Abstract
Apocynum venetum is a semi-shrubby perennial herb that not only prevents saline–alkaline land degradation but also produces leaves for medicinal uses. Although physiological changes during the seed germination of A. venetum in response to salt stress have been studied, the adaptive mechanism to salt conditions is still limited. Here, the physiological and transcriptional changes during seed germination under different NaCl treatments (0–300 mmol/L) were examined. The results showed that the seed germination rate was promoted at low NaCl concentrations (0–50 mmol/L) and inhibited with increased concentrations (100–300 mmol/L); the activity of antioxidant enzymes exhibited a significant increase from 0 (CK) to 150 mmol/L NaCl and a significant decrease from 150 to 300 mmol/L; and the content of osmolytes exhibited a significant increase with increased concentrations, while the protein content peaked at 100 mmol/L NaCl and then significantly decreased. A total of 1967 differentially expressed genes (DEGs) were generated during seed germination at 300 mmol/L NaCl versus (vs.) CK, with 1487 characterized genes (1293 up-regulated, UR; 194 down-regulated, DR) classified into 11 categories, including salt stress (29), stress response (146), primary metabolism (287), cell morphogenesis (156), transcription factor (TFs, 62), bio-signaling (173), transport (144), photosynthesis and energy (125), secondary metabolism (58), polynucleotide metabolism (21), and translation (286). The relative expression levels (RELs) of selected genes directly involved in salt stress and seed germination were observed to be consistent with the changes in antioxidant enzyme activities and osmolyte contents. These findings will provide useful references to improve seed germination and reveal the adaptive mechanism of A. venetum to saline–alkaline soils.
1. Introduction
Apocynum venetum L. (Apocynaceae) is a semi-shrubby perennial herb that is widely distributed in saline–alkaline soils and plays a critical role in preventing the land from degradation [1,2]. The leaves are used as a traditional Chinese medicine (TCM) for the treatment of cardiac disease, hypertension, and nephritis, which largely rely on the bioactive compounds such as flavonoids, organic acids, and polysaccharides [3,4,5].
In natural habitats, the plants have to face abiotic stresses, such as drought, extreme temperatures, and high salinity [6]. In saline–alkaline soils, the plants grow flourishingly due to their adaptive ability in a high salinity environment [7,8]. Seed germination is the initial stage throughout the life cycle and plays a determining role in the distribution of plant populations, especially under adverse abiotic conditions [9,10]. Currently, germination characteristics and physiological changes have been conducted during the seed germination of A. venetum in response to abiotic stresses. Specifically, salt and drought stress significantly inhibited seed germination [11,12,13,14]; fluctuated temperatures (10/25 and 15/30 °C, 12 h/12 h) were more in favor of seed germination than other temperatures [8]; a prolonged storage period significantly decreased seed germination [8,15]; and there was no significant effect of light on seed germination [8]. For the physiological changes in response to salt stress, the rate of electrolyte leakage, the content of osmolytes (e.g., soluble sugar, malondialdehyde (MDA), and proline (Pro)), and the activity of antioxidant enzymes (e.g., superoxide dismutase (SOD), ascorbateperoxidase (APX), catalase (CAT), and peroxidase (POD)) gradually increased with increased NaCl concentrations [13,16,17,18]. Meanwhile, salt stress induced the UR of genes involved in cation transport and antioxidants while inducing the DR of genes involved in cell wall structure [19].
Previous studies have found that there are distinct resistances for seed germination among different psammophytes under salt stress. For example, the seed germination of Halimodendron halodendron and Alhagi sparsifolia was significantly affected by the NaCl treatments [20]; the seed germination of Halogeton glomeratus and Cannabis sativa was inhibited by increased salt concentrations [21,22]. Extensive studies have demonstrated that plants respond and adapt to salt stress through various physiological, biochemical, and transcriptional processes, thereby acquiring stress tolerance [23,24]. For example, the content of soluble protein and Pro increased at an early stage and then decreased with the prolonging of salt stress, and the activity of SOD, POD, and CAT as well as the content of MDA in Broyssonetia papyrifera and Pogostemon cablin gradually increased with increased salt concentrations [25,26]. Hundreds of genes involved in abiotic tolerance as well as growth and development have also been identified in response to salt stress [27]. For example, the stress-responsive genes (e.g., DREB/CBFs), abiotic stress-related TFs (e.g., ZAT, WRKY, and NAC), and transporters (e.g., MATE and LeOPT1-like) were up-regulated in Arabidopsis under salt stress [28,29,30]. The above results indicate that salt stress triggers a wide range of responses, including physiological characteristics (e.g., seed germination and growth), cellular metabolism (e.g., osmolytes accumulation and enzyme activity), and molecular regulation (e.g., gene and protein expression).
To date, a low-salt environment improving the seed germination of A. venetum has been found, and the physiological changes have also been primarily conducted [16,17,18], while the adaptive mechanism to salt stress is still limited. In order to reveal the adaptive mechanism of A. venetum to salt stress in this study, we examined the changes in the rate of seed germination, the activity of antioxidant enzymes, the content of osmolytes, and the expression levels of genes in A. venetum at the seed germination stage under different NaCl treatments.
2. Results
2.1. Seed Germination Rate at Different NaCl Treatments
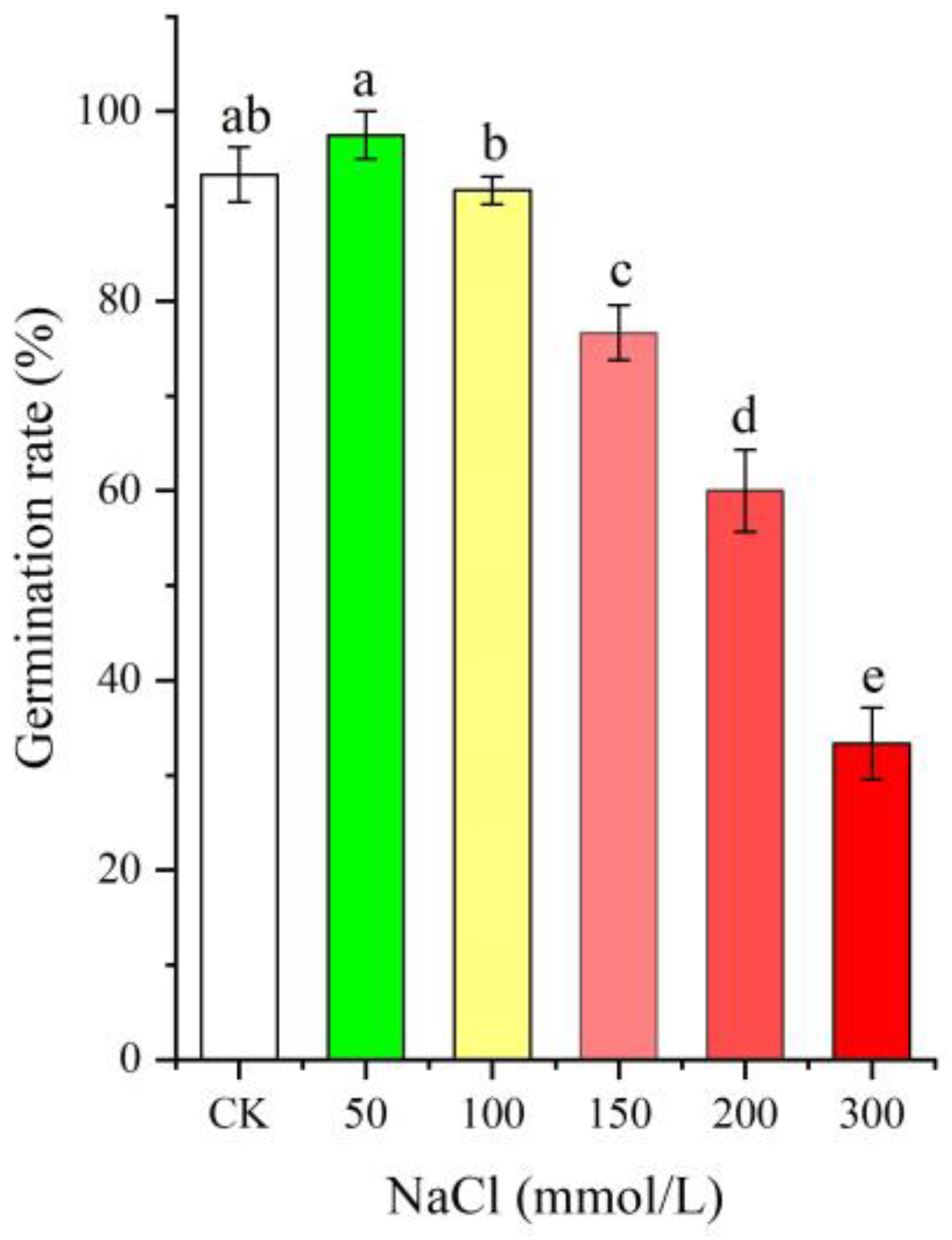
As shown in Figure 1, the seed germination rate was distinctly affected by different NaCl treatments (0–300 mmol/L, p < 0.05), with an increase at the low NaCl concentration (50 mmol/L), while there was a gradual decrease with increased NaCl concentrations (from 100 to 300 mmol/L) compared with CK.
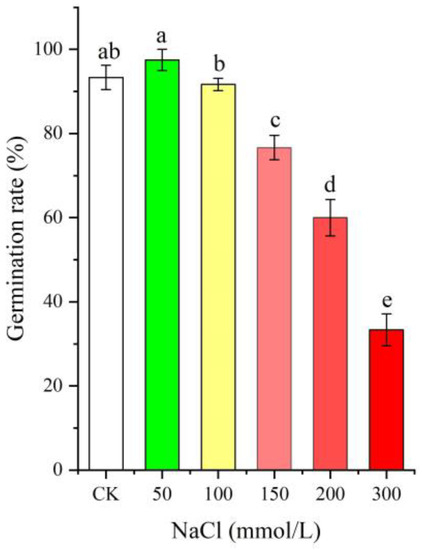
Figure 1.
Change in the seed germination rate of Apocynum venetum under different NaCl treatments. Different letters represent a significant difference (p < 0.05) among different NaCl treatments.
2.2. Antioxidant Enzyme Activities at Different NaCl Treatments
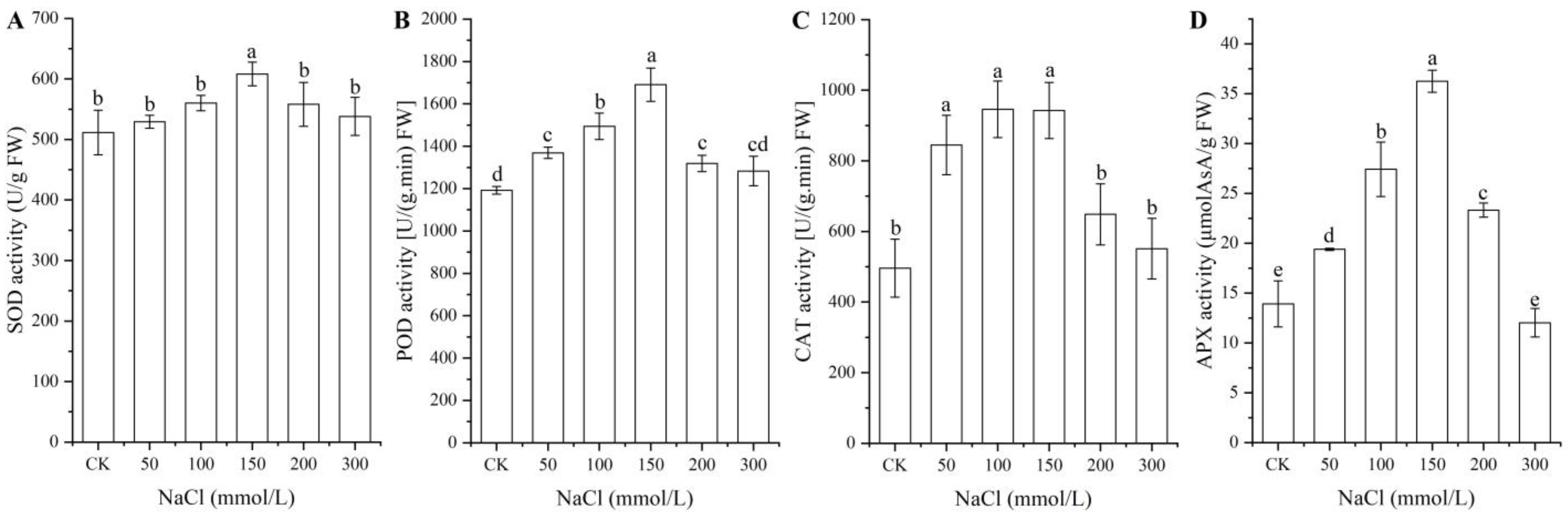
As shown in Figure 2, significant changes in the activities of the four antioxidant enzymes (i.e., SOD, POD, CAT, and APX) in seeds were observed under different NaCl treatments, with a significant increase with NaCl concentrations from 0 (CK) to 150 mmol/L and a significant decrease with NaCl concentrations from 150 to 300 mmol/L.
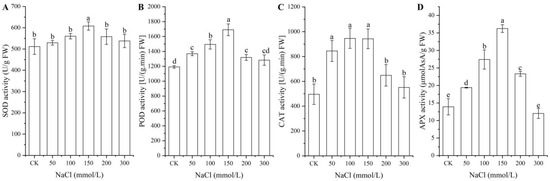
Figure 2.
Changes in the activities of the four antioxidant enzymes SOD (A), POD (B), CAT (C), and APX (D) in A. venetum seeds at different NaCl treatments. Different letters represent a significant difference (p < 0.05) among different NaCl treatments.
2.3. Osmolyte Contents under Different NaCl Treatments
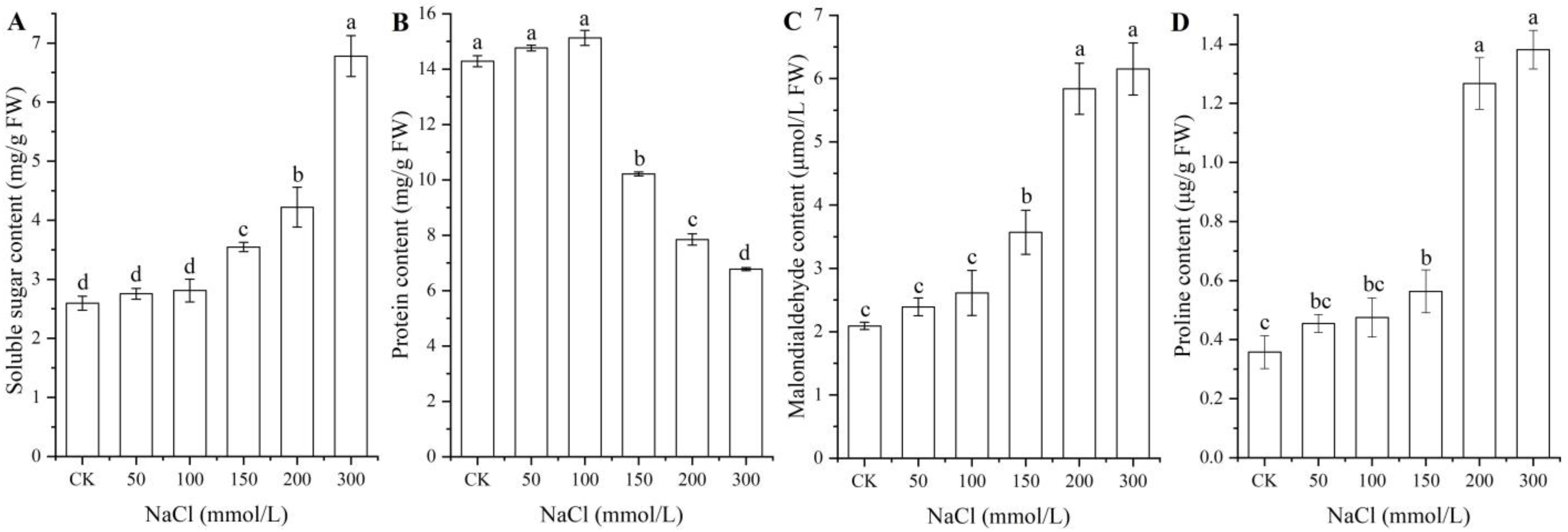
As shown in Figure 3, significant changes in osmolyte contents (i.e., soluble sugar, protein, MDA, and Pro) in seeds were observed under different NaCl treatments, with a significant increase for the contents of soluble sugar, MDA, and Pro with increased NaCl concentrations, while a significant decrease in the protein content was observed at NaCl concentrations from 100 to 300 mmol/L.
