This research investigated the impact of cork stoppers and screw caps on the aromatic profiles of sparkling wine after second fermentation and aging in a bottle. The yeast-lees contact time was 94 months.
3.1. Extraction of Aromatic Compounds versus Type of Closure
Two extraction methods (HP-SPME and TD) were used to obtain volatile compounds of sparkling wine after second fermentation and aging for 94 months. The obtained chromatograms of bottle-aged sparkling wine closured with closure A and isolated by TD and HP-SPME are shown in
Figure 2. A summary of the data obtained by both extraction methodologies is presented in
Table A1 (
Appendix A). Briefly, the number of identified compounds extracted from TD (65) was slightly higher than HP-SPME (53).
These results show that both methodologies are appropriate for the analysis of volatile compounds in sparkling wine. SPME and TD have been successfully used for the identification of aromatic compounds in wine [
25,
26,
36,
40,
53,
54,
55,
56] and sparkling wine [
23,
24,
57]. Therefore, TD required 30 mL of sample for extraction, while HP-SPME used 10 mL.
Furthermore, TD is a time-consuming process (2 h) and requires temperature for extracting volatile compounds. Both depended on the equilibrium partitioning of the volatile compounds between the solution (or sparkling wine) and gas phase. Then, the solutes were extracted from a liquid phase of an aqueous matrix or wine and migrated into a polymer phase; they fit inside the needle of a syringe-like device in the case of HP-SPME and filled a desorption tube in the case of TD. Then, in the case of TD, a thermal desorption-step process followed by a preconcentration step (TD) was performed using a cryotrap. The most important disadvantage of HP-SPME is the lack of sensitivity [
41]. In this study, TD extraction was designed with the aim of benefiting from the higher amount of sorbent, as well as the trap focusing, for the purpose of increasing the sensitivity.
The extracted compounds are classified into families, according to their chemical nature, as acids, alcohols, alkanes, esters, ethers, ketones and others.
Figure 3 shows the percentages of sparkling wine families of volatile compounds extracted by HP-SPME and TD, considering all analyzed closures. As can be seen, some differences were observed in terms of the percentages of the major families of volatiles. HP-SPME allowed for the more effective extraction of esters, considering both the percentage of area (66.1%) and the number of compounds (19). Furthermore, HP-SPME showed a higher proportion of ketones (1.8%) and others (26.2%). On the contrary, the proportion of alcohols (40.6%), ethers (17.0%), alkanes (2.6%), terpenes (3.0%) and acids (4.0%) was higher in TD extracts than HP-SPME (1.9%, 2.0%, 0.9%, 0.1% and 0.8%, respectively).
The most important group of compounds for sparkling wine aroma are ethyl esters of aliphatic acids [
23]. Although HP-SPME allowed for the extraction of a greater number of esters, TD and HP-SPME were appropriate methods for extracting common esters such as methyl 2,4-dimethylhexanoate, ethyl octanoate, ethyl decanoate, ethyl arachidate or ethyl hexanoate in the sparkling wine samples (
Table A1,
Appendix A). Other ester compounds extracted using HP-SPME and TD were ethyl 9-decenoate, diethyl succinate and ethyl butyrate. All these compounds have already been identified in sparkling wines [
23,
25,
58,
59,
60]. Diethyl succinate is a post-fermentation volatile formed during the aging of sparkling wines in contact with lees after second fermentation [
23]. For this reason, this compound is a signal of the evolution of sparkling wines during aging [
24,
25]. Overall, representative esters were detected by both extraction methodologies, but HP-SPME was the quickest and most straightforward method for extracting them.
Alcohols were the second-most-abundant family, with the highest levels of isoamyl alcohol in the case of TD extraction and phenylethyl alcohol in the case of HP-SPME. According to [
59], phenylethyl alcohol contributes to the sweet, floral, and honey-like aroma profile of sparkling wines. Both higher alcohols have already been identified in sparkling wines [
59]. Other representative alcohols such as 1-hexanol and 1-butanol, which are mostly produced during the pre-fermentation wine production process, were extracted using TD. Also, 1-hexanol is a representative alcohol characterized by “green” and “herbaceous” notes.
The different origins of the grapes and/or the winemaking conditions used are likely linked to the presence of fatty acids [
60]. The most significant acids identified were different depending on the use of TD or HP-SPME. Octanoic and decanoic acid were obtained by both methods, but the highest percentage of area was detected in the sparkling wines analyzed by HP-SPME (
Table A1,
Appendix A). We found that some compounds such as succinic acid and dimethyl caffeic acid were only detected by HP-SPME, and others like alkynyl stearic acid, 3-hydroxydodecanoic acid, acetic acid or aminomethanesulfonic acid were identified using TD.
