Ultrasonic Pretreatment Combined with Microwave-Assisted Hydrodistillation for Extraction of Essential Oil from Melaleuca bracteata ‘Revolution Gold’ Leaves Scales Induced by Cellulase-Inorganic Salt and Its Anti-Fungal Activity
Abstract
:1. Introduction
2. Material and Method
2.1. Materials
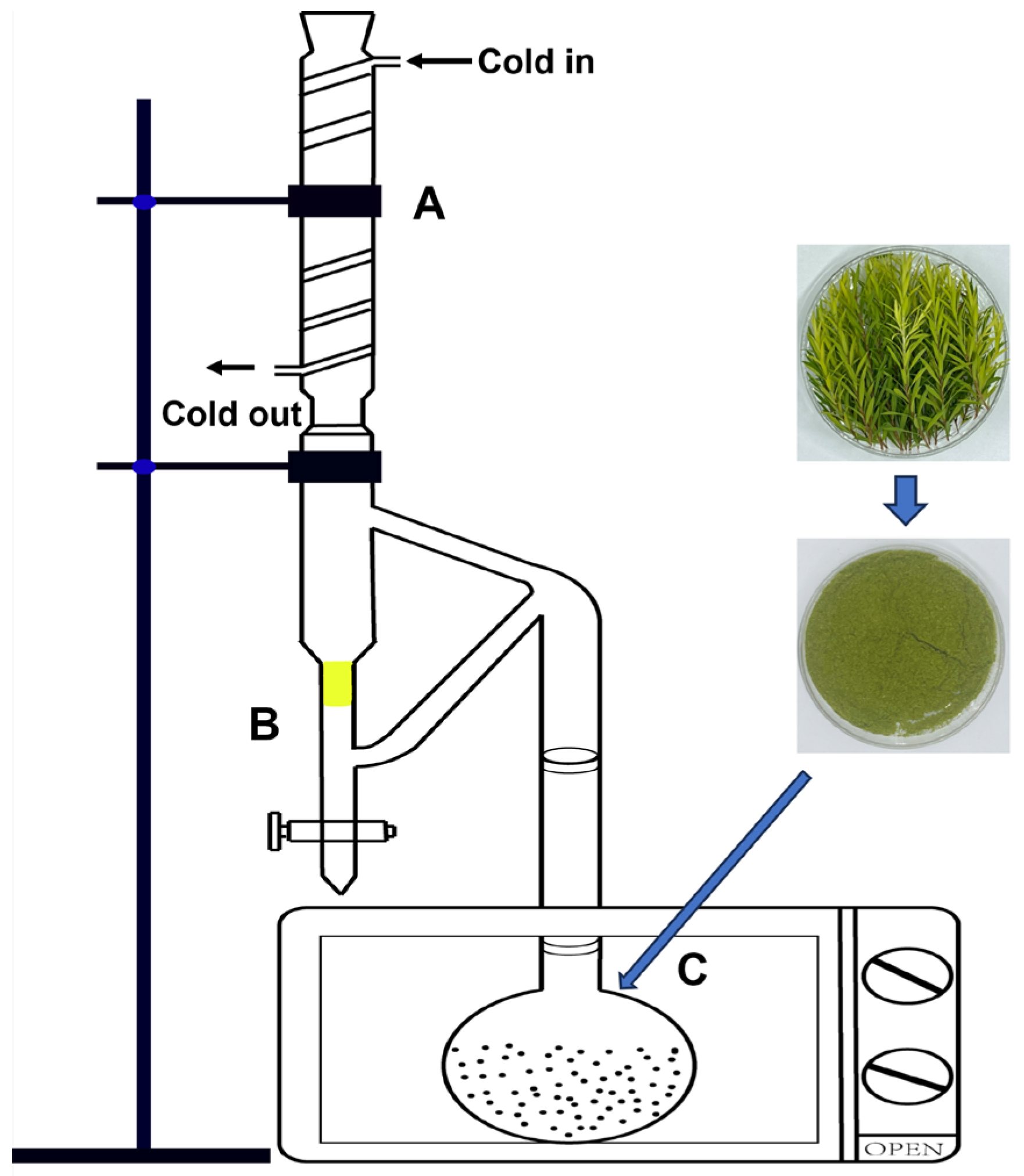
2.2. Essential Oil Extraction
2.3. Box–Behnken Design Optimizes the Extraction Rate of Essential Oils
2.4. Essential Oil Composition and Analysis
2.5. Fungal Inhibition Activity
2.5.1. Activation of Strains
2.5.2. Fungal Inhibition of Essential Oil
3. Results and Discussion
3.1. Influence of Various Factors on Extraction Rate
3.2. The Extraction Rate of Essential Oil Was Optimized by Response Surface
3.3. Analysis of Essential Oil by Gas Chromatography–Mass Spectrometry
3.4. Comparison with Reference Techniques
3.5. Study on Antifungal Activity of Essential Oils
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Carlier, J.; Robert, S.; Roussel, V.; Chilin-Charles, Y.; Lubin-Adjanoh, N.; Gilabert, A.; Abadie, C. Central American and Caribbean population history of the Pseudocercospora fijiensis fungus responsible for the latest worldwide pandemics on banana. Fungal Genet. Biol. 2021, 148, 103528. [Google Scholar] [CrossRef]
- Gebhardt, C. The historical role of species from the Solanaceae plant family in genetic research. Theor. Appl. Genet. 2016, 129, 2281–2294. [Google Scholar] [CrossRef] [PubMed]
- Seshadri, V.D.; Balasubramanian, B.; Al-Dhabi, N.A.; Esmail, G.A.; Arasu, M.V. Essential oils of Cinnamomum loureirii and Evolvulus alsinoides protect guava fruits from spoilage bacteria, fungi and insect (Pseudococcus longispinus). Ind. Crops Prod. 2020, 154, 112629. [Google Scholar] [CrossRef]
- Alakonya, A.E.; Kimunye, J.; Mahuku, G.; Amah, D.; Uwimana, B.; Brown, A.; Swennen, R. Progress in understanding Pseudocercospora banana pathogens and the development of resistant Musa germplasm. Plant Pathol. 2018, 67, 759–770. [Google Scholar] [CrossRef]
- Gaikwad, S.S.; Rumma, S.S.; Hangarge, M. Classification of Fungi Effected Psidium Guajava Leaves Using ML and DL Techniques BT—Computer Vision and Machine Intelligence Paradigms for SDGs; Kannan, R.J., Thampi, S.M., Wang, S.-H., Eds.; Springer Nature: Singapore, 2023; pp. 69–81. [Google Scholar]
- Damm, U.; Sun, Y.-C.; Huang, C.-J. Colletotrichum eriobotryae sp. nov. and C. nymphaeae, the anthracnose pathogens of loquat fruit in central Taiwan, and their sensitivity to azoxystrobin. Mycol. Prog. 2020, 19, 367–380. [Google Scholar] [CrossRef]