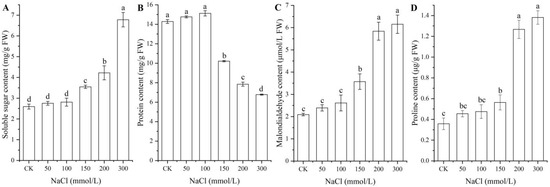
Figure 3.
Changes in the contents of soluble sugar (A), protein (B), MDA (C), and proline (D) in A. venetum seeds under different NaCl treatments. Different letters represent a significant difference (p < 0.05) among different NaCl treatments.
2.4. Transcriptomic Analysis
2.4.1. Global Gene Analysis
In order to find far more genes involved in salt tolerance, transcriptomic analysis was conducted on a higher salt concentration (300 mmol/L) instead of a lower salt concentration (50 mmol/L) vs. CK. After data filtering, 51.43 and 65.36 million high-quality reads were generated, and 42.12 and 53.26 million unique reads with 0.81 and 0.80 million multiple reads were mapped for CK and NaCl (300 mmol/L), respectively (Table 1; Figure S1).

Table 1.
Summary of sequencing data of seed germination under the CK and 300 mmol/L treatments (mean ± SD, n = 3).
A total of 52,305 unigenes were annotated on the Kyoto Encyclopedia of Genes and Genomes (KEGG, 34,198), Eukaryotic Orthologous Groups of proteins (KOG, 22,630), NCBI non-redundant protein (Nr, 36,077), and SwissProt (27,467) databases (Figure S2). Using the KEGG database, 1967 DEGs were enriched into 139 metabolism pathways such as global and overview maps, carbohydrate metabolism, and amino acid metabolism (Figure S3). Using the KOG database, 43.27% unigenes encoded the identified proteins that could be classified into 25 functional categories (Figure S4). Using the NR database, the top 10 species included: Coffea arabica, Carica papaya, Coffea eugenioides, Pistacia vera, Actinidia chinensis, Vitis vinifera, Citrus clementine, Theobroma cacao, Olea europaea, and Citrus sinensis (Figure S5). Using the SwissProt database, 52.51% unigenes were annotated to have biological functions. Using the Gene Ontology (GO) database, the DEGs were classified into three ontologies, including biological process (BP), cellular component (CC), and molecular function (MF) (Figure S6).
2.4.2. Identification of DEGs
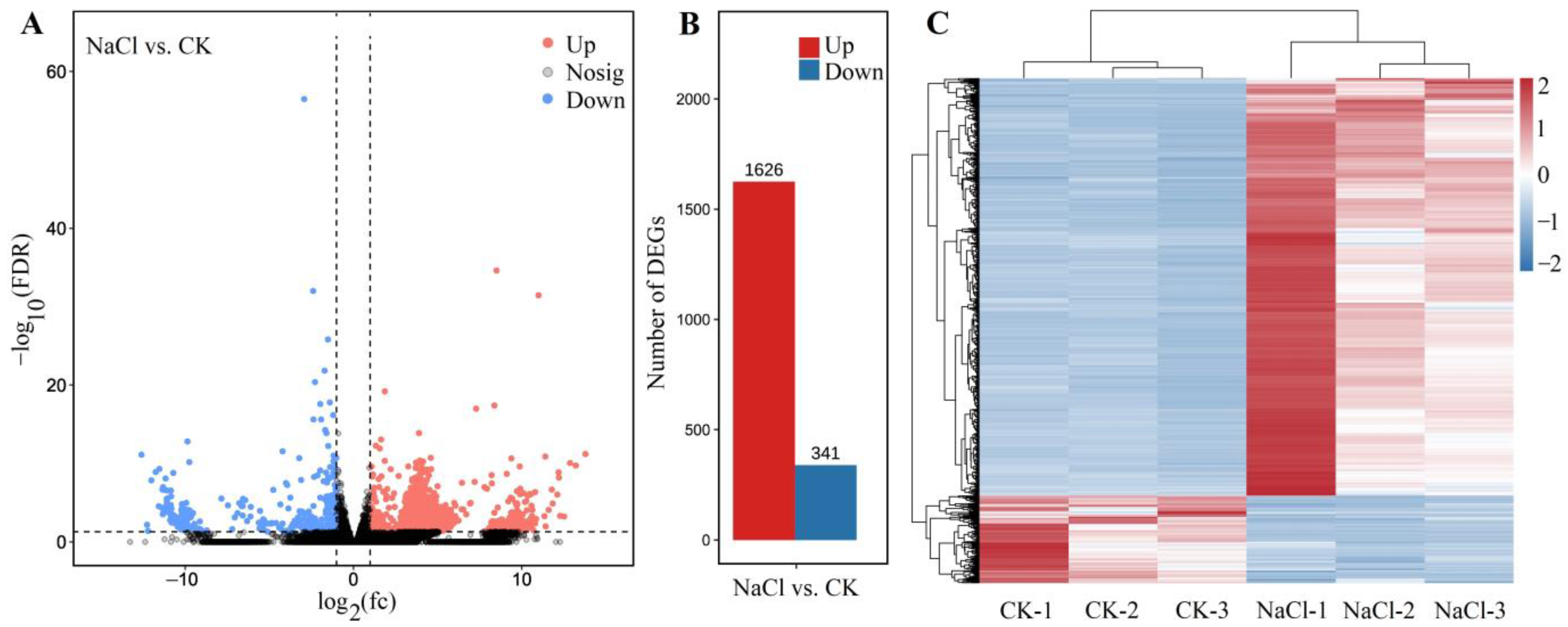
Among the 1967 DEGs, 1626 genes were UR and 341 genes were DR at 300 mmol/L NaCl vs. CK (Figure 4A,B), based on the principal component analysis (Figure S7) and Pearson correlation analysis (Figure S8). The cluster heat map of the 1967 DEGs is shown in Figure 4C. The distinct difference in the expression levels of genes between NaCl and CK treatments indicates that the data can be used for further analysis of gene biological functions.
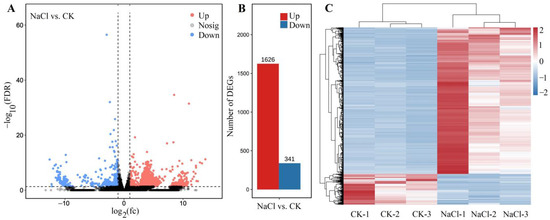
Figure 4.
Volcano plot of unigenes (A), number of DEGs (B), and cluster heat map of the DEGs (C) for NaCl (300 mmol/L) vs. CK. The black represent the genes without difference changes in the criteria of the false discovery rate (FDR) < 0.05 and |log2(fold-change)| > 1.
2.4.3. Distribution and Classification of DEGs
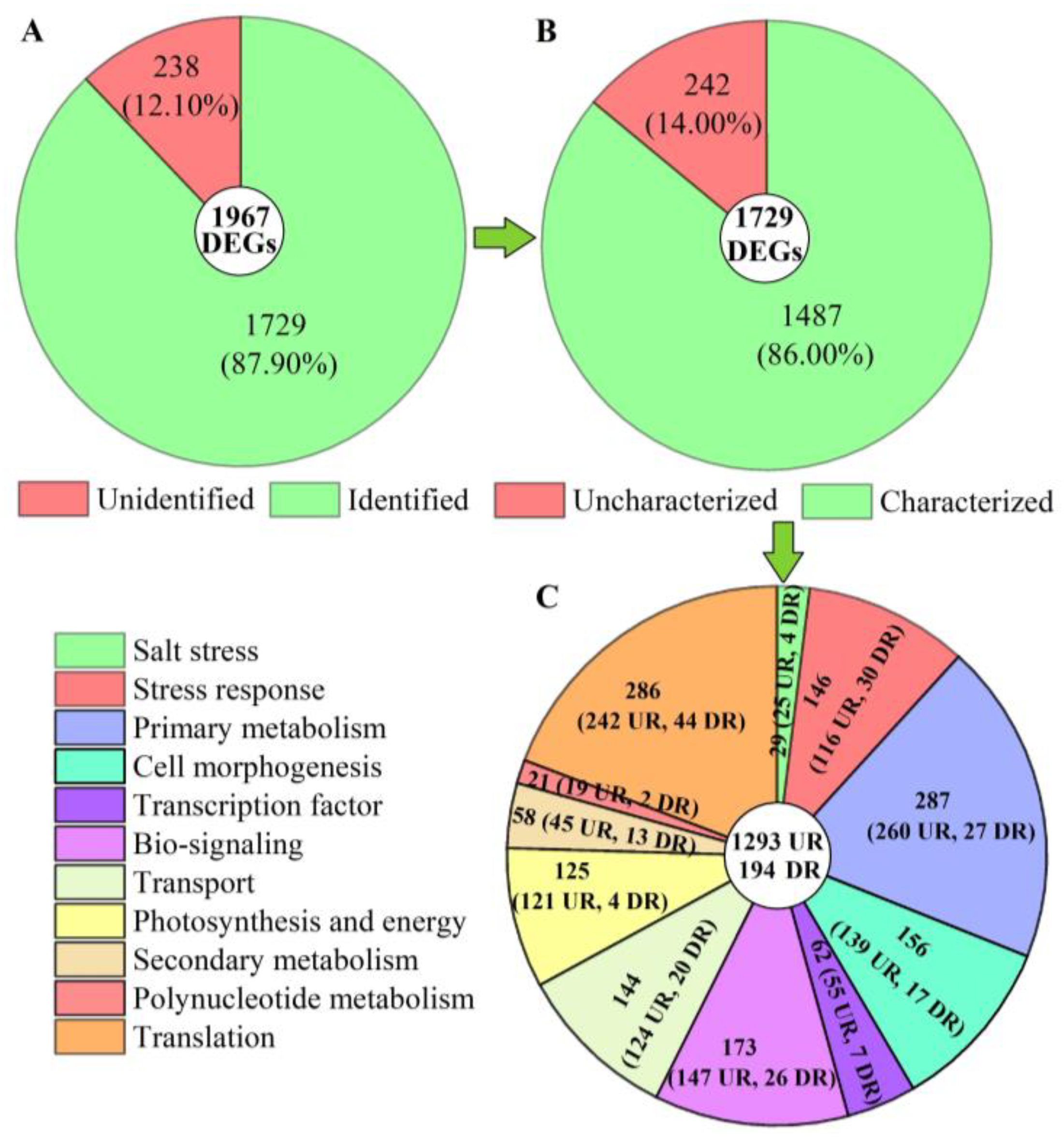
Of these 1967 DEGs, 1729 genes were identified from the KEGG, KOG, SwissProt, or GO databases (Figure 5A). Among the 1729 genes, 1487 genes with characterized biological functions were distributed into 1293 UR and 194 DR (Figure 5B). Based on their biological functions, the 1487 genes were classified into 11 categories, including salt stress (29), stress response (146), primary metabolism (287), cell morphogenesis (156), transcription factor (TFs, 62), bio-signaling (173), transport (144), photosynthesis and energy (125), secondary metabolism (58), polynucleotide metabolism (21), and translation (286) (Figure 5C).
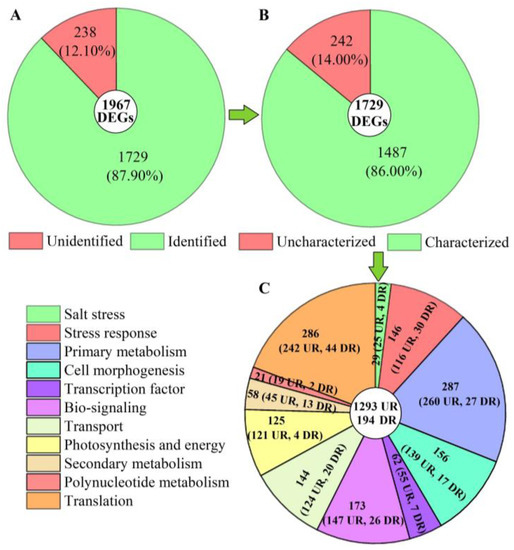
Figure 5.
Distribution and classification of DEGs at 300 mmol/L NaCl vs. CK. Abbreviations: UR, up-regulated, DR, down-regulated. Image (A) represents the distribution of unidentified and identified genes; image (B) represents the distribution of uncharacterized and characterized genes, and image (C) represents the classification of the characterized biological functional genes.
