Augmenting the Angiogenic Profile and Functionality of Cord Blood Endothelial Colony-Forming Cells by Indirect Priming with Bone-Marrow-Derived Mesenchymal Stromal Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Isolation of Cord-Blood-Derived Endothelial Colony-Forming Cells (ECFCs)
2.3. Characterization of ECFCs
2.3.1. Immunophenotyping
2.3.2. Immunocytochemistry Characterization
2.3.3. Isolation of Bone-Marrow-Derived Mesenchymal Stem Cells (BM-MSCs)
2.4. Characterization of BM-MSCs
2.4.1. Immunophenotyping
2.4.2. Trilineage Differentiation
2.5. In Vitro Co-Culture Assay
2.5.1. Indirect Co-Culture of ECFCs and BM-MSCs
2.5.2. Direct Co-Culture of ECFCs and BM-MSCs
2.6. Adhesion Assay
2.7. Cellular Proliferation Assay
2.8. Migration Assay
2.9. In Vitro Tube Formation Assay
2.10. Proteome Profiling
2.11. Statistical Analysis
3. Results
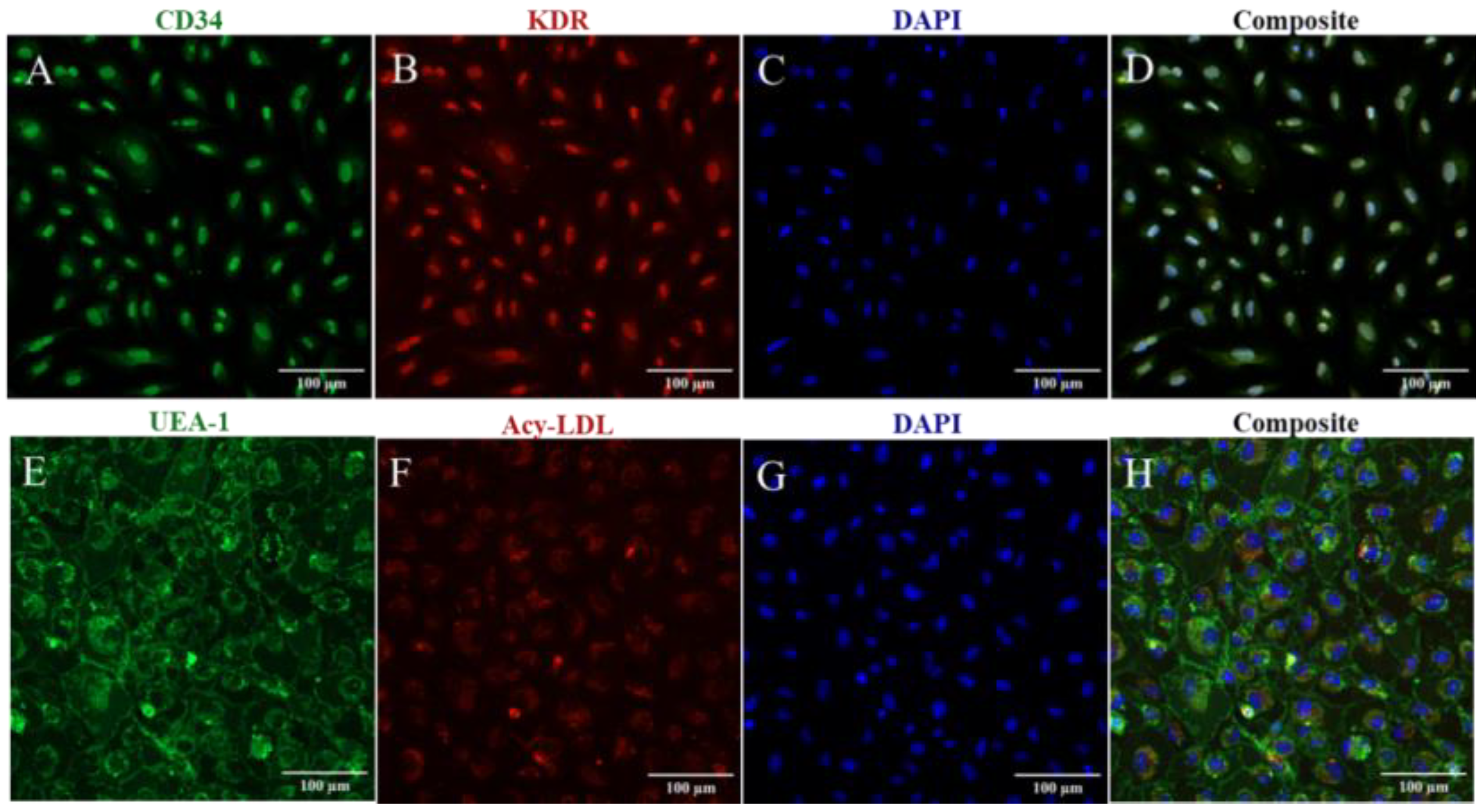
3.1. Structural and Functional Characterization of CB-ECFC
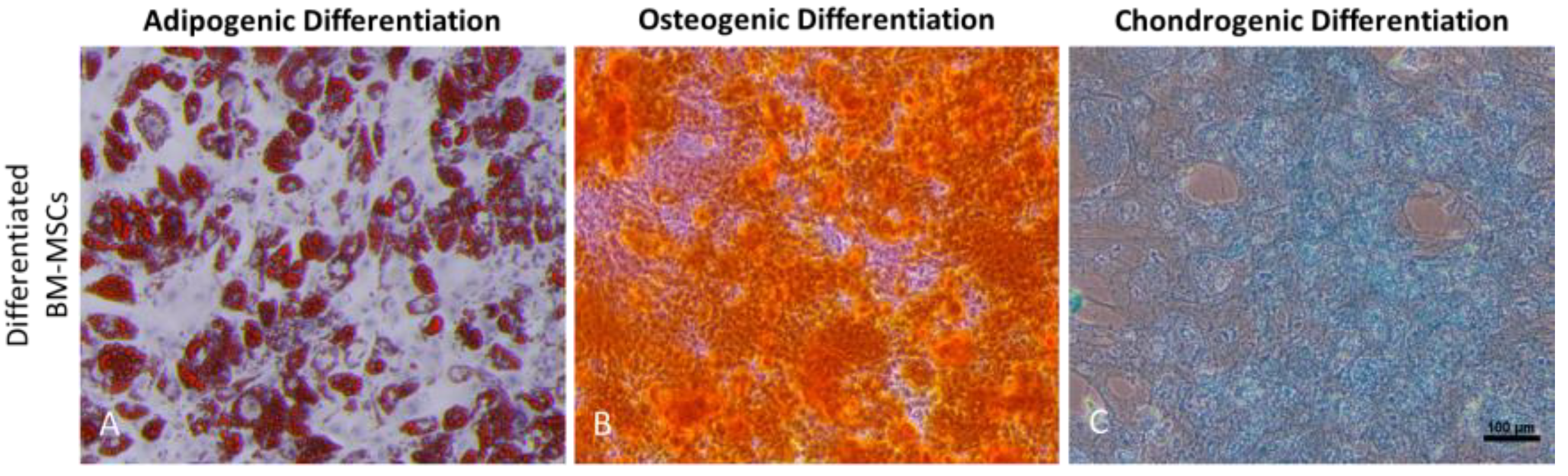
3.2. Isolation and Functional Characterization of BM-MSC
