Genome-Wide Association Analysis Identifies Candidate Genes Regulating Seed Number per Silique in Arabidopsis thaliana
Abstract
:1. Introduction
2. Results
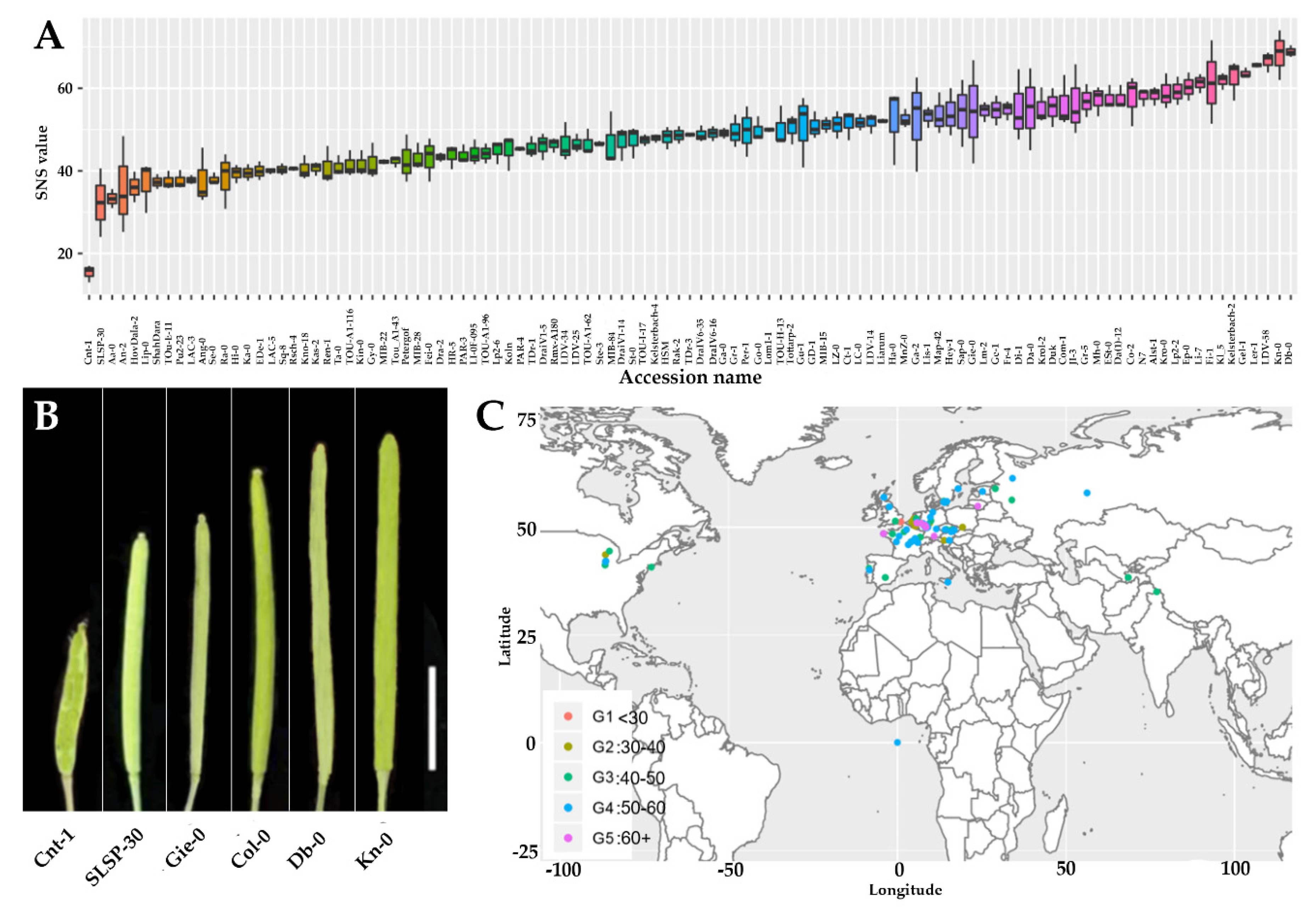
2.1. Seed Number Per Silique Vary Significantly Among Arabidopsis Accessions
2.2. 38 SNP-SNS Associations Indentified in GWAS
2.3. PIN3 Is a Positive Regulator of SNS
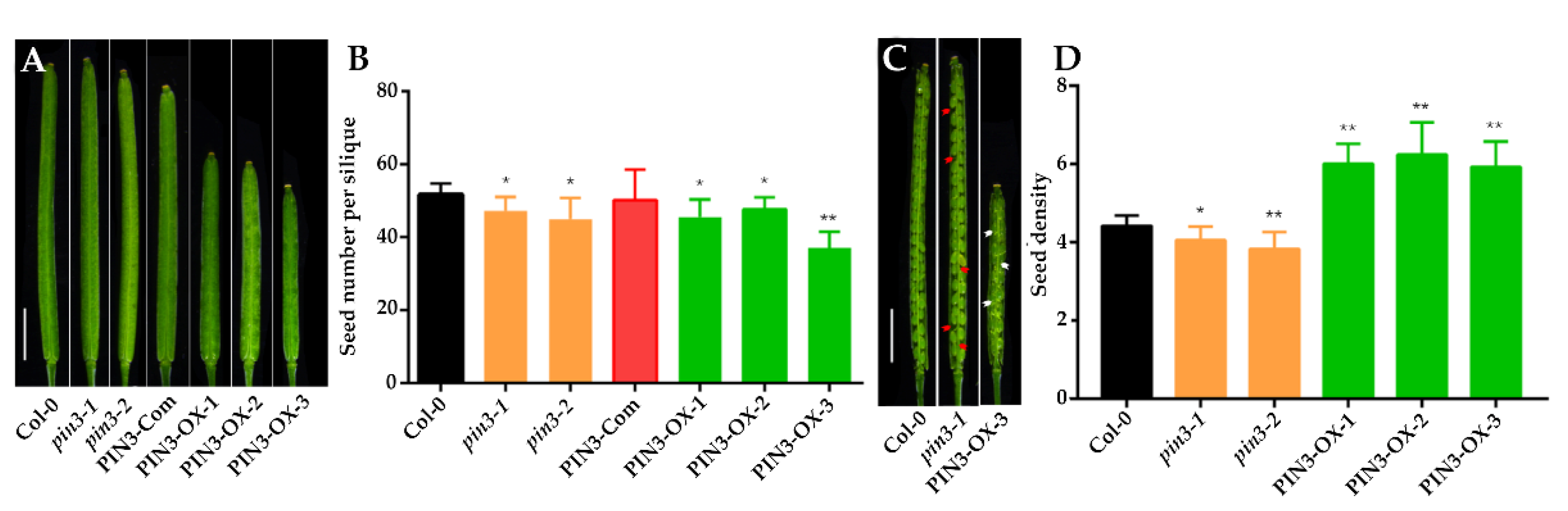
2.4. Overexpression of PIN3 Increases SNS
3. Discussion
4. Materials and Methods
4.1. Plant Material and Growth Conditions
4.2. Phenotypic Analysis of Seed Number
4.3. Genome-Wide Association Study
4.4. Vector Construction and Transformation Processes of Overexpression and Complementation Lines
4.5. Identification of Mutants, Overexpression and Complementary Lines
4.6. Quantitative Real-time RT-PCR
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| GWAS | Genome-wide association analysis |
| SNS | Seed number per silique |
| SD | Seed density |
| STK | SEEDSTICK |
| CMM | Carpel margin meristem |
| SNP | Single nucleotide polymorphisms |
| OD | Ovule density |
| TGW | Thousand grain weight |
References
- Tong, H.; Chu, C. Functional Specificities of Brassinosteroid and potential utilization for crop improvement. Trends Plant Sci. 2018, 23, 1016–1028. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, Y.; Wang, C.; Li, Z.; Liu, Q.; Xu, J.; Liao, J.; Wang, X.; Qu, L.; Chen, F.; et al. Overexpression of microRNA OsmiR397 improves rice yield by increasing grain size and promoting panicle branching. Nat. Biotechnol. 2013, 31, 848–852. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Jiang, W.; Hu, Y.; Wu, P.; Zhu, J.; Liang, W.; Wang, Z.; Lin, W. BR signal influences Arabidopsis ovule and seed number through regulating related genes expression by BZR. Mol. Plant 2013, 6, 456–469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, W.; Lin, W. Brassinosteroid functions in Arabidopsis seed development. Plant Signal. Behav. 2013, 8, e25928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theissen, G. Development of floral organ identity: Stories from the MADS house. Curr. Opin. Plant Biol. 2001, 4, 75–85. [Google Scholar] [CrossRef]
- Theissen, G.; Saedler, H. Floral quartets. Nature 2001, 409, 469–471. [Google Scholar] [CrossRef]
- Twell, D.; Park, S.K.; Hawkins, T.J.; Schubert, D.; Schmidt, R.; Smertenko, A.; Hussey, P.J. MOR1/GEM1 plays an essential role in the plant-specific cytokinetic phragmoplast. Nat. Cell Biol. 2002, 4, 711–714. [Google Scholar] [CrossRef] [Green Version]
