Poly(methyl methacrylate) with Oleic Acid as an Efficient Candida albicans Biofilm Repellent
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Physico-Chemicals Characterization of PMMA_OA
2.2.1. Fourier Transform Infrared Spectroscopy (FTIR)
2.2.2. Contact Angle Measurements
2.3. Antifungal Characterization of Composites
2.3.1. Biofilm Formation on PMMA_OA Composites
2.3.2. XTT Test on PMMA_OA Composites and in Biofilm Supernatant
2.4. Antifungal Susceptibility Test
2.4.1. Determination of Minimal Inhibitory Concentration (MIC)
2.4.2. Embedded Filamentation Test (EFT)
2.4.3. Statistic
3. Results
3.1. Physico-Chemicals Characterization
3.1.1. FTIR (Chemical Characterization of Composite Surface)
3.1.2. Water Contact Angle Measurement
3.2. Antifungal Characterization
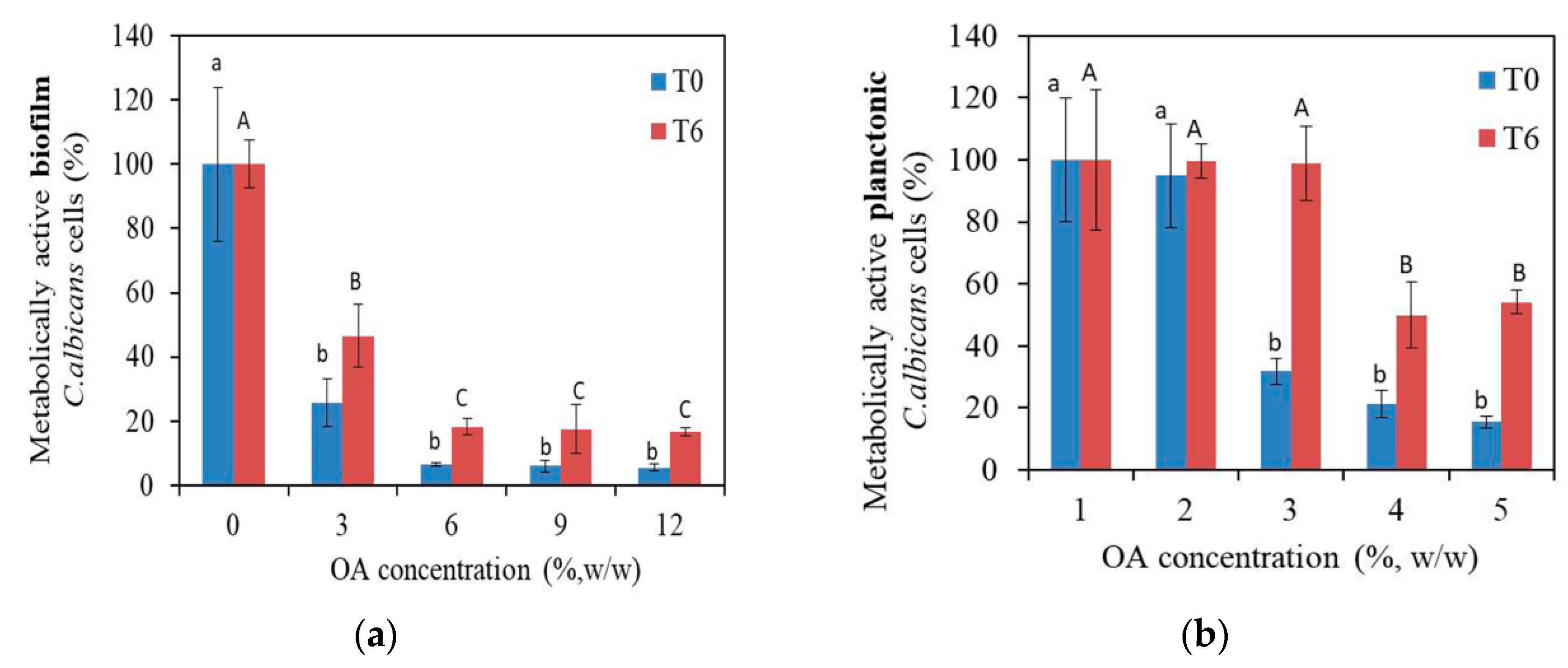
3.2.1. Antifungal Characterization of PMMA_OA Composites with XTT Test
3.2.2. Antifungal Susceptibility Test
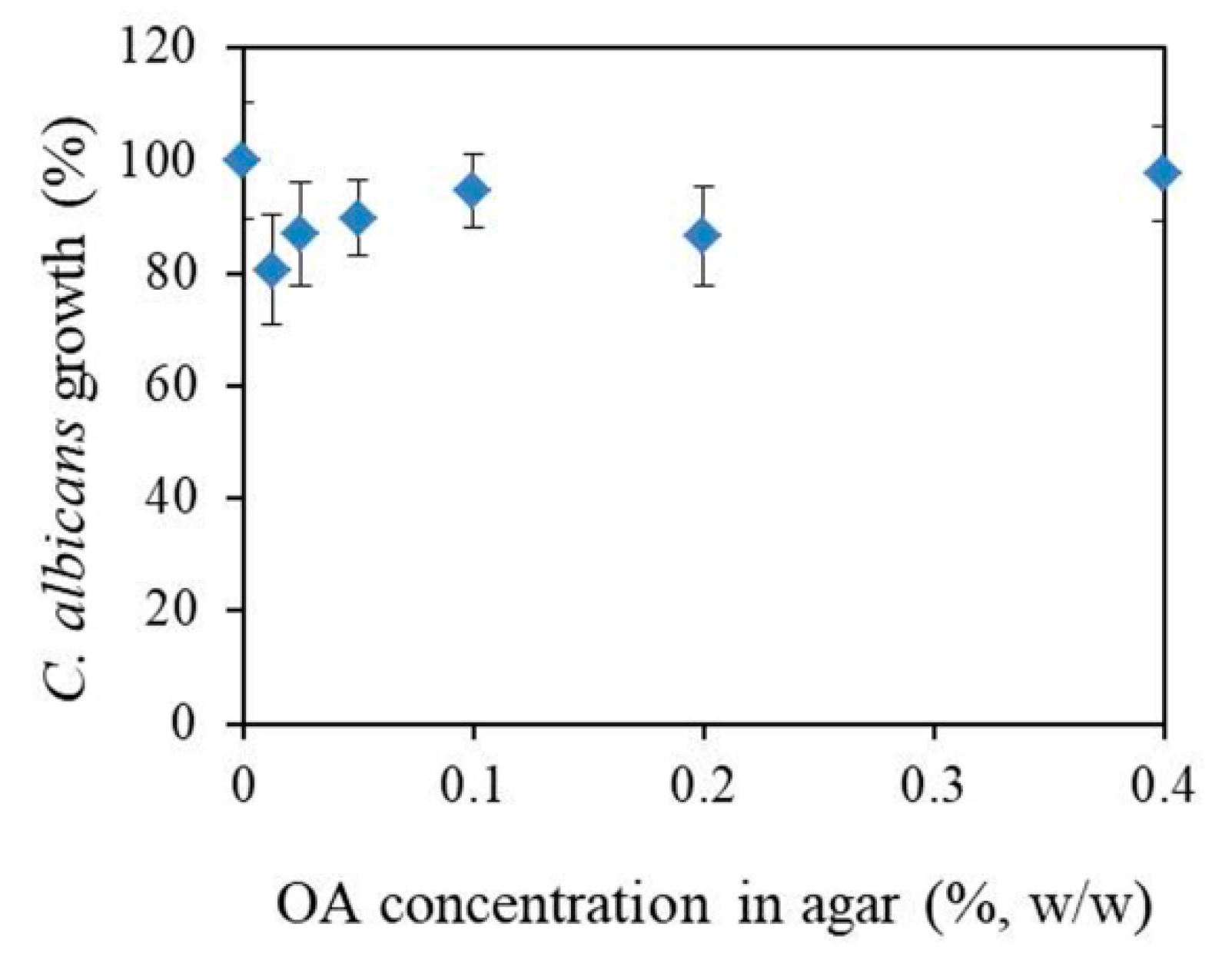
Determination of Minimal Inhibitory Concentration (MIC)
Embedded Filamentation Test (EFT)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Beltrán-Partida, E.; Valdez-Salas, B.; Curiel-Álvarez, M.; Castillo-Uribe, S.; Escamilla, A.; Nedev, N. Enhanced Antifungal Activity by Disinfected Titanium Dioxide Nanotubes via Reduced Nano-Adhesion Bonds. Mater. Sci. Eng. C 2017, 76, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Nobile, C.J.; Johnson, A.D. Candida Albicans Biofilms and Human Disease. Annu. Rev. Microbiol. 2015, 69, 71–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsui, C.; Kong, E.F.; Jabra-Rizk, M.A. Pathogenesis of Candida Albicans Biofilm. Pathog. Dis. 2016, 74, ftw018. [Google Scholar] [CrossRef] [Green Version]
- Petrović, M.; Hofmann, H.; Mionić Ebersold, M. PMMA-Oleic Acid Composites as Candida Biofilm Repellent; eCM Meeting Abstracts 2017, Collection 3; SSB+RM; European Cells and Materials/ECM, Empa: St.Gallen, Switzerland, 2017; p. 51. [Google Scholar]
- Cierech, M.; Szerszeń, M.; Wojnarowicz, J.; Łojkowski, W.; Kostrzewa-Janicka, J.; Mierzwińska-Nastalska, E. Preparation and Characterisation of Poly(Methyl Metacrylate)-Titanium Dioxide Nanocomposites for Denture Bases. Polymers 2020, 12, 2655. [Google Scholar] [CrossRef]
