1. Introduction
To meet the current food demand derived from population growth, agricultural fields have sought to increase their yields based on the use of conventional agricultural practices, such as the use of synthetic chemical fertilizers and pesticides, generating an unsustainable agroecosystem [
1,
2] as well as harming human health [
3]. In this sense, the development of sustainable strategies to reach food security worldwide is needed. One of the most promising alternatives to conventional agricultural practices is the use of beneficial microorganisms, such as the genus
Bacillus, which, due to their metabolic, physiologic, and ecological traits, are the most exploited genus in agricultural biotechnology [
4]. Currently,
Bacillus strains are the active ingredient in more than 85% of bacterial inoculants commercialized worldwide [
5], and several species of this genus are being studied to increase crop production and yields through mechanisms such as lipopeptide production, nitrogen fixation, siderophore production, phosphate solubilization, lytic enzymes, toxins, and inducing systemic resistance in plants [
6]. For this reason, bioprospecting for new strains and technologies of this genus with agro-biotechnological potential has been driven by the need to increase crop yields while minimizing the use of agrochemicals and improving soil fertility [
7].
In this context,
Bacillus cabrialesii subsp.
cabrialesii TE3
T, an endophytic bacteria isolated from a commercial wheat field in the Yaqui Valley located in México [
8,
9], can promote plant growth due to its ability to produce indoles, siderophores and solubilize phosphates [
7], and has a high tolerance to thermal, hydric, and saline stress conditions [
4,
10]. In addition, strain TE3
T acts as a biological control agent (BCA) [
11] against several pathogens of wheat (
Triticum turgidum L. subsp.
durum), such as
Bipolaris sorokiniana, the causal agent of spot blotch, root rot, foot rot, seedling blight, and seed rot [
12]. Also, it has been demonstrated that the antifungal activity of cell-free culture filtrate (CF) of strain TE3
T has strong antifungal activity against
B. sorokiniana [
8], reducing significantly (
p ≤ 0.05) the visual damage, as well as the number of lesions/cm
2 of
B. sorokiniana vs. the control treatment [
11], where the proposed metabolites involved in this biocontrol ability are surfactin, fengycin, and rhizocticin A [
13]. Thus, strain TE3
T is a promising alternative to mitigate the negative effects of this phytopathogen on wheat, where according to climate change projections,
B. sorokiniana is poised to emerge as a significant impediment to future wheat cultivation [
14]. However, strategies to improve its beneficial role in agroecosystems need to be explored to develop more efficient and cost-effective bacterial inoculants.
Mutation induction is a viable strategy for modifying the genome of bacterial strains to improve their bioactivity [
15]. The conventional random mutagenesis process is the simplest and most cost-effective technique for the improvement of bacterial capacities, either by physical and/or chemical mutagens [
16]. In this sense, Ultraviolet (UV) mutagenesis is generally the most used method for improving microbes such as bacteria [
17], due to the UV light being absorbed by many intracellular compounds in bacterial cells, but DNA suffers the most significant damage [
18]. After DNA is exposed to ultraviolet (UV) light, photoproducts such as cyclobutane-pyrimidine dimers (CPD) and pyrimidine (6-4)-pyrimidine adducts (6-4 PP) are formed at adjacent pyrimidine sites. Dinucleotides including cytosine are major sites of UV-induced mutagenesis in bacteria and eukaryotic cells, resulting mainly in C→T transitions [
18]. This damage often disrupts DNA replication, potentially leading to cell death or mutation, subsequently altering or compromising microbial traits. Examples of those mutations may manifest as shifts in colony size, cessation of sporulation, pigment loss, and a marked decrease in cell viability, known as the lethal effect [
19]. However, in some cases, mutations also improve the bioactivity of bacterial stains, which can be used in agro-biotechnological applications.
