In Vitro Propagation of Isoëtes sabatina (Isoetaceae): A Key Conservation Challenge for a Critically Endangered Quillwort
Abstract
:1. Introduction
2. Results
2.1. Spore Germination
2.2. Sporeling Development
2.2.1. Effect of the Substrate on Sporeling Emergence
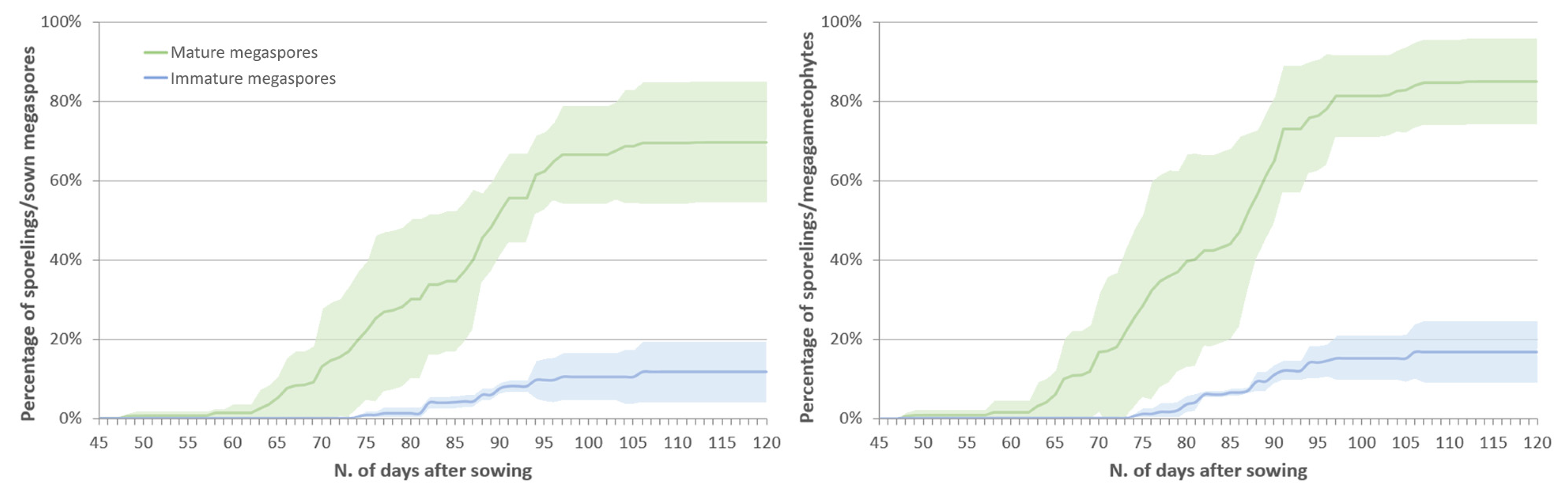
2.2.2. Effect of the Megaspores Developmental Stage on Sporeling Emergence
2.3. Transplants
3. Discussion
3.1. In Vitro Sporeling Production
3.2. The Conservation Challenges for I. sabatina
4. Materials and Methods
4.1. Study Area
4.2. Plant Material
4.3. Spore Collection Ex Situ
4.4. In vitro Mixed Culture of Mega- and Microspores
- (1)
- Water-agar (1%) medium, as described by [31];
- (2)
4.5. Evaluation of Spore Germination and Sporeling Development
4.6. Calculation of Sporeling Emergence Indices
- -
- Sporeling emergence percentage (SE, in percent), calculated on the number of sown megaspores, and the number of germinated megaspores (SEG, in percent);
- -
- Time for first sporeling emergence (T1, in days);
- -
- Mean daily emergence (MDE, in percent), calculated by the expression: , where Tn is the total number of days required to reach the final sporeling emergence percentage (SE);
- -
- Mean emergence time (MET, in days) indicates the average length of time required for maximum sporelings emergence and was calculated according to the following formula: , where ni is the number of sporelings newly emerged at time ti;
- -
- Mean emergence rate (MER, in day−1) is the average number of sporelings emerged per day, calculated as the reciprocal of MET;
- -
- Coefficient of variation of emergence time (CVt), calculated by the expression , where SDt is the standard deviation of the emergence time and MET is the mean emergence time;
- -
- Emergence Index (EI, number of sporelings per day), calculated by the expression , where ni is the number of sporelings emerged at time ti;
- -
- Peak value (PV, sporelings/day) is the accumulated number of sporelings at the point on the emergence curve at which the rate of emergence starts to decrease. It is calculated as the maximum quotient obtained by dividing successive cumulative emergence values by the respective incubation time;
- -
- Uncertainty of the sporeling emergence process (U, in bit), i.e., the uncertainty associated with the distribution of the relative frequency of emergence (fi), calculated through the formula: , being , where ni is the number of sporelings emerged at time ti. It can assume values spanning from 0 and log2n, with n being the total number of sporelings emerged.
