Insight into the Genome of Diverse Penicillium chrysogenum Strains: Specific Genes, Cluster Duplications and DNA Fragment Translocations
Abstract
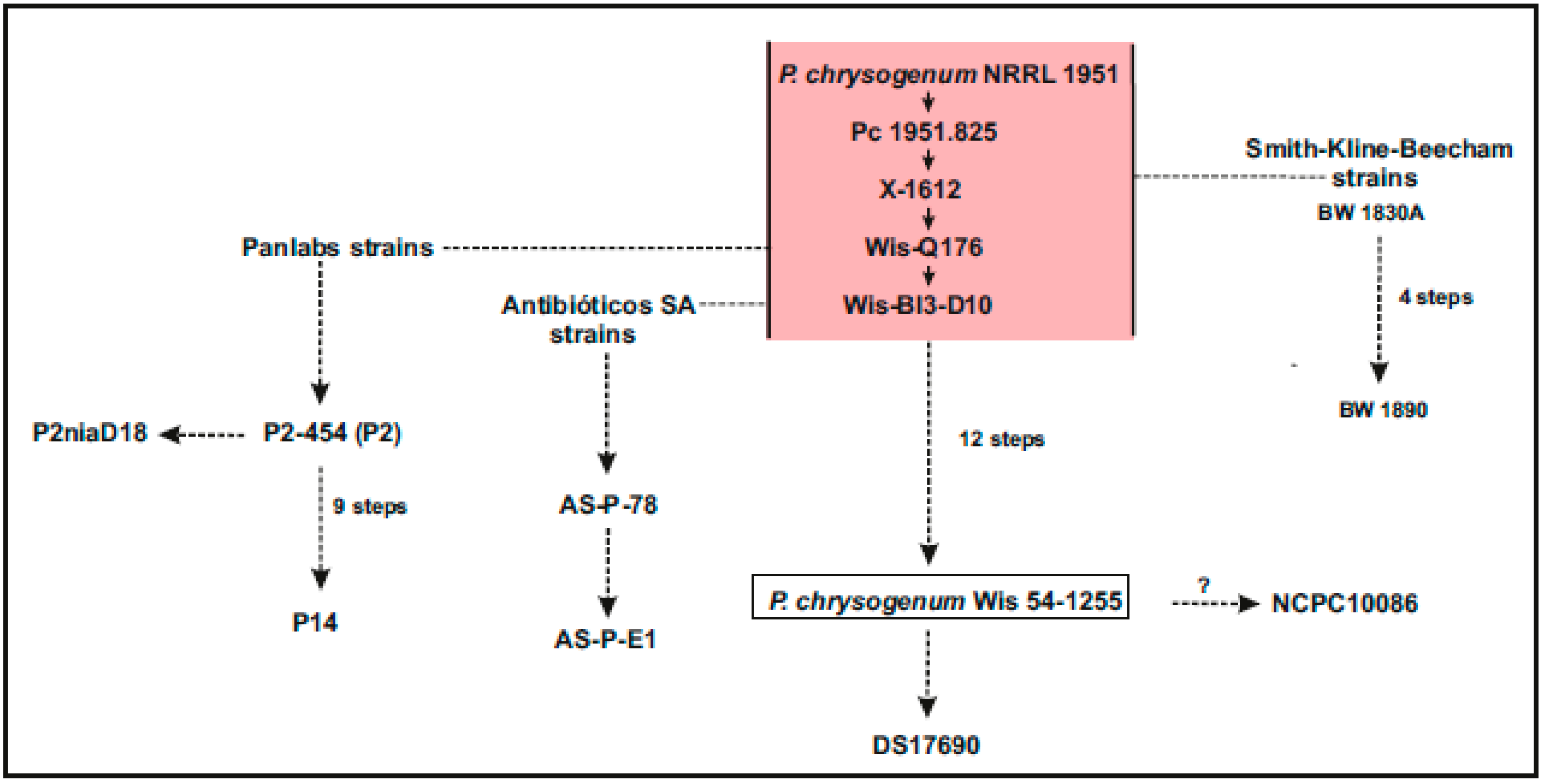
:1. Introduction: Penicillium chrysogenum as a Platform for the Biotechnology of Fungal Products
2. Unveiling the Content of the Genome of P. chrysogenum Wis54-1255
2.1. Reorganization of the Chromosome Architecture by DNA Fragments Translocation in the Strains P. chrysogenum Wis54-1255 and P. chrysogenum P2niaD18
2.2. Resolution of P. chrysogenum and P. notatum Chromosomes and Physical Map of the Encoding Genes
2.3. The Genome Sequence of the Wild Type Isolate P. chrysogenum Pc3 Contains Numerous Differences with Respect to P. chrysogenum NRRL1951
3. Genomes of Other Wild Type P. chrysogenum Strains
3.1. The Genome of the Wild Type P. chrysogenum KF-25 and Differences with P. chrysogenum Wis 54-1255
3.2. The Genome of P. chrysogenum HKF2: Potential for the Production of Prebiotics
4. Enigmas in the Genome of Industrial High Penicillin-Producing Strains
4.1. Mutations and Translocations in the Genomes of two High Penicillin Producing Strains Result in Drastic Changes in Gene Expression
4.2. The Genome of the Industrial Strain P. chrysogenum NCPC10086 Shows Small Differences with Other Industrial Strains
5. Concluding Remarks
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| SM | secondary metabolites |
| PK | polyketide |
| NRP | non-ribosomal peptide |
| PKS | polyketide synthase |
| NRPS | non-ribosomal peptide synthetase |
| ACV | δ-(l-α-aminoadipyl-l cysteinyl-d-valine) |
| ORF | open reading frame |
| SNP | single nucleotide polymorphism |
| CRISPR | Clustered Regularly Interspaced Short Palindromic Repeats |
References
- Houbraken, J.; Frisvad, J.C.; Seifert, K.A.; Overy, D.P.; Tuthill, D.M.; Valdez, J.G.; Samson, R.A. New penicillin-producing Penicillium species and an overview of section Chrysogena. Persoonia 2012, 29, 78–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scott, J.; Untereiner, W.A.; Wong, B.; Straus, N.A.; Malloch, D. Genotypic variation in Penicillium chrysogenum from indoor environments. Mycologia 2004, 96, 1095–1105. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A. On the antibacterial action of cultures of a Penicillium, with special reference to their use in the isolation of B. influenzae. Br. J. Exp. Pathol. 1929, 10, 226–236. [Google Scholar] [CrossRef]
- Aharonowitz, Y.; Cohen, G.; Martín, J.F. Penicillin and cephalosporin biosynthetic genes: Structure, organization, regulation, and evolution. Annu. Rev. Microbiol. 1992, 46, 461–495. [Google Scholar] [CrossRef]
- Martín, J.F.; Liras, P. Insights into the structure and molecular mechanisms of βlactam synthesizing enzymes in fungi. In Biotechnology of Microbial Enzymes; Brahmachati, G., Demain, A.L., Adrio, J.L., Eds.; Elsevier: New York, NY, USA, 2016; pp. 215–241. [Google Scholar]
- Laich, F.; Fierro, F.; Martín, J.F. Production of penicillin by fungi growing on food products: Identification of a complete penicillin gene cluster in Penicillium griseofulvum and a truncated cluster in Penicillium verrucosum. Appl. Environ. Microbiol. 2002, 68, 1211–1219. [Google Scholar] [CrossRef] [Green Version]