In the case of ether compounds, 2-(1,1-dimethylethyl)-3-methyloxirane and 1-Methyl-1-silacyclopentan-1-ol were identified. The latter was only detected using TD. Ethers are a type of volatile compound that significantly contribute to the intricate aroma of wine. They are mainly produced throughout the alcoholic fermentation, but additionally, they can also develop during the wine-aging process as the wine undergoes chemical reactions in barrels or bottles [
61]. Although ethers are less prevalent than esters, their presence enhances the wine’s aromatic complexity, offering scents that span from herbaceous to floral and spicy. Their interaction with other volatile and non-volatile elements in the wine further shapes their sensory perception and overall impact.
Ketones are a key group of volatile compounds that play a significant role in shaping the aroma profile of wine. Their presence has a crucial impact in the complexity of wine’s sensory attributes, although its concentration is lower compared to other volatiles like esters and alcohols. Ketones are primarily formed during the fermentation processes but also in the aging step [
62].
HP-SPME was the best method used to extract alpha-ionone or a C13-norisoprenoid that is known for its contribution to the aroma of fruits. Other ketones such as caprolactone or 2,3,4,5,6,6-hexamethylcyclohexa-2,4-dien-1-one were also detected using HP-SPME. On the contrary, 2,2-dimethyl-5-phenylfuran-3-one was extracted using TD. Lactones were also detected in sparkling wines due to the aging process because they are generated by the hydrolysis of the precursors.
Alkanes are naturally present in the waxy cuticle of grape skins. They can also form during the fermentation process, because yeast metabolism can produce them as secondary metabolites, or during the aging of wine, due to chemical reactions that can generate or modify alkane compounds. The number and nature of extracted alkanes depend on the extraction methodology. Levels of pentacosane, nonacosane, tetratriacontane, hexatriacontane, 3-choropentane and vinyl decanoate were obtained using HP-SPME. On the other hand, n-hexane, octathiocane, 5-methyl-hexadecane, hexadecane and octadecane were extracted by the TD method. Alkanes typically have a relatively low impact on the aroma profile of wine due to their low volatility. However, they can contribute to the background complexity and texture of the wine.
Terpene compounds, which are a major group of wine aroma compounds known for their floral aromas, were similarly identified using both HP-SPME and TD techniques. It seems that these groups of compounds are produced during the pressing of grapes and the settling process. Squalene is a natural triterpene widely distributed in nature, and it was extracted using both methodologies. Therefore, the TD method allowed for the extraction of another triterpene, Friedelin. This is a compound of the triterpenic fraction of cork, and it is described as a precursor of bioactive components for biomedical applications [
63]. D-limonene or a monoterpene was also obtained using TD, as in the case of TDN, or a C13-norisoprenoid. According to Torrens et al. 2010 [
26] and Francioli et al. 2003 [
64], TDN, which forms from the degradation of carotenoids, is affected by the aging process through acid-catalyzed reactions. Along with diethyl succinate and vitispirane, TDN can be used to distinguish sparkling wines aged for more than 20 months. However, beyond this, due to the relatively low concentrations of these compounds and the complexity of the matrix, their analyses require the previous fractionation and separation of volatile terpenes or a non-polar fraction from a polar fraction [
65].
There are several compounds classified as other. HP-SPME allowed for the extraction of most of them, such as lactamide or dimethylamine. Caprylic anhydride was obtained by both methods. Some of them have not been described previously.
According to our results, HS-SPME may a useful extraction method for esters, ketones and other compounds, while for the extraction of alcohol, acid, ethers, alkanes and terpenes, the TD methodology is better. It was initially expected that TD extraction would be more efficient due to the advantages mentioned earlier. Considering these results, a first step, with the aim of the optimization of the TD methodology, would be necessary to gear its strengths. Some desorption parameters, such as desorption time, desorption temperature, low trap cryo-temperature or high trap temperature, and its associations would affect the response of the volatile compounds [
43].
Ref. [
23] considered that HP-SPME allows for the extraction of the most representative polar compounds of a sparkling wine. In this study, HP-SPME enabled the extraction of several high-molecular-weight compounds, such as acetate, ethyl, and isoamyl esters, which appear to be characteristic aromas of sparkling wines with a short aging period. According to [
64], HP-SPME is proven to be an effective method for detecting diethyl succinate, TDN, hexanol, and other compounds associated with the autolysis process, as well as compounds inherent in the bouquet of long-aged sparkling wines.
