up-regulating the expression of TaSPS [73]. Carbon assimilation products are products of photosynthesis and substrates of plant respiration, which provide the carbon skeleton for plant growth and development and enhance plant resilience [74]. Cd-induced hexose accumulation is due to a reduction in the activity of key enzymes in the glycolytic pathway and a decrease in the translocation of sucrose to the depot cells, resulting in a decrease in the efficiency of hexose being oxidized and utilized. Proteomic screening was used to identify differentially expressed proteins in the leaves of wheat seedlings, both with and without NaHS pretreatment under conditions of drought stress, and found that H2S regulates many biochemical pathways, including energy and carbon metabolism, signal transduction, and antioxidant capacity. The expression of key genes of some metabolic pathways was consistent with the analysis of proteomic results [75]. Therefore, H2S can enhance the tolerance of wheat to abiotic stresses by increasing energy metabolism. Combining the results and discussion of this study, we propose the use of the working model of NaHS pretreatment to enhance Cd resistance in wheat under Cd stress (Figure 8).
fructose + uridine diphosphate glucose (UDPG). SuSy is the only enzyme among the enzymes involved in sucrose metabolism that can catalyze the reversible reaction of sucrose metabolism. Its activity affects both the synthesis and catabolism of sucrose [68]. In the process of sucrose synthesis, the function of SPS is to catalyze the conversion of fructose-6-phosphate and UDPG into sucrose. The activity of SPS in plants directly reflects the capacity for sucrose synthesis in plants [69]. NaHS pretreatment simultaneously up-regulated TaSPS and down-regulated TaSuSy expression compared to Cd treatment alone, which significantly increased sucrose synthesis. Invertase is involved in the decomposition of sucrose into glucose and fructose. This mainly occurs in the form of SAInv and A/NInv according to the optimal pH [70]. When plant cells respond to hexoses, these enzymes regulate the intracellular sucrose/hexose ratio [71]. In the present study, the genes (TaSAInv, TaA/NInv, and TaSPS) that catalyzed sucrose decomposition and synthesis were significantly up-regulated under the stress of Cd. The up-regulation of these genes was consistent with the increase in glucose, fructose, and sucrose content under the stress of Cd. Moreover, the expression of TaSPS was further up-regulated by NaHS pretreatment. The up-regulation of TaSPS may be why NaHS pretreatment further increased the soluble sugar in wheat leaves under the stress of Cd [72]. In addition, compared with the control, the expression of TaSuSy showed no significant change under Cd stress. However, NaHS pretreatment significantly down-regulated its expression, which is consistent with the variation trend of two invertase genes (TaSAInv/TaA/NInv) (Figure 6). Therefore, the level of Cd-induced hexoses accumulation was decreased. We can get a result from the above that the increase in fructose and glucose content in wheat seedling leaves under Cd stress was correlated with the up-regulation expression of TaSAInv and TaA/NInv [70]. NaHS pretreatment resulted in a significant reduction of fructose and glucose contents. An increase in sucrose content in wheat seedling leaves under Cd stress compared with Cd treatment alone, which was associated with down-regulating the expression of TaSAInv, TaA/NInv, TaSuSy, and up-regulating the expression of TaSPS [73]. Carbon assimilation products are products of photosynthesis and