Designing a Clean Label Fish Patty with Olive, Citric, Pomegranate, or Rosemary Extracts
Abstract
:1. Introduction
2. Results
2.1. Proximate Composition
2.2. Shelf-Life Study
2.3. Volatile Compounds
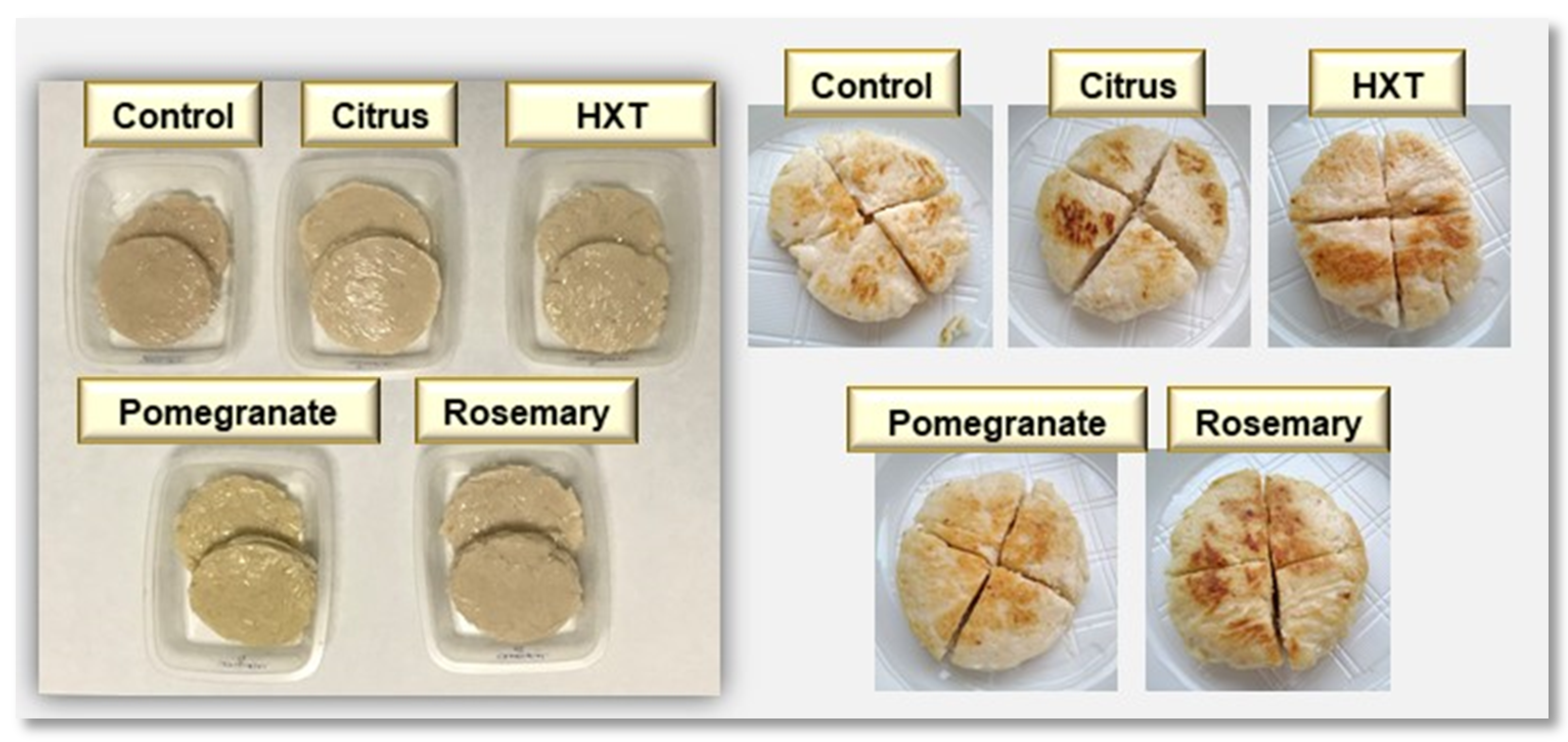
2.4. Sensory Analysis
3. Discussion
4. Materials and Methods
4.1. Natural Oils and Extracts
4.2. Fish Patty Manufacture and Sampling
4.3. Proximate Composition
Fatty Acids Profile
4.4. Shelf-Life Study
4.5. Sensory Analysis
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hosomi, R.; Yoshida, M.; Fukunaga, K. Seafood consumption and components for health. Glob. J. Health Sci. 2012, 4, 72–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martín Cerdeño, V.J. Consumo de pescados y mariscos en España. Un análisis de los perfiles de la demanda. Distrib. y Consum. 2017, 27, 5–18. [Google Scholar]
- Ehira, S.; Uchiyama, H. Freshness-Lowering Rates of Cod and Sea Bream Viewed from Changes in Bacterial Count, Total Volatile Base- and Trimethylamine-Nitrogen, and ATP Related Compounds. Nippon Suisan Gakkaishi 1974, 40, 479–487. [Google Scholar] [CrossRef] [Green Version]
- Nieto, G.; Martínez, L.; Castillo, J.; Ros, G. Hydroxytyrosol extracts, olive oil and walnuts as functional components in chicken sausages. J. Sci. Food Agric. 2017, 97, 3671–3771. [Google Scholar] [CrossRef]
- Nieto, G.; Martínez, L.; Castillo, J.; Ros, G. Effect of hydroxytyrosol, walnut and olive oil on nutritional profile of Low-Fat Chicken Frankfurters. Eur. J. Lipid Sci. Technol. 2017, 119, 1600518. [Google Scholar] [CrossRef]
- Martínez, L.; Ros, G.; Nieto, G. Effect of natural extracts obtained from food industry by-products on nutritional quality and shelf life of chicken nuggets enriched with organic Zn and Se provided in broiler diet. Poult. Sci. 2020, 99, 1491–1501. [Google Scholar] [CrossRef]
- Martínez, L.; Castillo, J.; Ros, G.; Nieto, G. Antioxidant and antimicrobial activity of rosemary, pomegranate and olive extracts in fish patties. Antioxidants 2019, 8, 86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martínez, L.; Bastida, P.; Castillo, J.; Ros, G.; Nieto, G. Green alternatives to synthetic antioxidants, antimicrobials, nitrates, and nitrites in clean label Spanish Chorizo. Antioxidants 2019, 8, 184. [Google Scholar] [CrossRef] [Green Version]
- Shah, M.A.; Bosco, S.J.D.; Mir, S.A. Plant extracts as natural antioxidants in meat and meat products. Meat Sci. 2014, 98, 21–33. [Google Scholar] [CrossRef]
- Karre, L.; Lopez, K.; Getty, K.J.K. Natural antioxidants in meat and poultry products. Meat Sci. 2013, 94, 220–227. [Google Scholar] [CrossRef]
- Yerlikaya, P.; Gokoglu, N.; Uran, H. Quality changes of fish patties produced from anchovy during refrigerated storage. Eur. Food Res. Technol. 2005, 220, 287–291. [Google Scholar] [CrossRef]
- Sehgal, H.S.; Shahi, M.; Sehgal, G.K.; Thind, S.S. Nutritional, microbial and organoleptic qualities of fish patties prepared from carp (Cyprinus carpio Linn.) of three weight groups. J. Food Sci. Technol. 2011, 48, 242–245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salgado, P.R.; López-Caballero, M.E.; Gómez-Guillén, M.C.; Mauri, A.N.; Montero, M.P. Sunflower protein films incorporated with clove essential oil have potential application for the preservation of fish patties. Food Hydrocoll. 2013, 33, 74–84. [Google Scholar] [CrossRef]