- Li, M.; Feng, W.; Yang, J.; Gao, Z.; Zhang, Z.; Zhang, W.; Wang, S.; Wang, W.; Gong, D.; Hu, M. First report of anthracnose caused by Colletotrichum siamense on avocado fruits in China. Crop Prot. 2022, 155, 105922. [Google Scholar] [CrossRef]
- Han, F.; Qi, Y.-K.; Zhang, Y.; Qi, J.-H.; Wang, J.-W.; Liu, W.; Zhang, L.W.; Ji, Y.-P.; Wang, Q.-H. First report of jujube anthracnose caused by Colletotrichum siamense in China. Plant Dis. 2023, 107, 2538. [Google Scholar] [CrossRef]
- Krishnan, S.; Kaari, M.; Sawhney, S.; Sheoran, N.; Gautam, R.K.; Mohan Das, M.; Kumar, A. First report of Colletotrichum siamense from Andaman and Nicobar Islands causing anthracnose in chilli. J. Plant Pathol. 2019, 101, 767. [Google Scholar] [CrossRef]
- Fuentes-Aragón, D.; Silva-Rojas, H.V.; Guarnaccia, V.; Mora-Aguilera, J.A.; Aranda-Ocampo, S.; Bautista-Martínez, N.; Téliz-Ortíz, D. Colletotrichum species causing anthracnose on avocado fruit in Mexico: Current status. Plant Pathol. 2020, 69, 1513–1528. [Google Scholar] [CrossRef]
- Aliya, S.S.S.; Nusaibah, S.A.; Mahyudin, M.M.; Yun, W.M.; Yusop, M.R. Colletotrichum siamense and Pestalotiopsis jesteri as potential pathogens of new rubber leaf spot disease via detached leaf assay. J. Rubber Res. 2022, 25, 195–212. [Google Scholar] [CrossRef]
- Eh Teet, S.; Hashim, N. Recent advances of application of optical imaging techniques for disease detection in fruits and vegetables: A review. Food Control. 2023, 152, 109849. [Google Scholar] [CrossRef]
- Duhan, J.S.; Kumar, R.; Kumar, N.; Kaur, P.; Nehra, K.; Duhan, S. Nanotechnology: The new perspective in precision agriculture. Biotechnol. Rep. 2017, 15, 11–23. [Google Scholar] [CrossRef]
- Tudi, M.; Daniel Ruan, H.; Wang, L.; Lyu, J.; Sadler, R.; Connell, D.; Chu, C.; Phung, D.T. Agriculture Development, Pesticide Application and Its Impact on the Environment. Int. J. Environ. Res. Public Health 2021, 18, 1112. [Google Scholar] [CrossRef]
- Garrigou, A.; Laurent, C.; Berthet, A.; Colosio, C.; Jas, N.; Daubas-Letourneux, V.; Jackson Filho, J.-M.; Jouzel, J.-N.; Samuel, O.; Baldi, I.; et al. Critical review of the role of PPE in the prevention of risks related to agricultural pesticide use. Saf. Sci. 2020, 123, 104527. [Google Scholar] [CrossRef]