At present, there are previously reported studies where, through induced random mutations, the biological potential of
Bacillus species was improved. For example,
Bacillus sp. ACT 1, mutated with UV irradiation and chemical methods using ethidium bromide, improved its growth kinetics and the biodegradation of Congo red [
20]. Also,
Bacillus amyloliquefaciens MF 510169 was subjected to ultraviolet (UV) light mutation to improve biodiesel production by increasing lipid accumulation. The cellular lipid content of the wild-type was improved by exposing it to UV-C (254 nm) and UV-A (365 nm) wavelengths for varying durations, ranging from 20 to 380 s [
17]. Another example of improved bioactivity is
Bacillus velezensis strain BSM54, which improved in its antifungal activity attributed to the UV mutation, as this strain produced high levels of iturin, as well as surfactin and fengycin. After UV irradiation iturin A and surfactin biosynthesis genes were expressed at higher levels in the mutant strain, and in a greenhouse assay, this mutant strain reduced more effectively (
p ≥ 0.05) the phytopathogens
Sclerotinia sclerotiorum (sclerotinia rot) and
Fusarium oxysporum vs. the wild-type strain [
21]. Thus, this study aimed to induce UV random mutations in
B. cabrialesii subsp.
cabrialesii TE3
T to improve traits involved in plant growth promotion and biological control against the phytopathogenic fungi
Bipolaris sorokiniana, by seeking to increase the production of diffusible metabolites or other activities such as space and nutrient competition.
3. Discussion
Mutagenesis involves a permanent change in one or more nucleotides along the DNA strand at a specific site and is considered a technique to alter the genetic sequence of a bacterial strain to induce its activity [
15]. Random mutagenesis by physical or chemical mutagens is a known tool to improve the capabilities of biocontrol agents and/or antifungal metabolite producers. Most mutagenic agents can damage DNA through various mechanisms, such as cleavage or deletion, addition, transversion, or base substitution. These processes can affect various pathways of the cellular machinery, generating genetic instability and, as a result, a potentially improved strain [
24].