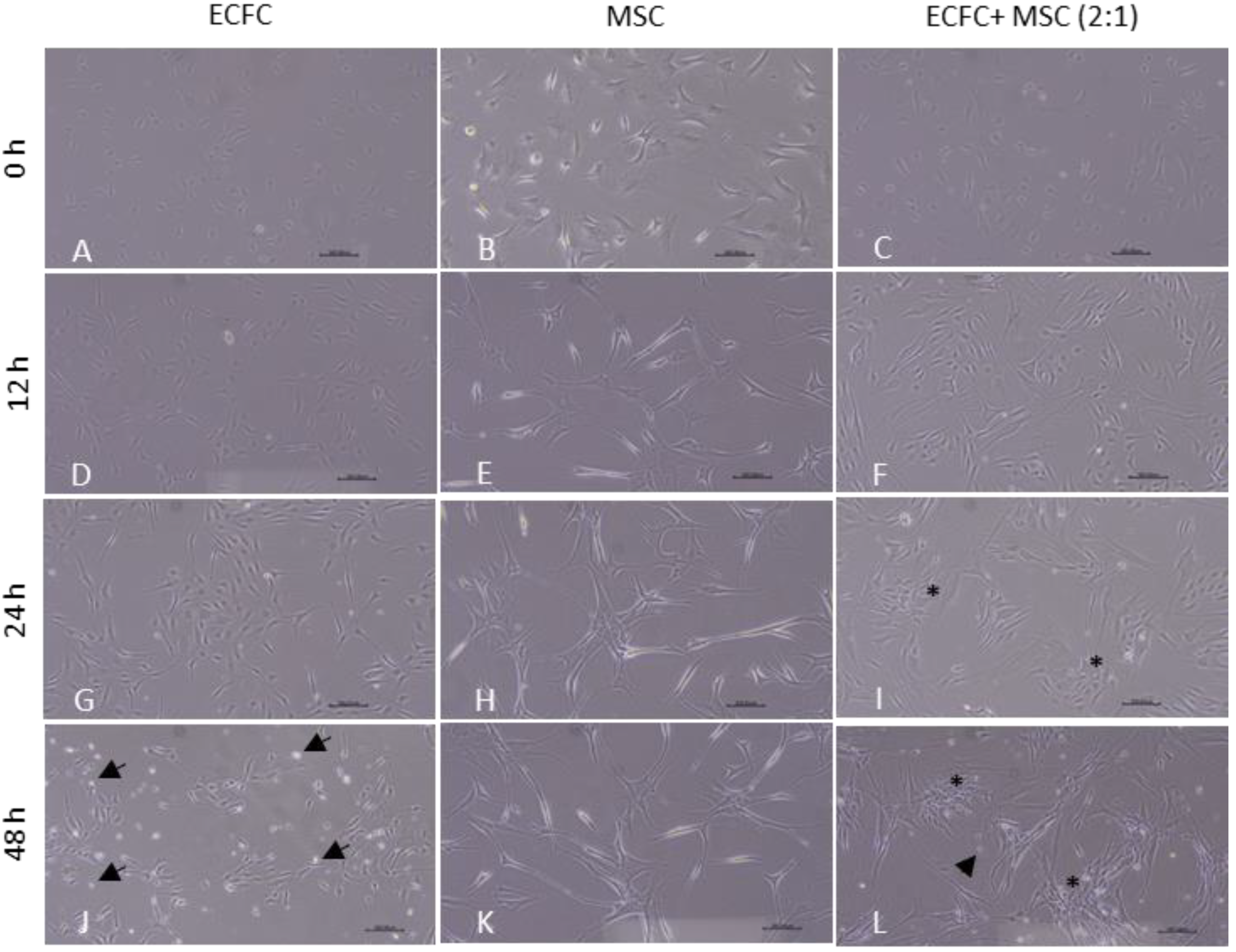
3.3. The BM-MSCs Stabilized and Imparted Fibroblast-like Morphology to CB-ECFCs
3.4. Priming CB-ECFCs with BM-MSCs Improves the Adhesion Ability of CB-ECFCs
3.5. Indirectly Primed CB-ECFCs Depicted Better Proliferative Potential Than Directly Primed CB-ECFCs
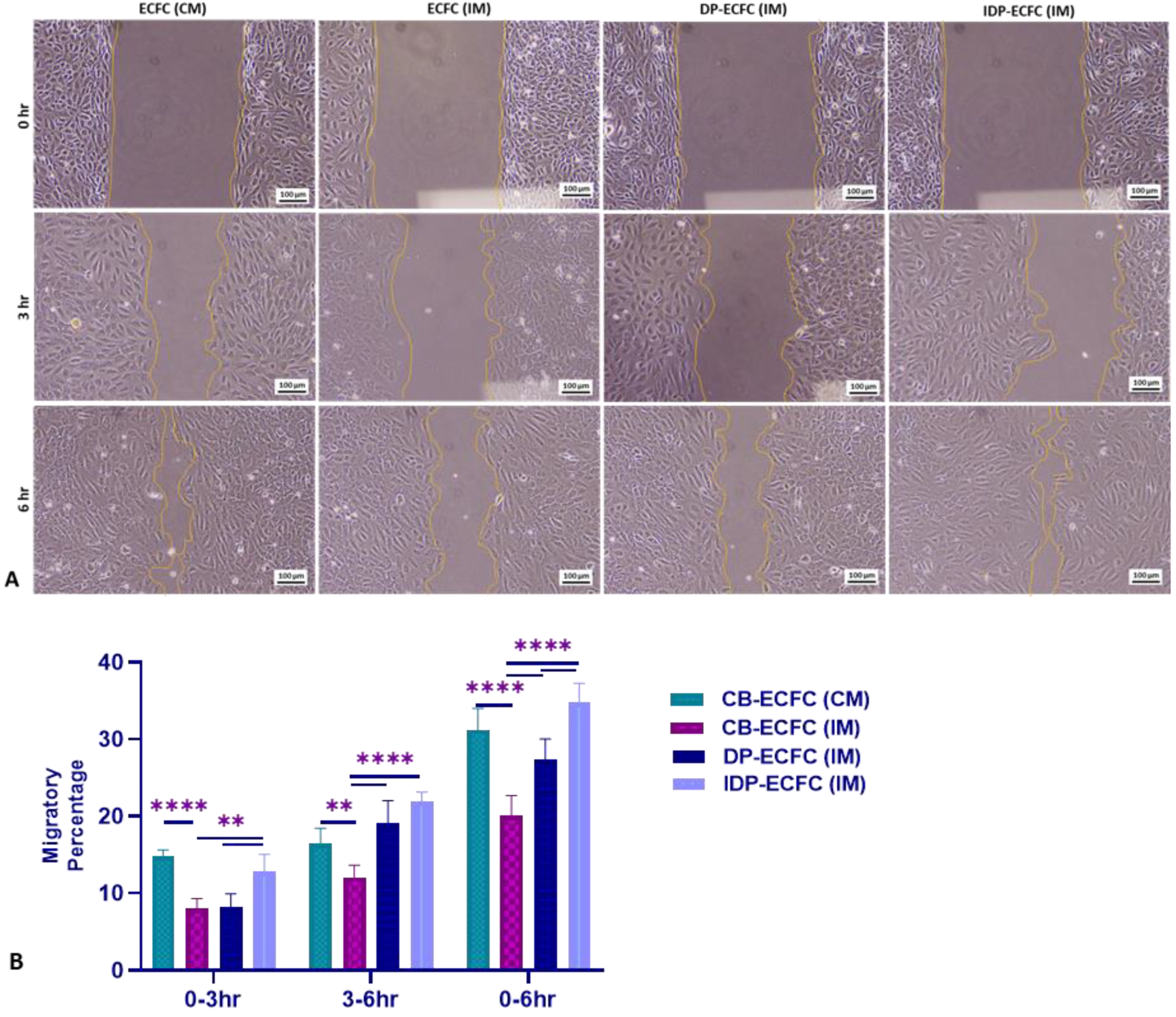
3.6. Priming CB-ECFCs with BM-MSCs Improves Their Migration Ability, with Indirectly Primed ECFCs Migrating Better Than the Directly Primed ECFCs
3.7. Priming CB-ECFCs with BM-MSCs Increases Their Angiogenic Potential