- Nowack, M.K.; Grini, P.E.; Jakoby, M.J.; Lafos, M.; Koncz, C.; Schnittger, A. A positive signal from the fertilization of the egg cell sets off endosperm proliferation in angiosperm embryogenesis. Nat. Genet. 2006, 38, 63–67. [Google Scholar] [CrossRef]
- Springer, P.S.; Holding, D.R.; Groover, A.; Yordan, C.; Martienssen, R.A. The essential Mcm7 protein PROLIFERA is localized to the nucleus of dividing cells during the G (1) phase and is required maternally for early Arabidopsis development. Development 2000, 127, 1815–1822. [Google Scholar]
- Moll, C.; Lyncker, L.V.; Zimmermann, S.; Kägi, C.; Baumann, N.; Twell, D.; Grossniklaus, U.; Gross-Hardt, R. CLO/GFA1 and ATO are novel regulators of gametic cell fate in plants. Plant J. 2008, 56, 913–921. [Google Scholar] [CrossRef]
- Chaudhury, A.M.; Koltunow, A.; Payne, T.; Luo, M.; Tucker, M.R.; Dennis, E.S.; Peacock, W.J. Control of Early Seed Development. Annu. Rev. Cell Dev. Biol. 2010, 17, 677–699. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, P.; Kapoor, S.; Tyagi, A.K. Transcription factors regulating the progression of monocot and dicot seed development. BioEssays 2011, 33, 189–202. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, V.V.; Surendrarao, A.; Liu, Z. APETALA1 and SEPALLATA3 interact with SEUSS to mediate transcription repression during flower development. Development 2006, 133, 3159–3166. [Google Scholar] [CrossRef] [Green Version]
- Franks, R.G.; Wang, C.; Levin, J.Z.; Liu, Z. SEUSS, a member of a novel family of plant regulatory proteins, represses floral homeotic gene expression with LEUNIG. Development 2002, 129, 253–263. [Google Scholar] [PubMed]
- Azhakanandam, S.; Nole-Wilson, S.; Bao, F.; Franks, R.G. SEUSS and AINTEGUMENTA mediate patterning and ovule initiation during gynoecium medial domain development. Plant Physiol. 2008, 146, 1165–1181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartrina, I.; Otto, E.; Strnad, M.; Werner, T.; Schmülling, T. Cytokinin regulates the activity of reproductive meristems, flower organ size, ovule formation, and thus seed yield in Arabidopsis thaliana. Plant Cell 2011, 23, 69–80. [Google Scholar] [CrossRef] [Green Version]
