3.1. The Determination of the Optimum Amount of Hydrophobic Fumed Silica
The inclusion of hydrophobic fumed silica serves a dual purpose in the context of our study. Firstly, it prevents the agglomeration of finer ADP particles, thereby ensuring the uniform dispersion of ADP throughout the fire extinguishing agent. Additionally, it enhances the flowability of ADP, facilitating its effective deployment. However, it is worth mentioning that the impact of fumed silica additions on the UDPA requirements for flame extinction was found to be negligible, as reported by Hamins et al. [
29]. Similarly, Rosser et al. [
30] observed that fumed silica did not exhibit sufficient inhibitory properties towards hydrocarbon combustion.
In our investigation, the extinguishing agent samples primarily rely on ADP as the active ingredient responsible for fire suppression. Consequently, the inclusion of fumed silica leads to a reduction in the mass fraction of ADP, as the amount of fumed silica increases. However, it is advisable to incorporate a small quantity of fumed silica, provided that the extinguishing agent satisfies the hydrophobic criteria. In our experimental setup, we prepared samples containing varying proportions of hydrophobic fumed silica (ranging from 2.0 to 10.0 wt.%), as detailed in
Table 1.
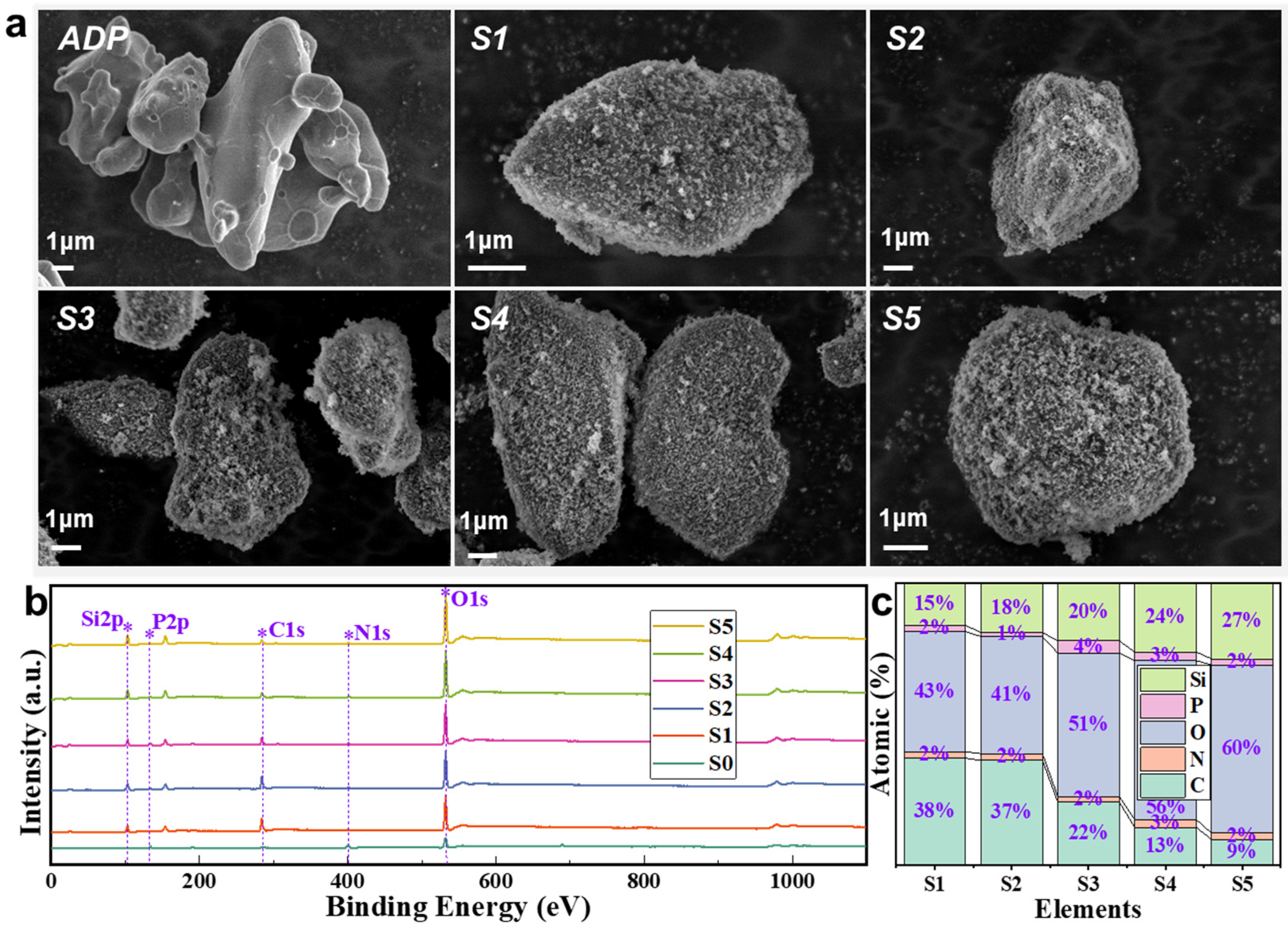
The microscopic morphology images of various samples are presented in
Figure 5a. The pulverized ADP particles are about 2–5 microns in size and exhibit an ellipsoidal shape with a smooth surface and no attachment. The fine ADP has a large specific surface energy, and the particles agglomerate under the action of electrostatic and van der Waals forces [
31]. The attached fine fumed silica particles in Samples S1–S5 are distinctly visible, giving rise to a ‘core-shell’ configuration. In order to ascertain the fumed silica content in the different samples, an XPS analysis was conducted, as depicted in
Figure 5b,c, owing to the varying incorporations of hydrophobic fumed silica among the samples. ADP serves as the primary substrate for powder preparation, thereby resulting in the manifestation of peaks corresponding to P 2p, C 1s, N 1s, and O 1s in all the samples. The pronounced characteristic of the powder samples impregnated with hydrophobic fumed silica is the presence of the Si element as an augmentation to the pure ADP powder. Notably, in comparison to the pure ADP powder, the other samples exhibit an additional peak (Si 2p) at 101.98 eV [
