Dynamic Regulation of Grapevine’s microRNAs in Response to Mycorrhizal Symbiosis and High Temperature
Abstract
:1. Introduction
2. Results
2.1. Mycorrhizal Inoculation Results in Better Plant Response to High Temperatures
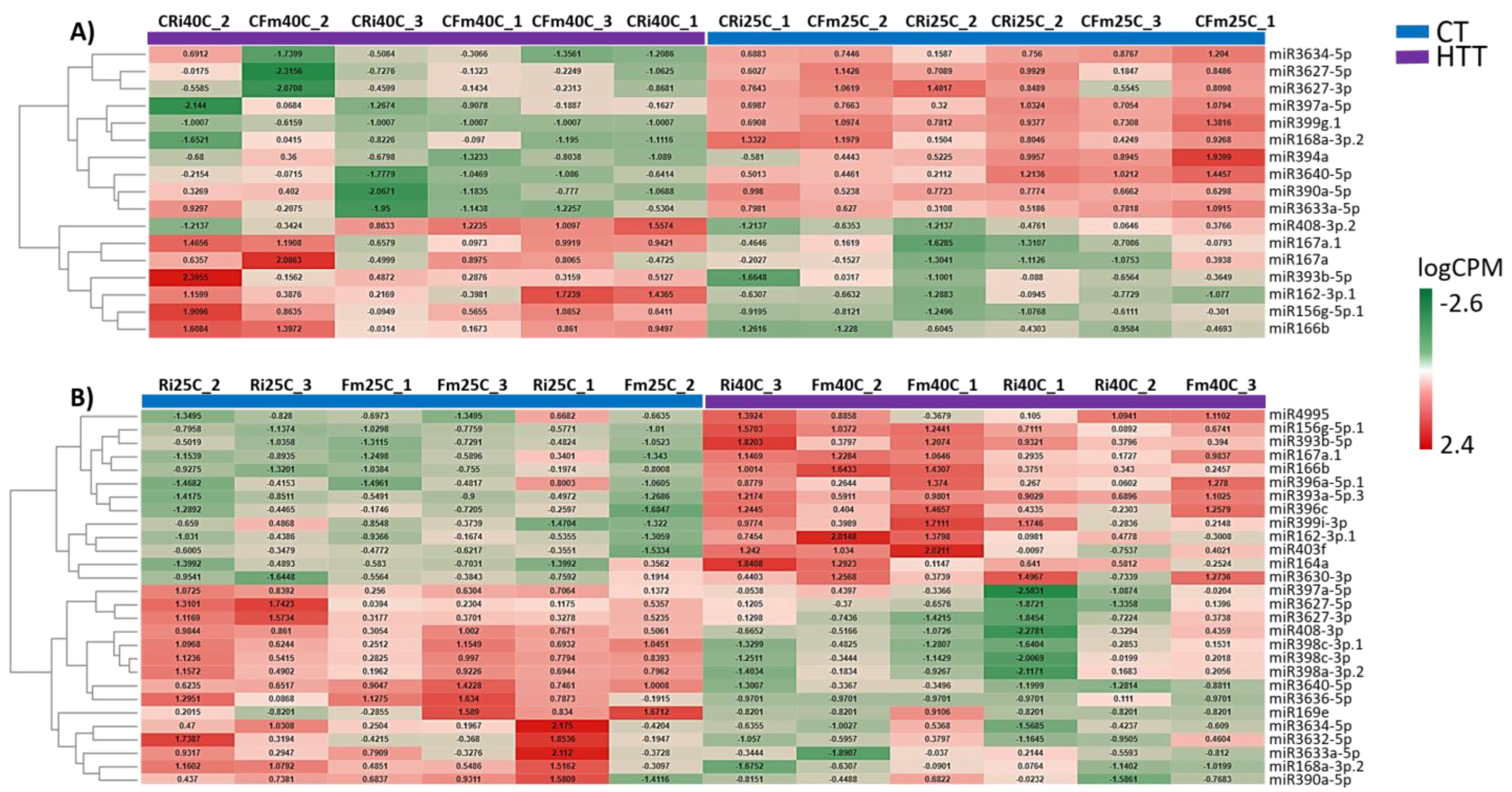
2.2. Grapevine miRNA Profiling
2.3. Heat- and AMF-Responsive miRNAs in Grapevine Leaves
2.4. Prediction of Grapevine miRNA Target Genes
2.5. GO and KEGG Functional Analyses of miRNA Target Genes
2.6. Prediction of Heat-Associated Regulatory Networks
3. Discussion
3.1. Mycorrhizal Plants Show Enhanced Response to High Temperature
3.2. Heat Stress Differently Affects miRNA Regulation in Mycorrhizal vs. Non-Mycorrhizal Grapevines
3.3. Mycorrhizal Species Influence microRNA Expression under Heat Stress
4. Materials and Methods
4.1. Plant Material and Growth Conditions
4.2. Grapevine Exposure to High Temperature Treatment
4.3. Small RNA Library Construction and Sequencing
4.4. Bioinformatic Analysis and miRNA Identification
4.5. Prediction of Novel miRNA
4.6. Target Prediction of Conserved and Novel miRNAs
4.7. Differential Expression Analysis of miRNAs
4.8. Validation of miRNA and Target Gene Expression by RT-qPCR
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schüβler, A.; Schwarzott, D.; Walker, C. A New Fungal Phylum, the Glomeromycota: Phylogeny and Evolution* *Dedicated to Manfred Kluge (Technische Universität Darmstadt) on the Occasion of His Retirement. Mycol. Res. 2001, 105, 1413–1421. [Google Scholar] [CrossRef] [Green Version]
- Smith, S.E.; Read, D. 4-Growth and Carbon Economy of Arbuscular Mycorrhizal Symbionts. In Mycorrhizal Symbiosis, 3rd ed.; Smith, S.E., Read, D., Eds.; Academic Press: Cambridge, MA, USA, 2008; pp. 117–144. ISBN 978-0-12-370526-6. [Google Scholar]
- Smith, S.E.; Read, D. 5-Mineral Nutrition, Toxic Element Accumulation and Water Relations of Arbuscular Mycorrhizal Plants. In Mycorrhizal Symbiosis, 3rd ed.; Smith, S.E., Read, D., Eds.; Academic Press: Cambridge, MA, USA, 2008; pp. 145–187. ISBN 978-0-12-370526-6. [Google Scholar]
- Campos, C.; Nobre, T.; Goss, M.J.; Faria, J.; Barrulas, P.; Carvalho, M. Transcriptome Analysis of Wheat Roots Reveals a Differential Regulation of Stress Responses Related to Arbuscular Mycorrhizal Fungi and Soil Disturbance. Biology 2019, 8, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mathur, S.; Tomar, R.S.; Jajoo, A. Arbuscular Mycorrhizal Fungi (AMF) Protects Photosynthetic Apparatus of Wheat under Drought Stress. Photosynth. Res. 2019, 139, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Maya, M.A.; Matsubara, Y. Influence of Arbuscular Mycorrhiza on the Growth and Antioxidative Activity in Cyclamen under Heat Stress. Mycorrhiza 2013, 23, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Bowles, T.M.; Barrios-Masias, F.H.; Carlisle, E.A.; Cavagnaro, T.R.; Jackson, L.E. Effects of Arbuscular Mycorrhizae on Tomato Yield, Nutrient Uptake, Water Relations, and Soil Carbon Dynamics under Deficit Irrigation in Field Conditions. Sci. Total Environ. 2016, 566–567, 1223–1234. [Google Scholar] [CrossRef] [Green Version]
- Augé, R.M.; Toler, H.D.; Saxton, A.M. Arbuscular Mycorrhizal Symbiosis and Osmotic Adjustment in Response to NaCl Stress: A Meta-Analysis. Front. Plant Sci. 2014, 5, 562. [Google Scholar] [CrossRef] [Green Version]
- Daynes, C.N.; Field, D.J.; Saleeba, J.A.; Cole, M.A.; McGee, P.A. Development and Stabilisation of Soil Structure via Interactions between Organic Matter, Arbuscular Mycorrhizal Fungi and Plant Roots. Soil Biol. Biochem. 2013, 57, 683–694. [Google Scholar] [CrossRef]