2.5. Specific Classification of DEGs and Validation of Expression Levels
2.5.1. DEGs Directly Associated with Salt Stress
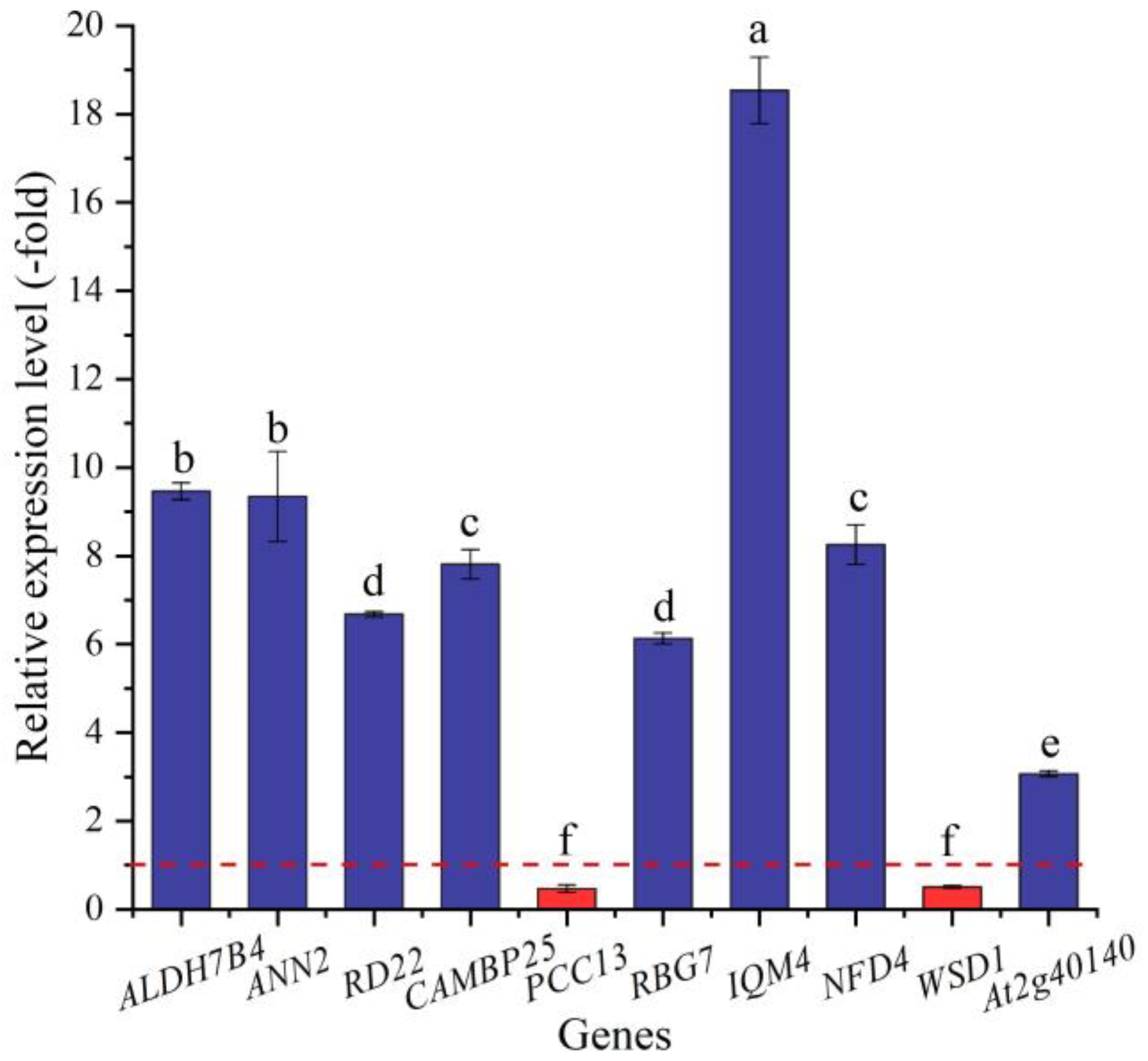
Based on the biological functions of proteins encoded by the DEGs, 29 genes (25 UR, 4 DR) were found to be directly associated with salt stress (e.g., ALDH7B4, ALDH10A8, and ANN2) (Table 2). The expression levels of 10 selected genes were validated by qRT-PCR, with 3.07- (At2g40140) to 18.54-fold (IQM4) UR for eight genes and 0.47- (PCC13) and 0.50-fold (WSD1) DR for two genes at the 300 mmol/L NaCl vs. CK (Figure 6). Meanwhile, the RELs were consistent with their Reads Per kb per Million (RPKM) values (Table 2).

Table 2.
Twenty-nine genes directly associated with salt stress at 300 mmol/L NaCl vs. CK.
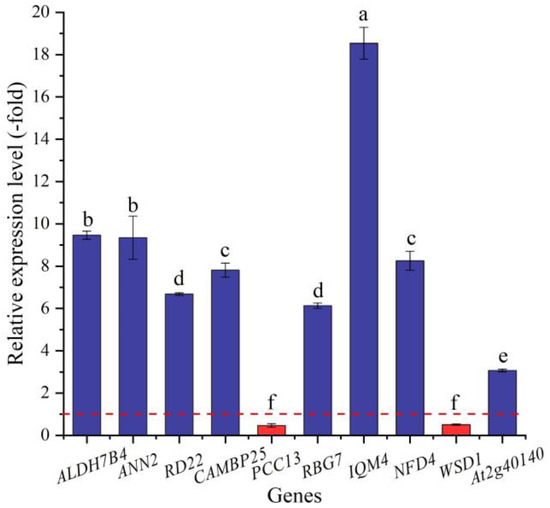
Figure 6.
The RELs of genes directly associated with salt stress in A. venetum seeds at 300 mmol/L NaCl vs. CK (mean ± SD, n = 3). Different letters represent a significant difference (p < 0.05) among different genes. The red dotted line differentiates UR (>1) and DR (<1).
2.5.2. DEGs Directly Associated with Antioxidant Enzymes
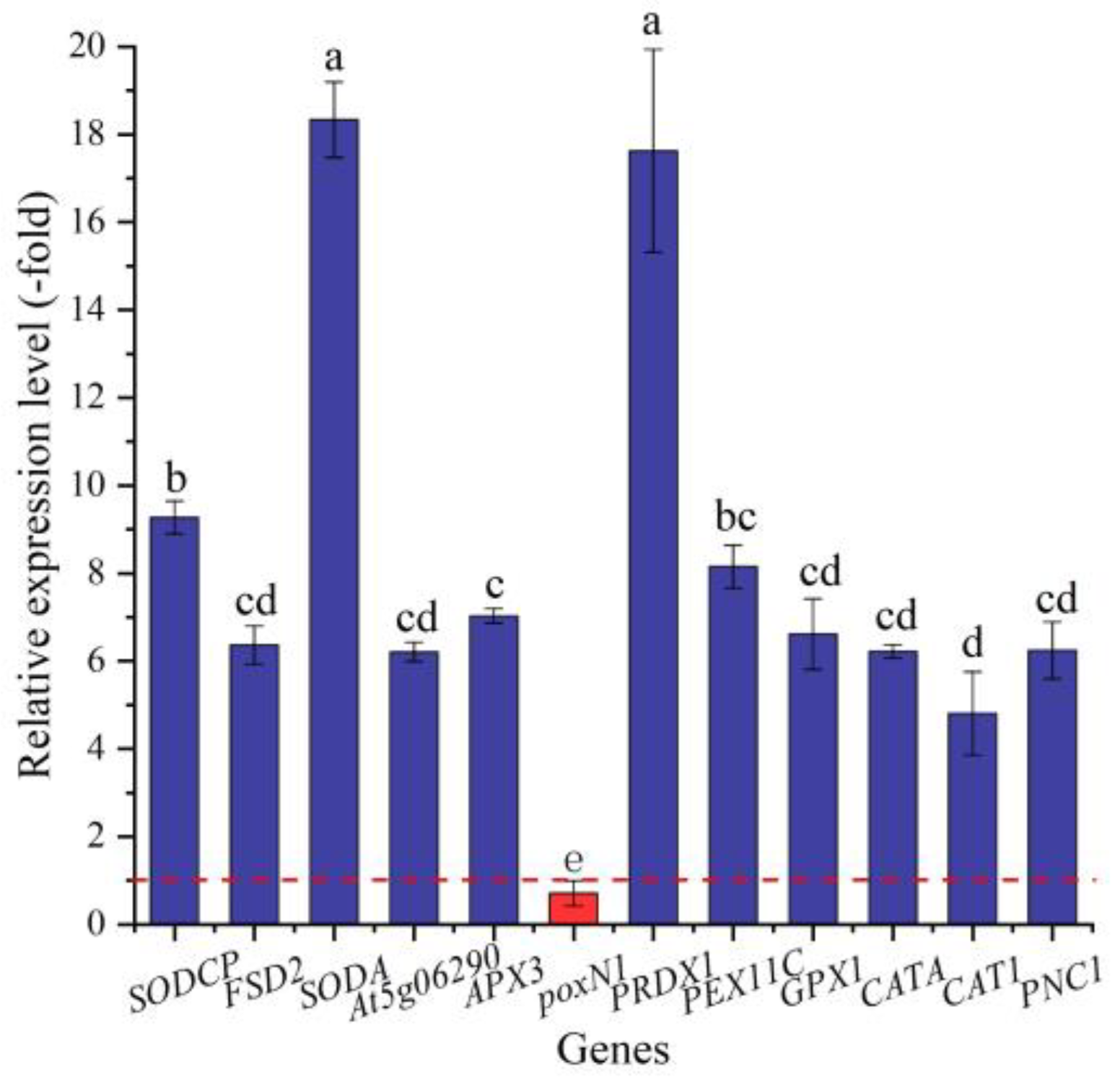
In this study, 23 genes (21 UR, 2 DR) directly associated with antioxidant enzymes, including SOD (SODCP, FSD2, and SODA), POD (e.g., At5g06290, PNC1, and APX3), and CAT (CATA, CAT1, and PNC1) (Table 3), were screened from the 146 DEGs involved in stress response, and another 123 DEGs were directly associated with stress responses such as temperature, pathogens, and oxidative stresses (Table S1). The expression levels of 12 selected genes were validated by qRT-PCR, with 4.81- (CAT1) to 18.34-fold (SODA) UR for 11 genes and 0.71-fold (poxN1) DR at 300 mmol/L NaCl vs. CK (Figure 7). Meanwhile, the RELs were consistent with their RPKM values (Table 3).

Table 3.
Twenty-three genes directly associated with antioxidant enzymes at the 300 mmol/L NaCl vs. CK.
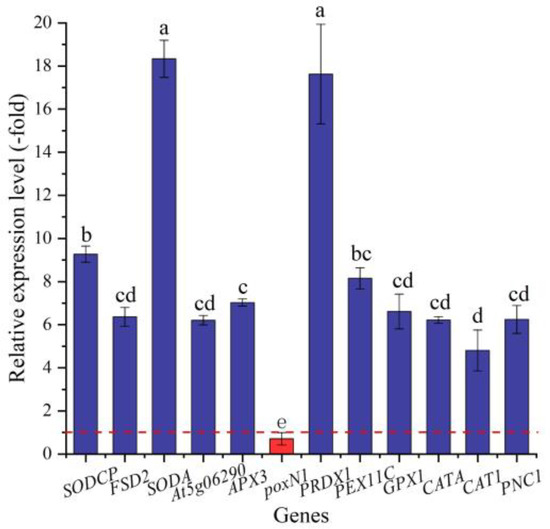
Figure 7.
The RELs of genes directly associated with antioxidant enzymes in A. venetum seeds at 300 mmol/L NaCl vs. CK (mean ± SD, n = 3). Different letters represent a significant difference (p < 0.05) among different genes. The red dotted line differentiates UR (>1) and DR (<1).
2.5.3. DEGs Directly Associated with Soluble Sugar and Protein Metabolism
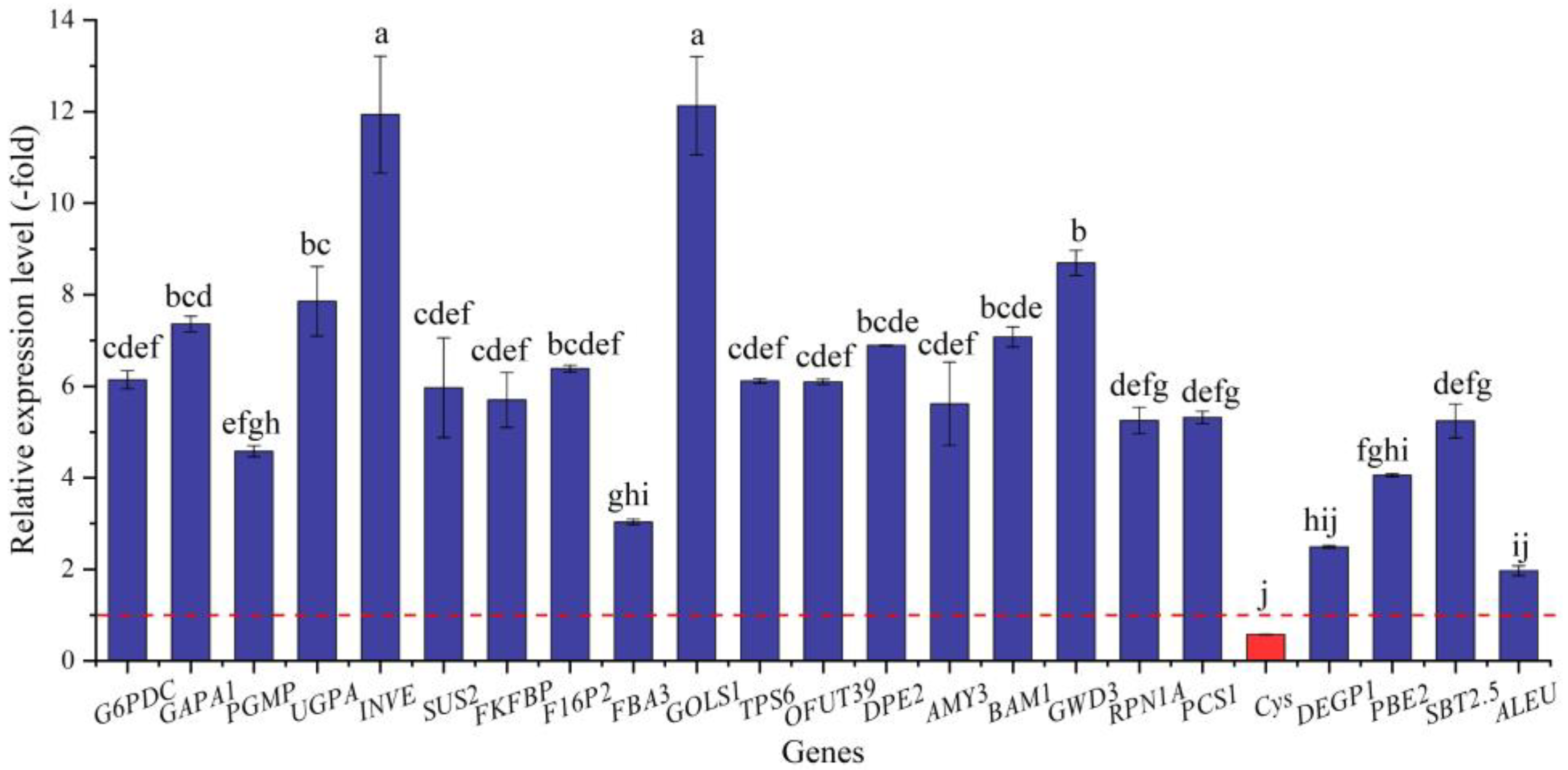
In this study, 79 genes (72 UR, 7 DR) directly associated with osmolytes, including soluble sugar (41 genes) and protein (38 genes), were screened from the 287 DEGs involved in primary metabolism; specifically, the 41 genes associated with soluble sugar metabolism included glucose (e.g., G6pc2, G6PDC, and GAPB), sucrose (INVE, SUS2, and SUS3), fructose (e.g., FKFBP, RHVI2, and FRK2), galactose (GOLS1 and GOLS2), trehalose (TPS6 and TPS7), fucose (OFUT19 and OFUT39), and starch (SBEI, DPE2, and AMY3), and the 38 genes associated with protein metabolism included PSMD2, RPN1A, RPN10, etc. (Table 4). The other 208 genes were associated with fatty acids, lipid metabolism, amino acids, etc. (Table S2). The expression levels of the 23 selected genes were validated by qRT-PCR, with 1.97- (ALEU) to 12.13-fold (GOLS1) UR for 22 genes and 0.57-fold (Cys) DR at 300 mmol/L NaCl vs. CK (Figure 8). Meanwhile, the RELs were consistent with their RPKM values (Table 4).

Table 4.
