Exogenous Melatonin Delays Dark-Induced Grape Leaf Senescence by Regulation of Antioxidant System and Senescence Associated Genes (SAGs)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and MT Treatment
2.2. Measurement of Chlorophyll Content
2.3. Determination of MDA Content and Electrolyte Leakage
2.4. Extraction and Antioxidant Enzymes Assay
2.5. Extraction and Analysis of Antioxidant Substances
2.6. Quantifications of O2− and H2O2
2.7. RNA Isolation and Quantitative Real-time PCR
2.8. Statistical Analysis
3. Results
3.1. MT Could Monitor Dark Yellowing in a Dose-Dependent Behavior
3.2. MT Could Decline the Accumulation of Oxidizing Agents
3.3. Antioxidant Enzyme Responses to MT Treatment
3.4. Effect of MT on the Ascorbate-Glutathione Cycle
3.5. MT Could Inhibit the Expression Levels of SAGs
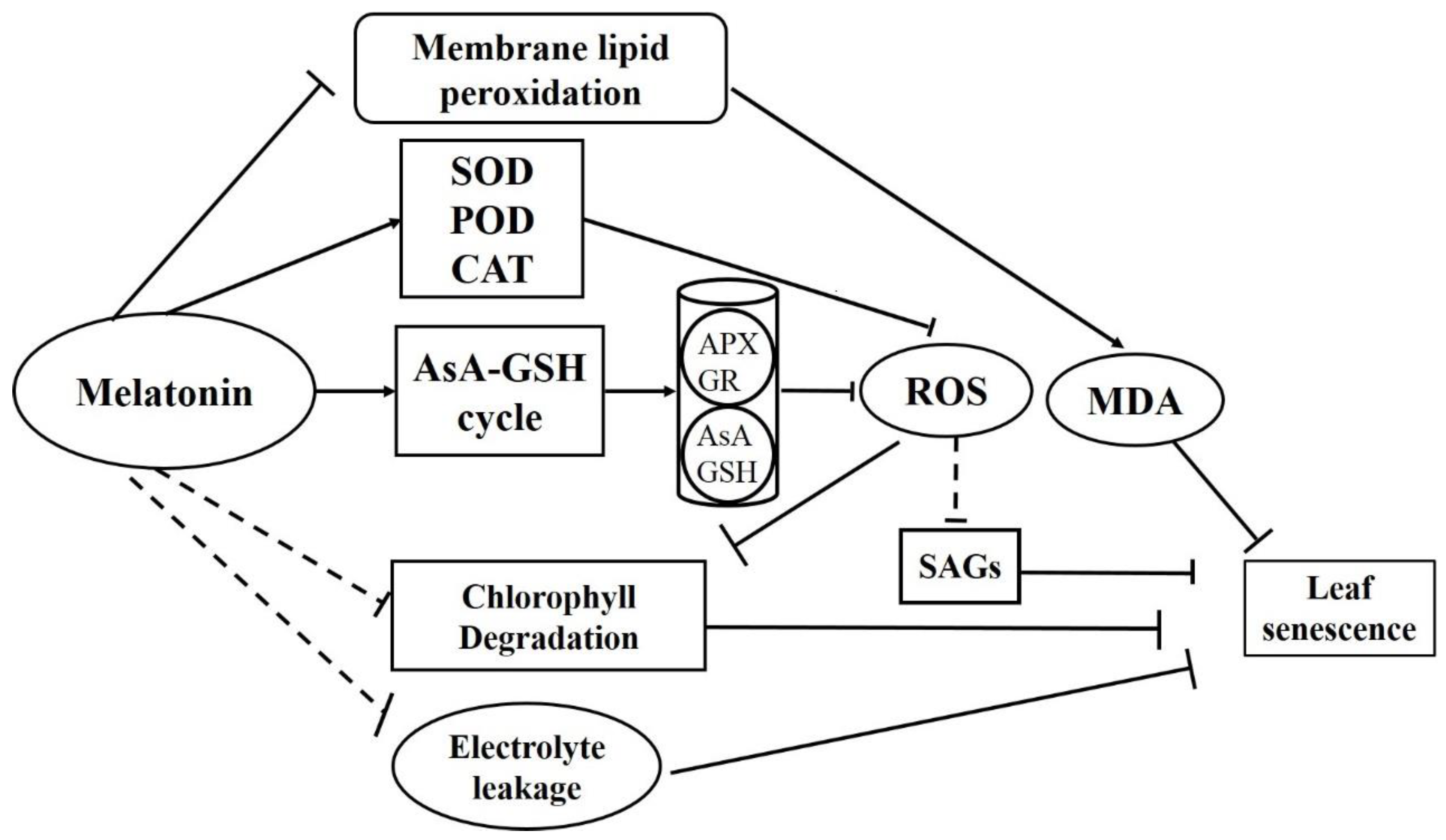
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lim, P.O.; Kim, H.J.; Nam, H.G. Leaf Senescence. Annu. Rev. Plant Biol. 2007, 58, 115–136. [Google Scholar] [CrossRef] [Green Version]
- Zhang, K.K.; Liu, F.Z.; Wang, X.D.; Shi, X.B.; Wang, B.L.; Zheng, X.C.; Ji, X.H.; Wang, H.B. Effects of supplementary light with different wavelengths on fruit quality of ‘Ruidu Xiangyu’ grape under promoted cultivation. Chin. J. Appl. Ecol. 2017, 28, 115–126. [Google Scholar]
- Li, C.X.; Chang, S.X.; Khalil-Ur-Rehman, M.; Xu, Z.G.; Tao, J.M. Effect of irradiating the leaf abaxial surface with supplemental light-emitting diode lights on grape photosynthesis: Supplemental light and grape photosynthesis. Aust. J. Grape Wine Res. 2017, 23, 58–65. [Google Scholar] [CrossRef]
- Wagstaff, C.; Malcolm, P.; Rafiq, A.; Leverentz, M.; Griffiths, G.; Thomas, B.; Stead, A.; Rogers, H. Programmed cell death (PCD) processes begin extremely early in Alstroemeria petal senescence. N. Phytol. 2010, 160, 49–59. [Google Scholar] [CrossRef]
- Van Doorn, W.G.; Woltering, E.J. Senescence and programmed cell death: Substance or semantics. J. Exp. Bot. 2004, 55, 2147–2153. [Google Scholar] [CrossRef]
- Yen, C.H.; Yang, C.H. Evidence for programmed cell death during leaf senescence in plants. Plant Cell Physiol. 1998, 39, 922–927. [Google Scholar] [CrossRef]
- Panda, D.; Sarkar, R.K. Natural leaf senescence: Probed by chlorophyll fluorescence, CO2 photosynthetic rate and antioxidant enzyme activities during grain filling in different rice cultivars. Physiol. Mol. Biol. Plants 2013, 19, 43–51. [Google Scholar] [CrossRef]
- Prochazkova, D.; Wilhelmova, N. Leaf senescence and activities of the antioxidant enzymes. Biol. Plant. 2007, 51, 401–406. [Google Scholar] [CrossRef]
- Jakhar, S.; Mukherjee, D. Chloroplast pigments, proteins, lipid peroxidation and activities of antioxidative enzymes during maturation and senescence of leaves and reproductive organs of Cajanus cajan L. Physiol. Mol. Biol. Plants 2014, 20, 171–180. [Google Scholar] [CrossRef]