- Brakhage, A.A.; Thön, M.; Spröte, P.; Scharf, D.H.; Al-Abdallah, Q.; Wolke, S.M.; Hortschansky, P. Aspects on evolution of fungal beta-lactam biosynthesis gene clusters and recruitment of trans-acting factors. Phytochemistry 2009, 70, 1801–1811. [Google Scholar] [CrossRef]
- Martín, J.F.; Liras, P. Transfer of Secondary Metabolite Gene Clusters: Assembly and Reorganization of the β-Lactam Gene Cluster from Bacteria to Fungi and Arthropods. In Horizontal Gene Transfer: Breaking Borders Between Living Kingdoms; Villa, T.G., Viñas, M., Eds.; Springer Nature: Cham Switzerland, 2018; pp. 337–361. [Google Scholar]
- Raper, K.B.; Alexander, D.F.; Coghill, R.D. Penicillin: Natural variation and penicillin production in Penicillium notatum and allied species. J. Bacteriol. 1944, 48, 639–659. [Google Scholar] [CrossRef] [Green Version]
- Houbraken, J.; Frisvad, J.C.; Samson, R.A. Fleming’s penicillin-producing strain is not Penicillium chrysogenum but P. rubens. IMA Fungus 2011, 2, 87–95. [Google Scholar] [CrossRef] [Green Version]
- Keller, N.P.; Turner, G.; Bennett, J.W. Fungal secondary metabolism -from biochemistry to genomics. Nat. Rev. Genet. 2005, 3, 937–947. [Google Scholar] [CrossRef]
- Bok, J.W.; Keller, N.P. LaeA, a regulator of secondary metabolism in Aspergillus spp. Eukaryot. Cell 2004, 3, 527–535. [Google Scholar] [CrossRef] [Green Version]
- Jami, M.S.; García-Estrada, C.; Barreiro, C.; Cuadrado, A.A.; Salehi-Najafabadi, Z.; Martín, J.F. The Penicillium chrysogenum extracellular proteome. Conversion from a food-rotting strain to a versatile cell factory for white biotechnology. Mol. Cell. Proteom. 2010, 9, 2729–2744. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guzmán-Chávez, F.; Zwahlen, R.D.; Bovenberg, R.A.L.; Driessen, A.J.M. Engineering of the Filamentous Fungus Penicillium chrysogenum as Cell Factory for Natural Products. Front. Microbiol. 2018, 9, 2768. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Sáiz, M.; Barredo, J.L.; Moreno, M.A.; Fernández-Cañón, J.M.; Peñalva, M.A.; Díez, B. Reduced function of a phenylacetate-oxidizing cytochrome p450 caused strong genetic improvement in early phylogeny of penicillin-producing strains. J. Bacteriol. 2001, 183, 5465–5471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodríguez-Sáiz, M.; Díez, B.; Barredo, J.L. Why did the Fleming strain fail in penicillin industry? Fungal Genet. Boil. 2005, 42, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Kosalková, K.; Rodríguez-Sáiz, M.; Barredo, J.L.; Martín, J.F. Binding of the PTA1 transcriptional activator to the divergent promoter region of the first two genes of the penicillin pathway in different Penicillium species. Curr. Genet. 2007, 52, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Elander, R.P. Strain improvement and preservation of beta-lactam producing microorganisms. In Antibiotics Containing the Beta-Lactam Structure; Demain, A.L., Solomon, N.A., Eds.; Springer: Berlin/Heidelberg, Germany, 1983; pp. 97–146. [Google Scholar]
