Assessment of Cassava Pollen Viability and Ovule Fertilizability under Red-Light, 6-Benzyl Adenine, and Silver Thiosulphate Treatments
Abstract
:1. Introduction
2. Results
2.1. Effects of RL and PGR Treatments on Pollen Characteristics and Their Capacity to Improve Fruit and Seed Set
2.2. Effect of RL and PGR Treatments on Pollen Stainability and Diameter
2.3. Effect of Exposing Flowers to RL on Ovule Fertilizability and Fruit and Seed Set Efficiency
2.4. Effect of Treating Flowers with PGR on Ovule Fertilizability and Fruit Set and Seed Set Efficiency
2.5. Comparison of Effects of RL and PGR Treatments on Pollen Viability and Fruit and Seed Set Efficiency
2.6. Pollen Ploidy and Its Relationship with Diameter and Viability in RL-Treated Flowers
3. Discussion
3.1. Effect of Extended Daylength with RL on Pollen Diameter, Stainability, and Ovule Fertilizability
3.2. Effect of PGR on Pollen Diameter, Stainability, and Ovule Fertilizability
3.3. Relationship between Ploidy, Diameter, and Viability of Pollen in RL-Treated Flowers
4. Materials and Methods
4.1. Plant Materials, Field Conditions and Treatments
4.2. Pollen Collection
4.3. Assessment of Pollen Germination, Stainability, and Measurement of Grain Diameter
4.3.1. Pollination
4.3.2. In Vivo Pollen Germination and Ovule Fertilization
4.3.3. In Vitro Pollen Stainability
4.3.4. Estimation of Pollen Grain Diameter
4.4. Assessment of Fruit and Seed Set Efficiency
4.5. Determination of Pollen Ploidy Level
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alves, A.A.C. Cassava botany and phyiology. In Cassava: Biology, Production and Utilization; Hillocks, R.J., Thresh, J.M., Bellotti, A.C., Eds.; CABI: Wallingford, UK, 2002; pp. 67–89. [Google Scholar]
- Fathima, A.A.; Sanitha, M.; Tripathi, L.; Muiruri, S. Cassava (Manihot esculenta) dual use for food and bioenergy: A review. Food Energy Secur. 2022, 12, e380. [Google Scholar] [CrossRef]
- Amelework, A.B.; Bairu, M.W.; Maema, O.; Venter, S.L.; Laing, M. Adoption and promotion of resilient crops for climate risk mitigation and import substitution: A case analysis of cassava for South African agriculture. Front. Sustain. Food Syst. 2021, 5, 617783. [Google Scholar] [CrossRef]
- Pushpalatha, R.; Gangadharan, B. Is cassava (Manihot esculenta Crantz) a climate “smart” crop? A review in the context of bridging future food demand gap. Trop. Plant Biol. 2020, 13, 201–211. [Google Scholar] [CrossRef]
- Jarvis, A.; Ramirez-Villegas, J.; Herrera Campo, B.V.; Navarro-Racines, C. Is cassava the answer to African climate change adaptation? Trop. Plant Biol. 2012, 5, 9–29. [Google Scholar] [CrossRef]