- Luo, L.; Dong, L.; Huang, Q.; Ma, S.; Fantke, P.; Li, J.; Jiang, J.; Fitzgerald, M.; Yang, J.; Jia, Z.; et al. Detection and risk assessments of multi-pesticides in 1771 cultivated herbal medicines by LC/MS-MS and GC/MS-MS. Chemosphere 2021, 262, 127477. [Google Scholar] [CrossRef] [PubMed]
- Hou, T.; Sana, S.S.; Li, H.; Xing, Y.; Nanda, A.; Netala, V.R.; Zhang, Z. Essential oils and its antibacterial, antifungal and anti-oxidant activity applications: A review. Food Biosci. 2022, 47, 101716. [Google Scholar] [CrossRef]
- Álvarez-Martínez, F.J.; Barrajón-Catalán, E.; Herranz-López, M.; Micol, V. Antibacterial plant compounds, extracts and essential oils: An updated review on their effects and putative mechanisms of action. Phytomedicine 2021, 90, 153626. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Lin, Y.; Qiu, Z.; Xiang, X.; Shao, D.; Li, Y.; Wu, S. Reference gene selection for qRT-PCR normalization of gene expression analysis in Melaleuca bracteata F. Muell. under abiotic stresses and hormonal stimuli. Sci. Hortic. 2023, 319, 112184. [Google Scholar] [CrossRef]
- Padalia, R.C.; Verma, R.S.; Chauhan, A.; Goswami, P.; Verma, S.K.; Darokar, M.P. Chemical composition of Melaleuca linarrifolia Sm. from India: A potential source of 1,8-cineole. Ind. Crops Prod. 2015, 63, 264–268. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Y.; Feng, Y.; Du, S.; Jia, L. Contact Toxicity and Repellent Efficacy of Essential Oil from Aerial Parts of Melaleuca bracteata and its Major Compositions against Three Kinds of Insects. J. Essent. Oil Bear. Plants 2021, 24, 349–359. [Google Scholar] [CrossRef]
- Yasin, M.; Younis, A.; Javed, T.; Akram, A.; Ahsan, M.; Shabbir, R.; Ali, M.M.; Tahir, A.; El-Ballat, E.M.; Sheteiwy, M.S.; et al. River Tea Tree Oil: Composition, Antimicrobial and Antioxidant Activities, and Potential Applications in Agriculture. Plants 2021, 10, 2105. [Google Scholar] [CrossRef]
- Bottoni, M.; Milani, F.; Mozzo, M.; Radice Kolloffel, D.A.; Papini, A.; Fratini, F.; Maggi, F.; Santagostini, L. Sub-Tissue Localization of Phytochemicals in Cinnamomum camphora (L.) J. Presl. Growing in Northern Italy. Plants 2021, 10, 1008. [Google Scholar] [CrossRef] [PubMed]
- Raman, G.; Gaikar, V.G. Microwave-Assisted Extraction of Piperine from Piper nigrum. Ind. Eng. Chem. Res. 2002, 41, 2521–2528. [Google Scholar] [CrossRef]