- López-Caballero, M.E.; Gómez-Guillén, M.C.; Pérez-Mateos, M.; Montero, P. A chitosan-gelatin blend as a coating for fish patties. Food Hydrocoll. 2005, 19, 303–311. [Google Scholar] [CrossRef] [Green Version]
- Nowzari, F.; Shábanpour, B.; Ojagh, S.M. Comparison of chitosan-gelatin composite and bilayer coating and film effect on the quality of refrigerated rainbow trout. Food Chem. 2013, 141, 1667–1672. [Google Scholar] [CrossRef] [PubMed]
- Martí, L. Evaluación De La Vida Útil De Hamburguesas Elaboradas a Base De Pescado Y Algas. Master’s Thesis, Universitat Politècnica de València, Valencia, Spain, 2010. [Google Scholar]
- Hleap Borrero, J.; Gutiérrez, A.; Rivera, L. Análisis microbiológico y sensorial de productos elaborados a partir de surimi de carduma (cetengraulis mysticetus) y plumuda (opisthonema spp.). Biotecnol. En El Sect. Agropecu. Y Agroind. BSAA 2010, 8, 58–65. [Google Scholar]
- Hleap, J.; Gutiérrez, A.; Rivera, L. Microbiological and sensory analysis of products made from surimi carduma (Cetengraulis mysticetus) and plumuda (Opisthonema spp.). Biotecnol. En El Sect. Agropecu. Y Agroind. 2010, 8, 57–65. [Google Scholar]
- Córser, P.I.; Ferrari, G.T.; De Martínez, Y.B.; Salas, E.M.; Cagnasso, M.A. Análisis proximal, perfil de ácidos grasos, aminoácidos esenciales y contenido de minerales en doce especies de pescado de importancia comercial en Venezuela. Arch. Latinoam. Nutr. 2000, 50, 187–194. [Google Scholar]
- Irmak, A.; Tokusoglu, O. Saturated and Unsaturated Fatty Acids Composition of Olive Oils Obtained from Less Salty Black Table Olives Preserved with Vacuum, MAP and Gamma Irradiation Technologies. J. Nutr. Food Sci. 2017, 7, 582. [Google Scholar] [CrossRef]
- Pereira De Melo, I.L.; Carvalho, E.; Filho, J.M.; Teixeira De Carvalho, B.; Mancini-Filho, J. Pomegranate Seed Oil (Punica Granatum, L.): A Source of Punicic Acid (Conjugated α-Linolenic Acid). J. Hum. Nutr. Food Sci. 2014, 2, 1–11. [Google Scholar]
- Moreiras, O.; Carbajal, A.; Cabrera, L.; Cuadrado, C. Tablas De Composicion De Alimentos (Ciencia Y Tecnica); Ediciones Pirámide: Madrid, Spain, 2016. [Google Scholar]
- Ministerio de Agricultura Pesca y Alimentación (MAPA). Informe del consumo de alimentación en España 2018; Gobierno de España: Madrid, Spain, 2018. [Google Scholar]
- Hebard, C.E.; Flick, G.J.; Martín, R.E. Occurrence and significance of trimethylamine oxide and its derivatives in fish and shellfish. In Chemistry and Biochemistry of Marine Food Products; Martin, R.E., Ed.; AVI publishing: Westport, CT, USA, 1982; pp. 149–304. [Google Scholar]
- Fuentes-Zaragoza, E.; Pérez-Álvarez, J.A.; Sánchez-Zapata, E. Efecto de la concentración de aditivos e ingredientes sobre el color de pastas de merluza (Merluccius australis) tratadas térmicamente. Opt. Pura y Apl. 2009, 42, 9–21. [Google Scholar]