Seventy-nine DEGs directly associated with soluble sugar and protein metabolism at 300 mmol/L NaCl vs. CK.
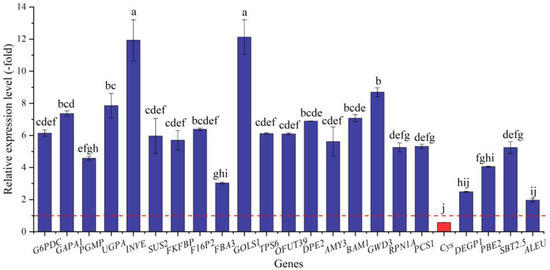
Figure 8.
The RELs of genes directly associated with soluble sugar and protein metabolism in A. venetum seeds at 300 mmol/L NaCl vs. CK (mean ± SD, n = 3). Different letters represent a significant difference (p < 0.05) among different genes. The red dotted line differentiates UR (>1) and DR (<1).
2.5.4. DEGs Directly Associated with Cell Morphogenesis for Seed Germination
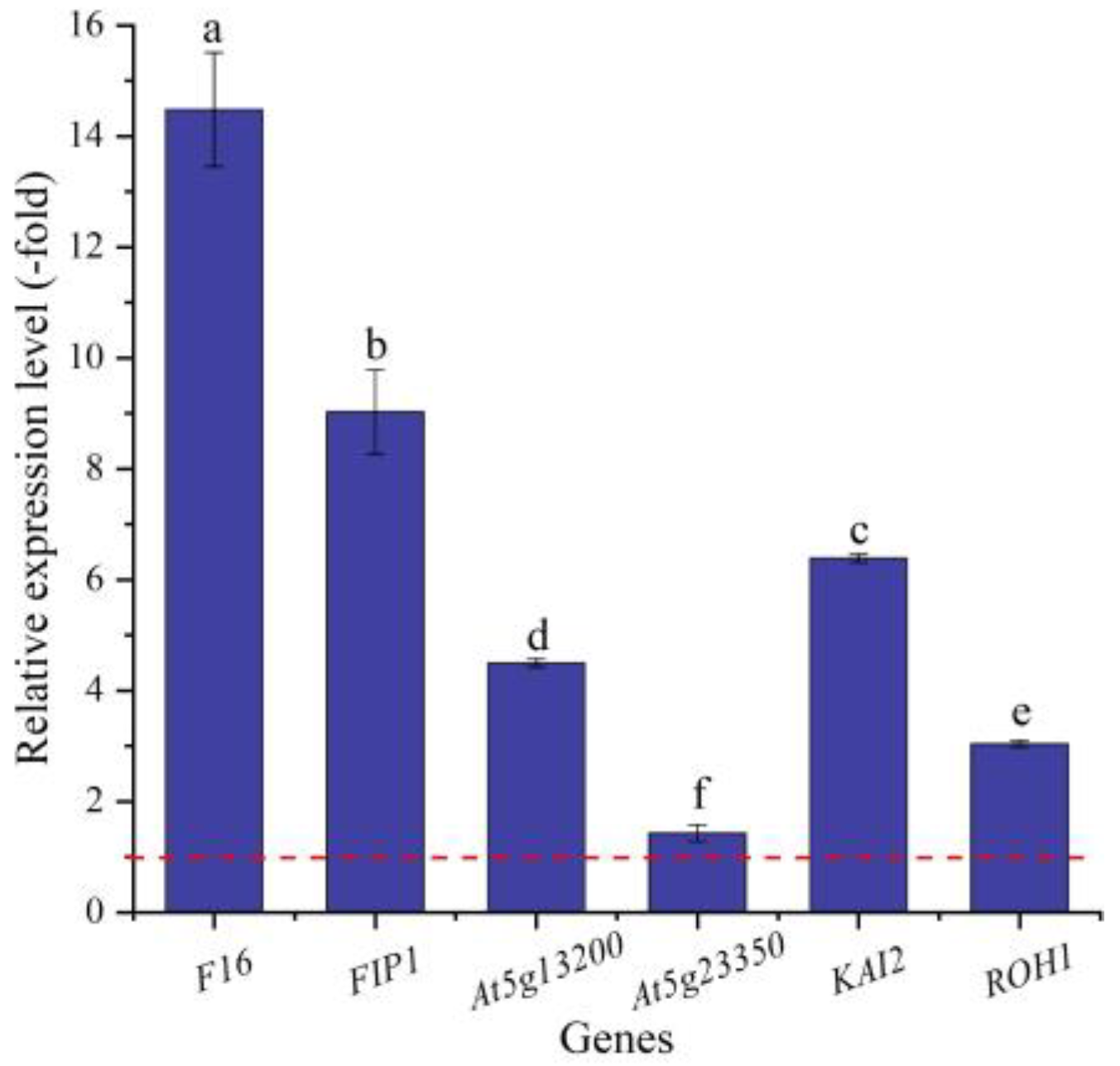
In this study, six genes (5 UR, 1 DR) directly associated with seed germination (i.e., F16, FIP1, At5g13200, At5g23350, KAI2, and ROH1) (Table 5) were screened from the 156 DEGs involved in cell morphogenesis. The other 150 genes were associated with flower development, cell wall organization, programmed cell death, etc. (Table S3). The expression levels of the six genes were validated by qRT-PCR, with 1.42- (At5g23350) to 14.48-fold (F16) UR at 300 mmol/L NaCl vs. CK (Figure 9). Meanwhile, the RELs were almost consistent with their RPKM values, except for the At5g23350 gene (Table 5).

Table 5.
Six DEGs directly associated with cell morphogenesis for seed germination at 300 mmol/L NaCl vs. CK.
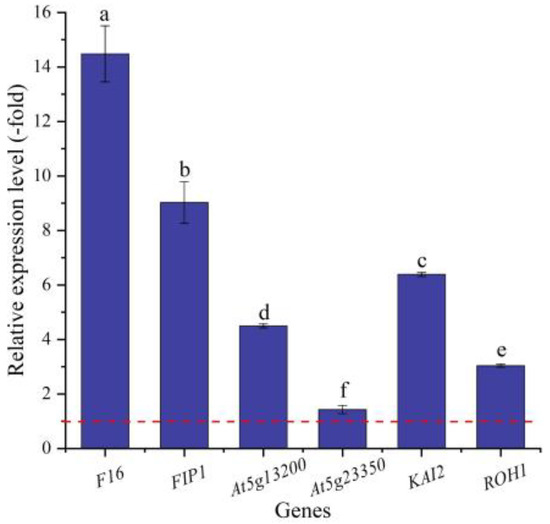
Figure 9.
The RELs of genes directly associated with cell morphogenesis for seed germination in A. venetum seeds at 300 mmol/L NaCl vs. CK (mean ± SD, n = 3). Different letters represent a significant difference (p < 0.05) among different genes. The red dotted line differentiates UR (>1) and DR (<1).
2.5.5. TFs Directly Associated with Stress Response and Seed Germination
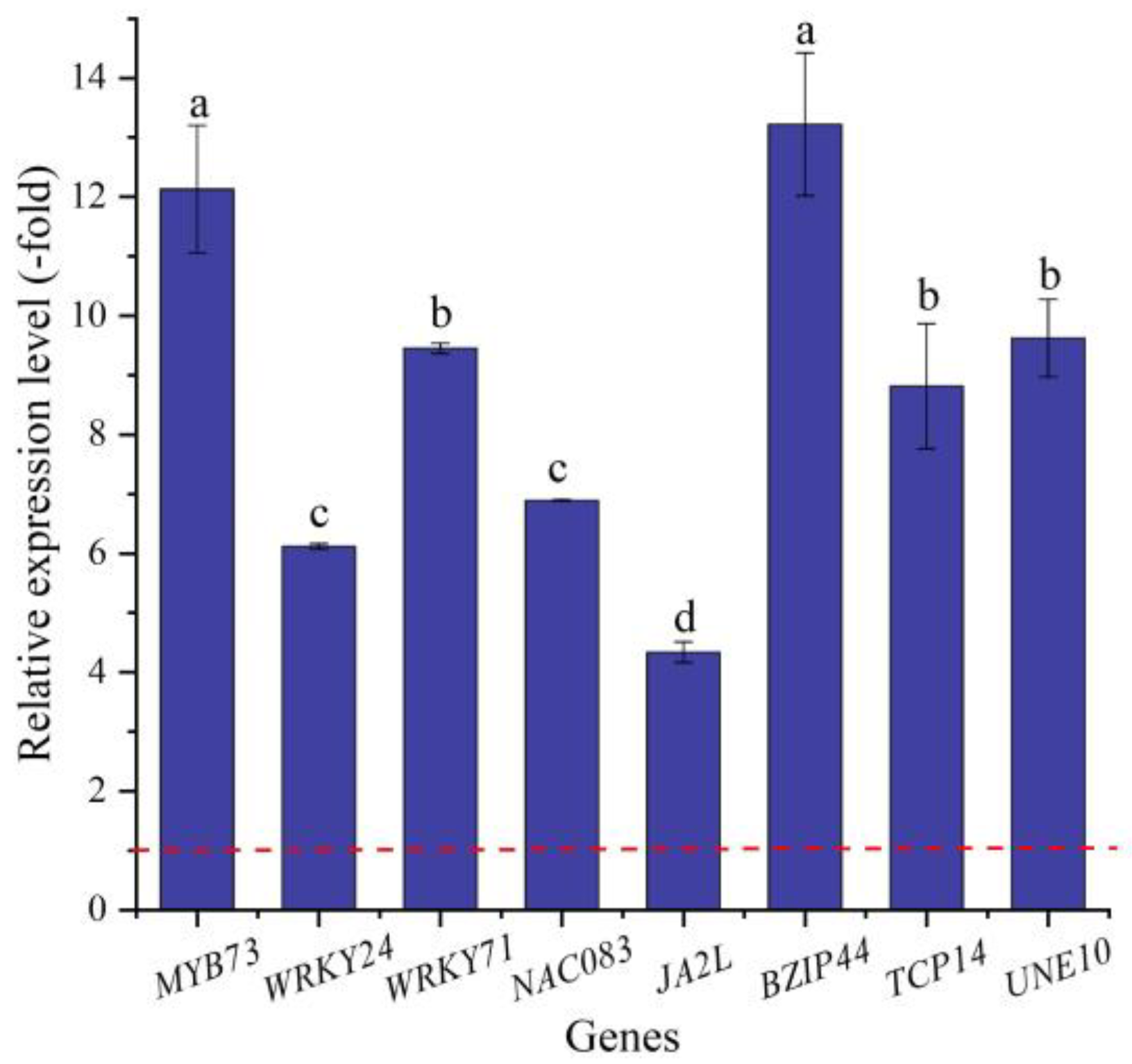
In this study, 16 TFs (15 UR, 1 DR) directly associated with stress response and seed germination, including MYB (MYB73 and MYB1R1), WRKY (e.g., WRKY4, WRKY23, and WRKY24), NAC (e.g., NAC083, NAC091, and NAC100), BZIP44, TCP14, and UNE10 (Table 6), were screened from the 62 TFs; the other 46 TFs were associated with biotic stress, the regulation of transcription by RNA polymerase II, flower development, etc. (Table S4). The expression levels of eight selected genes were validated by qRT-PCR, with 4.33- (JA2L) to 12.13-fold (MYB73) UR at 300 mmol/L NaCl vs. CK (Figure 10). Meanwhile, the RELs were consistent with their RPKM values (Table 6).

Table 6.
Sixteen TFs directly associated with stress response and seed germination at 300 mmol/L NaCl vs. CK.
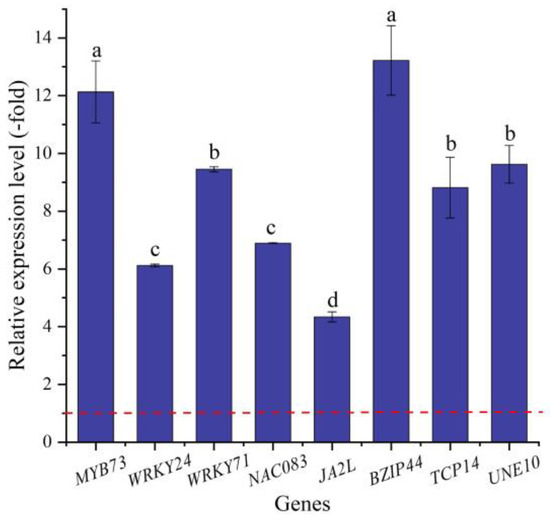
Figure 10.
The RELs of TFs directly associated with stress response and seed germination in A. venetum seeds at 300 mmol/L NaCl vs. CK (mean ± SD, n = 3). Different letters represent a significant difference (p < 0.05) among different genes. The red dotted line differentiates UR (>1) and DR (<1).
2.5.6. DEGs Directly Associated with Hormone Response
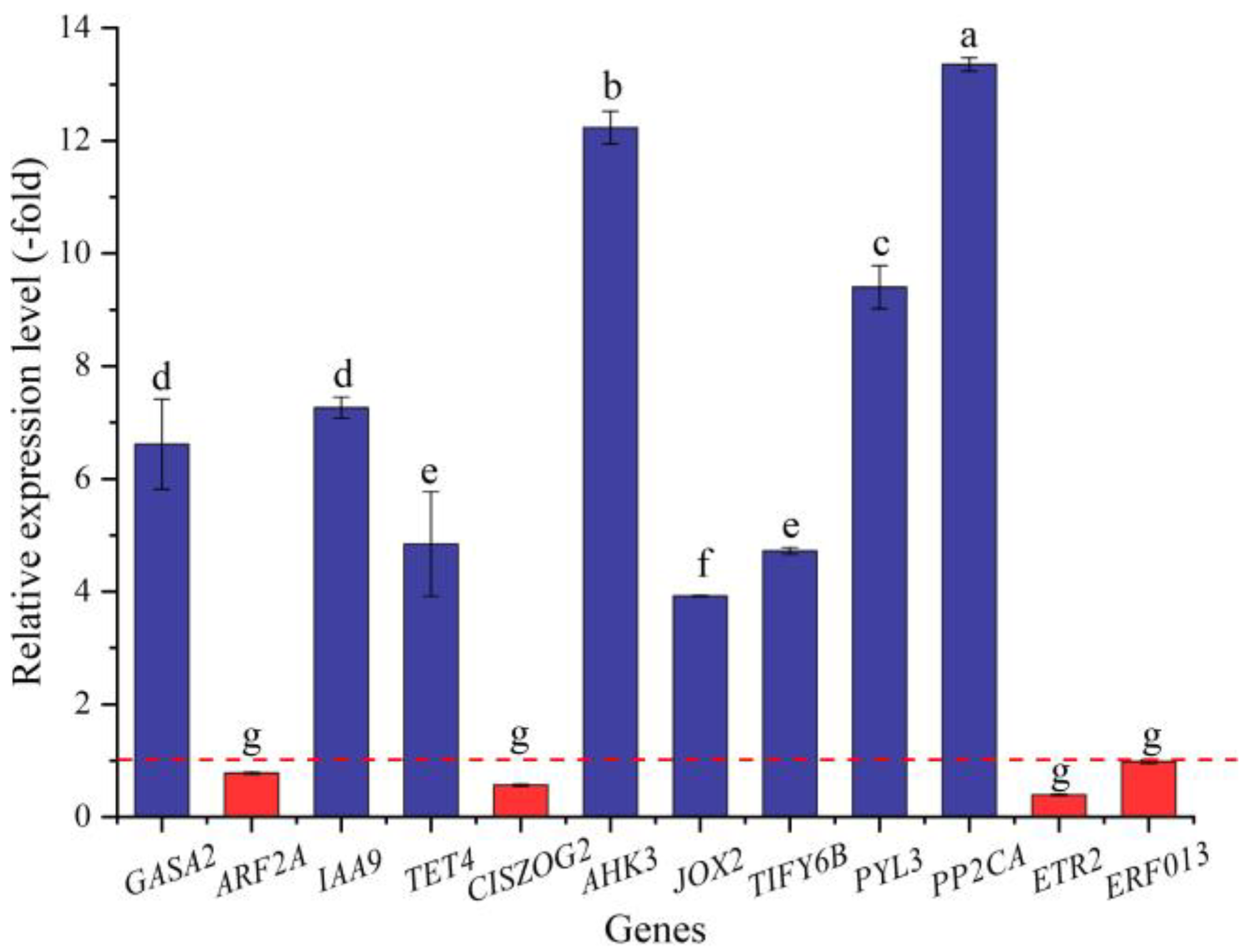
In this study, 36 genes (30 UR, 6 DR) directly associated with hormone response, including GA (GASA2), IAA (e.g., ABP19A, AUX22D, and AUX12KD), CTK (CISZOG2, AHK3, and ARR4), JA (JOX2 and TIFY6B), ABA (e.g., PYL3, PYL4, and GRDP1), and ETH (e.g., RAV1, ETR2, and ERF4) response (Table 7), were screened from the 173 DEGs involved in bio-signaling. The other 137 genes were associated with other bio-signals such as protein kinases, protein phosphatase, calcium sensors, etc. (Table S5). The expression levels of the 12 selected genes were validated by qRT-PCR, with 3.92- (JOX2) to 13.36-fold (PP2CA) UR for eight genes and 0.39- (ETR2) to 0.98-fold (ERF013) DR for four genes at 300 mmol/L NaCl vs. CK (Figure 11). Meanwhile, the RELs were almost consistent with their RPKM values (Table 7).