- Lein, J. The Panlabs penicillin strain improvement program. In Overproduction of Microbial Metabolites; Vanek, Z., Hostalek, Z., Eds.; Butterworths: Boston, MA, USA, 1986; pp. 105–139. [Google Scholar]
- Specht, T.; Dahlmann, T.A.; Zadra, I.; Kürnsteiner, H.; Kück, U. Complete sequencing and chromosome-scale genome assembly of the industrial progenitor strain P2niaD18 from the penicillin producer Penicillium chrysogenum. Genome Announc. 2014, e00577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Böhm, J.; Dahlmann, T.A.; Gümüşer, H.; Kück, U. A MAT1-2 wild-type strain from Penicillium chrysogenum: Functional mating-type locus characterization, genome sequencing and mating with an industrial penicillin-producing strain. Mol. Microbiol. 2015, 95, 859–874. [Google Scholar] [CrossRef] [Green Version]
- Newbert, R.W.; Barton, B.; Greaves, P.; Harper, J.; Turner, G. Analysis of a commercially improved Penicillium chrysogenum strain series: Involvement of recombinogenic regions in amplification and deletion of the penicillin biosynthesis gene cluster. J. Ind. Microbiol. Biotechnol. 1997, 19, 18–27. [Google Scholar] [CrossRef]
- Smidák, R.; Kralovicova, M.; Sevcikova, B.; Jakubcova, M.; Kormanec, J.; Timko, J.; Turna, J. Sequence analysis and gene amplification study of the penicillin biosynthesis gene cluster from different strains of Penicillium chrysogenum. Biologia 2010, 65, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Fierro, F.; Barredo, J.L.; Díez, B.; Gutiérrez, S.; Fernández, F.J.; Martín, J.F. The penicillin gene cluster is amplified in tandem repeats linked by conserved hexanucleotide sequences. Proc. Natl. Acad. Sci. USA 1995, 92, 6200–6204. [Google Scholar] [CrossRef] [Green Version]
- Peng, Q.; Yuan, Y.; Gao, M.; Chen, X.; Liu, B.; Liu, P.; Wu, Y.; Wu, D. Genomic characteristics and comparative genomics analysis of Penicillium chrysogenum KF-25. BMC Genom. 2014, 15, 144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gujar, V.V.; Fuke, P.; Khardenavis, A.A.; Purohit, H.J. Draft genome sequence of Penicillium chrysogenum strain HKF2, a fungus with potential for production of prebiotic synthesizing enzymes. 3 Biotech 2018, 8, 106. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.Q.; Zhong, J.; Zhao, Y.; Xiao, J.; Liu, J.; Dai, M.; Guizhen, Z.; Li, Z.; Jun, Y.; Jiayan, W.; et al. Genome sequencing of high-penicillin producing industrial strain of Penicillium chrysogenum. BMC Genom. 2014, 15, S11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salo, O.; Ries, M.; Medema, M.H.; Lankhorst, P.P.; Vreeken, R.J.; Bovenberg, R.A.L.; Driessen, A.J.M. Genomic mutational analysis of the impat ofthe classical strain improvement program on β-lactam producing Penicillium chrysogenum. BMC Genom. 2015, 16, 937. [Google Scholar] [CrossRef] [Green Version]
- Hersbach, G.J.M.; Van der Beek, C.P.; Van Dijck, P.W.M. The penicillins: Properties, biosynthesis, and fermentation. In Biotechnology of Industrial Antibiotics. Drugs and the Pharmaceutical Sciences; Vandamme, E.J., Ed.; Marcel Dekker: New York, NY, USA, 1984; Volume 22, pp. 45–140. [Google Scholar]
