Production of New Biopesticides from Cymbopogon citratus for the Control of Coffee Rust (Hemileia vastatrix) under Laboratory and Field Conditions
Abstract
:1. Introduction
2. Results
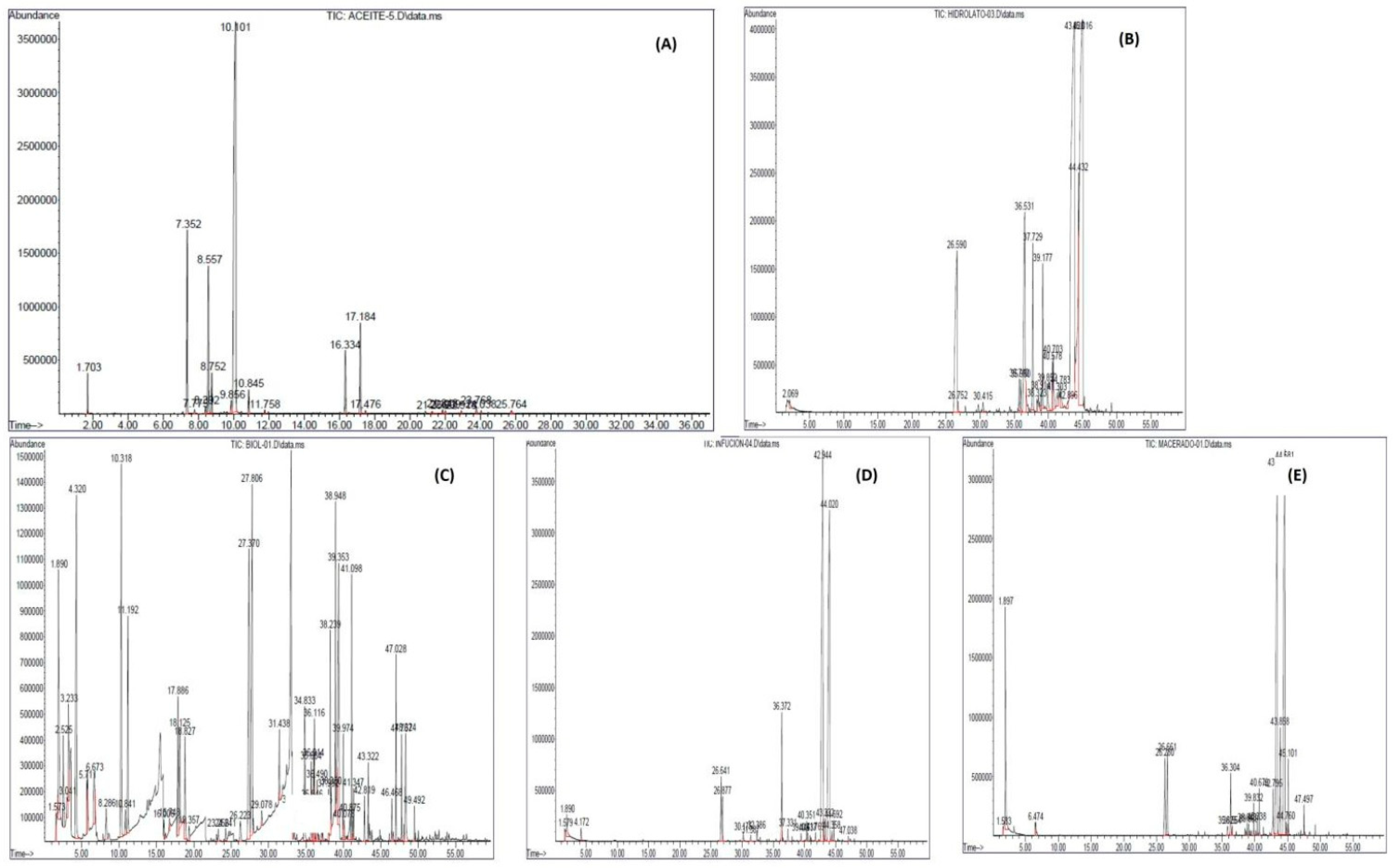
2.1. Chemical Composition of the Biopesticides Employed
2.2. Laboratory and Field Statistical Analysis
2.3. Spore Germination Behavior under Laboratory Conditions
2.4. Performance under Field Conditions
3. Discussion
4. Materials and Methods
4.1. Location of the Experimental Area in the Field
4.2. Field Experimental Design
4.3. Preparation of Biopesticides
4.4. Chromatographic Analysis of Biopesticides
4.5. Laboratory Tests
4.6. Field Trials
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Julca Otiniano, A.; Anyosa, C.E.; Manyari, Y.L.; Ventura, R.B.; Janampa, R.C.; Amez, S.B.; Costa, R.C. Una Revisión Sobre la Roya del Café (Hemileia vastatrix) Algunas Experiencias y Recomendaciones Para el Peru. Universidad Nacional Agraria La Molina. Instituto Regional de Desarrollo de Selva IRD-Selva. Sanidad Vegetal. 2013. Available online: http://repebis.upch.edu.pe/articulos/agro_enfoque/v28n189/a1.pdf (accessed on 5 July 2021).