- Bandeira e Sousa, M.; Andrade, L.R.B.; Souza, E.H.; Alves, A.A.C.; de Oliveira, E.J. Reproductive barriers in cassava: Factors and implications for genetic improvement. PLoS ONE 2021, 16, e0260576. [Google Scholar] [CrossRef]
- Ndubuisi, N.D.; Nathaniel, I.U.; Favour, E.; Chiedozie, E. Crossability and germinability potentials of some elite cassava progenitors. J. Plant Breed. Crop Sci. 2015, 7, 61–66. [Google Scholar] [CrossRef]
- Stephenson, A.G. Flower and fruit abortion: Proximate causes and ultimate functions. Annu. Rev. Ecol. Evol. Syst. 1981, 12, 253–279. [Google Scholar] [CrossRef]
- Palupi, E.R.; Owens, J.N.; Sadjad, S.; Solihin, D.D. Importance of fruit set, fruit abortion and pollination success in teak. Can. J. For. Res. 2010, 40, 2204–2214. [Google Scholar] [CrossRef]
- Rathod, V.; Behera, T.K.; Munshi, A.D.; Durgesh, K.; Jat, G.S.; Krishnan, B.; Sharma, N. Pollen viability and in vitro pollen germination studies in Momordica species and their intra and interspecific hybrids. Int. J. Chem. Stud. 2018, 6, 32–40. [Google Scholar]
- Lankinen, A.; Lindstrom, S.A.M.; D’Hertefeldt, T. Variable pollen viability and effects of pollen load size on components of seed set in cultivars and feral populations of oilseed rape. PLoS ONE 2018, 13, e0204407. [Google Scholar] [CrossRef]
- He, G.; Hu, F.; Ming, J.; Liu, C.; Yuan, S. Pollen viability and stigma receptivity in Lilium during anthesis. Euphytica 2017, 213, 231. [Google Scholar] [CrossRef]
- Impe, D.; Reitz, J.; Kopnick, C.; Rolletschek, H.; Borner, A.; Senula, A.; Nagel, M. Assessment of pollen viability for wheat. Front. Plant Sci. 2019, 10, 1588. [Google Scholar] [CrossRef] [PubMed]
- Younis, A.; Hwang, Y.J.; Lim, K.B. Exploitation of induced 2n-gametes for plant breeding. Plant Cell Rep. 2014, 33, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Farinati, S.; Draga, S.; Betto, A.; Palumbo, F.; Vannozzi, A.; Lucchin, M.; Barcaccia, G. Current insights and advances into plant male sterility: New precision breeding technology based on genome editing applications. Front. Plant Sci. 2023, 14, 1223861. [Google Scholar] [CrossRef] [PubMed]
- Gökbayrak, Z.; Engin, H. Effects of foliar-applied brassinosteroid on viability and in vitro germination of pollen collected from bisexual and functional male flowers of pomegranate. Int. J. Fruit Sci. 2018, 18, 226–230. [Google Scholar] [CrossRef]
- Dafni, A.; Firmage, D. Pollen viability and longevity. Plant Syst. Evol. 2000, 222, 113–132. [Google Scholar] [CrossRef]