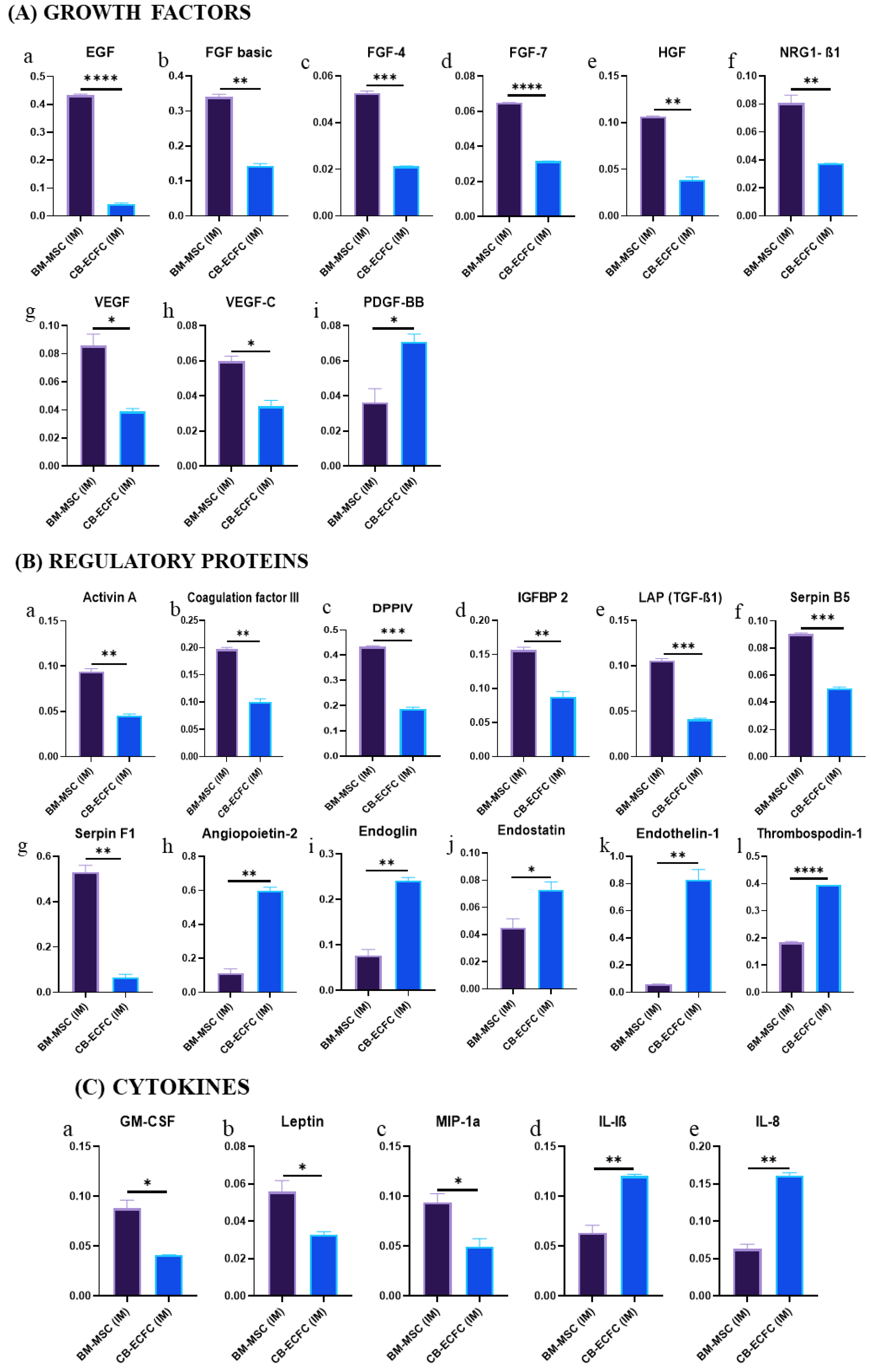
3.8. BM-MSCs Exhibit a Superior Angiogenic Profile Compared to CB-ECFCs
3.9. Starvation of CB-ECFCs Initiates Cellular Imbalance
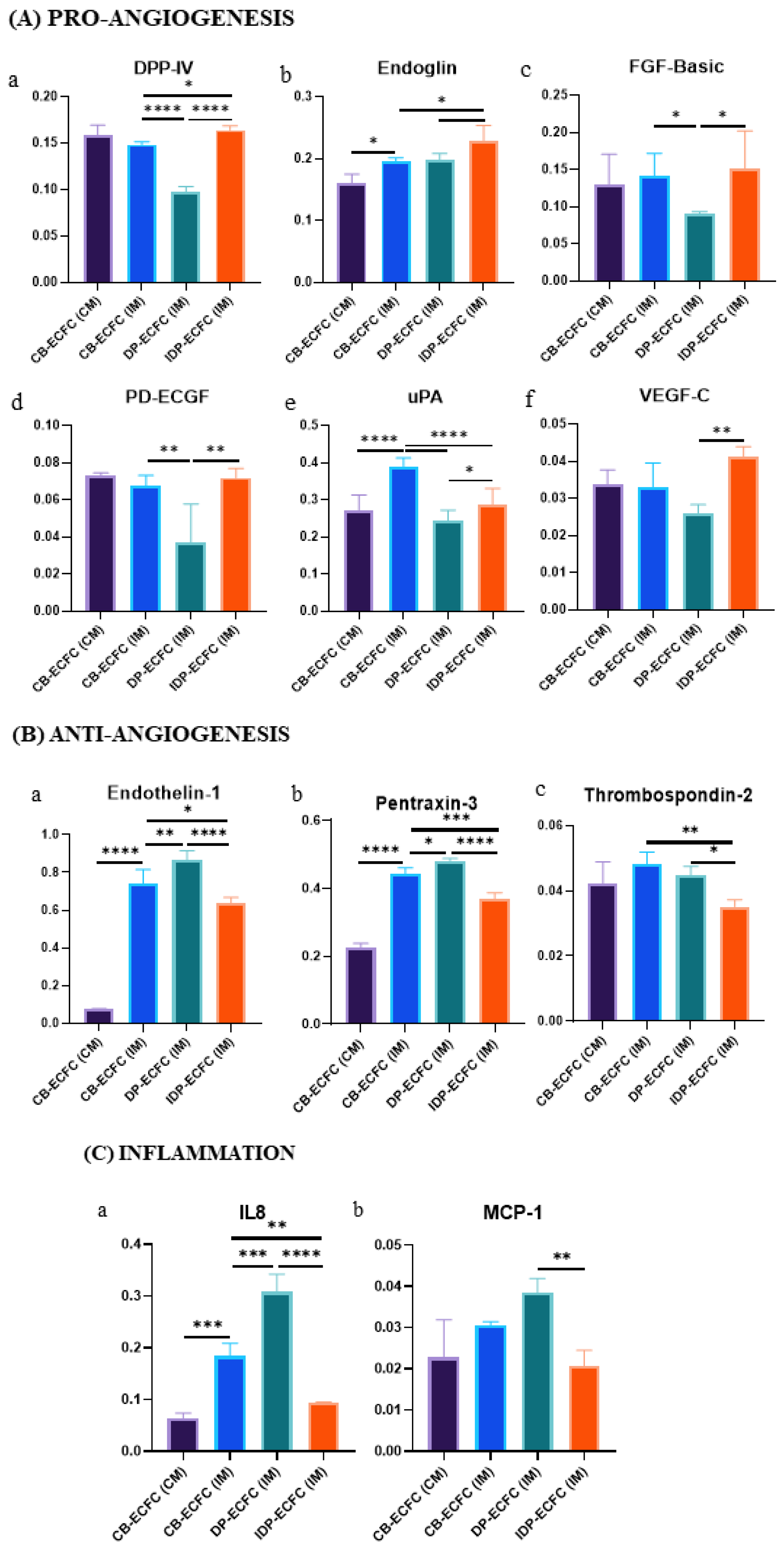
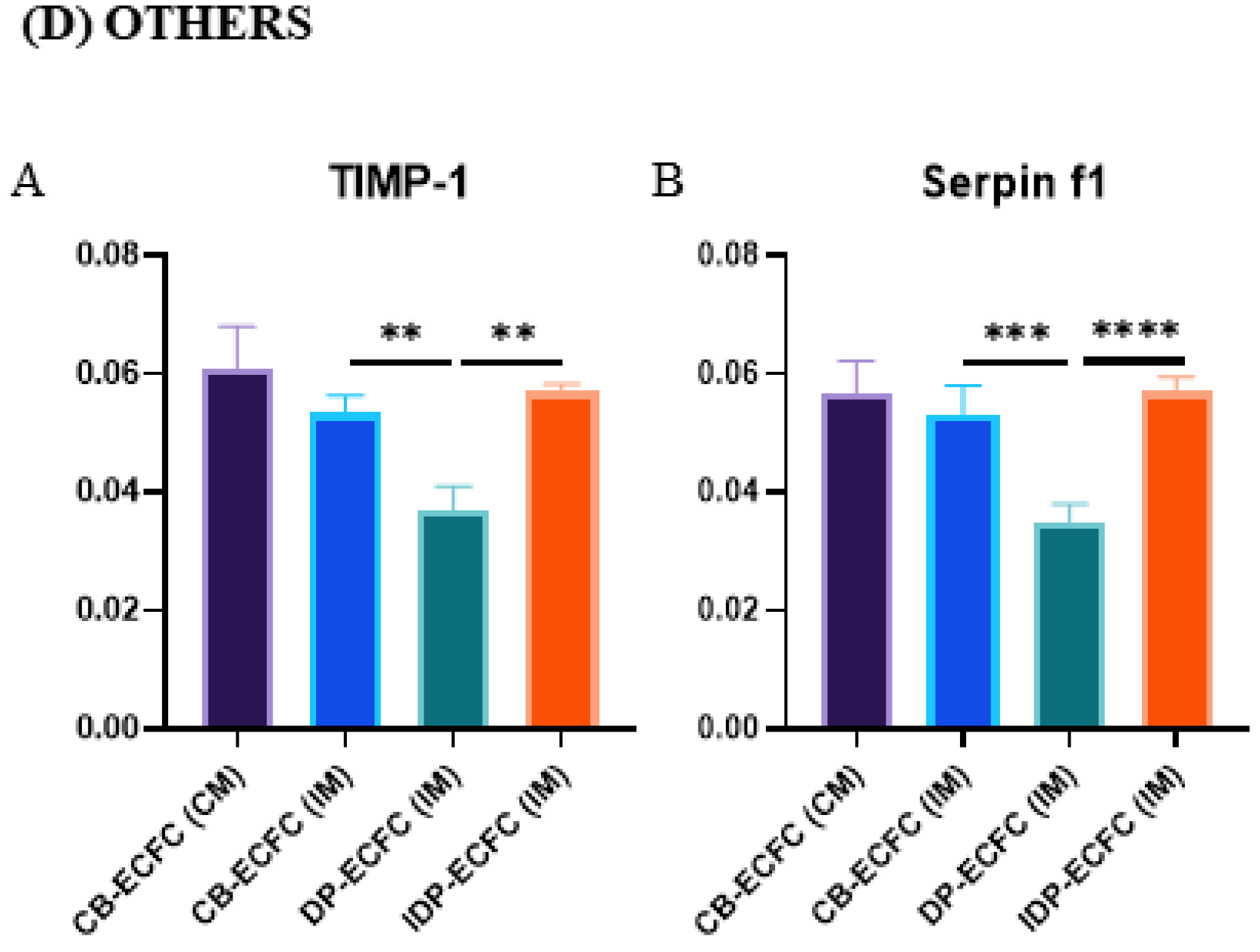
3.10. Direct and Indirect Priming Impart Different Angiogenic Stimuli
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Contributions of Risk Factors and Medical Care to Cardiovascular Mortality Trends-PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/26076950/ (accessed on 30 March 2023).
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef] [PubMed]
- Cardiovascular Diseases (CVDs). Available online: https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds) (accessed on 4 February 2023).
- Deaths Due to Heart Attacks up by 53% in 5 Years: NCRB|India News-Times of India. Available online: https://timesofindia.indiatimes.com/india/deaths-due-to-heart-attacks-up-by-53-in-5-years-ncrb/articleshow/77922046.cms (accessed on 4 February 2023).