- Elander, R.P. Industrial production of beta-lactam antibiotics. Appl. Microbiol. Biotechnol. 2003, 61, 385–392. [Google Scholar] [CrossRef]
- Fierro, F.; Montenegro, E.; Gutiérrez, S.; Martín, J.F. Mutants blocked in penicillin biosynthesis show a deletion of the entire penicillin gene cluster at a specific site within a conserved hexanucleotide sequence. Appl. Microbiol. Biotechnol. 1996, 44, 597–604. [Google Scholar] [CrossRef]
- Van den Berg, M.A.; Albang, R.; Albermann, K.; Badger, J.H.; Daran, J.M.; Driessen, A.J.; García-Estrada, C.; Fedorova, N.D.; Harris, D.M.; Heijne, W.H.; et al. Genome sequencing and analysis of the filamentous fungus Penicillium chrysogenum. Nat. Biotechnol. 2008, 26, 1161–1168. [Google Scholar] [CrossRef] [Green Version]
- Salo, O.; Guzmán-Chávez, F.; Ries, M.I.; Lankhorst, P.P.; Bovenberg, R.A.L.; Vreeken, R.J.; Driessen, A.J.M. Identification of a Polyketide Synthase Involved in Sorbicillin Biosynthesis by Penicillium chrysogenum. Appl. Environ. Microbiol. 2016, 82, 3971–3978. [Google Scholar] [CrossRef] [Green Version]
- Walz, M.; Kück, U. Polymorphic karyotypes in related Acremonium strains. Curr. Genet. 1991, 19, 73–76. [Google Scholar] [CrossRef]
- Gutiérrez, S.; Fierro, F.; Casqueiro, J.; Martín, J.F. Gene organization and plasticity of the ß-lactam genes in different filamentous fungi. Antonie Van Leeuwenhoek 1999, 75, 81–94. [Google Scholar] [CrossRef]
- Dahlmann, T.A.; Böhm, J.; Becker, K.; Kück, U. Sexual recombination as a tool for engineering industrial Penicillium chrysogenum strains. Curr. Genet. 2015, 61, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Fierro, F.; Gutiérrez, S.; Díez, B.; Martín, J.F. Resolution of four large chromosomes in penicillin-producing filamentous fungi: The penicillin gene cluster is located on chromosome II (9.6 Mb) in Penicillium notatum and chromosome I (10.4 Mb) in Penicillium chrysogenum. Mol. Genet. Genom. 1993, 241, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; van den Berg, M.A.; Covaleda, L.; Lu, H.; Santos, F.A.; Uhm, T.; Lee, M.K.; Wu, C.; Liu, S.; Zhang, H.B. Genome physical mapping from large-insert clones by fingerprint analysis with capillary electrophoresis: A robust physical map of Penicillium chrysogenum. Nucleic Acids Res. 2005, 33, e50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henk, D.A.; Eagle, C.E.; Brown, K.; Van den Berg, M.A.; Dyer, P.S.; Peterson, S.W.; Fisher, M.C. Speciation despite globally overlapping distributions in Penicillium chrysogenum: The population genetics of Alexander Fleming’s lucky fungus. Mol. Ecol. 2011, 20, 4288–4301. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, J.C.; Grijseels, S.; Prigent, S.; Ji, B.; Dainat, J.; Nielsen, K.F.; Frisvad, J.C.; Workman, M.; Nielsen, J. Global analysis of biosynthetic gene clusters reveals vast potential of secondary metabolite production in Penicillium species. Nat. Microbiol. 2017, 2, 17044. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.D.; Xu, J.; Rapior, S.; Jeewon, R. The amazing potential of fungi: 50 forms we can exploit fungi industrially. Fungal Divers. 2019, 97, 1–136. [Google Scholar] [CrossRef] [Green Version]