- Lee, M.-J.; Kim, M.-J.; Oh, S.-H.; Kwon, J.-S. Novel Dental Poly (Methyl Methacrylate) Containing Phytoncide for Antifungal Effect and Inhibition of Oral Multispecies Biofilm. Materials 2020, 13, 371. [Google Scholar] [CrossRef] [Green Version]
- Davies, D. Understanding Biofilm Resistance to Antibacterial Agents. Nat. Rev. Drug Discov. 2003, 2, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Nobile, C.J.; Fox, E.P.; Nett, J.E.; Sorrells, T.R.; Mitrovich, Q.M.; Hernday, A.D.; Tuch, B.B.; Andes, D.R.; Johnson, A.D. A Recently Evolved Transcriptional Network Controls Biofilm Development in Candida Albicans. Cell 2012, 148, 126–138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tobudic, S.; Kratzer, C.; Lassnigg, A.; Presterl, E. Antifungal Susceptibility of Candida Albicans in Biofilms. Mycoses 2012, 55, 199–204. [Google Scholar] [CrossRef]
- Shehabeldine, A.; El-Hamshary, H.; Hasanin, M.; El-Faham, A.; Al-Sahly, M. Enhancing the Antifungal Activity of Griseofulvin by Incorporation a Green Biopolymer-Based Nanocomposite. Polymers 2021, 13, 542. [Google Scholar] [CrossRef]
- Jose, A.; Coco, B.J.; Milligan, S.; Young, B.; Lappin, D.F.; Bagg, J.; Murray, C.; Ramage, G. Reducing the Incidence of Denture Stomatitis: Are Denture Cleansers Sufficient? J. Prosthodont. 2010, 19, 252–257. [Google Scholar] [CrossRef]
- Chladek, G.; Pakieła, K.; Pakieła, W.; Żmudzki, J.; Adamiak, M.; Krawczyk, C. Effect of Antibacterial Silver-Releasing Filler on the Physicochemical Properties of Poly(Methyl Methacrylate) Denture Base Material. Materials 2019, 12, 4146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Da Silva, P.M.; Acosta, E.J.; de Rezende Pinto, L.; Graeff, M.; Spolidorio, D.M.; Almeida, R.S.; Porto, V.C. Microscopical Analysis of Candida Albicans Biofilms on Heat-Polymerised Acrylic Resin after Chlorhexidine Gluconate and Sodium Hypochlorite Treatments. Mycoses 2011, 54, e712–e717. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, D.R.; Silva, S.; Negri, M.; Gorup, L.F.; De Camargo, E.R.; Oliveira, R.; Barbosa, D.D.; Henriques, M. Silver Nanoparticles: Influence of Stabilizing Agent and Diameter on Antifungal Activity against Candida Albicans and Candida Glabrata Biofilms. Lett. Appl. Microbiol. 2012, 54, 383–391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salerno, C.; Pascale, M.; Contaldo, M.; Esposito, V.; Busciolano, M.; Milillo, L.; Guida, A.; Petruzzi, M.; Serpico, R. Candida-Associated Denture Stomatitis. Med. Oral Patol. Oral Cir. Bucal 2011, 16, e139–e143. [Google Scholar] [CrossRef]