- Distelfeld, A.; Avni, R.; Fischer, A.M. Senescence, nutrient remobilization, and yield in wheat and barley. J. Exp. Bot. 2014, 65, 3783–3798. [Google Scholar] [CrossRef] [Green Version]
- Lim, P.O.; Woo, H.R.; Nam, H.G. Molecular genetics of leaf senescence in Arabidopsis. Trends Plant Sci. 2003, 8, 272–278. [Google Scholar] [CrossRef]
- Zhou, X.; Jiang, Y.J.; Yu, D.Q. WRKY22 transcription factor mediates dark-induced leaf senescence in Arabidopsis. Mol. Cells 2011, 31, 303–313. [Google Scholar] [CrossRef] [Green Version]
- Weaver, L.M.; Gan, S.S.; Quirino, B.; Amasino, R.M. A comparison of the expression patterns of several senescence-associated genes in response to stress and hormone treatment. Plant Mol. Biol. 1998, 37, 455–469. [Google Scholar] [CrossRef]
- Basuchaudhuri, P. Changes in leaves of mandarin orange during senescence. Indian J. Hortic. 1990, 47, 186–189. [Google Scholar]
- He, Y.; Gan, S. A gene encoding an acyl hydrolase is involved in leaf senescence in Arabidopsis. Plant Cell 2002, 14, 805–815. [Google Scholar] [CrossRef]
- Lee, S.; Seo, P.J.; Lee, H.J.; Park, C.M. A NAC transcription factor NTL4 promotes reactive oxygen species production during drought-induced leaf senescence in Arabidopsis. Plant J. 2012, 70, 14. [Google Scholar] [CrossRef]
- Ren, B.; Zhang, J.; Li, X.; Fan, X.; Dong, S.; Zhao, B.; Liu, P. Effect of waterlogging on leaf senescence characteristics of summer maize in the field. Chin. J. Appl. Ecol. 2014, 25, 1022–1028. [Google Scholar]
- Xiao, H.J.; Liu, K.K.; Li, D.W.; Arisha, M.H.; Chai, W.G.; Gong, Z.H. Cloning and characterization of the pepper CaPAO gene for defense responses to salt-induced leaf senescence. BMC Biotechnol. 2015, 15, 100. [Google Scholar] [CrossRef]
- Yang, S.D.; Seo, P.J.; Yoon, H.K.; Park, C.M. The Arabidopsis NAC transcription factor VNI2 integrates abscisic acid signals into leaf senescence via the COR/RD genes. Plant Cell 2011, 23, 2155–2168. [Google Scholar] [CrossRef]
- Xu, Y.; Huang, B. Heat-induced leaf senescence and hormonal changes for thermal bentgrass and turf-type bentgrass species differing in heat tolerance. J. Am. Soc. Hortic. Sci. 2007, 132, 185–192. [Google Scholar] [CrossRef]
- Chao, Z.; Yu, W.; Zhao, T.D.; Zhao, L. Global transcriptional analysis reveals the complex relationship between tea quality, leaf senescence and the responses to cold-drought combined stress in Camellia sinensis. Front. Plant Sci. 2016, 7, 1858. [Google Scholar]
- Zhao, L.J.; Huang, Y.X.; Paglia, K.; Vaniya, A.; Wancewicz, B. Metabolomics reveals the molecular mechanisms of copper induced cucumber leaf (Cucumis sativus) senescence. Environ. Sci. Technol. 2018, 52, 7092–7100. [Google Scholar] [CrossRef]
- Dube, B.K.; Sinha, P.; Shukla, K.; Chatterjee, C.; Pandey, V.K.; Rai, A.D. Involvement of excess cadmium on oxidative stress and other physiological parameters of eggplant. J. Plant Nutr. 2009, 32, 9. [Google Scholar] [CrossRef]