- Czeczuga, B.; Kłyszejko, B. Carotenoids in fish. XL. Carotenoids in fish from the Falklands region. Acta Ichthyol. Piscat. 1986, 16, 73–86. [Google Scholar] [CrossRef]
- Secci, G.; Parisi, G. From farm to fork: Lipid oxidation in fish products. A review. Ital. J. Anim. Sci. 2016, 15, 124–136. [Google Scholar] [CrossRef]
- Sun, Y.Q.; Tao, X.; Men, X.M.; Xu, Z.W.; Wang, T. In vitro and in vivo antioxidant activities of three major polyphenolic compounds in pomegranate peel: Ellagic acid, punicalin, and punicalagin. J. Integr. Agric. 2017, 16, 1808–1818. [Google Scholar] [CrossRef] [Green Version]
- Sarojini, A.; Raju, C.V.; Lakshmisha, I.P.; Amitha; Gajendra. Effect of pomegranate (Punica granatum) peel extract on lipid oxidation in sardine fish oil. J. Entomol. Zool. Stud. 2019, 7, 140–144. [Google Scholar]
- Ranucci, D.; Roila, R.; Andoni, E.; Braconi, P.; Branciari, R. Punica granatum and Citrus spp. Extract Mix Affects Spoilage Microorganisms Growth Rate in Vacuum-Packaged Cooked Sausages Made from Pork Meat, Emmer Wheat (Triticum dicoccum Schübler), Almond (Prunus dulcis Mill.) and Hazelnut (Corylus avellana L.). Foods 2019, 8, 664. [Google Scholar] [CrossRef] [Green Version]
- Kim, L.S.; Jin, S.K.; Kang, S.N.; Hur, I.C.; Choi, S.Y. Effect of olive-oil prepared tomato powder (OPTP) and refining lycopene on the physicochemical and sensory characteristics of seasoned raw pork during storage. Korean J. Food Sci. Anim. Resour. 2009, 29, 329–334. [Google Scholar] [CrossRef] [Green Version]
- Nieto, G.; Díaz, P.; Bañón, S.; Garrido, M.D. Effect on lamb meat quality of including thyme (Thymus zygis ssp. gracilis) leaves in ewes’ diet. Meat Sci. 2010, 85, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Djenane, D.; Sánchez-Escalante, A.; Beltrán, J.A.; Roncalés, P. Ability of α-tocopherol, taurine and rosemary, in combination with vitamin C, to increase the oxidative stability of beef steaks packaged in modified atmosphere. Food Chem. 2002, 76, 407–415. [Google Scholar] [CrossRef]
- Cuvelier, M.E.; Richard, H.; Berset, C. Antioxidative activity and phenolic composition of pilot-plant and commercial extracts of sage and rosemary. JAOCS J. Am. Oil Chem. Soc. 1996, 73, 645–652. [Google Scholar] [CrossRef]
- Zheng, W.; Wang, S.Y. Antioxidant activity and phenolic compounds in selected herbs. J. Agric. Food Chem. 2001, 49, 5165–5170. [Google Scholar] [CrossRef] [PubMed]
- Bensid, A.; Ucar, Y.; Bendeddouche, B.; Özogul, F. Effect of the icing with thyme, oregano and clove extracts on quality parameters of gutted and beheaded anchovy (Engraulis encrasicholus) during chilled storage. Food Chem. 2014, 145, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Özyurt, G.; Kuley, E.; Balikçi, E.; Kaçar, Ç.; Gökdogan, S.; Etyemez, M.; Özogul, F. Effect of the Icing with Rosemary Extract on the Oxidative Stability and Biogenic Amine Formation in Sardine (Sardinella aurita) During Chilled Storage. Food Bioprocess Technol. 2012, 5, 2777–2786. [Google Scholar] [CrossRef]