The behaviors of the cork stoppers and screw caps used in bottle aging, after the second fermentation of the sparkling wine samples, are shown in
Figure 4.
Esters was the most abundant family of compounds in both types of closures. Levels of some families of volatile compounds—for example alcohols, acids and others—are those with wide differences between the type of closures.
As previously mentioned, volatile esters represent one of the most significant groups of compounds due to their crucial role in shaping the volatile profile [
59]. They contribute to the presence of fruity and floral-like notes in the sparkling wine aroma [
1,
2,
18], and it appears that this is the result of the autolysis of the yeast. Some factors such as the yeast strain or fermentation conditions (temperature, nutrient content, or availability of oxygen) have already been linked to the formation of volatile esters [
1,
3]. Therefore, the type of closure would be related to the preservation of the levels of ester compounds in sparkling wine, improving the shelf life of the product. The percentage of the ester area is similar in both closures. Among the esters present in the analyzed sparkling wines, methyl 2,4-dimethylhexanoate was the most abundant ester and was only detected in screw-cap closures. Methyl esters in wine are related to yeast fermentation [
66]. Ethyl acetate and ethyl esters with a high molecular weight, such as ethyl octanoate (floral), ethyl hexanoate (fruity), ethyl decanote (floral), ethyl arachidate and diethyl succinate (overripe), were detected in all samples. As noted by [
66], these compounds can exhibit a synergistic effect, even when present in small quantities. In the case of the effect of the type of closure, ref. [
18] described the presence of greater amounts of several ethyl esters in sparkling wine closured using cork stoppers instead of micro-agglomerated stoppers. Diethyl succinate is regarded as a marker, primarily associated with the duration of cava storage in the cellar. This compound is defined as fruity or floral by tasters and is a marker of the evolution of sparkling wine because it is a post-fermentative volatile formed during the aging of sparkling wine in contact with lees from second fermentation [
1,
23]. Overall, diethyl succinate, TDN and hexanol seem to be compounds inherent in the bouquet of long-aged sparkling wines [
24,
25]. The first two were detected in higher amounts in bottles with cork stoppers than screw-cap closures (
Table A1,
Appendix A). Finally, sparkling wine acetates like ethyl acetate decrease along the ageing time of sparkling wine in contact with lees [
25]. However, a higher amount of this acetate was detected in products with screw-cap closures.
In the case of cork closures, the second-most-abundant family of compounds was alcohols, which are related to yeast metabolism [
1]. Sparkling wine alcohols such as isoamyl alcohol and phenylethyl alcohol were detected in bottles with both closures. The latter has influence on the sweet, rose and honey aroma structure of sparkling wines [
26,
67,
68], and isoamyl alcohol can influence wine aroma by adding “alcohol” and “nail polish” notes [
59]. Moreover, 1-hexanol was identified with both stoppers but in higher amounts with screw-cap closures. In the case of sparkling wines elaborated by traditional method, the alcohols augmented after alcoholic fermentation and remained almost constant after second fermentation and throughout aging. However, in the case of certain alcohols like 1-hexanol, they tended to increase, serving as a suitable ageing marker, as mentioned previously. Ref. [
18] described that the levels of 1-hexanol were also significantly influenced by the type of closure in a sparkling wine. In accordance with these authors, the lower levels of 1-hexanol in sparkling wine sealed with cork stoppers may be due to its oxidation to 3-hexenal, but this aldehyde was not detected in the volatile composition of our samples under our experimental conditions. Additionally, 1-butanol was only obtained in screw-cap samples. According to [
69], 1-hexanol contributes to the aroma of just-cut grass, and 1-butanol contributes to the medicinal aroma of wine.
Concerning the acids, the most common were detected in both groups. However, the most representative, like octanoic and decanoic acid, have been detected in higher amounts in sparkling wines closured using screw caps. These acids, depending on the concentration, can have a negative role in the development of wine sensory profiles [
26,
67]. Among ethers compounds, oxirane, 2-(1,1-dimethylethyl)-3-methyl- was identified in both groups of closures but was more abundant in screw-cap closures (
Table A1,
Appendix A). Furthermore, 1-Methyl-1-silacyclopentan-1-ol was only detected in screw-cap closures. Alkanes and ketones were determined in both closures, with results from cork stoppers being slightly higher than for screw caps.