32]. The Si elemental content on the particle surface in Samples S1–S5 escalates from 15% to 27% with the inclusion of hydrophobic fumed silica. The discrepancy in the Si elemental content between the samples lucidly demonstrates the variability in the quantities of hydrophobic fumed silica adhesions on their surfaces.
Firstly, we determined the water contact angle of the different samples. In
Figure 6, the water contact angle increases from 113.45° to 126.48° with an increase in fumed silica’s mass fraction from 2% to 6%. The hydrophobicity of the extinguishing agent doesn’t change much as the mass fraction of fumed silica increases from 6% to 10%. It can be obtained that the hydrophobicity of the UDPA reaches a good state when the mass fraction of fumed silica is above 6%.
Next, the flowability of the above five samples were studied. There is no clear definition of powder flowability and no uniform evaluation method. The common evaluation method is basically to test a single physical property of powders, such as the angle of repose, plate angle, compressibility, etc., to evaluate the flowability of powders based on the performance [
33]. Many testing methods exist, but not every method is suitable for UDPAs, so it is necessary to compare and select the evaluation method that is most suitable. The Carr flow index method is a more comprehensive method for evaluating powder flowability [
34]. Each performance test result is assigned an index with a maximum value of 25 based on a measurement of four sub-indices, which are the repose angle, compressibility, plate angle, and uniformity. The angle of repose refers to the angle formed by the free surface of the powder accumulation layer in static equilibrium with the horizontal plane. The smaller the angle of repose, the better the flowability of the powders. The ratio of the difference between the vibrational density and the bulk density of the sample to the vibrational density is the compressibility. The smaller the degree of compression, the better the flowability of the powder. The flat plate angle refers to the average of the angle between the free surface of the powders on the plate and the plate and the angle after being vibrated. The smaller the plate angle, the more mobile the powder. Uniformity is the ratio of particle diameter D
60 to D
10 and can be used to determine the size of mutual agglutination. In order to evaluate the flowability of the powder in a comprehensive way, the total value is obtained by adding points as the Carr flow index. The higher the value, the better the powder’s flowability.
