Analysis of Stored mRNA Degradation in Acceleratedly Aged Seeds of Wheat and Canola in Comparison to Arabidopsis
Abstract
:1. Introduction
2. Results
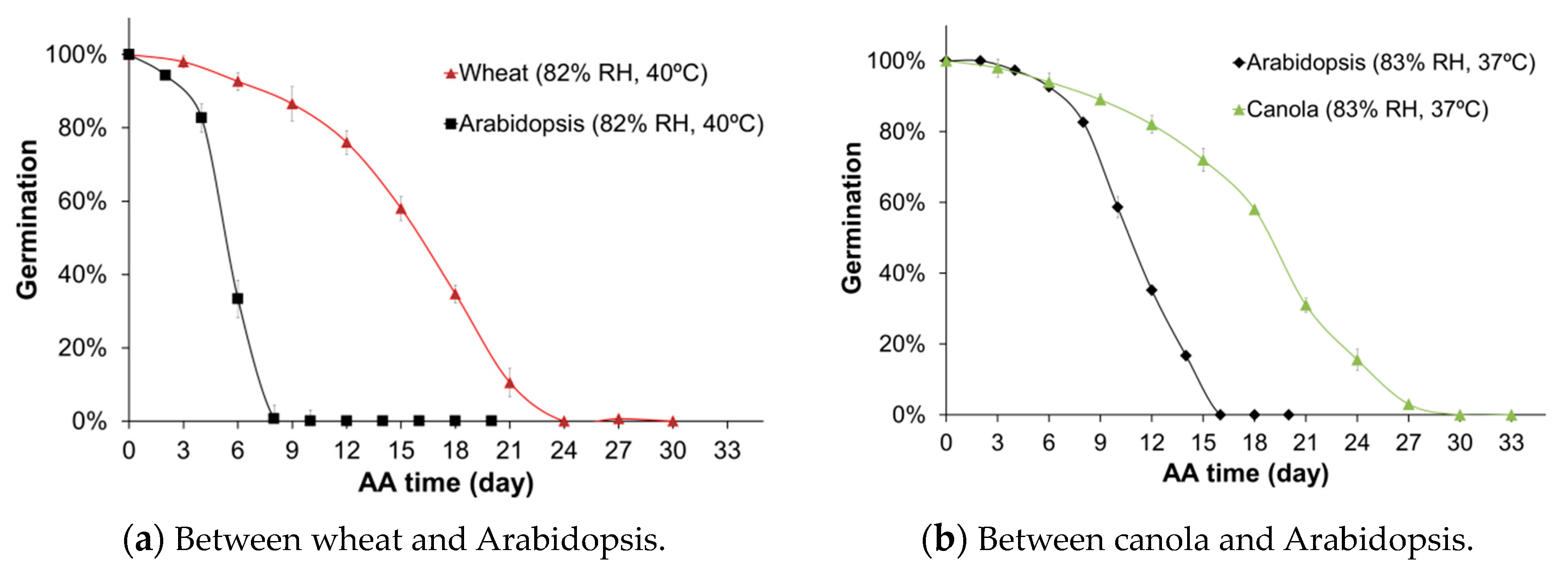
2.1. Germination Kinetics for Wheat, Canola, and Arabidopsis Seeds
2.2. Similar Quality of RNA Samples for Aged Wheat and Canola Seeds
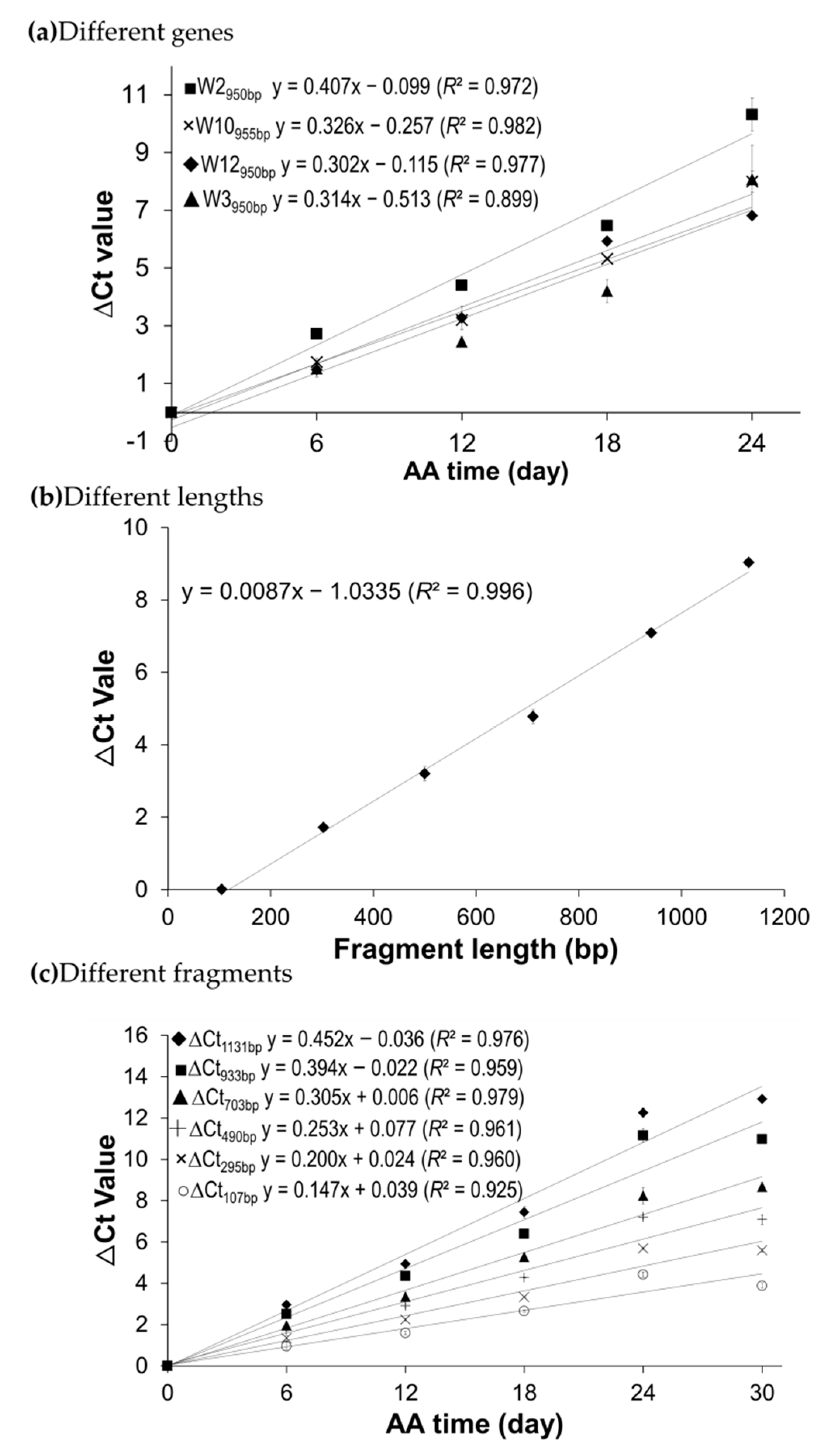
2.3. Correlation of mRNA Degradation with Aging Time and Fragment Length
2.4. Similar Rate of mRNA Degradation in Aged Seeds of Wheat, Canola and Arabidopsis
3. Discussion
3.1. Stored mRNAs Were Gradually Degraded at a Constant Rate over Aging Time in Wheat and Canola Seeds
3.2. The β Value Can Be Used to Describe the Rate of mRNA Degradation Quantitatively in Different Plants
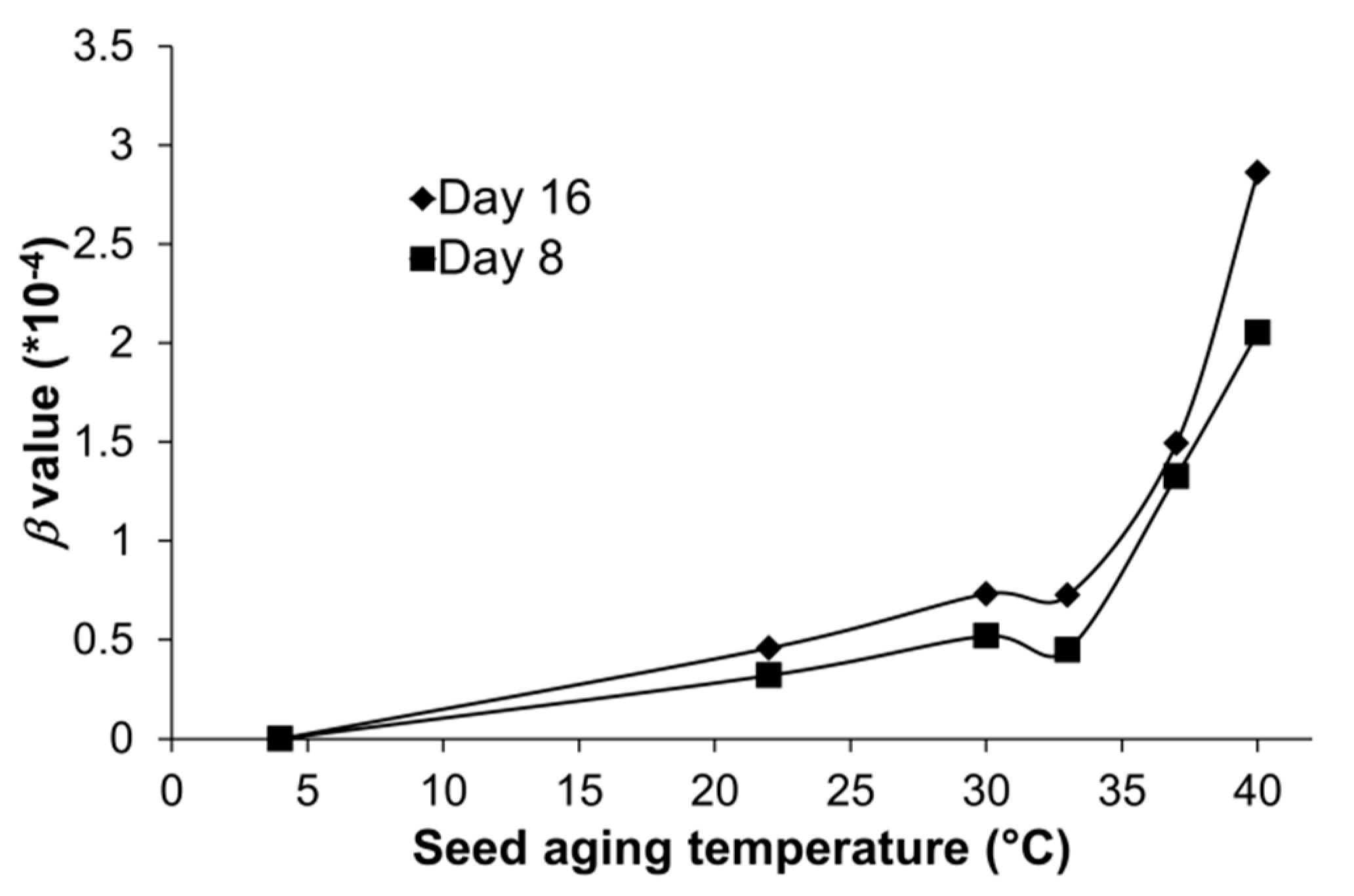
3.3. β Values for AA Seeds of Arabidopsis Increased Non-Linearly with High Temperatures
3.4. Practical Implications for Plant Germplasm Conservation
4. Materials and Methods
4.1. Comparing Seed Germination Kinetics in Wheat, Canola, and Arabidopsis Seeds
4.2. Total RNA Analysis for Differently Aged Seeds of Wheat, Canola, and Arabidopsis
4.3. qPCR Analysis of mRNA Degradation in Aged Seeds
4.4. Estimating the Rate of Stored mRNA Degradation in Aged Seeds
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Walters, C. Genebanking seeds from natural populations. Nat. Area J. 2015, 35, 98–105. [Google Scholar] [CrossRef]
- Fu, Y.-B.; Ahmed, Z.; Diederichsen, A. Towards a better monitoring of seed ageing under ex situ seed conservation. Conserv. Physiol. 2015, 3, cov206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hay, F.R.; Probert, R.J. Advances in seed conservation of wild plant species: A review of recent research. Conserv. Physiol. 2013, 1, cot030. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, D.; Pritchard, H.W.; Walters, C. Dry architecture: Towards the understanding of the variation of longevity in desiccation-tolerant germplasm. Seed Sci. Res. 2020, 30, 142–155. [Google Scholar] [CrossRef]
- Wiebach, J.; Nagel, M.; Börner, A.; Altmann, T.; Riewe, D. Age-dependent loss of seed viability is associated with increased lipid oxidation and hydrolysis. Plant Cell Environ. 2020, 43, 303–314. [Google Scholar] [CrossRef] [Green Version]
- Zinsmeister, J.; Leprince, O.; Buitink, J. Molecular and environmental factors regulating seed longevity. Biochem. J. 2020, 477, 305–323. [Google Scholar] [CrossRef]
- Zhou, W.; Chen, F.; Luo, X.; Dai, Y.; Yang, Y.; Zheng, C.; Yang, W.; Shu, K. A matter of life and death: Molecular, physiological, and environmental regulation of seed longevity. Plant Cell Environ. 2020, 43, 293–302. [Google Scholar] [CrossRef]