4.7. Transplants
- (1)
- Lake sediment was homogeneously distributed at the bottom of glass containers (20 × 10 × 10 cm), covered with a thin layer of sterilized quartzite sand to avoid the spread of debris according to [18], and then covered with distilled water up to a height of ca. 3 cm;
- (2)
- Quartzite sand was homogeneously distributed at the bottom of the glass container and covered with distilled water up to a height of ca. 3 cm;
- (3)
- Water-agar (1%) medium (4 cm high) in plant culture containers MagentaTM GA7 (7.6 × 7.6 × 10.2 cm), wetted with only 5 mL of distilled water.
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Troia, A.; Pereira, J.B.; Kim, C.; Taylor, W.C. The genus Isoëtes (Isoetaceae): A provisional checklist of the accepted and unresolved taxa. Phytotaxa 2016, 277, 101–145. [Google Scholar] [CrossRef]
- Brunton, D.F.; Troia, A. Global review of recent taxonomic research into Isoëtes (Isoetaceae), with implications for biogeography and conservation. Fern Gaz. 2018, 20, 309–333. [Google Scholar]
- Hoot, S.B.; Taylor, W.C.; Napier, N.S. Phylogeny and biogeography of Isoëtes (Isoëtaceae) based on nuclear and chloroplast DNA sequence data. Syst. Bot. 2006, 31, 449–460. [Google Scholar] [CrossRef]
- Seddon, B. Occurrence of Isoëtes echinospora in eutrophic lakes in Wales. Ecology 1965, 46, 747–748. [Google Scholar] [CrossRef]
- Hickey, R.J.; Macluf, C.; Taylor, W.C. A re-evaluation of Isoëtes savatieri Franchet in Argentina and Chile. Am. Fern J. 2003, 93, 126–136. [Google Scholar] [CrossRef]
- Liu, X.; Wang, J.-Y.; Wang, Q.-F. Current status and conservation strategies for Isoëtes in China: A case study for the conservation of threatened aquatic plants. Oryx 2005, 39, 335–338. [Google Scholar] [CrossRef] [Green Version]
- Kim, C.; Na, H.R.; Choi, H.-K. Genetic diversity and population structure of endangered Isoëtes coreana in South Korea based on RAPD analysis. Aquat. Bot. 2008, 89, 43–49. [Google Scholar] [CrossRef]
- Pereira, J.B.S.; Salino, A.; Arruda, A.; Stutzel, T. Two New species of Isoëtes (Isoetaceae) from northern Brazil. Phytotaxa 2016, 272, 141–148. [Google Scholar] [CrossRef] [Green Version]
- Troia, A.; Azzella, M.M. Isoëtes sabatina (Isoëtaceae, Lycopodiophyta), a new aquatic species from central Italy. Plant Biosyst. 2013, 147, 1052–1058. [Google Scholar] [CrossRef]
- Abeli, T.; Cauzzi, P.; Rossi, G.; Pistoja, F.; Mucciarelli, M. A gleam of hope for the critically endangered Isoëtes malinverniana: Use of small scale translocations to guide conservation planning. Aquat. Conserv. 2018, 28, 501–505. [Google Scholar] [CrossRef]
- Jacobsen, D.; Dangles, O. Ecology of High Altitude Waters; Oxford University Press: Oxford, UK, 2017; p. 284. [Google Scholar]
- Free, G.; Bowman, J.; McGarrigle, M.; Caroni, R.; Donnelly, K.; Tierney, D.; Trodd, W.; Little, R. The identification, characterization and conservation value of isoetid lakes in Ireland. Aquat. Conserv. 2009, 19, 264–273. [Google Scholar] [CrossRef]
- García Criado, M.; Väre, H.; Nieto, A.; Bento Elias, R.; Dyer, R.; Ivanenko, Y.; Ivanova, D.; Lansdown, R.; Molina, J.A.; Rouhan, G.; et al. European Red List of Lycopods and Ferns; IUCN: Brussels, Belgium, 2017; p. iv + 59. [Google Scholar]
