Abstract
To elucidate the currently unknown molecular mechanisms responsible for the similarity and difference during the acquirement of resistance against gemcitabine (GEM) and paclitaxel (PTX) in patients with pancreatic carcinoma, we examined two-dimensional (2D) and three-dimensional (3D) cultures of parent MIA PaCa-2 cells (MIA PaCa-2-PA) and their GEM resistance cell line (MIA PaCa-2-GR) and PTX resistance (MIA PaCa-2-PR). Using these cells, we examined 3D spheroid configurations and cellular metabolism, including mitochondrial and glycolytic functions, with a Seahorse bio-analyzer and RNA sequencing analysis. Compared to the MIA PaCa-2-PA, (1) the formation of the 3D spheroids of MIA PaCa-2-GR or -PR was much slower, and (2) their mitochondrial and glycolytic functions were greatly modulated in MIA PaCa-2-GR or -PR, and such metabolic changes were also different between their 2D and 3D culture conditions. RNA sequencing and bioinformatic analyses of the differentially expressed genes (DEGs) using an ingenuity pathway analysis (IPA) suggested that various modulatory factors related to epithelial –mesenchymal transition (EMT) including STAT3, GLI1, ZNF367, NKX3-2, ZIC2, IFIT2, HEY1 and FBLX, may be the possible upstream regulators and/or causal network master regulators responsible for the acquirement of drug resistance in MIA PaCa-2-GR and -PR. In addition, among the prominently altered DEGs (Log2 fold changes more than 6 or less than −6), FABP5, IQSEC3, and GASK1B were identified as unique genes associated with their antisense RNA or pseudogenes, and among these, FABP5 and GASK1B are known to function as modulators of cancerous EMT. Therefore, the observations reported herein suggest that modulations of cancerous EMT may be key molecular mechanisms that are responsible for inducing chemoresistance against GEM or PTX in MIA PaCa-2 cells.
1. Introduction
It is known that pancreatic ductal adenocarcinoma (PDAC) is a malignant tumor that has a poor prognosis, and in fact, even after undergoing potentially curative surgery, their 5-year survival rate is only approximately 15–25% [1,2,3]. In terms of the clinical treatment for most patients with PDAC, systemic chemotherapy is conducted regardless of the surgical treatment option. In the past decade, based upon evidence obtained from various clinical trials [4,5,6,7,8], a combination of gemcitabine (GEM) and an albumin nanoparticle conjugate of paclitaxel (nab-paclitaxel, n-PTX) [6,9] have emerged as a first-line therapy in patients with advanced PDAC. However, the response to this chemotherapy regimen is still poor because of the rapid acquirement of drug resistance in most patients [10]. Therefore, identifying the underlying molecular mechanisms responsible for causing such chemoresistance to be acquired, as well as additional candidate targets and compounds that can overcome these factors, are urgently required. However, for this purpose, the various in vitro drug screening methods using conventional two-dimensional (2D) planar cultures of cancer cell lines have been used, but using these cultures, it has not been successfully identified possible candidate drugs and compounds to translate into clinical applications [11,12,13,14]. In addition, recent studies have suggested that the molecular mechanisms responsible for inducing this chemoresistance are much more complicated because of the numerous genetic changes that are related to various cellular signaling pathways and responses of PDAC cells [15], in addition to drug transport [16] and the tumor microenvironment [17]. Therefore, developing a better understanding of those underlying mechanisms would make it possible to identify promising therapeutic strategies for overcoming this chemoresistance. To accomplish this, it will be necessary to develop more suitable in vitro models that replicate the biological characteristics of the PDAC tumor environment.
Three-dimensional (3D) cultures were developed in order to replicate in vitro tumor models more closely [18,19]. Among the various types of in vitro 3D cell culture models, an in vitro 3D spheroid model is the simplest and, thus, has been the most frequently used in studies related to not only cancerous but also non-cancerous related research fields [20,21]. In fact, 3D spheroid cultures of various PDAC cell lines are now recognized as a better in vitro model to mimic the tumor microenvironment in investigations related to tumor pathophysiology and chemoresistance, as well as in drug screening [22,23,24]. Our group recently independently developed various in vitro 3D spheroid models using various non-cancerous ocular-related cells [25,26,27,28] and rat cardiomyocytes, H9c2 cells [29], as well as cancerous cell lines including an A549 lung adenocarcinoma cell line [30], malignant melanoma cell lines [31] and oral squamous cell carcinoma (OSCC) cells [32]. These collective studies allowed us to conclude that the biological natures were significantly different between the 2D planar cultures and 3D spheroid cultures, even though we employed exactly the same experimental conditions except that different culture plates were used. Interestingly, we also found that the appearance of the 3D spheroids was also different between non-cancerous and cancerous cells, in that they were globe-shape [26,27,28] or non-globe shape [30,31,32], respectively. Furthermore, we also recognized that the appearances of the 3D spheroids were significantly diverse among malignant tumors even though they had the same origins, and the degree of difference was potentially correlated with cellular metabolic functions, pathological aspects, and/or cytotoxicity against anti-tumor drugs [30,31,32]. Considering these collective findings, we concluded that characteristic appearances of the cancerous 3D spheroid could be a potential indicator for evaluating the clinicopathological aspects of certain malignant tumors.
Therefore, in the current study, to elucidate the currently unidentified underlying molecular mechanisms responsible for the chemoresistance of PDAC against GEM or PTX, using a well-characterized PDCA cell line, MIA PaCa-2 as a parent cell line (MIA PaCa-2-PA), their corresponding chemoresistant cell lines against GEM (MIA PaCa-2-GR) or PTX (MIA PaCa-2-PR) were prepared. Thereafter, those were further cultured by 2D planar and 3D spheroid culture methods, and the resulting cultures were then subjected to a Seahorse real-time cellular metabolic analysis. In addition, three 2D cultured cell lines were also subjected to RNA sequencing analysis in an attempt to elucidate possible critical genes responsible for developing chemoresistance against GEM or PTX in PDAC.
2. Materials and Methods
The current study, which was conducted at the Sapporo Medical University Hospital, Japan, was approved by the institutional review board (IRB, registration number 342-3416) according to the tenets of the Declaration of Helsinki and national laws for using human-related carcinoma cell lines.
2.1. Preparations of Gemcitabine or Paclitaxel Resistance MIA PaCa-2 Cells
A pancreatic ductal carcinoma cell line, MIA PaCa-2 cells, was obtained from the American Type Culture Collection (Manassas, VA, USA). MIA PaCa-2 cells were cultured in 2D culture dishes at 37 °C in a 2D culture medium composed of HG-DMEM culture medium supplemented with 8 mg/L d-biotin, 4 mg/L calcium pantothenate, 100 U/mL penicillin, 100 μg/mL streptomycin (b.p. HG-DMEM), 10% CS were used as the parental line (MIA PaCa-2-PA). For generating drug resistance in MIA-PaCa-2 against GEM (MIA PaCa-2-GR) or PTX (MIA PaCa-2-PR), subcultures of the MIA PaCa-2-PA cells were exposed to incremental increases in GEM or PTX concentrations, starting with an IC50 dose (GEM: 26 nM, PTX: 232 nM) for six months. Finally, the MIA PaCa-2-GR or MIA PaCa-2-PR cells developed the capacity for proliferation when returned to a medium containing 2.6 µM GEM or 5.0 µM PTX, respectively.
2.2. 3D Cell Cultures of MIA PaCa-2-P, -GR or -PR Cells
MIA PaCa-2-PA, -GR, or -PR cells, as generated above, were 3D cells cultured by methods described in previous reports using 3T3-L1 preadipocytes and human orbital fibroblasts [26,33,34,35,36,37]. Briefly, these cells were each cultured in 2D culture dishes at 37 °C in a 2D culture medium containing 0.25% w/v Methocel A4M in the absence or presence of 2.6 μM GEM or 0.5 μM PTX until reaching approximately 90% confluence. After washing with a phosphate-buffered saline (PBS), the cells were detached by treatment with 0.25% Trypsin/EDTA, resuspended in the culture medium, and 28 μL of medium containing approximately 20,000 cells were added to each well of the drop culture plate (# HDP1385, Sigma-Aldrich, Burlington, MA, USA) (3D/Day 0) as described previously [26,34]. Thereafter, half of the culture medium was replaced with fresh medium in each well daily until Day 5 [35,36,37]. As a representative non-cancerous human cell line, human trabecular meshwork (HTM) cells (Applied Biological Materials Inc., Richmond, BC, Canada) [38] were also used. The 3D spheroid morphology was observed by a phase contrast microscope (Nikon ECLIPSE TS2; Tokyo, Japan), as described previously [35,36,37].
2.3. Real-Time Analysis of the Cellular Metabolic Functions
Oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) in 2D and 3D cultured MIA PaCa-2-P, -GR, or -PR cells were measured using a Seahorse XFe96 real-time metabolic analyzer (Agilent Technologies, Santa Clara, CA, USA). On the day of assay, an XFe96 Cell Culture Microplate (Agilent Technologies, #103794-100) was coated with Cell-Tak™ (Corning #354240, Corning, NY, USA). In brief, 200 μL of 2 mg/mL Cell-Tak in 5% acetic acid was added in 2.8 mL of 0.1 M sodium bicarbonate, and then 30 μL of this Cell-Tak Mix was placed in each well of a microplate and incubated for 1 h in a non-CO2 incubator at 37 °C. Following the incubation, Cell-Tak Mix was aspirated from the plate, and the plate was washed twice with 400 µL of sterile 37 °C water and allowed to air dry. Approximately 10,000 2D-cultured cells and six 3D-cultured spheroids were resuspended to a pre-warmed 50 μL Seahorse XF DMEM assay medium (pH 7.4, Agilent Technologies, #103575-100) containing 5.5 mM glucose, 2.0 mM glutamine, and 1.0 mM sodium pyruvate and were seeded onto each well in the pre-made Cell-Tak coated Seahorse assay plate. The plate was incubated in a CO2-free incubator at 37 °C for 1 h prior to the measurements.