- Ottaviano, E.; Mulcahy, D.L. Genetics of Angiosperm Pollen. Adv. Genet. 1989, 26, 1–64. [Google Scholar] [CrossRef]
- Akçal, A.; Gokbayrak, Z.; Engin, H. Determination of the effects of growth regulators on pollen viability and germination level of tulip. Acta Hortic. 2019, 1242, 549–552. [Google Scholar] [CrossRef]
- Fan, X.; Yang, Y.; Xu, Z. Effects of different ratio of red and blue light on flowering and fruiting of tomato. In Proceedings of the 4th International Conference on Agricultural and Food Science, Istanbul Turkey, 28–30 October 2021; IOP Publishing: Bristol, UK, 2021. [Google Scholar]
- Jha, C.V.; Patel, N.B.; Patel, V.A. Effects of the lights of different spectral composition on in vitro pollen germination and tube growth in Peltophorum. LSL 2011, 12, 396–402. [Google Scholar]
- Baguma, J.K.; Mukasa, S.B.; Nuwamanya, E.; Alicai, T.; Omongo, C.; Hyde, P.T.; Setter, T.L.; Ochwo-Ssemakula, M.; Esuma, W.; Kanaabi, M.; et al. Flowering and fruit-set in cassava under extended red-light photoperiod supplemented with plant-growth regulators and pruning. BMC Plant Biol. 2023, 23, 335. [Google Scholar] [CrossRef]
- Zebrowska, J. Factors affecting pollen grain viability in the strawberry (Fragaria × ananassa Duch.). J. Hortic. Sci. 2015, 72, 213–219. [Google Scholar] [CrossRef]
- Kelly, J.K.; Rasch, A.; Kalisz, S. A method to estimate pollen viability from pollen size variation. Am. J. Bot. 2002, 89, 1021–1023. [Google Scholar] [CrossRef] [PubMed]
- Czarnecki II, D.M.; Hershberger, A.J.; Robacker, C.D.; Clark, D.G.; Deng, Z. Ploidy levels and pollen stainability of Lantana camara cultivars and breeding lines. HortScience 2014, 49, 1271–1276. [Google Scholar] [CrossRef]
- Tomaszewska, P.; Kosina, R. Variability in the quality of pollen grains in oat amphiploids and their parental species. Braz. J. Bot. 2022, 45, 987–1000. [Google Scholar] [CrossRef]
- Peterson, R.; Slovin, J.P.; Chen, C. A simplified method for differential staining of aborted and non-aborted pollen grains. Int. J. Plant Biol. 2010, 1, e13. [Google Scholar] [CrossRef]
- Harshitha, H.; Chandrashekar, S.; Harishkumar, K.; Pradeep, K.C. Photoperiod manipulation in flowers and ornamentals for perpetual flowering. Pharma Innov. J. 2021, 10, 127–134. [Google Scholar]
- Demotes-Mainard, S.; Péron, T.; Corot, A.; Bertheloot, J.; Le Gourrierec, J.; Pelleschi-Travier, S.; Crespel, L.; Morel, P.; Huché-Thélier, L.; Boumaza, R.; et al. Plant responses to red and far-red lights, applications in horticulture. Environ. Exp. Bot. 2016, 121, 4–21. [Google Scholar] [CrossRef]