- Asahara, T.; Murohara, T.; Sullivan, A.; Silver, M.; van der Zee, R.; Li, T.; Witzenbichler, B.; Schatteman, G.; Isner, J.M. Isolation of Putative Progenitor Endothelial Cells for Angiogenesis. Science 1997, 275, 964–967. [Google Scholar] [CrossRef] [PubMed]
- Tasev, D.; Koolwijk, P.; van Hinsbergh, V.W.M. Therapeutic Potential of Human-Derived Endothelial Colony-Forming Cells in Animal Models. Tissue Eng. Part B Rev. 2016, 22, 371–382. [Google Scholar] [CrossRef] [PubMed]
- Chong, M.S.K.; Ng, W.K.; Chan, J.K.Y. Concise Review: Endothelial Progenitor Cells in Regenerative Medicine: Applications and Challenges. Stem Cells Transl. Med. 2016, 5, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Melero-Martin, J.M.; Khan, Z.A.; Picard, A.; Wu, X.; Paruchuri, S.; Bischoff, J. In Vivo Vasculogenic Potential of Human Blood-Derived Endothelial Progenitor Cells. Blood 2007, 109, 4761–4768. [Google Scholar] [CrossRef]
- Medina, R.J.; Barber, C.L.; Sabatier, F.; Dignat-George, F.; Melero-Martin, J.M.; Khosrotehrani, K.; Ohneda, O.; Randi, A.M.; Chan, J.K.Y.; Yamaguchi, T.; et al. Endothelial Progenitors: A Consensus Statement on Nomenclature. Stem Cells Transl. Med. 2017, 6, 1316–1320. [Google Scholar] [CrossRef]
- Mund, J.A.; Estes, M.L.; Yoder, M.C.; Ingram, D.A.; Case, J. Flow Cytometric Identification and Functional Characterization of Immature and Mature Circulating Endothelial Cells. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1045–1053. [Google Scholar] [CrossRef]
- Estes, M.L.; Mund, J.A.; Ingram, D.A.; Case, J. Identification of Endothelial Cells and Progenitor Cell Subsets in Human Peripheral Blood. Curr. Protoc. Cytom. 2010, 52, 9–33. [Google Scholar] [CrossRef]
- Asahara, T.; Kawamoto, A.; Masuda, H. Concise Review: Circulating Endothelial Progenitor Cells for Vascular Medicine. Stem Cells 2011, 29, 1650–1655. [Google Scholar] [CrossRef]
- Smadja, D.M.; Bièche, I.; Silvestre, J.-S.; Germain, S.; Cornet, A.; Laurendeau, I.; Duong-Van-Huyen, J.-P.; Emmerich, J.; Vidaud, M.; Aiach, M.; et al. Bone Morphogenetic Proteins 2 and 4 Are Selectively Expressed by Late Outgrowth Endothelial Progenitor Cells and Promote Neoangiogenesis. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 2137–2143. [Google Scholar] [CrossRef]
- Kang, K.-T.; Lin, R.-Z.; Kuppermann, D.; Melero-Martin, J.M.; Bischoff, J. Endothelial Colony Forming Cells and Mesenchymal Progenitor Cells Form Blood Vessels and Increase Blood Flow in Ischemic Muscle. Sci. Rep. 2017, 7, 770. [Google Scholar] [CrossRef]
- Liao, G.; Zheng, K.; Shorr, R.; Allan, D.S. Human Endothelial Colony-Forming Cells in Regenerative Therapy: A Systematic Review of Controlled Preclinical Animal Studies. Stem Cells Transl. Med. 2020, 9, 1344–1352. [Google Scholar] [CrossRef]
- Schwarz, T.M.; Leicht, S.F.; Radic, T.; Rodriguez-Arabaolaza, I.; Hermann, P.C.; Berger, F.; Saif, J.; Böcker, W.; Ellwart, J.W.; Aicher, A.; et al. Vascular Incorporation of Endothelial Colony-Forming Cells Is Essential for Functional Recovery of Murine Ischemic Tissue Following Cell Therapy. Arterioscler. Thromb. Vasc. Biol. 2012, 32, e13–e21. [Google Scholar] [CrossRef]
- Bouvard, C.; Gafsou, B.; Dizier, B.; Galy-Fauroux, I.; Lokajczyk, A.; Boisson-Vidal, C.; Fischer, A.-M.; Helley, D. A6-Integrin Subunit Plays a Major Role in the Proangiogenic Properties of Endothelial Progenitor Cells. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 1569–1575. [Google Scholar] [CrossRef]
- Lee, S.H.; Lee, J.H.; Asahara, T.; Kim, Y.S.; Jeong, H.C.; Ahn, Y.; Jung, J.S.; Kwon, S.-M. Genistein Promotes Endothelial Colony-Forming Cell (ECFC) Bioactivities and Cardiac Regeneration in Myocardial Infarction. PLoS ONE 2014, 9, e96155. [Google Scholar] [CrossRef]
- Saif, J.; Schwarz, T.M.; Chau, D.Y.; Henstock, J.; Sami, P.; Leicht, S.F.; Hermann, P.C.; Alcala, S.; Mulero, F.; Shakesheff, K.M.; et al. Combination of Injectable Multiple Growth Factor-Releasing Scaffolds and Cell Therapy as an Advanced Modality to Enhance Tissue Neovascularization. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 1897–1904. [Google Scholar] [CrossRef]
- Palii, C.G.; Vulesevic, B.; Fraineau, S.; Pranckeviciene, E.; Griffith, A.J.; Chu, A.; Faralli, H.; Li, Y.; McNeill, B.; Sun, J.; et al. Trichostatin A Enhances Vascular Repair by Injected Human Endothelial Progenitors through Increasing the Expression of TAL1-Dependent Genes. Cell Stem Cell 2014, 14, 644–657. [Google Scholar] [CrossRef]
- Keighron, C.; Lyons, C.J.; Creane, M.; O’Brien, T.; Liew, A. Recent Advances in Endothelial Progenitor Cells toward Their Use in Clinical Translation. Front. Med. 2018, 5, 354. [Google Scholar] [CrossRef]