Mutation induction using UV, X-rays, γ-rays, lasers, neutrons, and chemophoresis are commonly used techniques for microorganisms [
25]. UV radiation is the most essential and efficient physical method to obtain broad-spectrum mutations. Compared to chemical mutagenesis, the use of UV light as a mutagen is considered safer [
26]. This bacterial mutation induction by UV irradiation ensured that different types of mutations would be generated, which could lead to improving the metabolic and ecological roles of promising wild-type strains [
27,
28].
The wild-type strain TE3
T was isolated from wheat and was reported as a promising biological control agent against
Bipolaris sorokiniana [
8,
11] and plant growth promoter for wheat crops [
4,
29]. Then, in 2019, this strain was reported as a novel
Bacillus species,
Bacillus cabrialesii by de los Santos-Villalobos et al., 2019 [
9]; then in 2023, this strain was the first subspecies of
B. cabrialesii,
B. cabrialesii subsp.
cabrialesii, with TE3
T being the type strain. This strain has been studied over the past five years as a promising active component of bacterial inoculants [
7]. Thus, this work aimed to improve traits involved in plant growth promotion and biological control of
Bacillus cabrialesii subsp.
cabrialesii TE3
T by UV-induced random mutations.
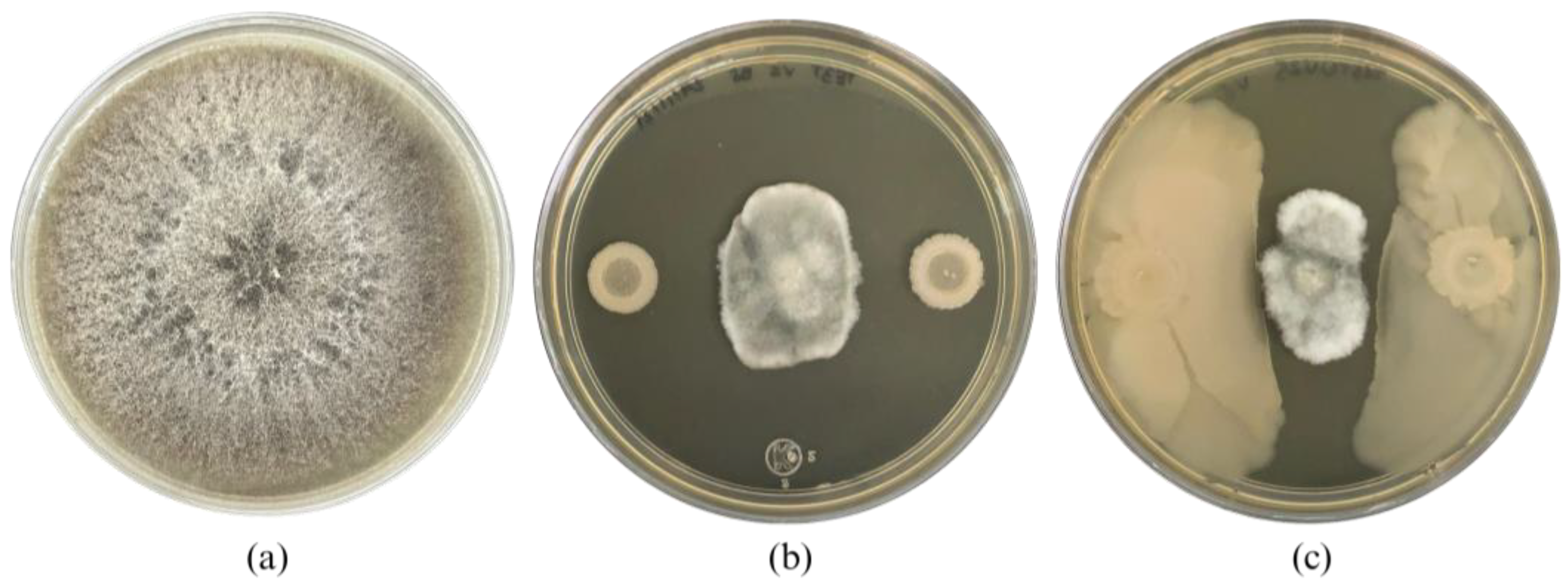
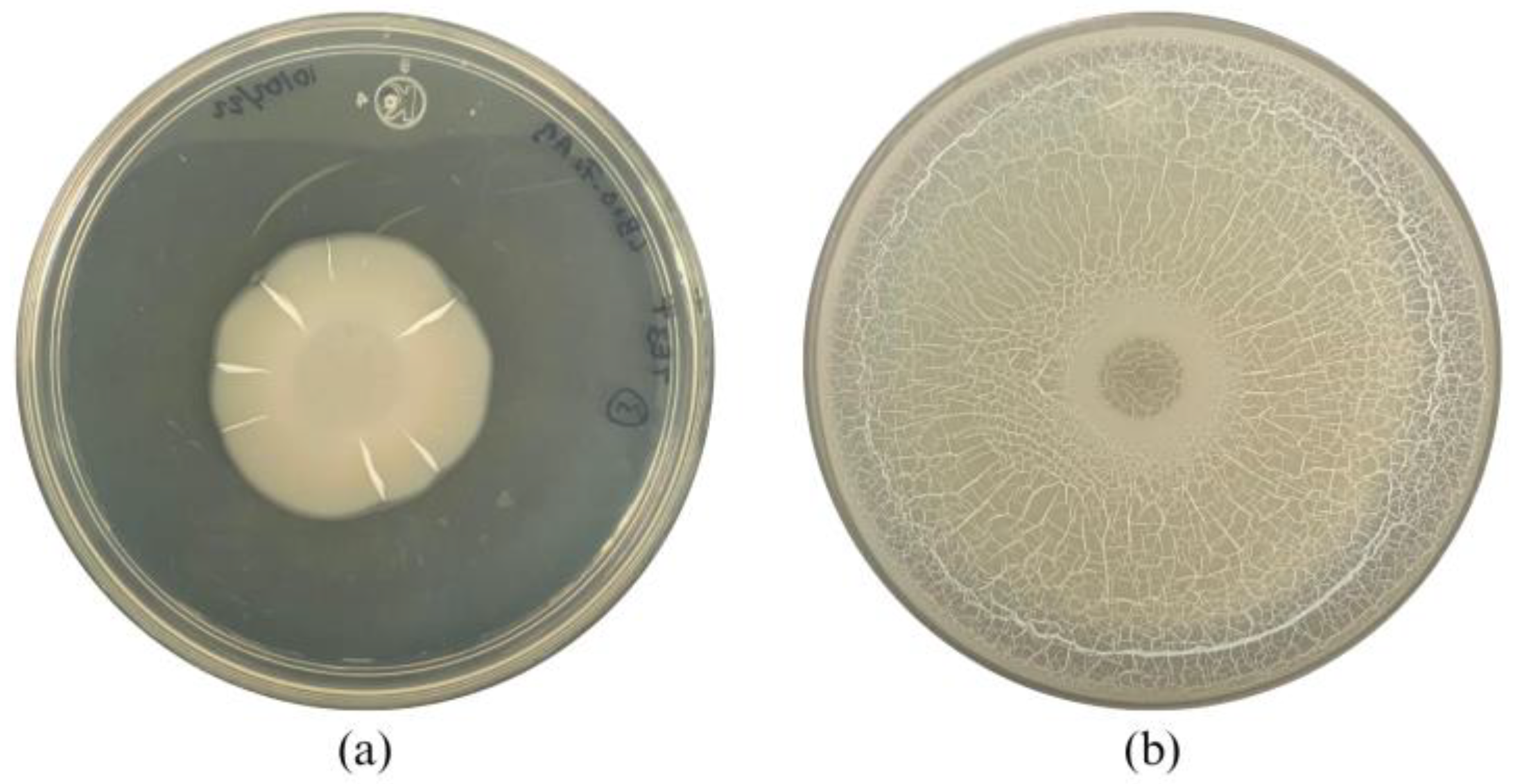
After 10 min of UV irradiation and incubation at 27 °C for 24 h, a bacterial colony named TE3
T-UV25 showed significant differences in morphology compared with the wild-type strain TE3
T. This interesting trait observed in strain TE3
T-UV25 was the swarming motility (
Figure 1 and
Figure 3, and
Table 2), which is associated with quick and successful growth to colonize nutrient-rich environments [
30], improving its ability to control phytopathogenic fungi [
31]. Thus, strain TE3
T-UV25 showed increased inhibition of
Bipolaris sorokiniana by 53.7% compared to the wild-type strain by both space and nutrient competition and diffusible metabolites (
Figure 1 and
Figure 2, and
Table 2). However, wild-type strain TE3
T has been previously investigated and its biocontrol capabilities were only associated with the production of diffusible metabolites [
13,
32]; thus, in addition to competition by swarming, the remaining ability of strain TE3