- Dhawan, A.K.; Malik, C.P. Effect of growth regulators and light on pollen germination and pollen tube growth in Pinus roxburghii Sarg. Ann. Bot. 1981, 47, 239–248. [Google Scholar] [CrossRef]
- Kimball, J.W. Photoperiodism and phytochrome. In Biology; LibreTexts: Berkeley, CA, USA, 2020. [Google Scholar]
- Tripathi, S.; Hoang, Q.T.N.; Han, Y.J.; Kim, J.I. Regulation of photomorphogenic development by plant phytochromes. Int. J. Mol. Sci. 2019, 20, 6165. [Google Scholar] [CrossRef]
- Wu, F.; Hanzawa, Y. Photoperiodic control of flowering in plants. In Handbook of Plant and Crop Physiology; CRC Press: Boca Raton, FL, USA, 2014; pp. 33–62. [Google Scholar] [CrossRef]
- Li, J.; Li, G.; Wang, H.; Deng, X.W. Phytochrome signaling mechanisms. Arab. Book 2011, 9, e0148. [Google Scholar] [CrossRef]
- Elena, B. Aspects of the pollen grains diameter variability and the pollen viability to some sunflower genotypes. J. Hortic. For. Biotechnol. 2013, 17, 161–165. [Google Scholar]
- Roulston, T.a.H.; Cane, J.H.; Buchmann, S.L. What governs protein content of pollen: Pollinator preferences, pollen–pistil interactions, or phylogeny? Ecol. Monogr. 2000, 70, 617–643. [Google Scholar] [CrossRef]
- Hyde, P.T.; Setter, T.L. Long-day photoperiod and cool temperature induce flowering in cassava: Expression of signaling genes. Front. Plant Sci. 2022, 13, 973206. [Google Scholar] [CrossRef] [PubMed]
- Dias, J.P.T. Plant growth regulators in horticulture: Practices and perspectives. Biotecnol. Veg. 2019, 19, 3–14. [Google Scholar]
- Aliyu, O.M.; Adeigbe, O.O.; Awopetu, J.A. Foliar application of the exogenous plant hormones at pre-blooming stage improves flowering and fruiting in cashew (Anacardium occidentale L.). J. Crop Sci. Biotechnol. 2011, 14, 143–150. [Google Scholar] [CrossRef]
- Oluwasanya, D.; Esan, O.; Hyde, P.T.; Kulakow, P.; Setter, T.L. Flower development in cassava is feminized by cytokinin, while proliferation is stimulated by anti-ethylene and pruning: Transcriptome responses. Front. Plant Sci. 2021, 12, 666266. [Google Scholar] [CrossRef] [PubMed]
- Hyde, P.T.; Guan, X.; Abreu, V.; Setter, T.L. The anti-ethylene growth regulator silver thiosulfate (STS) increases flower production and longevity in cassava (Manihot esculenta Crantz). Plant Growth Regul. 2020, 90, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Chhun, T.; Aya, K.; Asano, K.; Yamamoto, E.; Morinaka, Y.; Watanabe, M.; Kitano, H.; Ashikari, M.; Matsuoka, M.; Ueguchi-Tanaka, M. Gibberellin regulates pollen viability and pollen tube growth in rice. Plant Cell 2007, 19, 3876–3888. [Google Scholar] [CrossRef] [PubMed]