- Moreno-Ruiz, G. Nueva variedad de café de porte alto resistente a la roya del cafeto. Cenicafé 2002, 53, 132–143. Available online: https://www.cenicafe.org/es/publications/arc053(02)132-143.pdf (accessed on 6 July 2020).
- Herrera Monroy, S.; Castro Brindis, R.; Pérez Moreno, J.; Valdés Velarde, E. Diversidad endomicorrícica en plantas de café (Coffea arabica L.) infestadas con roya (Hemileia vastatrix). Nova Sci. 2019, 11, 102–123. [Google Scholar] [CrossRef] [Green Version]
- Bustamante, J.; Roa, S.; Casanova, A.; Roso, L. Líneas de café resistentes a la roya en una localidad del Estado Táchira, Venezuela. Agron. Trop. 2004, 54, 75–91. Available online: http://ve.scielo.org/scielo.php?script=sci_arttext&pid=S0002-192X2004000100006&lng=es&tlng=es (accessed on 6 August 2020).
- Morales-Antonio, M.; Santiago-Martínez, G.; Vásquez-López, A.; Rodríguez-Ortiz, G.; Soto-Castro, D.; Lozano-Trejo, S.; Castañeda-Hidalgo, E. Uredospore germination of Hemileia vastatrix and its inhibition by the effect of plant extracts in vitro. Int. J. Agric. Nat. Resour. 2021, 48, 108–114. [Google Scholar] [CrossRef]
- Avelino, J.; Rivas, G. La Roya Anaranjada Del Cafeto. 2013. Available online: https://hal.archives-ouvertes.fr/hal-01071036 (accessed on 6 July 2020).
- Hernández-Martínez, G.; Velázquez-Premio, T. Análisis integral sobre la roya del café y su control. Rinderesu 2016, 1, 92–99. Available online: http://rinderesu.com/index.php/rinderesu/article/view/9/pdf (accessed on 6 July 2020).
- Sandoval-Islas, J.S.; Osada-Kawasoe, S.; Vivar-Flores, H.; Benítez-Riquelme, I. Correlación entre resistencia en plántula y resistencia en planta adulta a la roya amarilla y a la escaldadura de la cebada. Agrociencia 1999, 33, 415–422. Available online: https://agrociencia-colpos.mx/index.php/agrociencia/article/view/1627. (accessed on 6 July 2020).
- Rodríguez-García, M.F.; González-González, M.; Huerta-Espino, J.; Solano-Hernández, S. Evaluación de fungicidas para el control de la roya lineal amarilla en cebada. Rev. Mex. De Fitopatol. 2021, 37, S89. Available online: https://www.smf.org.mx/rmf/suplemento/docs/Volumen372019/S372019.pdf (accessed on 6 July 2020).
