Senescence-Associated Vacuoles, a Specific Lytic Compartment for Degradation of Chloroplast Proteins?
Abstract
:1. An Overview of Leaf Senescence
2. Chloroplast Degradation in Senescing Leaf Cells
3. Involvement of the Central Vacuole
4. Senescence-Associated Vacuoles: Contents, Characteristics, Markers
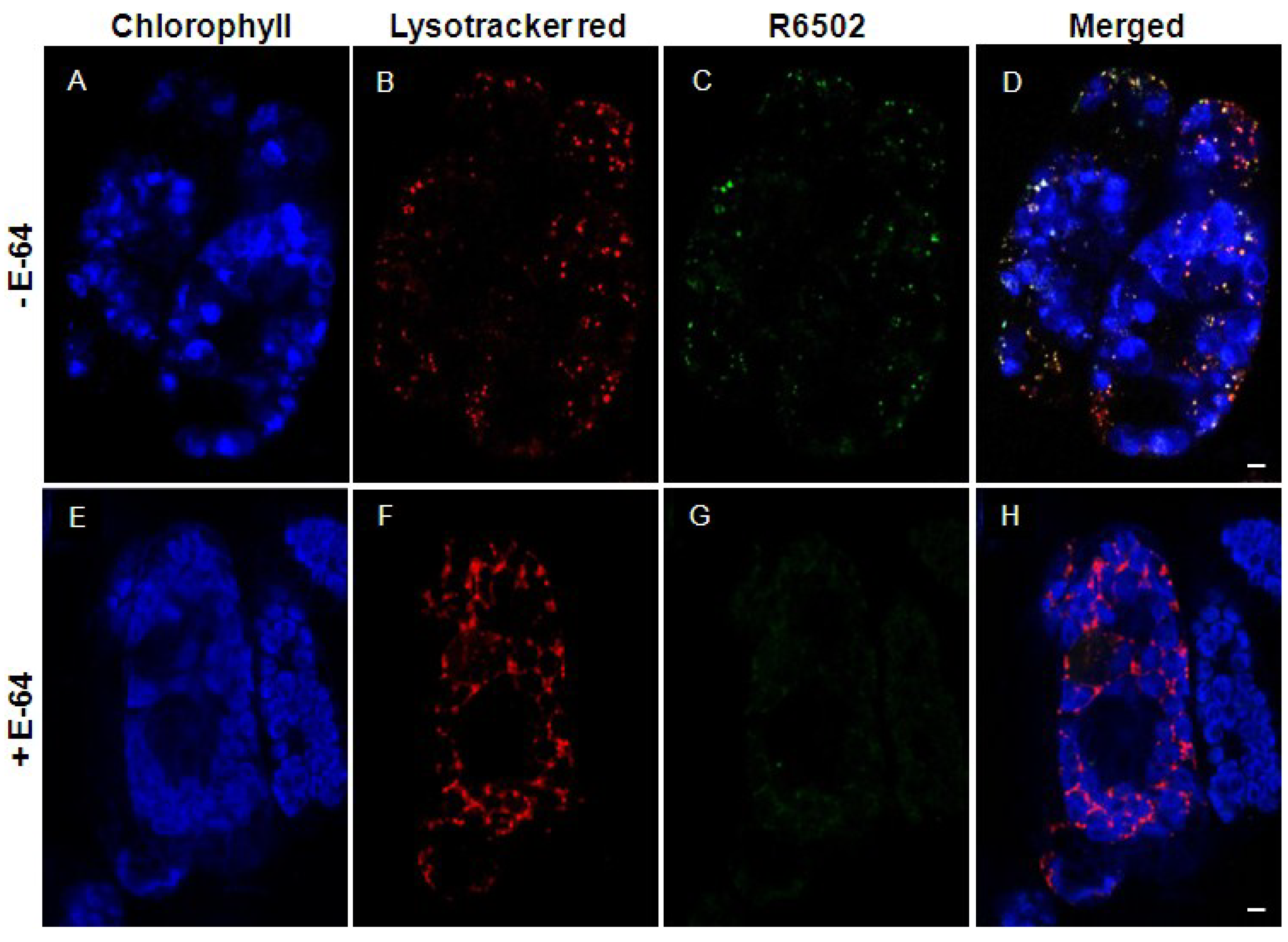

5. Involvement of SAVs in Chloroplast Protein Degradation