Table 2 gives the measured data for the angle of repose, compression, flatness angle, and homogeneity of the five samples, as well as the Carr flow index obtained. It can be directly obtained from
Figure 7 that the S3 and S4 samples have the highest Carr flow index and the best flowability of the powder, i.e., the best flowability of the extinguishing agent with a 6% and 8% mass fraction of fumed silica added. In combination with the water contact angle, the amount of fumed silica added should be as small as possible to ensure hydrophobicity, and the optimum amount of fumed silica added was finally determined to be 6 wt.%. When the level of fumed silica additive is insufficient to fully cover the surface of ADP particles, the ability of fumed silica particles to act as separators for the ADP particles is compromised. This, in turn, allows for the interaction force between ADP particles to continue being chiefly determined by direct contact, thereby impeding enhancements in the flowability of the extinguishing agent powder. Both Anthony [
35] and Shinohara [
36] have demonstrated that particles with irregular shapes have a propensity to interlock, consequently leading to increased internal friction.
Lattice spacing within the ADP particles is expanded when uniformly adhered with appropriate fumed silica additives, resulting in a reduced interaction force between the particles [
27,
37]. Additionally, the hydrophobic fumed silica particles adhered to the surface of ADP particles create an effect akin to having a ‘thin film’ covering the particles, thereby performing the function of a ‘lubricant’. This ‘lubricant’ ultimately diminishes the mutual friction occurring on the surface of ADP particles, thus effecting an improvement in the fluidity of the powder fire extinguishing agent.
The excessive addition of fumed silica leads to surplus silica particles being trapped in the interstices between the ADP particles. These excess fumed silica particles form branched chains and bridges among the ADP particles, impeding the relative movement of ADP particles. Thus, inter-particle flow hindrance results since the ADP particles become tightly interconnected through chains or bridges that are challenging to disengage, thus impairing flowability. Consequently, the optimal flow properties of the powder are observed when a 6% mass fraction of fumed silica is added, thereby reducing inter-particle cohesion and dynamic flow resistance.
3.2. Determination of Optimum Amount of Three Functional Additives to Promote Flowability of UDPA
Based on the determination of 6% hydrophobic fumed silica as the most suitable addition, perlite, magnesium stearate, and bentonite were proposed to be further added as functional additives to improve the flowability of the UDPA. The mass fraction of functional additives in different samples is presented in
Table 3. The P, M, and B in the samples stand for perlite, magnesium stearate, and bentonite, respectively.