- Jarajapu, Y.P.; Hazra, S.; Segal, M.; LiCalzi, S.; Jhadao, C.; Qian, K.; Mitter, S.K.; Raizada, M.K.; Boulton, M.E.; Grant, M.B. Vasoreparative Dysfunction of CD34+ Cells in Diabetic Individuals Involves Hypoxic Desensitization and Impaired Autocrine/Paracrine Mechanisms. PLoS ONE 2014, 9, e93965. [Google Scholar]
- Simoncini, S.; Toupance, S.; Labat, C.; Gautier, S.; Dumoulin, C.; Arnaud, L.; Stathopoulou, M.G.; Visvikis-Siest, S.; Rossi, P.M.; Benetos, A.; et al. Functional Impairment of Endothelial Colony Forming Cells (ECFC) in Patients with Severe Atherosclerotic Cardiovascular Disease (ASCVD). Int. J. Mol. Sci. 2022, 23, 8969. [Google Scholar] [CrossRef]
- Chan, K.H.; Simpson, P.J.L.; Yong, A.S.; Dunn, L.L.; Chawantanpipat, C.; Hsu, C.; Yu, Y.; Keech, A.C.; Celermajer, D.S.; Ng, M.K.C. The Relationship between Endothelial Progenitor Cell Populations and Epicardial and Microvascular Coronary Disease—A Cellular, Angiographic and Physiologic Study. PLoS ONE 2014, 9, e93980. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-W.; Lo, H.-H.; Chiu, Y.-L.; Chang, S.-J.; Huang, P.-H.; Liao, K.-H.; Tasi, C.-F.; Wu, C.-H.; Tsai, T.-N.; Cheng, C.-C.; et al. Dysregulated MiR-361-5p/VEGF Axis in the Plasma and Endothelial Progenitor Cells of Patients with Coronary Artery Disease. PLoS ONE 2014, 9, e98070. [Google Scholar] [CrossRef] [PubMed]
- Bone Marrow Origin of Endothelial Progenitor Cells Responsible for Postnatal Vasculogenesis in Physiological and Pathological Neovascularization|Circulation Research. Available online: https://www.ahajournals.org/doi/10.1161/01.res.85.3.221?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed (accessed on 5 February 2023).
- Armulik, A.; Genové, G.; Betsholtz, C. Pericytes: Developmental, Physiological, and Pathological Perspectives, Problems, and Promises. Dev. Cell 2011, 21, 193–215. [Google Scholar] [CrossRef] [PubMed]
- Crisan, M.; Yap, S.; Casteilla, L.; Chen, C.-W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; et al. A Perivascular Origin for Mesenchymal Stem Cells in Multiple Human Organs. Cell Stem Cell 2008, 3, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Armulik, A.; Abramsson, A.; Betsholtz, C. Endothelial/Pericyte Interactions. Circ. Res. 2005, 97, 512–523. [Google Scholar] [CrossRef]
- Souidi, N.; Stolk, M.; Rudeck, J.; Strunk, D.; Schallmoser, K.; Volk, H.-D.; Seifert, M. Stromal Cells Act as Guardians for Endothelial Progenitors by Reducing Their Immunogenicity After Co-Transplantation. Stem Cells 2017, 35, 1233–1245. [Google Scholar] [CrossRef]
- Allen, P.; Kang, K.-T.; Bischoff, J. Rapid Onset of Perfused Blood Vessels after Implantation of ECFCs and MPCs in Collagen, PuraMatrix and Fibrin Provisional Matrices. J. Tissue Eng. Regen. Med. 2015, 9, 632–636. [Google Scholar] [CrossRef]
- Shafiee, A.; Patel, J.; Yi Wong, H.; Donovan, P.; Hutmacher, D.W.; Fisk, N.M.; Khosrotehrani, K. Priming of Endothelial Colony-forming Cells in a Mesenchymal Niche Improves Engraftment and Vasculogenic Potential by Initiating Mesenchymal Transition Orchestrated by NOTCH Signaling. FASEB J. 2017, 31, 610–624. [Google Scholar] [CrossRef]
- Tepper, O.M.; Galiano, R.D.; Capla, J.M.; Kalka, C.; Gagne, P.J.; Jacobowitz, G.R.; Levine, J.P.; Gurtner, G.C. Human Endothelial Progenitor Cells from Type II Diabetics Exhibit Impaired Proliferation, Adhesion, and Incorporation into Vascular Structures. Circulation 2002, 106, 2781–2786. [Google Scholar] [CrossRef]
- Tran, P.M.; Tang, S.S.; Salgado-Pabón, W. Staphylococcus Aureus β-Toxin Exerts Anti-Angiogenic Effects by Inhibiting Re-Endothelialization and Neovessel Formation. Front. Microbiol. 2022, 13, 840236. [Google Scholar] [CrossRef]
- Popescu, S.; Preda, M.B.; Marinescu, C.I.; Simionescu, M.; Burlacu, A. Dual Stem Cell Therapy Improves the Myocardial Recovery Post-Infarction through Reciprocal Modulation of Cell Functions. Int. J. Mol. Sci. 2021, 22, 5631. [Google Scholar] [CrossRef]
- Pinho, A.G.; Cibrão, J.R.; Silva, N.A.; Monteiro, S.; Salgado, A.J. Cell Secretome: Basic Insights and Therapeutic Opportunities for CNS Disorders. Pharmaceuticals 2020, 13, 31. [Google Scholar] [CrossRef]
- Antimisiaris, S.G.; Mourtas, S.; Marazioti, A. Exosomes and Exosome-Inspired Vesicles for Targeted Drug Delivery. Pharmaceutics 2018, 10, 218. [Google Scholar] [CrossRef]
- Rascher-Eggstein, G.; Liebner, S.; Wolburg, H. The Blood–Brain Barrier in the Human Glioma. In Blood-Spinal Cord and Brain Barriers in Health and Disease; Elsevier: Amsterdam, The Netherlands, 2004; pp. 561–576. ISBN 978-0-12-639011-7. [Google Scholar]
- Vascular Apoptosis and Involution in Gliomas Precede Neovascularization: A Novel Concept for Glioma Growth and Angiogenesis-Laboratory Investigation. Available online: https://li.uscap.org/article/S0023-6837(22)02147-X/fulltext (accessed on 22 February 2023).