substrates of plant respiration, which provide the carbon skeleton for plant growth and development and enhance plant resilience [74]. Cd-induced hexose accumulation is due to a reduction in the activity of key enzymes in the glycolytic pathway and a decrease in the translocation of sucrose to the depot cells, resulting in a decrease in the efficiency of hexose being oxidized and utilized. Proteomic screening was used to identify differentially expressed proteins in the leaves of wheat seedlings, both with and without NaHS pretreatment under conditions of drought stress, and found that H2S regulates many biochemical pathways, including energy and carbon metabolism, signal transduction, and antioxidant capacity. The expression of key genes of some metabolic pathways was consistent with the analysis of proteomic results [75]. Therefore, H2S can enhance the tolerance of wheat to abiotic stresses by increasing energy metabolism. Combining the results and discussion of this study, we propose the use of the working model of NaHS pretreatment to enhance Cd resistance in wheat under Cd stress (Figure 8).5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zulfiqar, U.; Jiang, W.T.; Wang, X.K.; Hussain, S.; Ahmad, M.; Maqsood, M.F.; Ali, N.; Ishfaq, M.; Kaleem, M.; Haider, F.U.; et al. Cadmium Phytotoxicity, Tolerance, and Advanced Remediation Approaches in Agricultural Soils; A Comprehensive Review. Front. Plant Sci. 2022, 13, 773815. [Google Scholar] [CrossRef] [PubMed]
- Haider, F.U.; Cai, L.Q.; Coulter, J.A.; Cheema, S.A.; Wu, J.; Zhang, R.Z.; Ma, W.J.; Farooq, M. Cadmium toxicity in plants: Impacts and remediation strategies. Ecotoxicol. Environ. Saf. 2021, 211, 111887. [Google Scholar] [CrossRef] [PubMed]
- Clemens, S.; Aarts, M.G.M.; Thomine, S.; Verbruggen, N. Plant science: The key to preventing slow cadmium poisoning. Trends Plant Sci. 2013, 18, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Kubier, A.; Wilkin, R.T.; Pichler, T. Cadmium in soils and groundwater: A review. Appl. Geochem. 2019, 108, 104388. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, C.; Liu, Y.; Yu, K.; Zhou, Y. GmHMA3 sequesters Cd to the root endoplasmic reticulum to limit translocation to the stems in soybean. Plant Sci. 2018, 270, 23–29. [Google Scholar] [CrossRef]
- Xie, Y.; Li, X.; Liu, X.; Amombo, E.; Chen, L.; Fu, J. Application of Aspergillus aculeatus to rice roots reduces Cd concentration in grain. Plant Soil 2018, 422, 409–422. [Google Scholar] [CrossRef]
- Zhou, J.; Hao, M.; Liu, Y.; Huang, G.; Fu, Q.; Zhu, J.; Hu, H. Effects of exogenous sulfur on growth and Cd uptake in Chinese cabbage (Brassica campestris spp. pekinensis) in Cd-contaminated soil. Environ. Sci. Pollut. Res. 2018, 25, 15823–15829. [Google Scholar] [CrossRef]
- Huang, H.; Li, M.; Rizwan, M.; Dai, Z.; Yuan, Y.; Hossain, M.M.; Cao, M.; Xiong, S.; Tu, S. Synergistic effect of silicon and selenium on the alleviation of cadmium toxicity in rice plants. J. Hazard. Mater. 2021, 401, 123393. [Google Scholar] [CrossRef]
- Lei, G.J.; Sun, L.; Sun, Y.; Zhu, X.F.; Li, G.X.; Zheng, S.J. Jasmonic acid alleviates cadmium toxicity in Arabidopsis via suppression of cadmium uptake and translocation. J. Integr. Plant Biol. 2020, 62, 218–227. [Google Scholar] [CrossRef]
- Singh, S.; Prasad, S.M.; Sharma, S.; Dubey, N.K.; Ramawat, N.; Prasad, R.; Singh, V.P.; Tripathi, D.K.; Chauhan, D.K. Silicon and nitric oxide-mediated mechanisms of cadmium toxicity alleviation in wheat seedlings. Physiol. Plant. 2022, 174, e13065. [Google Scholar] [CrossRef]
