Abstract
This paper studies the global dynamics of a cholera model incorporating age structures and general infection rates. First, we explore the existence and point dissipativeness of the orbit and analyze the asymptotical smoothness. Then, we perform rigorous mathematical analysis on the existence and local stability of equilibria. Based on the uniform persistence, we further investigate the global behavior of the cholera infection model. The results of theoretical analysis are well confirmed by numerical simulations. This research generalizes some known results and provides deeper insights into the dynamics of cholera propagation.
MSC:
92D30; 34K20
1. Introduction
Cholera is a serious infectious disease that is caused by the bacterium Vibrio cholera. Due to the cholera toxin produced by the bacterium Vibrio cholera, it is characterized by severe symptoms, including acute diarrhea, vomiting, hypotension and a weak pulse. Without proper medical treatment, it can cause dehydration and death within hours. This disease peaks in summer and its propagation among humans depends on direct person-to-person contact, as well as indirect contact through contaminated food and water [1,2]. Due to the lack of clean food and water, cholera can spread quickly in regions with poor sanitation conditions and has long been a threat to the public health of human society. In 2018, it was estimated that there were 2.9 million burden cases worldwide, with a death toll of around 95,000, which corresponds to dozens of countries and regions [3].
A mathematical model of cholera propagation was first proposed to study cholera infection in 1973 around the Mediterranean region [4]. Henceforward, there have been numerous studies on the dynamics of cholera infection models. Tien and Earn [2] established a water-borne infectious disease model including multiple propagation paths. Control strategies, such as vaccination, were also considered in cholera models in Posny et al. [5] to inhibit the propagation of epidemics. Recently, by combining the cholera infection around aquatic regions, as well as the interaction between the bacteriophage and the cholera bacterium, researchers constructed a refined cholera infection model and provided reasonable cholera control strategies [6]. Considering environmental uncertainties and stochastic factors, researchers also studied a cholera system with respect to the stochastic differential equation and confirmed the decisive effect of the stochastic basic reproduction number on the system [7].
Age structure, incorporating the age structure of the pathogen the infection age of individuals, is a significant characteristic in the cholera model [8,9,10,11]. A cholera model with bilinear incidence rates including two age structures was introduced and discussed in the work of Brauer et al. [12] and was further investigated in the work of Wang and Zhang [13]. The relative compactness of the orbits and the uniform persistence of the system were explored in [13]. The local stability of disease-free equilibrium and endemic equilibrium was analyzed in [13] and global stability was studied in [12]. Furthermore, a cholera transmission model incorporating vaccination age was analyzed in [14]. Actually, incidence rates are influenced by the complicated connections between susceptible individuals and the infected individuals/pathons. Various nonlinear incidence rates have been considered by researchers [15,16,17,18,19].
Inspired by the above works, we aim to discuss an age-structured cholera model. At time t, let and stand for the number of susceptible individuals and infected individuals with infection age a, with representing the quantity of aquatic cholera pathogens at the age of b. Then, the infectivity of infected individuals and the total infectivity of the cholera pathogen at time t can be measured by and , in which kernel functions denote the infectivity of infected individuals and pathogens at corresponding ages. In this manuscript, we consider the following cholera model, taking general incidence rates into account, which is a generation of the model in Brauer et al. [12].
with boundary conditions
and the initial condition
where is a functional space equipped with the norm
In model (1), denotes the recruitment of the susceptible, and represents the natural death rate of individuals. describes the removal rate of pathogens at age b and describes the pathogen shedding rate of an infected patient with infection age a. , where is the disease-related death rate and accounts for the recovery rate of infected individuals at infection age a. and represent the direct and indirect transmission of cholera. For system (1), we make the following assumptions.
Assumption 1.
(I) The functions are bounded, integrable and Lipschitz-continuous. Denote and as essential upper and lower bound of for .
- (II)
- There exists one positive constant satisfying for .
- (III)
- and are Lipschitz-continuous on with , and , for .