- Nuccio, E.E.; Hodge, A.; Pett-Ridge, J.; Herman, D.J.; Weber, P.K.; Firestone, M.K. An Arbuscular Mycorrhizal Fungus Significantly Modifies the Soil Bacterial Community and Nitrogen Cycling during Litter Decomposition. Environ. Microbiol. 2013, 15, 1870–1881. [Google Scholar] [CrossRef]
- Schmitz, A.M.; Harrison, M.J. Signaling Events during Initiation of Arbuscular Mycorrhizal Symbiosis. J. Integr. Plant Biol. 2014, 56, 250–261. [Google Scholar] [CrossRef]
- Vangelisti, A.; Natali, L.; Bernardi, R.; Sbrana, C.; Turrini, A.; Hassani-Pak, K.; Hughes, D.; Cavallini, A.; Giovannetti, M.; Giordani, T. Transcriptome Changes Induced by Arbuscular Mycorrhizal Fungi in Sunflower (Helianthus annuus L.) Roots. Sci. Rep. 2018, 8, 4. [Google Scholar] [CrossRef] [Green Version]
- Schweiger, R.; Baier, M.C.; Persicke, M.; Müller, C. High Specificity in Plant Leaf Metabolic Responses to Arbuscular Mycorrhiza. Nat. Commun. 2014, 5, 3886. [Google Scholar] [CrossRef] [Green Version]
- Salvioli, A.; Zouari, I.; Chalot, M.; Bonfante, P. The Arbuscular Mycorrhizal Status Has an Impact on the Transcriptome Profile and Amino Acid Composition of Tomato Fruit. BMC Plant Biol. 2012, 12, 44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zouari, I.; Salvioli, A.; Chialva, M.; Novero, M.; Miozzi, L.; Tenore, G.C.; Bagnaresi, P.; Bonfante, P. From Root to Fruit: RNA-Seq Analysis Shows That Arbuscular Mycorrhizal Symbiosis May Affect Tomato Fruit Metabolism. BMC Genom. 2014, 15, 221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiorilli, V.; Vannini, C.; Ortolani, F.; Garcia-Seco, D.; Chiapello, M.; Novero, M.; Domingo, G.; Terzi, V.; Morcia, C.; Bagnaresi, P.; et al. Omics Approaches Revealed How Arbuscular Mycorrhizal Symbiosis Enhances Yield and Resistance to Leaf Pathogen in Wheat. Sci. Rep. 2018, 8, 9625. [Google Scholar] [CrossRef] [Green Version]
- Borges, F.; Martienssen, R.A. The Expanding World of Small RNAs in Plants. Nat. Rev. Mol. Cell Biol. 2015, 16, 727–741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, B.; Pan, X.; Cannon, C.H.; Cobb, G.P.; Anderson, T.A. Conservation and Divergence of Plant MicroRNA Genes. Plant J. 2006, 46, 243–259. [Google Scholar] [CrossRef] [PubMed]
- Spanudakis, E.; Jackson, S. The Role of MicroRNAs in the Control of Flowering Time. J. Exp. Bot. 2014, 65, 365–380. [Google Scholar] [CrossRef]
- Lan, Y.; Su, N.; Shen, Y.; Zhang, R.; Wu, F.; Cheng, Z.; Wang, J.; Zhang, X.; Guo, X.; Lei, C.; et al. Identification of Novel MiRNAs and MiRNA Expression Profiling during Grain Development in Indica Rice. BMC Genom. 2012, 13, 264. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Meng, X.; Dobrovolskaya, O.B.; Orlov, Y.L.; Chen, M. Non-Coding RNAs and Their Roles in Stress Response in Plants. Genom. Proteom. Bioinform. 2017, 15, 301–312. [Google Scholar] [CrossRef]
- Pagano, L.; Rossi, R.; Paesano, L.; Marmiroli, N.; Marmiroli, M. MiRNA Regulation and Stress Adaptation in Plants. Environ. Exp. Bot. 2021, 184, 104369. [Google Scholar] [CrossRef]
- Devers, E.A.; Branscheid, A.; May, P.; Krajinski, F. Stars and Symbiosis: MicroRNA- and MicroRNA*-Mediated Transcript Cleavage Involved in Arbuscular Mycorrhizal Symbiosis. Plant Physiol. 2011, 156, 1990–2010. [Google Scholar] [CrossRef] [Green Version]