- Wang, H.B.; Wang, S.; Wang, X.D. Effects of light quality on leaf senescence and endogenous hormones content in grapevine under protected cultivation. Chin. J. Appl. Ecol. 2017, 28, 3535–3543. [Google Scholar]
- Causin, H.F.; Jauregui, R.N.; Barneix, A.J. The effect of light spectral quality on leaf senescence and oxidative stress in wheat. Plant Sci. 2006, 171, 24–33. [Google Scholar] [CrossRef]
- Zhao, J.Q.; Zhao, F.Q.; Jian, G.L.; Ye, Y.X.; Zhang, J.S.; Li, J.S.; Qi, F.J. Intensified Alternaria spot disease under potassium deficiency conditions results in acceleration of cotton (Gossypium hirsutum L.) leaf senescence. Aust. J. Crop Sci. 2013, 7, 241–248. [Google Scholar]
- Ding, N.; Jiang, Y.M.; Peng, F.T.; Chen, Q.; Wang, F.L.; Zhou, E.D. Effect of topdressing nitrogen application on leaf senescence and 15 N-urea of absorption, distribution and utilization in potted ‘Fuji ’apple. Sci. Agric. Sin. 2012, 45, 4025–4031. [Google Scholar]
- Liu, W.; Zhang, W.W.; Zheng, N.; Zhai, W.B.; Qi, F.J. Study of cotton leaf senescence induced by Alternaria alternata infection. Methods Mol. Biol. 2018, 1744, 161–171. [Google Scholar]
- Land, C.J.; Lawrence, K.S.; Burmester, C.H.; Meyer, B. Cultivar, irrigation, and soil contribution to the enhancement of verticillium wilt disease in cotton. Crop Prot. 2017, 96, 1–6. [Google Scholar] [CrossRef]
- Ghanem, M.E.; Ghars, M.A.; Frettinger, P.; Perez-Alfocea, F.; Lutts, S.; Wathelet, J.P.; du Jardin, P.; Fauconnier, M.L. Organdependent oxylipin signature in leaves and roots of salinized tomato plants (Solanum lycopersicum). J. Plant Physiol. 2012, 169, 1090–1101. [Google Scholar] [CrossRef]
- Rosenthal, S.I. Effects of air temperature, photoperiod and leaf age on foliar senescence of western larch (Larix occidentalis Nutt.) in environmentally controlled chambers. Plant Cell Environ. 2010, 19, 1057–1065. [Google Scholar] [CrossRef]
- Guo, Y.F. Hormone treatments in studying leaf senescence. Methods Mol. Biol. 2018, 1744, 125–132. [Google Scholar]
- Shen, F.F.; Yu, S.X.; Fan, S.; Li, J.; Huang, Z.M. The relationship between hormone and membrance lipid peroxidation in cotton leaf during senescence. J. Plant Physiol. Mol. Biol. 2003, 29, 589–592. [Google Scholar]
- Xiao, X.O.; Zeng, Y.M.; Cao, B.H.; Lei, J.J.; Chen, Q.H.; Meng, C.M.; Cheng, Y.J. PSAG12-IPT overexpression in eggplant delays leaf senescence and induces abiotic stress tolerance. J. Hortic. Sci. Biotechnol. 2017, 92, 349–357. [Google Scholar] [CrossRef]
- Li, Z.H.; Zhao, Y.; Liu, X.C.; Jiang, Z.Q.; Peng, J.Y.; Jin, J.P.; Guo, H.W.; Luo, J.C. Construction of the leaf senescence database and functional assessment of senescence-associated genes. Methods Mol. Biol. 2017, 1533, 315–333. [Google Scholar]
- Ma, X.M.; Zhang, Y.J.; Turečková, V.; Xue, G.P.; Fernie, A.R.; Mueller-Roeber, B.; Balazadeh, S. The NAC transcription factor SlNAP2 regulates leaf senescence and fruit yield in tomato. Plant Physiol. 2018, 177, 00292. [Google Scholar] [CrossRef]