- Marx, F.; Binder, U.; Leiter, E.; Pocsi, I. The Penicillium chrysogenum antifungal protein PAF, a promising tool for the development of new antifungal therapies and fungal cell biology studies. Mol. Life Sci. 2007, 65, 445–454. [Google Scholar] [CrossRef]
- Rodríguez-Martín, A.; Acosta, R.; Liddell, S.; Nuñez, F.; Benito, M.J.; Asensio, M.A. Characterization of the novel antifungal protein PgAFP and the encoding gene of Penicillium chrysogenum. Peptides 2010, 31, 541–547. [Google Scholar] [CrossRef]
- Leiter, E.; Szappanos, H.; Oberparleiter, C.; Kaiserer, L.; Csernoch, L.; Pusztahelyi, T.; Emri, T.; Pocsi, I.; Salvenmoser, W.; Marx, F. Antifungal protein PAF severely affects the integrity of the plasma membrane of Aspergillus nidulans and induces an apoptosis-like phenotype. Antimicrob. Agents Chemother. 2005, 49, 2445–2453. [Google Scholar] [CrossRef] [Green Version]
- Hegedus, N.; Leiter, E.; Kovacs, B.; Tomori, V.; Kwon, N.J.; Emri, T.; Marx, F.; Batta, G.; Csernoch, L.; Haas, H.; et al. The small molecular mass antifungal protein of Penicillium chrysogenum -a mechanism of action oriented review. J. Basic Microbiol. 2011, 51, 561–571. [Google Scholar] [CrossRef] [Green Version]
- Shaaban, M.; Palmer, J.M.; El-Naggar, W.A.; El-Sokkary, M.A.; Habib, E.-S.; Keller, N.P. Involvement of transposon-like elements in penicillin gene cluster regulation. Fungal Genet. Boil. 2010, 47, 423–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, S.; Goyal, A. Functional oligosaccharides: Production, properties and applications. World J. Microbiol. Biotechnol. 2010, 27, 1119–1128. [Google Scholar] [CrossRef]
- Patel, S.; Goyal, A. The current trends and future perspectives of prebiotics research: A review. 3 Biotech 2012, 2, 115–125. [Google Scholar] [CrossRef] [Green Version]
- Barredo, J.L.; Díez, B.; Alvarez, E.; Martín, J.F. Large amplification of a 35-kb DNA fragment carrying two penicillin biosynthetic genes in high penicillin producing strains of Penicillium chrysogenum. Curr. Genet. 1989, 16, 453–459. [Google Scholar] [CrossRef]
- Díez, B.; Gutiérrez, S.; Barredo, J.L.; van Solingen, P.; van der Voort, L.H.; Lucía, H.M.; Martín, J.F. The cluster of penicillin biosynthetic genes. J. Boil. Chem. 1990, 265, 16358–16365. [Google Scholar]
- Fierro, F.; Martín, J.F. Molecular mechanism of chromosomal rearrangement in fungi. Crit. Rev. Microbiol. 1999, 25, 1–17. [Google Scholar] [CrossRef]
- Jami, M.S.; Barreiro, C.; García-Estrada, C.; Martín, J.F. Proteome analysis of the penicillin producer Penicillium chrysogenum: Characterization of protein changes during the industrial strain improvement. Mol. Cell. Proteom. 2010, 9, 1182–1198. [Google Scholar] [CrossRef] [Green Version]