- Terceros, G.C.; Resentini, F.; Cucinotta, M.; Manrique, S.; Colombo, L.; Mendes, M.A. The importance of cytokinins during reproductive development in Arabidopsis and beyond. Int. J. Mol. Sci. 2020, 21, 8161. [Google Scholar] [CrossRef]
- Emery, R.J.N.; Kisiala, A. The roles of cytokinins in plants and their response to environmental stimuli. Plants 2020, 9, 1158. [Google Scholar] [CrossRef]
- Bairwa, S.; Mishra, J.S. Effect of NAA, BA and kinetin on yield of african marigold (Tagetes erecta Linn.). Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 1236–1241. [Google Scholar] [CrossRef]
- Rosliani, R.; Palupi, E.R.; Hilman, Y. Benzyl amino purine and boron application for improving production and quality of true shallots seed (Allium cepa var. ascalonicum) in highlands. J. Hort. 2012, 22, 242–250. [Google Scholar]
- Owen, L.C.; Suchoff, D.H.; Chen, H. A novel method for stimulating Cannabis sativa L. male flowers from female plants. Plants 2023, 12, 3371. [Google Scholar] [CrossRef] [PubMed]
- DiMatteo, J.; Kurtz, L.; Lubell-Brand, J.D. Pollen appearance and in vitro germination varies for five strains of female hemp masculinized using silver thiosulfate. HortScience 2020, 55, 547–549. [Google Scholar] [CrossRef]
- Ram, M.H.Y.; Sett, R. Induction of fertile male flowers in genetically female Cannabis sativa plants by silver nitrate and silver thiosulphate anionic complex. Theor. Appl. Genet. 1982, 62, 369–375. [Google Scholar]
- Flajsman, M.; Slapnik, M.; Murovec, J. Production of feminized seeds of high CBD Cannabis sativa L. by manipulation of sex expression and its application to breeding. Front. Plant Sci. 2021, 12, 718092. [Google Scholar] [CrossRef] [PubMed]
- Veen, H. Silver thiosulphate: An experimrntal tool in plant science. Sci. Hortic. 1983, 20, 211–224. [Google Scholar] [CrossRef]
- Li, Q.; Chen, P.; Tang, H.; Zeng, F.; Li, X. Integrated transcriptome and hormone analyses provide insights into silver thiosulfate-induced “maleness” responses in the floral sex differentiation of pumpkin (Cucurbita moschata D.). Front. Genet. 2022, 13, 960027. [Google Scholar] [CrossRef]
- Gautam, P.; Terfa, M.T.; Olsen, J.E.; Torre, S. Red and blue light effects on morphology and flowering of Petunia × hybrida. Sci. Hortic. 2015, 184, 171–178. [Google Scholar] [CrossRef]