- Franco-Vega, A.; López-Malo, A.; Palou, E.; Ramírez-Corona, N. Effect of imidazolium ionic liquids as microwave absorption media for the intensification of microwave-assisted extraction of Citrus sinensis peel essential oils. Chem. Eng. Process. Process Intensif. 2021, 160, 108277. [Google Scholar] [CrossRef]
- Liu, Z.; Li, H.; Zhu, Z.; Huang, D.; Qi, Y.; Ma, C.; Zou, Z.; Ni, H. Cinnamomum camphora fruit peel as a source of essential oil extracted using the solvent-free microwave-assisted method compared with conventional hydrodistillation. LWT 2022, 153, 112549. [Google Scholar] [CrossRef]
- Hou, K.; Bao, M.; Wang, L.; Zhang, H.; Yang, L.; Zhao, H.; Wang, Z. Aqueous enzymatic pretreatment ionic liquid–lithium salt based microwave–assisted extraction of essential oil and procyanidins from pinecones of Pinus koraiensis. J. Clean. Prod. 2019, 236, 117581. [Google Scholar] [CrossRef]
- Mahmoudi, H.; Marzouki, M.; M’Rabet, Y.; Mezni, M.; Ait Ouazzou, A.; Hosni, K. Enzyme pretreatment improves the recovery of bioactive phytochemicals from sweet basil (Ocimum basilicum L.) leaves and their hydrodistilled residue by-products, and potentiates their biological activities. Arab. J. Chem. 2020, 13, 6451–6460. [Google Scholar] [CrossRef]
- Xie, J.; Chen, Y.; Cai, G.; Cai, R.; Hu, Z.; Wang, H. Tree Visualization by One Table (tvBOT): A web application for visualizing, modifying and annotating phylogenetic trees. Nucleic Acids Res. 2023, 51, W587–W592. [Google Scholar] [CrossRef]
- Chen, F.; Su, X.; Gao, J.; Liu, Y.; Zhang, Q.; Luo, D. A modified strategy to improve the dissolution of flavonoids from Artemisiae Argyi Folium using ultrasonic-assisted enzyme-deep eutectic solvent system. J. Chromatogr. A 2023, 1707, 464282. [Google Scholar] [CrossRef] [PubMed]
- Kapadiya, S.M.; Parikh, J.; Desai, M.A. A greener approach towards isolating clove oil from buds of Syzygium aromaticum using microwave radiation. Ind. Crops Prod. 2018, 112, 626–632. [Google Scholar] [CrossRef]
- Gao, H.; Zhao, R.; Fan, C.; Yang, Y.; Wang, Z.; Gu, H.; Yang, L.; Tian, H.; Zhang, Q.; Zhang, X. An efficient approach to obtain essential oil, oleanoic acid, 3-O-acetyloleanolic acid and betulinic acid from fresh leaves of Melaleuca bracteata F. Muell. “Revolution Gold” using microwave and gravity-assisted solvent-free distillation followed by extra. Ind. Crops Prod. 2023, 203, 117178. [Google Scholar] [CrossRef]