- Colville, L.; Pritchard, H.W. Seed life span and food security. New Phytol. 2019, 224, 557–562. [Google Scholar] [CrossRef] [Green Version]
- Ballesteros, D.; Walters, C. Solid-state biology and seed longevity: A mechanical analysis of glasses in pea and soybean embryonic axes. Front. Plant Sci. 2019, 10, 920. [Google Scholar] [CrossRef] [Green Version]
- Roach, T.; Nagel, M.; Börner, A.; Eberle, C.; Kranner, I. Changes in tocochromanols and glutathione reveal differences in the mechanisms of seed ageing under seedbank conditions and controlled deterioration in Barley. Environ. Exp. Bot. 2018, 156, 8–15. [Google Scholar] [CrossRef]
- Sano, N.; Rajjou, L.; North, H.M.; Debeaujon, I.; Marion-Poll, A.; Seo, M. Staying alive: Molecular aspects of seed longevity. Plant Cell Physiol. 2016, 57, 660–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurek, K.; Plitta-Michalak, B.; Ratajczak, E. Reactive oxygen species as potential drivers of the seed aging process. Plants 2019, 8, 174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, M.; He, D.; Teng, H.; Chen, L.; Song, H.; Huang, Q. Physiological and proteomic analyses of coix seed aging during storage. Food Chem. 2018, 260, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Bai, B.; van Der Horst, S.; Cordewener, J.H.; America, T.A.; Hanson, J.; Bentsink, L. Seed stored mRNAs that are specifically associated to monosomes are translationally regulated during germination. Plant Physiol. 2020, 182, 378–392. [Google Scholar] [CrossRef] [Green Version]
- FAO. The Second Report on the State of the World’s Plant Genetic Resources for Food and Agriculture; FAO: Rome, Italy, 2010. [Google Scholar]
- Walters, C.; Wheeler, L.M.; Grotenhuis, J.M. Longevity of seeds stored in a genebank: Species characteristics. Seed Sci. Res. 2005, 15, 1–20. [Google Scholar] [CrossRef]
- Van Treuren, R.; de Groot, E.C.; van Hintum, T.J. Preservation of seed viability during 25 years of storage under standard genebank conditions. Genet. Resour. Crop Evol. 2013, 60, 1407–1421. [Google Scholar] [CrossRef]
- FAO. Genebank Standards for Plant Genetic Resources for Food and Agriculture. Available online: http://www.fao.org/3/a-i3704e.pdf (accessed on 29 August 2020).
- Pritchard, H.W.; Dickie, J. Predicting seed longevity: The use and abuse of seed viability equations. In Seed Conservation: Turning Science into Practice; Smith, R.D., Dickie, J.B., Linington, S.H., Pritchard, H.W., Probert, R.J., Eds.; Royal Botanic Gardens: Kew, UK, 2003; pp. 653–721. [Google Scholar]