- Ulum, F.B.; Costa Castro, C.; Horandl, E. Ploidy-dependent effects of light stress on the mode of reproduction in the Ranunculus auricomus Complex (Ranunculaceae). Front. Plant Sci. 2020, 11, 104. [Google Scholar] [CrossRef]
- Joubès, J.; Chevalier, C. Endoreduplication in higher plants. In The Plant Cell Cycle; Springer: Berlin/Heidelberg, Germany, 2000; pp. 191–201. [Google Scholar] [CrossRef]
- Traas, J.; Hulskamp, M.; Gendreau, E.; Hofte, H. Endoreduplication and development: Rule without dividing? Curr. Opin. Plant Biol. 1998, 1, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Larkins, B.A.; Dilkes, B.P.; Dante, R.A.; Coelho, C.M.; Woo, Y.M.; Liu, Y. Investigating the hows and whys of DNA endoreduplication. J. Exp. Bot. 2001, 52, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.-H.; Liu, P.; Liu, M.-J. Advances in ploidy breeding of fruit trees. Acta Hortic. Sin. 2012, 39, 1639. [Google Scholar]
- Zhao, R.; Xu, L.; Xu, X.; Li, Y.; Xiao, S.; Yuan, D. Comparative study on pollen viability of Camellia oleifera at four ploidy levels. Agronomy 2022, 12, 2592. [Google Scholar] [CrossRef]
- Jan, F.; Schüler, L.; Behling, H. Trends of pollen grain size variation in C3 and C4 Poaceae species using pollen morphology for future assessment of grassland ecosystem dynamics. Grana 2014, 54, 129–145. [Google Scholar] [CrossRef]
- Rotreklová, O.; Krahulcová, A. Estimating paternal efficiency in an agamic polyploid complex: Pollen stainability and variation in pollen size related to reproduction mode, ploidy level and hybridogenous origin in Pilosella (Asteraceae). Folia Geobot. 2016, 51, 175–186. [Google Scholar] [CrossRef]
- Souza-Pérez, M.; Mourelle, D.; Trujillo, C.; Borges, A.; Speroni, G. Pollen grain performance in Psidium cattleyanum (Myrtaceae): A pseudogamous polyploid species. Flora 2021, 281, 151863. [Google Scholar] [CrossRef]
- Sanders, H. Polyploidy and pollen grain size: Is there a correlation? Grad. Rev. 2021, 1, 15. [Google Scholar]
- Alexander, L. Ploidy level influences pollen tube growth and seed viability in interploidy crosses of Hydrangea macrophylla. Front. Plant Sci. 2020, 11, 100. [Google Scholar] [CrossRef]
- Abubakar, M.; Setter, T.; Gibson, P.; Baguma, Y.; Edema, R.; Pariyo, A.; Hyde, P.; Mukasa, S. Effect of plant growth regulators on flowering and seed set in cassava (Manihot esculenta Crantz) in Uganda. In Proceedings of the World Congress on Root and Tuber Crops, GCP21-III and ISTRC, Nanning, China, 10–15 January 2016. [Google Scholar]