- Oh, M.J.; Kim, C.; Na, H.R.; Shin, H.; Liu, J.R.; Choi, H.-K.; Kim, S.W. High frequency sporophytes regeneration from the spore culture of the endangered aquatic fern Isoëtes coreana. Am. J. Plant Sci. 2013, 4, 14–20. [Google Scholar] [CrossRef] [Green Version]
- Christenhusz, M.; Bento Elias, R.; Dyer, R.; Ivanenko, Y.; Rouhan, G.; Rumsey, F.; Väre, H. Isoëtes sabatina. In The IUCN Red List of Threatened Species 2017; E.T83515150A83515211; IUCN: Gland, Switzerland, 2017. [Google Scholar] [CrossRef]
- Orsenigo, S.; Montagnani, C.; Fenu, G.; Gargano, D.; Peruzzi, L.; Abeli, T.; Alessandrini, A.; Bacchetta, G.; Bartolucci, F.; Bovio, M.; et al. Red Listing plants under full national responsibility: Extinction risks and threats in the vascular flora endemic to Italy. Biol. Conserv. 2018, 224, 213–222. [Google Scholar] [CrossRef]
- Barni, E.; Minuzzo, C.; Gatto, F.; Lonati, M.; Abeli, T.; Amosso, C.; Rossi, G.; Siniscalco, C. Estimating influence of environmental quality and management of channels on survival of a threatened endemic quillwort. Aquat. Bot. 2013, 107, 39–46. [Google Scholar] [CrossRef] [Green Version]
- Caldeira, C.F.; Abranches, C.B.; Gasutauer, M.; Ramos, S.; Guimarães, J.T.F.; Pereira, J.B.S.; Siqueira, J.O. Sporeling regeneration and ex situ growth of Isoëtes cangae (Isoetaceae): Initial steps towards the conservation of a rare Amazonian quillwort. Aquat. Bot. 2019, 152, 51–58. [Google Scholar] [CrossRef]
- Giuffrida, A.; Taylor, M. Romans threatened with water rationing as Italy’s heatwave drags on. The Guardian. 2017. Available online: https://www.theguardian.com/world/2017/jul/24/rome-water-rationing-italy-heatwave (accessed on 13 June 2020).
- Horowitz, J. Rome, City of Ancient Aqueducts, Faces Water Rationing. The New York Times. 2017. Available online: https://www.nytimes.com/2017/07/27/world/europe/rome-water-shortage.html (accessed on 13 June 2020).
- Baccetti, N.; Bellucci, V.; Bernabei, S.; Bianco, P.; Braca, G.; Bussettini, M.; Cascone, C.; Ciccarese, L.; D’Antoni, S.; Grignetti, A.; et al. Analisi e Valutazione Dello Stato Ambientale del Lago di Bracciano Riferito All’estate 2017; Rapporto ISPRA; ISPRA: Roma, Italy, 2017; Available online: www.isprambiente.gov.it/files2017/notizie/Relazione_ISPRA_Bracciano_18ottobre2017.pdf (accessed on 13 June 2020). (In Italian)
- Hoerling, M.; Eischeid, J.; Perlwitz, J.; Quan, X.; Zhang, T.; Pegion, P. On the increased frequency of Mediterranean drought. J. Clim. 2012, 25, 2146–2161. [Google Scholar] [CrossRef] [Green Version]
- Planton, S.; Lionello, P.; Artale, V.; Aznar, R.; Carrillo, A.; Colin, J.; Congedi, L.; Dubois, C.; Elizalde, A.; Gualdi, S.; et al. Chapter 8. The Climate of the Mediterranean Region in Future Climate Projections. In The Climate of the Mediterranean Region; Lionello, P., Ed.; Elsevier: Amsterdam, The Netherlands, 2012; pp. 449–502. [Google Scholar] [CrossRef] [Green Version]
- Magrini, S.; Olmati, C.; Onofri, S.; Scoppola, A. Recovery of viable germplasm from herbarium specimens of Osmunda regalis L. Am. Fern J. 2010, 100, 159–166. [Google Scholar] [CrossRef]
- Magrini, S. Herbaria as useful spore banks for integrated conservation strategies of pteridophytic diversity. Plant Biosyst. 2011, 145, 635–637. [Google Scholar] [CrossRef]
- Magrini, S.; Scoppola, A. Agravitropic growth of the early leaves of apogamous sporophytes of Dryopteris tyrrhena. Am. Fern J. 2012, 102, 181–183. [Google Scholar] [CrossRef]