- Fu, Y.-B.; Yang, M.H.; Horbach, C.; Kessler, D.; Diederichsen, A.; You, F.M.; Wang, H. Patterns of SSR variation in bread wheat (Triticum aestivum L.) seeds under ex situ genebank storage and accelerated ageing. Genet. Resour. Crop Evol. 2017, 64, 277–290. [Google Scholar] [CrossRef]
- Groot, S.P.; de Groot, L.; Kodde, J.; van Treuren, R. Prolonging the longevity of ex situ conserved seeds by storage under anoxia. Plant Genet. Resour. 2014, 13, 18–26. [Google Scholar] [CrossRef] [Green Version]
- Walters, C.; Ballesteros, D.; Vertucci, V.A. Structural mechanics of seed deterioration: Standing the test of time. Plant Sci. 2010, 179, 565–573. [Google Scholar] [CrossRef]
- Ballesteros, D.; Walters, C. Detailed characterization of mechanical properties and molecular mobility within dry seed glasses: Relevance to the physiology of dry biological systems. Plant J. 2011, 68, 607–619. [Google Scholar] [CrossRef]
- Kranner, I.; Kastberger, G.; Hartbauer, M.; Pritchard, H.W. Noninvasive diagnosis of seed viability using infrared thermography. Proc. Natl. Acad. Sci. USA 2010, 107, 3912–3917. [Google Scholar] [CrossRef] [Green Version]
- Colville, L.; Bradley, E.L.; Lloyd, A.S.; Pritchard, H.W.; Castle, L.; Kranner, I. Volatile fingerprints of seeds of four species indicate the involvement of alcoholic fermentation, lipid peroxidation, and maillard reactions in seed deterioration during ageing and desiccation stress. J. Exp. Bot. 2012, 63, 6519–6530. [Google Scholar] [CrossRef] [PubMed]
- Umarani, R.; Bhaskaran, M.; Vanitha, C.; Tilak, M. Fingerprinting of volatile organic compounds for quick assessment of vigour status of seeds. Seed Sci. Res. 2020, 30, 112–121. [Google Scholar] [CrossRef]
- Mira, S.; Hill, L.M.; González-Benito, M.E.; Ibáñez, M.A.; Walters, C. Volatile emission in dry seeds as a way to probe chemical reactions during initial asymptomatic deterioration. J. Exp. Bot. 2016, 67, 1783–1793. [Google Scholar] [CrossRef] [PubMed]
- Kranner, I.; Birtić, S.; Anderson, K.M.; Pritchard, H.W. Glutathione half-cell reduction potential: A universal stress marker and modulator of programmed cell death? Free Radical. Biol. Med. 2006, 40, 2155–2165. [Google Scholar] [CrossRef] [PubMed]
- Fleming, M.B.; Hill, L.M.; Walters, C. The kinetics of ageing in dry-stored seeds: A comparison of viability loss and RNA degradation in unique legacy seed collections. Ann. Bot. 2019, 123, 1133–1146. [Google Scholar] [CrossRef] [Green Version]