- Liu, P.; Zhang, S.B.; Zhou, B.; Luo, X.; Zhou, X.F.; Cai, B.; Jin, Y.H.; Niu, D.; Lin, J.X.; Cao, X.F.; et al. The histone H3K4 demethylase JMJ16 represses leaf senescence in Arabidopsis. Plant Cell 2019, 31, 430–443. [Google Scholar] [CrossRef]
- Li, Z.H.; Guo, H.W. Ethylene treatment in studying leaf senescence in Arabidopsis. Methods Mol. Biol. 2018, 1744, 105–112. [Google Scholar]
- Liang, C.Z.; Chu, C.C. Towards understanding abscisic acid-mediated leaf senescence. Sci. China Life Sci. 2015, 58, 506–508. [Google Scholar] [CrossRef] [Green Version]
- Morris, K.; -Mackerness, S.A.-H.; Page, T.; John, F.C.; Murphy, A.M.; Carr, J.P.; Buchanan-Wollaston, V. Salicylic acid has a role in regulating gene expression during leaf senescence. Plant J. 2000, 23, 677–685. [Google Scholar] [CrossRef]
- Hu, Y.R.; Jiang, Y.J.; Han, X.; Wang, H.P.; Pan, J.J.; Yu, D.Q. Jasmonate regulates leaf senescence and tolerance to cold stress: Crosstalk with other phytohormones. J. Exp. Bot. 2017, 68, 1361–1369. [Google Scholar] [CrossRef]
- Back, A.; Richmond, A.E. Interrelations between gibberellic acid, cytokinins and abscisic acid in retarding leaf senescence. Physiol. Plant. 1971, 24, 76–79. [Google Scholar] [CrossRef]
- Gören, N.; Çağ, S. The effect of indole-3-acetic acid and benzyladenine on sequential leaf senescence on Helianthus annuus L. seedlings. Biotechnol. Biotechnol. Equip. 2007, 21, 322–327. [Google Scholar] [CrossRef]
- Kim, J.I.; Murphy, A.S.; Baek, D.; Lee, S.W.; Yun, D.J.; Bressan, R.A.; Narasimhan, M.L. YUCCA6 over-expression demonstrates auxin function in delaying leaf senescence in Arabidopsis thaliana. J. Exp. Bot. 2011, 62, 3981–3992. [Google Scholar] [CrossRef]
- Back, A.; Richmond, A. An interaction between the effects of kinetin and gibberellin in retarding leaf senescence. Physiol. Plant. 2010, 22, 1207–1216. [Google Scholar] [CrossRef]
- Masi, L.D.; Castaldo, D.; Pignone, D.; Servillo, L.; Facchiano, A. Experimental evidence and in silico identification of tryptophan decarboxylase in Citrus Genus. Molecules 2017, 22, 272. [Google Scholar] [CrossRef]
- Gong, B.; Shi, Q.H. Review of melatonin in horticultural crop. Sci. Agric. Sin. 2017, 50, 2326–2337. [Google Scholar]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin promotes adventitious and lateral root regeneration in etiolated hypocotyls of Lupinus albus L. J. Pineal Res. 2007, 42, 147–152. [Google Scholar] [CrossRef]
- Feng, L.J. Effects of plant growth substances on seed germination, plant growth development of rape. Ph.D. Thesis, Huazhong Agricultural University, Wuhan, China, 2015. [Google Scholar]
- Vitalini, S.; Gardana, C.; Zanzotto, A.; Simonetti, P.; Faoro, F.; Fico, G.; Iriti, M. The presence of melatonin in grapevine (Vitis vinifera L.) berry tissues. J. Pineal Res. 2011, 51, 331–337. [Google Scholar] [CrossRef]