- Higuchi, M.; Pischke, M.S.; Mähönen, A.P.; Miyawaki, K.; Hashimoto, Y.; Seki, M.; Kobayashi, M.; Shinozaki, K.; Kato, T.; Tabata, S.; et al. In planta functions of the Arabidopsis cytokinin receptor family. Proc. Natl. Acad. Sci. USA 2004, 101, 8821–8826. [Google Scholar] [CrossRef] [Green Version]
- Bencivenga, S.; Simonini, S.; Benková, E.; Colombo, L. The transcription factors BEL1 and SPL are required for cytokinin and auxin signaling during ovule development in Arabidopsis. Plant Cell 2012, 24, 2886–2897. [Google Scholar] [CrossRef] [Green Version]
- Sita, K.; Sehgal, A.; Kumar, J.; Kumar, S.; Singh, S.; Siddique, K.H.M.; Nayyar, H. Identification of high-temperature tolerant Lentil (Lens culinaris Medik.) genotypes through leaf and pollen traits. Front. Plant Sci. 2017, 8, 744. [Google Scholar] [CrossRef] [Green Version]
- Jiang, Y.; Lahlali, R.; Karunakaran, C.; Warkentin, T.D.; Davis, A.R.; Bueckert, R.A. Pollen, ovules, and pollination in pea: Success, failure, and resilience in heat. Plant Cell Environ. 2019, 42, 354–372. [Google Scholar] [CrossRef] [Green Version]
- Bowman, J.L.; Smyth, D.R.; Meyerowitz, E.M. Genetic interactions among floral homeotic genes of Arabidopsis. Development 1991, 112, 1–20. [Google Scholar] [PubMed]
- Coen, E.S.; Meyerowitz, E.M. The war of the whorls: Genetic interactions controlling flower development. Nature 1991, 353, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Favaro, R.; Pinyopich, A.; Battaglia, R.; Kooiker, M.; Borghi, L.; Ditta, G.; Yanofsky, M.F.; Kater, M.M.; Colombo, L. MADS-Box Protein Complexes Control Carpel and Ovule Development in Arabidopsis. Plant Cell 2003, 15, 2603–2611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinyopich, A.; Ditta, G.S.; Savidge, B.; Liljegren, S.J.; Baumann, E.; Wisman, E.; Yanofsky, M.F. Assessing the redundancy of MADS-box genes during carpel and ovule development. Nature 2003, 424, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Kooiker, M.; Airoldi, C.A.; Losa, A.; Manzotti, P.S.; Finzi, L.; Kater, M.M.; Colombo, L. BASIC PENTACYSTEINE1, a GA binding protein that induces conformational changes in the regulatory region of the homeotic Arabidopsis gene SEEDSTICK. Plant Cell 2005, 17, 722–729. [Google Scholar] [CrossRef] [Green Version]
- Zu, S.; Jiang, Y.; Hu, L.; Zhang, Y.; Chang, J.; Xue, H.; Lin, W. Effective Modulating Brassinosteroids signal to study their specific regulation of reproductive development and enhance yield. Front. Plant Sci. 2019, 10, 980. [Google Scholar] [CrossRef] [Green Version]