T-UV25 to biosynthesize diffusible metabolites was studied. In this sense, the cell-free supernatant of strain TE3
T-UV25 after 72 h showed a growth inhibition of 99.9% against
B. sorokiniana, indicating that this strain can inhibit the fungal growth by two mechanisms, (i) competition by nutrient and space, and (ii) biosynthesis of diffusible metabolites. This ability has been reported by Villa-Rodríguez (2021) where it was demonstrated that the lipopeptide complex of
Bacillus cabrialesii subsp.
cabrialesii TE3
T inhibited the growth of
Bipolaris sorokiniana by 89.77% [
11]. Both mutant and wild-type strains continue to be positive for siderophore production.
On the other hand, the mutant strain TE3
T-UV25 not only increased its biological control capacity but also maintained its growth-promoting properties in wheat. The results show that there are no significant differences in biometric parameters between the mutant (TE3
T-UV25) and the wild-type strain (TE3
T). However, significant differences were observed compared to the non-inoculated treatment (
Table 2), mainly highlighting stem length (44.7%), root length (22.6%), stem dry weight (42.9%) and root dry weight (22.6%). This increase in wheat biomass production by mutant strain TE3
T-UV25 is similar to values observed for the wild-type strain TE3
T as is shown in this work, as well as those demonstrated in earlier experiments by Robles Montoya et al. (2019) [
33]. In this analysis, the results demonstrate the ability of the mutant strain to stimulate wheat growth by increasing plant nutrient availability, as well as the production of plant growth regulators, such as IAA and indoles [
34]. The genus
Bacillus is one of the genera with the highest biocontrol and growth promotion capacity [
35], being recognized as one of the most abundant and universally present in the soil, with high growth promotion attributes [
36], which is consistent with the traits of the obtained mutant strain. However, there is currently little information regarding UV mutagenesis-induced PGPB to improve its ability to combat phytopathogens and promote plant growth [
27]. Despite this, UV mutation is categorized as an important technique to improve functional traits in bacterial strains [
37], but there is a lack of regulations for its use in the industry [
38].