OCR and ECAR were measured in an XFe96 extracellular flux analyzer at the baseline and after the following sequential injections of 2.0 μM oligomycin, 5.0 μM
carbonyl cyanide-p-trifluoromethoxyphenylhydrazone (FCCP), a mixture of 1.0 μM rotenone, 1.0 μM antimycin A, and 10 mM 2-deoxyglucose (2DG). The OCR and ECAR values were normalized for the number of protein contents assessed by a BCA protein assay (TaKaRa, Otsu, Japan) per well by lysing the cells of the wells in which the measurements were completed with 10 μL of CelLytic™ MT Cell Lysis Reagent (Sigma-Aldrich). Key parameters of mitochondrial and glycolytic functions were calculated as follows: Basal respiration = OCR at baseline − OCR after adding R/A; ATP-linked respiration = OCR at baseline − OCR after adding oligomycin; Maximal respiration = OCR after adding FCCP − OCR after adding R/A; Glycolytic capacity = ECAR after adding oligomycin − ECAR at baseline; Glycolytic reserve = ECAR after adding oligomycin − ECAR after adding 2DG.
2.4. RNA Sequencing, Gene Function, and Analysis of Pathways
Total RNA was isolated from 2D confluent cells of MIA PaCa-2-PA, -GR, or -PR in a 150 mm dish as described above (n = 3) using an RNeasy mini kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions and were then subjected to an RNA sequencing analysis as described recently [30]. Briefly, after the RNA content and quality were checked to make sure that the RNA quality was suitable for RNA sequencing, ribosomal RNA was removed from total RNA using NEBNext® Poly(A) mRNA Magnetic Isolation Module (Cat. # E7490, New England BioLabs, Ipswich, MA, USA). The rRNA-depleted RNA was then processed to convert to cDNA using a TruSeq RNA Sample Preparation Kit (Illumina, San Diego, CA, USA) and final sequence-ready libraries with the NEBNext Ultra II RNA library prep kit (Cat. #E7760, New England BioLabs). After checking their quality and quantity using an Agilent 2100 Bioanalyzer and KAPA Library Quantification Kit (KAPA Biosystems, Wilmington, MA, USA), respectively, they were subjected to NovaSeq 6000 and GenoLab M sequencing in the PE150 mode. Sequence data were filtered by removing the adapter sequence, ambiguous nucleotides, and low-quality sequences using software (FastQC, version 0.11.7) as quality control by an Agilent 2100 Bioanalyzer (Agilent, CA, USA) and Trimmomatic (version 0.38) were mapped to the reference genome sequence (GRCh38) using HISAT2 tools software [39]. The read counts for each respective gene and statistical analysis were analyzed by featureCounts (version 1.6.3) and DESeq2 (version 1.24.0), respectively. Statistical significance was determined by an empirical analysis, and genes with fold-change ≥ 2.0 and FDR-adjusted p-value < 0.05 and q < 0.08 were assigned as differentially expressed genes (DEG).
To predict possible upstream transcriptional regulators, DEGs were interpreted using the upstream regulator and causal network regulator functions of the ingenuity pathway analysis (IPA, Qiagen, accessed on 27 July 2023. https://digitalinsights.qiagen.com/products-overview/discovery-insights-portfolio/analysis-and-visualization/qiagen-ipa/) [40].
2.5. Other Analytical Methods
For drug sensitivity measurements, cells were seeded in 96-well plates at a density of 3 × 103 cells/well and allowed to attach for 24 h, after which they were cultured for 72 hrs with 0–100 μM GEM or PTX. Cell viability was evaluated by a WST-1 assay (Premix WST-1 Cell Proliferation Assay; Takara Bio, Otsu, Japan) and Infinite M1000 PRO microplate reader (Tecan Japan, Kawasaki, Japan). Absorbance was measured at 450 nm to determine cell viability.
Quantitative PCR using specific primers (Table S1) was conducted with Quant Studio 3 (Applied Biosystems, Foster City, CA, USA). RNA was extracted using TRIzol Reagent (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s protocol, and 1 µg of total RNA was reverse transcribed with a SuperScript VILO cDNA synthesis kit (Thermo Fisher Scientific). The analysis was conducted in quadruplicate using a POWER UP SYBR Green Master Mix (Thermo Fisher Scientific). Transcript levels were normalized to β-actin expression.
Statistical analyses using the Graph Pad Prism 8 (GraphPad Software, San Diego, CA, USA) were performed as demonstrated in a previous report [35]. All statistical analyses were performed using the Graph Pad Prism 8 (GraphPad Software, San Diego, CA). The statistical difference between groups was determined using a Students’ t-test for two-group comparison, or two-ANOVA followed by a Tukey’s multiple comparison test. Data are expressed as the arithmetic mean ± the standard error of the mean (SEM).
3. Results
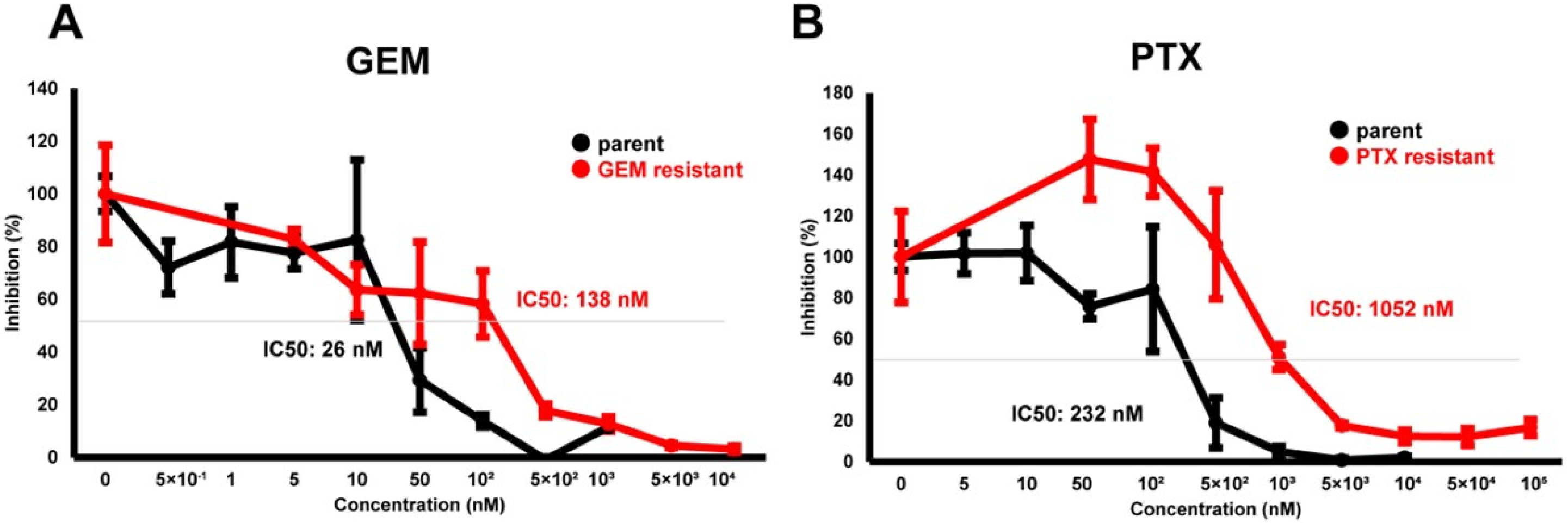
To elucidate biological similarities and differences upon acquiring chemoresistance of PDCA against various anti-cancer drugs, a well-characterized cell line, MIA PaCa-2, was used. Initially, using MIA PaCa-2-PA and standard first-line anti-tumor drugs, GEM or PTX, MIA PaCa-2-GR, and -PR were prepared. As shown in Figure 1, chemoresistance against GEM or PTX were apparently obtained in the MIA PaCa-2-GR (GEM IC 50 = 138 nM) or MIA PaCa-2-PR (PTX IC50 = 1052 nM), while this was not the case for MIA PaCa-2-PA (GEM IC50 = 26 nM, PTX IC50 = 232 nM), respectively.
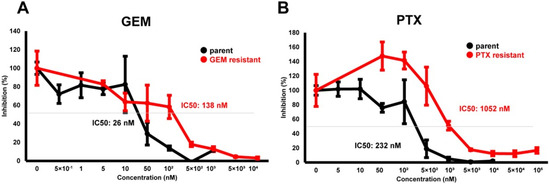
Figure 1.
Cytotoxic analysis of chemoresistant MIA PaCa-2 cells. To determine the cytotoxicity against GEM (A) or PTX (B) in MIA PaCa-2-PA, MIA PaCa-2-GR, and MIA PaCa-2-PR, survival living cells detected using a WST-1 assay were plotted (n = 3).