6. SAVs and Rubisco-Containing Bodies, Is There a Link?


7. Future Directions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Noodén, L.D.; Guiamet, J.J.; John, I. Whole plant senescence. In Plant Cell Death Processes; Noodén, L.D., Ed.; Academic Press: San Diego, CA, USA, 2004; pp. 227–244. [Google Scholar]
- Li, P.O.; Kim, H.J.; Nam, H.G. Leaf senescence. Annu. Rev. Plant. Biol. 2007, 58, 115–136. [Google Scholar]
- Masclaux-Daubresse, C.; Reisdorf-Cren, M.; Orsel, M. Leaf nitrogen remobilisation for plant development and grain filling. Plant Biol. 2008, 10, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Mae, T. Leaf senescence and nitrogen metabolism. In Plant Cell Death Processes; Noodén, L.D., Ed.; Academic Press: San Diego, CA, USA, 2004; pp. 157–168. [Google Scholar]
- Killingbeck, K.T. Nutrient resorption. In Plant Cell Death Processes; Noodén, L.D., Ed.; Academic Press: San Diego, CA, USA, 2004; pp. 215–226. [Google Scholar]
- Peoples, M.B.; Dalling, M.J. The interplay between proteolysis and amino acid metabolism during senescence and nitrogen reallocation. In Senescence and Aging in Plants; Noodén, L.D., Leopold, A.C., Eds.; Academic Press: San Diego, CA, USA, 1988; pp. 181–217. [Google Scholar]
- Vergutz, L.; Manzoni, S.; Portorato, A.; Ferreira Novais, R.; Jackson, M.B. Global resorption efficiencies and concentrations of carbon and nutrients in leaves of terrestrial plants. Ecol. Monogr. 2012, 82, 205–220. [Google Scholar] [CrossRef]
- Martínez, D.E.; Guiamet, J.J. Senescence-related changes in the leaf apoplast. J. Plant Growth Regul. 2014, 33, 44–55. [Google Scholar] [CrossRef]
- Del Río, L.A.; Pastori, G.M.; Palma, J.M.; Sandalio, L.M.; Sevilla, F.; Corpas, F.J.; Jiménez, A.; López-Huerta, E.; Hernández, J.A. The activated oxygen role of peroxisomes in senescence. Plant Physiol. 1998, 116, 1195–1200. [Google Scholar] [CrossRef] [PubMed]
- Keech, O.; Pesquet, E.; Ahad, A.; Askne, A.; Nordvall, D.; Vodnala, S.M.; Tuominen, H.; Hurry, V.; Dizengremel, P.; Gardeström, P. The different fates of mitochondria and chloroplasts during dark-induced senescence in Arabidopsis leaves. Plant Cell Environ. 2007, 30, 1523–1534. [Google Scholar] [CrossRef] [PubMed]
- Guiamét, J.J.; Luquez, V.M. Leaf senescence and related processes. In Crops: Growth, Quality, and Biotechnology; Dris, R., Ed.; WFL Publisher: Helsinki, Finland, 2005; pp. 1238–1253. [Google Scholar]
- Krupinska, K. Fate and activities of plastids during leaf senescence. In The Structure and Function of Plastids; Wise, R.R., Hoober, J.K., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 433–449. [Google Scholar]
- Gepstein, S. Photosynthesis. In Senescence and Aging in Plants; Noodén, L.D., Leopold, A.C., Eds.; Academic Press: San Diego, CA, USA, 1988; pp. 85–109. [Google Scholar]
- Costa, M.L.; Martínez, D.E.; Gomez, F.M.; Carrión, C.; Guiamét, J.J. Chloroplast protein degradation: Involvement of senescence-associated vacuoles. In Advances in Photosynthesis and Respiration Series; Biswal, B., Krupinska, K., Biswal, U.C., Eds.; Springer Science + Business Media: Dordrecht, The Netherlands, 2013; pp. 417–433. [Google Scholar]
- Meir, S.; Philosoph-Hadas, S. Metabolism of polar lipids during senescence of watercress leaves. Plant Physiol. Biochem. 1995, 33, 241–249. [Google Scholar]
- Ougham, H.; Hörtensteiner, S.; Armstead, I.; Donninson, I.; King, I.; Thomas, H.; Mur, L. The control of chlorophyll catabolism and the status of yellowing as a biomarker of leaf senescence. Plant Biol. 2008, 10, 4–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hörtensteiner, S.; Kräutler, B. Chlorophyll breakdown in higher plants. Biochim. Biophys. Acta 2011, 1807, 977–988. [Google Scholar] [CrossRef]