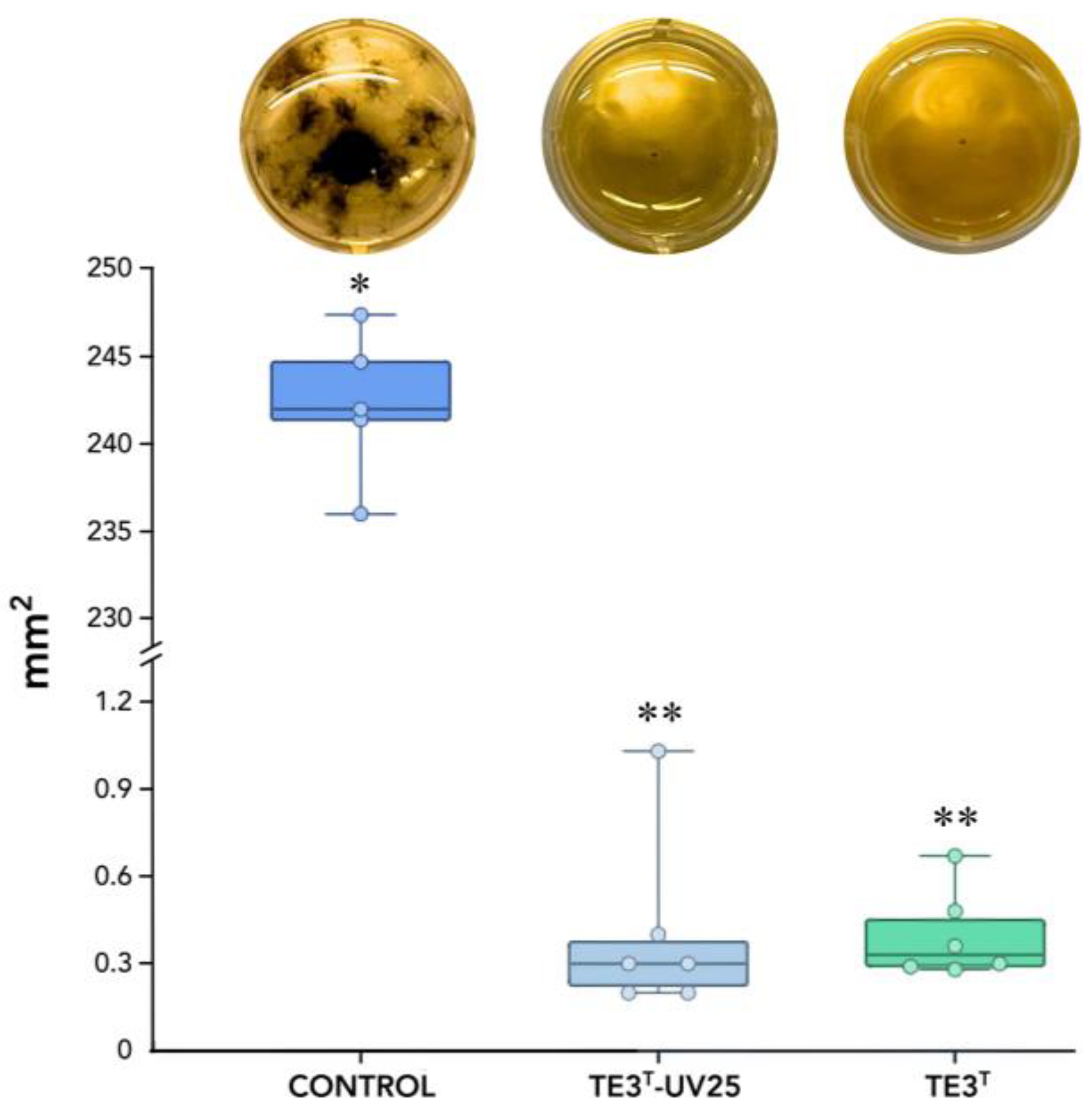
The metabolic characterization of mutant strain TE3
T-UV25 showed that the optimal pH, tolerance to NaCl, and the use of citrate as a carbon source were different vs. wild-type strains. On the other hand,
B. cabrialesii subps.
cabrialesii TE3
T is a non-hemolytic strain (γ-type); however, strain TE3
T-UV25 exhibited erythrocyte breakage (β-hemolysis). The ability to solubilize phosphates is of great relevance to their function as plant growth promoters because they play an essential role in the phosphorus (P) cycle and increase P uptake in rhizosphere soils [
28]. Most phosphate solubilizing bacteria (PSB) produce indole-3-acetic acid (IAA), which enables plant cell growth and RNA/protein synthesis and increases plant growth [