- Veiga, T.; Nijland, J.G.; Driessen, A.J.; Bovenberg, R.A.; Touw, H.; van den Berg, M.A.; Pronk, J.T.; Daran, J.M. Impact of velvet complex on transcriptome and penicillin G production in glucose-limited chemostat cultures of a β-lactam high-producing Penicillium chrysogenum strain. OMICS 2012, 16, 320–333. [Google Scholar] [CrossRef] [Green Version]
- Terfehr, D.; Dahlmann, T.A.; Kück, U. Transcriptome analysis of the two unrelated fungal β-lactam producers Acremonium chrysogenum and Penicillium chrysogenum: Velvet-regulated genes are major targets during conventional strain improvement programs. BMC Genom. 2017, 18, 272. [Google Scholar] [CrossRef] [Green Version]
- Martín, J.F. Key role of LaeA and velvet complex proteins on expression of β-lactam and PR-toxin genes in Penicillium chrysogenum: Cross-talk regulation of secondary metabolite pathways. J. Ind. Microbiol. Biotechnol. 2016, 44, 525–535. [Google Scholar] [CrossRef]
- Cohen, G.; Argaman, A.; Schreiber, R.; Mislovati, M.; Aharonowitz, Y. The thioredoxin system of Penicillium chrysogenum and its possible role in penicillin biosynthesis. J. Bacteriol. 1994, 176, 973–984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theilgaard, H.A.; Nielsen, J. Metabolic control analysis of the penicillin biosynthetic pathway: The influence of the LLD-ACV:bisACV ratio on the flux control. Antonie Van Leeuwenhoek 1999, 75, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Huber, A.; Hajdu, D.; Bratschun-Khan, D.; Gáspári, Z.; Varbanov, M.; Philippot, S.; Fizil, Á.; Czajlik, A.; Kele, Z.; Sonderegger, C.; et al. New Antimicrobial Potential and Structural Properties of PAFB: A Cationic, Cysteine-Rich Protein from Penicillium chrysogenum Q176. Sci. Rep. 2018, 8, 1751. [Google Scholar] [CrossRef] [PubMed]
- Sonderegger, C.; Galgóczy, L.; Garrigues, S.; Fizil, Á.; Borics, A.; Manzanares, P.; Hegedüs, N.; Huber, A.; Marcos, J.F.; Batta, G.; et al. A Penicillium chrysogenum-based expression system for the production of small, cysteine-rich antifungal proteins for structural and functional analyses. Microb. Cell Factories 2016, 15, 192. [Google Scholar] [CrossRef] [Green Version]
- McLean, K.J.; Hans, M.; Meijrink, B.; van Scheppingen, W.B.; Vollebregt, A.; Tee, K.L.; van der Laan, J.M.; Leys, D.; Munro, A.W.; van den Berg, M.A. Single-step fermentative production of the cholesterol-lowering drug pravastatin via reprogramming of Penicillium chrysogenum. Proc. Natl. Acad. Sci. USA 2015, 112, 2847–2852. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fierro, F.; García-Estrada, C.; Castillo, N.I.; Rodríguez, R.; Velasco-Conde, T.; Martín, J.F. Transcriptional and Bioinformatic Analysis of the 56.8 Kb DNA Region Amplified in Tandem Repeats Containing the Penicillin Gene Cluster in Penicillium chrysogenum. Fungal Genet. Boil. 2006, 43, 618–629. [Google Scholar]
- Martín, J.F.; Liras, P. Novel Antimicrobial and other Bioactive Metabolites obtained from silent gene clusters. In Antibiotics, Current Innovations and Future Trends; Sánchez, S., Demain, A.L., Eds.; Caister Academic Press: Poole, UK, 2015; pp. 271–292. [Google Scholar]