- Sun, X.; Cao, Z.; Yeh, C.-K.; Sun, Y. Antifungal Activity, Biofilm-Controlling Effect, and Biocompatibility of Poly(N-Vinyl-2-Pyrrolidinone)-Grafted Denture Materials. Colloids Surf. B Biointerfaces 2013, 110, 96–104. [Google Scholar] [CrossRef] [Green Version]
- Wady, A.F.; Machado, A.L.; Zucolotto, V.; Zamperini, C.A.; Berni, E.; Vergani, C.E. Evaluation of Candida Albicans Adhesion and Biofilm Formation on a Denture Base Acrylic Resin Containing Silver Nanoparticles. J. Appl. Microbiol. 2012, 112, 1163–1172. [Google Scholar] [CrossRef] [Green Version]
- Wen, J.; Jiang, F.; Yeh, C.-K.; Sun, Y. Controlling Fungal Biofilms with Functional Drug Delivery Denture Biomaterials. Colloids Surf. B Biointerfaces 2016, 140, 19–27. [Google Scholar] [CrossRef] [Green Version]
- Bajunaid, S.O.; Baras, B.H.; Balhaddad, A.A.; Weir, M.D.; Xu, H.H. Antibiofilm and Protein-Repellent Polymethylmethacrylate Denture Base Acrylic Resin for Treatment of Denture Stomatitis. Materials 2021, 14, 1067. [Google Scholar] [CrossRef]
- Salim, N.; Silikas, N.; Satterthwaite, J.D.; Moore, C.; Ramage, G.; Rautemaa, R. Chlorhexidine-Impregnated PEM/THFM Polymer Exhibits Superior Activity to Fluconazole-Impregnated Polymer against Candida Albicans Biofilm Formation. Int. J. Antimicrob. Agents 2013, 41, 193–196. [Google Scholar] [CrossRef]
- Muthamil, S.; Prasath, K.G.; Priya, A.; Precilla, P.; Pandian, S.K. Global Proteomic Analysis Deciphers the Mechanism of Action of Plant Derived Oleic Acid against Candida Albicans Virulence and Biofilm Formation. Sci. Rep. 2020, 10, 5113. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.-H.; Kim, Y.-G.; Khadke, S.K.; Lee, J. Antibiofilm and Antifungal Activities of Medium-Chain Fatty Acids against Candida Albicans via Mimicking of the Quorum-Sensing Molecule Farnesol. Microb. Biotechnol. 2021, 14, 1353–1366. [Google Scholar] [CrossRef] [PubMed]