39]. According to Valenzuela-Aragon (2019),
B. cabrialesii subsp.
cabrialesii TE3
T can produce indoles (1.4 ± 0.1 ppm) and solubilize phosphate (43.2 ± 1.7%). In this study, the mutant strain maintained the metabolic traits of producing 5.4 ± 0.1 ppm of indoles; however, the capacity to solubilize phosphates (24.4 ± 1.3%) was significantly reduced but not missed [
4]. Within the genome of the wild-type strain TE3
T, 14 CDSs related to phosphate solubilization can be found, in contrast to the mutated strain in which only 11 of these CDSs were detected. The three missing CDSs are associated with the enzyme phosphoenolpyruvate mutase (EC 5.4.2.9) and phosphoenolpyruvate decarboxylase (EC 4.1.1.82), which are key enzymes in the catalysis of C-P bond formation by the intramolecular rearrangement of phosphoenolpyruvate to phosphoenolpyruvate (PnPy) [
40]. However, despite the mutation affecting these three CDSs, plant growth promotion was not significantly impacted, indicating that this characteristic may not be entirely responsible for the observed trait.
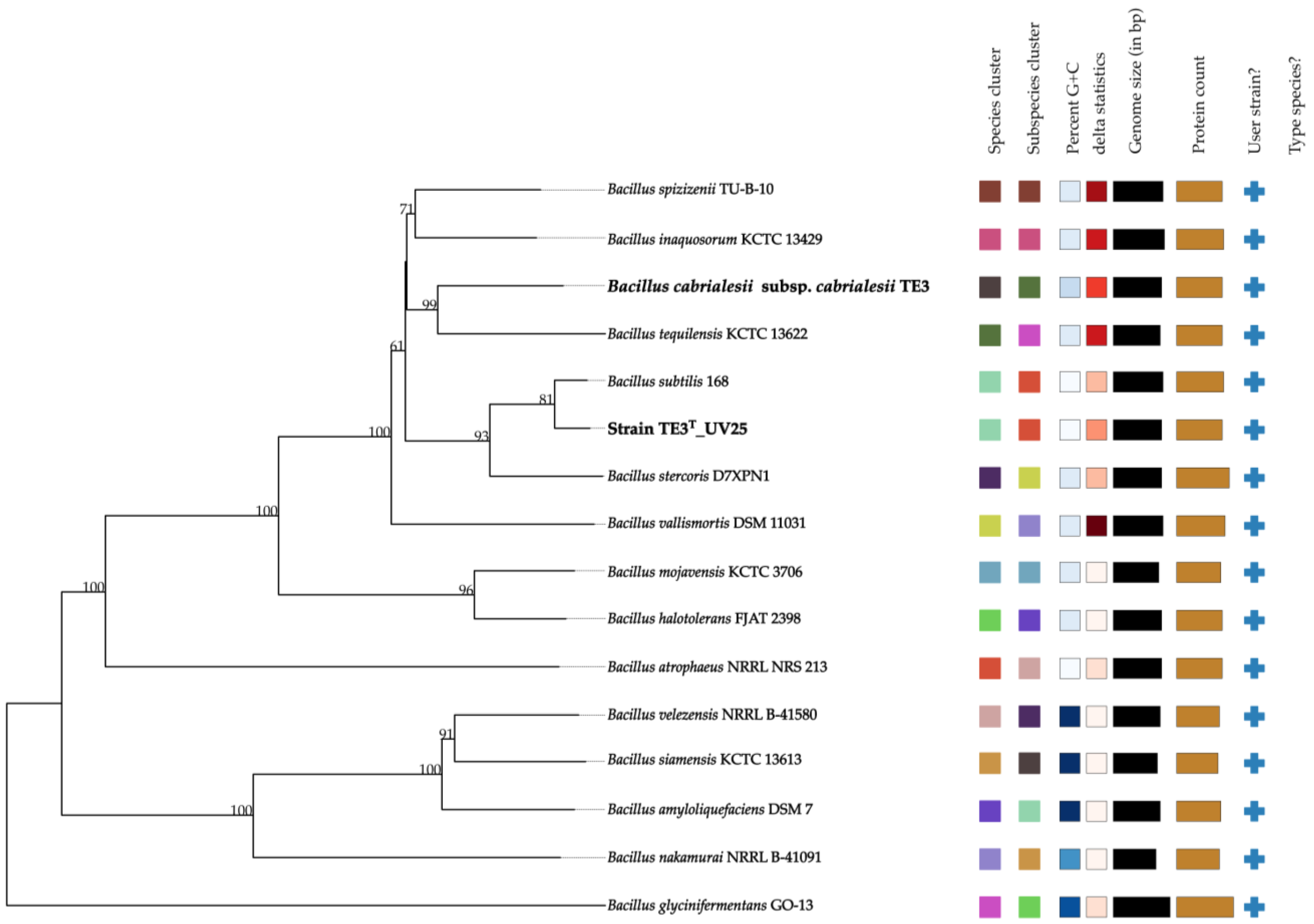
Regarding the genomic analyses, the genome of mutant strain TE3
T-UV25 is 54,478 bp larger compared to the genome of the wild-type strain TE3
T (from 1,824,296 bp to 1,818,624 bp), as well as a decrease in the G + C content of 0.69% was observed. These alterations in the genome of mutant strain TE3
T-UV25 implied not only changes at morphological, metabolically, and ecological levels, as mentioned before, but also changes in the OGRIs values (