In this paper, for an age-infection model, we analyze the qualitative behavior by means of the Lyapunov functional method [20,21,22]. By considering the routes of the spread from the pathogen to the susceptible group and from the infected group to the susceptible group spread with generalized infection functions, we form a unified theoretical structure to present the propagation features of the epidemic. The basic reproduction number is defined as the threshold value, determining whether the epidemic dies out or not. Specifically, the cholera epidemic withers away if , whereas if , the disease persists at the endemic level.
The plan of this article is as follows. We give some preliminaries in the next section. In Section 3, we explore the existence and local stability of equilibria. In Section 4, we construct Lyapunov functionals to discuss the global stability of equilibria. In Section 5, we perform numerical simulations. Section 6 presents brief conclusions and a discussion.
2. Preliminaries
2.1. Existence and Uniqueness of Solutions
The standard theory for age-dependent models [8,11] can be applied to establish the existence and uniqueness of solutions for system (1) with boundary conditions (2) and initial condition (3). For this, we introduce the following Banach spaces
In order to formulate system (1) as an abstract Cauchy problem [23], we define the following linear operator, where ,
and the nonlinear operator
Similarly to the proof process in [24,25], we can verify that operator is a Hille–Yosida operator [23].
Let . System (1) can be expressed by the following abstract cauchy problem:
Let . We have the following theorem by [23,26]:
Theorem 1.
There exists one unique determined semiflow on such that for any , there exists one unique continuous map , acting as an integrated solution of the Cauchy problem, that is
2.2. Point Dissipativeness
Let
Then we have the following proposition:
Theorem 2.
Ξ is a positive invariant set under the semiflow . Moreover, the semiflow is point-dissipative and attracts all the positive solutions of system (1) in .
Proof.
From the first equation of (1), we have . Due to , we have . Note that
Combining the first equation of (1), one yields
Since , we have . Thus, it follows that
This implies that .
Hence, and this implies that is a positively invariant set and attracts all positive solutions of (1). □
From Theorem 2, we obtain the following result.
Proposition 1.
If and with some sufficiently large constant B, then for , we have the following propositions
- (i)
- , , ;
- (ii)
- , .
Proof.
From the boundedness of system (1), we can find a constant B such that proposition (i) holds. Due to Assumption 1 (III), we further have
and
This completes the proof. □
2.3. Asymptotical Smoothness and Global Attractor
From Equations (2) and (3), using the method presented in [8] to integrate the second and the third equations in (1) along the characteristic lines , we have
and
where
denote the fraction at which an infected cell and virus survive up to age a and b.
In order to explore the existence of an attractor, we first analyze the asymptotical smoothness of semiflow . For this, we present the following proposition.
Proposition 2.
The functions , and are Lipschitz-continuous.
Proof.
Here we give the proof of being Lipschitz-continuous. From Assumption 1, there exists a positive constant such that . Then, combining Proposition 1, it holds that
Let ; we have
Hence, is Lipschitz-continuous with the coefficient . Through similar verification, the functions and are Lipschitz-continuous with coefficients and . □
For the asymptotical smoothness of the semiflow, the following lemma [27] is necessary.
Lemma 1.
The semiflow is asymptotically smooth if there are maps Ψ, such that and the following holds for any bounded closed set , which is forward invariant under : (i); (ii) There exists such that has compact closure for each .
For condition (ii) of Lemma 1, we introduce the following lemma [27].
Lemma 2.
A set has a compact closure if the following conditions hold: (i) ; (ii) uniformly in ; (iii) uniformly in ; (iv) uniformly in .
Based on Lemmas 1 and 2, we investigate the asymptotical smoothness.
Theorem 3.
The semiflow generated by (1) is asymptotically smooth.
Proof.
Define the maps and such that , with
where
Firstly, we show that map satisfies condition (i) of Lemma 1. For satisfying , letting and , we have
This shows that as , which indicates that approaches 0 with uniform exponential speed. Thus, the proof of Lemma 1 (i) is completed.
Then, we verify that Lemma 2 holds. Using Proposition 1, we can verify that conditions (i), (ii) and (iv) of Lemma 2 hold since
Finally, we focus on condition (iii) of Lemma 2. For sufficiently small , we have
where
Note that
Let be the Lipschitz coefficient of . Then, the following holds:
Similarly, we have
does not rely on the initial condition . Thus, Lemma 2 holds. Hence, remains in a pre-compact subset in , and so does . We thus accomplish the proof. □
Based on the above preparations, the following results hold due to Theorem 3.4.6 of Hale [28].