Table 7.
Thirty-six DEGs directly associated with hormone response at 300 mmol/L NaCl vs. CK.
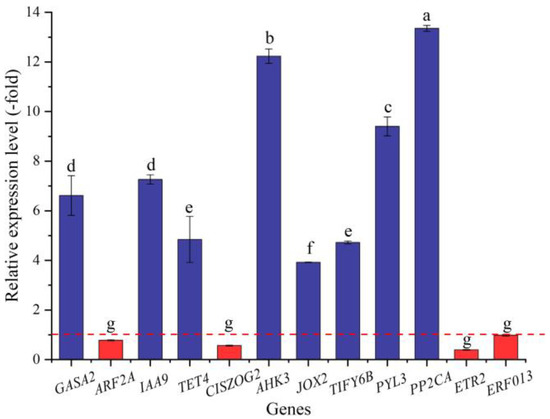
Figure 11.
The RELs of genes directly associated with hormone response in A. venetum seeds at 300 mmol/L NaCl vs. CK (mean ± SD, n = 3). Different letters represent a significant difference (p < 0.05) among different genes. The red dotted line differentiates UR (>1) and DR (<1).
2.5.7. DEGs Directly Associated with Ion Transport
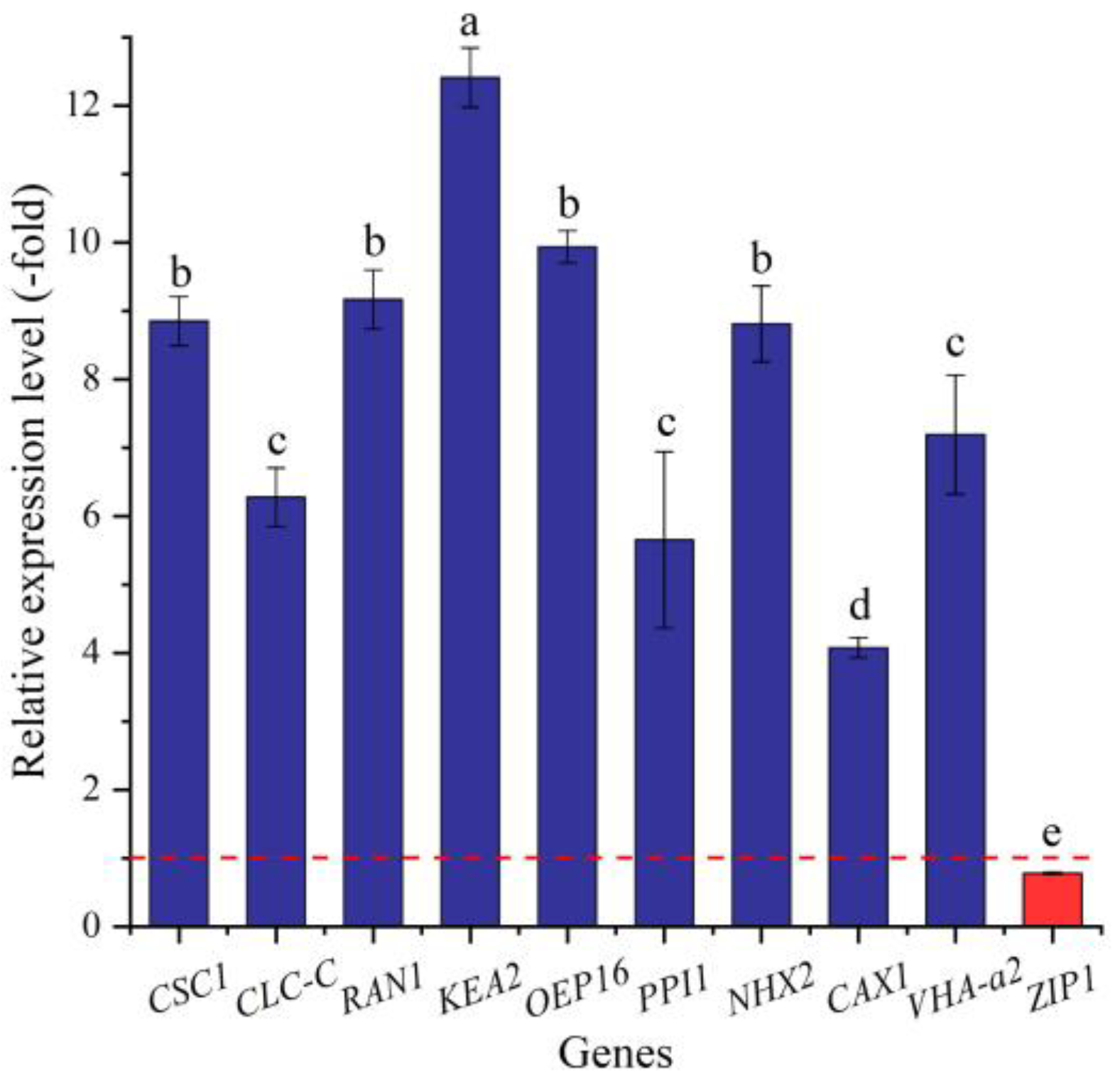
In this study, 34 genes (27 UR, 7 DR) directly associated with ion transport (e.g., AHA10, CSC1, and CLC-B) (Table 8) were screened from the 144 DEGs involved in transport, and the other 110 genes were associated with other kinds of transport such as sugar transport, protein transport, amino acid transport, etc. (Table S6). The expression levels of the 10 selected genes were validated by qRT-PCR, with 4.07- (CAX1) to 12.41-fold (KEA2) UR for nine genes and 0.78-fold (ZIP1) DR at 300 mmol/L NaCl vs. CK (Figure 12). Meanwhile, the RELs were consistent with their RPKM values (Table 8).

Table 8.
Thirty-four DEGs directly associated with ion transport at 300 mmol/L NaCl vs. CK.
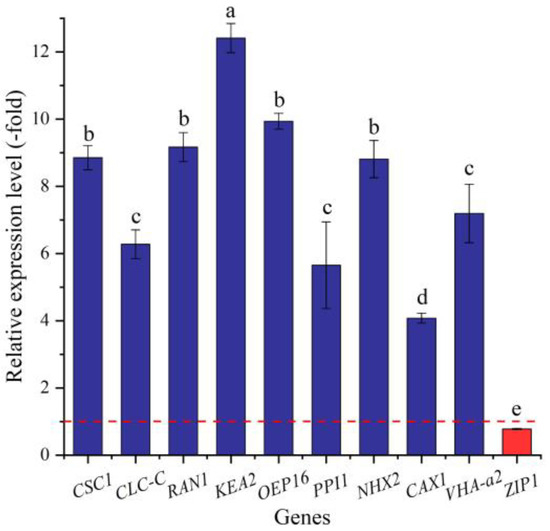
Figure 12.
The RELs of genes directly associated with ion transport in A. venetum seeds at 300 mmol/L NaCl vs. CK (mean ± SD, n = 3). Different letters represent a significant difference (p < 0.05) among different genes. The red dotted line differentiates UR (>1) and DR (<1).
2.5.8. DEGs Associated with Other Biological Functions
There are 490 DEGs involved in other biological functions, including photosynthesis and energy (125 genes, Table S7), secondary metabolism (58 genes, Table S8), polynucleotide metabolism (21 genes, Table S9), and translation (286 genes, Table S10). These genes may also participate in the seed germination of A. venetum in response to salt stress.
3. Discussion
Under both natural and agricultural conditions, plants are frequently exposed to unfavorable environments such as salt stress, extreme temperatures, and drought stress, which restrict plant distribution and reproduction, while plant adaptations that are genetically determined confer stress resistance [31]. Salt stress is a major abiotic stress that hinders seed germination, growth and development, and flowering and fruiting by driving various physiological and biochemical changes such as nutrition uptake, photosynthesis, and cellular metabolism [32,33,34,35,36,37]. In this study, we found that salt stress significantly affected the seed germination of A. venetum by changing the activity of antioxidant enzymes (i.e., SOD, POD, CAT, and APX), the content of osmolytes (i.e., soluble sugar, protein, MDA, and Pro), and the expression levels of genes associated with salt-stress response and seed germination.
Extensive experiments have demonstrated that stress resistance in response to salt stress is involved in the changes in physiological parameters (e.g., seed germination rate, plant growth, and flower development), antioxidant enzyme activities (e.g., SOD, POD, and CAT), and osmolyte contents (e.g., soluble protein, MDA, and Pro) [23,38]. For example, the seed germination of A. venetum decreases sharply under salt stress [11,12,13,14]. Salt stress causes oxidative stress that is responsible for the generation of reactive oxygen species (ROS), which are highly reactive and cause damage to biomolecules (e.g., lipids, proteins, and nucleic acids). To minimize the effect of oxidative stress, the ROS generation and increased activity of many antioxidant enzymes (e.g., SOD, CAT, and APX) during salt stress can protect plants from damage [23,39,40,41,42,43,44]. Under salt stress, ion imbalance and water deficiency induce osmotic stress, which triggers osmotic signaling pathways that regulate related genes’ expression, the enzyme activity of osmolyte biosynthesis, and the contents of osmolytes (e.g., soluble sugar, MDA, and Pro) [17,24,45].
In this study, we found that a low NaCl concentration (50 mmol/L) could promote the seed germination of A. venetum by adjusting the antioxidant enzyme activities and osmolyte contents. Actually, salt response is not only involved in the changes in phenotypes, antioxidant enzymes, and osmolytes but also in transcriptional alternations [46,47]. Here, 1487 DEGs (1293 UR and 194 DR) were observed during the seed germination of A. venetum at 300 mmol/L NaCl vs. CK, with 11 categories classified, including response to salt, stress response, primary metabolism, cell morphogenesis, TFs, bio-signaling, transport, photosynthesis and energy, secondary metabolism, polynucleotide metabolism, and translation.
Specifically, a total of 29 genes were directly associated with salt stress. For example, ALDH7B4 is activated by high salinity, dehydration, and ABA in a tissue-specific manner [48]; annexin (ANNs) in Arabidopsis is regulated by exposure to salt, drought, and extreme temperature conditions [49]; and RD22 is induced by salt stress and water deficit during the early and middle stages of seed development [50]. CAMBP25 is induced in Arabidopsis seedlings exposed to high salinity, dehydration, and low temperature [51]. PCC13 is abundantly expressed in dried leaves and abscisic acid-treated dried callus and is involved in the response to salt stress [52]; RBG7 plays a role in altering the germination and seedling growth of Arabidopsis under various stresses [53]; IQM4 plays a key role in modulating the responses to salt, ABA, and osmotic stress during seed germination and post-germination growth [54]; NFD4 mediates the transport of Na+ and K+ into the vacuole, influences plant development, and contributes to salt tolerance [55]; WSD1 is involved in the accumulation of wax esters in response to salt, drought, and ABA [56]; and At2g40140 plays important roles in modulating the tolerance of Arabidopsis to salt stress [57]. In this study, most of the genes involved in salt stress were UR, which may play a determining role in obtaining the ability of seed germination of A. venetum in saline–alkaline soils.
The primary effect of salt stress is hyperionic and hyperosmotic stresses, as well as, in severe cases, oxidative stress in plants. Oxidative stress is responsible for the generation of ROS, which are deleterious to plants. To minimize the effect of oxidative stress, plants have developed an efficient system of antioxidant enzymes (e.g., SOD, CAT, and APX) that can protect plants from damage [39,42,43,58,59,60,61]. Here, 23 genes were directly associated with antioxidant enzyme activities. For example, the SOD (SODCP, FSD2, and SODA) is a critical enzyme responsible for the elimination of superoxide radicals and is considered to be a key antioxidant in aerobic cells [23]; the POD (At5g06290, APX3, poxN1, PRDX1, PEX11C, and GPX1) catalyzes the reduction of hydrogen peroxide and plays a role in cell protection against oxidative stress by detoxifying peroxides [62]; and the CAT (CATA, CAT1, and PNC1) protects cells from the toxic effects of hydrogen peroxide [63]. In this study, most of the genes encoding SOD, POD, and CAT were UR, which is in accordance with the increased activities of SOD, POD, and CAT, which can reduce oxidative stress under salt stress.