- Avelino, J.; Cristancho, M.; Georgiou, S.; Imbach, P.; Aguilar, L.; Bornemann, G.; Laderach, P.; Anzueto, F.; Hruska, A.J.; Morales, C. Las crisis de la roya del café en Colombia y Centroamérica (2008–2013): Impactos, posibles causas y propuestas de solución. Segur. Aliment. 2015, 7, 303–321. [Google Scholar] [CrossRef] [Green Version]
- Gamarra Gamarra, D.P.; Torres Suarez, G.; Villar Quiñonez, C.M.; McTaggart, A.R.; Carrasco Lozano, E.C. Relación filogenética de roya del café en la selva central de Perú. Acta Agronómica 2022, 70, 155–162. [Google Scholar] [CrossRef]
- Fisher, K.; Phillips, C. Potential antimicrobial uses of essential oils in food: Is citrus the answer? Trends Food Sci. Technol. 2008, 19, 156–164. [Google Scholar] [CrossRef]
- Zhang, Y.-P.; Wang, X.; Shen, Y.; Thakur, K.; Zhang, J.-G.; Hu, F.; Wei, Z.-J. Preparation and Characterization of Bio-Nanocomposites Film of Chitosan and Montmorillonite Incorporated with Ginger Essential Oil and Its Application in Chilled Beef Preservation. Antibiotics 2021, 10, 796. [Google Scholar] [CrossRef] [PubMed]
- Di Vaio, C.; Graziani, G.; Gaspari, A.; Scaglione, G.; Nocerino, S.; Ritieni, A. Essential oils content and antioxidant properties of peel ethanol extract in 18 lemon cultivars. Sci. Hortic. 2010, 126, 50–55. [Google Scholar] [CrossRef]
- Marei, G.I.K.; Rasoul, M.A.A.; Abdelgaleil, S.A.M. Comparative antifungal activities and biochemical effects of monoterpenes on plant pathogenic fungi. Pestic. Biochem. Physiol. 2012, 103, 56–61. [Google Scholar] [CrossRef]
- Guédez, C.; Cañizalez, L.; Avendaño, L.; Scorza, J.; Castillo, C.; Olivar, R.; Méndez, Y.; Sánchez, L. Actividad antifúngica del aceite esencial de naranja (Citrus sinensis L.) sobre hongos postcosecha en frutos de lechosa (Carica papaya L.). Rev. De La Soc. Venez. De Microbiol. 2014, 34, 81–87. Available online: http://ve.scielo.org/scielo.php?script=sci_arttext&pid=S1315-25562014000200007&lng=es&tlng=es. (accessed on 6 July 2020).
- He, L.L.; Zhao, Y.; Fan, L.M.; Zhan, J.J.; Tao, L.H.; Yang, Y.H.; Su, F.W.; Chen, Q.B.; Ye, M. In vitro and in vivo antifungal activity of Cymbopogon citrates essential oils from different climate conditions against Botrytis cinerea. Sci. Hortic. 2023, 308, 111544. [Google Scholar] [CrossRef]
- Rodríguez, A.T.; Morales, D.; Ramírez, M.A. Efecto de extractos vegetales sobre el crecimiento in vitro de hongos fitopatógenos. Cultiv. Trop. 2000, 21, 79–82. Available online: https://www.redalyc.org/articulo.oa?id=193215024014 (accessed on 6 July 2020).
- Rodríguez-García, M.F.; González-González, M.; Huerta-Espino, J.; Solano-Hernández, S. Evaluación de fungicidas contra roya amarilla (Puccinia striiformis f. sp. hordei) en seis variedades de cebada. Rev. Mex. De Fitopatol. 2021, 39, 414–434. [Google Scholar] [CrossRef]
- Reddy, P.R.K.; Elghandour, M.M.M.Y.; Salem, A.Z.M.; Yasaswini, D.; Reddy, P.P.R.; Reddy, A.N.; Hyder, I. Plant secondary metabolites as feed additives in calves for antimicrobial stewardship. Anim. Feed Sci. Technol. 2020, 264, 114469. [Google Scholar] [CrossRef]
- Singh, A.; Singh, D.K.; Mishra, T.N.; Agarwal, R.A. Molluskicides of plant origin. Biol. Agri. Horti. 1996, 13, 205–252. [Google Scholar] [CrossRef]
- Leng, P.; Zhang, Z.; Pan, G.; Zhao, M. Applications and development trends in biopesticides. Afr. J. Biotechnol. 2011, 10, 19864–19873. [Google Scholar] [CrossRef]
- Rojas, J.; Ronceros, S.; Palacios, O.; Sevilla, C. Efecto anti-Trypanosoma cruzi del aceite esencial de Cymbopogon citratus (DC) Stapf (hierba luisa) en ratones Balb/c. An. De La Fac. De Med. 2012, 73, 7–12. Available online: http://www.scielo.org.pe/scielo.php?script=sci_arttext&pid=S1025-55832012000100002&lng=es&tlng=es. (accessed on 6 July 2020). [CrossRef] [Green Version]
- Negrelle, R.R.B.; Gomes, E.C. Cymbopogon citratus (DC.) Stapf: Chemical composition and biological activities. Rev. Bras. Pl. Med. 2007, 9, 80–92. Available online: https://www1.ibb.unesp.br/Home/Departamentos/Botanica/RBPM-RevistaBrasileiradePlantasMedicinais/revisao_v9_n1.pdf (accessed on 6 July 2021).