Theorem 4.
The semi-flow has a global attractor in , which attracts all bound subsets of .
3. Existence and Local Stability of Equilibria
3.1. Equilibria and Basic Reproductive Number
System (1) possesses two equilibria at most in . Besides the infection-free equilibrium with , there possibly exists an infection equilibrium in , satisfying the following equations
where and .
From the second and third equations of system (11), we have
Let
We can further obtain
and
Thus, combining equations , (13) and (14), we have
Let . Then, we yield , and
Define the basic reproduction number of system (1) as
When , and there exists at least one . Then, we obtain
and
Thus, there exists one unique positive equilibrium . This yields the following theorem.
Theorem 5.
System (1) always exists a disease-free steady state . Furthermore, another endemic steady state exists if .
3.2. Local Stability of Equilibria
The global asymptotical stability of equilibria is conducive to forecasting the trends of epidemics [29,30,31,32,33,34,35]. For this, we first focus on the local stability by exploring the corresponding characteristic equations.
Theorem 6.
The infection-free equilibrium is locally asymptotically stable when . The infection equilibrium is locally asymptotically stable when .
Proof.
The characteristic equation for the linearized part of system (1) with boundary conditions (2) on is
where
Then, if , all roots of the characteristic equation (16) have negative parts. If not, that is, if there exists a such that , then
This is a contradiction with equation (16). Thus, the infection-free equilibrium is locally asymptotically stable when .
Similarly, for , combining the linearization of the system on , the corresponding characteristic equation of the linearization for system (1) is
Now we assume that system (17) has one characteristic root with a positive real root. Since and , we have
This is a contradiction with Equation (17). Thus, is locally asymptotically stable when . □
4. Global Stability of Equilibria
For the proof of the global attractiveness of equilibria, we apply the Lyapunov functional method. For the invariance principle, we have investigated the relative compactness of the orbits. For the well-posedness of Lyapunov functionals, the uniform persistence of system should also be discussed.
4.1. Uniform Persistence
In this section, we aim to investigate the uniform persistence of system (1). Define
and .
Lemma 3.
The subsets M and are both positively invariant under the semiflow generated by system (1) on , that is,
Moreover, for each as .
Proof.
Let . For any , we have
For any , we have . Thus, and then we have . Thus, M is positively invariant.
In the following, we try to prove that . For any , we have
and
Thus, and . □
Then we obtain the following theorem by means of [36].
Theorem 7.
If , then the semiflow is uniformly persistent with respect to the pair , that is, there exists , such that .
Proof.
We need to verify that , where
Suppose there exists . Then, there exists a , such that
Since M is an invariant set, we have
Since , we have . Thus, for , there exists , such that
Let , where
Then we have
Since , and
we further have for a sufficiently large t,
This indicates that is a non-decreasing function for a sufficiently large t. Hence, for a sufficiently large t, , which prevents the orbits from converging to as . This contradicts . □
4.2. Global Stability of the Infection-Free Equilibrium
This subsection explores the global stability of the infection-free equilibrium .
Theorem 8.
is globally asymptotically stable when .
Proof.
Define the Liapunov function , with
Since and we further have
Thus, when , . The largest invariant set of is singleton . Hence, due to the invariance principle [37], is globally asymptotically stable when . □
4.3. Global Stability of the Infection Equilibrium
In this subsection, we focus on the global stability of the infection equilibrium . To this end, we introduce a function h defined by
In order to ensure that and are well-defined, we have shown that and are bounded below and above through the above uniform persistence analysis. In the following, we prove the following result.
Theorem 9.
The infection equilibrium is globally asymptotically stable when .
Proof.