- Gooderham, N.J.; Cohen, S.M.; Eisenbrand, G.; Fukushima, S.; Guengerich, F.P.; Hecht, S.S.; Rietjens, I.M.C.M.; Rosol, T.J.; Davidsen, J.M.; Harman, C.L.; et al. FEMA GRAS assessment of natural flavor complexes: Clove, cinnamon leaf and West Indian bay leaf-derived flavoring ingredients. Food Chem. Toxicol. 2020, 145, 111585. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Wu, S.; Fu, X.; Pan, H. De Novo Biosynthesis of Methyl Cinnamate in Engineered Escherichia coli. J. Agric. Food Chem. 2022, 70, 7736–7741. [Google Scholar] [CrossRef] [PubMed]
- Sosa, L.; Espinoza, L.C.; Valarezo, E.; Bozal, N.; Calpena, A.; Fábrega, M.-J.; Baldomà, L.; Rincón, M.; Mallandrich, M. Therapeutic Applications of Essential Oils from Native and Cultivated Ecuadorian Plants: Cutaneous Candidiasis and Dermal Anti-Inflammatory Activity. Molecules 2023, 28, 5903. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Liu, Z.; Gu, H.; Yang, L.; Liu, T.; Tian, H. Preparation and characterization of a modified Canna starch as a wall material for the encapsulation of methyleugenol improves its antifungal activity against Fusarium trichothecioides. Food Chem. 2023, 433, 137324. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Cai, J.; Chen, H.; Zhong, Q.; Hou, Y.; Chen, W.; Chen, W. Antibacterial activity and mechanism of linalool against Pseudomonas aeruginosa. Microb. Pathog. 2020, 141, 103980. [Google Scholar] [CrossRef]
- Tomazoni, E.Z.; Pauletti, G.F.; da Silva Ribeiro, R.T.; Moura, S.; Schwambach, J. In vitro and in vivo activity of essential oils extracted from Eucalyptus staigeriana, Eucalyptus globulus and Cinnamomum camphora against Alternaria solani Sorauer causing early blight in tomato. Sci. Hortic. 2017, 223, 72–77. [Google Scholar] [CrossRef]




| Run | BBD Experiments | ANOVA | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A (W) | B (min) | C (μmol) | YEO (mL/kg) | Source of Variation | Sum of Squares | Degree of Freedom | Mean Square | F Value | p Value | |||
| 1 | 576 | 50 | 60 | 9.31 | Model | 24.59 | 9 | 2.73 | 13.78 | 0.0011 * | ||
| 2 | 576 | 50 | 60 | 9.82 | A | 1.44 | 1 | 1.44 | 7.29 | 0.0306 | ||
| 3 | 576 | 50 | 60 | 9.73 | B | 3.51 | 1 | 3.51 | 17.71 | 0.0040 | ||
| 4 | 720 | 60 | 60 | 6.98 | C | 0.061 | 1 | 0.06 | 0.31 | 0.5956 | ||
| 5 | 720 | 40 | 60 | 7.92 | AB | 0 | 1 | 0.00 | 0.00 | 1.0000 | ||
| 6 | 720 | 50 | 75 | 6.00 | AC | 4.41 | 1 | 4.41 | 22.25 | 0.0022 | ||
| 7 | 576 | 40 | 45 | 8.11 | BC | 0.20 | 1 | 0.20 | 1.02 | 0.3458 | ||
| 8 | 720 | 50 | 45 | 8.22 | A2 | 2.78 | 1 | 2.78 | 14.02 | 0.0072 | ||
| 9 | 432 | 50 | 45 | 7.63 | B2 | 6.98 | 1 | 6.98 | 35.21 | 0.0006 | ||