- Petrović, M.; Bonvin, D.; Hofmann, H.; Mionić Ebersold, M. Fungicidal PMMA-Undecylenic Acid Composites. Int. J. Mol. Sci. 2018, 19, 184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chandra, J.; Mukherjee, P.K.; Leidich, S.D.; Faddoul, F.F.; Hoyer, L.L.; Douglas, L.J.; Ghannoum, M.A. Antifungal Resistance of Candidal Biofilms Formed on Denture Acrylic in Vitro. J. Dent. Res. 2001, 80, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Chandra, J.; Kuhn, D.M.; Mukherjee, P.K.; Hoyer, L.L.; McCormick, T.; Ghannoum, M.A. Biofilm Formation by the Fungal Pathogen Candida Albicans: Development, Architecture, and Drug Resistance. J. Bacteriol. 2001, 183, 5385–5394. [Google Scholar] [CrossRef] [Green Version]
- Pierce, C.G.; Uppuluri, P.; Tristan, A.R.; Wormley, F.L.; Mowat, E.; Ramage, G.; Lopez-Ribot, J.L. A Simple and Reproducible 96-Well Plate-Based Method for the Formation of Fungal Biofilms and Its Application to Antifungal Susceptibility Testing. Nat. Protoc. 2008, 3, 1494–1500. [Google Scholar] [CrossRef]
- Ferreira, J.A.G.; Carr, J.H.; Starling, C.E.F.; Resende, M.A.d.; Donlan, R.M. Biofilm Formation and Effect of Caspofungin on Biofilm Structure of Candida Species Bloodstream Isolates. Antimicrob. Agents Chemother. 2009, 53, 4377–4384. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, A.; Khan, A.; Akhtar, F.; Yousuf, S.; Xess, I.; Khan, L.A.; Manzoor, N. Fungicidal Activity of Thymol and Carvacrol by Disrupting Ergosterol Biosynthesis and Membrane Integrity against Candida. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 41–50. [Google Scholar] [CrossRef]
- Wiegand, I.; Hilpert, K.; Hancock, R.E. Agar and Broth Dilution Methods to Determine the Minimal Inhibitory Concentration (MIC) of Antimicrobial Substances. Nat. Protoc. 2008, 3, 163–175. [Google Scholar] [CrossRef]
- Duan, G.; Zhang, C.; Li, A.; Yang, X.; Lu, L.; Wang, X. Preparation and Characterization of Mesoporous Zirconia Made by Using a Poly (Methyl Methacrylate) Template. Nanoscale Res. Lett. 2008, 3, 118–122. [Google Scholar] [CrossRef] [Green Version]
- Totu, E.E.; Nechifor, A.C.; Nechifor, G.; Aboul-Enein, H.Y.; Cristache, C.M. Poly(Methyl Methacrylate) with TiO2 Nanoparticles Inclusion for Stereolitographic Complete Denture Manufacturing—The Fututre in Dental Care for Elderly Edentulous Patients? J. Dent. 2017, 59, 68–77. [Google Scholar] [CrossRef]