Saline and alkaline ions can reduce the soil water potential, which makes it difficult for plants to absorb water, and then causes osmotic stress [35]. Previous studies have demonstrated that genes related to soluble sugar and protein metabolism play critical roles in response to salt stress in other plants. For example, the trehalose-6-phosphate synthase (TPS) gene plays a critical role in Citrullus lanatus’ response to salt stress [64]; the OsGolS1 gene is significantly up-regulated in Oryza sativa ssp. japonica under salt stress [65]; and the overexpression of MsTRX in tobacco induced the upregulation of beta-amylase 1 (BAM1) under salt stress [66]. In this study, 79 genes were directly associated with soluble sugar and protein metabolism, and 41 genes participate in soluble sugar metabolism. For example, G6PDC is involved in the synthesis of D-ribulose 5-phosphate from D-glucose 6-phosphate [67]; GAPA1 is involved in the synthesis of pyruvate from D-glyceraldehyde 3-phosphate [68]; PGMP is involved in both the breakdown and synthesis of glucose [69]; UGPA is involved in the UDP-glucose metabolic process [69]; INVE participates in cleaving sucrose into glucose and fructose and is associated with the assimilation of nitrogen to control the sucrose to hexose ratio [70]; SUS2 is involved in providing UDP-glucose and fructose for various metabolic pathways [71]; FKFBP is involved in the synthesis and degradation of fructose 2,6-bisphosphate [72]; F16P2 and FBA3 are involved in the fructose 1,6-bisphosphate metabolic process [69]; GOLS1 is involved in the synthesis of raffinose family oligosaccharides and promotes plant stress tolerance [73]; TPS6 is involved in the trehalose biosynthetic process [74]; OFUT39 is involved in the fucose metabolic process [75]; DPE2 is involved in the starch catabolic process and an intermediate on the pathway by which starch is converted to sucrose [76]; AMY3 is involved in stress-induced starch degradation [77]; BAM1 plays a role in the starch degradation and maltose metabolism [78]; and GWD3 is involved in starch degradation and mediates the incorporation of phosphate into starch-like phospho-alpha-glucan [79]. The other 38 genes participate in protein metabolism. For example, RPN1A is involved in the ATP-dependent degradation of ubiquitinated proteins [80]; PCS1 is involved in proteolysis and possesses peptidase activity toward casein [69,81]; Cys is involved in the protein catabolic process and shows the highest affinity for Pro [82]; DEGP1 is involved in the degradation of damaged proteins [83,84]; PBE2 is involved in the proteasomal protein catabolic process and shows an ATP-dependent proteolytic activity [69]; and SBT2.5 and ALEU are involved in proteolysis and show serine-type peptidase activity [69,85]. In this study, most of the genes involved in soluble sugar and protein metabolism were UR, which is in accordance with the increased content of soluble sugar and protein during seed germination.
For the biological functions of the six genes directly associated with seed germination, F16 is involved in seed trichome differentiation and expressed in the early phase of cotton fiber development [86]; GEM-like proteins (i.e., FIP1, At5g13200, and At5g23350) are involved in seed germination and play a novel role in regulating the reproductive development of plants [87]; KAI2 is involved in seed germination and seedling development [88]; and ROH1 is involved in seed coat development [89]. Thus, the differential expression of these genes could play important roles in seed germination under salt stress.
TFs are emerged as key regulators in various signaling networks and play significant roles by improving the growth and development of plants under stress conditions [90]. Previous studies have demonstrated that genes related to TFs play critical roles in response to salt stress in other plants. For example, the WRKY TFs of cotton play a significant role in the regulation of abiotic stresses (i.e., salt, drought, and extreme temperatures) [91]; MYB TFs are involved in the responses to different abiotic stresses, such as salt, cold, and drought, and the FvMYB82 gene probably plays an important role in the response to salt and cold stresses in Arabidopsis thaliana by regulating downstream related genes [92]; and NAC TFs play vital roles in plant development and responses to various abiotic stresses, and the ThNAC4 gene of Tamarix hispida is involved in salt and osmotic stress tolerance [93]. In this study, 16 TFs were directly associated with stress response and seed germination. Specifically for the stress response, MYB73 is involved in the salt-stress response [94]; WRKY TFs (WRKY24 and WRKY71) play critical regulatory roles in the plant alkaline metal (sodium) salt-stress response [95]; NAC083 is involved in xylem vessel formation and mediates signaling crosstalk between the salt-stress response and leaf-aging process [96]; and JA2L with MYC2 forms a transcription module that regulates wounding-responsive genes [97]. For seed germination, BZIP44 is involved in the positive regulation of seed germination through MAN7 gene activation [98]; TCP14 is involved in the regulation of seed germination and regulates the activation of embryonic growth potential during seed germination [99]; and UNE10 negatively regulates seed germination [100]. Here, their up-regulation may play a critical role in conferring the seed germination of A. venetum under salt stress.
As is known, endogenous hormones play critical roles in plant growth and development. Previous studies have demonstrated that genes related to hormone signaling play critical roles in response to salt stress in other plants. For example, SmGASA4 was found to be positively regulated by Gibberellin (GA) and significantly enhanced plant resistance to salt, drought, and paclobutrazol (PBZ) stress in Salvia miltiorrhiza [101]; Aux/IAA proteins in auxin and ARF transcription factors directly regulate auxin-responsive gene expression and OsIAA24 and OsIAA20 are up-regulated in rice under high salt stress [102]; genes related to Cytokinin (CK) and ethylene (ET) were involved in alleviating the root damage of Tamarix ramosissima under NaCl stress [103]. In this study, 36 genes were directly associated with hormone response. Specifically, GASA2 plays a role in the promotion of GA responses during seed germination, flowering, and seed maturation [104]; ARF2A, IAA9, and TET4 act as auxin-responsive genes and regulate vegetative growth, lateral root formation, and flower organ senescence [105,106]; CISZOG2 may regulate active versus storage forms of cytokinin (CTK) and can have an impact on seed growth [107]; AHK3 acts as a redundant positive regulator of CTK signaling that regulates many developmental processes such as seed germination, cell division, and shoot promotion [108]; JOX2 prevents the over-accumulation of jasmonate (JA) under stress responses [109]; TIFY6B acts as a repressor of JA responses and is involved in the regulation of defense responses [110]; PYL3 is involved in ABA signaling during seed germination and abiotic stress responses [111]; PP2CA acts as a major negative regulator of ABA responses during seed germination and cold acclimation [112]; ETR2 acts as a redundant negative regulator of ethylene (ETH) signaling [113]; and ERF013 may be involved in the regulation of gene expression by stress factors and by the components of stress signal transduction pathways [114]. Their differential expression may also play critical roles in promoting the seed germination of A. venetum.
Saline and alkaline stress can induce ion toxicity in plants when too many toxic ions enter plant cells, which can harm the plant cytoplasm and organelles; among them, Na+ is the main toxicity ion due to the similarity in size of the hydrated ionic radii of Na+ and K+, which makes them difficult to discriminate [115]. The salt overly sensitive (SOS) regulatory pathway regulates ion homeostasis by modulating Na+/H+ antiporter activity during salt stress [116]. Previous studies have demonstrated that genes related to ion transport play critical roles in the response to salt stress in other plants. For example, CLC-c is involved in the response to salt stress tolerance and seed germination in Gossypium hirsutum [117]; K+ efflux transporters (KEAs) were expressed under abiotic stress (salt, heat, and drought) in Cajanus cajan [118]; the Zipper (Zip) gene family participates in plant growth and development and the ability to cope with outside environment stresses, which may potentially regulate seed germination and stress resistance in Miscanthus sinensis [119]. In this study, 34 genes were directly associated with ion transport. For example, CSC1 acts as an osmosensitive calcium-permeable cation channel and is activated by hyperosmotic shock after NaCl treatment [120,121]; CLC-C is involved in ion transmembrane transport [122]; RAN1 is involved in copper import into the cell and acts by delivering copper to create functional hormone receptors [123]; KEA2 transports K+ and Cs+ preferentially relative to Na+ or Li+ [124]; OEP16 is involved in ion transport and acts as a voltage-dependent high-conductance channel [125]; PPI1 is involved in proton transport and promotes AHA1 plasma membrane ATPase activity by binding to a site different from the 14-3-3 binding site [126]; NHX2 is involved in the vacuolar ion compartmentalization necessary for cell volume regulation and cytoplasmic Na+ detoxification [127]; CAX1 translocates Ca2+ and other metal ions into vacuoles using the proton gradient formed by H+-ATPase and H+-pyrophosphatase [128]; VHA-a2 is involved in vacuolar nutrient storage and in the tolerance to some toxic ions, as well as catalyzing the translocation of protons across membranes [129]; and ZIP1 mediates zinc uptake from the rhizosphere and may also transport copper and cadmium ions [130]. Their differential expression may also play critical roles in promoting the seed germination of A. venetum under salt stress.
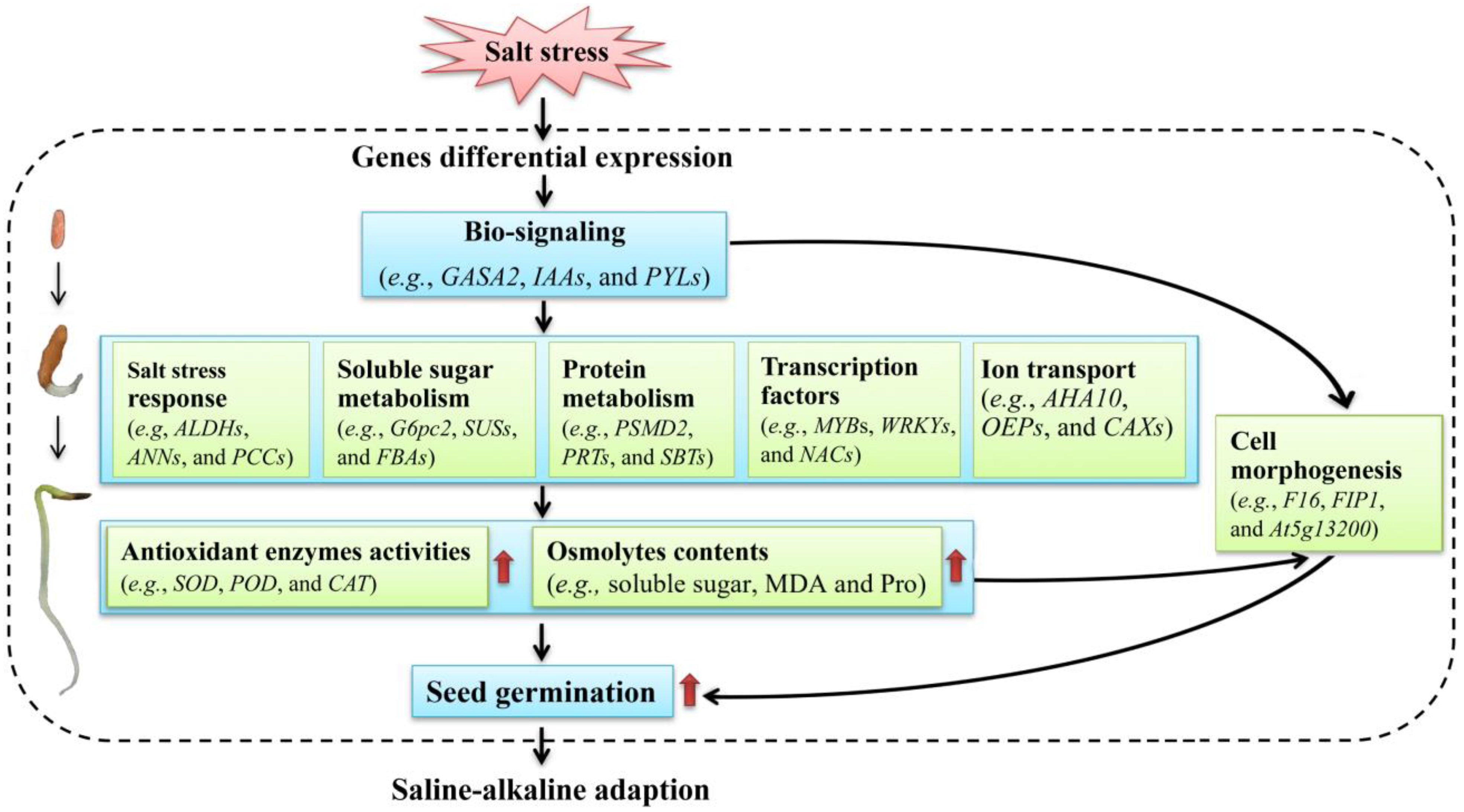
Based on the above studies on the physiological and transcriptional changes, a model of the low-salt-stress-enhanced seed germination of A. venetum is proposed (Figure 13). Briefly, when seeds are stimulated by salt stress, gene regulation is performed and related genes are differentially expressed; then, bio-signaling such as the hormone response (e.g., GASA2, IAAs, and PYLs) is generated; subsequently, the genes related to salt-stress responses (e.g., ALDHs, ANNs, and PCCs), soluble sugar metabolism (e.g., G6pc2, SUSs, and FBAs), protein metabolism (e.g., PSMD2, PRTs, and SBTs), TFs (e.g., MYBs, WRKYs, and NACs), and ion transport (e.g., AHA10, OEPs, and CAXs) are up-regulated, which promotes the antioxidant enzyme activities (e.g., SOD, POD, and CAT) and osmolyte contents (e.g., soluble sugar, MDA, and Pro) that protect the cells from salt-stress-induced membrane injury; finally, these changes are in favor of building the cell morphogenesis, enhancing seed germination, and conferring the ability to adapt to the saline–alkaline environment.
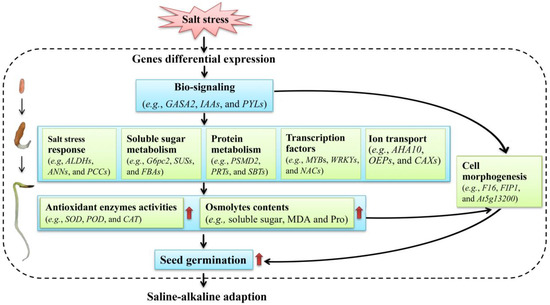
Figure 13.
A proposed model of the low-salt-stress-enhanced seed germination of A. venetum.
4. Materials and Methods
4.1. Plant Materials
Mature seeds of Apocynum venetum L. were collected from a resource nursery located at Alxa Left Banner, China (1050 m a.s.l.; E 103°42′14.77″, N 38°18′5.62″) in September 2020. Seeds were air dried at room temperature, stored at 4 °C in air-tight bags in the dark for two months, then cleaned with tap water and successively immersed in 50 °C water for 15 min and sanitized in 70% ethanol for 20 s. After being rinsed with sterile water 5 times, 30 seeds were sown in a Petri dish (15 cm diameter, two layers of gauze on the bottom) containing 10 mL of NaCl solution with 0 (CK), 50, 100, 150, 200, and 300 mmol/L. These doses were selected based on the published literature [11,16,17]. Each treatment had 40 independent biological replicates, which were made in 40 Petri dishes (30 seeds per dish).
4.2. Measurement of Germination Rate
After germination in a dark growth chamber at 22 °C for 3 days, the seed germination rate was measured. Three seeds were randomly collected from the 40 independent biological replicates for each NaCl treatment.
4.3. Determination of Antioxidant Enzyme Activities
The activities of 4 antioxidant enzymes including SOD, POD, CAT, and APX in seed embryos were determined using a spectrometer (Evolution 201, Thermo Scientific, Waltham, MA, USA). Briefly, SOD activity was determined based on the ability to inhibit the photochemical reduction of nitro blue tetrazolium chloride: extracts (100 μL) were added to the reaction, and the absorbance reading was taken at 560 nm [131]. POD activity was determined by the guaiacol colorimetric method: extracts (1000 μL) were added to the reaction and the absorbance reading was taken at 470 nm [132]. CAT activity was determined by the UV absorption method: extracts (100 μL) were added to the reaction and the absorbance reading was taken at 240 nm [133]. APX activity was determined based on the decrease in the absorbance of the oxidized ascorbate: extracts (100 μL) were added to the reaction and the absorbance reading was taken at 290 nm [134]. Each determination contained three biological replicates (a total of 120 samples = 3 seeds × 40 independent biological replicates) and each biological replicate had three technical replicates.