- Pelaz, S.; Ditta, G.S.; Baumann, E.; Wisman, E.; Yanofsky, M.F. B and C floral organ identity functions require SEPALLATA MADS-box genes. Nature 2000, 405, 200–203. [Google Scholar] [CrossRef]
- Pelaz, S.; Gustafson-Brown, C.; Kohalmi, S.E.; Crosby, W.L.; Yanofsky, M.F. APETALA1 and SEPALLATA3 interact to promote flower development. Plant J. 2001, 26, 385–394. [Google Scholar] [CrossRef]
- Tzeng, T.; Hsiao, C.; Chi, P.; Yang, C. Two lily SEPALLATA-like genes cause different effects on floral formation and floral transition in Arabidopsis. Plant Physiol. 2003, 133, 1091–1101. [Google Scholar] [CrossRef] [Green Version]
- Ferrario, S.; Immink, R.G.; Angenent, G.C. Conservation and diversity in flower land. Curr. Opin Plant Biol. 2003, 7, 84–91. [Google Scholar] [CrossRef]
- Reyes-Olalde, J.I.; Zuñiga-Mayo, V.M.; Chávez Montes, R.A.; Marsch-Martníez, N.; de Folter, S. Inside the gynoecium: At the carpel margin. Trends Plant Sci. 2013, 18, 644–655. [Google Scholar] [CrossRef] [PubMed]
- Cucinotta, M.; Colombo, L.; Roig-Villanova, I. Ovule development, a new model for lateral organ formation. Front. Plant Sci. 2014, 5, 117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hagen, G.; Guilfoyle, T. Auxin-responsive gene expression: Genes, promoters and regulatory factors. Plant Mol. Biol. 2002, 49, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Venglat, S.P.; Sawhney, V.K. Benzylaminopurine induces phenocopies of floral meristem and organ identity mutants in wild-type Arabidopsis plants. Planta 1996, 198, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Kunst, L.; Klenz, J.E.; Martinez-Zapater, J.; Haughn, G.W. AP2 Gene determines the identity of perianth organs in flowers of Arabidopsis thaliana. Plant Cell 1989, 1, 1195–1208. [Google Scholar] [CrossRef] [PubMed]
- Jofuku, K.D.; den Boer, B.G.; Van Montagu, M.; Okamuro, J.K. Control of Arabidopsis flower and seed development by the homeotic gene APETALA2. Plant Cell 1994, 6, 1211–1225. [Google Scholar] [PubMed] [Green Version]
- Galbiati, F.; Roy, D.S.; Simonini, S.; Cucinotta, M.; Ceccato, L.; Cuesta, C.; Simaskova, M.; Benkova, E.; Kamiuchi, Y.; Aida, M.; et al. An integrative model of the control of ovule primordia formation. Plant J. 2013, 76, 446–455. [Google Scholar] [CrossRef]
- Nottingham Arabidopsis Stock Centre (NASC). Available online: http://arabidopsis.info/ (accessed on 10 October 2018).