- Lauressergues, D.; Delaux, P.-M.; Formey, D.; Lelandais-Brière, C.; Fort, S.; Cottaz, S.; Bécard, G.; Niebel, A.; Roux, C.; Combier, J.-P. The MicroRNA MiR171h Modulates Arbuscular Mycorrhizal Colonization of Medicago Truncatula by Targeting NSP2: MiR171h Regulates Fungal Colonization. Plant J. 2012, 72, 512–522. [Google Scholar] [CrossRef] [PubMed]
- Mewalal, R.; Yin, H.; Hu, R.; Jawdy, S.; Vion, P.; Tuskan, G.A.; Le Tacon, F.; Labbé, J.L.; Yang, X. Identification of Populus Small RNAs Responsive to Mutualistic Interactions With Mycorrhizal Fungi, Laccaria Bicolor and Rhizophagus Irregularis. Front. Microbiol. 2019, 10, 515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, P.; Wu, Y.; Liu, C.-C.; Liu, L.-W.; Ma, F.-F.; Wu, X.-Y.; Wu, M.; Hang, Y.-Y.; Chen, J.-Q.; Shao, Z.-Q.; et al. Identification of Arbuscular Mycorrhiza (AM)-Responsive MicroRNAs in Tomato. Front. Plant Sci. 2016, 7, 429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Formey, D.; Sallet, E.; Lelandais-Brière, C.; Ben, C.; Bustos-Sanmamed, P.; Niebel, A.; Frugier, F.; Combier, J.P.; Debellé, F.; Hartmann, C.; et al. The Small RNA Diversity from Medicago Truncatula Roots under Biotic Interactions Evidences the Environmental Plasticity of the MiRNAome. Genome Biol. 2014, 15, 457. [Google Scholar] [CrossRef]
- Guan, Q.; Lu, X.; Zeng, H.; Zhang, Y.; Zhu, J. Heat Stress Induction of MiR398 Triggers a Regulatory Loop That Is Critical for Thermotolerance in Arabidopsis. Plant J. 2013, 74, 840–851. [Google Scholar] [CrossRef]
- Li, A.-L.; Wen, Z.; Yang, K.; Wen, X.-P. Conserved MiR396b-GRF Regulation Is Involved in Abiotic Stress Responses in Pitaya (Hylocereus polyrhizus). Int. J. Mol. Sci. 2019, 20, 2501. [Google Scholar] [CrossRef] [Green Version]
- Ravichandran, S.; Ragupathy, R.; Edwards, T.; Domaratzki, M.; Cloutier, S. MicroRNA-Guided Regulation of Heat Stress Response in Wheat. BMC Genom. 2019, 20, 488. [Google Scholar] [CrossRef] [Green Version]
- Samad, A.F.A.; Sajad, M.; Nazaruddin, N.; Fauzi, I.A.; Murad, A.M.A.; Zainal, Z.; Ismail, I. MicroRNA and Transcription Factor: Key Players in Plant Regulatory Network. Front. Plant Sci. 2017, 8, 565. [Google Scholar] [CrossRef] [Green Version]
- Stief, A.; Altmann, S.; Hoffmann, K.; Pant, B.D.; Scheible, W.-R.; Bäurle, I. Arabidopsis MiR156 Regulates Tolerance to Recurring Environmental Stress through SPL Transcription Factors. Plant Cell 2014, 26, 1792–1807. [Google Scholar] [CrossRef] [Green Version]
- Hivrale, V.; Zheng, Y.; Puli, C.O.R.; Jagadeeswaran, G.; Gowdu, K.; Kakani, V.G.; Barakat, A.; Sunkar, R. Characterization of Drought- and Heat-Responsive MicroRNAs in Switchgrass. Plant Sci. 2016, 242, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Trouvelot, S.; Bonneau, L.; Redecker, D.; van Tuinen, D.; Adrian, M.; Wipf, D. Arbuscular Mycorrhiza Symbiosis in Viticulture: A Review. Agron. Sustain. Dev. 2015, 35, 1449–1467. [Google Scholar] [CrossRef] [Green Version]
- Carvalho, L.C.; Coito, J.L.; Colaço, S.; Sangiogo, M.; Amâncio, S. Heat Stress in Grapevine: The Pros and Cons of Acclimation. Plant Cell Environ. 2015, 38, 777–789. [Google Scholar] [CrossRef] [PubMed]
- Venios, X.; Korkas, E.; Nisiotou, A.; Banilas, G. Grapevine Responses to Heat Stress and Global Warming. Plants 2020, 9, 1754. [Google Scholar] [CrossRef]
- Nogales, A.; Rottier, E.; Campos, C.; Victorino, G.; Costa, J.M.; Coito, J.L.; Pereira, H.S.; Viegas, W.; Lopes, C. The Effects of Field Inoculation of Arbuscular Mycorrhizal Fungi through Rye Donor Plants on Grapevine Performance and Soil Properties. Agric. Ecosyst. Environ. 2021, 313, 107369. [Google Scholar] [CrossRef]