Aziridinylethylamine and hydroxyurea were presented only in screw-cap closures in higher amounts. This first is related to a fishy flavor [
70]. Corlumine, N-Methylcalycotomine, phenol and emulphor were detected in screw-cap closures, while 12-O-Acetylingol 8-tiglate, 2-Myristynoyl pantetheine, 6,7-Dimethoxy-1,4-dimethyl-1,3-quinoxalinedithione and longifolenaldehyde were identified in cork stoppers. Also, the highest amount of dimethylamine or a volatile amine with secondary amino groups [
71] was detected in screw-cap closures.
3.2. Effect of the Type of Closure through Compositional Data Analysis
Based on the results obtained, the type of closure used during the second fermentation can influence the aroma composition of the resulting sparkling wine. This impact may result from the desorption of volatile compounds from the closures into the wine [
8,
18] or be related to the level of oxidation. In the case of still wine, ref. [
72] described that wines sealed with different types of closures for four years differed significantly in their content of some volatile chemicals, such as 1-butanol, 2-phenylethanol, 2-nonanol and ethyl decanoate. Additionally, ref. [
73] discovered that eight volatile compounds (isoamyl acetate, ethyl decanoate, nonanoic acid, n-decanoic acid, undecanoic acid, 2-furancarboxylic acid, dodecanoic acid, and phenylacetaldehyde) played a role in differentiating wine closures and were linked to the oxidation level of Cabernet Sauvignon wines.
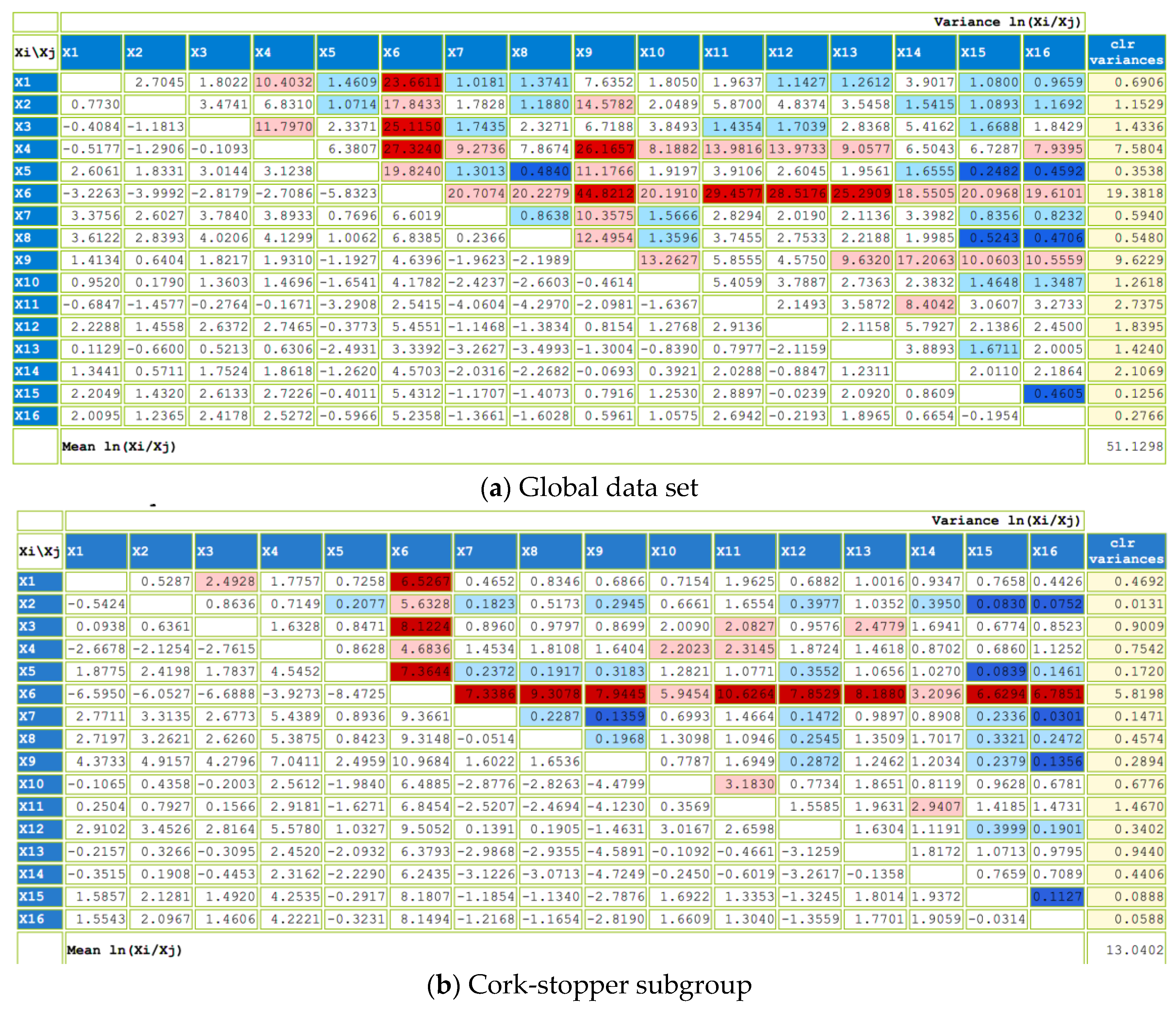
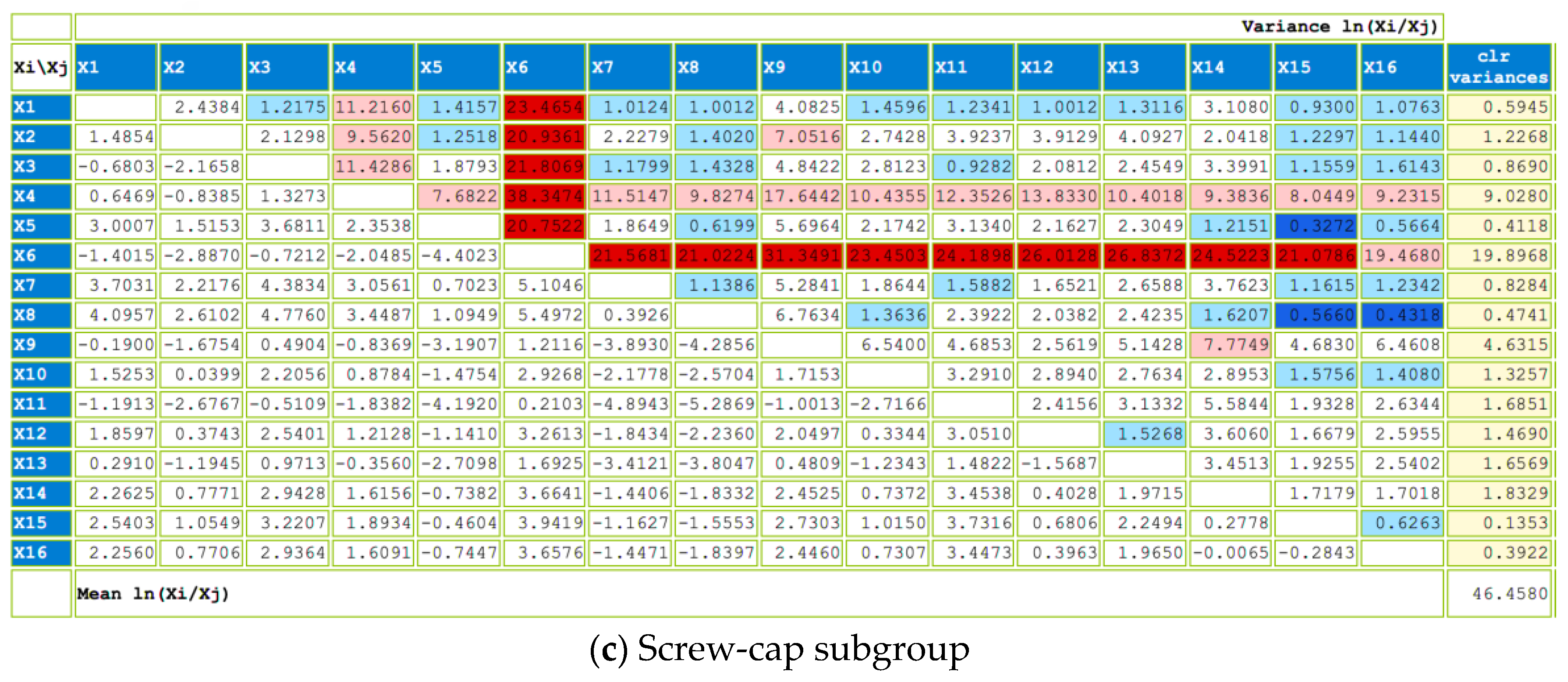
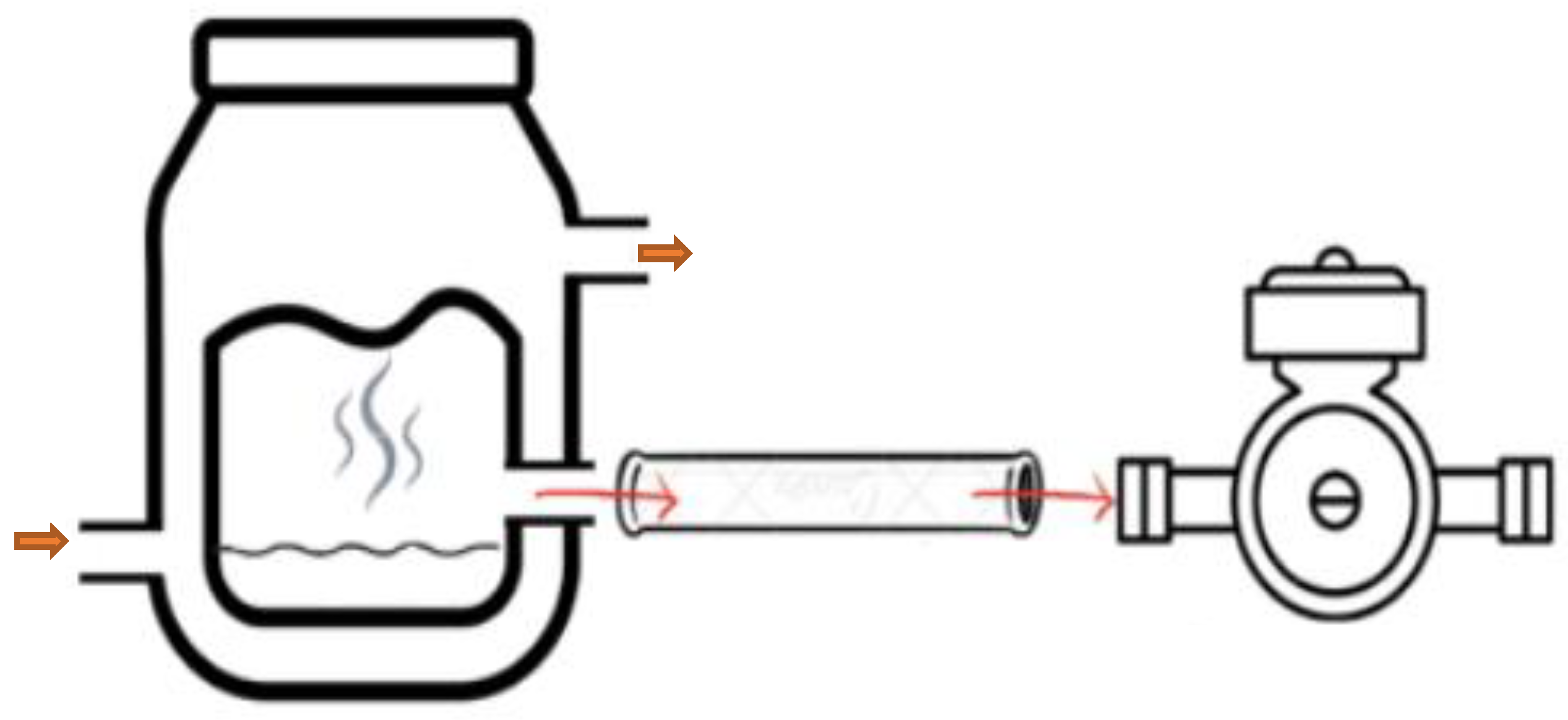
The impact of six different closures used in bottle aging after second fermentation on the volatile composition of sparkling wine has been evaluated using the compositional analysis of a data matrix that included the compounds extracted by HP-SPME. First, two groups were analyzed: cork stoppers (cork 1 and cork 2) and screw-cap closures (from CC1 to CC4).
We used a compositional data set with n = 37 stoppers (n = 13 group cork and n = 24 group screw cap) and D = 57 volatile compounds. Due to the substantial number of zeros, we first selected the volatile compounds detected in more than five stoppers—that is, with a percentage of zeros below 80%. This reduced the number of parts of our composition to D = 16, and their names are listed in
Table 3. Additionally, the corresponding percentage of replaced zeros and the notation used in the figures to avoid the lengthy names of certain compounds are also provided. According to the nature of these zeros, the log-ratio EM imputation algorithm was applied [
52].