Define a Lyapunov function , where
with
Here, we make some preparations. Firstly, since , we have
Thus,
Similarly, we have
Then, calculating the derivative of along system (1) gives
Because of equation (22), we obtain
Due to the fact that and , we have
Since
we further have
Similarly, we have
We introduce
Then we can verify that . Combining equations (24), (25) and (26), we can transfer as follows:
where
Since and , we have
Due to and , we obtain
Since , we obtain
and
Then, we have
Due to , and
we have
Thus, combining and yields
Then, due to and
we further obtain
5. Numerical Simulations
In this section, as a special case for the age-infection model (1), we consider the following model:
with the initial condition (3) and the following boundary conditions
From Theorems 8 and 9, we obtain the following corollary:
Corollary 1.
To verify the result, we perform numerical simulations. Following [6,7] and references therein, with some assumptions, we adopt the following coefficients, for ,
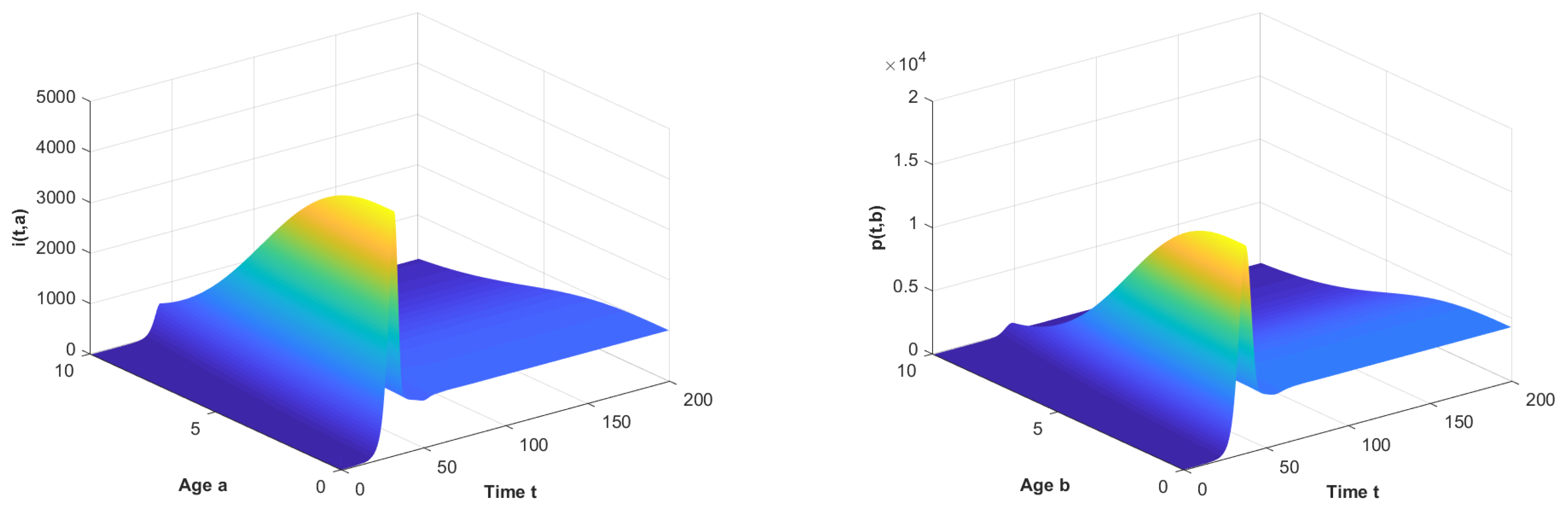
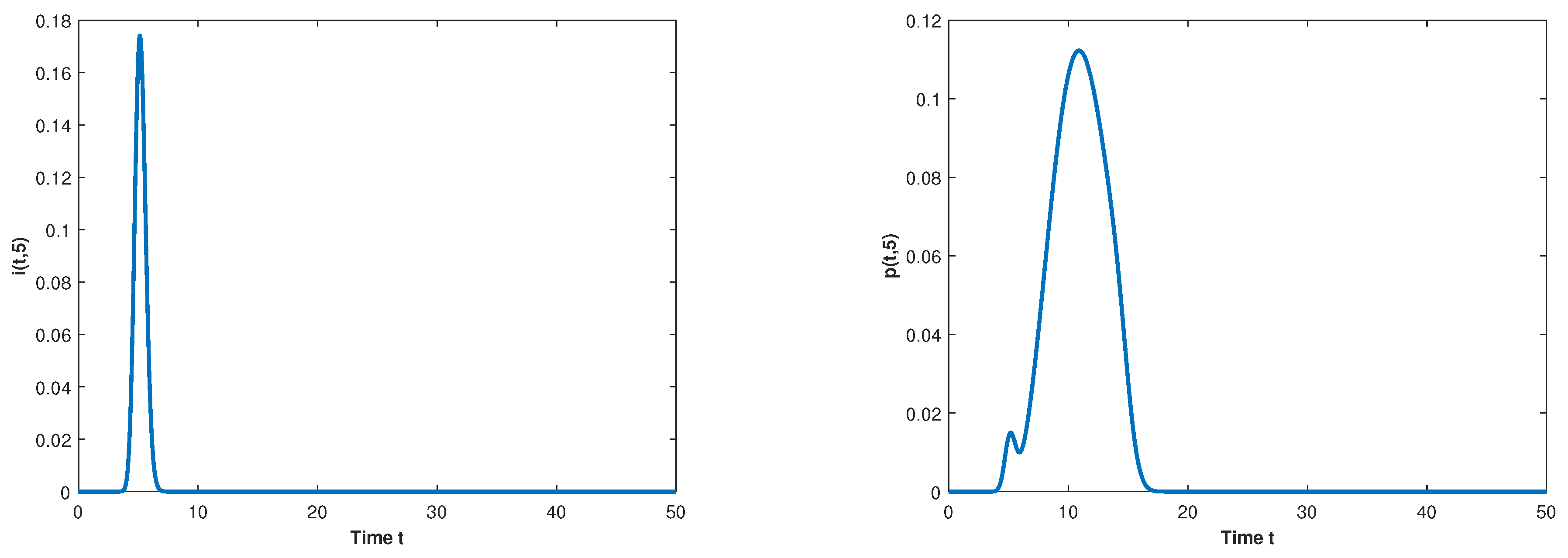
Let and observe the dynamical behavior of the model when q varies. Let decrease to . The globally asymptotically stable changes to be unstable and the epidemic is inhibited effectively, which can be seen in Figure 1 and Figure 2.
Figure 1.
The long-term dynamical behavior of and as .
Figure 2.
The long-term dynamical behavior of and for as .
6. Conclusions and Discussion
In this paper, an age-structured model of cholera infection was explored. By considering general infection functions, the discussion provided in this paper serves as a generalization and supplement to the work presented in F. Brauer et al. [12]. We applied the Lyapunov functional method to show that the global stability of equilibria are determined by the basic reproduction number . The infection-free equilibrium is globally asymptotically stable if is less than one, whereas a globally asymptotically stable infection equilibrium emerges if is greater than one. This shows that both the direct contact with infected individuals and indirect pathogen infection have vital effects on cholera epidemics. It is significant to implement effective treatment for infected individuals and to clean pathogens from contaminated water in a timely fashion. More specifically, for the critical case when equals one, further bifurcation studies are needed.
In our model, vaccinated individuals and vaccination age have not been incorporated, which play vital effects on the spread of cholera. Furthermore, the immigration of infected individuals plays a significant role in the outbreak and infection of cholera. For the actual control and elimination of cholera, it is necessary to take into account the effects of vaccination and immigration [5,38]. Thus, our future work will consider these factors and focus on their effects on cholera transmission. In addition to qualitative analyses, tremendous amounts of works on numerical methods have been proposed and developed to deal with various epidemic models [39,40,41], which provide us with more aspects and methods to analyze in relation to this model.
Funding
This research was funded by Fundamental Research Funds of Beijing Municipal Education Commission (Grant Number: 110052972027/141) and North China University of Technology Research Fund Program for Young Scholars (Grant Number: 110051360002).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The author declares no conflict of interest.