- Torres, N.; Antolín, M.C.; Goicoechea, N. Arbuscular Mycorrhizal Symbiosis as a Promising Resource for Improving Berry Quality in Grapevines Under Changing Environments. Front. Plant Sci. 2018, 9, 897. [Google Scholar] [CrossRef] [Green Version]
- Nogales, A.; Ribeiro, H.; Nogales-Bueno, J.; Hansen, L.D.; Gonçalves, E.F.; Coito, J.L.; Rato, A.E.; Peixe, A.; Viegas, W.; Cardoso, H. Response of Mycorrhizal ’Touriga Nacional‘ Variety Grapevines to High Temperatures Measured by Calorespirometry and Near-Infrared Spectroscopy. Plants 2020, 9, 1499. [Google Scholar] [CrossRef]
- Campos, C.; Cardoso, H.; Nogales, A.; Svensson, J.; Lopez-Ráez, J.A.; Pozo, M.J.; Nobre, T.; Schneider, C.; Arnholdt-Schmitt, B. Intra and Inter-Spore Variability in Rhizophagus Irregularis AOX Gene. PLoS ONE 2015, 10, e0142339. [Google Scholar] [CrossRef] [Green Version]
- Amiri, R.; Nikbakht, A.; Rahimmalek, M.; Hosseini, H. Variation in the Essential Oil Composition, Antioxidant Capacity, and Physiological Characteristics of Pelargonium Graveolens L. Inoculated with Two Species of Mycorrhizal Fungi Under Water Deficit Conditions. J. Plant Growth Regul. 2017, 36, 502–515. [Google Scholar] [CrossRef]
- Peñuelas, J.; Munné-Bosch, S.; Llusià, J.; Filella, I. Leaf Reflectance and Photo- and Antioxidant Protection in Field-Grown Summer-Stressed Phillyrea Angustifolia. Optical Signals of Oxidative Stress? New Phytol. 2004, 162, 115–124. [Google Scholar] [CrossRef] [Green Version]
- De Cannière, S.; Vereecken, H.; Defourny, P.; Jonard, F. Remote Sensing of Instantaneous Drought Stress at Canopy Level Using Sun-Induced Chlorophyll Fluorescence and Canopy Reflectance. Remote Sens. 2022, 14, 2642. [Google Scholar] [CrossRef]
- Tavares, C.J.; Ribeiro Junior, W.Q.; Ramos, M.L.G.; Pereira, L.F.; Casari, R.A.d.C.N.; Pereira, A.F.; de Sousa, C.A.F.; da Silva, A.R.; Neto, S.P.d.S.; Mertz-Henning, L.M. Water Stress Alters Morphophysiological, Grain Quality and Vegetation Indices of Soybean Cultivars. Plants 2022, 11, 559. [Google Scholar] [CrossRef] [PubMed]
- Axtell, M.J.; Meyers, B.C. Revisiting Criteria for Plant MicroRNA Annotation in the Era of Big Data. Plant Cell 2018, 30, 272–284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chitarra, W.; Pagliarani, C.; Abbà, S.; Boccacci, P.; Birello, G.; Rossi, M.; Palmano, S.; Marzachì, C.; Perrone, I.; Gambino, G. MiRVIT: A Novel MiRNA Database and Its Application to Uncover Vitis Responses to Flavescence Dorée Infection. Front. Plant Sci. 2018, 9, 1034. [Google Scholar] [CrossRef] [PubMed]
- Pantaleo, V.; Szittya, G.; Moxon, S.; Miozzi, L.; Moulton, V.; Dalmay, T.; Burgyan, J. Identification of Grapevine MicroRNAs and Their Targets Using High-Throughput Sequencing and Degradome Analysis: Grapevine MicroRNAs and Their Targets. Plant J. 2010, 62, 960–976. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Yang, Y.; Shi, H.; Wang, Y.; Ren, F. Small RNA and Degradome Deep Sequencing Reveal Respective Roles of Cold-Related MicroRNAs across Chinese Wild Grapevine and Cultivated Grapevine. BMC Genom. 2019, 20, 740. [Google Scholar] [CrossRef] [Green Version]
- Urban, J.; Ingwers, M.W.; McGuire, M.A.; Teskey, R.O. Increase in Leaf Temperature Opens Stomata and Decouples Net Photosynthesis from Stomatal Conductance in Pinus Taeda and Populus Deltoides x Nigra. J. Exp. Bot. 2017, 68, 1757–1767. [Google Scholar] [CrossRef] [Green Version]