- Beyra, A.; León, M.; Iglesias, E.; Ferrándiz, D.; Herrera, R.; Volpato, G.; Godínez, D.; Guimarais, M.; Álvarez, R. Estudios etnobotánicos sobre plantas medicinales en la provincia de Camagüey (Cuba). An. Jardín Botánico De Madr. 2004, 61, 185–204. [Google Scholar]
- Macia, J.; García, M.; Vidaurre, J. An ethnobotanical survey of medicinal plants commercialized in the markets of La Paz and El alto, Bolivia. J. Ethnopharmacol. 2005, 97, 337–350. [Google Scholar] [CrossRef]
- Goñi, P.; López, P.; Sánchez, C.; Gómez-Lus, R.; Becerril, R.; Nerín, C.N. Antimicrobial activity in the vapor phase of a combination of cinnamon and clove essential oils. Food Chemestry 2009, 116, 982–989. [Google Scholar] [CrossRef]
- Tofiño-Rivera, A.; Ortega-Cuadros, M.; Galvis-Pareja, D.E.; Jiménez-Rios, H.; Merini, L.J.; Martínez-Pabón, M.C. Effect of Lippia alba and Cymbopogon citratus essential oils on biofilms of Streptococcus mutans and cytotoxicity in CHO cells. J. Ethnopharmacol. 2016, 194, 749–754. [Google Scholar] [CrossRef] [Green Version]
- Vázquez-Briones, M.C.; Guerrero-Beltrán, J.Á. Efecto del aceite esencial de Cymbopogon citratus sobre propiedades fisicoquímicas en películas de quitosano. Sci. Agropecu. 2017, 8, 401–409. [Google Scholar] [CrossRef] [Green Version]
- Mongkolsilp, S.; Pongbupakit, I.; Sae-Lee, N.; Sitthithaworn, W. Radical scavenging activity and total phenolic content of medicinal plants used in primary health care. SWU J. Pharm. Sci. 2004, 9, 32–35. [Google Scholar]
- Tiwari, M.; Dwivedi, U.; Kakkar, P. Suppression ofoxidative stress and pro-inflammatory mediators by Cymbopogon citratus D Stapf extract in lipopolysaccharide stimulated murine alveolar macrophages. Food Chem. Toxicol. 2010, 48, 2913–2919. [Google Scholar] [CrossRef]
- Nwachukwu, I.; Allison, L.; Chinakwe, E.; Nwadiaro, P. Studies on the effects Cymbopogon citratus, Ceiba pentandra and Loranthus bengwelensis extracts on species of dermatophytes. J. Am. Sci. 2008, 4, 58–67. [Google Scholar]
- Wright, S.C.; Maree, J.E.; Sibanyoni, M. Treatment of oral thrush in HIV/AIDS patients with lemon juice and lemon grass (Cymbopogon citratus) and gentian violet. Phytomedicine Int. J. Phytother. Phytopharm. 2009, 16, 118–124. [Google Scholar] [CrossRef]
- Okigbo, R.; Mmeka, C. Antimicrobial effects of three tropical plant extracts on staphyolococcus aureus, Escherichia coli and Candida albicans. Afr. J. Trad. Complement. Altern. Med. 2008, 5, 226–229. [Google Scholar] [CrossRef] [Green Version]
- Pushpanathan, T.; Jebanesan, A.; Govindarajan, M. Larvicidal, ovicidal and repellent activities of Cymbopogan citratus Stapf (Graminae) essential oil against the filarial mosquito Culex quinquefasciatus (Say) (Diptera: Culicidae). Trop. Biomed. 2006, 23, 208–212. [Google Scholar] [PubMed]