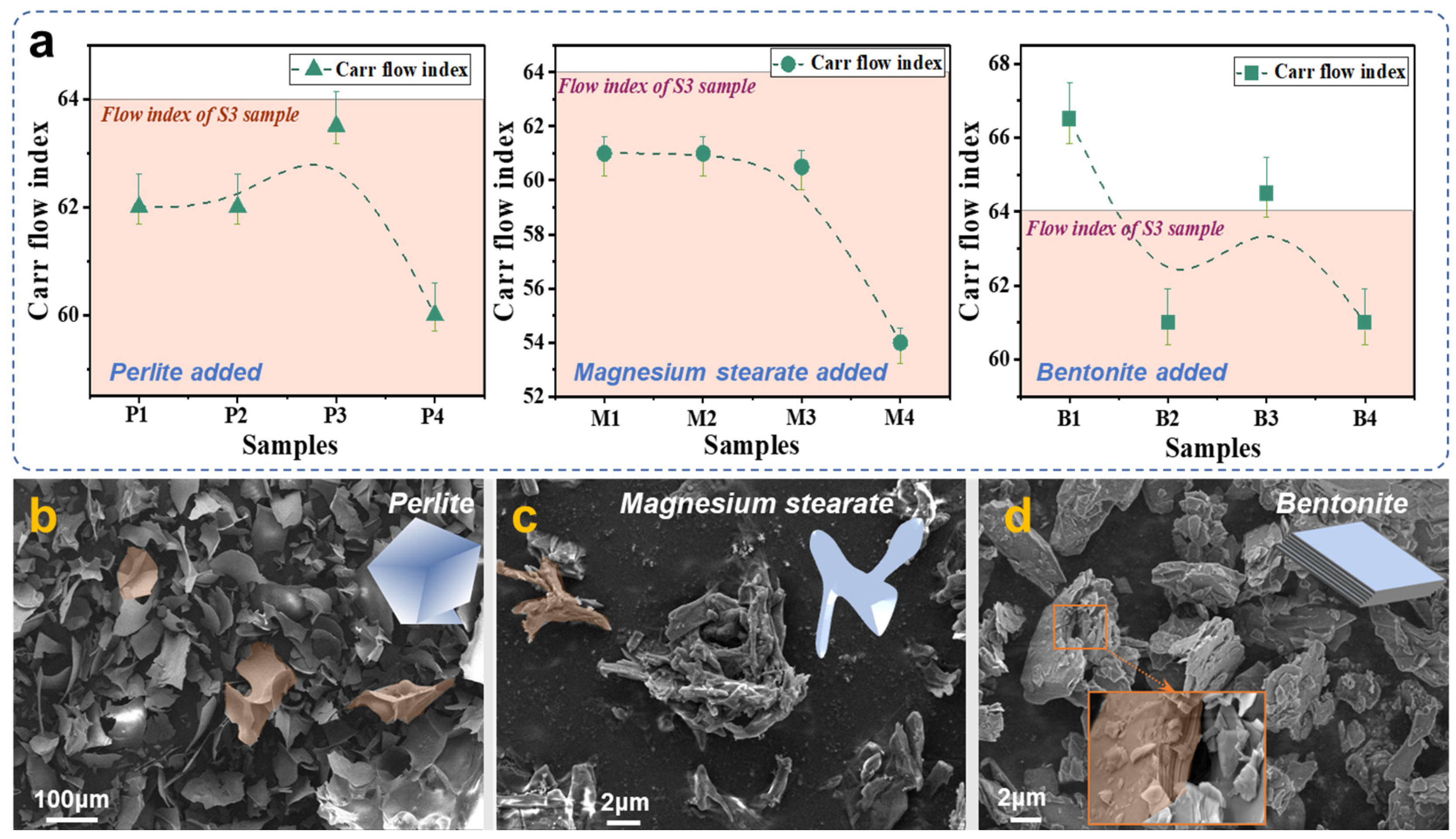
As shown in
Figure 8a, the deduced Carr flow index varies with the different mass fraction of perlite added.
Table 4 shows the different indexes to characterize the flowability of powder samples. In the case of a single index, the value is not monotonically increasing or decreasing. The P3 sample, which has 6 wt.% fumed silica and 1.0 wt.% perlite added, has the highest Carr flow index and the best flowability. However, the Carr flow index of the P3 sample is lower than that of the S3 sample.
For the samples with magnesium stearate added, as the mass fraction of magnesium stearate increases, the repose angle increases and the plate angle decreases, shown in
Table 5. The regularity of the compressibility and uniformity was not obvious, but extreme points were observed in all M4 samples. Powder samples became less flowable as magnesium stearate was added, resulting in a decreasing Carr flow index. It is also found that the Carr flow index of the M1 to M4 samples is lower than that of S3, which indicates that the addition of magnesium stearate reduced the flowability of the powder samples instead. The results suggest that magnesium stearate may not be suitable as a functional additive to promote the flowability of ADP powder.