- Liu, J.L.; Zhang, R.M.; Sun, Y.K.; Liu, Z.Y.; Jin, W.; Sun, Y. The beneficial effects of exogenous melatonin on tomato fruit properties. Sci. Hortic. 2016, 207, 14–20. [Google Scholar] [CrossRef]
- Ding, F.; Liu, B.; Zhang, S.X. Exogenous melatonin ameliorates cold-induced damage in tomato plants. Sci. Hortic. 2017, 219, 264–271. [Google Scholar] [CrossRef]
- Meng, J.F.; Xu, T.F.; Wang, Z.Z.; Fang, Y.L.; Xi, Z.M.; Zhang, Z.W. The ameliorative effects of exogenous melatonin on grape cuttings under water-deficient stress: Antioxidant metabolites, leaf anatomy, and chloroplast morphology. J. Pineal Res. 2014, 57, 200–212. [Google Scholar] [CrossRef]
- Kabiri, R.; Hatami, A.; Oloumi, H.; Naghizadeh, M.; Nasibi, F.; Tahmasebi, Z. Foliar application of melatonin induces tolerance to drought stress in Moldavian balm plants (Dracocephalum moldavica) through regulating the antioxidant system. Folia Hortic. 2018, 30, 155–167. [Google Scholar] [CrossRef]
- Wang, L.Y.; Liu, J.L.; Wang, W.X.; Sun, Y. Exogenous melatonin improves growth and photosynthetic capacity of cucumber under salinity-induced stress. Photosynthetica 2016, 54, 19–27. [Google Scholar] [CrossRef]
- Zhang, J.; Li, H.B.; Xu, B.; Li, J.; Huang, B.R. Exogenous melatonin suppresses dark-induced leaf senescence by activating the superoxide dismutase-catalase antioxidant pathway and down-regulating chlorophyll degradation in excised leaves of perennial ryegrass (Lolium perenne L.). Front. Plant Sci. 2016, 7, 1500. [Google Scholar] [CrossRef]
- Liang, D.; Shen, Y.Q.; Ni, Z.Y.; Wang, Q.; Lei, Z.; Xu, N.Q.; Deng, Q.X.; Lin, L.J.; Wang, J.; Lv, X.L.; et al. Exogenous melatonin application delays senescence of kiwifruit leaves by regulating the antioxidant capacity and biosynthesis of flavonoids. Front. Plant Sci. 2018, 9, 426. [Google Scholar] [CrossRef]
- Wang, P.; Yin, L.H.; Liang, D.; Li, C.; Ma, F.W.; Yue, Z.Y. Delayed senescence of apple leaves by exogenous melatonin treatment: Toward regulating the ascorbate–glutathione cycle. J. Pineal Res. 2012, 53, 11–20. [Google Scholar] [CrossRef]
- Munne-Bosch, S.; Alegre, L. Plant aging increases oxidative stress in chloroplasts. Planta 2002, 214, 608–615. [Google Scholar]
- Thompson, J.E.; Barber, R.F. The role of free radicals in senescence and wounding. N. Phytol. 1987, 105, 317–344. [Google Scholar] [CrossRef]
- Khanna-Chopra, R. Leaf senescence and abiotic stresses share reactive oxygen species-mediated chloroplast degradation. Protoplasma 2012, 249, 469–481. [Google Scholar] [CrossRef]
- Wang, P.; Sun, X.; Li, C.; Wei, Z.W.; Liang, D.; Ma, F.W. Long-term exogenous application of melatonin delays drought-induced leaf senescence in apple. J. Pineal Res. 2013, 54, 292–302. [Google Scholar] [CrossRef]
- Rodriguez, C.; Mayo, J.C.; Sainz, R.M.; Antolín, I.; Herrera, F.; Martín, V.; Reiter, R.J. Regulation of antioxidant enzymes: A significant role for melatonin. J. Pineal Res. 2004, 36, 1–9. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.-X.; Galano, A. Melatonin reduces lipid peroxidation and membrane viscosity. Front. Physiol. 2014, 5, 377. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Wellburn, A.R. Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem. Soc. Trans. 1983, 11, 591–592. [Google Scholar] [CrossRef]