- Al-Reza, S.M.; Bajpai, V.K.; Kang, S.C. Antioxidant and antilisterial effect of seed essential oil and organic extracts from Zizyphus jujuba. Food Chem. Toxicol. 2009, 47, 2374–2380. [Google Scholar] [CrossRef] [PubMed]
- Dan, H.A.; Barroso, A.L.L.; Dan, L.G.M.; Finotte, T.R.; Feldkircher, C.A. Adjuvantes multifuncionais associados ao herbicida glyphosate no controle de Digitaria insularis. Glob. Sci. Technol. 2010, 3, 30–38. [Google Scholar]
- Otiniano, A.J.; Ventura, R.B.; Huamán, L.A.; Vera, N.J.; Cepero, V.C.; Amez, S.B. Relación entre la incidencia y la severidad de la roya del café (Hemileia vastatrix) en san ramón, Chanchamayo, Perú. Cienc. E Investig. 2019, 4, 1–9. [Google Scholar] [CrossRef]
- Plaus Albado, E.; Saez Flores, G.; Grabiel Ataucusi, S. Chemical composition and antibacterial activity of Origanum vulgare (oregano) essential oil. Rev. Médica Hered. 2001, 12, 16–19. [Google Scholar]
- Chang, Y.; Harmon, P.F.; Treadwell, D.D.; Carrillo, D.; Sarkhosh, A.; Brecht, J.K. Biocontrol Potential of Essential Oils in Organic Horticulture Systems: From Farm to Fork. Front. Nutr. 2022, 8, 805138. [Google Scholar] [CrossRef]
- Yan, J.; Wu, H.; Chen, K.; Feng, J.; Zhang, Y. Antifungal Activities and Mode of Action of Cymbopogon citratus, Thymus vulgraris, and Origanum heracleoticum Essential Oil Vapors against Botrytis cinerea and Their Potential Application to Control Postharvest Strawberry Gray Mold. Foods 2021, 10, 2451. [Google Scholar] [CrossRef]
- Wu, Y.X.; Zhang, Y.D.; Li, N.; Wu, D.D.; Li, Q.M.; Chen, Y.Z.; Zhang, G.C.; Yang, J. Inhibitory effect and mechanism of action of juniper essential oil on gray mold in cherry tomatoes. Front. Microbiol. 2022, 13, 1000526. [Google Scholar] [CrossRef]
- El Abdali, Y.; Agour, A.; Allali, A.; Bourhia, M.; El Moussaoui, A.; Eloutassi, N.; Salamatullah, A.M.; Alzahrani, A.; Ouahmane, L.; Aboul-Soud, M.A.M.; et al. Lavandula dentata L.: Phytochemical Analysis, Antioxidant, Antifungal and Insecticidal Activities of Its Essential Oil. Plants 2022, 11, 311. [Google Scholar] [CrossRef]
- Tomazoni, L.Z.; Griggio, G.S.; Broilo, E.P.; Ribeiro, R.T.S.; Soares, G.L.G.; Schwambach, J. Screening for inhibitory activity of essential oils on fungal tomato pathogen Stemphylium solani Weber. Biocatal. Agric. Biotechnol. 2018, 16, 364–372. [Google Scholar] [CrossRef]
- Armenta, J.E.; Bejarano, D.A.; López, L.G.; Cruz, M.A. Evaluación cualitativa deextractos maceradoscon diferentes solventes para la extracción de glicoalcaloides y flavonoides de Dioscórea bulbifera en condiciones estándar obtenidos en los llanos orientales de Colombia. Entre Cienc. E Ing. 2020, 14, 59–65. [Google Scholar] [CrossRef]
- Guevara, D.I.; Ramos, B.M.; Karell, B.J.R.; Santana, D.P. Optimization of the process of obtaining an aqueous extract of cocoa husk. CENIC Chem. Sci. J. 2022, 53, 60–71. Available online: http://scielo.sld.cu/pdf/rccq/v53n1/2221-2442-rccq-53-01-60.pdf (accessed on 6 July 2021).