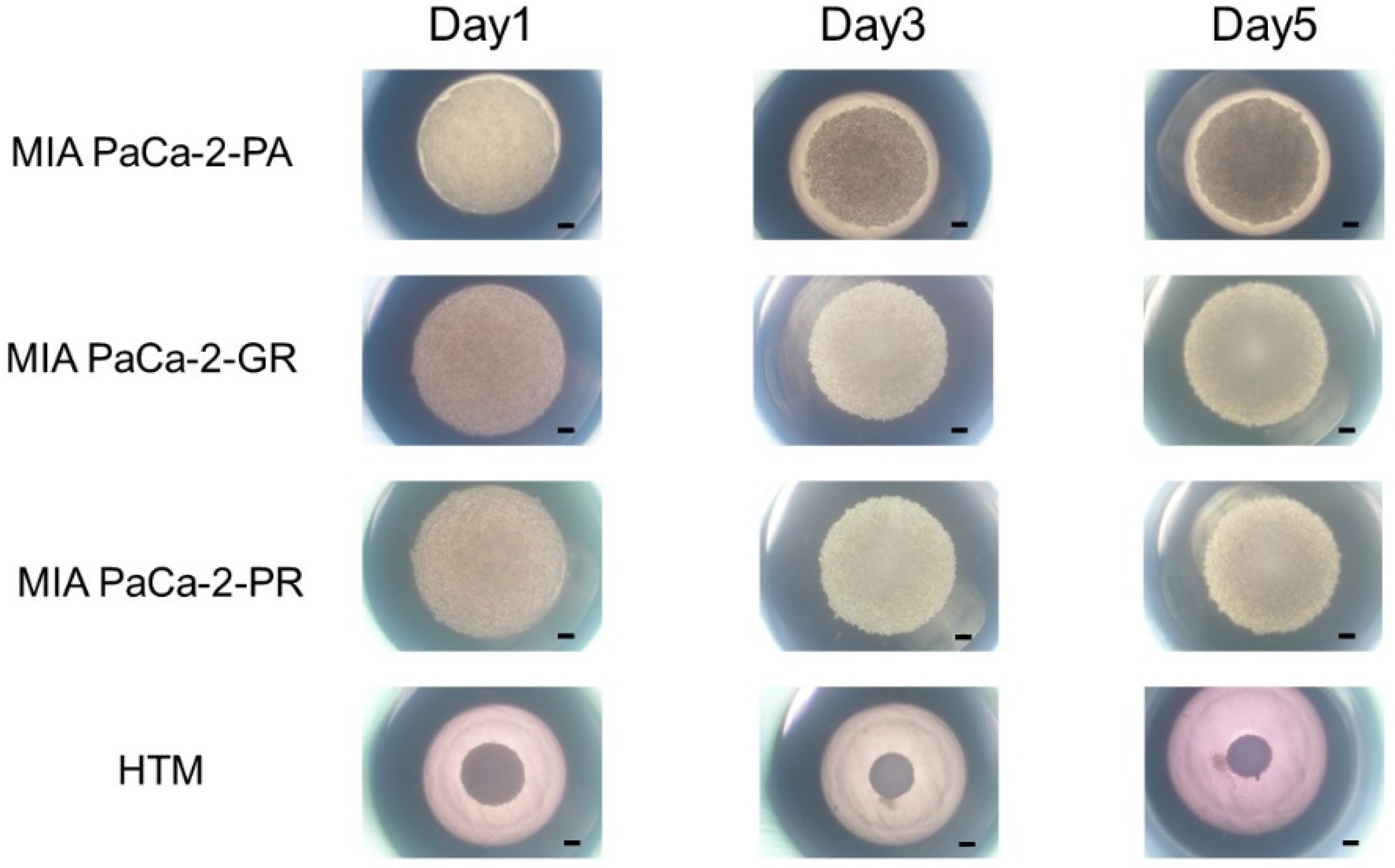
In our recent study, we reported that the 3D spheroid configurations were significantly diverse among malignant tumor cells even though they had the same origin, and these biological diversities were confirmed by Seahorse cellular metabolic measurements [31]. Therefore, we concluded that 3D spheroid cultures of malignant tumors might be quite useful for evaluating diverse biological aspects among various malignant tumors. In the current investigation, using this methodology, unidentified biological similarities and differences among three cell lines, MIA PaCa-2-PA, -GR, and -PR, were studied. As shown in Figure 2, the cells that were placed into each well of the 3D drop culture all began to coalesce after one day, as was typically observed in the 3D spheroid cultures of none cancerous and cancerous cells [26,27,28,30,31,41], but the sizes were apparently larger in MIA PaCa-2 cell lines. However, unexpectedly, the progression for forming 3D spheroids in the MIA PaCa-2-PA was extremely slow and did not progress to the formation of a solid 3D spheroid as is typically recognized within the non-cancerous and cancerous cells [26,27,28,30,31,41] after 5 days of culture. Such slower 3D spheroid formation was more evident in the chemoresistant cell lines, MIA PaCa-2-GR, and -PR.
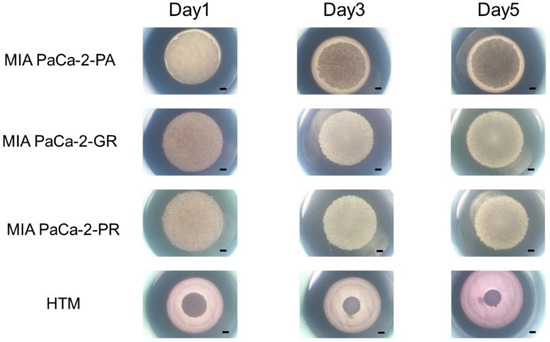
Figure 2.
3D spheroid cultures of MIA PaCA-2-PA and -GR or -PR. Approximately 20,000 cells of MIA PaCa-2-PA, MIA PaCa-2-GR, or MIA PaCa-2-PR were subjected to a 3D drop cell culture to form 3D spheroids. As a representative non-cancerous human cell line, 20,000 cells of human trabecular meshwork (HTM) were also subjected to the 3D spheroid culture. Representative phase contrast microscopy images of these cells on Day 1, 3, and 5. Scale Bar: 100 μm.
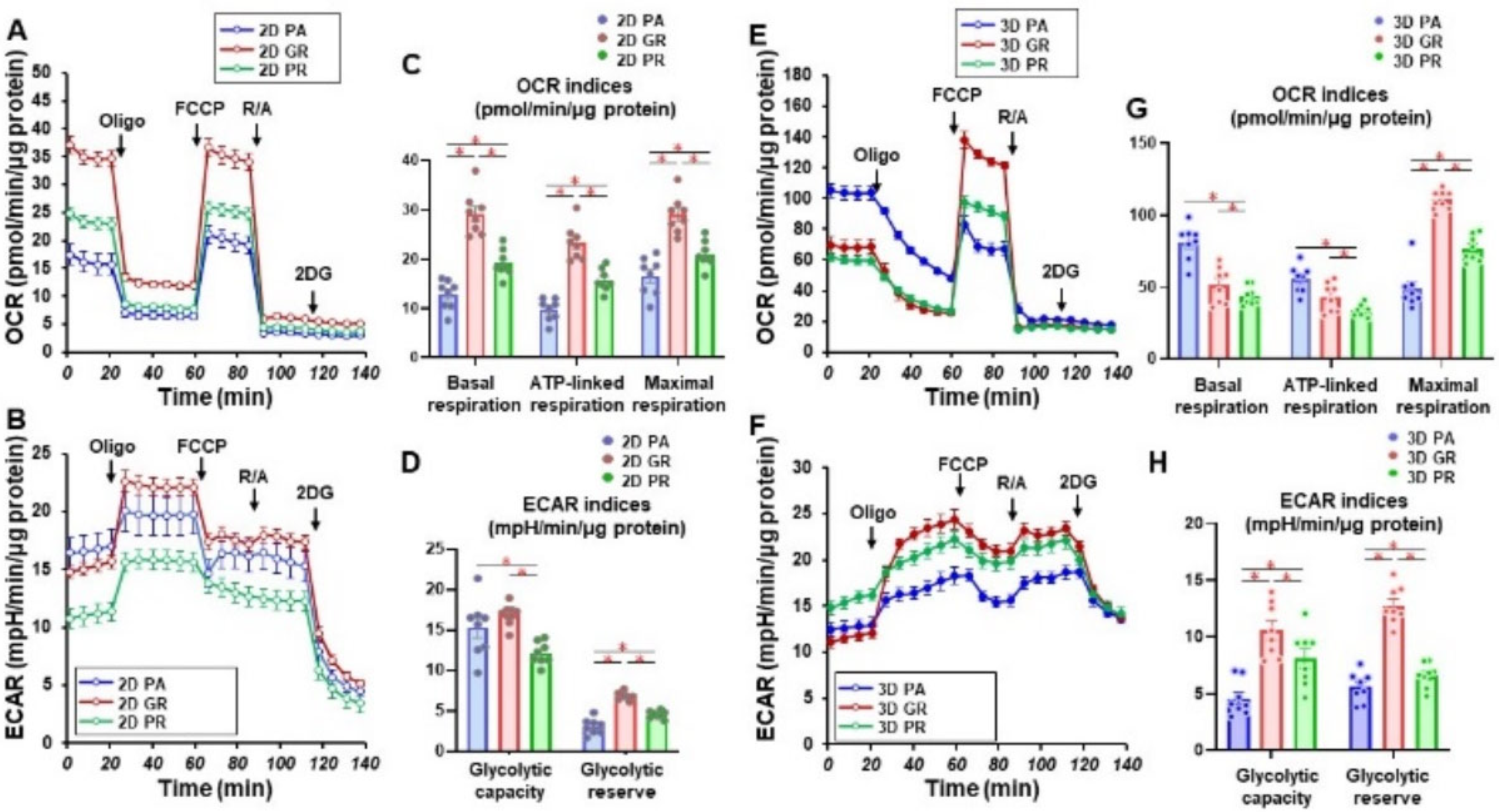
We then studied the cellular metabolic characteristics of the 2D and 3D cultured three cell lines, MIA PaCa-2-PA, -GR, and -PR. As shown in Figure 3, as compared with MIA PaCa-2-PA, the mitochondrial (OCR) and glycolytic functions (ECAR) of MIA PaCa-2-GR and -PR were significantly modulated, and those changes were also different between 2D and 3D cultures. That is, (1) in the 2D culture, both OCR and ECAR indices were substantially increased in the order of MIA PaCa-2-PR and -GR, and (2) in the 3D cell cultures, ECAR indices were similarly modulated as the 2D cultured cells, but within the OCR indices, basal respiration and ATP-linked respiration were markedly decreased in the order of MIA PaCa-2-GR and -PR. Therefore, these collective observations indicate that (1) biological aspects are greatly modulated on acquiring chemoresistance, and these aspects were also different between GEM and PTX, and (2) even though solid 3D spheroids were not generated, significant alterations of the biological functions of MIA PaCa-2 related cells were induced in the 3D cultures, as compared with the corresponding 2D planar cell cultures.
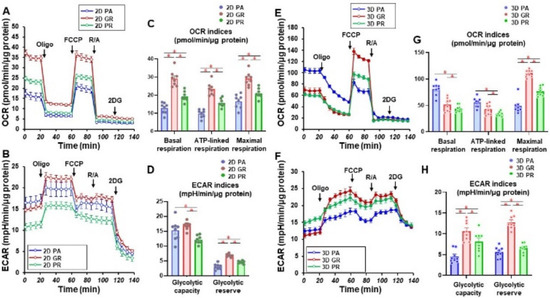
Figure 3.