- Cao, J.K.; Jiang, W.B.; Zhao, Y.M. Physiological and Biochemical Experiments of Postharvest Fruits and Vegetables; China Light Industrial Press Ltd.: Beijing, China, 2007; Volume 9, pp. 154–156. [Google Scholar]
- Dionisio-Sese, M.L.; Tobita, S. Antioxidant responses of rice seedlings to salinity stress. Plant Sci. 1998, 135, 1–9. [Google Scholar] [CrossRef]
- Kakkar, P.S.; Das, B.; Viswanathan, P.N. A modified spectrophotometric assay of superoxide dimutase. Indian J. Biochem. Biol. 1984, 21, 130–132. [Google Scholar]
- Rao, M.; Paliyath, G.; Ormrod, D.P. Ultraviolet-B- and ozone-induced biochemical changes in antioxidant enzymes of Arabidopsis thaliana. Plant Physiol. 1996, 110, 125–136. [Google Scholar] [CrossRef]
- Dhindsa, R.S.; Plumb-Dhindsa, P.; Thorpe, T.A. Leaf senescence: Correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J. Exp. Bot. 1981, 32, 93–101. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar]
- Carlberg, I.; Mannervik, B. Glutathione reductase. Method Enzymol. 1985, 113, 484–490. [Google Scholar]
- Kampfenkel, K.; Vanmontagu, M.; Inze, D. Extraction and determination of ascorbate and dehydroascorbate from plant tissue. Anal. Biochem. 1995, 225, 165–167. [Google Scholar] [CrossRef] [PubMed]
- Griffith, O.W. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal. Biochem. 1980, 106, 207–212. [Google Scholar] [CrossRef]
- Zhang, Z.K.; Huber, D.J.; Qu, H.X.; Wang, H.; Huang, Z.H.; Huang, H.; Jiang, Y.M. Enzymatic browning and antioxidant activities in harvested litchi fruit as influenced by apple polyphenols. Food Chem. 2015, 171, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Yahmed, B.J.; de Oliveira, T.M.; Novillo, P.; Quinones, A.; Forner, M.A.; Salvador, A.; Froelicher, M.; Mimoun, M.B.; Talon, M.; Ollitrault, P.; et al. A simple, fast and inexpensive method to assess salt stress tolerance of aerial plant part: Investigations in the mandarin group. J. Plant Physiol. 2016, 190, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Espinoza, C.; Medina, C.; Somerville, S.; Arce-Johnson, P. Senescence-associated genes induced during compatible viral interactions with grapevine and Arabidopsis. J. Exp. Bot. 2007, 58, 3197–3212. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.Q.; Shi, S.T.; Dou, F.F.; Song, Y.; Ma, F.W. Exogenous melatonin alleviates alkaline stress in Malus hupehensis Rehd. by regulating the biosynthesis of polyamines. Molecules 2017, 22, 1542. [Google Scholar] [CrossRef]
- Bowler, C.; Montagu, M.V.; Inze, D. Superoxide dismutase and stress tolerance. Annu. Rev. Plant Biol. 1992, 43, 81–116. [Google Scholar] [CrossRef]