- Wang, C.L.; Deng, Y.Z.; Liu, Z.S.; Liao, W.B. Hydrogen Sulfide in Plants: Crosstalk with Other Signal Molecules in Response to Abiotic Stresses. Int. J. Mol. Sci. 2021, 22, 12068. [Google Scholar] [CrossRef]
- Cui, Q.; Liu, D.; Chen, H.; Qiu, T.; Zhao, S.; Duan, C.; Cui, Y.; Zhu, X.; Chao, H.; Wang, Y.; et al. Synergistic interplay between Azospirillum brasilense and exogenous signaling molecule H2S promotes Cd stress resistance and growth in pak choi (Brassica chinensis L.). J. Hazard. Mater. 2023, 444, 130425. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.; Liu, Z.; Long, Y.; Liang, Y.; Jin, Z.; Zhang, L.; Liu, D.; Li, H.; Zhai, J.; Pei, Y. The Ca2+/calmodulin2-binding transcription factor TGA3 elevates LCD expression and H2S production to bolster Cr6+ tolerance in Arabidopsis. Plant J. 2017, 91, 1038–1050. [Google Scholar] [CrossRef] [Green Version]
- Ali, B.; Gill, R.A.; Yang, S.; Gill, M.B.; Ali, S.; Rafiq, M.T.; Zhou, W. Hydrogen sulfide alleviates cadmium-induced morpho-physiological and ultrastructural changes in Brassica napus. Ecotoxicol. Environ. Saf. 2014, 110, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Bharwana, S.A.; Ali, S.; Farooq, M.A.; Ali, B.; Iqbal, N.; Abbas, F.; Ahmad, M.S.A. Hydrogen sulfide ameliorates lead-induced morphological, photosynthetic, oxidative damages and biochemical changes in cotton. Environ. Sci. Pollut. Res. 2014, 21, 717–731. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wang, R.; Zhang, X.; Yu, Y.; Zhao, R.; Li, Z.; Chen, S. Hydrogen sulfide alleviates cadmium toxicity through regulations of cadmium transport across the plasma and vacuolar membranes in Populus euphratica cells. Plant Physiol. Biochem. 2013, 65, 67–74. [Google Scholar] [CrossRef]
- Gururani, M.A.; Venkatesh, J.; Lam-Son Phan, T. Regulation of Photosynthesis during Abiotic Stress-Induced Photoinhibition. Mol. Plant 2015, 8, 1304–1320. [Google Scholar] [CrossRef] [Green Version]
- Alzahrani, Y.; Kusvuran, A.; Alharby, H.F.; Kusvuran, S.; Rady, M.M. The defensive role of silicon in wheat against stress conditions induced by drought, salinity or cadmium. Ecotoxicol. Environ. Saf. 2018, 154, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, M.; Turkyilmaz Unal, B.; Garcia-Caparros, P.; Khursheed, A.; Gul, A.; Hasanuzzaman, M. Osmoregulation and its actions during the drought stress in plants. Physiol. Plant. 2021, 172, 1321–1335. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-G.; Ding, X.-J.; Du, P.-F. Hydrogen sulfide donor sodium hydrosulfide-improved heat tolerance in maize and involvement of proline. J. Plant Physiol. 2013, 170, 741–747. [Google Scholar] [CrossRef]
- Hilal, B.; Khan, T.A.; Fariduddin, Q. Recent advances and mechanistic interactions of hydrogen sulfide with plant growth regulators in relation to abiotic stress tolerance in plants. Plant Physiol. Biochem. 2023, 196, 1065–1083. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Cheng, X.; Pan, N.; Huang, W.; Shi, L.; Lu, W. Foliar Spraying of Mannose Alleviates Cadmium Stress by Changing the Subcellular Distribution and Chemical Forms of Cadmium in Wheat Root. Agronomy 2022, 12, 2273. [Google Scholar] [CrossRef]
- Zhang, D.Z.; Liu, J.J.; Zhang, Y.B.; Wang, H.R.; Wei, S.W.; Zhang, X.; Zhang, D.; Ma, H.S.; Ding, Q.; Ma, L.J. Morphophysiological, proteomic and metabolomic analyses reveal cadmium tolerance mechanism in common wheat (Triticum aestivum L.). J. Hazard. Mater. 2023, 445, 130499. [Google Scholar] [CrossRef] [PubMed]
- Duan, B.; Ma, Y.; Jiang, M.; Yang, F.; Ni, L.; Lu, W. Improvement of photosynthesis in rice (Oryza sativa L.) as a result of an increase in stomatal aperture and density by exogenous hydrogen sulfide treatment. Plant Growth Regul. 2015, 75, 33–44. [Google Scholar] [CrossRef]