Measurement of mitochondrial and glycolytic functions of MIA PaCa-2-PA and -GR or -PR. Real-time metabolic function analysis by an XFe96 Extracellular Flux Analyzer of the 2D- or 3D-cultured MIA PaCa-2 cells in fresh preparations (n = 8). (A) Measurement of OCR in 2D-cultured cells. (B) Measurement of ECAR in 2D-cultured cells. (C) Key parameters in mitochondrial function in 2D-cultured cells. (D) Key parameters in glycolytic function in 2D-cultured cells. (E) Measurement of OCR in 3D-cultured spheroids. (F) Measurement of ECAR in 3D-cultured spheroids. (G) Key parameters in the mitochondrial function in 3D-cultured spheroids. (H) Key parameters in glycolytic function in 3D-cultured spheroids. OCR, oxygen consumption rate; ECAR, extracellular acidification rate; Oligo, oligomycin; FCCP, carbonyl cyanide p-trifluoromethoxyphenylhydrazone; R/A, otenone/antimycin A; 2DG, 2-deoxyglucose. * p < 0.05 (one-way ANOVA followed by a Tukey’s muliple comparison test).
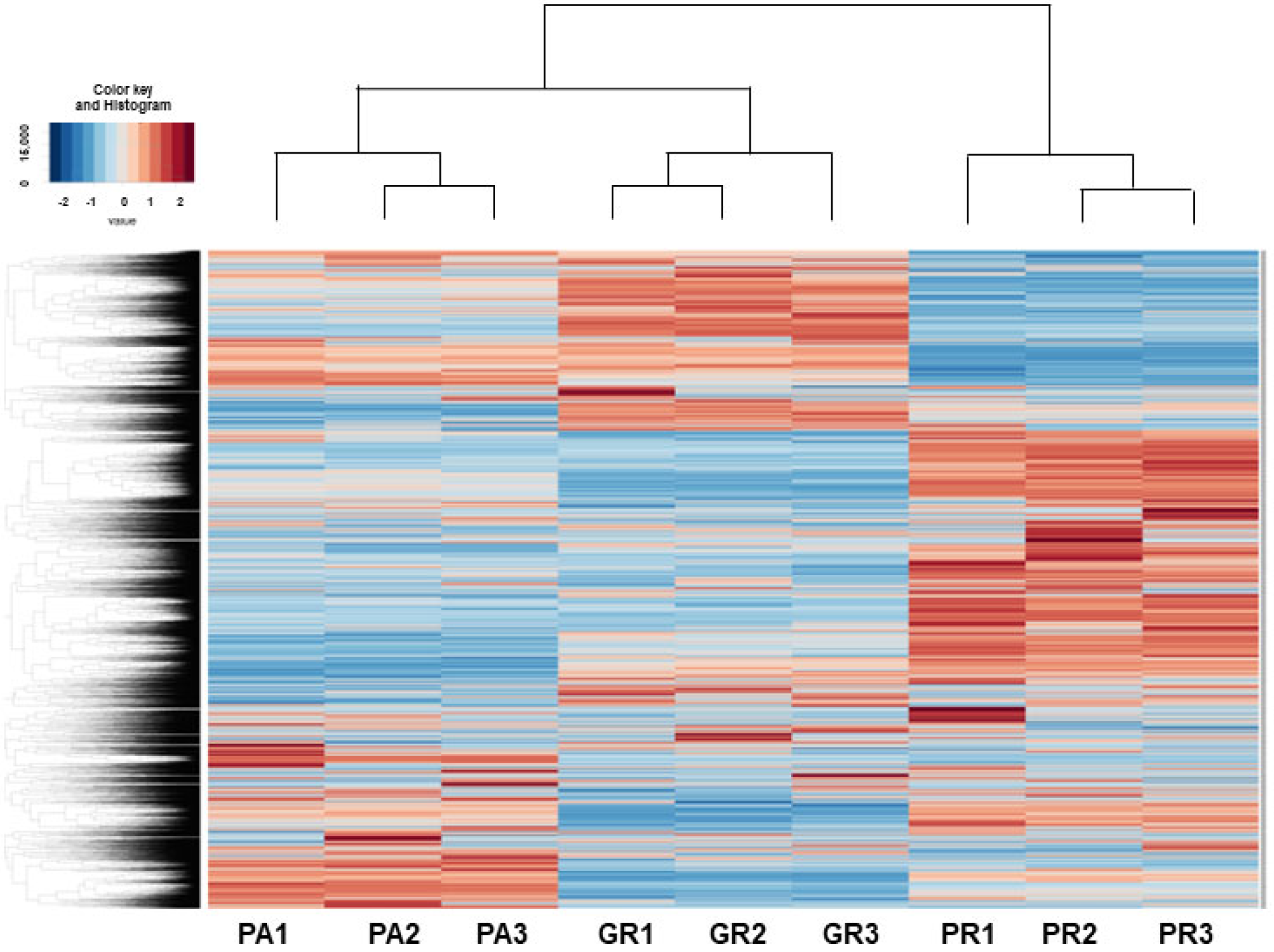
RNA sequencing analyses were performed in an attempt to elucidate the currently unidentified mechanisms responsible for inducing such characteristic biological alterations upon acquiring chemoresistance against GEM or PTX in the MIA PaCa-2 cells. As shown in the heatmap (Figure 4) and MA and volcano plots (Figures S1–S3), 578, 991, or 1319 significantly up-regulated and 890, 873 or 800 down-regulated differentially expressed genes (DEGs) were identified between MIA PaCa2-PA vs. -GR, MIA PaCa-2-PA vs. -PR or MIA PaCa2-GR vs. -PR, respectively, with a significance level of <0.05 (FDR) and an absolute fold-change ≥2 was identified (the list of all of the up-regulated and down-regulated DEGs is attached in a Supplemental excel file). Among these DEGs, the most prominently up-regulated or down-regulated DEGs (Log2 fold change of more than 6 or less than −6, respectively) were compared between MIA PaCa-2-PA vs. -GR and MIA PaCa-2-PA vs. -PR. As shown in Table 1 and Table 2, a total of three up-regulated DEGs (DACH1, VCAM1, and PACRG) and 11 down-regulated DEGs (TGFBR2, DOC2B, VCAN, MDFIC, LRP1B, GLIS3, MMP1, DSC2, ARHGDIB, ARGGAP15 and ZNF488) were commonly detected. We, therefore, speculated that these genes are most likely involved in the molecular mechanisms responsible for inducing chemoresistance of MIA PaCa-2 cells regardless of different anti-tumor drugs. Alternatively, a pair of FABP5 and FABP5P7, IQSEC3 and IQSEC3-AS1, and GASK1B and GASK1B-AS1 were identified only in the MIA PaCa-2-PA vs. -GR or MIA PaCa-2-PA vs. -PR, respectively. We speculate that these unique DEGs that were detected in one of both chemoresistant MIA PaCa-2 cells may be key regulatory factors for acquiring chemoresistance against GEM or PTX.
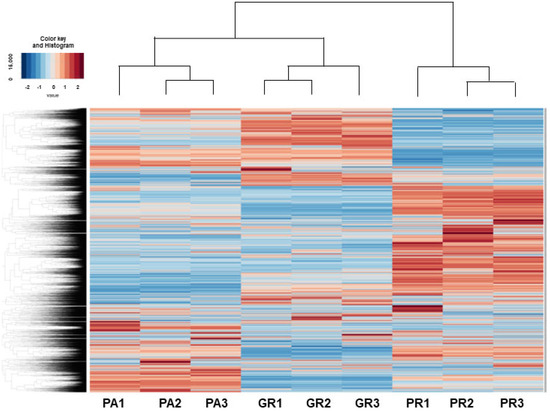
Figure 4.
Heatmap for DEGs between 2D cultured MIA PaCA-2-PA and -2-GR or -PR.

Table 1.
Significant up-regulated DEGs upon chemoresistance against GEM or PTX (Log2Fold change more than 6).

Table 2.
Significant down-regulated DEGs upon chemoresistance against GEM or PTX (Log2Fold).