- Weaver, L.M.; Amasino, R.M. Senescence is induced in individually darkened Arabidopsis leaves, but inhibited in whole darkened plants. Plant Physiol. 2001, 127, 876–886. [Google Scholar] [CrossRef]
- Buchanan-Wollaston, V.; Page, T.; Harrison, E.; Breeze, E.; Lim, P.O.; Nam, H.G.; Lin, J.F.; Wu, S.H.; Swidzinski, J.; Ishizaki, K.; et al. Comparative transcriptome analysis reveals significant differences in gene expression and signaling pathways between developmental and dark/starvation-induced senescence in Arabidopsis. Plant J. 2005, 42, 567–585. [Google Scholar] [CrossRef]
- Nooden, L.D.; Penney, J.P. Correlative controls of senescence and plant death in Arabidopsis thaliana (Brassicaceae). J. Exp. Bot. 2001, 52, 2151–2159. [Google Scholar] [CrossRef]
- Hortensteiner, S.; Krautler, B. Chlorophyll breakdown in higher plants. Biochim. Biophys. Acta 2011, 1807, 977–988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ke, X.W.; Xu, P.; Yin, L.H.; Wang, Z.H.; Zhang, P.P.; Diao, J.J.; Zuo, Y.H.; Zheng, D.F. Exogenous application of melatonin delays leaf senescence in Adzuki bean. J. Heilongjiang Bayi Agric. Univ. 2015, 27, 52–55. [Google Scholar]
- Zhang, Z.P.; Miao, M.M.; Wang, C.L. Effects of ALA on photosynthesis, antioxidant enzyme activity, and gene expression, and regulation of proline accumulation in tomato seedlings under NaCl stress. J. Plant Growth Regul. 2015, 34, 637–650. [Google Scholar] [CrossRef]
- Tan, D.X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Reiter, R.J. One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species? J. Pineal Res. 2007, 42, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Melchiorri, D.; Sewerynek, E.; Poeggeler, B.; Barlow-Walden, L.; Chuang, J.; Ortiz, G.G.; Acuña-Castroviejo, D. A review of the evidence supporting melatonin’s role as an antioxidant. J. Pineal Res. 2010, 18, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Chen, Y.; Tan, D.X.; Reiter, R.J.; Chan, Z.; He, C. Melatonin induces nitric oxide and the potential mechanisms relate to innate immunity against bacterial pathogen infection in Arabidopsis. J. Pineal Res. 2015, 59, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.A.; Islam, F.; Yang, C.; Nawaz, A.; Athar, H.R.; Gill, R.A.; Ali, B.; Song, W.J.; Zhou, W.J. Methyl jasmonate alleviates arsenic-induced oxidative damage and modulates the ascorbate-glutathione cycle in oilseed rape roots. Plant Growth Regul. 2017, 84, 135–148. [Google Scholar] [CrossRef]
- Li, L.J.; Lu, X.C.; Ma, H.Y.; Lyu, D.G. Jasmonic acid regulates the ascorbate–glutathione cycle in Malus baccata Borkh. roots under low root-zone temperature. Acta Physiol. Plant. 2017, 39, 174. [Google Scholar] [CrossRef]
- Mir, B.A.; Mir, S.A.; Khazir, J.; Tonfack, L.B.; Cowan, D.A.; Vyasa, D.; Koula, S. Cold stress affects antioxidative response and accumulation of medicinally important withanolides in Withania somnifera (L.) dunal. Ind. Crop Prod. 2015, 74, 1008–1016. [Google Scholar] [CrossRef]