- Martínez, L.; Jongberg, S.; Ros, G.; Skibsted, L.H.; Nieto, G. Plant derived ingredients rich in nitrates or phenolics for protection of pork against protein oxidation. Food Res. Int. 2020, 129, 108789. [Google Scholar] [CrossRef] [PubMed]
- Gillett, M.B.; Suko, J.R.; Santoso, F.O.; Yancey, P.H. Elevated levels of trimethylamine oxide in muscles of deep-sea gadiform teleosts: A high-pressure adaptation? J. Exp. Zool. 1997, 279, 386–391. [Google Scholar] [CrossRef]
- Ghaly, A.E.; Dave, D.; Budge, S.; Brooks, M.S. Fish spoilage mechanisms and preservation techniques: Review. Am. J. Appl. Sci. 2010, 7, 859–877. [Google Scholar] [CrossRef] [Green Version]
- Huss, H.H. El Pescado Fresco: Su Calidad Y Cambios De Su Calidad; FAO Documento Técnico Pesca Ministerio Pesca; FAO: Rome, Italy, 1998; Volume 348, pp. 1–144. [Google Scholar]
- Dalgaard, P.; Gram, L.; Huss, H.H. Spoilage and shelf-life of cod fillets packed in vacuum or modified atmospheres. Int. J. Food Microbiol. 1993, 19, 283–294. [Google Scholar] [CrossRef]
- Noseda, B.; Islam, M.T.; Eriksson, M.; Heyndrickx, M.; De Reu, K.; Van Langenhove, H.; Devlieghere, F. Microbiological spoilage of vacuum and modified atmosphere packaged Vietnamese Pangasius hypophthalmus fillets. Food Microbiol. 2012, 30, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Peters, E.P.; Schlakman, G.J.; Yang, E.N. Production of Malonic Acid through the Fermentation of Glucose; Senior Design Reports; SAE: Ahmedabad, India, 2018; Volume 107. [Google Scholar]
- Asensio, C.M.; Quiroga, P.R.; Huang, Q.; Nepote, V.; Grosso, N.R. Fatty acids, volatile compounds and microbial quality preservation with an oregano nanoemulsion to extend the shelf life of hake (Merluccius hubbsi) burgers. Int. J. Food Sci. Technol. 2019, 54, 149–160. [Google Scholar] [CrossRef] [Green Version]
- Surette, M.E.; Gill, T.A.; LeBlanc, P.J. Biochemical basis of postmortem nucleotide catabolism in cod (Gadus morhua) and its relationship to spoilage. J. Agric. Food Chem. 1988, 36, 19–22. [Google Scholar] [CrossRef]
- Harpaz, S.; Glatman, L.; Drabkin, V.; Gelman, A. Effects of herbal essential oils used to extend the shelf life of freshwater-reared Asian sea bass fish (Lates calcarifer). J. Food Prot. 2003, 66, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Shapira, R.; Mimran, E. Isolation and characterization of Escherichia coli mutants exhibiting altered response to thymol. Microb. Drug Resist. 2007, 13, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, V.K.; Baek, K.H.; Kang, S.C. Control of Salmonella in foods by using essential oils: A review. Food Res. Int. 2012, 45, 722–734. [Google Scholar] [CrossRef]
- Del Nobile, M.A.; Corbo, M.R.; Speranza, B.; Sinigaglia, M.; Conte, A.; Caroprese, M. Combined effect of MAP and active compounds on fresh blue fish burger. Int. J. Food Microbiol. 2009, 135, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Tassou, C.C.; Drosinos, E.H.; Nychas, G.J.E. Inhibition of resident microbial flora and pathogen inocula on cold fresh fish fillets in olive oil, oregano, and lemon juice under modified atmosphere or air. J. Food Prot. 1996, 59, 31–34. [Google Scholar] [CrossRef]