- Fleming, M.B.; Richards, C.M.; Walters, C. Decline in RNA integrity of dry-stored soybean seeds correlates with loss of germination potential. J. Exp. Bot. 2017, 68, 2219–2230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Priestley, D.A. Seed Aging; Cornell University Press: Ithaca, NY, USA, 1986; pp. 39–197. [Google Scholar]
- Rajjou, L.; Debeaujon, I. Seed longevity: Survival and maintenance of high germination ability of dry seeds. C. R. Biol. 2008, 331, 796–805. [Google Scholar] [CrossRef] [PubMed]
- Pukacka, S.; Ratajczak, E. Ascorbate and glutathione metabolism during development and desiccation of orthodox and recalcitrant seeds of the genus Acer. Funct. Plant Biol. 2007, 34, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Møller, I.M. Plant mitochondria and oxidative stress: Electron transport, NADPH turnover, and metabolism of reactive oygen species. Annu. Rev. Plant Biol. 2001, 52, 561–591. [Google Scholar] [CrossRef] [Green Version]
- Spanò, C.; Bottega, S.; Lorenzi, R.; Grilli, I. Ageing in embryos from wheat grains stored at different temperatures: Oxidative stress and antioxidant response. Funct. Plant Biol. 2011, 38, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Prieto-Dapena, P.; Castaño, R.; Almoguera, C.; Jordano, J. Improved resistance to controlled deterioration in transgenic seeds. Plant Physiol. 2006, 142, 1102–1112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Almoguera, C.; Prieto-Dapena, P.; Díaz-Martín, J.; Espinosa, J.M.; Carranco, R.; Jordano, J. The HaDREB2 transcription factor enhances basal thermotolerance and longevity of seeds through functional interaction with HaHSFA9. BMC Plant Biol. 2009, 9, 75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogé, L.; Bourdais, G.; Bove, J.; Collet, B.; Godin, B.; Granier, F.; Boutin, J.P.; Job, D.; Jullien, M.; Grappin, P. Protein repair L-isoaspartyl methyltransferase1 is involved in both seed longevity and germination vigor in Arabidopsis. Plant Cell 2008, 20, 3022–3037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, H.; Chu, P.; Zhou, Y.; Li, Y.; Liu, J.; Ding, Y.; Tsang, E.W.T.; Jiang, L.; Wu, K.; Huang, S. Overexpression of AtOGG1, a DNA glycosylase/AP lyase, enhances seed longevity and abiotic stress tolerance in Arabidopsis. J. Exp. Bot. 2012, 63, 4107–4121. [Google Scholar] [CrossRef] [Green Version]
- Kimura, M.; Nambara, E. Stored and neosynthesized mRNA in Arabidopsis seeds: Effects of cycloheximide and controlled deterioration treatment on the resumption of transcription during imbibition. Plant Mol. Biol. 2010, 73, 119–129. [Google Scholar] [CrossRef]