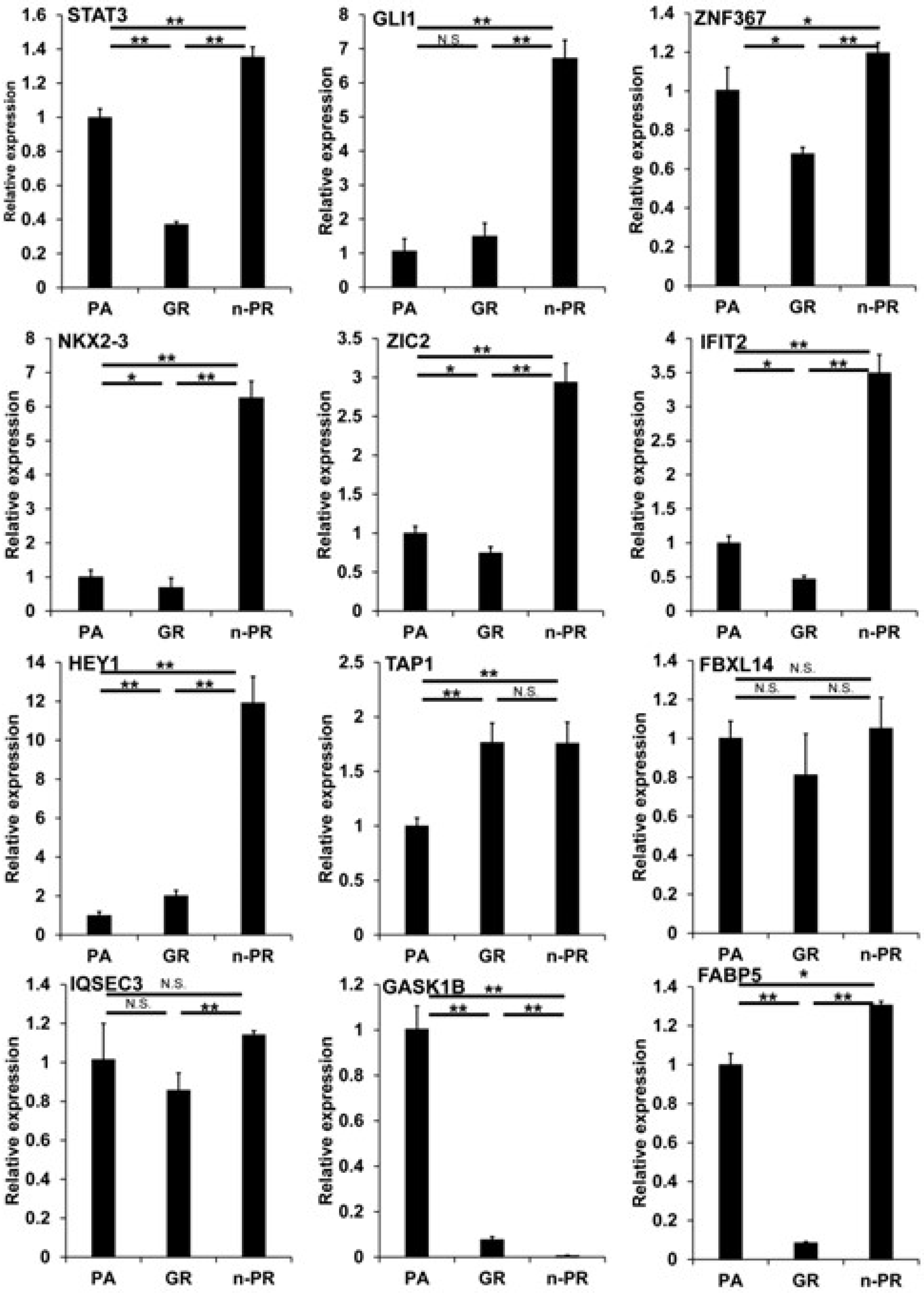
To examine these issues further, we conducted an Ingenuity Pathway Analysis (IPA, Qiagen, Redwood City, CA) to estimate possible up-stream regulators and causal network master regulators between MIA PaCa-2-PA vs. -GR (PA vs. GR), MIA PaCa-2-PA vs. -2 PR (PA vs. PR) or MIA PaCa-2-GR vs. -PR (GR vs. PR), respectively. As shown in Table 3, the results indicated that 2 or 4 (PA vs. GR), 3 or 8 (PA vs. PR), and 3 or 9 (GR vs. PR) candidate genes were estimated as possible upstream regulators or causal network masters, respectively. It was speculated that the observed DEGs and their related estimations should be included in two possible mechanisms, that is, (1) biological deteriorations and/or cellular damage caused by GEM or PTX, and (2) newly acquired biological activities for survival in the presence of GEM or PTX. Taking into account the possible roles of cancer progression of each up-stream regulator and the causal network master regulators, which have already been elucidated (Table 3), we rationally speculate that IFIT2-related signaling or STAT3, GLI1, ZNF367, NKX3-2, ZIC2, HEY1, TAP1, and FBXL14 related signaling represent possible candidates involved in the underlying molecular mechanisms causing chemoresistance against GEM or PTX, respectively, in addition to three possible factors, i.e., FABP5, IQSEC3, and GASK1B as above. Among these candidate genes, the qPCR analysis (Figure 5) confirmed that IFIT2 and FABP5 or STAT3, GLI1, NKX3-2, ZIC2, HEY1, TAP1, and GASK1B may be truly possible master regulators for inducing GEM or PTX resistance in MIA PaCa-2 cells.

Table 3.
Possible upstream regulators and causal network master regulators.
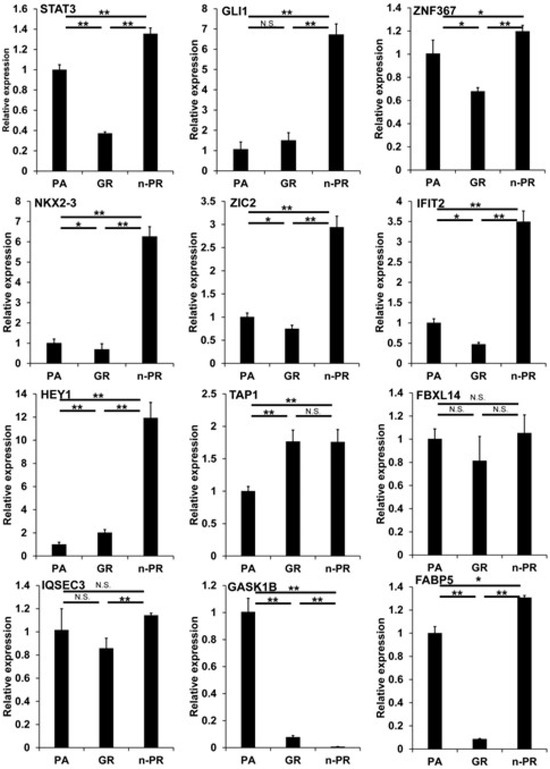
Figure 5.
qPCR analysis of several candidate regulatory genes among 2D cultured MIA capa-2-PA, -GR and -PR. Among the 2D cultured cells obtained from 2D cultured MIA PaCa-2-PA, -GR and -PR, the mRNA expression of STAT3, GLI1, ZNF367, NKX3-2, ZIC2, IFIT2, NEY1, TAP1, FBXL14, IQSEC3, GASK1B and FABP5 were evaluated by a qPCR procedure. All experiments were performed in triplicate, each of which involved the use of freshly prepared 2D structures (n = 3) in each experimental condition. * p < 0.05, ** p < 0.01, N.S. not significant.
4. Discussion
It is known that GEM, n-PTX, and other anti-cancer drugs are effective in the treatment of patients with advanced and metastatic PDAC, but acquiring chemoresistance to these drugs seriously deteriorates their effectiveness. However, although those underlying molecular mechanisms have not yet been fully identified, various transcription factors and signaling pathways involved in nucleoside metabolism are possible candidates for being involved in the development of such chemoresistance [61,62,63,64]. Theoretically, possible underlying molecular mechanisms for causing acquired chemoresistance include drug transport, drug-induced effects on various enzymes and others. It is likely that drug transport, activation, and metabolism are precisely regulated by numerous enzymes, and therefore, acquiring chemoresistance is thought to be regulated by various additional factors, including the tumor microenvironment, EMT, microRNA, and others [64]. Among these mechanisms, it is well known that the EMT phase converts phenotypes into tumor cells in which aggressive EMT changes are evoked and thus associated with their morphological changes as well as various alterations in genome and protein levels. Alternatively, it has also been reported that such mesenchymal transcription factors are pivotal factors in the induction of chemoresistance [64]. In fact, in addition to two key transcription factors, Snail and Twist, various signaling pathways could also be responsible factors such as Notch, tumor necrosis factor-alpha (TNFα), transforming growth factor beta (TGF-β), and hypoxia-inducible factor-1 alpha (HIF1α), which are involved in the induction of EMT in pancreatic cancer cells [65]. Quite interestingly, in the current study, all of the up-stream regulators and/or causal network master regulators estimated by the IPA analysis of the RNA sequencing, that is, STAT3 [42], GLI1 [43], ZNF367 [53], NKX3-2 [47], ZIC2 [51] IFIT2 [46], HEY1 [55] and FBXL14 [60] were identified as being directly or indirectly related to the EMT mechanisms of cancerous cells. Furthermore, among four factors, FABP5, IQSEC3, GASK1B, and SCN1A were identified as unique genes associated with their antisense RNA or pseudogene among the prominently altered DEGs (Log2 fold changes more than 6 or less than −6), FABP5 [66] and GASK1B [67] are also identified as modulators of cancerous EMT. Alternatively, despite the lack of evidence of any apparent correlation with cancerous EMT, IQSEC3 was identified as a novel prognostic marker for breast cancer patients [68], and it is known that SCN1A is a possible factor in the development of chemoresistance in esophageal adenocarcinoma [69]. Furthermore, VCAM1 was identified as the top 7 and the top 2 significant up-regulated DEG in MIA-GR and MIA-PR, respectively (Table 1). In fact, VCAM1 has been suggested as a factor in estimating poor patient prognosis and can promote tumor metastasis by inducing EMT in cancer [70]. In pancreatic cancer, EMT is known to lead to acquiring the characteristics of cancer stemness and enhance chemotherapy resistance through multiple different ways [71]. For instance, it is reported that the recapitulation of the fibrotic rigidities in pancreatic cancer tissues promotes elements of EMT, and the stiffness induces chemoresistance in pancreatic cancer cells [72]. Based on these findings, we suggested that VCAM1 is a potential regulator of the acquired resistance to GEM and n-PTX, which needs further investigation in the future. Therefore, these collective observations suggest that modulations of the cancerous EMT phenotype may be the main factor in the underlying molecular mechanisms for the induction of chemoresistance against GEM or PTX in MIA PaCa-2 cells.
Recent studies related to the field of cancer biology have pointed to the biological importance of the tumor surrounding environment (TSE) in addition to cancerous cells themselves because of their great influence on tumorigenesis, progression, metastasis, and drug sensitivities [73,74,75,76]. To study this further, in contrast to the conventional in vitro 2D planar cell culture models, in vitro 3D cell culture models will be required because of their high potential for replicating the physiological and spatial local environments of cancerous cells. In fact, such 3D cell culture methods are being more frequently applied for testing not only concerning drug efficacy but also in determining suitable dosages for chemotherapy [77,78,79,80,81]. Among the numerous in vitro 3D cell culture methods [20,21], we successfully produced various simple in vitro 3D spheroids using non-cancerous cells [26,27,28] as well as cancer cells [30,31] and found that the physical properties of the 3D spheroid represent potentially new indicators for estimating the biological nature of cancerous cells such as malignancy and drug efficacies [30,31,32,41]. In the current investigation, unfortunately, we were only successful in producing pre-matured 3D spheroids but not solid 3D spheroids, which are usually generated from most non-cancerous [26,27,28] and cancerous cells [30,31] using MIA PaCa-2 cells. However, similar to our results, 3D spheroid cultures of PDAC-related cells such as PANC-1 and MIA PaCa-2 were extremely difficult to produce, and in fact, much longer culture periods in excess of 10 days were required to obtain quite soft 3D spheroids [82,83,84]. Alternatively, to produce more solid 3D spheroids, co-culturing with other cells, such as cancer-associated fibroblasts (CAF), is required in addition to PDAC [84,85]. However, in the current study, the mitochondrial and glycolytic functions of the 3D MIA PaCa-2 spheroids were significantly different from those of their 2D cultured cells, and these differences in the cellular metabolic functions between 2D and 3D cultured cells closely resembled 3T3-L1 cells [33]. Therefore, even though such soft and pre-mature 3D spheroids were formed, their biological natures may already have been altered as the solid 3D spheroid. Collectively, 3D spatial environments rather than solid formation may be required for developing a complete understanding of the biological significance of the in vitro 3D spheroid models.