Table 4), indicating that its taxonomic affiliation changed from
B. cabrialesii subsp.
cabrialesii to
B. subtilis. Although studies on the improvement of microbial bioactivities through mutation induction have been reported, no research was found that reports that the magnitude of the mutations generated would cause a change of species, as is this case. Then as it was confirmed by the OrthoVenn diagram, in which through the comparative genomics of the platform orthologous groups were identified and annotated, inferring evolution and phylogenetic relationships between the type strain and its mutant and
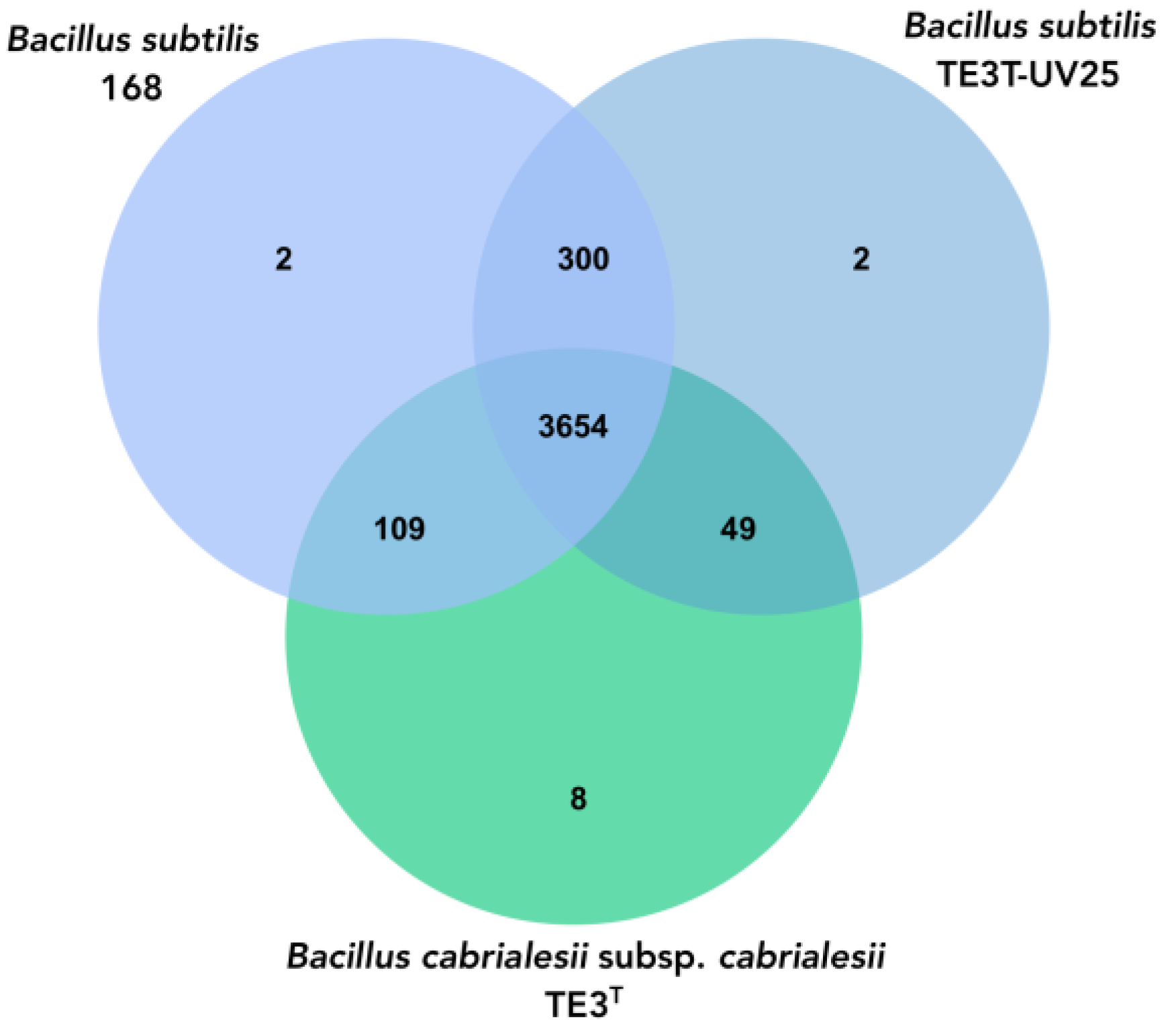
Bacillus subtilis, to which it was affiliated through the OGRIs and TYGS. Orthologous genes diverge by evolutionary speciation. Thus, orthologous genes are those shared by two or more bacteria that have an equivalent biological function and tend to be more conserved [
41]. The three species shared 3564 orthologous genes and between
Bacillus cabrialesii subsp.