- Hyde, P.; Abreu, V.; Setter, T. Anti-ethylene growth regulator treatment enhances flower set in cassava. In Proceedings of the World Congress on Root and Tuber Crops, GCP21-III and ISTRC, Nanning, China, 10–15 January 2016. [Google Scholar]
- Baguma, J.K.; Mukasa, S.B.; Kawuki, R.; Tugume, A.K.; Buttibwa, M.; Nalela, P.; Eyokia, M.; Oshaba, B.; Ceballos, H.; Lentini, Z.; et al. Fruit set and plant regeneration in cassava following interspecific pollination with castor bean. Afr. Crop Sci. J. 2019, 27, 99–118. [Google Scholar] [CrossRef]
- Kron, P.; Husband, B.C. Using flow cytometry to estimate pollen DNA content: Improved methodology and applications. Ann. Bot. 2012, 110, 1067–1078. [Google Scholar] [CrossRef] [PubMed]
- R-Core-Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
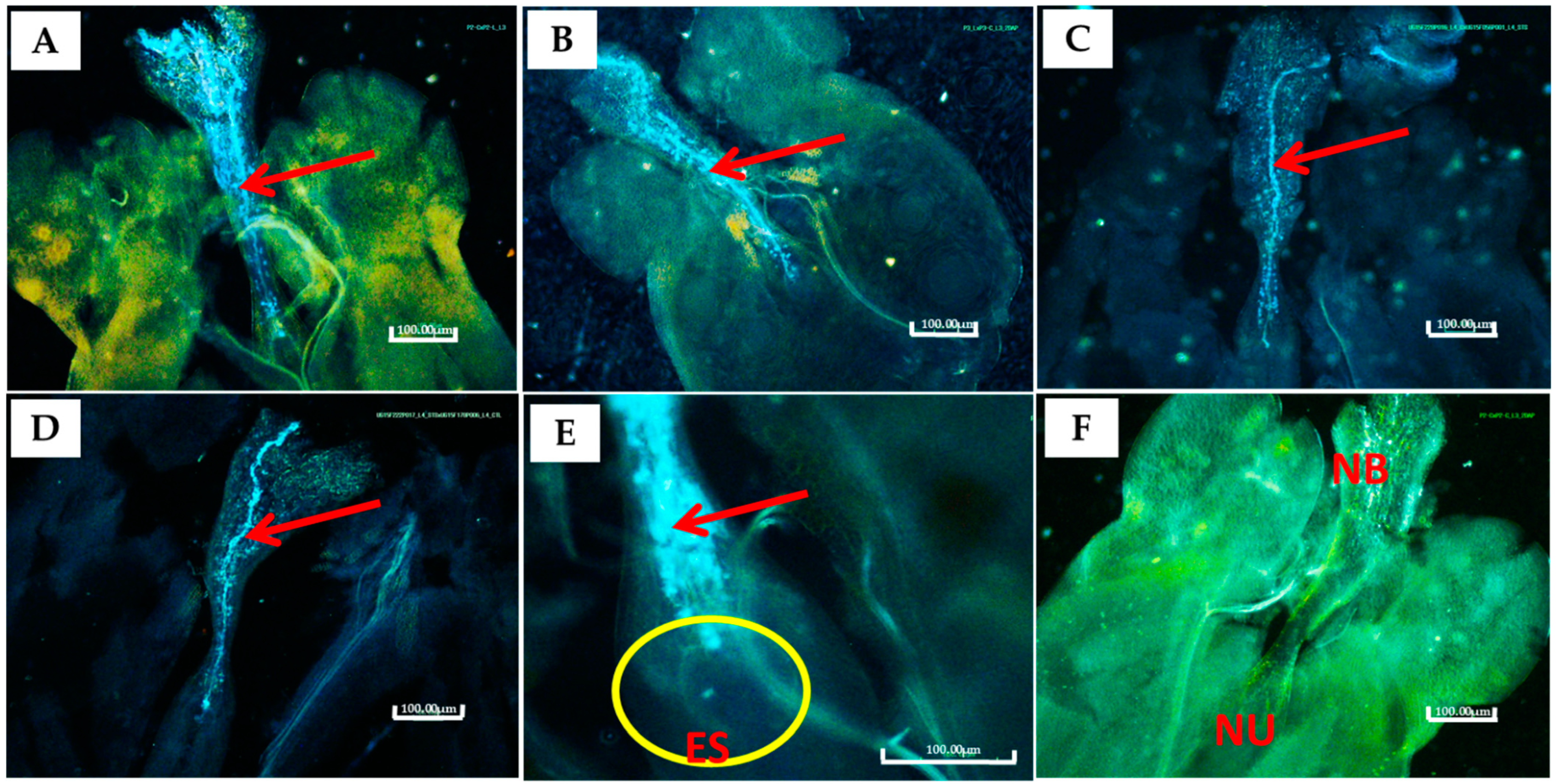



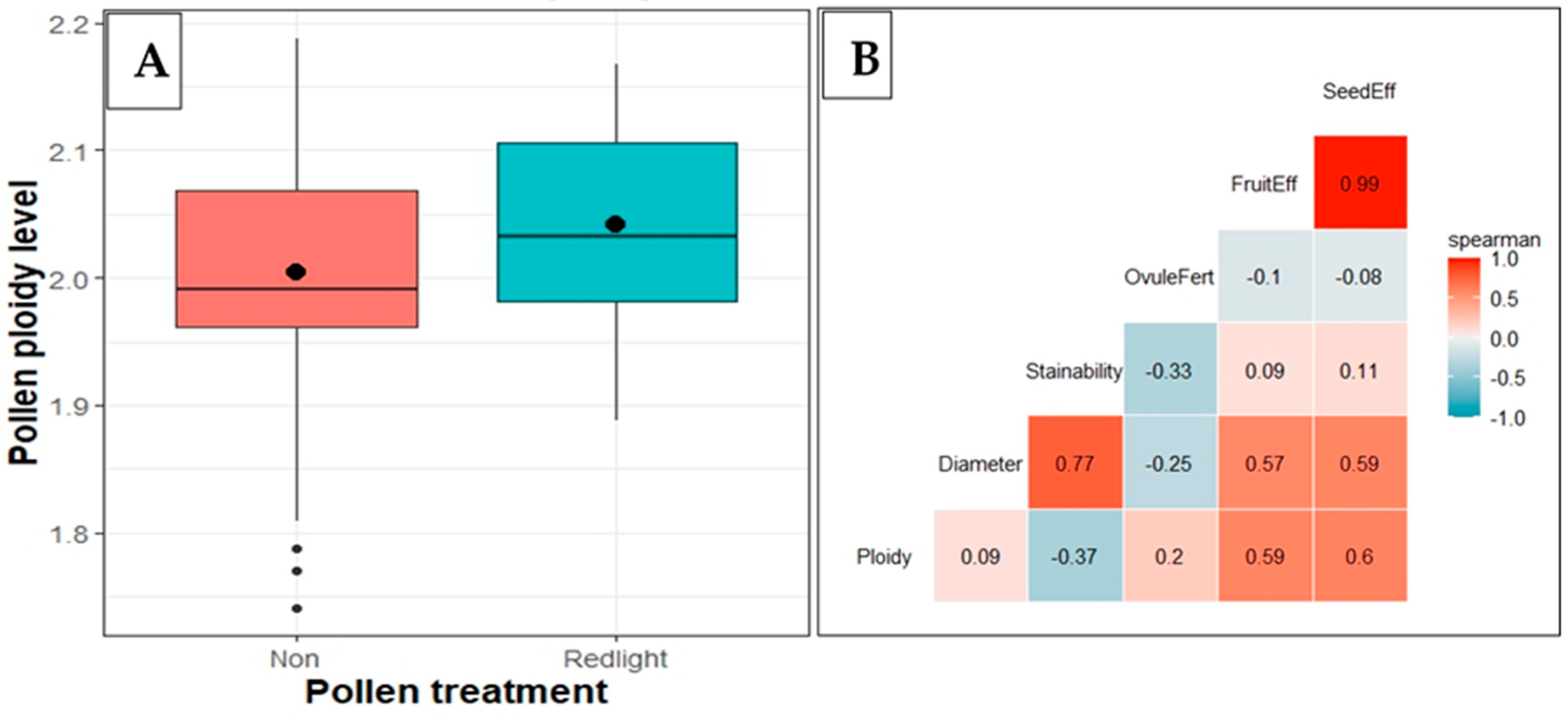


| Variable | Pollen Diameter (µm) | Pollen Stainability (%) | Ovules Fertilized (%) | Fruit Set Efficiency (%) | Seed Set Efficiency (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Treatment | RL | PGR | Ctl | RL | PGR | Ctl | RL | PGR | Ctl | RL | PGR | Ctl | RL | PGR | Ctl |
| Mean | 148.5 | 142.4 | 145.6 | 93.0 | 82.1 | 78.5 | 57.0 | 65.0 | 44.4 | 71.8 | 51.9 | 44.6 | 49.7 | 37.3 | 27.7 |
| SEM | 0.7 | 1.0 | 1.2 | 1.4 | 1.4 | 1.5 | 4.2 | 4.5 | 6.0 | 2.2 | 4.3 | 4.2 | 2.3 | 4.7 | 3.0 |
| CI | 1.4 | 1.9 | 2.4 | 2.7 | 2.8 | 2.9 | 8.3 | 8.9 | 12.2 | 4.4 | 8.8 | 9.2 | 4.6 | 9.5 | 6.7 |
| Variance | 83.5 | 150.7 | 191.9 | 305.5 | 318.7 | 284.3 | 1856.2 | 1381.9 | 1714.9 | 368.3 | 511.0 | 209.9 | 393.2 | 606.0 | 111.0 |
| Std | 9.1 | 12.3 | 13.9 | 17.5 | 17.9 | 16.9 | 43.1 | 37.2 | 41.4 | 19.2 | 22.6 | 14.5 | 19.8 | 24.6 | 10.5 |
| CV | 0.1 | 0.1 | 0.1 | 0.2 | 0.2 | 0.2 | 0.8 | 0.6 | 1.2 | 0.3 | 0.5 | 0.5 | 0.5 | 0.7 | 0.6 |
| Significance (α = 0.05) | ns | *** | *** | ns | ns | *** | *** | *** | *** | *** | |||||
| Treatment | Pollen Diameter (µm) | Pollen Stainability (%) |
|---|---|---|
| PGR | 142.4 b | 82.1 b |
| Red light | 148.5 a | 93.0 a |
| Control (Non) | 145.6 b | 78.5 b |
| Treatment | Ovules Fertilised (%) | Fruit Set Efficiency (%) | Seed Set Efficiency (%) | |||
|---|---|---|---|---|---|---|
| Female | Male | Female | Male | Female | Male | |
| Red light | 56.9 a | 64.3 a | 71.8 a | 71.1 a | 49.7 a | 51.8 a |
| Control | 52.0 b | 47.7 b | 51.5 b | 53.7 b | 31.7 b | 31.9 b |
| Significance (α = 0.05) | *** | *** | *** | *** | *** | *** |
| Genotype | Ovules Fertilised (%) | Fruit Set Efficiency (%) | Seed Set Efficiency (%) | |||
|---|---|---|---|---|---|---|
| Female | Male | Female | Male | Female | Male | |