- Han, M.; Cao, B.L.; Liu, S.S.; Xu, K. Effects of rootstock and scion interactions on ascorbate-glutathione cycle in tomato seedlings under low temperature stress. Acta Hortic. Sin. 2019, 46, 65–73. [Google Scholar]
- Chen, L.G.; Xiang, S.; Chen, Y.; Li, D.B.; Yu, D.Q. Arabidopsis WRKY45 interacts with the DELLA protein RGL1 to positively regulate age-triggered leaf senescence. Mol. Plant 2017, 10, 1174–1189. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.; Liu, J.T.; Yang, L.F.; Xie, G.S. Survey of the rubber tree genome reveals a high number of cysteine protease-encoding genes homologous to Arabidopsis SAG12. PLoS ONE 2017, 12, e0171725. [Google Scholar] [CrossRef] [PubMed]
- Noh, Y.S.; Amasino, R.M. Identification of a promoter region responsible for the senescence-specific expression of SAG12. Plant Mol. Biol. 1999, 41, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Rosenvasser, S.; Mayak, S.; Friedman, H. Increase in reactive oxygen species (ROS) and in senescence-associated gene transcript (SAG) levels during dark-induced senescence of Pelargonium cuttings, and the effect of gibberellic acid. Plant Sci. 2006, 170, 873–879. [Google Scholar] [CrossRef]
- Navabpour, S.; Morris, K.; Allen, R.; Harrison, E.; Mackerness, S.A.-H.; Buchanan-Wollaston, V. Expression of senescence-enhanced genes in response to oxidative stress. J. Exp. Bot. 2003, 54, 2285–2292. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.Q.; Loake, G.J.; Chu, C.C. Cross-talk of nitric oxide and reactive oxygen species in plant programed cell death. Front. Plant Sci. 2013, 4, 314. [Google Scholar] [CrossRef] [PubMed] [Green Version]






| Gene Name | Accession Number | Forward Primer Sequence(5′–3′) | Reverse Primer Sequence(5′–3′) |
|---|---|---|---|
| SAG12-Vv | XM002284937.3 | TGAAGGATGCAATGGGGGAC | TCTGCCATCGGAAGCTTTGT |
| SAG13-Vv | XM002282719.4 | TCCTACAAGTGTTTGTGAACGC | ATAGTGGAGCCATCCCCTGA |
| Ubiquitin-Vv | XM003634272.3 | GCTCGCTGTTTTGCAGTTCTAC | AACATAGGTGAGGCCGCACTT |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, X.; Xu, S.; Mu, D.; Sadeghnezhad, E.; Li, Q.; Ma, Z.; Zhao, L.; Zhang, Q.; Wang, L. Exogenous Melatonin Delays Dark-Induced Grape Leaf Senescence by Regulation of Antioxidant System and Senescence Associated Genes (SAGs). Plants 2019, 8, 366. https://doi.org/10.3390/plants8100366
Shi X, Xu S, Mu D, Sadeghnezhad E, Li Q, Ma Z, Zhao L, Zhang Q, Wang L. Exogenous Melatonin Delays Dark-Induced Grape Leaf Senescence by Regulation of Antioxidant System and Senescence Associated Genes (SAGs). Plants. 2019; 8(10):366. https://doi.org/10.3390/plants8100366
Chicago/Turabian StyleShi, Xingyun, Shanshan Xu, Desheng Mu, Ehsan Sadeghnezhad, Qiang Li, Zonghuan Ma, Lianxin Zhao, Qinde Zhang, and Lixin Wang. 2019. "Exogenous Melatonin Delays Dark-Induced Grape Leaf Senescence by Regulation of Antioxidant System and Senescence Associated Genes (SAGs)" Plants 8, no. 10: 366. https://doi.org/10.3390/plants8100366