cabrialesii TE3
T and its mutant TE3
T-UV25, only share 49 genes, unlike the species with which it was affiliated,
Bacillus subtilis, which shares 300 genes, confirming the taxonomic change and affiliation.
In addition, the genome mining of mutant strain TE3
T-UV25 revealed seven BGCs encoding secondary metabolites that confer strong antimicrobial properties (
Table 5). For example, (i) Surfactin, a biosurfactant produced by large multifunctional non-ribosomal peptide synthetases (NRPSs), is crucial in combating bacterial plant diseases and inhibiting filamentous fungi. It also shows efficacy against diverse multidrug-resistant bacteria. This metabolite plays roles in cell adhesion, biofilm formation, and hemolytic activity [
42,
43,
44,
45]. Furthermore, surfactin is essential for swarming motility by reducing surface tension and acting as a wetting agent [
46]; (ii) Bacillaene, a linear polyketide/non-ribosomal peptide, which enhanced the biocontrol efficiency of
Bacillus strains, and have the ability to inhibit the growth of bacteria and showing strong antifungal activity [
47]; (iii) Fengycin, a cyclic lipopeptide synthesized by NRPSs that exhibit antibacterial and antifungal activity against Gram-positive bacteria and a broad-spectrum of filamentous fungi, respectively [
48,
49], and also it can be involved in the development of induced systemic resistance [
50]; (iv) Bacillibactin, a non-ribosomal peptide produced by NRPSs, is a catechol siderophore that binds iron with high affinity, and allows cells to obtain small amounts of iron from the environment, moreover it has a potential antibacterial and antifungal activity against plant pathogens [
44,
51,
52]; (v) Subtilosin A, a cyclic peptide with antimicrobial activity against a wide range of bacteria, both Gram-positive, Gram-negative, aerobes and anaerobes [
53,
54]; (vi) Bacilysin, a non-ribosomally synthesized dipeptide antibiotic [
55] that causes cell lysis in bacteria and fungi, and has also been related with the inhibition of algae [
56]; and (vii) Pulcherriminic acid, which was only detected in the genome of mutant strain TE3
T-UV25. This cyclodipeptide is one of the siderophores produced by
B. subtilis, that acts as an iron-chelating molecule and when ligated with Fe (III) forms pulcherrimin [
57]. The pulcherriminic acid can modulate iron availability and decrease oxidative stress, increasing survival and contributing to biofilm formation, while inter-species competition. In addition, it has been reported that pulcherriminic acid has an important role in the biocontrol of pathogens, this is related to iron competition [
58], causing other microorganisms unable to acquire the required iron for growth acting as a protective agent [
59]. Finally, Rhizocticin A was detected only in the genome of the wild-type strain, which has been reported with antifungal activity [
60]; however, in metabolomics analysis realized previously in
Bacillus cabrialesii subsp.
cabrialesii TE3
T, this metabolite was not identified under-tested culture conditions (28 °C and 180 rpm) and therefore it is not involved in the antifungal activity exhibited against the phytopathogen
Bipolaris sorokiniana TPQ3 [
11].