The values of the whole center and subgroups center (cork and screw-cap closures) are presented in
Table A2 (
Appendix A). Overall, the most abundant part is ethyl hexanoate
, which is also observed in the screw-cap subgroup. However, in the cork subgroup, the most abundant part is ethyl octanoate
. Both are ester compounds—the most abundant family of compounds detected in sparkling wine samples (
Figure 5 and
Figure 6). To better understand these results, a geometric-mean bar plot comparing the compositional center of the entire sample with the compositional center of cork stoppers and screw-cap subgroups is shown in
Figure 5. The volatile compounds with a larger relative difference compared to the global center were dimethylamine
and ethyl octanoate
. It is clear from
Figure 5 that the proportion amount of dimethylamine
was higher in screw-cap closures than cork stoppers relative to the entire sample. Dimethylamine primarily forms through the decarboxylation of amino acids at various stages of vinification. Since high concentrations of amines are difficult to remove, it is crucial to monitor their production. Amines with secondary amino groups, like dimethylamine, can lead to the formation of nitrosamines or other harmful substances [
41]. On the contrary, ethyl octanoate
was higher in the cork-stopper group. This ester compound is responsible for fruity and floral-like aromas, as commented previously. A similar pattern was observed for other volatile compounds such as isoamyl alcohol
, lactamide
and 4-(2,3,6-Trimethylphenyl)-1,3-butadiene
. On the other hand, the bar plot suggests that the values of octanoic acid
and alpha-ionone
are very similar in the two subgroups.
A non-parametric MANOVA [
74] contrast applied to the clr-scores data set confirms significant differences (
p < 0.05) between the centers of cork-stopper and screw-cap closures. In this case, the homogeneity cannot be accepted, but the screw-cap group has large dispersion and a large number of samples, and the test became conservative [
75]. For comparing the centers of the two groups, the bootstrap 95% percentile interval for each part is provided (
Figure 6).
Figure 6 shows that only intervals of the parts 1,1,5-Trimethyl-1,2-dihydronaphthalene
and octanoic acid
have a zero value (horizontal dashed line), which means no differences between the two types of closures. All the other parts do not contain the zero value indicating differences. The larger ones in the dimethylamine
and ethyl octanoate
parts are also observed in
Figure 5.
The variability of the data is displayed in
Figure A1 (
Appendix A), within the variation matrix, illustrating both the variance and the mean of each pairwise log-ratio. The highest variances are highlighted using a red-shaded background, while the lowest are indicated with a blue-shaded background. Additionally, the total variance and the variance of each clr component are also provided. Globally, the total variance is 51.13. Screw-cap closures exhibit higher total variance (46.46) compared to cork stoppers (13.04), showing heterogeneity between groups. Examining the pairwise log-ratios, it becomes evident that both globally and within each subgroup, the log-ratios with the highest variance are those containing the dimethylamine
part. However, in the screw-cap subgroup, log-ratios containing the carbon dioxide
part also display high variability. Furthermore, in both subgroups and globally, the log-ratios involving phenylethyl alcohol
and alpha-ionone
exhibit low variability.
Figure 7 shows the compositional clr-biplot, with cork closures in red and screw closures in blue, simplifying a comparison between the subgroups. The biplot retains a relevant portion of the total variability (78.34%), ensuring the high quality of the representation. In
Figure 7, the conclusion mirrors earlier findings, highlighting a clear separation between subgroups and the higher variability of the screw-cap closures group (blue). We can see the volatile compounds’ characteristics associated with cork closures, such as
,
and
, as well as other compounds’ characteristics linked to screw-cap closures such as
and
, exhibiting opposite directions relative to the PC1 axis.