- Duc, N.H.; Csintalan, Z.; Posta, K. Arbuscular Mycorrhizal Fungi Mitigate Negative Effects of Combined Drought and Heat Stress on Tomato Plants. Plant Physiol. Biochem. 2018, 132, 297–307. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, D.; Liu, G.; Hussain, S.; Teng, Y. Influence of Heat Stress on Leaf Ultrastructure, Photosynthetic Performance, and Ascorbate Peroxidase Gene Expression of Two Pear Cultivars (Pyrus Pyrifolia). J. Zhejiang Univ. Sci. B 2013, 14, 1070–1083. [Google Scholar] [CrossRef] [Green Version]
- Feng, B.; Liu, P.; Li, G.; Dong, S.T.; Wang, F.H.; Kong, L.A.; Zhang, J.W. Effect of Heat Stress on the Photosynthetic Characteristics in Flag Leaves at the Grain-Filling Stage of Different Heat-Resistant Winter Wheat Varieties. J. Agron. Crop. Sci. 2014, 200, 143–155. [Google Scholar] [CrossRef]
- O’Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front. Endocrinol. 2018, 9, 402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Budak, H.; Akpinar, B.A. Plant MiRNAs: Biogenesis, Organization and Origins. Funct. Integr. Genom. 2015, 15, 523–531. [Google Scholar] [CrossRef]
- Fard, E.M.; Moradi, S.; Salekdeh, N.N.; Bakhshi, B.; Ghaffari, M.R.; Zeinalabedini, M.; Salekdeh, G.H. Plant IsomiRs: Origins, Biogenesis, and Biological Functions. Genomics 2020, 112, 3382–3395. [Google Scholar] [CrossRef] [PubMed]
- Baev, V.; Milev, I.; Naydenov, M.; Vachev, T.; Apostolova, E.; Mehterov, N.; Gozmanva, M.; Minkov, G.; Sablok, G.; Yahubyan, G. Insight into Small RNA Abundance and Expression in High- and Low-Temperature Stress Response Using Deep Sequencing in Arabidopsis. Plant Physiol. Biochem. 2014, 84, 105–114. [Google Scholar] [CrossRef]
- Perdiguero, P.; Rodrigues, A.S.; Chaves, I.; Costa, B.; Alves, A.; María, N.; Vélez, M.D.; Díaz-Sala, C.; Cervera, M.T.; Miguel, C.M. Comprehensive Analysis of the IsomiRome in the Vegetative Organs of the Conifer Pinus Pinaster under Contrasting Water Availability. Plant Cell Environ. 2021, 44, 706–728. [Google Scholar] [CrossRef] [PubMed]
- Sablok, G.; Srivastva, A.K.; Suprasanna, P.; Baev, V.; Ralph, P.J. IsomiRs: Increasing Evidences of IsomiRs Complexity in Plant Stress Functional Biology. Front. Plant Sci. 2015, 6, 949. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balyan, S.; Joseph, S.V.; Jain, R.; Mutum, R.D.; Raghuvanshi, S. Investigation into the MiRNA/5′ IsomiRNAs Function and Drought-Mediated MiRNA Processing in Rice. Funct. Integr. Genom. 2020, 20, 509–522. [Google Scholar] [CrossRef]
- Pandey, P.; Wang, M.; Baldwin, I.T.; Pandey, S.P.; Groten, K. Complex Regulation of MicroRNAs in Roots of Competitively-Grown Isogenic Nicotiana Attenuata Plants with Different Capacities to Interact with Arbuscular Mycorrhizal Fungi. BMC Genom. 2018, 19, 937. [Google Scholar] [CrossRef]
- Xu, Y.; Zhu, S.; Liu, F.; Wang, W.; Wang, X.; Han, G.; Cheng, B. Identification of Arbuscular Mycorrhiza Fungi Responsive MicroRNAs and Their Regulatory Network in Maize. Int. J. Mol. Sci. 2018, 19, 3201. [Google Scholar] [CrossRef] [Green Version]
- Pacak, A.; Barciszewska-Pacak, M.; Swida-Barteczka, A.; Kruszka, K.; Sega, P.; Milanowska, K.; Jakobsen, I.; Jarmolowski, A.; Szweykowska-Kulinska, Z. Heat Stress Affects Pi-Related Genes Expression and Inorganic Phosphate Deposition/Accumulation in Barley. Front. Plant Sci. 2016, 7, 926. [Google Scholar] [CrossRef] [Green Version]
- Szentpéteri, V.; Mayer, Z.; Posta, K. Mycorrhizal Symbiosis-Induced Abiotic Stress Mitigation through Phosphate Transporters in Solanum lycopersicum L. Plant Growth Regul. 2022. [Google Scholar] [CrossRef]