- Fleming, M.B.; Patterson, E.L.; Reeves, P.A.; Richards, C.M.; Gaines, T.A.; Walters, C. Exploring the fate of mRNA in aging seeds: Protection, destruction, or slow decay? J. Exp. Bot. 2018, 69, 4309–4321. [Google Scholar] [CrossRef]
- Wilson, D.O., Jr.; Mcdonald, M.B., Jr. The lipid-peroxidation model of seed aging. Seed Sci. Technol. 1986, 14, 269–300. [Google Scholar]
- Loiseau, J.; Ly Vu, B.; Macherel, M.H.; Deunff, Y.L. Seed lipoxygenases: Occurrence and functions. Seed Sci. Res. 2001, 11, 199–211. [Google Scholar]
- Seo, Y.S.; Kim, E.Y.; Kim, W.T. The Arabidopsis sn-1-specific mitochondrial acylhydrolase AtDLAH is positively correlated with seed viability. J. Exp. Bot. 2011, 62, 5683–5698. [Google Scholar] [CrossRef] [Green Version]
- Zhao, L.; Wang, S.; Fu, Y.-B.; Wang, H. Arabidopsis seed stored mRNAs are degraded constantly over aging time with the rate of degradation depending on aging conditions, as revealed by new quantification methods. Front. Plant Sci. 2020, 10, 1764. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Jordan, M.C.; Ayele, B.T. Microarray dataset of after-ripening induced mRNA oxidation in wheat seeds. Data Brief 2018, 21, 852–855. [Google Scholar] [CrossRef] [PubMed]
- Basbouss-Serhal, I.; Pateyron, S.; Cochet, F.; Leymarie, J.; Bailly, C. 5′ to 3′ mRNA decay contributes to the regulation of Arabidopsis seed germination by dormancy. Plant Physiol. 2017, 173, 1709–1723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Comai, L.; Dietrich, R.A.; Maslyar, D.J.; Baden, C.S.; Harada, J.J. Coordinate expression of transcriptionally regulated isocitrate lyase and malate synthase genes in Brassica napus L. Plant Cell 1989, 1, 293–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajjou, L.; Gallardo, K.; Debeaujon, I.; Vandekerckhove, J.; Job, C.; Job, D. The effect of α-Amanitin on the Arabidopsis seed proteome highlights the distinct roles of stored and neosynthesized mRNAs during germination. Plant Physiol. 2004, 134, 1598–1613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sano, N.; Ono, H.; Murata, K.; Yamada, T.; Hirasawa, T.; Kanekatsu, M. Accumulation of long-lived mRNAs associated with germination in embryos during seed development of rice. J. Exp. Bot. 2015, 66, 4035–4046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sano, N.; Permana, H.; Kumada, R.; Shinozaki, Y.; Tanabata, T.; Yamada, T.; Hirasawa, T.; Kanekatsu, M. Proteomic analysis of embryonic proteins synthesized from long-lived mRNAs during germination of rice seeds. Plant Cell Physiol. 2012, 53, 687–698. [Google Scholar] [CrossRef]