- Lin, Z.; Wang, Y.L.; Cheng, L.S.; Zhou, L.L.; Xu, Q.T.; Liu, D.C.; Deng, X.Y.; Mei, F.Z.; Zhou, Z.Q. Mutual regulation of ROS accumulation and cell autophagy in wheat roots under hypoxia stress. Plant Physiol. Biochem. 2021, 158, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tan, Z.Q.; Hu, L.Y.; Wang, S.H.; Luo, J.P.; Jones, R.L. Hydrogen Sulfide Alleviates Aluminum Toxicity in Germinating Wheat Seedlings. J. Integr. Plant Biol. 2010, 52, 556–567. [Google Scholar] [CrossRef]
- Ye, Z.-P.; Suggett, D.J.; Robakowski, P.; Kang, H.-J. A mechanistic model for the photosynthesis-light response based on the photosynthetic electron transport of photosystem II in C3 and C4 species. New Phytol. 2013, 199, 110–120. [Google Scholar] [CrossRef]
- Kaur, N.; Kaur, H.; Mavi, G.S. Assessment of nutritional and quality traits in biofortified bread wheat genotypes. Food Chem. 2020, 302, 125342. [Google Scholar] [CrossRef]
- Ali, S.; Xu, Y.; Jia, Q.; Ma, X.; Ahmad, I.; Adnan, M.; Gerard, R.; Ren, X.; Zhang, P.; Cai, T.; et al. Interactive effects of plastic film mulching with supplemental irrigation on winter wheat photosynthesis, chlorophyll fluorescence and yield under simulated precipitation conditions. Agric. Water Manag. 2018, 207, 1–14. [Google Scholar] [CrossRef]
- Yaashikaa, P.R.; Kumar, P.S.; Jeevanantham, S.; Saravanan, R. A review on bioremediation approach for heavy metal detoxification and accumulation in plants. Environ. Pollut. 2022, 301, 119035. [Google Scholar] [CrossRef]
- Goncharuk, E.A.; Zagoskina, N.V. Heavy Metals, Their Phytotoxicity, and the Role of Phenolic Antioxidants in Plant Stress Responses with Focus on Cadmium: Review. Molecules 2023, 28, 3921. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Ali, S.; Rizwan, M.; Rehman, M.Z.u.; Javed, M.R.; Imran, M.; Chatha, S.A.S.; Nazir, R. Zinc oxide nanoparticles alter the wheat physiological response and reduce the cadmium uptake by plants. Environ. Pollut. 2018, 242, 1518–1526. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.; Irfan, M.; Sattar, A.; Hussain, S.; Ullah, S.; Abbas, T.; Ur-Rehman, H.; Nawaz, F.; Al-Hashimi, A.; Elshikh, M.S.; et al. Alleviation of Cadmium Stress in Wheat through the Combined Application of Boron and Biochar via Regulating Morpho-Physiological and Antioxidant Defense Mechanisms. Agronomy 2022, 12, 434. [Google Scholar] [CrossRef]
- Yang, M.; Qin, B.-p.; Ma, X.-l.; Wang, P.; Li, M.-l.; Chen, L.-l.; Chen, L.-t.; Sun, A.-q.; Wang, Z.-l.; Yin, Y.-p. Foliar application of sodium hydrosulfide (NaHS), a hydrogen sulfide (H2S) donor, can protect seedlings against heat stress in wheat (Triticum aestivum L.). J. Integr. Agric. 2016, 15, 2745–2758. [Google Scholar] [CrossRef] [Green Version]
- Yan, L.-J.; Allen, D.C. Cadmium-Induced Kidney Injury: Oxidative Damage as a Unifying Mechanism. Biomolecules 2021, 11, 1575. [Google Scholar] [CrossRef] [PubMed]
- Sabella, E.; Luvisi, A.; Genga, A.; De Bellis, L.; Aprile, A. Molecular Responses to Cadmium Exposure in Two Contrasting Durum Wheat Genotypes. Int. J. Mol. Sci. 2021, 22, 7343. [Google Scholar] [CrossRef]