- Mejlholm, O.; Dalgaard, P. Antimicrobial effect of essential oils on the seafood spoilage micro-organism Photobacterium phosphoreum in liquid media and fish products. Lett. Appl. Microbiol. 2002, 34, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, B.S.M.; Yamazaki, K.; Miyashita, K.; Il-Shik, S.; Dong-Suk, C.; Suzuki, T. Bacterial microflora of carp (Cyprinus carpio) and its shelf-life extension by essential oil compounds. Food Microbiol. 2004, 21, 657–666. [Google Scholar] [CrossRef]
- Casiot, C.; Egal, M.; Elbaz-Poulichet, F.; Bruneel, O.; Bancon-Montigny, C.; Cordier, M.A.; Gomez, E.; Aliaume, C. Hydrological and geochemical control of metals and arsenic in a Mediterranean river contaminated by acid mine drainage (the Amous River, France); preliminary assessment of impacts on fish (Leuciscus cephalus). Appl. Geochem. 2009, 24, 787–799. [Google Scholar] [CrossRef]
- Olmedo, P.; Pla, A.; Hernández, A.F.; Barbier, F.; Ayouni, L.; Gila, F. Determination of toxic elements (mercury, cadmium, lead, tin and arsenic) in fish and shellfish samples. Risk assessment for the consumers. Environ. Int. 2013, 59, 63–72. [Google Scholar] [CrossRef]
- Neff, J.M. Ecotoxicology of arsenic in the marine environment. Environ. Toxicol. Chem. 2009, 16, 917–927. [Google Scholar] [CrossRef]
- Van der Oost, R.; Beyer, J.; Vermeulen, N.P.E. Fish bioaccumulation and biomarkers in environmental risk assessment: A review. Environ. Toxicol. Pharmacol. 2003, 13, 57–149. [Google Scholar] [CrossRef]
- Hinck, J.E.; Blazer, V.S.; Denslow, N.D.; Echols, K.R.; Gale, R.W.; Wieser, C.; May, T.W.; Ellersieck, M.; Coyle, J.J.; Tillitt, D.E. Chemical contaminants, health indicators, and reproductive biomarker responses in fish from rivers in the Southeastern United States. Sci. Total Environ. 2008, 390, 538–557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahn, J.; Grün, I.U.; Mustapha, A. Effects of plant extracts on microbial growth, color change, and lipid oxidation in cooked beef. Food Microbiol. 2007, 24, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Brunton, N.P.; Cronin, D.A.; Monahan, F.J.; Durcan, R. A comparison of solid-phase microextraction (SPME) fibres for measurement of hexanal and pentanal in cooked turkey. Food Chem. 2000, 68, 339–345. [Google Scholar] [CrossRef]
- Mi, H.B.; Guo, X.; Li, J.R. Effect of 6-gingerol as natural antioxidant on the lipid oxidation in red drum fillets during refrigerated storage. LWT-Food Sci. Technol. 2016, 74, 70–76. [Google Scholar] [CrossRef]
- Shahidi, F.; Yun, J.; Rubin, L.J.; Wood, D.F. The Hexanal Content as an Indicator of Oxidative Stability and Flavour Acceptability in Cooked Ground Pork. Can. Inst. Food Sci. Technol. J. 1987, 20, 104–106. [Google Scholar] [CrossRef]
- Angelo, A.J.S.; Crippen, K.L.; Dupuy, H.P.; James, C. Chemical and Sensory Studies of Antioxidant-Treated Beef. J. Food Sci. 1990, 55, 1501–1505. [Google Scholar] [CrossRef]
- Kerler, J.; Grosch, W. Character impact odorants of boiled chicken: Changes during refrigerated storage and reheating. Eur. Food Res. Technol. 1997, 205, 232–238. [Google Scholar] [CrossRef]