- Magrini, S.; Scoppola, A. First results from conservation studies of chlorophyllous spores of the Royal fern (Osmunda regalis, Osmundaceae). Cryobiology 2012, 64, 65–69. [Google Scholar] [CrossRef]
- Fay, M.F. In what situations is in vitro culture appropriate to plant conservations? Biodivers. Conserv. 1994, 3, 176–183. [Google Scholar] [CrossRef]
- Sarasan, V.; Cripps, R.; Ramsay, M.M.; Atherton, C.; McMichen, M.; Prendergast, G.; Rowntree, J.K. Conservation in vitro of threatened plants—Progress in the past decade. In Vitro Cell. Dev. Biol. Plant 2006, 42, 206–214. [Google Scholar] [CrossRef]
- Engelmann, F. Use of biotechnologies for the conservation of plant biodiversity. In Vitro Cell. Dev. Biol. Plant 2011, 47, 5–16. [Google Scholar] [CrossRef]
- Abeli, T.; Mucciarelli, M. Notes on the Natural History and reproductive biology of Isoëtes malinverniana. Am. Fern J. 2010, 100, 235–237. [Google Scholar] [CrossRef]
- Čtvrtlíková, M.; Znachor, P.; Vrba, J. The effect of temperature on the phenology of germination of Isoëtes lacustris. Preslia 2014, 86, 279–292. [Google Scholar]
- Čtvrtlíková, M.; Znachor, P.; Nedoma, J.; Vrba, J. The effect of temperature on the phenology of germination of Isoëtes echinospora. Preslia 2012, 84, 141–153. [Google Scholar]
- Kott, L.S.; Britton, D.M. A comparative study of spore germination of some Isoëtes species of northeastern North America. Can. J. Bot. 1982, 60, 1679–1687. [Google Scholar] [CrossRef]
- La Motte, C. Morphology of the Megagametophyte and the Embryo Sporophyte of Isoetes lithophila. Am. J. Bot. 1933, 20, 217–233. [Google Scholar] [CrossRef]
- Karrfalt, E. Some Observations on the Reproductive Anatomy of Isoetes andicola. Am. Fern J. 1999, 89, 198–203. [Google Scholar] [CrossRef]
- Kienitz-Gerloff, F. Über Wachstum und Zelltheilung in der Entwickelung des Embryos von Isoetes lacustris. Bot. Zeit. 1881, 39, 761–770. [Google Scholar]
- Reid, A.J.; Carlson, A.K.; Creed, I.F.; Eliason, E.J.; Gell, P.A.; Johnson, P.T.J.; Kidd, K.A.; MacCormack, T.J.; Olden, J.D.; Ormerod, S.J.; et al. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biol. Rev. 2019, 94, 849–873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Işik, K. Rare and endemic species: Why are they prone to extinction? Turk. J. Bot. 2011, 35, 411–417. [Google Scholar] [CrossRef]
- Gilpin, M.E.; Soulé, M.E. Minimum viable populations: The processes of species extinctions. In Conservation Biology: The Science of Scarcity and Diversity; Soulé, M.E., Ed.; Sinauer: Sunderland, MA, USA, 1986; pp. 13–34. [Google Scholar]
- Lamont, B.B.; Klinkhamer, P.G.L.; Witkowski, E.T.F. Population fragmentation may reduce fertility to zero in Banksia goodii—A demonstration of the Allee effect. Oecologia 1993, 94, 446–450. [Google Scholar] [CrossRef]
- Coelho, N.; Gonçalves, S.; Romano, A. Endemic plant species conservation: Biotechnological approaches. Plants 2020, 9, 345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barnicoat, H.; Cripps, R.; Kendon, J.; Sarasan, V. Conservation in vitro of rare and threatened ferns—Case studies of biodiversity hotspot and island species. In Vitro Cell. Dev. Biol. Plant 2011, 47, 37–45. [Google Scholar] [CrossRef]