Table 6 shows the flow and fluidization behaviors of samples with bentonite added, including the indexes of the repose angle, compressibility, plate angle, and uniformity. The Carr flow index was deduced from the above indexes. Statistically, the compressibility trend is the closest to the Carr flow index trend, with B1 having the highest flowability and B3 having the second highest. The Carr flow index is shown to decrease overall as more bentonite is added, indicating that the flowability decreases. The B1 sample, which had 6 wt.% fumed silica and 0.5 wt.% bentonite added, has the highest Carr flow index and the best flowability. Comparing with the Carr flow index of the S3 sample, the B1 and B3 samples have higher indexes than that of the S3 sample. The result shows that the optimum mass fraction of bentonite added is 0.5%, while more might decrease the flowability of ADP powders.
Figure 8b–d illustrate the micro-morphological traits of the three functional additives—perlite, magnesium stearate, and bentonite. Perlite, in its original state, exhibits a porous nature and assumes a fragmented form with rhombic angles upon comminution. The introduction of such a structural functional additive curtails the slippage of ADP particles against each other, thereby mitigating the flowability of ADP powder. Magnesium stearate particles are minute, approximately 2 μm, and tend to agglomerate, indicating pronounced van der Waals forces and inter-particle cohesion. This phenomenon possibly accounts for the absence of flow-promoting characteristics upon its integration into ADP powders. Conversely, bentonite, with a particle size of roughly 5 μm, demonstrates a more dispersed and notably flaky structure. Coupled with the assessment of the Carr flow index measurement results, it becomes apparent that additives characterized by a flaky structure circumvent the compromised flowability arising from the irregular shape of the ADP particles. Nevertheless, there lies a threshold for the quantity of functional additives, as an excessive amount may exert detrimental effects on flowability owing to increased friction. Therefore, the incorporation of 0.5 wt.% bentonite into ADP allows for the observation of an optimal flow effect.
3.3. The Experiments of Fire Extinguishing and Acute Inhalation Toxicity