- Ullrich, F.; Grosch, W. Identification of the most intense volatile flavour compounds formed during autoxidation of linoleic acid. Z. Lebensm. Unters. Forsch. 1987, 184, 277–282. [Google Scholar] [CrossRef]
- Nieto, G.; Bañón, S.; Garrido, M.D. Incorporation of thyme leaves in the diet of pregnant and lactating ewes: Effect on the fatty acid profile of lamb. Small Rum. Res. 2012, 105, 140–147. [Google Scholar] [CrossRef]
- Nieto, G.; Bañón, S.; Garrido, M.D. Administration of distillate thyme leaves in to the diet of Segureña ewes: Effect on lamb meat at quality. Animal 2012, 6, 2048–2056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nieto, G. Incorporation of by-products of rosemary and thyme in the diet of ewes: Effect on the fatty acid profile of lamb. Eur. Food Res. Technol. 2013, 236, 379–389. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 17th ed.; Association of Official Analyticial Chemistry: Washington, DC, USA, 2002. [Google Scholar]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Wang, L.L.; Xiong, Y.L. Inhibition of lipid oxidation in cooked beef patties by hydrolyzed potato protein is related to its reducing and radical scavenging ability. J. Agric. Food Chem. 2005, 53, 9186–9192. [Google Scholar] [CrossRef] [PubMed]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]




| Samples | Moisture (%) | Ash (%) | Protein (%) | Fat (%) | ALA (%) | DHA (%) |
|---|---|---|---|---|---|---|
| Control | 78.99 ± 1.02 | 2.87 ± 0.04 | 14.84 ± 0.63 | 1.11 ± 0.06 c | 0.05 ± 0.00 c | 0.09 ± 0.00 d |
| Ct | 79.08 ± 1.03 | 2.72 ± 0.12 | 15.24 ± 0.42 | 1.46 ± 0.05 b | 11.20 ± 1.06 a | 1.93 ± 0.09 a |
| HXT | 77.24 ± 0.83 | 2.98 ± 0.16 | 15.99 ± 0.41 | 1.62 ± 0.04 a | 12.50 ± 0.79 a | 1.34 ± 0.04 c |
| P | 77.52 ± 1.25 | 3.29 ± 0.05 | 15.39 ± 0.54 | 1.67 ± 0.09 a | 11.80 ± 0.95 a | 1.22 ± 0.08 c |
| R | 77.63 ± 0.92 | 3.14 ± 0.04 | 15.87 ± 0.34 | 1.78 ± 0.10 a | 9.48 ± 0.52 b | 1.51 ± 0.05 b |
| Samples | Fe | K | Mg | Na | Se | Zn | P |
|---|---|---|---|---|---|---|---|
| Control | 0.7 a 5% | 534 a 15.3% | 68 a 20% | 409 e 20.5% | 0.03 45% | 0.4 a 2.6% | 214 d 30.6% |
| Ct | 0.5 ab 3.9% | 288 e 8.2% | 39 d 11.5% | 651 d 32.5% | 0.02 28.8% | 0.2 c 1.6% | 209 e 29.9% |
| HXT | 0.3 b 1.8% | 349 b 10% | 49 b 14.4% | 751 c 37.6% | 0.022 35.8% | 0.3 b 1.7% | 281 b 40.1% |
| P | 0.7 a 5.1% | 305 c 8.7% | 42 c 12.4% | 818 b 41% | 0.02 25.6% | 0.2 e 1.4% | 272 c 38.9% |
| R | 0.4 b 3.5% | 300 d 8.5% | 40 cd 11.8% | 846 a 42% | 0.02 30.2% | 0.2 d 1.5% | 292 a 41.8% |
| RDA (mg/day) | 10–18 | 3500 | 330–350 | <2000 | 0.055–0.070 | 15 | 700 |
| Sample | Days Refrigerated Storage at 4 °C | ||||
|---|---|---|---|---|---|
| 0 | 4 | 7 | 11 | 14 | |
| pH | |||||
| Control Ct HXT P R | 6.8 ± 0.0 az 6.8 ± 0.0 az 6.8 ± 0.0 az 6.8 ± 0.0 bz 6.8 ± 0.1 abz | 7.0 ± 0.0 ayz 7.1 ± 0.0 ayz 7.0 ± 0.1 ayz 6.8 ± 0.1 byz 6.8 ± 0.0 abyz | 8.0 ± 0.1 ax 7.6 ± 0.0 ax 7.4 ± 0.0 ax 6.8 ± 0.0 bx 7.0 ± 0.1 abx | 7.3 ± 0.0 axy 7.2 ± 0.0 axy 7.3 ± 0.0 axy 6.7 ± 0.0 bxy 7.3 ± 0.0 abxy | 7.1 ± 0.0 axyz 7.2 ± 0.0 axyz 7.2 ± 0.1 axyz 6.6 ± 0.1 bxyz 7.1 ± 0.0 abxyz |
| Colour Parameters: L* | |||||