- Krupinska, K.; Humbeck, K. Photosynthesis and chloroplast breakdown. In Plant Cell Death Processes; Noodén, L.D., Ed.; Academic Press: San Diego, CA, USA, 2004; pp. 169–187. [Google Scholar]
- Kamachi, K.; Yamaya, T.; Hayakawa, T.; Mae, T.; Ojima, K. Changes in cytosolic glutamine synthetase polypeptide and its mRNA in a leaf blade of rice plants during natural senescence. Plant Physiol. 1992, 98, 1323–1329. [Google Scholar] [CrossRef]
- Schmutz, D.; Wyss, H.-R.; Brunold, C. Activity of sulfate-assimilating enzymes in primary leaves of Phaseolus vulgaris L. during dark-induced senescence. Z. Pflanzenphysiol. 1983, 110, 211–219. [Google Scholar] [CrossRef]
- Hidema, J.; Makino, A.; Mae, T.; Ojima, K. Photosynthetic characteristics of rice leaves aged under different irradiances from full expansion through senescence. Plant Physiol. 1991, 97, 1287–1293. [Google Scholar] [CrossRef] [PubMed]
- Zaltsman, A.; Ori, N.; Adam, Z. Two types of FtsH subunits are required for chloroplast biogenesis and Photosystem II repair in Arabidopsis. Plant Cell 2005, 17, 2782–2790. [Google Scholar] [CrossRef] [PubMed]
- Kapri-Pardes, E.; Naveh, L.; Adam, Z. The thylakoid lumen protease DegP1 is involved in the repair of photosystem II from photoinhibition in Arabidopsis. Plant Cell 2007, 19, 1039–1047. [Google Scholar] [CrossRef]
- Sun, X.; Peng, L.; Guo, J.; Chi, W.; Ma, J.; Lu, C.; Zhang, L. Formation of Deg5 and Deg8 complexes and their involvement in the degradation of photodamaged Photosystem II reaction center D1 protein in Arabidopsis. Plant Cell 2007, 19, 1347–1361. [Google Scholar] [CrossRef]
- Kato, Y.; Miura, E.; Ido, K.; Ifuku, K.; Sakamoto, W. The variegated mutants lacking chloroplastic FtsHs are defective in D1 degradation and accumulate reactive oxygen species. Plant Physiol. 2009, 151, 1790–1801. [Google Scholar]
- Sun, X.; Fu, T.; Chen, N.; Guo, J.; Ma, J.; Zou, M.; Lu, C.; Zhang, L. The stromal chloroplast Deg7 protease participates in the repair of photosystem II after photoinhibition in Arabidopsis. Plant Physiol. 2010, 152, 1263–1273. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.-H.; Webster, J.; Adam, Z.; Lindahl, M.; Andersson, B. Induction of acclimative proteolysis of the light-harvesting chlorophyll a/b protein of photosystem II in response to elevated light intensities. Plant Physiol. 1998, 118, 827–834. [Google Scholar] [CrossRef]
- Zelisko, A.; Jackowski, G. Senescence-dependent degradation of Lhcb3 is mediated by a thylakoid membrane-bound protease. J. Plant Physiol. 2004, 161, 1157–1170. [Google Scholar] [CrossRef]
- Zelisko, A.; García-Lorenzo, M.; Jackowski, G.; Jansson, S.; Funk, C. AtFtsH6 is involved in the degradation of the light-harvesting complex II during high-light acclimation and senescence. Proc. Natl. Acad. Sci. USA 2005, 102, 13690–13704. [Google Scholar] [CrossRef]
- Guiamét, J.J.; Tyystjärvi, E.; Tyystjärvi, T.; John, I.; Kairavuo, M.; Pichersky, P.; Noodén, L.D. Photoinhibition and loss of photosystem II reaction center proteins during senescence of soybean leaves. Enhancement of photoinhibition by the “stay-green” mutation cytG. Physiol. Plant 2002, 115, 468–478. [Google Scholar] [CrossRef]