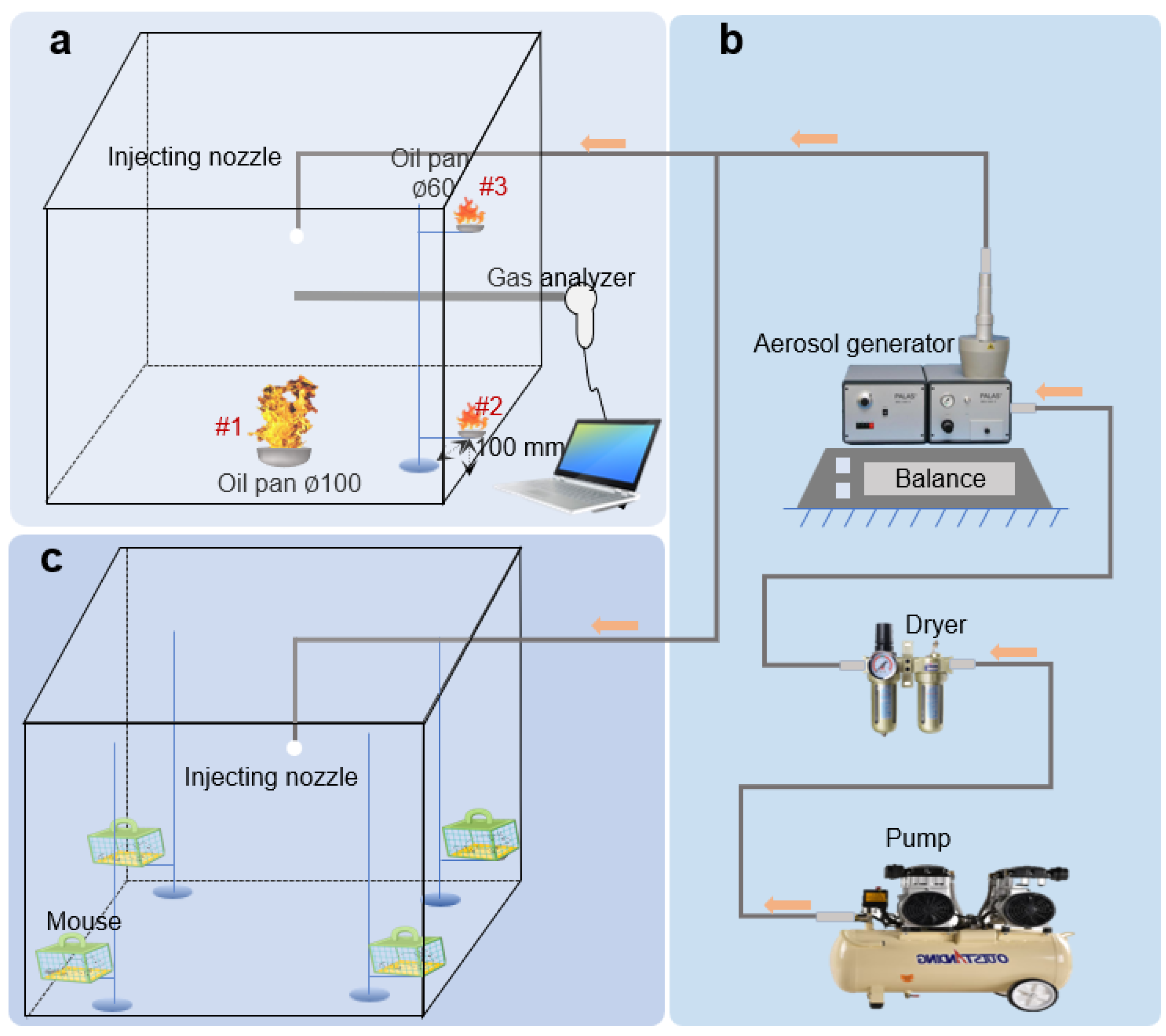
Sample B1 exhibited superior flowability in all the samples and was utilized for fire suppression as well as acute inhalation investigations. As shown in
Figure 9a, during one of the fire suppression experiments, the gas analyzer recorded data reflecting an increase in smoke temperature and CO content at the onset of the experiment, concurrently with a decrease in oxygen content due to the sustained combustion of the flame. Throughout the three fire extinguishing experiments, the lowest observed oxygen content was 17.2%, 18.0%, and 18.3%, which exceeded the self-extinguishing oxygen content threshold of 14%. When the UDPA was released, there was a marked rise in CO content as the flame combustion was suppressed. The carrier gas stream for the UDPA was air, leading to a gradual increase in oxygen content. Approximately 60 s after the deployment of the UDPA, the flame was fully extinguished. Across the three fire extinguishing experiments (
Figure 9b was given as the sample), it was noted that flame #3 was extinguished first, followed by flames #1 and #2 in sequence. The observed phenomenon may be attributed to the flow of gas within the experimental chamber. Under the influence of the primary flame, the heated air rose upwards, formed swirling patterns, and entrained around the periphery of the flame, leading to a higher concentration at the #3 position. Upon the continuous injection of the UDPA, the concentration at positions #1 and #2 gradually increased to the extinguishing levels and subsequently ceased. The amount of powder consumed during the three fire extinguishing experiments was 38.0 g, 42.2 g, and 44.4 g, with an average minimum extinguishing concentration of 41.5 g/m
3, which was better than the reported values of 79.8 g/m
3 and 60~160 g/m
3 [
38,
39].
Shown in
Figure 9c, in the acute inhalation experiment, the mice were removed from their cages 10 min after 120 g of the UDPA had been introduced into the chamber and found to be in good condition, with no fatalities and normal escape abilities. With sustained care and observation over the next three days, all four mice remained alive and exhibited either stable or slightly increased body weights. These results attest to the absence of the acute inhalation toxicity of the UDPA.