4.4. Determination of Osmolytes Content
The contents of 4 osmolytes including soluble sugar, protein, MDA, and Pro in seed embryos were determined using a spectrometer (Evolution 201, Thermo Scientific, Waltham, MA, USA). Briefly, the soluble sugar content was determined by the phenolsulfuric acid method: extracts (1000 μL) were added to the reaction and the absorbance reading was taken at 485 nm [135]. Protein content was determined by the comassie brilliant blue colorimetric method: extracts (800 μL) were added to the reaction and the absorbance reading was taken at 595 nm [136]. MDA content was determined by the thiobarbituric acid reaction: extracts (7000 μL) were added to the reaction and absorbance readings were taken at 532, 600, and 450 nm [137]. Pro content was determined by the sulfosalicylic acid-acid ninhydrin method: extracts (3000 μL) were added to the reaction and the absorbance reading was taken at 520 nm [138]. Each determination contained three biological replicates (a total of 120 samples = 3 seeds × 40 independent biological replicates) and each biological replicate had three technical replicates.
4.5. Transcriptomic Analysis
4.5.1. RNA Extraction, cDNA Library Construction, and Illumina Sequencing
Total RNA samples of CK and 300 mmol/L NaCl treatments with 3×40 biological replicates were extracted using an RNA kit (R6827, Omega Bio-Tek, Norcross, GA, USA). The RNA amount was quantified using a NanoDrop ND1000 spectrophotometer (Nanodrop Technologies, Wilmington, NC, USA), and the quality was determined using an Agilent 2100 Bioanalyzer (Agilent Technologies, Palo Alto, CA, USA). One cDNA library was constructed from the total RNA samples of CK and 300 mmol/L NaCl treatments, and the processes of enrichment, fragmentation, reverse transcription, the synthesis of the second-strand cDNA, and the purification of cDNA fragments were conducted as previously described [139]. Reads were generated using an Illumina HiSeqTM 4000 platform (Gene Denovo Biotechnology Co., Ltd., Guangzhou, China).
4.5.2. Reads Filtration, Assembly, Unigene Expression Analysis, and Basic Annotation
Raw reads were filtered using a FASTQ system to obtain high-quality clean reads by removing reads containing adapters, more than 10% of unknown nucleotides (N), and more than 50% of low-quality (Q-value ≤ 20) bases [140]. Clean reads were assembled using Trinity [141]. The expression level of each transcript was normalized to the RPKM values. The differential expression analysis of transcripts was performed using DESeq2 software [142] between the CK and 300 mmol/L NaCl treatments with the criteria of the false discovery rate (FDR) < 0.05 and |log2(fold-change)| > 1. The function of DEGs was annotated using BLAST against the databases, including Nr, KEGG, KOG, SwissProt, and GO, with an e-value ≤ 10−5 as a threshold [143].
4.5.3. qRT-PCR Validation
The primer sequences (Table S11) were designed via a primer blast in NCBI and synthesized by reverse transcription (Sangon Biotech Co., Ltd., Shanghai, China). First, cDNA was synthesized using an RT Kit (KR116, Tiangen, Beijing, China). PCR amplification was performed using a SuperReal PreMix (FP205, Tiangen, Beijing, China). The melting curve was analyzed at 72 °C for 34 s. The Actin gene was used as a reference control [14]. The RELs of genes were calculated using a 2−ΔΔCt method (Ct, cycle threshold value of target gene) according to the following formula [144]. The validation of qRT-PCR contained three biological replicates (a total of 120 samples = 3 seeds × 40 independent biological replicates) and each biological replicate had three technical replicates.
ΔCt Test gene = Ct Test gene − Ct Reference gene
ΔCt Control gene = Ct Control gene − Ct Reference gene
−ΔΔCt = −(ΔCt Test gene − ΔCt Control gene)
Relative gene expression fold (Test gene/Control gene) = 2−ΔΔCt
4.6. Statistical Analysis
SPSS 22.0 software was used for statistical analysis, and one-way analysis of variance (ANOVA) by Duncan’s test was performed for statistical comparisons with p < 0.05 considered statistically significant.
5. Conclusions
This research reveals that salt stress affects the seed germination of A. venetum. The seed germination of A. venetum is promoted at low NaCl concentrations such as 50 mmol/L, and significant changes in antioxidant enzyme activities, osmolyte contents, and genes expression levels in A. venetum play critical roles in regulating seed germination under different salt stresses. These findings indicate that A. venetum plants can adapt to salt stress in saline–alkaline soils by integrating physiological and transcriptional responses. The specific roles of key genes in conferring the ability to salt resistance will require further investigation.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms24043623/s1.
Author Contributions
X.L.: data curation, formal analysis, and writing—original draft preparation; J.L.: data curation, formal analysis, and validation; H.S.: formal analysis, methodology, and validation; P.S.: formal analysis and methodology; Z.Z.: formal analysis; M.L.: conceptualization, funding acquisition, and writing—review and editing; H.X.: resources, supervision, and writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the State Key Laboratory of Aridland Crop Science Gansu Agricultural University (GSCS-2021-Z03).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets are publicly available at NCBI (https://www.ncbi.nlm.nih.gov/bioproject/PRJNA847749, accessed on 2 August 2022), and Sequence Read Archive (SRA) accession: CK (SRR20341324, SRR20341330, and SRR20341336), and 300 mmol/L NaCl (SRR20341332, SRR20341333, and SRR20341334).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Editorial Committee of the Flora of China of Chinese Academy of Science. Flora of China; Science Press: Beijing, China, 1977; p. 157. (In Chinese) [Google Scholar]
- Gao, G.; Chen, P.; Chen, J.; Chen, K.; Wang, X.; Abubakar, A.S.; Liu, N.; Yu, C.; Zhu, A. Genomic survey, transcriptome, and metabolome analysis of Apocynum venetum and Apocynum hendersonii to reveal major flavonoid biosynthesis pathways. Metabolites 2019, 9, 296. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, C.; Zhang, Z.; Wang, J.; Wu, G.; Li, S. Comprehensive separation and identification of chemical constituents from Apocynum venetum leaves by high performance counter-current chromatography and high performance liquid chromatography coupled with mass spectrometry. J. Chromatogr. 2010, 878, 3149–3155. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Zhang, X.; Wang, T.; Hu, J. Botany, traditional uses, phytochemistry and pharmacology of Apocynum venetum L. (Luobuma): A review. J. Ethnopharmacol. 2012, 141, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Huang, G.; Tan, F.; Zhou, X.; Mu, J.; Zhao, X. In vitro analysis of antioxidant, anticancer, and bioactive components of Apocynum venetum tea extracts. J. Food Qual. 2019, 2019, 2465341. [Google Scholar] [CrossRef]
- Xu, Z.; Zhou, J.; Zhang, C.; Li, Y. Review of current research and utilization status of Apocynum venetum germplasm in China. Chin. Bull. Bot. 2018, 53, 382–390. [Google Scholar]
- Ning, J.F.; Zheng, Q.S.; Yang, S.H.; Zou, X.Z.; Sun, L.L.; Chen, Y. Impact of high salt stress on Apocynum venetum growth and ionic homeostasis. Chin. J. Appl. Ecol. 2010, 21, 325–330. [Google Scholar]
- Jiang, L.; She, C.W.; Tian, C.Y.; Tanveer, M.; Wang, L. Storage period and different abiotic factors regulate seed germination of two Apocynum species-cash crops in arid saline regions in the northwestern China. Front. Plant Sci. 2021, 12, 671157. [Google Scholar] [CrossRef]
- Ren, J.; Yu, F.K.; Tao, L. Research advances on the germination of desert plants under stress. Bull Bot. Res. 2011, 31, 121–128. [Google Scholar]
- Campobenedetto, C.; Grange, E.; Mannino, G.; van Arkel, J.; Beekwilder, J.; Karlova, R.; Garabello, C.; Contartese, V.; Bertea1, C.M. A biostimulant seed treatment improved heat stress tolerance during cucumber seed germination by acting on the antioxidant system and glyoxylate cycle. Front. Plant Sci. 2020, 11, 836. [Google Scholar] [CrossRef]
- Zhang, X.; Li, R.; Shi, F. Effects of salt stress on the seed germination of Apocynum venetum. Acta Sci. Univ. Nankaiensis 2007, 40, 13–18. [Google Scholar]
- Yu, D.H.; Xu, H.L.; Chang, S.L. Effect of NaCl stress on seed germination and seedling growth of Luobuma. Hubei Agric. Sci. 2008, 47, 772–775. [Google Scholar]
- Xu, Z.; Zhou, J.; Ren, T.; Du, H.; Liu, H.; Li, Y.; Zhang, C. Salt stress decreases seedling growth and development but increases quercetin and kaempferol content in Apocynum venetum. Plant Biol. 2020, 22, 813–821. [Google Scholar] [CrossRef]
- Wu, Z.B.; Chang, P.X.; Zhao, J.; Wang, W.S.; Cui, X.W.; Li, M.F. Physiological and transcriptional responses of seed germination to moderate drought in Apocynum venetum. Front. Ecol. Evol. 2022, 10, 975771. [Google Scholar] [CrossRef]
- Liu, Z.H.; Wang, N.; Chen, L.Y.; Geng, L. Measurement and analysis of physiological and biochemical indexes during natural aging on Apocynum seed. Seed 2015, 34, 43–46. [Google Scholar]
- Ning, J.F.; Zheng, Q.S.; Zou, X.Z.; Sun, L.L.; Yao, Y.; Chen, Y.; Wu, J.L.; Wei, L. PhysiologicaI responses of Apocynum venetum to different levels of salt stress. Chin. Bull. Bot. 2010, 45, 889–897. [Google Scholar]
- Fang, J.W.; Wu, Y.; Liu, Z.H. Effects of salt stress on seed germination and physiological characteristics of Apocynum venetum. Crops 2018, 4, 167–174. [Google Scholar]
- Yan, M.X.; Bai, R.; Cui, J.X.; Zhang, Y.Q.; Wang, C.F.; Wu, P.; Bing, Z.; Jin, B. Effects of mixed saline alkali stress on physiological characteristics of Apocynum venetum. Bull. Agric. Sci. Technol. 2021, 8, 153–157. [Google Scholar]
- Xu, Z.C.; Wang, M.; Ren, T.T.; Li, K.Y.; Li, Y.Q.; Marowa, P.; Zhang, C.S. Comparative transcriptome analysis reveals the molecular mechanism of salt tolerance in Apocynum venetum. Plant Physiol. Bioch. 2021, 167, 816–830. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Xu, H.L.; Shi, S.B.; Li, J.M. Effect of water supply, salinity and buried depth on the seed germination of Halimodendron halodendron and Alhagi sparsifolia. Arid Land Geogr. 2008, 31, 687–692. [Google Scholar]
- Khan, M.A.; Gul, B.; Weber, D.J. Effect of germination regulating chemicals on seed germination of Halogeton glomeratus for alleviation of salinity stress. Pak. J. Bot. 2009, 41, 1205–1212. [Google Scholar]
- Hu, R.H.; Liu, H.; Liu, F.H. Seed germination of hemp (Cannabis sativa L.) cultivars responds differently to the stress of salt type and concentration. Ind. Crops Prod. 2018, 123, 254–261. [Google Scholar] [CrossRef]
- Rasool, S.; Ahmad, A.; Siddiqi, T.O.; Ahmad, P. Changes in growth, lipid peroxidation and some key antioxidant enzymes in chickpea genotypes under salt stress. Acta Physiol. Plant. 2013, 35, 1039–1050. [Google Scholar] [CrossRef]
- Zhao, S.S.; Zhang, Q.K.; Liu, M.Y.; Zhou, H.P.; Ma, C.L.; Wang, P.P. Regulation of plant responses to salt stress. Int. J. Mol. Sci. 2021, 22, 4609. [Google Scholar] [CrossRef]
- Li, R.J.; Zhang, M.; Jiang, Z.P.; Huang, L.B.; Wang, B.S.; Ji, Y.H.; Fang, Y.M. Effects of NaCl stress on antioxidant enzyme activities and isoenzyme pattern of Broyssonetia papyrifera Plantlets. Sci. Silvae Sin. 2009, 45, 40–47. [Google Scholar]
- Guo, Q.S.; Wu, Y.G.; Lin, Y.F.; Zheng, H.Q. Effect of NaCl stress on growth and antioxidant systems of Pogostemon cablin. China J. Chin. Mater. Med. 2009, 34, 530–534. [Google Scholar]
- Chan, Z.; Loescher, W.; Grumet, R. Transcriptional variation in response to salt stress in commonly used Arabidopsis thaliana accessions. Plant Physiol. Biochem. 2013, 73, 189–201. [Google Scholar] [CrossRef]
- Vogel, J.T.; Zarka, D.G.; Van Buskirk, H.A.; Fowler, S.G.; Thomashow, M.F. Roles of the CBF2 and ZAT12 transcription factors in configuring the low temperature transcriptome of Arabidopsis. Plant J. 2005, 41, 195–211. [Google Scholar] [CrossRef]
- Van Buskirk, H.A.; Thomashow, M.F. Arabidopsis transcription factors regulating cold acclimation. Physiol. Plant. 2006, 126, 72–80. [Google Scholar] [CrossRef]
- Jiang, Y.; Deyholos, M.K. Comprehensive transcriptional profiling of NaCl-stressed Arabidopsis roots reveals novel classes of responsive genes. BMC Plant Biol. 2006, 6, 25. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Plant Physiology. In Responses and Adaptations to Abiotic Stress; Radin, J., Bressan, R., Drew, M., Hasegawa, P., Locy, R., Mickelbart, M., Salt, D., Eds.; Sinauer Associates Inc.: Sunderland, MA, USA, 2010; pp. 620–621. [Google Scholar]
- Quan, R.; Lin, H.; Mendoza, I.; Zhang, Y.; Cao, W.; Yang, Y.; Shang, M.; Chen, S.; Pardo, J.M.; Guo, Y. SCABP8/CBL10, a putative calcium sensor, interacts with the protein kinase SOS2 to protect Arabidopsis shoots from salt stress. Plant Cell 2007, 19, 1415–1431. [Google Scholar] [CrossRef]
- Park, H.J.; Kim, W.Y.; Yun, D.J. A role for GIGANTEA. Plant Signal. Behav. 2013, 8, e24820. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Hoehenwarter, W. Changes in the phosphosproteome and metabolome link early signaling events to rearrangement of photosynthesis and central metabolism in salinity and oxidative stress response in Arabidopsis. Plant Physiol. 2015, 69, 3021–3033. [Google Scholar] [CrossRef]