- Acosta, M.J.; Hurtado, B.A.; Arango, B.O.; Alvarez, S.D.; Salazar, G.C. Efecto de abonos orgánicos a partir de subproductos del fique en la producción de maíz. Biotecnol. En El Sect. Agropecu. Y Agroind. 2013, 11, 94–102. Available online: http://www.scielo.org.co/scielo.php?script=sci_arttext&pid=S1692-35612013000100012&lng=en&tlng=. (accessed on 6 July 2021).
- Adorno Chavez, M.; Soilán Duarte, L.C. Effect of fungicides on anthracnose (Colletotrichum fragariae Brooks) on strawberry plants [Fragaria × ananassa (Duchesne ex Weston) Duchesne ex Rozier]. Investig. Agrar. 2018, 20, 38–50. [Google Scholar] [CrossRef]
- SENASA. Norma para la ejecución y remisión de información de actividades del programa manejo integrado de plagas del cafeto. In National Agricultural Health Service; Ministry of Agrarian Development and Irrigation: Lima, Peru, 2013. [Google Scholar]
- Campbell, C.L.; Madden, L.V. Introduction to Plant Disease Epidemiology; John Wiley and Sons: New York, NY, USA, 1990; 532p. [Google Scholar]
- Bhering, L.L. Rbio: A Tool For Biometric And Statistical Analysis Using The R Platform. Crop Breed. Appl. Biotechnol. 2017, 17, 187–190. [Google Scholar] [CrossRef] [Green Version]




| FV 1 | GL 2 | Field | GL | Laboratory | |||
|---|---|---|---|---|---|---|---|
| Incidence (%) | Severity (%) | AUDPC 3 | GLight (%) | GDark (%) | |||
| Biopesticides (B) | 4 | 3338 *** | 854 *** | 5 *** | 4 | 3 | 3 |
| Concentration (C) | 3 | 56582 *** | 62370 *** | 4 *** | 3 | 4 *** | 5 *** |
| Evaluation (E) | 3 | 15929 *** | 1010 *** | ||||
| B × C | 12 | 485 *** | 191 *** | 2 *** | 12 | 2 | 2 |
| B × E | 12 | 329 *** | 142 *** | ||||
| C × E | 9 | 3381 *** | 1709 *** | ||||
| B × C × E | 36 | 48 *** | 30 *** | ||||
| Waste | 160 | 0.37 | 0.27 | 3 | 40 | 5 | 4 |
| CV (%) | 1.39 | 1.99 | 1.31 | 26.75 | 16.03 | ||
| Biopesticides/Concentration | I Week | II Week | ||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 15 | 20 | 25 | 0 | 15 | 20 | 25 | |
| Essential oil | 81.7 Aa | 39.7 Bc | 30.7 Cd | 26.7 Of | 86.77 Aa | 10.4 Be | 5.3 Cd | 1.0 Of |
| Biol | 81.7 Aa | 78.3 Ba | 67.9 Cb | 49.2 Dc | 86.77 Aa | 63.4 Ba | 55.3 Ca | 35.4 Da |
| Macerated | 81.7 Aa | 79.1 Ba | 69.2 Cab | 53.3 Db | 86.77 Aa | 49.0 Bd | 38.9 Cc | 28.7 Dc |
| Hydrolat | 81.7 Aa | 79.6 Ba | 69.8 Ca | 54.9 Da | 86.77 Aa | 57.0 Bb | 45.6 Cb | 30.3 Db |
| Infusion | 81.7 Aa | 75.5 Bb | 58.0 Cc | 45.7 Dd | 86.77 Aa | 55.4 Bc | 45.4 Cb | 21.6 Dd |
| Biopesticides/Concentration | III Week | IV Week | ||||||
| 0 | 15 | 20 | 25 | 0 | 15 | 20 | 25 | |
| Essential oil | 89.73 Aa | 1.0 Bd | 0.7 Be | 0.2 Bc | 95.67 Aa | 0.0 Bd | 0.0 Bd | 0.0 Bb |