- Bolpagni, R.; Laini, A.; Azzella, M.M. Short-term dynamics of submerged aquatic vegetation diversity and abundance in deep lakes. Appl. Veg. Sci. 2016, 19, 711–723. [Google Scholar] [CrossRef]
- Azzella, M.M.; Bresciani, M.; Nizzoli, N.; Bolpagni, R. Aquatic vegetation in deep lakes: Macrophyte co-occurrence patterns and environmental determinants. J. Limnol. 2017, 76, 97–108. [Google Scholar] [CrossRef] [Green Version]
- Taylor, W.C.; Luebke, N.T. Germinating spores and growing sporelings of aquatic Isoëtes. Am. Fern J. 1986, 76, 21–24. [Google Scholar] [CrossRef]
- Niklas, K.J. Embryo morphology and seedling evolution. In Seedling Ecology and Evolution; Leck, M.A., Parker, V.T., Simpson, R.L., Eds.; Cambridge University Press: Cambridge, UK, 2008; pp. 103–129. [Google Scholar]
- Wardlaw, C.W. Physiology of embryonic development in cormophytes. In Encyclopedia of Plant Physiology XV Part 1. Differentiation and Development; Addicott, F.T., Lang, A., Ruhland, W., Eds.; Springer: Berlin, Germany, 1965; pp. 844–965. [Google Scholar]
- Ranal, M.A.; Santana, D.G. How and why to measure the germination process? Rev. Bras. Bot. 2006, 29, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Ranal, M.A.; Garcia De Santana, D.; Resende Ferreira, W.; Mendes-Rodrigues, C. Calculating germination measurements and organizing spreadsheets. Rev. Bras. Bot. 2009, 32, 849–855. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann, W.A.; Poorter, H. Avoiding bias in calculations of Relative Growth Rate. Ann. Bot. 2002, 90, 37–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]




| Substrates | Distilled Water | Water-Agar Medium | ||
|---|---|---|---|---|
| Megaspore Developmental Stage | Mature (n = 104) | Mature (n = 361) | Immature (n = 205) | |
| G (%) | 80.1 ± 5.0 a | 82.1 ± 13.1 a | 65.6 ± 16.1 b | * |
| TG (days) | 41.7 ± 1.5 a | 48.3 ± 9.3 a | 77.0 ± 6.7 b | ** |
| Sporeling Emergence Indices | Distilled Water | Water-Agar Medium | ||
|---|---|---|---|---|
| Mature Megaspores | Mature Megaspores | Immature Megaspores | ||
| SE (%) | 64.5 ± 8.3a | 69.6 ± 15.5a | 11.6 ± 7.9b | ** |
| SEG (%) | 80.4 ± 5.4a | 85.0 ± 12.7a | 16.7 ± 7.9b | ** |
| T1 (day) | 52.7 ± 3.1a | 59.5 ± 8.5a | 85.0 ± 6.7b | * |
| MDE (% sporelings/day) | 0.8 ± 0.2a | 0.6 ± 0.1b | 0.1 ± 0.1c | *** |
| MET (day) | - | 82.9 ± 6.7 | 87.9 ± 5.0 | ns |
| MER (day−1) | - | 0.012 ± 0.001 | 0.011 ± 0.001 | ns |
| CVt (%) | - | 12.1 ± 1.8 | 6.9 ± 4.3 | * |
| EI (n. sporelings/day) | - | 0.8 ± 0.4 | 0.1 ± 0.1 | * |
| PV (day−1) | - | 0.70 ± 0.11 | 0.12 ± 0.07 | *** |
| U (bit) | - | 3.1 ± 0.3 | 1.7 ± 1.1 | * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magrini, S.; Azzella, M.M.; Bolpagni, R.; Zucconi, L. In Vitro Propagation of Isoëtes sabatina (Isoetaceae): A Key Conservation Challenge for a Critically Endangered Quillwort. Plants 2020, 9, 887. https://doi.org/10.3390/plants9070887
Magrini S, Azzella MM, Bolpagni R, Zucconi L. In Vitro Propagation of Isoëtes sabatina (Isoetaceae): A Key Conservation Challenge for a Critically Endangered Quillwort. Plants. 2020; 9(7):887. https://doi.org/10.3390/plants9070887
Chicago/Turabian StyleMagrini, Sara, Mattia M. Azzella, Rossano Bolpagni, and Laura Zucconi. 2020. "In Vitro Propagation of Isoëtes sabatina (Isoetaceae): A Key Conservation Challenge for a Critically Endangered Quillwort" Plants 9, no. 7: 887. https://doi.org/10.3390/plants9070887