- Baxter, I.; Brazelton, J.N.; Yu, D.; Huang, Y.; Lahner, B.; Yakubova, E.; Li, Y.; Bergelson, J.; Borevitz, J.O.; Nordborg, M.; et al. A coastal cline in sodium accumulation in Arabidopsis thaliana is driven by natural variation of the sodium transporter AtHKT1;1. PLoS Genet. 2010, 6, e1001193. [Google Scholar] [CrossRef] [Green Version]
- Platt, A.; Horton, M.; Huang, Y.; Li, Y.; Anastasio, A.E.; Mulyati, N.W.; Agren, J.; Bossdorf, O.; Byers, D.; Donohue, K.; et al. The scale of population structure in Arabidopsis thaliana. PLoS Genet. 2010, 6, e1000843. [Google Scholar] [CrossRef] [Green Version]
- Lippert, C.; Listgarten, L.; Liu, Y.; Kadie, C.M.; Davidson, R.I.; Heckerman, D. FaST linear mixed models for genome-wide association studies. Nat. Methods 2011, 8, 833–835. [Google Scholar] [CrossRef]
- TAIR Database. Available online: https://www.arabidopsis.org/ (accessed on 15 March 2020).
- Abbas, M.; Hernández-García, J.; Pollmann, S.; Samodelov, S.L.; Kolb, M.; Friml, J.; Hammes, U.Z.; Zurbriggen, M.D.; Blázquez, M.A.; Alabadía, D. Auxin methylation is required for differential growth in Arabidopsis. Proc. Natl. Acad. Sci. USA 2018, 115, 6864–6869. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grones, P.; Abas, M.; Hajný, J.; Jones, A.; Waidmann, S.; Kleine-Vehn, J.; Friml, J. PID/WAG-mediated phosphorylation of the Arabidopsis PIN3 auxin transporter mediates polarity switches during gravitropism. Sci. Rep. 2018, 8, 10279. [Google Scholar] [CrossRef] [PubMed]
- Winter, D.; Vinegar, B.; Nahal, H.; Ammar, R.; Wilson, G.V.; Provart, N.J. An “Electronic Fluorescent Pictograph” Browser for Exploring and Analyzing Large-Scale Biological Data Sets. PLoS ONE 2007, 2, e718. [Google Scholar] [CrossRef] [PubMed]
- T-DNA Express: Arabidopsis Gene Mapping Tool. Available online: http://signal.salk.edu/cgi-bin/tdnaexpress (accessed on 4 January 2019).