- Li, G.-Z.; Wang, Y.-Y.; Liu, J.; Liu, H.-T.; Liu, H.-P.; Kang, G.-Z. Exogenous melatonin mitigates cadmium toxicity through ascorbic acid and glutathione pathway in wheat. Ecotoxicol. Environ. Saf. 2022, 237, 113533. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, P.; Krasylenko, Y.; Zeiner, A.; Samaj, J.; Takac, T. Signaling Toward Reactive Oxygen Species-Scavenging Enzymes in Plants. Front. Plant Sci. 2021, 11, 618835. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Liu, H.; Nie, Z.; Gao, W.; Li, C.; Lin, Y.; Zhao, P. AsA-GSH Cycle and Antioxidant Enzymes Play Important Roles in Cd Tolerance of Wheat. Bull. Environ. Contam. Toxicol. 2018, 101, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Raza, A.; Tabassum, J.; Mubarik, M.S.; Anwar, S.; Zahra, N.; Sharif, Y.; Hafeez, M.B.; Zhang, C.; Corpas, F.J.; Chen, H. Hydrogen sulfide: An emerging component against abiotic stress in plants. Plant Biol. 2022, 24, 540–558. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Qiao, Z.; Zhang, L.; Li, H.; Pei, Y. Hydrogen sulfide and proline cooperate to alleviate cadmium stress in foxtail millet seedlings. Plant Physiol. Biochem. 2016, 109, 293–299. [Google Scholar] [CrossRef]
- Wagle, P.; Zhang, Y.; Jin, C.; Xiao, X. Comparison of solar-induced chlorophyll fluorescence, light-use efficiency, and process-based GPP models in maize. Ecol. Appl. 2016, 26, 1211–1222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinnamaneni, S.R.; Anapalli, S.S.; Reddy, K.N. Photosynthetic Response of Soybean and Cotton to Different Irrigation Regimes and Planting Geometries. Front. Plant Sci. 2022, 13, 894706. [Google Scholar] [CrossRef]
- da Silva Cunha, L.F.; de Oliveira, V.P.; do Nascimento, A.W.S.; da Silva, B.R.S.; Batista, B.L.; Alsahli, A.A.; Lobato, A.K.d.S. Leaf application of 24-epibrassinolide mitigates cadmium toxicity in youngEucalyptus urophyllaplants by modulating leaf anatomy and gas exchange. Physiol. Plant. 2021, 173, 67–87. [Google Scholar] [CrossRef]
- Yadav, M.R.; Choudhary, M.; Singh, J.; Lal, M.K.; Jha, P.K.; Udawat, P.; Gupta, N.K.; Rajput, V.D.; Garg, N.K.; Maheshwari, C.; et al. Impacts, Tolerance, Adaptation, and Mitigation of Heat Stress on Wheat under Changing Climates. Int. J. Mol. Sci. 2022, 23, 2838. [Google Scholar] [CrossRef]
- Chu, J.; Zhu, F.; Chen, X.; Liang, H.; Wang, R.; Wang, X.; Huang, X. Effects of cadmium on photosynthesis of Schima superba young plant detected by chlorophyll fluorescence. Environ. Sci. Pollut. Res. 2018, 25, 10679–10687. [Google Scholar] [CrossRef]
- Tang, G.; Tang, J.; Huang, J.; Lu, M.; Zhang, X.; Yang, Y.; Sun, S.; Chen, Y.; Dou, X. Passivating Agents Relieved Cu and Cd Pollution on Maize Growth. J. Soil Sci. Plant Nutr. 2023, 23, 2030–2038. [Google Scholar] [CrossRef]
- Shi, G.; Liu, C.; Cai, Q.; Liu, Q.; Hou, C. Cadmium Accumulation and Tolerance of Two Safflower Cultivars in Relation to Photosynthesis and Antioxidantive Enzymes. Bull. Environ. Contam. Toxicol. 2010, 85, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Teng, Z.; Zheng, W.; Jiang, S.; Hong, S.-B.; Zhu, Z.; Zang, Y. Role of melatonin in promoting plant growth by regulating carbon assimilation and ATP accumulation. Plant Sci. 2022, 319, 111276. [Google Scholar] [CrossRef] [PubMed]
- Ozfidan-Konakci, C.; Arikan, B.; Elbasan, F.; Cavusoglu, H.; Yildiztugay, E.; Alp, F.N. The biphasic responses of nanomaterial fullerene on stomatal movement, water status, chlorophyll a fluorescence transient, radical scavenging system and aquaporin-related gene expression in Zea mays under cobalt stress. Sci. Total Environ. 2022, 826, 154213. [Google Scholar] [CrossRef]
- Piao, L.; Wang, Y.; Liu, X.; Sun, G.; Zhang, S.; Yan, J.; Chen, Y.; Meng, Y.; Li, M.; Gu, W. Exogenous Hemin alleviated cadmium stress in maize (Zea mays L.) by enhancing leaf photosynthesis, AsA-GSH cycle and polyamine metabolism. Front. Plant Sci. 2022, 13, 993675. [Google Scholar] [CrossRef] [PubMed]