| 10 | 576 | 60 | 75 | 6.21 | C2 | 3.70 | 1 | 3.70 | 18.67 | 0.0035 | ||
| 11 | 576 | 50 | 60 | 9.90 | Residual | 1.39 | 7 | 0.19 | ||||
| 12 | 432 | 50 | 75 | 9.60 | Lack of fit | 1.07 | 3 | 0.36 | 4.45 | 0.0917 | ||
| 13 | 576 | 60 | 45 | 6.90 | Pure error | 0.32 | 4 | 0.08 | ||||
| 14 | 432 | 60 | 60 | 7.16 | Cor total | 25.98 | 16 | |||||
| 15 | 576 | 40 | 75 | 8.36 | Credibility analysis of the regression equations | |||||||
| 16 | 432 | 40 | 60 | 8.18 | Index mark | SD | Mean | C.V.% | R2 | Adjust R2 | Predicted R2 | AP |
| 17 | 576 | 50 | 60 | 9.32 | Y | 0.4452 | 8.17 | 5.45 | 0.9466 | 0.8779 | 0.3232 | 9.7009 |
| Number | Retention Time (min) | Compound Name A | RI B | Cas # | Similarity (%) | Area Percentage (%) C |
|---|---|---|---|---|---|---|
| 1 | 3.81 | 2-Hexenal, (E)- | 848 | 006728-26-3 | 98 | 0.02 |
| 2 | 7.69 | O-Cymene | 1017 | 000527-84-4 | 97 | 0.03 |
| 3 | 7.80 | D-Limonene | 1024 | 005989-27-5 | 99 | 0.02 |
| 4 | 7.89 | Benzyl alcohol | 1033 | 000100-51-6 | 95 | 0.03 |
| 5 | 9.46 | Terpinolene | 1083 | 000586-62-9 | 95 | 0.02 |
| 6 | 9.76 | Linalool | 1097 | 000078-70-6 | 97 | 0.76 |
| 7 | 11.18 | Isopulegol | 1146 | 000089-79-2 | 98 | 0.03 |
| 8 | 11.44 | Citronellal | 1153 | 000106-23-0 | 96 | 0.02 |
| 9 | 12.34 | Terpinen-4-ol | 1171 | 000562-74-3 | 90 | 0.19 |
| 10 | 12.65 | P-cymenol | 1181 | 001197-01-9 | 91 | 0.15 |
| 11 | 12.89 | α-Terpineol | 1187 | 010482-56-1 | 87 | 0.90 |
| 12 | 13.23 | Estragole | 1196 | 000140-67-0 | 98 | 0.65 |
| 13 | 14.64 | Citronellol | 1212 | 000106-22-9 | 98 | 0.71 |
| 14 | 15.34 | β-Citral | 1236 | 000106-26-3 | 87 | 0.03 |
| 15 | 16.13 | P-Chavicol | 1254 | 000501-92-8 | 95 | 0.03 |
| 16 | 16.86 | Citral | 1276 | 005392-40-5 | 96 | 0.06 |
| 17 | 19.41 | Methyl geranate | 1326 | 002349-14-6 | 83 | 0.21 |
| 18 | 20.51 | α-Cubebene | 1339 | 017699-14-8 | 98 | 0.02 |
| 19 | 20.74 | 2,6-Octadiene, 2,6-dimethyl- | 1351 | 002792-39-4 | 95 | 0.15 |
| 20 | 20.85 | Chavibetol | 1362 | 000501-19-9 | 98 | 0.79 |
| 21 | 21.60 | α-Copaene | 1376 | 1000360-33-0 | 99 | 0.11 |
| 22 | 21.97 | Methyl cinnamate | 1388 | 000103-26-4 | 95 | 12.62 |
| 23 | 23.14 | Methyleugenol | 1408 | 000093-15-2 | 98 | 76.53 |
| 24 | 23.39 | Caryophyllene | 1420 | 000087-44-5 | 96 | 0.55 |
| 25 | 23.80 | β-Cubebene | 1425 | 013744-15-5 | 93 | 0.06 |
| 26 | 24.16 | 10 s,11 s-Himachala-3(12),4-diene | 1437 | 060909-28-6 | 86 | 0.02 |
| 27 | 24.97 | Alloaromadendrene | 1463 | 025246-27-9 | 99 | 0.08 |