| Biol | 89.73 Aa | 37.8 Ba | 21.5 Ca | 14.7 Da | 95.67 Aa | 19.5 Ba | 9.8 Ca | 5.7 Da |
| Macerated | 89.73 Aa | 22.7 Bc | 18.0 Cc | 7.8 Db | 95.67 Aa | 7.7 Bc | 1.7 Cc | 0.2 Db |
| Hydrolat | 89.73 Aa | 31.1 Bb | 15.0 Cd | 8.9 Db | 95.67 Aa | 9.0 Bc | 4.4 Cb | 0.7 Db |
| Infusion | 89.73 Aa | 31.2 Bb | 19.9 Cb | 8.3 Db | 95.67 Aa | 11.1 Bb | 3.7 Cb | 0.4 Db |
| Biopesticides/Concentration | Concentration 0 | Concentration 15 | ||||||
|---|---|---|---|---|---|---|---|---|
| I Week | II Week | III Week | IV Week | I Week | II Week | III Week | IV Week | |
| Essential oil | 81.7 D | 86.8 C | 89.7 B | 95.7 A | 39.7 A | 10.4 B | 1.0 C | 0.0 C |
| Biol | 81.7 D | 86.8 C | 89.7 B | 95.7 A | 78.3 A | 63.4 B | 37.8 C | 19.5 D |
| Macerated | 81.7 D | 86.8 C | 89.7 B | 95.7 A | 79.1 A | 49.0 B | 22.7 C | 7.7 D |
| Hydrolat | 81.7 D | 86.8 C | 89.7 B | 95.7 A | 79.6 A | 57.0 B | 31.1 C | 9.0 D |
| Infusion | 81.7 D | 86.8 C | 89.7 B | 95.7 A | 75.5 A | 55.4 B | 31.2 C | 11.1 D |
| Biopesticides/Concentration | Concentration 20 | Concentration 25 | ||||||
| I Week | II Week | III Week | IV Week | I Week | II Week | III Week | IV Week | |
| Essential oil | 30.7 A | 5.3 B | 0.7 C | 0.0 C | 26.7 A | 1.0 B | 0.2 B | 0.0 B |
| Biol | 67.9 A | 55.3 B | 21.5 C | 9.8 D | 49.2 A | 35.4 B | 14.7 C | 5.7 D |
| Macerated | 69.2 A | 38.9 B | 18.0 C | 1.7 D | 53.3 A | 28.7 B | 7.8 C | 0.2 D |
| Hydrolat | 69.8 A | 45,6 B | 15.0 C | 4.4 D | 54.9 A | 30.3 B | 8.9 C | 0.7 D |
| Infusion | 58.0 A | 45.4 B | 19.9 C | 3.7 D | 45.7 A | 21.6 B | 8.3 C | 0.4 D |
| Biopesticides/Concentration | I Week | II Week | ||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 15 | 20 | 25 | 0 | 15 | 20 | 25 | |
| Essential oil | 55.6 Aa | 5.0 Bc | 4.0 Bc | 2.0 Cc | 80.0 Aa | 2.0 Bc | 2.0 Bc | 0.0 Cc |
| Biol | 55.6 Aa | 25.0 Bb | 25.0 Bb | 10.0 Cb | 80.0 Aa | 10.0 Bb | 10.0 Bb | 5.0 Cb |
| Macerated | 55.6 Aa | 40 Ba | 40.0 Ba | 25.0 Ca | 80.0 Aa | 25.0 Ba | 25.0 Ba | 10.0 Ca |
| Hydrolat | 55.6 Aa | 40 Ba | 25.0 Cb | 10.0 Db | 80.0 Aa | 25.0 Ba | 10.0 Cb | 5.0 Db |
| Infusion | 55.6 Aa | 40 Ba | 25.0 Cb | 10.0 Db | 80.0 Aa | 25.0 Ba | 10.0 Cb | 5.0 Db |
| Biopesticides/Concentration | III Week | IV Week | ||||||
| 0 | 15 | 20 | 25 | 0 | 15 | 20 | 25 | |
| Essential oil | 80.0 Aa | 0.0 Bc | 0.0 Bc | 0.0 Bc | 80.0 Aa | 0.0 Bc | 0.0 Bc | 0.0 Ba |
| Biol | 80.0 Aa | 5.0 Bb | 5.0 Bb | 2.0 Cb | 80.0 Aa | 2.0 Bb | 2.0 Bb | 0.7 Ca |
| Macerated | 80.0 Aa | 10.0 Ba | 8.3 Ca | 5.0 Da | 80.0 Aa | 5.0 Ba | 4.0 Ba | 0.7 Ca |
| Hydrolat | 80.0 Aa | 10.0 Ba | 5.0 Cb | 2.0 Db | 80.0 Aa | 5.0 Ba | 2.0 Cb | 0.0 Da |