| Control Ct HXT P R | 72.3 ± 0.3 by 80.0 ± 1.2 ay 87.8 ± 2.3 ay 82.3 ± 0.9 ay 81.9 ± 1.1 ay | 65.0 ± 0.8 bz 73.9 ± 1.3 az 73.6 ± 0.8 az 70.9 ± 0.5 abz 71.7 ± 0.4 az | 62.8 ± 0.6 bz 72.2 ± 0.4 az 71.8 ± 0.3 az 70.4 ± 0.5 az 70.2 ± 0.4 az | 64.7 ± 0.5 bz 72.3 ± 0.5 az 72.8 ± 0.7 az 69.5 ± 0.8 az 70.3 ± 0.6 az | 64.5 ± 0.2 bz 73.0 ± 0.6 az 72.5 ± 0.3 az 69.2 ± 0.4 az 69.8 ± 0.3 az |
| a* | |||||
| Control Ct HXT P R | 3.5 ± 0.0 az 2.4 ± 0.1 bz 1.9 ± 0.1 bz 0.7 ± 0.1 cz 2.6 ± 0.0 bz | 3.7 ± 0.1 ayz 2.4 ± 0.1 byz 2.1 ± 0.0 byz 1.4 ± 0.1 cyz 2.7 ± 0.0 byz | 4.7 ± 0.1 ay 3.4 ± 0.1 by 3.0 ± 0.1 by 1.9 ± 0.1 cy 3.3 ± 0.0 by | 4.2 ± 0.1 ayz 3.4 ± 0.8 byz 2.9 ± 0.0 byz 2.2 ± 0.2 cyz 3.3 ± 0.1 byz | 3.7 ± 0.1 ayz 2.4 ± 0.1 byz 2.4 ± 0.1 byz 1.5 ± 0.0 cyz 2.8 ± 0.0 byz |
| b* | |||||
| Control Ct HXT P R | 11.4 ± 0.1 c 11.9 ± 0.4 b 11.9 ± 0.4 c 18.7 ± 0.4 a 12.1 ± 0.4 c | 11.3 ± 0.2 c 13.7 ± 0.1 b 11.1 ± 0.2 c 17.0 ± 0.3 a 11.1 ± 0.7 c | 12.2 ± 0.3 c 14.8 ± 0.1 b 13.0 ± 0.3 c 16.2 ± 0.2 a 11.5 ± 0.1 c | 12.1 ± 0.2 c 14.6 ± 0.4 b 12.4 ± 0.3 c 16.5 ± 0.3 a 10.6 ± 0.5 c | 11.0 ± 0.1 c 13.8 ± 0.3 b 12.4 ± 0.1 c 15.7 ± 0.2 a 11.2 ± 0.2 c |
| Sample | Days Refrigerated Storage | ||||
|---|---|---|---|---|---|
| 0 | 4 | 7 | 11 | 14 | |
| TBARs (mg MDA/kg) | |||||
| Control Ct HXT P R | 0.3 ± 0.01 x 0.2 ± 0.01 x 0.2 ± 0.00 x 0.2 ± 0.01 x 0.3 ± 0.01 x | 0.6 ± 0.02 ay 0.6 ± 0.01 az 0.4 ± 0.01 bcy 0.3 ± 0.01 cy 0.5 ± 0.01 byz | 0.6 ± 0.01 ay 0.6 ± 0.01 ayz 0.5 ± 0.00 bcyz 0.4 ± 0.01 cy 0.5 ± 0.01 bcyz | 0.6 ± 0.01 by 0.7 ± 0.01 ayz 0.6 ± 0.01 bcz 0.5 ± 0.01 cyz 0.5 ± 0.01 cyz | 0.8 ± 0.01 az 0.7 ± 0.01 byz 0.6 ± 0.01 cz 0.6 ± 0.01 cz 0.6 ± 0.00 cz |
| Thiol groups concentration (mmol thiol/mg protein) | |||||
| Control Ct HXT P R | 37.5 ± 0.1 w 32.9 ± 0.0 w 34.7 ± 0.1 w 32.0 ± 0.0 w 32.6 ± 0.0 w | 32.0 ± 0.0 x 21.4 ± 0.0 x 18.6 ± 0.1 x 26.8 ± 0.0 x 27.0 ± 0.0 x | 26.7 ± 0.0 xy 16.4 ± 0.1 xy 17.2 ± 0.1 xy 24.1 ± 0.1 xy 18.1 ± 0.0 xy | 15.7 ± 0.0 yz 15.4 ± 0.0 yz 18.6 ± 0.0 yz 20.7 ± 0.1 yz 13.5 ± 0.4 yz | 10.1 ± 0.0 z 12.9 ± 0.1 z 14.7 ± 0.1 z 16.7 ± 0.0 z 11.0 ± 0.0 z |
| TMA | |||||
| Control Ct HXT P R | 1.0 ± 0.0 z 0.5 ± 0.0 z 0.2 ± 0.1 z 0.9 ± 0.0 z 0.6 ± 0.1 z | 4.5 ± 0.0 yz 4.7 ± 0.0 yz 4.4 ± 0.0 yz 1.5 ± 0.2 yz 3.5 ± 0.0 yz | 7.8 ± 0.0 y 7.0 ± 0.1 y 6.3 ± 0.0 y 2.9 ± 0.0 y 5.1 ± 0.1 y | 17.3 ± 0.0 x 16.1 ± 0.0 x 10.8 ± 0.0 x 7.1 ± 0.0 x 9.2 ± 0.1 x | 18.3 ± 0.0 x 17.3 ± 0.0 x 15.4 ± 0.1 x 10.7 ± 0.0 x 11.7 ± 0.0 x |
| TVB-N | |||||
| Control Ct HXT P R | 4.0 ± 0.1 z 5.2 ± 0.1 z 4.6 ± 0.0 z 4.4 ± 0.1 z 4.8 ± 0.0 z | 32.2 ± 0.1 z 23.9 ± 0.1 z 27.9 ± 0.0 z 7.5 ± 0.0 z 14.2 ± 0.0 z | 87.9 ± 0.2 y 81.8 ± 0.1 y 50.6 ± 0.1 y 12.4 ± 0.1 y 42.2 ± 0.1 y | 110.0 ± 0.1 x 114.4 ± 0.0 x 108.1 ± 0.0 x 34.8 ± 0.1 x 91.1 ± 0.0 x | 115.2 ± 0.1 x 118.4 ± 0.0 x 120.6 ± 0.0 x 49.7 ± 0.0 x 100.9 ± 0.0 x |
| Volatile Compounds | Sample | Day 0 | Day 11 | Volatile Compounds | Sample | Day 0 | Day 11 |
|---|---|---|---|---|---|---|---|
| Spices | Microbiological degradation | ||||||
| Eucalyptol | Control | 0.66 ± 0.01 b | 0.56 ± 0.02 b | Propanoic acid | Control | nd | 2.05 ± 0.01 |