| 28 | 25.48 | γ-Cadinene | 1470 | 039029-41-9 | 83 | 0.03 |
| 29 | 25.60 | γ-Muurolene | 1477 | 030021-74-0 | 98 | 0.08 |
| 30 | 25.76 | D-Germacrene | 1487 | 023986-74-5 | 98 | 0.66 |
| 31 | 26.45 | Methylisoeugenol | 1492 | 000093-16-3 | 96 | 0.08 |
| 32 | 26.56 | α-Muurolene | 1493 | 031983-22-9 | 99 | 0.06 |
| 33 | 27.15 | α-Amorphene | 1495 | 000483-75-0 | 97 | 0.06 |
| 34 | 27.57 | Calamenene | 1511 | 000483-77-2 | 96 | 0.66 |
| 35 | 27.98 | Cadinadiene-1,4 | 1524 | 016728-99-7 | 98 | 0.10 |
| 36 | 29.31 | Elemicin | 1558 | 000487-11-6 | 97 | 0.35 |
| 37 | 30.26 | Espatulenol | 1566 | 006750-60-3 | 99 | 0.27 |
| 38 | 31.57 | Ledol | 1580 | 000577-27-5 | 98 | 0.05 |
| 39 | 33.99 | α-Cadinol | 1651 | 000481-34-5 | 99 | 0.18 |
| 40 | 36.91 | Methyl tri-O-methylgallate | 1669 | 001916-07-0 | 98 | 0.42 |
| 41 | 38.27 | Benzyl Benzoate | 1753 | 000120-51-4 | 93 | 0.03 |
| Total | 97.82 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.; Zhang, X.; Zeng, F.; Chang, J.; Liu, Z. Ultrasonic Pretreatment Combined with Microwave-Assisted Hydrodistillation for Extraction of Essential Oil from Melaleuca bracteata ‘Revolution Gold’ Leaves Scales Induced by Cellulase-Inorganic Salt and Its Anti-Fungal Activity. Separations 2024, 11, 147. https://doi.org/10.3390/separations11050147
Huang Y, Zhang X, Zeng F, Chang J, Liu Z. Ultrasonic Pretreatment Combined with Microwave-Assisted Hydrodistillation for Extraction of Essential Oil from Melaleuca bracteata ‘Revolution Gold’ Leaves Scales Induced by Cellulase-Inorganic Salt and Its Anti-Fungal Activity. Separations. 2024; 11(5):147. https://doi.org/10.3390/separations11050147
Chicago/Turabian StyleHuang, Yan, Xiaonan Zhang, Fajian Zeng, Jinmei Chang, and Zhiwei Liu. 2024. "Ultrasonic Pretreatment Combined with Microwave-Assisted Hydrodistillation for Extraction of Essential Oil from Melaleuca bracteata ‘Revolution Gold’ Leaves Scales Induced by Cellulase-Inorganic Salt and Its Anti-Fungal Activity" Separations 11, no. 5: 147. https://doi.org/10.3390/separations11050147
APA StyleHuang, Y., Zhang, X., Zeng, F., Chang, J., & Liu, Z. (2024). Ultrasonic Pretreatment Combined with Microwave-Assisted Hydrodistillation for Extraction of Essential Oil from Melaleuca bracteata ‘Revolution Gold’ Leaves Scales Induced by Cellulase-Inorganic Salt and Its Anti-Fungal Activity. Separations, 11(5), 147. https://doi.org/10.3390/separations11050147