The present study showed that metabolic capacities were significantly increased in MIA PaCa-2-GR and MIA PaCa-2-PR compared to those in MIA PaCa-2-PA. Such increased metabolic capacities were observed in both 2D- and 3D-culture conditions. Activation in metabolic pathways has been reported as one phenotype of metabolic plasticity, a finding that can be observed in cancer cells that are resistant to chemotherapy [86], which is consistent with the findings in the present study. The molecular mechanisms underlying chemotherapy resistance-induced metabolic alteration are presumably multifactorial, but the most plausible interpretation would be a compensatory response of cancer cells to secure energy for survival against anti-cancer agents. Indeed, it has also been reported that EMT can activate both oxidative phosphorylation and glycolysis [87]. Interestingly, the degree of increased metabolic capacity in MIA-PaCa-2-GR was milder than that in MIA-PaCa-2-PR. The finding that the gene expression level of FABP5, an important lipid chaperon for the activation of intracellular metabolism in cancer cells [88,89], was markedly different between MIA-PaCa-2-GR and MIA-PaCa-2-PR might be one of the explanations for the differences in metabolic capacities between two cells. Nevertheless, an enhanced metabolic capacity in chemotherapy-resistant cells may play a significant role in cell survival and function. The assessment of cellular metabolism in the acquisition of chemotherapy resistance may be an important factor in the selection of anti-cancer therapies.
In conclusion, our current observations using RNA sequencings suggest that modulations of cancerous EMT may be key underlying molecular mechanisms that are responsible for inducing chemoresistance against GEM or PTX in MIA PaCa-2 cells. However, the 3D spheroid appearance and cellular metabolic aspects of the GEM or PTX-resistant MIA PaCa-2 were significantly different from each other, and possible upstream and causal network master regulators inducing their chemoresistance estimated by IPA analysis were also different. Therefore, these collective observations suggest that the acquirement of chemoresistance against GEM or PTX may associate diverse modulations of the cancerous EMT, and thus, additional investigation to elucidate these unidentified issues will be required as our next project.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/biomedicines12051011/s1, Figure S1: M-A plot and volcano plot for MIA capa-2-PA vs. -GR., Figure S2: M-A plot and volcano plot for MIA capa-2-PA vs. -GR., Figure S3: M-A plot and volcano plot for MIA capa-2-GR vs. -PR.; Table S1: primers for qPCR.
Author Contributions
H.N. designed and performed experiments, analyzed data, and wrote the paper; M.W. designed and performed experiments, analyzed data, and wrote the paper; T.S. designed and performed experiments, analyzed data, and wrote the paper; F.H., A.U., J.M., M.F. and H.O. analyzed the data, K.T. designed experiments, analyzed the data, and wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Lim, J.E.; Chien, M.W.; Earle, C.C. Prognostic factors following curative resection for pancreatic adenocarcinoma: A population-based, linked database analysis of 396 patients. Ann. Surg. 2003, 237, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.X.; Zhao, C.F.; Chen, W.B.; Liu, Q.C.; Li, Q.W.; Lin, Y.Y.; Gao, F. Pancreatic cancer: A review of epidemiology, trend, and risk factors. World J. Gastroenterol. 2021, 27, 4298–4321. [Google Scholar] [CrossRef] [PubMed]
- Sohn, T.A.; Yeo, C.J.; Cameron, J.L.; Koniaris, L.; Kaushal, S.; Abrams, R.A.; Sauter, P.K.; Coleman, J.; Hruban, R.H.; Lillemoe, K.D. Resected adenocarcinoma of the pancreas-616 patients: Results, outcomes, and prognostic indicators. J. Gastrointest. Surg. 2000, 4, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Neoptolemos, J.P.; Palmer, D.H.; Ghaneh, P.; Psarelli, E.E.; Valle, J.W.; Halloran, C.M.; Faluyi, O.; O’Reilly, D.A.; Cunningham, D.; Wadsley, J.; et al. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): A multicentre, open-label, randomised, phase 3 trial. Lancet 2017, 389, 1011–1024. [Google Scholar] [CrossRef] [PubMed]
- Conroy, T.; Hammel, P.; Hebbar, M.; Ben Abdelghani, M.; Wei, A.C.; Raoul, J.L.; Choné, L.; Francois, E.; Artru, P.; Biagi, J.J.; et al. FOLFIRINOX or Gemcitabine as Adjuvant Therapy for Pancreatic Cancer. N. Engl. J. Med. 2018, 379, 2395–2406. [Google Scholar] [CrossRef] [PubMed]
- Von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, E.G.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, D.; El-Maraghi, R.H.; Hammel, P.; Heinemann, V.; Kunzmann, V.; Sastre, J.; Scheithauer, W.; Siena, S.; Tabernero, J.; Teixeira, L.; et al. nab-Paclitaxel plus gemcitabine for metastatic pancreatic cancer: Long-term survival from a phase III trial. J. Natl. Cancer Inst. 2015, 107. [Google Scholar] [CrossRef]
- Tempero, M.A.; Pelzer, U.; O’Reilly, E.M.; Winter, J.; Oh, D.Y.; Li, C.P.; Tortora, G.; Chang, H.M.; Lopez, C.D.; Bekaii-Saab, T.; et al. Adjuvant nab-Paclitaxel + Gemcitabine in Resected Pancreatic Ductal Adenocarcinoma: Results From a Randomized, Open-Label, Phase III Trial. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2023, 41, 2007–2019. [Google Scholar] [CrossRef] [PubMed]
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.L.; Gourgou-Bourgade, S.; de la Fouchardière, C.; et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef]
- Jain, A.; Bhardwaj, V. Therapeutic resistance in pancreatic ductal adenocarcinoma: Current challenges and future opportunities. World J. Gastroenterol. 2021, 27, 6527–6550. [Google Scholar] [CrossRef]
- Miyabayashi, K.; Nakagawa, H.; Koike, K. Molecular and Phenotypic Profiling for Precision Medicine in Pancreatic Cancer: Current Advances and Future Perspectives. Front. Oncol. 2021, 11, 682872. [Google Scholar] [CrossRef] [PubMed]
- Gündel, B.; Liu, X.; Löhr, M.; Heuchel, R. Pancreatic Ductal Adenocarcinoma: Preclinical in vitro and ex vivo Models. Front. Cell Dev. Biol. 2021, 9, 741162. [Google Scholar] [CrossRef]
- Sharma, S.V.; Haber, D.A.; Settleman, J. Cell line-based platforms to evaluate the therapeutic efficacy of candidate anticancer agents. Nat. Rev. Cancer 2010, 10, 241–253. [Google Scholar] [CrossRef] [PubMed]
- Bleijs, M.; van de Wetering, M.; Clevers, H.; Drost, J. Xenograft and organoid model systems in cancer research. Embo J. 2019, 38, e101654. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.; Zhang, X.; Parsons, D.W.; Lin, J.C.; Leary, R.J.; Angenendt, P.; Mankoo, P.; Carter, H.; Kamiyama, H.; Jimeno, A.; et al. Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science 2008, 321, 1801–1806. [Google Scholar] [CrossRef] [PubMed]
- Hagmann, W.; Jesnowski, R.; Löhr, J.M. Interdependence of gemcitabine treatment, transporter expression, and resistance in human pancreatic carcinoma cells. Neoplasia 2010, 12, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, Y.; Ahmad, A.; Banerjee, S.; Azmi, A.S.; Kong, D.; Sarkar, F.H. Pancreatic cancer: Understanding and overcoming chemoresistance. Nature reviews. Gastroenterol. Hepatol. 2011, 8, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Habanjar, O.; Diab-Assaf, M.; Caldefie-Chezet, F.; Delort, L. 3D Cell Culture Systems: Tumor Application, Advantages, and Disadvantages. Int. J. Mol. Sci. 2021, 22, 12200. [Google Scholar] [CrossRef] [PubMed]
- Mueller-Klieser, W. Multicellular spheroids. A review on cellular aggregates in cancer research. J. Cancer Res. Clin. Oncol. 1987, 113, 101–122. [Google Scholar] [CrossRef]
- Santini, M.T.; Rainaldi, G. Three-dimensional spheroid model in tumor biology. Pathobiol. J. Immunopathol. Mol. Cell. Biol. 1999, 67, 148–157. [Google Scholar] [CrossRef]
- Katt, M.E.; Placone, A.L.; Wong, A.D.; Xu, Z.S.; Searson, P.C. In Vitro Tumor Models: Advantages, Disadvantages, Variables, and Selecting the Right Platform. Front. Bioeng. Biotechnol. 2016, 4, 12. [Google Scholar] [CrossRef]