- Zhai, H.; Feng, Z.; Du, X.; Song, Y.; Liu, X.; Qi, Z.; Song, L.; Li, J.; Li, L.; Peng, H.; et al. A novel allele of TaGW2-A1 is located in a finely mapped QTL that increases grain weight but decreases grain number in wheat (Triticum aestivum L.). Theor. Appl. Genet. 2018, 131, 539–553. [Google Scholar] [CrossRef] [Green Version]
- Guan, P.; Di, N.; Mu, Q.; Shen, X.; Wang, Y.; Wang, X.; Yu, K.; Song, W.; Chen, Y.; Xin, M.; et al. Use of near-isogenic lines to precisely map and validate a major QTL for grain weight on chromosome 4AL in bread wheat (Triticum aestivum L.). Theor. Appl. Genet. 2019, 132, 2367–2379. [Google Scholar] [CrossRef]
- Wang, Y.; Hou, Y.; Gu, H.; Kang, D.; Chen, Z.; Liu, J.; Qu, L. The Arabidopsis anaphase-promoting complex/cyclosome subunit 1 is critical for both female gametogenesis and embryogenesis. J. Integr. Plant Biol. 2012, 55, 64–74. [Google Scholar] [CrossRef]
- Makkena, S.; Lee, E.; Sack, F.D.; Lamb, R.S. The R2R3 MYB transcription factors FOUR LIPS and MYB88 regulate female reproductive development. J. Exp. Bot. 2012, 63, 5545–5558. [Google Scholar] [CrossRef] [Green Version]
- Schneitz, K.; Hulskamp, M.; PruittR, E. Wild-type ovule development in Arabidopsis thaliana-A light microscope study of cleared whole-mount tissue. Plant J. 1995, 7, 731–749. [Google Scholar] [CrossRef]
- Christensen, C.A.; King, E.J.; Jordan, J.R.; Drews, G.N. Megagametogenesis in Arabidopsis wild type and the Gf mutant sexual. Plant Reprod. 1997, 10, 49–64. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, Y.; Qin, G.; Tsuge, T.; Sakaguchi, N.; Luo, G.; Sun, K.; Shi, D.; Aki, S.; Zheng, N.; et al. Targeted degradation of the cyclin-dependent kinase inhibitor ICK4/KRP6 by RING-Type E3 ligases is essential for mitotic cell cycle progression during Arabidopsis gametogenesis. Plant Cell 2008, 20, 1538–1554. [Google Scholar] [CrossRef] [Green Version]
- Springer, P.S.; McCombie, W.R.; Sundaresan, V.; Martienssen, R.A. Gene trap tagging of PROLIFERA, an essential MCM2-3-5-like gene in Arabidopsis. Science 1995, 268, 877–880. [Google Scholar] [CrossRef] [PubMed]
- Capron, A.; Serralbo, O.; Fülöp, K.; Frugier, F.; Parrnentier, Y.; Dong, A.; Lecureuil, A.; Guerche, P.; Kondorosi, E.; Scheres, B. The Arabidopsis anaphase-promoting complex or cyclosome: Molecular and genetic characterization of the APC2 subunit. Plant Cell 2019, 31, 1386–1387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwee, H.S.; Sundaresan, V. The Nomega gene required for female gametophyte development encodes the putative APC6/CDC16 component of the Anaphase promoting complex in Arabidopsis. Plant J. 2003, 36, 853–866. [Google Scholar] [CrossRef]
- Lee, Y.J.; Li, Y.; Liu, B. Two Arabidopsis phragmoplast-associated kinesins play a critical role in cytokinesis during male gametogenesis. Plant Cell 2007, 19, 2595–2605. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; McCormick, S. Sidecar pollen, an Arabidopsis thaliana male gametophytic mutant with aberrant cell divisions during pollen development. Development 1996, 122, 3243–3253. [Google Scholar] [PubMed]
- Mukhtar, M.S.; Liu, X.; Somssich, I.E. Elucidating the role of WRKY27 in male sterility in Arabidopsis. Plant Signal. Behav. 2018, 13, e1363945. [Google Scholar] [CrossRef] [Green Version]
- Yuan, J.; Kessler, S.A. A genome-wide association study reveals a novel regulator of ovule number and fertility in Arabidopsis thaliana. PLoS Genet. 2019, 15, e1007934. [Google Scholar] [CrossRef] [Green Version]