- Wagner, R.; Aigner, H.; Pruzinská, A.; Jänkänpää, H.J.; Jansson, S.; Funk, C. Fitness analysis of Arabidopsis thaliana mutants depleted of FtsH metalloproteases and characterization of three FtsH6 deletion mutants exposed to high light stress, senescence and chilling. New Phytol. 2011, 191, 449–458. [Google Scholar] [CrossRef]
- Martínez, D.E.; Costa, M.; Guiamet, J.J. Senescence associated degradation of chloroplast proteins inside and outside the organelle. Plant Biol. 2008, 10, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Chiba, A.; Ishida, H.; Nishizawa, N.K.; Makino, A.; Mae, T. Exclusion of ribulose-1,5-bisphosphate carboxylase/oxygenase from chloroplasts by specific bodies in naturally senescing leaves of wheat. Plant Cell Physiol. 2003, 44, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Ishida, H.; Yoshimoto, K.; Izumi, M.; Reisen, D.; Yano, Y.; Makino, A.; Ohsumi, Y.; Hanson, M.R.; Mae, T. Mobilization of rubisco and stroma-localized fluorescent proteins of chloroplast to the vacuole by an ATG gene-dependent autophagic process. Plant Physiol. 2008, 148, 142–155. [Google Scholar] [CrossRef] [PubMed]
- Martínez, D.E.; Costa, M.L.; Gomez, F.M.; Otegui, M.S.; Guiamet, J.J. “Senescence-associated vacuoles” are involved in the degradation of chloroplast proteins in tobacco leaves. Plant J. 2008, 56, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Wittenbach, V.A. Subcellular localization of proteases in wheat and corn mesophyll protoplasts. Plant Physiol. 1981, 67, 969–972. [Google Scholar] [CrossRef]
- Wittenbah, V.A.; Lin, W.; Herbert, R.H. Vacuolar localization of proteases and degradation of chloroplasts in mesophyll protoplasts from senescing primary wheat leaves. Plant Physiol. 1982, 69, 98–102. [Google Scholar] [CrossRef]
- Srivalli, B.; Bharti, S.; Kanna-Chopra, R. Vacuole cysteine proteases and ribulose-1,5-bisphosphate carboxylase/oxygenase degradation during monocarpic senescence in cowpea leaves. Photosynthetica 2001, 39, 87–93. [Google Scholar] [CrossRef]
- De Deepesh, N. Plant Cell Vacuoles. An Introduction; CSIRO Publishing: Collingwood, Australia, 2000; pp. 1–304. [Google Scholar]
- Müntz, K. Protein dynamics and proteolysis in plant vacuoles. J. Exp. Bot. 2007, 58, 2391–2407. [Google Scholar] [CrossRef]
- Hensel, L.L.; Grbic, V.; Baumgarten, D.A.; Bleecker, A.B. Developmental and age-related processes that influence the longevity and senescence of photosynthetic tissues in Arabidopsis. Plant Cell 1993, 5, 553–564. [Google Scholar] [CrossRef] [PubMed]
- Drake, R.; John, I.; Farrell, A.; Cooper, W.; Schuch, W.; Grierson, D. Isolation and analysis of cDNAs encoding tomato cysteine proteases expressed during leaf senescence. Plant Mol. Biol. 1996, 30, 755–767. [Google Scholar] [CrossRef] [PubMed]
- Martínez, D.E.; Bartoli, C.G.; Grbic, V.; Guiamet, J.J. Vacuolar cysteine proteases of wheat (Triticum aestivum L.) are common to leaf senescence induced by different factors. J. Exp. Bot. 2007, 58, 1099–1107. [Google Scholar] [CrossRef] [PubMed]
- Minimikawa, T.; Toyooka, K.; Okamoto, T.; Hara-Nishimura, I.; Nishimura, M. Degradation of ribulose-bisphosphate carboxylase by vacuolar enzymes of senescing French bean leaves: Immunocytochemical and ultrastructural observations. Protoplasma 2001, 218, 144–153. [Google Scholar]
- Niwa, Y.; Kato, T.; Tabata, S.; Seki, M.; Kobayashi, M.; Shinozaki, K.; Moriyasu, Y. Disposal of chloroplasts with abnormal function into the vacuole in Arabidopsis thaliana cotyledon cells. Protoplasma 2004, 223, 229–232. [Google Scholar]
- Wada, S.; Ishida, H.; Izumi, M.; Yoshimoto, K.; Ohsumi, Y.; Mae, T.; Makino, A. Autophagy plays a role in chloroplast degradation during senescence in individually darkened leaves. Plant Physiol. 2009, 149, 885–893. [Google Scholar]