- Nakabayashi, K.; Okamoto, M.; Koshiba, T.; Kamiya, Y.; Nambara, E. Genome-wide profiling of stored mRNA in Arabidopsis thaliana seed germination: Epigenetic and genetic regulation of transcription in seed. Plant J. 2005, 41, 697–709. [Google Scholar] [CrossRef]
- Barciszewski, J.; Barciszewska, M.Z.; Siboska, G.; Rattan, S.I.; Clark, B.F. Some unusual nucleic acid bases are products of hydroxyl radical oxidation of DNA and RNA. Mol. Biol. Rep. 1999, 26, 231–238. [Google Scholar] [CrossRef]
- Job, D.; Bailly, C. Role of protein and mRNA oxidation in seed dormancy and germination. Front. Plant Sci. 2013, 4, 77. [Google Scholar]
- Ventura, L.; Dona, M.; Macovei, A.; Carbonera, D.; Buttafava, A.; Mondoni, A.; Rossi, G.; Balestrazzi, A. Understanding the molecular pathways associated with seed vigor. Plant Phsiol. Bioch. 2012, 60, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Rushton, P.J.; Bray, C.M. Stored and de novo synthesized polyadenylated RNA and loss of vigor and viability in wheat seed. Plant Sci. 1987, 51, 51–59. [Google Scholar] [CrossRef]
- Brocklehurst, P.A.; Fraser, R.S.S. Ribosomal RNA integrity and rate of seed germination. Planta 1980, 148, 417–421. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.; Bryant, J.A.; Brocklehurst, P.A. Changes in levels and integrity of ribosomal RNA during seed maturation and germination in carrot (Daucus carota L.). J. Exp. Bot. 1987, 38, 1343–1350. [Google Scholar] [CrossRef]
- Reuzeau, C.; Cavalie, G. Changes in RNA and protein metabolism associated with alterations in the germination efficiency of sunflower seeds. Ann. Bot. 1997, 80, 131–137. [Google Scholar] [CrossRef] [Green Version]
- Kranner, I.; Chen, H.; Pritchard, H.W.; Pearce, S.R.; Birtić, S. Inter-nucleosomal DNA fragmentation and loss of RNA integrity during seed ageing. Plant Growth Regul. 2011, 63, 63–72. [Google Scholar] [CrossRef]
- Sharma, S.N.; Maheshwari, A.; Sharma, C.; Shukla, N. Gene expression patterns regulating the seed metabolism in relation to deterioration/ageing of primed mung bean (Vigna radiata L.) seeds. Plant Physiol. Biochem. 2018, 124, 40–49. [Google Scholar] [CrossRef]
- Imbeaud, S.; Graudens, E.; Boulanger, V.; Barlet, X.; Zaborski, P.; Eveno, E.; Mueller, O.; Schroeder, A.; Auffray, C. Towards standardization of RNA quality assessment using user-independent classifiers of microcapillary electrophoresis traces. Nucleic Acids Res. 2005, 33, e56. [Google Scholar] [CrossRef] [Green Version]