References
- Hartley, D.M.; Morris, J.G., Jr.; Smith, D.L. Hyperinfectivity: A critical element in the ability of V. cholerae to cause epidemics? PLoS Med. 2006, 3, 63–69. [Google Scholar] [CrossRef]
- Tien, J.H.; Earn, D.J.D. Multiple transmission pathways and disease dynamics in a waterborne pathogen model. Bull. Math. Biol. 2010, 72, 1506–1533. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Cholera Prevention and Control, May 2018. Available online: www.who.int (accessed on 16 April 2021).
- Capasso, V.; Paveri-Fontana, S.L. A mathematical model for the 1973 cholera epidemic in the european mediterranean region. Revue d’épidémiologie et de Santé Publiqué 1979, 27, 121. [Google Scholar]
- Posny, D.; Wang, J.; Mukandavire, Z.; Modnak, C. Analyzing transmission dynamics of cholera with public health interventions. Math. Biosci. 2015, 264, 38–53. [Google Scholar] [CrossRef]
- Misra, A.K.; Gupta, A.; Venturino, E. Cholera dynamics with bacteriophage infection: A mathematical study. Chaos Solitons Fractals 2016, 91, 610–621. [Google Scholar] [CrossRef]
- Phan, T.A.; Tian, J.P.; Wang, B. Dynamics of cholera epidemic models in fluctuating environments. Stoch. Dyn. 2021, 21, 2150011. [Google Scholar] [CrossRef]
- Brauer, F.; Driessche, P.v.; Wu, J. (Eds.) Mathematical Epidemiology; Lecture Notes in Mathematics; Springer: Berlin/Heidelberg, Germany, 2008; Volume 1945. [Google Scholar]
- Iannelli, M. Mathematical Theory of Age-Structured Population Dynamics; Appl. Math. Monogr. CNR 7; Giadini Editori e Stampator: Pisa, Italy, 1994. [Google Scholar]
- Kuniya, T. Global stability analysis with a discretization approach for an age-structured multigroup SIR epidemic model. Nonlinear Anal. RWA 2011, 12, 2640–2655. [Google Scholar] [CrossRef]
- Webb, G.F. Theory of Nonlinear Age-Dependent Population Dynamics; Marcel Dekker: New York, NY, USA; Basel, Switzerland, 1985. [Google Scholar]
- Brauer, F.; Shuai, Z.; van den Driessche, P. Dynamics of an age-of-infection cholera model. Math. Biosci. Eng. 2013, 10, 1335–1349. [Google Scholar]
- Wang, J.; Zhang, R.; Kuniya, T. A note on dynamics of an age-of-infection chorela model. Math. Biosci. Eng. 2016, 13, 227–247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cai, L.; Fan, G.; Yang, C.; Wang, J. Modeling and analyzing cholera transmission dynamics with vaccination age. J. Frankl. Inst. 2020, 357, 8008–8034. [Google Scholar] [CrossRef]
- Huang, G.; Takeuchi, Y.; Ma, W.; Wei, D. Global stability for delay SIR and SEIR epidemic models with nonlinear incidence rate. Bull. Math. Biol. 2010, 72, 1192–1207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, G.; Yokoi, H.; Takeuchi, Y.; Kajiwara, T.; Sasaki, T. Impact of intracellular delay, immune activation delay and nonlinear incidence on viral dynamics. Jpn. J. Ind. Appl. Math. 2011, 28, 383–411. [Google Scholar] [CrossRef]
- Korobeinikov, A.; Maini, P.K. Nonlinear incidence and stability of infectious disease models. Math. Med. Biol. 2005, 22, 113–128. [Google Scholar] [CrossRef] [Green Version]
- Xu, S. Global stability of the virus dynamics model with Crowley-Martin functional response. Electron. J. Qual. Theory Differ. Equ. 2012, 9, 1–10. [Google Scholar] [CrossRef]
- Zhou, X.; Cui, J. Global stability of the viral dynamics with Crowley-Martin functional response. Bull. Korean Math. Soc. 2011, 48, 555–574. [Google Scholar] [CrossRef] [Green Version]
- Huang, G.; Liu, X.; Takeuchi, Y. Lyapunov functions and global stability for age-structured HIV infection model. SIAM J. Appl. Math. 2012, 72, 25–38. [Google Scholar] [CrossRef]