- Evans, M.; Rus, A.M.; Belanger, E.M.; Kimoto, M.; Brusslan, J.A. Dismantling of Arabidopsis thaliana mesophyll cell chloroplasts during natural leaf senescence. Plant Biol. 2010, 12, 1–12. [Google Scholar]
- Matile, P. The vacuole and cell senescence. Adv. Bot. Res. 1997, 25, 87–112. [Google Scholar]
- Ono, Y.; Wada, S.; Izumi, M.; Makino, A.; Ishida, H. Evidence for contribution of autophagy to Rubisco degradation during leaf senescence in Arabidopsis thaliana. Plant Cell Environ. 2013, 36, 1147–1159. [Google Scholar] [CrossRef]
- Swanson, S.J.; Bethke, P.C.; Jones, R.L. Barley aleurone cells contain two types of vacuoles: Characterization of lytic organelles by means of fluorescent probes. Plant Cell 1998, 10, 685–698. [Google Scholar] [PubMed]
- Epimashko, S.; Meckel, T.; Fischer-Schliebs, E.; Lüttge, U.; Thiel, G. Two functional different vacuoles for static and dynamic purposes in one plant mesophyll leaf cell. Plant J. 2004, 37, 294–300. [Google Scholar]
- Frigerio, L.; Hinz, G.; Robinson, D.G. Multiple vacuoles in plant cells: Rule or exception? Traffic 2008, 9, 1564–1570. [Google Scholar] [CrossRef]
- Otegui, M.; Noh, Y.-S.; Martínez, D.; Vila-Petroff, M.; Staehelin, A.; Amasino, R.; Guiamet, J.J. Senescence-associated vacuoles with intense proteolytic activity develop in senescing leaves of Arabidopsis and soybean. Plant J. 2005, 41, 831–844. [Google Scholar] [CrossRef]
- Carrión, C.A.; Costa, M.L.; Martínez, D.E.; Mohr, C.; Humbeck, K.; Guiamet, J.J. In vivo inhibition of cysteine proteases provides evidence for the involvement of “senescence-associated vacuoles” in chloroplast protein degradation during dark-induced senescence of tobacco leaves. J. Exp. Bot. 2013, 64, 4967–4980. [Google Scholar] [CrossRef]
- Peptidase substrates. Available online: http://www.lifetechnologies.com/ar/es/home/references/molecular-probes-the-handbook/enzyme-substrates/detecting-peptidases-and-proteases.html#head1 (accessed on 7 October 2014).
- Greenbaum, D.; Medzihradsky, K.; Burlingame, A.; Bogyo, M. Epoxide electrophiles as activity-dependent cysteine protease profiling and discovery tools. Chem. Biol. 2000, 7, 569–581. [Google Scholar] [CrossRef] [PubMed]
- Van der Hoorn, R.A.L.; Leeuwenbergh, M.A.; Bogyo, M.; Joosten, M.H.A.; Peck, S.C. Activity profiling of papain-like cystein proteases in plants. Plant Physiol. 2004, 135, 1170–1178. [Google Scholar] [CrossRef] [PubMed]
- Lohman, K.N.; Gan, S.; John, L.M.; Amasino, R.M. Molecular analysis of natural leaf senescence in Arabidopsis thaliana. Physiol. Plant. 1994, 92, 322–328. [Google Scholar] [CrossRef]
- Noh, Y.-S.; Amasino, R.M. Identification of a promoter region responsible for the senescence-specific expression of SAG12. Plant Mol. Biol. 1999, 41, 181–194. [Google Scholar] [CrossRef]
- Grbic, V. SAG2 and SAG12 protein expression in senescing Arabidopsis plants. Physiol. Plant. 2003, 119, 1–7. [Google Scholar]
- Köhler, R.H.; Cao, J.; Zipfel, W.R.; Webb, W.W.; Hanson, M.R. Exchange of protein molecules through connections between higher plant plastids. Science 1997, 276, 2039–2042. [Google Scholar] [CrossRef] [PubMed]
- Thoenen, M.; Herrmann, B.; Feller, U. Senescence in wheat leaves: Is a cysteine endopeptidase involved in the degradation of the large subunit of Rubisco? Acta Physiol. Plant. 2007, 29, 339–350. [Google Scholar]
- Carrión, C.A.; Instituto de Fisiología Vegetal (INFIVE, CONICET-UNLP), La Plata, Argentina. Unpublished data. 2013.