- Holash, J.; Maisonpierre, P.C.; Compton, D.; Boland, P.; Alexander, C.R.; Zagzag, D.; Yancopoulos, G.D.; Wiegand, S.J. Vessel Cooption, Regression, and Growth in Tumors Mediated by Angiopoietins and VEGF. Science 1999, 284, 1994–1998. [Google Scholar] [CrossRef]
- Endothelin: Function and Purpose. Available online: https://my.clevelandclinic.org/health/articles/23139-endothelin (accessed on 22 February 2023).
- Kim, Y.-M.; Hwang, S.; Kim, Y.-M.; Pyun, B.-J.; Kim, T.-Y.; Lee, S.-T.; Gho, Y.S.; Kwon, Y.-G. Endostatin Blocks Vascular Endothelial Growth Factor-Mediated Signaling via Direct Interaction with KDR/Flk-1. J. Biol. Chem. 2002, 277, 27872–27879. [Google Scholar] [CrossRef]
- Lawler, P.R.; Lawler, J. Molecular Basis for the Regulation of Angiogenesis by Thrombospondin-1 and -2. Cold Spring Harb. Perspect. Med. 2012, 2, a006627. [Google Scholar] [CrossRef]
- Miller, D.W.; Graulich, W.; Karges, B.; Stahl, S.; Ernst, M.; Ramaswamy, A.; Sedlacek, H.H.; Müller, R.; Adamkiewicz, J. Elevated Expression of Endoglin, a Component of the TGF-Beta-Receptor Complex, Correlates with Proliferation of Tumor Endothelial Cells. Int. J. Cancer 1999, 81, 568–572. [Google Scholar] [CrossRef]
- Kopczyńska, E.; Makarewicz, R. Endoglin—A Marker of Vascular Endothelial Cell Proliferation in Cancer. Contemp. Oncol. 2012, 16, 68–71. [Google Scholar] [CrossRef]
- Dutta, S.; Bandyopadhyay, C.; Bottero, V.; Veettil, M.V.; Wilson, L.; Pins, M.R.; Johnson, K.E.; Warshall, C.; Chandran, B. Angiogenin Interacts with the Plasminogen Activation System at the Cell Surface of Breast Cancer Cells to Regulate Plasmin Formation and Cell Migration. Mol. Oncol. 2014, 8, 483–507. [Google Scholar] [CrossRef]
- Domínguez-Vivero, C.; Leira, Y.; López-Ferreiro, A.; Saavedra, M.; Rodríguez-Osorio, X.; Sobrino, T.; Campos, F.; Castillo, J.; Leira, R. Pentraxin 3 (PTX3): A Molecular Marker of Endothelial Dysfunction in Chronic Migraine. J. Clin. Med. 2020, 9, 849. [Google Scholar] [CrossRef]
- Zlibut, A.; Bocsan, I.C.; Agoston-Coldea, L. Chapter Five-Pentraxin-3 and Endothelial Dysfunction. In Advances in Clinical Chemistry; Makowski, G.S., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 91, pp. 163–179. [Google Scholar]








Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bansal, A.; Singh, A.; Nag, T.C.; Sharma, D.; Garg, B.; Bhatla, N.; Choudhury, S.D.; Ramakrishnan, L. Augmenting the Angiogenic Profile and Functionality of Cord Blood Endothelial Colony-Forming Cells by Indirect Priming with Bone-Marrow-Derived Mesenchymal Stromal Cells. Biomedicines 2023, 11, 1372. https://doi.org/10.3390/biomedicines11051372
Bansal A, Singh A, Nag TC, Sharma D, Garg B, Bhatla N, Choudhury SD, Ramakrishnan L. Augmenting the Angiogenic Profile and Functionality of Cord Blood Endothelial Colony-Forming Cells by Indirect Priming with Bone-Marrow-Derived Mesenchymal Stromal Cells. Biomedicines. 2023; 11(5):1372. https://doi.org/10.3390/biomedicines11051372
Chicago/Turabian StyleBansal, Ashutosh, Archna Singh, Tapas Chandra Nag, Devyani Sharma, Bhavuk Garg, Neerja Bhatla, Saumitra Dey Choudhury, and Lakshmy Ramakrishnan. 2023. "Augmenting the Angiogenic Profile and Functionality of Cord Blood Endothelial Colony-Forming Cells by Indirect Priming with Bone-Marrow-Derived Mesenchymal Stromal Cells" Biomedicines 11, no. 5: 1372. https://doi.org/10.3390/biomedicines11051372
APA StyleBansal, A., Singh, A., Nag, T. C., Sharma, D., Garg, B., Bhatla, N., Choudhury, S. D., & Ramakrishnan, L. (2023). Augmenting the Angiogenic Profile and Functionality of Cord Blood Endothelial Colony-Forming Cells by Indirect Priming with Bone-Marrow-Derived Mesenchymal Stromal Cells. Biomedicines, 11(5), 1372. https://doi.org/10.3390/biomedicines11051372