- Li, X.; Xie, X.; Li, J.; Cui, Y.; Hou, Y.; Zhai, L.; Wang, X.; Fu, Y.; Liu, R.; Bian, S. Conservation and Diversification of the MiR166 Family in Soybean and Potential Roles of Newly Identified MiR166s. BMC Plant Biol. 2017, 17, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ariel, F.D.; Manavella, P.A.; Dezar, C.A.; Chan, R.L. The True Story of the HD-Zip Family. Trends Plant Sci. 2007, 12, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Ribichich, K.F.; Chiozza, M.; Ávalos-Britez, S.; Cabello, J.V.; Arce, A.L.; Watson, G.; Arias, C.; Portapila, M.; Trucco, F.; Otegui, M.E.; et al. Successful Field Performance in Warm and Dry Environments of Soybean Expressing the Sunflower Transcription Factor HB4. J. Exp. Bot. 2020, 71, 3142–3156. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Zhuang, L.; Zhang, J.; Yu, J.; Yang, Z.; Huang, B. Identification and Characterization of Novel Homeodomain Leucine Zipper (HD-Zip) Transcription Factors Associated with Heat Tolerance in Perennial Ryegrass. Environ. Exp. Bot. 2019, 160, 1–11. [Google Scholar] [CrossRef]
- Kumar, R.R.; Pathak, H.; Sharma, S.K.; Kala, Y.K.; Nirjal, M.K.; Singh, G.P.; Goswami, S.; Rai, R.D. Novel and Conserved Heat-Responsive MicroRNAs in Wheat (Triticum aestivum L.). Funct. Integr. Genom. 2015, 15, 323–348. [Google Scholar] [CrossRef]
- Harberd, N.P.; King, K.E.; Carol, P.; Cowling, R.J.; Peng, J.; Richards, D.E. Gibberellin: Inhibitor of an Inhibitor Of...? BioEssays 1998, 20, 1001–1008. [Google Scholar] [CrossRef]
- Bazin, J.; Khan, G.A.; Combier, J.-P.; Bustos-Sanmamed, P.; Debernardi, J.M.; Rodriguez, R.; Sorin, C.; Palatnik, J.; Hartmann, C.; Crespi, M.; et al. MiR396 Affects Mycorrhization and Root Meristem Activity in the Legume Medicago Truncatula. Plant J. 2013, 74, 920–934. [Google Scholar] [CrossRef]
- Liebsch, D.; Palatnik, J.F. MicroRNA MiR396, GRF Transcription Factors and GIF Co-Regulators: A Conserved Plant Growth Regulatory Module with Potential for Breeding and Biotechnology. Curr. Opin. Plant Biol. 2020, 53, 31–42. [Google Scholar] [CrossRef]
- Zhao, Y.; Xie, J.; Wang, S.; Xu, W.; Chen, S.; Song, X.; Lu, M.; El-Kassaby, Y.A.; Zhang, D. Synonymous Mutation in Growth Regulating Factor 15 of MiR396a Target Sites Enhances Photosynthetic Efficiency and Heat Tolerance in Poplar. J. Exp. Bot. 2021, 72, 4502–4519. [Google Scholar] [CrossRef]
- Shahnejat-Bushehri, S.; Mueller-Roeber, B.; Balazadeh, S. Arabidopsis NAC Transcription Factor JUNGBRUNNEN1 Affects Thermomemory-Associated Genes and Enhances Heat Stress Tolerance in Primed and Unprimed Conditions. Null 2012, 7, 1518–1521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, C.; Ding, Y.; Liu, H. MiR398 and Plant Stress Responses. Physiol. Plant 2011, 143, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Yu, X.; Ottosen, C.-O.; Zhang, T.; Wu, Z.; Zhao, T. Unique MiRNAs and Their Targets in Tomato Leaf Responding to Combined Drought and Heat Stress. BMC Plant Biol. 2020, 20, 107. [Google Scholar] [CrossRef] [Green Version]
- Song, F.; He, C.; Yan, X.; Bai, F.; Pan, Z.; Deng, X.; Xiao, S. Small RNA Profiling Reveals Involvement of MicroRNA-Mediated Gene Regulation in Response to Mycorrhizal Symbiosis in Poncirus Trifoliata L. Raf. Tree Genet. Genomes 2018, 14, 42. [Google Scholar] [CrossRef]
- Li, Z.; Wu, N.; Meng, S.; Wu, F.; Liu, T. Arbuscular Mycorrhizal Fungi (AMF) Enhance the Tolerance of Euonymus Maackii Rupr. at a Moderate Level of Salinity. PLoS ONE 2020, 15, e0231497. [Google Scholar] [CrossRef]