| Ct | 9.25 ± 0.05 a | 3.24 ± 0.06 a | Ct | nd | 3.43 ± 0.02 | ||
| R | 10.74 ± 0.10 a | 5.80 ± 0.02 a | R | nd | 3.26 ± 0.02 | ||
| P | nd | nd | P | nd | 4.01 ± 0.02 | ||
| HXT | nd | nd | HXT | nd | 2.35 ± 0.01 | ||
| Limonene | Control | 0.80 ± 0.02 cd | 0.90 ± 0.01 cd | Malonic acid | Control | 3.54 ± 0.02 b | 3.01 ± 0.03 b |
| Ct | 2.80 ± 0.01 a | 2.25 ± 0.01 a | Ct | 3.61 ± 0.03 b | 3.53 ± 0.04 b | ||
| R | 1.52 ± 0.01 b | 1.27 ± 0.01 b | R | 2.05 ± 0.03 b | 2.48 ± 0.03 b | ||
| P | 1.10 ± 0.03 bc | 1.39 ± 0.01 bc | P | 6.91 ± 0.08 a | 6.84 ± 0.08 a | ||
| HXT | 0.78 ± 0.01 d | 0.33 ± 0.01 d | HXT | 7.44 ± 0.09 a | 4.79 ± 0.05 a | ||
| Fish degradation | |||||||
| Methylamine | Control | 1.14 ± 0.02 | 9.55 ± 0.02 | Toluene | Control | 1.52 ± 0.01 | 0.91 ± 0.01 |
| Ct | 1.85 ± 0.02 | 10.93 ± 0.2 | Ct | 4.94 ± 0.05 | 2.00 ± 0.02 | ||
| R | 1.75 ± 0.02 | 5.60 ± 0.05 | R | 3.15 ± 0.01 | 1.77 ± 0.02 | ||
| P | 1.29 ± 0.02 | 1.96 ± 0.01 | P | 4.77 ± 0.02 | 2.52 ± 0.01 | ||
| HXT | 2.11 ± 0.03 | 7.67 ± 0.06 | HXT | 5.56 ± 0.04 | 1.97 ± 0.02 | ||
| Lipid autooxidation | Sea Pollution Agents | ||||||
| 1-butanol | Control | nd | 6.93 ± 0.03 e | Arsenous acid | Control | 6.59 ± 0.04 | 7.57 ± 0.01 |
| Ct | nd | 15.25 ± 0.10 c | Ct | 8.88 ± 0.06 | 4.76 ± 0.05 | ||
| R | nd | 17.16 ± 0.20 b | R | 6.55 ± 0.05 | 3.46 ± 0.02 | ||
| P | nd | 23.26 ± 0.20 a | P | 8.31 ± 0.05 | 4.77 ± 0.03 | ||
| HXT | nd | 13.06 ± 0.12 d | HXT | 7.87 ± 0.08 | 4.78 ± 0.04 | ||
| 2-nonanone | Control | nd | nd | Furan | Control | 0.29 ± 0.01 b | 0.16 ± 0.01 b |
| Ct | 2.23 ± 0.01 ab | 1.13 ± 0.01 ab | Ct | 0.37 ± 0.01 b | 0.22 ± 0.01 b | ||
| R | 1.50 ± 0.02 b | 0.98 ± 0.01 b | R | 0.33 ± 0.01 b | 0.16 ± 0.01 b | ||
| P | 2.18 ± 0.03 a | 1.81 ± 0.01 a | P | 0.51 ± 0.01 a | 0.46 ± 0.01 a | ||
| HXT | 1.24 ± 0.02 ab | 1.33 ± 0.03 ab | HXT | 0.47 ± 0.01 ab | 0.27 ± 0.01 ab | ||
| Ingredients | Control | Ct | HXT | P | R |
|---|---|---|---|---|---|
| Hake (g) | 852 | 852 | 852 | 852 | 852 |
| Water (ml) | 100 | 100 | 100 | 100 | 100 |
| Commercial mix (g) | 48 | ||||
| Salt (g) | 19 | 19 | 19 | 19 | |
| Fibers (g) | 25 | 25 | 25 | 25 | |
| Soy albumin (g) | 14 | 14 | 14 | 14 | |
| Essential oils (ml): | |||||
| Ecoflax ® | 5.7 | 5.7 | 5.7 | 5.7 | |
| Biomega Tech Algae ® | 5.7 | 5.7 | 5.7 | 5.7 | |
| Natural extracts (ppm): | |||||
| Acerola | 200 | 200 | 200 | 200 | |
| Ct | 200 | ||||
| HXT | 200 | ||||
| P | 200 | ||||
| R | 200 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Zamora, L.; Ros, G.; Nieto, G. Designing a Clean Label Fish Patty with Olive, Citric, Pomegranate, or Rosemary Extracts. Plants 2020, 9, 659. https://doi.org/10.3390/plants9050659
Martínez-Zamora L, Ros G, Nieto G. Designing a Clean Label Fish Patty with Olive, Citric, Pomegranate, or Rosemary Extracts. Plants. 2020; 9(5):659. https://doi.org/10.3390/plants9050659
Chicago/Turabian StyleMartínez-Zamora, Lorena, Gaspar Ros, and Gema Nieto. 2020. "Designing a Clean Label Fish Patty with Olive, Citric, Pomegranate, or Rosemary Extracts" Plants 9, no. 5: 659. https://doi.org/10.3390/plants9050659