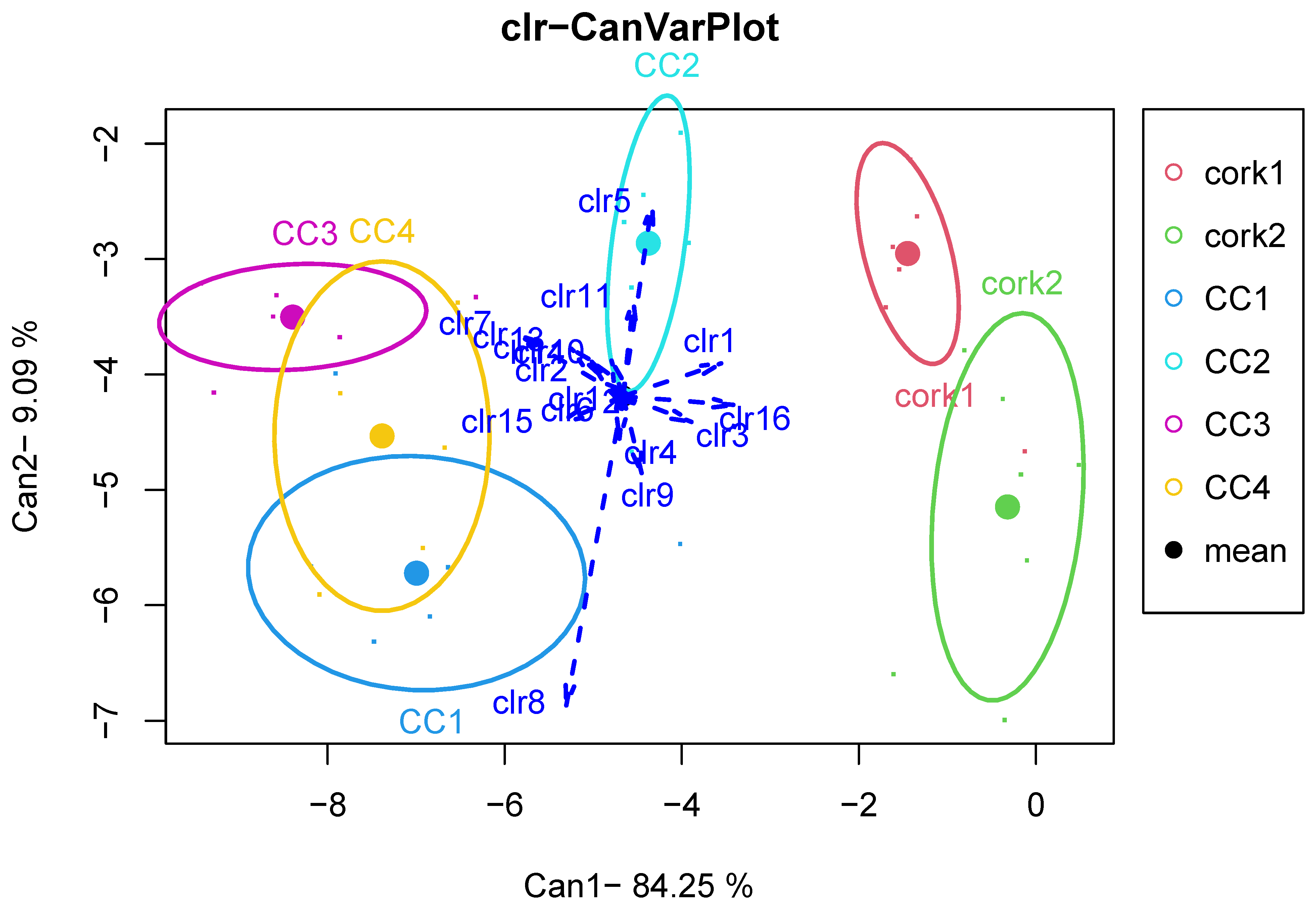
A second compositional data analysis was carried out using all types of stoppers as subgroups (n = 6 for Cork1, CC1, CC2, CC3, CC4 and n = 7 for Cork2). The centers of each subgroup, displayed in
Table A3 (
Appendix A), and the geometric-mean bar plot in
Figure 8, allow us to compare the compositional mean of the entire sample with the compositional mean of each type of closure.
Broadly speaking,
Figure 8 shows a similar pattern to
Figure 5. That is, the pattern stated between the two groups of closures in
Figure 5 is, respectively, reproduced in each set of subgroups. Indeed, the non-parametric MANOVA test applied separately to the subgroups does not allow us to confirm significant differences between the two cork-stopper groups (
p = 0.148) and the four screw-cap groups (
p = 0.377). In this case, the robustness of the test is guaranteed because we have balanced designs [
75]. A visual inspection of
Figure 9, where the first canonical variate retains 84.25% of the variability, allows us to perceive some differences between the agglomerated cork stopper with 31 mm of diameter (cork1) and the agglomerated cork stopper with 32 mm of diameter (cork2), as well as between Saranex (CC2) and the other screw-cap groups—polyethylene screw cap (CC1), Daraform (CC3) and Saranex plus araldite (CC4)—although a larger dataset is needed to confirm the potential differences.