- Moreno, K.; García-Miranda, J.; Hernández-Navarro, C.; Ruiz-Guillén, F.; Aguilera-Camacho, L.; Lesso, R.; Arizmendi-Morquecho, A. Preparation and Performance Evaluation of PMMA/HA Nanocomposite as Bulk Material. J. Compos. Mater. 2015, 49, 1345–1353. [Google Scholar] [CrossRef]
- Elshereksi, N.W.; Ghazali, M.J.; Muchtar, A.; Azhari, C.H. Studies on the Effects of Titanate and Silane Coupling Agents on the Performance of Poly (Methyl Methacrylate)/Barium Titanate Denture Base Nanocomposites. J. Dent. 2017, 56, 121–132. [Google Scholar] [CrossRef]
- Hong, J.; Yamaoka-Koseki, S.; Yasumoto, K. Determination of Palmitic Acid, Oleic Acid and Linoleic Acid by Near-Infrared Transflectance Spectroscopy in Edible Oils. Food Sci. Technol. Int. 1996, 2, 146–149. [Google Scholar] [CrossRef] [Green Version]
- Lee, D.H.; Condrate, R.A. FTIR Spectral Characterization of Thin Film Coatings of Oleic Acid on Glasses: I. Coatings on Glasses from Ethyl Alcohol. J. Mater. Sci. 1999, 34, 139–146. [Google Scholar] [CrossRef]
- Wu, N.; Fu, L.; Su, M.; Aslam, M.; Wong, K.C.; Dravid, V.P. Interaction of Fatty Acid Monolayers with Cobalt Nanoparticles. Nano Lett. 2004, 4, 383–386. [Google Scholar] [CrossRef]
- Nájera, J.J.; Horn, A.B. Infrared Spectroscopic Study of the Effect of Oleic Acid on the Deliquescence Behaviour of Ammonium Sulfate Aerosol Particles. Phys. Chem. Chem. Phys. PCCP 2009, 11, 483–494. [Google Scholar] [CrossRef]
- Lindsay, A.K.; Deveau, A.; Piispanen, A.E.; Hogan, D.A. Farnesol and Cyclic AMP Signaling Effects on the Hypha-to-Yeast Transition in Candida Albicans. Eukaryot. Cell 2012, 11, 1219–1225. [Google Scholar] [CrossRef] [Green Version]
- Pendrak, M.L.; Roberts, D.D. Hbr1 Activates and Represses Hyphal Growth in Candida Albicans and Regulates Fungal Morphogenesis under Embedded Conditions. PLoS ONE 2015, 10, e0126919. [Google Scholar] [CrossRef]
- Mangal, U.; Kim, J.-Y.; Seo, J.-Y.; Kwon, J.-S.; Choi, S.-H. Novel Poly(Methyl Methacrylate) Containing Nanodiamond to Improve the Mechanical Properties and Fungal Resistance. Materials 2019, 12, 3438. [Google Scholar] [CrossRef] [Green Version]