- Mubarakshina, M.M.; Ivanov, B.N. The production and scavenging of reactive oxygen species in the plastoquinone pool of chloroplast thylakoid membranes. Physiol. Plant. 2010, 140, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Nomani, L.; Zehra, A.; Choudhary, S.; Wani, K.I.; Naeem, M.; Siddiqui, M.H.; Khan, M.M.A.; Aftab, T. Exogenous hydrogen sulphide alleviates copper stress impacts in Artemisia annua L.: Growth, antioxidant metabolism, glandular trichome development and artemisinin biosynthesis. Plant Biol. 2022, 24, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Chaput, V.; Martin, A.; Lejay, L. Redox metabolism: The hidden player in carbon and nitrogen signaling? J. Exp. Bot. 2020, 71, 3816–3826. [Google Scholar] [CrossRef]
- Xu, Y.; Fu, X. Reprogramming of Plant Central Metabolism in Response to Abiotic Stresses: A Metabolomics View. Int. J. Mol. Sci. 2022, 23, 5716. [Google Scholar] [CrossRef]
- Shi, H.-T.; Li, R.-J.; Cai, W.; Liu, W.; Wang, C.-L.; Lu, Y.-T. Increasing Nitric Oxide Content in Arabidopsis thaliana by Expressing Rat Neuronal Nitric Oxide Synthase Resulted in Enhanced Stress Tolerance. Plant Cell Physiol. 2012, 53, 344–357. [Google Scholar] [CrossRef]
- Anjum, S.A.; Ashraf, U.; Khan, I.; Tanveer, M.; Ali, M.; Hussain, I.; Wang, L.C. Chromium and Aluminum Phytotoxicity in Maize: Morpho-Physiological Responses and Metal Uptake. Clean-Soil Air Water 2016, 44, 1075–1084. [Google Scholar] [CrossRef]
- Zhou, J.; Cheng, K.; Huang, G.; Chen, G.; Zhou, S.; Huang, Y.; Zhang, J.; Duan, H.; Fan, H. Effects of exogenous 3-indoleacetic acid and cadmium stress on the physiological and biochemical characteristics of Cinnamomum camphora. Ecotoxicol. Environ. Saf. 2020, 191, 109998. [Google Scholar] [CrossRef]
- Keunen, E.; Peshev, D.; Vangronsveld, J.; Van den Ende, W.; Cuypers, A. Plant sugars are crucial players in the oxidative challenge during abiotic stress: Extending the traditional concept. Plant Cell Environ. 2013, 36, 1242–1255. [Google Scholar] [CrossRef]
- Sami, F.; Yusuf, M.; Faizan, M.; Faraz, A.; Hayat, S. Role of sugars under abiotic stress. Plant Physiol. Biochem. 2016, 109, 54–61. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, X.; Chen, H.; Xu, X. Physiology and proteomics reveal Fulvic acid mitigates Cadmium adverse effects on growth and photosynthetic properties of lettuce. Plant Sci. 2022, 323, 111418. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Liu, T.; Zhao, Y.; He, Y.; Yang, M. Elevated atmospheric CO2 affected photosynthetic products in wheat seedlings and biological activity in rhizosphere soil under cadmium stress. Environ. Sci. Pollut. Res. 2016, 23, 514–526. [Google Scholar] [CrossRef] [PubMed]
- Sellami, S.; Le Hir, R.; Thorpe, M.R.; Vilaine, F.; Wolff, N.; Brini, F.; Dinant, S. Salinity Effects on Sugar Homeostasis and Vascular Anatomy in the Stem of the Arabidopsis Thaliana Inflorescence. Int. J. Mol. Sci. 2019, 20, 3167. [Google Scholar] [CrossRef] [Green Version]
- Jia, X.; Zhao, Y.; Wang, W.; He, Y. Elevated temperature altered photosynthetic products in wheat seedlings and organic compounds and biological activity in rhizopshere soil under cadmium stress. Sci. Rep. 2015, 5, 14426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonzalez-Fernandez, C.; Ballesteros, M. Linking microalgae and cyanobacteria culture conditions and key-enzymes for carbohydrate accumulation. Biotechnol. Adv. 2012, 30, 1655–1661. [Google Scholar] [CrossRef] [PubMed]
- Jian, M.; Zhang, D.; Wang, X.; Wei, S.; Zhao, Y.; Ding, Q.; Han, Y.; Ma, L. Differential expression pattern of the proteome in response to cadmium stress based on proteomics analysis of wheat roots. BMC Genom. 2020, 21, 343. [Google Scholar] [CrossRef]