- Pohl, C.; Kiel, J.A.; Driessen, A.J.; Bovenberg, R.A.; Nygård, Y. CRISPR/Cas9 Based Genome Editing of Penicillium chrysogenum. ACS Synth. Biol. 2016, 5, 754–764. [Google Scholar] [CrossRef]
| Strain | Copies of the Penicillin Gene Cluster | Source and Reference |
|---|---|---|
| P. notatum * | 1 | [3] |
| P. chrysogenum NRRL1951 * | 1 | [9] |
| P. chrysogenum Wis54-1255 * | 1 | [18] |
| P. chrysogenum P2 * | 2 | [19] |
| P. chrysogenum P2niaD18 * | 2 | [20] |
| P. chrysogenum Pc3 | 1 | [21] |
| P. chrysogenum BW series * | Up to 50 | Beecham Corp, UK [22] |
| P. chrysogenum NMU2/40 * | 4 | Biotika AS [23] |
| P. chrysogenum B14 * | 6 | Biotika AS, Slovakia [23] |
| P. chrysogenum WE1 | 1 | Inst. of Mol. Biol., Slovak Acad. Sci., Bratislava [23] |
| P. chrysogenum ASP-78 * | 5–6 | Antibióticos SA, Spain [24] |
| P. chrysogenum ASP-E1 * | 12–14 | Antibióticos SA, Spain [24] |
| P. chrysogenum KF25 | 1 | [25] |
| P. chrysogenum HKF2 | unknown | [26] |
| P. chrysogenum NCPC10086 * | 8 | [27] |
| P. chrysogenum DS17690 * | 8 | [28] |
| Characteristics | Penicillium chrysogenum strain | |||||
|---|---|---|---|---|---|---|
| Wis54-1255 | KF-25 | HKF2 | NCPC10086 | P2niaD18 | Pc3 | |
| Genome size (Mb) | 32.2 | 29.92 | 31.48 | 32.2 | 32.49 | 32.2 |
| GC content (%) | 48.9 | 49.0 | 53.21 | 48.9 | NA | 49 |
| Gene number | 12943 | 9804 | 11243 | 13290 | 11839 | 11460 |
| Average gene size (pb) | 1.515 | 1.573 | NA | 1.499 | NA | NA |
| Introns/gene | 2.2 | 2.2 | NA | NA | NA | NA |
| Exons/gene | 3.0 | 3.2 | 3.15 | NA | NA | NA |
| tRNA (Number) | 145 | 112 | 188 | NA | 188 | 213 |
| rRNA (number) | 28 | 29 | NA | NA | NA | 49 |
| Accession Number | AM920416 AM920464 | SRP022930 | MUXA00000000 | APKG00000000 | JMSF00000000 | JPDR00000000 |
| Strain | Chromosome I | Chromosome II | Chromosome III | Chromosome IV | Size Determination Method | Reference |
|---|---|---|---|---|---|---|
| P. notatum | 10.8 Mb | 9.6 Mb | 6.3 Mb | 5.4 Mb | PFGE | [37] |
| P. chrysogenum NRRL 1951 | 10.4 Mb | 9.6 Mb | 7.3 Mb | 6.8 Mb | PFGE | [37] |
| P. chrysogenum Wis-54-1255 | 10.3 Mb | 9.7 Mb | 7.2 Mb | 6.3 Mb | PFGE | [37] |
| P. chrysogenum Wis-54-1255 | 10,350,089 bp | 9,488,591 bp | 6,943,310 bp | 5,586,572 bp | Sequencing | [32,20] |
| P. chrysogenum P2niaD18 | 13,597,116 | 10,455,537 | 5,401,030 | 3,043,715 | Sequencing | [20] |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martín, J.F. Insight into the Genome of Diverse Penicillium chrysogenum Strains: Specific Genes, Cluster Duplications and DNA Fragment Translocations. Int. J. Mol. Sci. 2020, 21, 3936. https://doi.org/10.3390/ijms21113936
Martín JF. Insight into the Genome of Diverse Penicillium chrysogenum Strains: Specific Genes, Cluster Duplications and DNA Fragment Translocations. International Journal of Molecular Sciences. 2020; 21(11):3936. https://doi.org/10.3390/ijms21113936
Chicago/Turabian StyleMartín, Juan F. 2020. "Insight into the Genome of Diverse Penicillium chrysogenum Strains: Specific Genes, Cluster Duplications and DNA Fragment Translocations" International Journal of Molecular Sciences 21, no. 11: 3936. https://doi.org/10.3390/ijms21113936