- Bennett, J.E.; Izumikawa, K.; Marr, K.A. Mechanism of Increased Fluconazole Resistance in Candida Glabrata during Prophylaxis. Antimicrob. Agents Chemother. 2004, 48, 1773–1777. [Google Scholar] [CrossRef] [Green Version]
- Mathé, L.; Dijck, P.V. Recent Insights into Candida Albicans Biofilm Resistance Mechanisms. Curr. Genet. 2013, 59, 251–264. [Google Scholar] [CrossRef] [Green Version]
- Garland, E.R.; Rosen, E.P.; Clarke, L.I.; Baer, T. Structure of Submonolayer Oleic Acid Coverages on Inorganic Aerosol Particles: Evidence of Island Formation. Phys. Chem. Chem. Phys. 2008, 10, 3156–3161. [Google Scholar] [CrossRef] [PubMed]
- Chandra, J.; Patel, J.D.; Li, J.; Zhou, G.; Mukherjee, P.K.; McCormick, T.S.; Anderson, J.M.; Ghannoum, M.A. Modification of Surface Properties of Biomaterials Influences the Ability of Candida Albicans to Form Biofilms. Appl. Environ. Microbiol. 2005, 71, 8795–8801. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carrillo Pérez, C.; Cavia Camarero, M.D.; Alonso de la Torre, S. Role of Oleic Acid in Immune System; Mechanism of Action: A Review. Nutr. Hosp. 2012, 27, 978–990. [Google Scholar] [CrossRef] [Green Version]
- Sales-Campos, H.; Reis de Souza, P.; Crema Peghini, B.; Santana da Silva, J.; Ribeiro Cardoso, C. An Overview of the Modulatory Effects of Oleic Acid in Health and Disease. Mini Rev. Med. Chem. 2013, 13, 201–210. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrović, M.; Randjelović, M.; Igić, M.; Randjelović, M.; Arsić Arsenijević, V.; Mionić Ebersold, M.; Otašević, S.; Milošević, I. Poly(methyl methacrylate) with Oleic Acid as an Efficient Candida albicans Biofilm Repellent. Materials 2022, 15, 3750. https://doi.org/10.3390/ma15113750
Petrović M, Randjelović M, Igić M, Randjelović M, Arsić Arsenijević V, Mionić Ebersold M, Otašević S, Milošević I. Poly(methyl methacrylate) with Oleic Acid as an Efficient Candida albicans Biofilm Repellent. Materials. 2022; 15(11):3750. https://doi.org/10.3390/ma15113750
Chicago/Turabian StylePetrović, Milica, Marina Randjelović, Marko Igić, Milica Randjelović, Valentina Arsić Arsenijević, Marijana Mionić Ebersold, Suzana Otašević, and Irena Milošević. 2022. "Poly(methyl methacrylate) with Oleic Acid as an Efficient Candida albicans Biofilm Repellent" Materials 15, no. 11: 3750. https://doi.org/10.3390/ma15113750