- Li, Z.-Q.; Zhang, Y.; Li, H.; Su, T.-T.; Liu, C.-G.; Han, Z.-C.; Wang, A.-Y.; Zhu, J.-B. Genome-Wide Characterization and Expression Analysis Provide Basis to the Biological Function of Cotton FBA Genes. Front. Plant Sci. 2021, 12, 696698. [Google Scholar] [CrossRef]
- Stein, O.; Granot, D. An Overview of Sucrose Synthases in Plants. Front. Plant Sci. 2019, 10, 95. [Google Scholar] [CrossRef] [Green Version]
- Vargas, W.A.; Salerno, G.L. The Cinderella story of sucrose hydrolysis: Alkaline/neutral invertases, from cyanobacteria to unforeseen roles in plant cytosol and organelles. Plant Sci. 2010, 178, 1–8. [Google Scholar] [CrossRef]
- Zhao, M.; Meng, Y.; Wang, Y.; Sun, G.; Liu, X.; Li, J.; Wei, S.; Gu, W. Exogenous Hemin alleviates cadmium stress in maize by enhancing sucrose and nitrogen metabolism and regulating endogenous hormones. Int. J. Phytorem. 2023, 25, 368–380. [Google Scholar] [CrossRef]
- Aslani, L.; Gholami, M.; Mobli, M.; Ehsanzadeh, P.; Bertin, N. Decreased sink/source ratio enhances hexose transport in the fruits of greenhouse tomatoes: Integration of gene expression and biochemical analyses. Physiol. Plant. 2020, 170, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Guo, H.; Wang, S.; Li, J.; Wang, Y.; Xing, B. Carbon dots alleviate the toxicity of cadmium ions (Cd2+) toward wheat seedlings. Environ. Sci.-Nano 2019, 6, 1493–1506. [Google Scholar] [CrossRef]
- Ul-Allah, S.; Rehman, A.; Hussain, M.; Farooq, M. Fiber yield and quality in cotton under drought: Effects and management. Agric. Water Manag. 2021, 255, 106994. [Google Scholar] [CrossRef]
- Wang, L.; Yao, Y.; Wang, J.; Cui, J.; Wang, X.; Li, X.; Li, Y.; Ma, L. Metabolomics analysis reveal the molecular responses of high CO2 concentration improve resistance to Pb stress of Oryza sativa L. seedlings. Ecotoxicol. Environ. Saf. 2023, 251, 114515. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Han, Q.; Ma, D.; Hou, J.; Huang, X.; Wang, C.; Xie, Y.; Kang, G.; Guo, T. Characterizing Physiological and Proteomic Analysis of the Action of H2S to Mitigate Drought Stress in Young Seedling of Wheat. Plant Mol. Biol. Rep. 2018, 36, 45–57. [Google Scholar] [CrossRef]







| Gene Name | Forward Primer | Reverse Primer |
|---|---|---|
| TaRBCL | 5′-CGGTAGCTTCAGGTGGTATTC-3′ | 5′-GGATGTCCTAAAGTTCCTCCAC-3′ |
| TaRBCS | 5′-CAGCAACGGTGGAAGGAT-3′ | 5′-GGTGGCAAGTAGGACAGG-3′ |
| TaCpFBA | 5′-GCAGAAGGTGTGGGCGGAG-3′ | 5′-AGCGTCTGCCTCCAACCTC-3′ |
| TaPRK | 5′-TGTTGAGAGCCACCTAAGC-3′ | 5′-GAAGAGACCTGTTCCATTGTTG-3′ |
| TaSuSy | 5′-CCGACAAGGAGAAGTATG-3′ | 5′-CGAGTTCACTAACATTCAC-3′ |
| TaSPS | 5′-ATCGTCACGCTCGCTCAA-3′ | 5′-AGTCATCTTCCTGCCAAAATTACA-3′ |
| TaSAInv | 5′-AACGTCACAAGGCTCGTCGTCGT-3′ | 5′-ATGTAGGCCTGATTGTAGGAGGAGT-3′ |
| TaA/N-Inv | 5′-CACTGGAGCGTAAGAGGTCATT-3′ | 5′-CCACACTATCAAAGCCGTCAT-3′ |
| TaActin | 5′-CCTTAGTACCTTCCAACAGATGT-3′ | 5′-CCAGACAACTCGCAACTTAGA-3′ |
| Items | Con | Con + NaHS | Cd | Cd + NaHS |
|---|---|---|---|---|
| AQE | 0.072 ± 0.002 a | 0.074 ± 0.001 a | 0.039 ± 0.002 c | 0.052 ± 0.001 b |
| Rd (μmol CO2 m−2 s−1) | 1.119 ± 0.053 b | 1.268 ± 0.056 a | 0.614 ± 0.016 d | 0.948 ± 0.040 c |
| Pmax (μmol CO2 m−2 s−1) | 19.69 ± 0.178 b | 21.33 ± 0.265 a | 15.47 ± 0.273 d | 17.97 ± 0.050 c |
| LCP (μmol CO2 m−2 s−1) | 15.83 ± 0.578 c | 17.53 ± 0.589 ab | 16.08 ± 0.468 bc | 18.37 ± 0.907 a |
| LSP (μmol CO2 m−2 s−1) | 875.6 ± 22.10 ab | 893.1 ± 16.34 a | 782.1 ± 15.43 c | 845.2 ± 0.186 b |
| Amax (μmol CO2 m−2 s−1) | 27.41 ± 0.126 ab | 27.54 ± 0.283 a | 24.04 ± 0.422 c | 26.74 ± 0.261 b |
| Γ (μmol CO2 m−2 s−1) | 53.96 ± 0.226 c | 56.39 ± 0.148 b | 61.94 ± 1.533 a | 60.72 ± 0.409 a |