- Wu, N.; Li, Z.; Wu, F.; Tang, M. Comparative Photochemistry Activity and Antioxidant Responses in Male and Female Populus Cathayana Cuttings Inoculated with Arbuscular Mycorrhizal Fungi under Salt. Sci. Rep. 2016, 6, 37663. [Google Scholar] [CrossRef]
- Yeasmin, R.; Bonser, S.P.; Motoki, S.; Nishihara, E. Arbuscular Mycorrhiza Influences Growth and Nutrient Uptake of Asparagus (Asparagus officinalis L.) under Heat Stress. HortScience 2019, 54, 846–850. [Google Scholar] [CrossRef] [Green Version]
- Yang, H.; Zhao, Y.; Chen, N.; Liu, Y.; Yang, S.; Du, H.; Wang, W.; Wu, J.; Tai, F.; Chen, F.; et al. A New Adenylyl Cyclase, Putative Disease-Resistance RPP13-like Protein 3, Participates in Abscisic Acid-Mediated Resistance to Heat Stress in Maize. J. Exp. Bot. 2021, 72, 283–301. [Google Scholar] [CrossRef]
- Wang, L.; Sun, S.; Jin, J.; Fu, D.; Yang, X.; Weng, X.; Xu, C.; Li, X.; Xiao, J.; Zhang, Q. Coordinated Regulation of Vegetative and Reproductive Branching in Rice. Proc. Natl. Acad. Sci. USA 2015, 112, 15504–15509. [Google Scholar] [CrossRef] [Green Version]
- Xu, M.; Hu, T.; Zhao, J.; Park, M.-Y.; Earley, K.W.; Wu, G.; Yang, L.; Poethig, R.S. Developmental Functions of MiR156-Regulated SQUAMOSA PROMOTER BINDING PROTEIN-LIKE (SPL) Genes in Arabidopsis Thaliana. PLoS Genet. 2016, 12, e1006263. [Google Scholar] [CrossRef] [Green Version]
- Sun, X.; Fan, G.; Su, L.; Wang, W.; Liang, Z.; Li, S.; Xin, H. Identification of Cold-Inducible MicroRNAs in Grapevine. Front. Plant Sci. 2015, 6, 595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Q.; Luo, L.; Zheng, L. Lignins: Biosynthesis and Biological Functions in Plants. Int. J. Mol. Sci. 2018, 19, 335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tisserant, E.; Malbreil, M.; Kuo, A.; Kohler, A.; Symeonidi, A.; Balestrini, R.; Charron, P.; Duensing, N.; Frei dit Frey, N.; Gianinazzi-Pearson, V.; et al. Genome of an Arbuscular Mycorrhizal Fungus Provides Insight into the Oldest Plant Symbiosis. Proc. Natl. Acad. Sci. USA 2013, 110, 20117–20122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, E.; Sharma, R.; Borah, P.; Jain, M.; Khurana, J.P. Emerging Roles of Auxin in Abiotic Stress Responses. In Elucidation of Abiotic Stress Signaling in Plants: Functional Genomics Perspectives, Volume 1; Pandey, G.K., Ed.; Springer: New York, NY, USA, 2015; pp. 299–328. ISBN 978-1-4939-2211-6. [Google Scholar]
- Tromas, A.; Paque, S.; Stierlé, V.; Quettier, A.-L.; Muller, P.; Lechner, E.; Genschik, P.; Perrot-Rechenmann, C. Auxin-Binding Protein 1 Is a Negative Regulator of the SCFTIR1/AFB Pathway. Nat. Commun. 2013, 4, 2496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones-Rhoades, M.W.; Bartel, D.P. Computational Identification of Plant MicroRNAs and Their Targets, Including a Stress-Induced MiRNA. Mol. Cell 2004, 14, 787–799. [Google Scholar] [CrossRef]
- Zhou, S.-M.; Kong, X.-Z.; Kang, H.-H.; Sun, X.-D.; Wang, W. The Involvement of Wheat F-Box Protein Gene TaFBA1 in the Oxidative Stress Tolerance of Plants. PLoS ONE 2015, 10, e0122117. [Google Scholar] [CrossRef] [Green Version]
- Hernandez, Y.; Goswami, K.; Sanan-Mishra, N. Stress Induced Dynamic Adjustment of Conserved MiR164:NAC Module. Plant-Environ. Interact. 2020, 1, 134–151. [Google Scholar] [CrossRef]
- Magalhães, N. Tratado de Viticultura: A Videira, A Vinha e o “Terroir”, 2nd ed.; Esfera Poética: Lisboa, Portugal, 2015; ISBN 978-989-98207-3-9. [Google Scholar]