| UG15F178P001 | 65.1 a | 66.7 a | 54.7 b | 58.1 c | 37.0 c | 41.2 a |
| UG15F180P005 | 25.4 c | 53.0 b | 53.8 b | 59.5 bc | 30.1 d | 39.4 ab |
| UG15F192P012 | 57.1 b | 43.3 c | 63.9 a | 65.9 a | 49.6 a | 40.7 a |
| UG15F302P016 | 62.2 a | 56.9 b | 64.8 a | 60.1 b | 40.5 b | 38.4 b |
| Significance (α = 0.05) | ns | *** | *** | *** | *** | *** |
| Treatment | Ovules Fertilized (%) | Fruit Set Efficiency (%) | Seed Set Efficiency (%) | |||
|---|---|---|---|---|---|---|
| Female | Male | Female | Male | Female | Male | |
| PGR | 64.9 a | 60.9 a | 51.9 a | 54.7 a | 37.3 a | 40.4 a |
| Control | 42.9 b | 59.4 a | 23.2 b | 31.3 b | 17.7 b | 19.6 b |
| Significance (α = 0.05) | *** | ns | *** | *** | *** | *** |
| Genotype | Ovules Fertilized (%) | Fruit Set Efficiency (%) | Seed Set Efficiency (%) | |||
|---|---|---|---|---|---|---|
| Female | Male | Female | Male | Female | Male | |
| NASE14 | - | 90.0 a | 49.5 b | 55.7 b | 30.7 bc | 42.0 ab |
| UG15F039P015 | - | 46.7 de | - | 68.6 a | - | 52.3 a |
| UG15F056P001 | - | 79.2 b | 79.5 a | 35.9 e | 57.3 a | 30.2 cde |
| UG15F192P012 | 81.5 a | 33.4 e | 72.2 a | 41.8 cde | 68.4 a | 25.1 de |
| UG15F222P017 | 48.7 c | - | 32.8 d | 58.6 ab | 25.0 c | 26.4 de |
| UG15F228P016 | 57.5 b | - | 40.4 c | - | 23.8 c | - |
| UG15F302P016 | 83.3 a | 41.7 e | 49.5 b | 63.1 a | 36.0 b | 43.7 ab |
| Significance (α = 0.05) | * | ns | * | *** | *** | *** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baguma, J.K.; Mukasa, S.B.; Ochwo-Ssemakula, M.; Nuwamanya, E.; Iragaba, P.; Wembabazi, E.; Kanaabi, M.; Hyde, P.T.; Setter, T.L.; Alicai, T.; et al. Assessment of Cassava Pollen Viability and Ovule Fertilizability under Red-Light, 6-Benzyl Adenine, and Silver Thiosulphate Treatments. Plants 2024, 13, 1988. https://doi.org/10.3390/plants13141988
Baguma JK, Mukasa SB, Ochwo-Ssemakula M, Nuwamanya E, Iragaba P, Wembabazi E, Kanaabi M, Hyde PT, Setter TL, Alicai T, et al. Assessment of Cassava Pollen Viability and Ovule Fertilizability under Red-Light, 6-Benzyl Adenine, and Silver Thiosulphate Treatments. Plants. 2024; 13(14):1988. https://doi.org/10.3390/plants13141988
Chicago/Turabian StyleBaguma, Julius K., Settumba B. Mukasa, Mildred Ochwo-Ssemakula, Ephraim Nuwamanya, Paula Iragaba, Enoch Wembabazi, Michael Kanaabi, Peter T. Hyde, Tim L. Setter, Titus Alicai, and et al. 2024. "Assessment of Cassava Pollen Viability and Ovule Fertilizability under Red-Light, 6-Benzyl Adenine, and Silver Thiosulphate Treatments" Plants 13, no. 14: 1988. https://doi.org/10.3390/plants13141988