| Infusion | 80.0 Aa | 10.0 Ba | 5.0 Cb | 3.0 Db | 80.0 Aa | 5.0 Ba | 2.0 Cb | 0.7 Da |
| Biopesticides/Concentration | Concentration 0 | Concentration 15 | ||||||
|---|---|---|---|---|---|---|---|---|
| I Week | II Week | III Week | IV Week | I Week | II Week | III Week | IV Week | |
| Essential oil | 55.6 B | 80.0 A | 80.0 A | 80.0 A | 5.0 A | 2.0 B | 0.0 C | 0.0 C |
| Biol | 55.6 B | 80.0 A | 80.0 A | 80.0 A | 25.0 A | 10.0 B | 5.0 C | 2.0 D |
| Macerated | 55.6 B | 80.0 A | 80.0 A | 80.0 A | 40.0 A | 25.0 B | 10.0 C | 5.0 D |
| Hydrolat | 55.6 B | 80.0 A | 80.0 A | 80.0 A | 40.0 A | 25.0 B | 10.0 C | 5.0 D |
| Infusion | 55.6 B | 80.0 A | 80.0 A | 80.0 A | 40.0 A | 25.0 B | 10.0 C | 5.0 D |
| Biopesticides/Concentration | Concentration 20 | Concentration 25 | ||||||
| I Week | II Week | III Week | IV Week | I Week | II Week | III Week | IV Week | |
| Essential oil | 4.0 A | 2.0 B | 0.0 C | 0.0 C | 2.0 A | 0.0 B | 0.0 B | 0.0 B |
| Biol | 25.0 A | 10.0 B | 5.0 C | 2.0 D | 10.0 A | 5.0 B | 2.0 C | 0.7 D |
| Macerated | 40.0 A | 25.0 B | 8.3 C | 4.0 D | 25.0 A | 10.0 B | 5.0 C | 0.7 D |
| Hydrolat | 25.0 A | 10.0 B | 5.0 C | 2.0 D | 10.0 A | 5.0 B | 2.0 C | 0.0 D |
| Infusion | 25.0 A | 10.0 B | 5.0 C | 2.0 D | 10.0 A | 5.0 B | 3.0 C | 0.7 D |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morales-Aranibar, L.; Yucra, F.E.Y.; Estrada, N.M.P.; Flores, P.Q.; Zevallos, R.N.M.; Zegarra, J.C.L.; Trujillo, U.P.; Aranibar, C.G.M.; Gonzales, H.H.S.; Aguilera, J.G.; et al. Production of New Biopesticides from Cymbopogon citratus for the Control of Coffee Rust (Hemileia vastatrix) under Laboratory and Field Conditions. Plants 2023, 12, 1166. https://doi.org/10.3390/plants12051166
Morales-Aranibar L, Yucra FEY, Estrada NMP, Flores PQ, Zevallos RNM, Zegarra JCL, Trujillo UP, Aranibar CGM, Gonzales HHS, Aguilera JG, et al. Production of New Biopesticides from Cymbopogon citratus for the Control of Coffee Rust (Hemileia vastatrix) under Laboratory and Field Conditions. Plants. 2023; 12(5):1166. https://doi.org/10.3390/plants12051166
Chicago/Turabian StyleMorales-Aranibar, Luis, Francisca Elena Yucra Yucra, Nivia Marisol Pilares Estrada, Policarpo Quispe Flores, Renatto Nicolino Motta Zevallos, Juan Carlos Llanos Zegarra, Uriel Palomino Trujillo, Carlos Genaro Morales Aranibar, Hebert Hernán Soto Gonzales, Jorge González Aguilera, and et al. 2023. "Production of New Biopesticides from Cymbopogon citratus for the Control of Coffee Rust (Hemileia vastatrix) under Laboratory and Field Conditions" Plants 12, no. 5: 1166. https://doi.org/10.3390/plants12051166