- Sipos, B.; Möser, S.; Kalthoff, H.; Török, V.; Löhr, M.; Klöppel, G. A comprehensive characterization of pancreatic ductal carcinoma cell lines: Towards the establishment of an in vitro research platform. Virchows Arch. Int. J. Pathol. 2003, 442, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Barrera, A.M.; Menter, D.G.; Abbruzzese, J.L.; Reddy, S.A. Establishment of three-dimensional cultures of human pancreatic duct epithelial cells. Biochem. Biophys. Res. Commun. 2007, 358, 698–703. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Longati, P.; Jia, X.; Eimer, J.; Wagman, A.; Witt, M.R.; Rehnmark, S.; Verbeke, C.; Toftgård, R.; Löhr, M.; Heuchel, R.L. 3D pancreatic carcinoma spheroids induce a matrix-rich, chemoresistant phenotype offering a better model for drug testing. BMC Cancer 2013, 13, 95. [Google Scholar] [CrossRef]
- Katayama, H.; Furuhashi, M.; Umetsu, A.; Hikage, F.; Watanabe, M.; Ohguro, H.; Ida, Y. Modulation of the Physical Properties of 3D Spheroids Derived from Human Scleral Stroma Fibroblasts (HSSFs) with Different Axial Lengths Obtained from Surgical Patients. Curr. Issues Mol. Biol. 2021, 43, 1715–1725. [Google Scholar] [CrossRef] [PubMed]
- Hikage, F.; Atkins, S.; Kahana, A.; Smith, T.J.; Chun, T.H. HIF2A-LOX Pathway Promotes Fibrotic Tissue Remodeling in Thyroid-Associated Orbitopathy. Endocrinology 2019, 160, 20–35. [Google Scholar] [CrossRef]
- Watanabe, M.; Ida, Y.; Furuhashi, M.; Tsugeno, Y.; Ohguro, H.; Hikage, F. Screening of the Drug-Induced Effects of Prostaglandin EP2 and FP Agonists on 3D Cultures of Dexamethasone-Treated Human Trabecular Meshwork Cells. Biomedicines 2021, 9, 930. [Google Scholar] [CrossRef] [PubMed]
- Oouchi, Y.; Watanabe, M.; Ida, Y.; Ohguro, H.; Hikage, F. Rosiglitasone and ROCK Inhibitors Modulate Fibrogenetic Changes in TGF-β2 Treated Human Conjunctival Fibroblasts (HconF) in Different Manners. Int. J. Mol. Sci. 2021, 22, 335. [Google Scholar] [CrossRef]
- Watanabe, M.; Yano, T.; Sato, T.; Umetsu, A.; Higashide, M.; Furuhashi, M.; Ohguro, H. mTOR Inhibitors Modulate the Physical Properties of 3D Spheroids Derived from H9c2 Cells. Int. J. Mol. Sci. 2023, 24, 11459. [Google Scholar] [CrossRef]
- Ichioka, H.; Hirohashi, Y.; Sato, T.; Furuhashi, M.; Watanabe, M.; Ida, Y.; Hikage, F.; Torigoe, T.; Ohguro, H. G-Protein-Coupled Receptors Mediate Modulations of Cell Viability and Drug Sensitivity by Aberrantly Expressed Recoverin 3 within A549 Cells. Int. J. Mol. Sci. 2023, 24, 771. [Google Scholar] [CrossRef]
- Ohguro, H.; Watanabe, M.; Sato, T.; Hikage, F.; Furuhashi, M.; Okura, M.; Hida, T.; Uhara, H. 3D Spheroid Configurations Are Possible Indictors for Evaluating the Pathophysiology of Melanoma Cell Lines. Cells 2023, 12, 759. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, S.; Nishikiori, N.; Sato, T.; Watanabe, M.; Umetsu, A.; Tsugeno, Y.; Hikage, F.; Sasaya, T.; Kato, H.; Ogi, K.; et al. Three-Dimensional Spheroid Configurations and Cellular Metabolic Properties of Oral Squamous Carcinomas Are Possible Pharmacological and Pathological Indicators. Cancers 2023, 15, 2793. [Google Scholar] [CrossRef] [PubMed]
- Ohguro, H.; Ida, Y.; Hikage, F.; Umetsu, A.; Ichioka, H.; Watanabe, M.; Furuhashi, M. STAT3 Is the Master Regulator for the Forming of 3D Spheroids of 3T3-L1 Preadipocytes. Cells 2022, 11, 300. [Google Scholar] [CrossRef] [PubMed]
- Akama, T.; Leung, B.M.; Labuz, J.; Takayama, S.; Chun, T.H. Designing 3-D Adipospheres for Quantitative Metabolic Study. Methods Mol. Biol. 2017, 1566, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Ida, Y.; Hikage, F.; Itoh, K.; Ida, H.; Ohguro, H. Prostaglandin F2α agonist-induced suppression of 3T3-L1 cell adipogenesis affects spatial formation of extra-cellular matrix. Sci. Rep. 2020, 10, 7958. [Google Scholar] [CrossRef] [PubMed]
- Ida, Y.; Hikage, F.; Umetsu, A.; Ida, H.; Ohguro, H. Omidenepag, a non-prostanoid EP2 receptor agonist, induces enlargement of the 3D organoid of 3T3-L1 cells. Sci. Rep. 2020, 10, 16018. [Google Scholar] [CrossRef] [PubMed]
- Itoh, K.; Hikage, F.; Ida, Y.; Ohguro, H. Prostaglandin F2α Agonists Negatively Modulate the Size of 3D Organoids from Primary Human Orbital Fibroblasts. Investig. Ophthalmol. Vis. Sci. 2020, 61, 13. [Google Scholar] [CrossRef]
- Keller, K.E.; Bhattacharya, S.K.; Borrás, T.; Brunner, T.M.; Chansangpetch, S.; Clark, A.F.; Dismuke, W.M.; Du, Y.; Elliott, M.H.; Ethier, C.R.; et al. Consensus recommendations for trabecular meshwork cell isolation, characterization and culture. Exp. Eye Res. 2018, 171, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef]
- Krämer, A.; Green, J.; Pollard, J., Jr.; Tugendreich, S. Causal analysis approaches in Ingenuity Pathway Analysis. Bioinformatics 2014, 30, 523–530. [Google Scholar] [CrossRef]
- Marconi, A.; Quadri, M.; Saltari, A.; Pincelli, C. Progress in melanoma modelling in vitro. Exp. Dermatol. 2018, 27, 578–586. [Google Scholar] [CrossRef]
- Sadrkhanloo, M.; Entezari, M.; Orouei, S.; Ghollasi, M.; Fathi, N.; Rezaei, S.; Hejazi, E.S.; Kakavand, A.; Saebfar, H.; Hashemi, M.; et al. STAT3-EMT axis in tumors: Modulation of cancer metastasis, stemness and therapy response. Pharmacol. Res. 2022, 182, 106311. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.T.; Wu, K.J. Epigenetic regulation of epithelial-mesenchymal transition: Focusing on hypoxia and TGF-β signaling. J. Biomed. Sci. 2020, 27, 39. [Google Scholar] [CrossRef]
- Bankhead, A., 3rd; McMaster, T.; Wang, Y.; Boonstra, P.S.; Palmbos, P.L. TP63 isoform expression is linked with distinct clinical outcomes in cancer. EBioMedicine 2020, 51, 102561. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Xiao, L.; Rong, H.; Ou, Z.; Cai, T.; Liu, N.; Li, B.; Zhang, L.; Wu, F.; Lan, T.; et al. Single-cell profiling of tumor-infiltrating TCF1/TCF7(+) T cells reveals a T lymphocyte subset associated with tertiary lymphoid structures/organs and a superior prognosis in oral cancer. Oral. Oncol. 2021, 119, 105348. [Google Scholar] [CrossRef]
- Chen, J.; Liu, Y.; Zhu, Y.; Chen, Y.; Feng, J.; Jiang, T.; Zheng, X.; Chen, L.; Jiang, J. STAT1/IFIT2 signaling pathway is involved in PD-L1-mediated epithelial-to-mesenchymal transition in human esophageal cancer. Clin. Transl. Oncol. 2022, 24, 927–940. [Google Scholar] [CrossRef]
- Ouyang, S.; Zhu, G.; Ouyang, L.; Luo, Y.; Zhou, R.; Pan, C.; Bin, J.; Liao, Y.; Liao, W. Bapx1 mediates transforming growth factor-β- induced epithelial-mesenchymal transition and promotes a malignancy phenotype of gastric cancer cells. Biochem. Biophys. Res. Commun. 2017, 486, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, C.; Hu, Y.; Li, X.; Jin, S.; Liu, O.; Gou, R.; Zhuang, Y.; Guo, Q.; Nie, X.; et al. ZNF703 promotes tumor progression in ovarian cancer by interacting with HE4 and epigenetically regulating PEA15. J. Exp. Clin. Cancer Res. 2020, 39, 264. [Google Scholar] [CrossRef] [PubMed]
- Bersten, D.C.; Sullivan, A.E.; Peet, D.J.; Whitelaw, M.L. bHLH-PAS proteins in cancer. Nature reviews. Cancer 2013, 13, 827–841. [Google Scholar] [CrossRef]
- Geng, P.; Ou, J.; Li, J.; Liao, Y.; Wang, N.; Sa, R.; Xiang, L.; Liang, H. Genetic Association Between NFKBIA -881A>G Polymorphism and Cancer Susceptibility. Medicine 2015, 94, e1024. [Google Scholar] [CrossRef]
- Jiang, Z.; Zhang, Y.; Chen, X.; Wu, P.; Chen, D. Inactivation of the Wnt/β-catenin signaling pathway underlies inhibitory role of microRNA-129-5p in epithelial-mesenchymal transition and angiogenesis of prostate cancer by targeting ZIC2. Cancer Cell Int. 2019, 19, 271. [Google Scholar] [CrossRef] [PubMed]
- Steerenberg, P.A.; Vendrik, C.P.; de Jong, W.H.; de Groot, G.; Fichtinger-Schepman, A.M.; Scheefhals, A.P.; Schornagel, J.H. Resistance and cross-resistance of the IgM immunocytoma in the LOU/M Wsl rat for cisplatin, carboplatin, and iproplatin. Cancer Chemother. Pharmacol. 1988, 22, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.; Zhang, L.; Boufraqech, M.; Liu-Chittenden, Y.; Bussey, K.; Demeure, M.J.; Wu, X.; Su, L.; Pacak, K.; Stratakis, C.A.; et al. ZNF367 inhibits cancer progression and is targeted by miR-195. PLoS ONE 2014, 9, e101423. [Google Scholar] [CrossRef]
- Teng, Y.; Loveless, R.; Benson, E.M.; Sun, L.; Shull, A.Y.; Shay, C. SHOX2 cooperates with STAT3 to promote breast cancer metastasis through the transcriptional activation of WASF3. J. Exp. Clin. Cancer Res. 2021, 40, 274. [Google Scholar] [CrossRef]
- Fukusumi, T.; Guo, T.W.; Sakai, A.; Ando, M.; Ren, S.; Haft, S.; Liu, C.; Amornphimoltham, P.; Gutkind, J.S.; Califano, J.A. The NOTCH4-HEY1 Pathway Induces Epithelial-Mesenchymal Transition in Head and Neck Squamous Cell Carcinoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2018, 24, 619–633. [Google Scholar] [CrossRef]