| CE (μmol CO2 m−2 s−1) | 0.175 ± 0.005 a | 0.178 ± 0.005 a | 0.090 ± 0.007 b | 0.099 ± 0.002 b |
| Rp (μmol CO2 m−2 s−1) | 8.062 ± 0.216 a | 8.494 ± 0.227 a | 5.017 ± 0.204 c | 5.537 ± 0.139 b |
| Parameters | Description |
|---|---|
| Fo | The initial (minimum) fluorescence intensity at 20 µs after dark adaptation |
| Fm | The maximum fluorescence intensity |
| Tfm | Time to reach maximal fluorescence intensity Fm |
| Fv/Fm | The maximal PSII photochemistry efficiency |
| Vj | Relative variable fluorescence intensity at J-step |
| Vi | Relative variable fluorescence intensity at I-step |
| dVG/dto | The net rate of reaction center is closed at 100 μs |
| dV/dto | The net rate of reaction center is closed at 300 μs |
| PI(abs) | Performance index on absorption basis |
| PI(total) | Performance index for energy conservation from exciton to the reduction of PSI end acceptors |
| Wk | The degree of damage to oxygen-evolving center |
| ψEo | Probability that a trapped exciton moves an electron into the electron transport chain beyond Q−A (at t = 0) |
| φEo | Quantum yield (at t = 0) for electron transport |
| φRo | Quantum yield for reduction of end electron acceptors at PSI side |
| φDo | Quantum yield of dissipated energy |
| Mo | Approximated initial slope of the fluorescence transient |
| ABS/RC | Absorption flux per reaction center |
| TRo/RC | Trapped energy flux per reaction center |
| ETo/RC | Electron transport flux per reaction center |
| DIo/RC | Dissipated energy flux per reaction center |
| RC/CSo | Density of RCs per excited cross-section (at t = 0) |
| ABS/CSo | Absorption flux per excited cross-section (at t = 0) |
| TRo/CSo | Trapped energy flux per excited cross-section (at t = 0) |
| ETo/CSo | Electron transport flux per excited cross-section (at t = 0) |
| DIo/CSo | Dissipated energy flux per excited cross-section (at t = 0) |
| RC/CSo | Density of RCs per excited cross-section (at t = t Fm) |
| ABS/CSo | Absorption flux per excited cross-section (at t = t Fm) |
| TRo/CSo | Trapped energy flux per excited cross-section (at t = t Fm) |
| ETo/CSo | Electron transport flux per excited cross-section (at t = t Fm) |
| DIo/CSo | Dissipated energy flux per excited cross-section (at t = t Fm) |
| SFI(abs) | Structural function index |
| PI(ABS/CSo/CSm) | The performance indices were based on absorbed light energy (ABS)/basal fluorescence (Fo)/maximum fluorescence (Fm) |
| D.F. | Drive force photosynthesis |
| Treatments | Fructose Content (mg g−1 FW) | Glucose Content (mg g−1 FW) | Sucrose Content (mg g−1 FW) |
|---|---|---|---|
| Con | 8.78 ± 0.84 bc | 6.37 ± 0.45 b | 11.22 ± 0.91 b |
| Con + NaHS | 8.49 ± 0.47 c | 5.93 ± 0.25 b | 11.62 ± 0.65 b |
| Cd | 11.38 ± 0.54 a | 9.08 ± 0.61 a | 12.68 ± 0.3 b |
| Cd + NaHS | 10.06 ± 0.29 b | 6.79 ± 0.28 b | 14.29 ± 0.45 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, X.; Zhang, B.; Pan, N.; Cheng, X.; Lu, W. Hydrogen Sulfide Alleviates Cadmium Stress by Enhancing Photosynthetic Efficiency and Regulating Sugar Metabolism in Wheat Seedlings. Plants 2023, 12, 2413. https://doi.org/10.3390/plants12132413
Zheng X, Zhang B, Pan N, Cheng X, Lu W. Hydrogen Sulfide Alleviates Cadmium Stress by Enhancing Photosynthetic Efficiency and Regulating Sugar Metabolism in Wheat Seedlings. Plants. 2023; 12(13):2413. https://doi.org/10.3390/plants12132413
Chicago/Turabian StyleZheng, Xiang, Bei Zhang, Ni Pan, Xue Cheng, and Wei Lu. 2023. "Hydrogen Sulfide Alleviates Cadmium Stress by Enhancing Photosynthetic Efficiency and Regulating Sugar Metabolism in Wheat Seedlings" Plants 12, no. 13: 2413. https://doi.org/10.3390/plants12132413