- Schroeder, A.; Mueller, O.; Stocker, S.; Salowsky, R.; Leiber, M.; Gassmann, M.; Lightfoot, S.; Menzel, W.; Granzow, M.; Ragg, T. The RIN: An RNA integrity number for assigning integrity values to RNA measurements. BMC Mol. Biol. 2006, 7, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Buitink, J.; Leprince, O. Intracellular glasses and seed survival in the dry state. C.R. Biol. 2008, 331, 788–795. [Google Scholar] [CrossRef]
- Horvath, S.; Raj, K. DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nat. Rev. Genet. 2018, 19, 371. [Google Scholar] [CrossRef] [PubMed]
- Ganguli, S.; Sen-Mandi, S. Effects of aging on amylase activity and scutellar cell structure during imbibition in wheat seed. Ann. Bot. 1993, 71, 411–416. [Google Scholar] [CrossRef]
- Krishnan, P.; Nagarajan, S.; Moharir, A.V. Thermodynamic characterisation of seed deterioration during storage under accelerated ageing conditions. Biosyst. Eng. 2004, 89, 425–433. [Google Scholar] [CrossRef]
- Menezes, V.O.; Lopes, S.J.; Tedesco, S.B.; Henning, F.A.; Zen, H.D.; Mertz, L.M. Cytogenetic analysis of wheat seeds submitted to artificial ageing stress. J. Seed Sci. 2014, 36, 71–78. [Google Scholar] [CrossRef] [Green Version]
- Sugliani, M.; Rajjou, L.; Clerkx, E.J.; Koornneef, M.; Soppe, W.J. Natural modifiers of seed longevity in the Arabidopsis mutants abscisic acid insensitive3-5 (abi3-5) and leafy cotyledon1-3 (lec1-3). New Phytol. 2009, 184, 898–908. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and Bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Onate-Sanchez, L.; Vicente-Carbajosa, J. DNA-free RNA isolation protocols for Arabidopsis thaliana, including seeds and siliques. BMC Res. Notes 2008, 1, 93. [Google Scholar] [CrossRef] [Green Version]


| AA Day | Wheat | Canola | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 6 | 12 | 18 | 24 | 30 | 0 | 6 | 12 | 18 | 24 | 30 | ||
| Germination Percentage | 100.0 | 92.7 | 76.0 | 34.7 | 0.0 | 0.0 | 100.0 | 94.0 | 89.0 | 58.0 | 15.5 | 0.0 | |
| Yield (ng/μL) | a | 228.0 | 144.0 | 444.0 | 212.1 | 122.0 | 217.9 | 363.0 | 390.0 | 374.0 | 334.7 | 173.0 | 593.0 |
| b | 347.0 | 551.0 | 338.0 | 296.3 | 335.2 | 276.3 | 354.0 | 415.0 | 478.6 | 347.6 | 238.0 | 601.0 | |
| A280/260 | a | 2.02 | 2.02 | 2.03 | 2.07 | 2.05 | 2.10 | 2.10 | 2.11 | 2.04 | 2.07 | 1.95 | 2.10 |
| b | 1.98 | 1.98 | 2.06 | 2.01 | 2.09 | 2.08 | 2.09 | 2.11 | 2.07 | 2.10 | 2.03 | 2.13 | |
| A280/230 | a | 2.27 | 2.24 | 2.23 | 2.21 | 2.13 | 2.20 | 2.40 | 2.31 | 2.23 | 2.31 | 2.44 | 2.21 |