- Sorensen, A.M.; Kroeber, S.; Saedler, H. The ABNORMAL GAMETOPHYTES (AGM) gene product of Arabidopsis demonstrates a role in mitosis during gamete development. Plant Cell Physiol. 2004, 45, 905–913. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Hao, N.; Li, H.; Ge, D.; Du, Y.; Liu, R.; Wen, C.; Li, Y.; Zhang, X.; Wu, T. PINOID is required for lateral organ morphogenesis and ovule development in cucumber. J. Exp. Bot. 2019, 70, 5715–5730. [Google Scholar] [CrossRef]
- Zeng, J.; Zhang, M.; Hou, L.; Bai, W.; Yan, X.; Hou, N.; Wang, H.; Huang, J.; Zhao, J.; Pei, Y. Cytokinin inhibits cotton fiber initiation by disrupting PIN3a-mediated asymmetric accumulation of auxin in the ovule epidermis. J. Exp. Bot. 2019, 70, 3139–3151. [Google Scholar] [CrossRef]
- Xu, M.; Tang, D.; Cheng, X.; Zhang, J.; Tang, Y.; Tao, Q.; Shi, W.; You, A.; Gu, M.; Cheng, N.; et al. OsPINOID regulates stigma and ovule initiation through maintenance of the floral meristem by auxin signaling. Plant Physiol. 2019, 180, 952–965. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Olalde, J.I.; Zúñiga-Mayo, V.M.; Serwatowska, J.; Chavez Montes, R.A.; Lozano-Sotomayor, P.; Herrera-Ubaldo, H.; Gonzalez-Aguilera, K.L.; Ballester, P.; Ripoll, J.J.; Ezquer, I.; et al. The bHLH transcription factor SPATULA enables cytokinin signaling, and both activate auxin biosynthesis and transport genes at the medial domain of the gynoecium. PLoS Genet. 2017, 13, e1006726. [Google Scholar] [CrossRef] [PubMed]
- Ceccato, L.; Masiero, S.; Roy, D.S.; Bencivenga, S.; Roig-Villanova, I.; Ditengou, F.A.; Palme, K.; Simon, R.; Colombo, L. Maternal control of PIN1 is required for female gametophyte development in Arabidopsis. PLoS ONE 2013, 8, e66148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Friml, J.; Wiśniewska, J.; Benková, E.; Mendgen, K.; Palme, K. Lateral relocation of auxin efflux regulator PIN3 mediates tropism in Arabidopsis. Nature 2002, 415, 806–809. [Google Scholar] [CrossRef] [Green Version]
- Chen, Q.; Liu, Y.; Maere, S.; Lee, E.; Isterdael, G.V.; Xie, Z.; Xuan, W.; Lucas, J.; Vassileva, V.; Kitakura, S.; et al. A coherent transcriptional feed-forward motif model for mediating auxin-sensitive PIN3 expression during lateral root development. Nat. Commun. 2015, 6, 8821. [Google Scholar] [CrossRef] [Green Version]
- Chao Laboratory. Available online: http://sippe.ac.cn/dycao/ (accessed on 10 March 2020).
- Lempe, J.; Balasubramanian, S.; Sureshkumar, S.; Singh, A.; Schmid, M.; Weigel, D. Diversity of flowering responses in wild Arabidopsis thaliana strains. PLoS Genet. 2005, 1, 109–118. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, H.-L.; Hong, J.; Jiang, Y.-T.; Yu, S.-X.; Zhang, Y.-J.; Shi, J.-X.; Lin, W.-H. Genome-Wide Association Analysis Identifies Candidate Genes Regulating Seed Number per Silique in Arabidopsis thaliana. Plants 2020, 9, 585. https://doi.org/10.3390/plants9050585
Jiang H-L, Hong J, Jiang Y-T, Yu S-X, Zhang Y-J, Shi J-X, Lin W-H. Genome-Wide Association Analysis Identifies Candidate Genes Regulating Seed Number per Silique in Arabidopsis thaliana. Plants. 2020; 9(5):585. https://doi.org/10.3390/plants9050585
Chicago/Turabian StyleJiang, Huan-Li, Jun Hong, Yu-Tong Jiang, Shi-Xia Yu, Yan-Jie Zhang, Jian-Xin Shi, and Wen-Hui Lin. 2020. "Genome-Wide Association Analysis Identifies Candidate Genes Regulating Seed Number per Silique in Arabidopsis thaliana" Plants 9, no. 5: 585. https://doi.org/10.3390/plants9050585