- Tamura, K.; Shimada, T.; Ono, E.; Tanaka, Y.; Nagatani, A.; Higashi, S.-I.; Watanabe, M.; Nishimura, M.; Hara-Nishimura, I. Why green fluorescent fusion proteins have not been observed in the vacuoles of higher plants. Plant J. 2003, 35, 545–555. [Google Scholar] [CrossRef]
- Dettmer, J.; Hong-Hemesdorf, A.; Stierhof, Y.-D.; Schumacher, K. Vacuolar H+-ATPase activity is required for endocytic and secretory trafficking in Arabidopsis. Plant Cell 2006, 18, 715–730. [Google Scholar] [CrossRef]
- Doelling, J.H.; Walker, J.M.; Friedman, E.M.; Thompson, A.R.; Vierstra, R.D. The APG8/12-activating enzyme APG7 is required for proper nutrient recycling and senescence in Arabidopsis thaliana. J. Biol. Chem. 2002, 277, 33105–33114. [Google Scholar] [CrossRef]
- Thompson, A.R.; Doelling, J.H.; Suttangkakul, A.; Vierstra, R.D. Autophagic nutrient recycling in Arabidopsis directed by the ATG8 and ATG12 conjugation pathways. Plant Physiol. 2005, 138, 2097–2110. [Google Scholar] [CrossRef]
- Liu, Y.; Bassham, D.C. Autophagy: Pathways for self-eating in plant cells. Ann. Rev. Plant Biol. 2012, 63, 215–237. [Google Scholar] [CrossRef]
- Lee, T.A.; Vande Wetering, S.W.; Bruslan, J.A. Stromal protein degradation is incomplete in Arabidopsis thaliana autophagy mutants undergoing natural senescence. BMC Res. Notes 2013, 6, 17. [Google Scholar] [CrossRef] [PubMed]
- Guiboileau, A.; Avila-Ospina, L.; Yoshimoto, K.; Soulay, F.; Azzopardi, M.; Marmagne, M.; Lothier, J.; Masclaux-Daubresse, C. Physiological and metabolic consequences of autophagy deficiency for the management of nitrogen and protein resources in Arabidopsis leaves depending on nitrate availability. New Phytol. 2013, 199, 683–694. [Google Scholar]
- Yoshimoto, K.; Jikumaru, Y.; Kamiya, Y.; Kusano, M.; Consonni, C.; Panstruga, R.; Ohsuni, Y.; Shirasu, K. Autophagy negatively regulates cell death by controlling NPR1-dependent salicylic acid signaling during senescence and the innate immune response in Arabidopsis. Plant Cell 2009, 21, 2914–2927. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carrión, C.A.; Martínez, D.E.; Costa, M.L.; Guiamet, J.J. Senescence-Associated Vacuoles, a Specific Lytic Compartment for Degradation of Chloroplast Proteins? Plants 2014, 3, 498-512. https://doi.org/10.3390/plants3040498
Carrión CA, Martínez DE, Costa ML, Guiamet JJ. Senescence-Associated Vacuoles, a Specific Lytic Compartment for Degradation of Chloroplast Proteins? Plants. 2014; 3(4):498-512. https://doi.org/10.3390/plants3040498
Chicago/Turabian StyleCarrión, Cristian A., Dana E. Martínez, M. Lorenza Costa, and Juan José Guiamet. 2014. "Senescence-Associated Vacuoles, a Specific Lytic Compartment for Degradation of Chloroplast Proteins?" Plants 3, no. 4: 498-512. https://doi.org/10.3390/plants3040498