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method for Growing Plants without Soil; California Agricultural Experiment Station: Berkeley, CA, USA, 1950. [Google Scholar]
- Peñuelas, J.; Filella, I.; Gamon, J.A. Assessment of Photosynthetic Radiation-Use Efficiency with Spectral Reflectance. New Phytol. 1995, 131, 291–296. [Google Scholar] [CrossRef]
- Phillips, J.M.; Hayman, D.S. Improved Procedures for Clearing Roots and Staining Parasitic and Vesicular-Arbuscular Mycorrhizal Fungi for Rapid Assessment of Infection. Trans. Br. Mycol. Soc. 1970, 55, 158–161, IN16–IN18. [Google Scholar] [CrossRef]
- Giovannetti, M.; Mosse, B. AN EVALUATION OF TECHNIQUES FOR MEASURING VESICULAR ARBUSCULAR MYCORRHIZAL INFECTION IN ROOTS. New Phytol. 1980, 84, 489–500. [Google Scholar] [CrossRef]
- Stocks, M.B.; Mohorianu, I.; Beckers, M.; Paicu, C.; Moxon, S.; Thody, J.; Dalmay, T.; Moulton, V. The UEA SRNA Workbench (Version 4.4): A Comprehensive Suite of Tools for Analyzing MiRNAs and SRNAs. Bioinformatics 2018, 34, 3382–3384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, B.H.; Pan, X.P.; Cox, S.B.; Cobb, G.P.; Anderson, T.A. Evidence That MiRNAs Are Different from Other RNAs. Cell. Mol. Life Sci. 2006, 63, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Zhuang, Z.; Zhao, P.X. PsRNATarget: A Plant Small RNA Target Analysis Server (2017 Release). Nucleic Acids Res. 2018, 46, W49–W54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szklarczyk, D.; Morris, J.H.; Cook, H.; Kuhn, M.; Wyder, S.; Simonovic, M.; Santos, A.; Doncheva, N.T.; Roth, A.; Bork, P.; et al. The STRING Database in 2017: Quality-Controlled Protein-Protein Association Networks, Made Broadly Accessible. Nucleic Acids Res. 2017, 45, D362–D368. [Google Scholar] [CrossRef] [PubMed]
- Campos, C.; Carvalho, M.; Brígido, C.; Goss, M.J.; Nobre, T. Symbiosis Specificity of the Preceding Host Plant Can Dominate but Not Obliterate the Association Between Wheat and Its Arbuscular Mycorrhizal Fungal Partners. Front. Microbiol. 2018, 9, 2920. [Google Scholar] [CrossRef] [PubMed]
- Vandesompele, J.; De Preter, K.; Pattyn, F.; Poppe, B.; Van Roy, N.; De Paepe, A.; Speleman, F. Accurate Normalization of Real-Time Quantitative RT-PCR Data by Geometric Averaging of Multiple Internal Control Genes. Genome Biol. 2002, 3, research0034.1. [Google Scholar] [CrossRef] [Green Version]





| Factor | Pn | gs | E | WI | CHLI | NDVI | PRI |
|---|---|---|---|---|---|---|---|
| AMF inoculation | 0.015 * | 0.279 | 0.320 | 0.777 | 0.032 * | 0.485 | 0.222 |
| Temperature | 0.269 | 0.274 | 0.001 * | <0.001 * | 0.133 | 0.036 * | <0.001 * |
| Interaction | 0.990 | 0.713 | 0.658 | 0.699 | 0.347 | 0.715 | 0.707 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campos, C.; Coito, J.L.; Cardoso, H.; Marques da Silva, J.; Pereira, H.S.; Viegas, W.; Nogales, A. Dynamic Regulation of Grapevine’s microRNAs in Response to Mycorrhizal Symbiosis and High Temperature. Plants 2023, 12, 982. https://doi.org/10.3390/plants12050982
Campos C, Coito JL, Cardoso H, Marques da Silva J, Pereira HS, Viegas W, Nogales A. Dynamic Regulation of Grapevine’s microRNAs in Response to Mycorrhizal Symbiosis and High Temperature. Plants. 2023; 12(5):982. https://doi.org/10.3390/plants12050982
Chicago/Turabian StyleCampos, Catarina, João Lucas Coito, Hélia Cardoso, Jorge Marques da Silva, Helena Sofia Pereira, Wanda Viegas, and Amaia Nogales. 2023. "Dynamic Regulation of Grapevine’s microRNAs in Response to Mycorrhizal Symbiosis and High Temperature" Plants 12, no. 5: 982. https://doi.org/10.3390/plants12050982