- Magal, P.; McCluskey, C.C.; Webb, G.F. Lyapunov functional and global asymptotic stability for an infection-age model. Appl. Anal. 2010, 89, 1109–1140. [Google Scholar] [CrossRef]
- Melnik, A.V.; Korobeinikov, A. Lyapunov functions and global stability for SIR and SEIR models with age-dependent susceptibility. Math. Biosci. Eng. 2013, 10, 369–378. [Google Scholar]
- Magal, P.; Ruan, S. Theory and Applications of Abstract Semilinear Cauchy Problems; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Liu, S.; Zhang, R. On an Age-Structured Hepatitis B Virus Infection Model with HBV DNA-Containing Capsids. Bull. Malays. Math. Sci. Soc. 2021, 44, 1345–1370. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, S. Global dynamics of an age-structured within-host viral infection model with cell-to-cell transmission and general humoral immunity response. Math. Biosci. Eng. 2020, 17, 1450–1478. [Google Scholar] [CrossRef]
- Magal, P.; Thieme, H. Eventual compactness for a semiflow generated by an age-structured models. Commun. Pure Appl. Anal. 2004, 3, 695–727. [Google Scholar] [CrossRef]
- Smith, H.L.; Thieme, H.R. Dynamical Systems and Population Persistence; American Mathematical Society: Providence, RI, USA, 2011. [Google Scholar]
- Hale, J.K. Asymptotic Behavior of Dissipative Systems, Mathematical Surveys and Monographs; American Mathematical Society: Providence, RI, USA, 1988; Volume 25. [Google Scholar]
- Georgescu, P.; Hsieh, Y.H. Global stability for a virus dynamics model with nonlinear incidence of infection and removal. SIAM J. Appl. Math. 2006, 67, 337–353. [Google Scholar] [CrossRef] [Green Version]
- Huang, G.; Ma, W.; Takeuchi, Y. Global properties for virus dynamics model with Beddington-DeAngelis functional response. Appl. Math. Lett. 2009, 22, 1690–1693. [Google Scholar] [CrossRef] [Green Version]
- Korobeinikov, A. Global properties of basic virus dynamics models. Bull. Math. Biol. 2004, 66, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Leenheer, P.D.; Smith, H.L. Virus dynamics: A global analysis. SIAM J. Appl. Math. 2003, 63, 1313–1327. [Google Scholar]
- McCluskey, C.C. Global stability for an SEI model of infectious disease with age structure and immigration of infecteds. Math. Biosci. Eng. 2016, 13, 381–400. [Google Scholar] [CrossRef]
- Shuai, Z.; van den Driessche, P. Global dynamics of cholera models with differential infectivity. Math. Biosci. 2010, 234, 118–126. [Google Scholar] [CrossRef]
- Vargas-De-León, C.; Esteva, L.; Korobeinikov, A. Age-dependency in host-vector models: The global analysis. Appl. Math. Comput. 2014, 243, 969–981. [Google Scholar] [CrossRef]
- Hale, J.; Waltman, P. Persistence in infinite-dimensional systems. SIAM J. Math. Anal. 1989, 20, 388–395. [Google Scholar] [CrossRef]
- Walker, J.A. Dynamical Systems and Evolution Equations; Plenum Press: New York, NY, USA; London, UK, 1980. [Google Scholar]
- Brauer, F.; van den Driessche, P. Models for transmission of disease with immigration of infectives. Math. Biosci. 2001, 171, 143–154. [Google Scholar] [CrossRef]
- Khalsaraei, M.M.; Shokri, A.; Ramos, H.; Heydari, S. A positive and elementary stable nonstandard explicit scheme for a mathematical model of the influenza disease. Math. Comput. Simul. 2021, 182, 397–410. [Google Scholar] [CrossRef]
- Jódar, L.; Villanueva, R.J.; Arenas, A.J.; González, G.C. Nonstandard numerical methods for a mathematical model for influenza disease. Math. Comput. Simul. 2008, 79, 622–633. [Google Scholar] [CrossRef]
- Mickens, R.E. Numerical integration of population models satisfying conservation laws: NSFD methods. Biol. Dyn. 2007, 1, 1751–1766. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).