- Zhu, Z.; Hou, Q.; Wang, B.; Li, C.; Liu, L.; Gong, W.; Chai, J.; Guo, H.; Jia, Y. FKBP4 regulates 5-fluorouracil sensitivity in colon cancer by controlling mitochondrial respiration. Life Sci. Alliance 2022, 5. [Google Scholar] [CrossRef]
- Liu, Q.; Li, A.; Tian, Y.; Wu, J.D.; Liu, Y.; Li, T.; Chen, Y.; Han, X.; Wu, K. The CXCL8-CXCR1/2 pathways in cancer. Cytokine Growth Factor. Rev. 2016, 31, 61–71. [Google Scholar] [CrossRef]
- Wu, G.; Wu, F.; Zhou, Y.Q.; Lu, W.; Hu, F.L.; Fan, X. Silencing of TRAF5 enhances necroptosis in hepatocellular carcinoma by inhibiting LTBR-mediated NF-κB signaling. PeerJ 2023, 11, e15551. [Google Scholar] [CrossRef] [PubMed]
- Tu, Z.; Li, K.; Ji, Q.; Huang, Y.; Lv, S.; Li, J.; Wu, L.; Huang, K.; Zhu, X. Pan-cancer analysis: Predictive role of TAP1 in cancer prognosis and response to immunotherapy. BMC Cancer 2023, 23, 133. [Google Scholar] [CrossRef]
- Zhao, B.; Huo, W.; Yu, X.; Shi, X.; Lv, L.; Yang, Y.; Kang, J.; Li, S.; Wu, H. USP13 promotes breast cancer metastasis through FBXL14-induced Twist1 ubiquitination. Cell. Oncol. 2023, 46, 717–733. [Google Scholar] [CrossRef]
- Ireland, L.; Santos, A.; Ahmed, M.S.; Rainer, C.; Nielsen, S.R.; Quaranta, V.; Weyer-Czernilofsky, U.; Engle, D.D.; Perez-Mancera, P.A.; Coupland, S.E.; et al. Chemoresistance in Pancreatic Cancer Is Driven by Stroma-Derived Insulin-Like Growth Factors. Cancer Res. 2016, 76, 6851–6863. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.K.; Purohit, V.; Mehla, K.; Gunda, V.; Chaika, N.V.; Vernucci, E.; King, R.J.; Abrego, J.; Goode, G.D.; Dasgupta, A.; et al. MUC1 and HIF-1alpha Signaling Crosstalk Induces Anabolic Glucose Metabolism to Impart Gemcitabine Resistance to Pancreatic Cancer. Cancer Cell 2017, 32, 71–87.e77. [Google Scholar] [CrossRef] [PubMed]
- Dauer, P.; Nomura, A.; Saluja, A.; Banerjee, S. Microenvironment in determining chemo-resistance in pancreatic cancer: Neighborhood matters. Pancreatol. Off. J. Int. Assoc. Pancreatol. 2017, 17, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Zeng, S.; Pöttler, M.; Lan, B.; Grützmann, R.; Pilarsky, C.; Yang, H. Chemoresistance in Pancreatic Cancer. Int. J. Mol. Sci. 2019, 20, 4504. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Yelle, N.; Venugopal, C.; Singh, S.K. EMT: Mechanisms and therapeutic implications. Pharmacol. Ther. 2018, 182, 80–94. [Google Scholar] [CrossRef] [PubMed]
- De Mattos-Arruda, L.; Cortes, J.; Blanco-Heredia, J.; Tiezzi, D.G.; Villacampa, G.; Gonçalves-Ribeiro, S.; Paré, L.; Souza, C.A.; Ortega, V.; Sammut, S.J.; et al. The temporal mutational and immune tumour microenvironment remodelling of HER2-negative primary breast cancers. NPJ Breast Cancer 2021, 7, 73. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.Y.; Chang, G.C.; Chen, Y.J.; Hsu, Y.C.; Hsiao, Y.J.; Su, K.Y.; Chen, H.Y.; Lin, C.Y.; Chen, J.S.; Chen, Y.J.; et al. FAM198B Is Associated with Prolonged Survival and Inhibits Metastasis in Lung Adenocarcinoma via Blockage of ERK-Mediated MMP-1 Expression. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2018, 24, 916–926. [Google Scholar] [CrossRef]
- Mo, W.; Ding, Y.; Zhao, S.; Zou, D.; Ding, X. Identification of a 6-gene signature for the survival prediction of breast cancer patients based on integrated multi-omics data analysis. PLoS ONE 2020, 15, e0241924. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Zhang, D.; Qian, X.; Wei, H.; Zhou, L.; Ding, C.; Pan, W.; Ye, Q. Analysis of cancer-promoting genes related to chemotherapy resistance in esophageal squamous cell carcinoma. Ann. Transl. Med. 2022, 10, 92. [Google Scholar] [CrossRef]
- Huang, Z.; Wu, C.; Zhou, W.; Lu, S.; Tan, Y.; Wu, Z.; You, R.; Stalin, A.; Guo, F.; Zhang, J.; et al. Compound Kushen Injection inhibits epithelial-mesenchymal transition of gastric carcinoma by regulating VCAM1 induced by the TNF signaling pathway. Phytomed. Int. J. Phytother. Phytopharm. 2023, 118, 154984. [Google Scholar] [CrossRef]
- Tangsiri, M.; Hheidari, A.; Liaghat, M.; Razlansari, M.; Ebrahimi, N.; Akbari, A.; Varnosfaderani, S.M.N.; Maleki-Sheikhabadi, F.; Norouzi, A.; Bakhtiyari, M.; et al. Promising applications of nanotechnology in inhibiting chemo-resistance in solid tumors by targeting epithelial-mesenchymal transition (EMT). Biomed. Pharmacother. Biomed. Pharmacother. 2024, 170, 115973. [Google Scholar] [CrossRef] [PubMed]
- Rice, A.J.; Cortes, E.; Lachowski, D.; Cheung, B.C.H.; Karim, S.A.; Morton, J.P.; Del Río Hernández, A. Matrix stiffness induces epithelial-mesenchymal transition and promotes chemoresistance in pancreatic cancer cells. Oncogenesis 2017, 6, e352. [Google Scholar] [CrossRef] [PubMed]
- Biffi, G.; Tuveson, D.A. Diversity and Biology of Cancer-Associated Fibroblasts. Physiol. Rev. 2021, 101, 147–176. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; McAndrews, K.M.; Kalluri, R. Clinical and therapeutic relevance of cancer-associated fibroblasts. Nat. Rev.. Clin. Oncol. 2021, 18, 792–804. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 2016, 16, 582–598. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.Y.; Kang, S.H.; Oh, S.Y.; Lee, K.Y.; Lee, H.J.; Gum, S.; Kwon, T.G.; Kim, J.W.; Lee, S.T.; Hong, Y.J.; et al. Differential Angiogenic Potential of 3-Dimension Spheroid of HNSCC Cells in Mouse Xenograft. Int. J. Mol. Sci. 2021, 22, 8245. [Google Scholar] [CrossRef] [PubMed]
- Jensen, C.; Teng, Y. Is It Time to Start Transitioning From 2D to 3D Cell Culture? Front. Mol. Biosci. 2020, 7, 33. [Google Scholar] [CrossRef]
- Miki, Y.; Ono, K.; Hata, S.; Suzuki, T.; Kumamoto, H.; Sasano, H. The advantages of co-culture over mono cell culture in simulating in vivo environment. J. Steroid Biochem. Mol. Biol. 2012, 131, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Shang, M.; Soon, R.H.; Lim, C.T.; Khoo, B.L.; Han, J. Microfluidic modelling of the tumor microenvironment for anti-cancer drug development. Lab. Chip 2019, 19, 369–386. [Google Scholar] [CrossRef]
- Yamada, K.M.; Cukierman, E. Modeling tissue morphogenesis and cancer in 3D. Cell 2007, 130, 601–610. [Google Scholar] [CrossRef]
- Zanoni, M.; Pignatta, S.; Arienti, C.; Bonafè, M.; Tesei, A. Anticancer drug discovery using multicellular tumor spheroid models. Expert. Opin. Drug Discov. 2019, 14, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Paškevičiūtė, M.; Petrikaitė, V. Differences of statin activity in 2D and 3D pancreatic cancer cell cultures. Drug Des. Dev. Ther. 2017, 11, 3273–3280. [Google Scholar] [CrossRef] [PubMed]
- Madsen, N.H.; Nielsen, B.S.; Nhat, S.L.; Skov, S.; Gad, M.; Larsen, J. Monocyte Infiltration and Differentiation in 3D Multicellular Spheroid Cancer Models. Pathogens 2021, 10, 969. [Google Scholar] [CrossRef] [PubMed]
- Krulikas, L.J.; McDonald, I.M.; Lee, B.; Okumu, D.O.; East, M.P.; Gilbert, T.S.K.; Herring, L.E.; Golitz, B.T.; Wells, C.I.; Axtman, A.D.; et al. Application of Integrated Drug Screening/Kinome Analysis to Identify Inhibitors of Gemcitabine-Resistant Pancreatic Cancer Cell Growth. SLAS Discov. Adv. Life Sci. R. D 2018, 23, 850–861. [Google Scholar] [CrossRef] [PubMed]
- Brumskill, S.; Barrera, L.N.; Calcraft, P.; Phillips, C.; Costello, E. Inclusion of cancer-associated fibroblasts in drug screening assays to evaluate pancreatic cancer resistance to therapeutic drugs. J. Physiol. Biochem. 2023, 79, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Desbats, M.A.; Giacomini, I.; Prayer-Galetti, T.; Montopoli, M. Metabolic Plasticity in Chemotherapy Resistance. Front. Oncol. 2020, 10, 281. [Google Scholar] [CrossRef]
- Jia, D.; Park, J.H.; Kaur, H.; Jung, K.H.; Yang, S.; Tripathi, S.; Galbraith, M.; Deng, Y.; Jolly, M.K.; Kaipparettu, B.A. Towards decoding the coupled decision-making of metabolism and epithelial-to-mesenchymal transition in cancer. Br. J. Cancer 2021, 124, 1902–1911. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.; Jeong, D.W.; Park, J.W.; Lee, K.W.; Fukuda, J.; Chun, Y.S. Fatty-acid-induced FABP5/HIF-1 reprograms lipid metabolism and enhances the proliferation of liver cancer cells. Commun. Biol. 2020, 3, 638. [Google Scholar] [CrossRef]
- Zhang, C.; Liao, Y.; Liu, P.; Du, Q.; Liang, Y.; Ooi, S.; Qin, S.; He, S.; Yao, S.; Wang, W. FABP5 promotes lymph node metastasis in cervical cancer by reprogramming fatty acid metabolism. Theranostics 2020, 10, 6561–6580. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).