| b | 2.30 | 2.32 | 2.24 | 2.23 | 2.25 | 2.10 | 2.38 | 2.32 | 2.32 | 2.37 | 2.39 | 2.34 | |
| Fragment | β Value (Break per Nucleotide per Day × 10−4) | |||||
|---|---|---|---|---|---|---|
| Day 6 | Day 12 | Day 18 | Day 24 | Average | Overall | |
| W2950bp | 3.30 ± 0.10 | 2.68 ± 0.04 | 2.62 ± 0.06 | 3.14 ± 0.18 | 2.94 ± 0.15 | 2.26 ± 0.51 |
| W3950bp | 1.86 ± 0.22 | 1.49 ± 0.11 | 1.71 ± 0.16 | 2.46 ± 0.36 | 1.88 ± 0.18 | |
| W10955bp | 2.12 ± 0.15 | 1.94 ± 0.03 | 2.15 ± 0.02 | 2.42 ± 0.11 | 2.16 ± 0.09 | |
| W12950bp | 1.80 ± 0.31 | 1.99 ± 0.25 | 2.41 ± 0.06 | 2.07 ± 0.02 | 2.07 ± 0.11 | |
| Average | 2.27 ± 0.30 | 2.03 ± 0.21 | 2.22 ± 0.17 | 2.52 ± 0.19 | ||
| Fragment | β Value (Break per Nucleotide per Day × 10−4) | ||||||
|---|---|---|---|---|---|---|---|
| Day 6 | Day 12 | Day 18 | Day 24 | Day 30 | Average | Overall | |
| Bn111490bp | 1.73 ± 0.04 | 1.54 ± 0.06 | 1.28 ± 0.04 | 1.17 ± 0.04 | 1.33 ± 0.06 | 1.41 ± 0.05 | 1.37 ± 0.31 |
| Bn121500bp | 2.46 ± 0.24 | 1.84 ± 0.04 | 1.54 ± 0.07 | 1.35 ± 0.03 | 1.40 ± 0.05 | 1.72 ± 0.09 | |
| Bn131486bp | 1.51 ± 0.05 | 1.44 ± 0.02 | 1.15 ± 0.02 | 1.03 ± 0.00 | 1.29 ± 0.02 | 1.28 ± 0.02 | |
| Bn141500bp | 1.44 ± 0.10 | 1.20 ± 0.10 | 1.02 ± 0.02 | 0.89 ± 0.02 | 1.20 ± 0.02 | 1.15 ± 0.05 | |
| Bn171500bp | 1.35 ± 0.20 | 1.39 ± 0.01 | 1.17 ± 0.03 | 1.44 ± 0.01 | 1.12 ± 0.06 | 1.29 ± 0.06 | |
| Average | 1.70 ± 0.13 | 1.48 ± 0.05 | 1.23 ± 0.04 | 1.18 ± 0.02 | 1.27 ± 0.04 | ||
| Gene | AA Days | β Value (Break per Nucleotide per Day × 10−4) | ||||
|---|---|---|---|---|---|---|
| 22 °C | 30 °C | 33 °C | 37 °C | 40 °C | ||
| At1G743102000bp | 8 | 0.32 ± 0.06 | 0.61 ± 0.07 | 0.37 ± 0.05 | 1.33 ± 0.11 | 2.05 ± 0.15 |
| 16 | 0.46 ± 0.09 | 0.73 ± 0.06 | 0.73 ± 0.03 | 1.49 ± 0.08 | 2.86 ± 0.24 | |
| Overall | 0.39 ± 0.10 | 0.67 ± 0.09 | 0.55 ± 0.18 | 1.41 ± 0.13 | 2.46 ± 0.45 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, L.; Wang, H.; Fu, Y.-B. Analysis of Stored mRNA Degradation in Acceleratedly Aged Seeds of Wheat and Canola in Comparison to Arabidopsis. Plants 2020, 9, 1707. https://doi.org/10.3390/plants9121707
Zhao L, Wang H, Fu Y-B. Analysis of Stored mRNA Degradation in Acceleratedly Aged Seeds of Wheat and Canola in Comparison to Arabidopsis. Plants. 2020; 9(12):1707. https://doi.org/10.3390/plants9121707
Chicago/Turabian StyleZhao, Liang, Hong Wang, and Yong-Bi Fu. 2020. "Analysis of Stored mRNA Degradation in Acceleratedly Aged Seeds of Wheat and Canola in Comparison to Arabidopsis" Plants 9, no. 12: 1707. https://doi.org/10.3390/plants9121707