- Liu, D.; Ma, Y.; Rui, M.M.; Lv, X.C.; Chen, R.J.; Chen, X.Y.; Wang, Y.Z. Is high pH the key factor of alkali stress on plant growth and physiology? A case study with Wheat (Triticum aestivum L.) seedlings. Agronomy 2022, 12, 1820. [Google Scholar] [CrossRef]
- Van Zelm, E.; Zhang, Y.; Testerink, C. Salt tolerance mechanisms of plants. Annu. Rev. Plant Biol. 2020, 71, 403–433. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z. Plant abiotic stress: New insights into the factors that activate and modulate plant responses. J. Integr. Plant Biol. 2021, 63, 429–430. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Gao, J.; Wu, X.; Li, Q.; Wang, J.; Ding, P.; Lai, X. Effect of salt treatment on growth, isoenzymes and metabolites of Andrographis paniculata (Burm. f.) Nees. Acta Physiol. Plant. 2015, 37, 35. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Ann. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Tuteja, N.; Ahmad, P.; Panda, B.B.; Tuteja, R. Genotoxic stress in plants: Shedding light on DNA damage, repair and DNA repair helicases. Mutat. Res. 2009, 681, 134–149. [Google Scholar] [CrossRef]
- Zheng, Y.H.; Xu, X.B.; Wang, M.Y.; Zheng, X.H.; Li, Z.J.; Jiang, G.M. Responses of salt-tolerant and intolerant wheat genotypes to sodium chloride: Photosynthesis, antioxidants activities, and yield. Photosynthetica 2009, 47, 87–94. [Google Scholar] [CrossRef]
- Ahmad, P.; Prasad, M.N.V. Environmental Adaptations and Stress Tolerance in Plants in the Era of Climate Change; Springer Science Business Media, LLC: New York, NY, USA, 2012. [Google Scholar]
- Ahmad, P.; Prasad, M.N.V. Abiotic Stress Responses in Plants: Metabolism, Productivity and Sustainability; Springer Science Business Media, LLC: New York, NY, USA, 2012. [Google Scholar]
- Koca, H.; Ozdemir, F.; Turkan, I. Effect of salt stress on lipid peroxidation and superoxide dismutase and peroxidase activities of Lycopersicon esculentum and L. pennellii. Biol. Plant 2006, 50, 745–748. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, Y. Elucidating the molecular mechanisms mediating plant salt-stress responses. New Phytol. 2017, 217, 523–539. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Zhang, H.; Song, C.; Zhu, J.K.; Shabala, S. Mechanisms of plant responses and adaptation to soil salinity. Innovation 2020, 1, 100017. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.M.; Zhu, J.H.; Gong, Z.Z.; Zhu, J.K. Abiotic stress responses in plants. Nat. Rev. Genet. 2022, 23, 104–119. [Google Scholar] [CrossRef]
- Kirch, H.H.; Schlingensiepen, S.; Kotchoni, S.; Sunkar, R.; Bartels, D. Detailed expression analysis of selected genes of the aldehyde dehydrogenase (ALDH) gene superfamily in Arabidopsis thaliana. Plant Mol. Biol. 2005, 57, 315–332. [Google Scholar] [CrossRef] [PubMed]
- Cantero, A.; Barthakur, S.; Bushart, T.J.; Chou, S.; Morgan, R.O.; Fernandez, M.P.; Clark, G.B.; Roux, S.J. Expression profiling of the Arabidopsis annexin gene family during germination, de-etiolation and abiotic stress. Plant Physiol. Biochem. 2006, 44, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. The plant hormone abscisic acid mediates the drought-induced expression but not the seed-specific expression of rd22, a gene responsive to dehydration stress in Arabidopsis thaliana. Mol. Gen. Genet. 1993, 238, 17–25. [Google Scholar] [CrossRef]
- Perruc, E.; Charpenteau, M.; Ramirez, B.C.; Jauneau, A.; Galaud, J.P.; Ranjeva, R.; Ranty, B. A novel calmodulin-binding protein functions as a negative regulator of osmotic stress tolerance in Arabidopsis thaliana seedlings. Plant J. 2004, 38, 410–420. [Google Scholar] [CrossRef]
- Piatkowski, D.; Schneider, K.; Salamini, F.; Bartels, D. Characterization of five abscisic acid-responsive cDNA clones isolated from the desiccation-tolerant plant Craterostigma plantagineum and their relationship to other water-stress genes. Plant Physiol. 1990, 94, 1682–1688. [Google Scholar] [CrossRef]
- Kwak, K.J.; Kim, Y.O.; Kang, H. Characterization of transgenic Arabidopsis plants overexpressing GR-RBP4 under high salinity, dehydration, or cold stress. J. Exp. Bot. 2005, 56, 3007–3016. [Google Scholar] [CrossRef]
- Zhou, Y.P.; Wu, J.H.; Xiao, W.H.; Chen, W.; Chen, Q.H.; Fan, T.; Xie, C.P.; Tian, C.E. Arabidopsis IQM4, a novel calmodulin-binding protein, is involved with seed dormancy and germination in Arabidopsis. Front. Plant Sci. 2018, 9, 721. [Google Scholar] [CrossRef]
- Sottosanto, J.B.; Saranga, Y.; Blumwald, E. Impact of AtNHX1, a vacuolar Na+/H+ antiporter, upon gene expression during short- and long-term salt stress in Arabidopsis thaliana. BMC Plant Biol. 2007, 7, 18. [Google Scholar] [CrossRef] [PubMed]
- Patwari, P.; Salewski, V.; Gutbrod, K.; Kreszies, T.; Dresen-Scholz, B.; Peisker, H.; Steiner, U.; Meyer, A.J.; Schreiber, L.; Doermann, P. Surface wax esters contribute to drought tolerance in Arabidopsis. Plant J. 2019, 98, 727–744. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Jiang, H.; Xu, Y.; Li, H.; Wu, X.; Xie, Q.; Li, C. The CCCH-type zinc finger proteins AtSZF1 and AtSZF2 regulate salt stress responses in Arabidopsis. Plant Cell Physiol. 2007, 48, 1148–1158. [Google Scholar] [CrossRef]
- Ahmad, P.; Jhon, R.; Sarwat, M.; Umar, S. Responses of proline, lipid peroxidation and antioxidative enzymes in two varieties of Pisum sativum L. under salt stress. Int. J. Plant Prod. 2008, 2, 353–366. [Google Scholar]
- Ahmad, P.; Sarwat, M.; Sharma, S. Reactive oxygen species, antioxidants and signaling in plants. J. Plant Biol. 2008, 51, 167–173. [Google Scholar] [CrossRef]
- Ahmad, P.; Jaleel, C.A.; Salem, M.A.; Nabi, G.; Sharma, S. Roles of enzymatic and non-enzymatic antioxidants in plants during abiotic stress. Crit. Rev. Biotechnol. 2010, 30, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Azooz, M.M.; Youssef, A.M.; Ahmad, P. Evaluation of salicylic acid (SA) application on growth, osmotic solutes and antioxidant enzyme activities on broad bean seedlings grown under diluted seawater. Int. J. Plant. Physiol. Biochem. 2011, 3, 253–264. [Google Scholar]
- Broin, M.; Cuine, S.; Eymery, F.; Rey, P. The plastidic 2-cysteine peroxiredoxin is a target for a thioredoxin involved in the protection of the photosynthetic apparatus against oxidative damage. Plant Cell 2002, 14, 1417–1432. [Google Scholar] [CrossRef]
- Foyer, C.H.; Descourvieres, P.; Kunert, K.J. Protection against oxygen radicals: An important defense mechanism studied in transgenic plants. Plant Cell Environ. 1994, 17, 507–523. [Google Scholar] [CrossRef]
- Yuan, G.; Liu, J.; An, G.; Li, W.; Si, W.; Sun, D.; Zhu, Y. Genome-wide identification and characterization of the trehalose-6-phosphate synthetase (TPS) gene family in Watermelon (Citrullus lanatus) and their transcriptional responses to salt stress. Int. J. Mol. Sci. 2022, 23, 276. [Google Scholar] [CrossRef]
- Kong, W.; Gong, Z.; Zhong, H.; Zhang, Y.; Zhao, G.; Gautam, M.; Deng, X.; Liu, C.; Zhang, C.; Li, Y. Expansion and evolutionary patterns of glycosyltransferase family 8 in gramineae crop genomes and their expression under salt and cold stresses in Oryza sativa ssp. japonica. Biomolecules 2019, 9, 188. [Google Scholar] [CrossRef]
- Duan, X.; Wang, Z.; Zhang, Y.; Li, H.; Yang, M.; Yin, H.; Cui, J.; Chai, H.; Gao, Y.; Hu, G.; et al. Overexpression of a thioredoxin-protein-encoding gene, MsTRX, from Medicago sativa enhances salt tolerance to transgenic tobacco. Agronomy 2022, 12, 1467. [Google Scholar] [CrossRef]
- Wenderoth, I.; Scheibe, R.; von Schaewen, A. Identification of the cysteine residues involved in redox modification of plant plastidic glucose-6-phosphate dehydrogenase. J. Biol. Chem. 1997, 272, 26985–26990. [Google Scholar] [CrossRef] [PubMed]
- Holtgrefe, S.; Gohlke, J.; Starmann, J.; Druce, S.; Klocke, S.; Altmann, B.; Wojtera, J.; Lindermayr, C.; Scheibe, R. Regulation of plant cytosolic glyceraldehyde 3-phosphate dehydrogenase isoforms by thiol modifications. Physiol. Plant. 2008, 133, 211–228. [Google Scholar] [CrossRef] [PubMed]
- Gaudet, P.; Livstone, M.S.; Lewis, S.E.; Thomas, P.D. Phylogenetic-based propagation of functional annotations within the Gene Ontology consortium. Brief. Bioinform. 2011, 12, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Tamoi, M.; Tabuchi, T.; Demuratani, M.; Otori, K.; Tanabe, N.; Maruta, T.; Shigeoka, S. Point mutation of a plastidic invertase inhibits development of the photosynthetic apparatus and enhances nitrate assimilation in sugar-treated Arabidopsis seedlings. J. Biol. Chem. 2010, 285, 15399–15407. [Google Scholar] [CrossRef]
- Angeles-Nunez, J.G.; Tiessen, A. Arabidopsis sucrose synthase 2 and 3 modulate metabolic homeostasis and direct carbon towards starch synthesis in developing seeds. Planta 2010, 232, 701–718. [Google Scholar] [CrossRef]
- Villadsen, D.; Rung, J.H.; Draborg, H.; Nielsen, T.H. Structure and heterologous expression of a gene encoding fructose-6-phosphate,2-kinase/fructose-2,6-bisphosphatase from Arabidopsis thaliana. Biochim. Biophys. Acta. 2000, 1492, 406–413. [Google Scholar] [CrossRef]
- Nishizawa, A.; Yabuta, Y.; Shigeoka, S. Galactinol and raffinose constitute a novel function to protect plants from oxidative damage. Plant Physiol. 2008, 147, 1251–1263. [Google Scholar] [CrossRef]
- Chary, S.N.; Hicks, G.R.; Choi, Y.G.; Carter, D.; Raikhel, N.V. Trehalose-6-phosphate synthase/phosphatase regulates cell shape and plant architecture in Arabidopsis. Plant Physiol. 2008, 146, 97–107. [Google Scholar] [CrossRef]
- Shi, S.; Stanley, P. Protein O-fucosyltransferase 1 is an essential component of Notch signaling pathways. Proc. Natl. Acad. Sci. USA 2003, 100, 5234–5239. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.M.; Zeeman, S.C.; Smith, S.M. Starch degradation. Annu. Rev. Plant Biol. 2005, 56, 73–98. [Google Scholar] [CrossRef] [PubMed]
- Thalmann, M.; Pazmino, D.; Seung, D.; Horrer, D.; Nigro, A.; Meier, T.; Koelling, K.; Pfeifhofer, H.W.; Zeeman, S.C.; Santelia, D. Regulation of leaf starch degradation by abscisic acid is important for osmotic stress tolerance in plants. Plant Cell 2016, 28, 1860–1878. [Google Scholar] [CrossRef]
- Edner, C.; Li, J.; Albrecht, T.; Mahlow, S.; Hejazi, M.; Hussain, H.; Kaplan, F.; Guy, C.; Smith, S.M.; Steup, M.; et al. Glucan, water dikinase activity stimulates breakdown of starch granules by plastidial beta-amylases. Plant Physiol. 2007, 145, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Baunsgaard, L.; Luetken, H.; Mikkelsen, R.; Glaring, M.A.; Pham, T.T.; Blennow, A. A novel isoform of glucan, water dikinase phosphorylates pre-phosphorylated alpha-glucans and is involved in starch degradation in Arabidopsis. Plant J. 2005, 41, 595–605. [Google Scholar] [CrossRef]
- Fu, H.; Reis, N.; Lee, Y.; Glickman, M.H.; Vierstra, R. Subunit interaction maps for the regulatory particle of the 26S proteasome and the COP9 signalosome. EMBO J. 2001, 20, 7096–7107. [Google Scholar] [CrossRef]
- Ge, X.; Dietrich, C.; Matsuno, M.; Li, G.; Berg, H.; Xia, Y. An Arabidopsis aspartic protease functions as an anti-cell-death component in reproduction and embryogenesis. EMBO Rep. 2005, 6, 282–288. [Google Scholar] [CrossRef]
- Aoki, H.; Ahsan, M.N.; Watabe, S. Molecular cloning and functional characterization of crustapain: A distinct cysteine proteinase with unique substrate specificity from northern shrimp Pandalus borealis. J. Biochem. 2003, 133, 799–810. [Google Scholar] [CrossRef]
- Itzhaki, H.; Naveh, L.; Lindahl, M.; Cook, M.; Adam, Z. Identification and characterization of DegP, a serine protease associated with the luminal side of the thylakoid membrane. J. Biol. Chem. 1998, 273, 7094–7098. [Google Scholar] [CrossRef]
- Zienkiewicz, M.; Ferenc, A.; Wasilewska, W.; Romanowska, E. High light stimulates Deg1-dependent cleavage of the minor LHCII antenna proteins CP26 and CP29 and the PsbS protein in Arabidopsis thaliana. Planta 2012, 235, 279–288. [Google Scholar] [CrossRef]
- Golldack, D.; Vera, P.; Dietz, K.J. Expression of subtilisin-like serine proteases in Arabidopsis thaliana is cell-specific and responds to jasmonic acid and heavy metals with developmental differences. Physiol. Plantarum 2003, 118, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.R.; Liu, J.Y.; Du, X.M. Molecular cloning and characterization of cotton cDNAs expressed in developing fiber cells. Biosci. Biotechnol. Biochem. 2001, 65, 2789–2793. [Google Scholar] [CrossRef]
- Baron, K.N.; Schroeder, D.F.; Stasolla, C. GEm-Related 5 (GER5), an ABA and stress-responsive GRAM domain protein regulating seed development and inflorescence architecture. Plant Sci. 2014, 223, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.D.; Ni, M. Hyposensitive to light, an alpha/beta fold protein, acts downstream of elongated hypocotyl 5 to regulate seedling de-etiolation. Mol. Plant 2011, 4, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Kulich, I.; Cole, R.; Drdova, E.; Cvrckova, F.; Soukup, A.; Fowler, J.; Zarsky, V. Arabidopsis exocyst subunits SEC8 and EXO70A1 and exocyst interactor ROH1 are involved in the localized deposition of seed coat pectin. New Phytol. 2010, 188, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; An, L.; Li, F.; Ahmad, W.; Aslam, M.; Ul Haq, M.Z.; Yan, Y.; Ahmad, R.M. Wide-range portrayal of AP2/ERF transcription factor family in Maize (Zea mays L.) development and stress responses. Genes 2023, 14, 194. [Google Scholar] [CrossRef]
- Guo, X.; Ullah, A.; Siuta, D.; Kukfisz, B.; Iqbal, S. Role of WRKY transcription factors in regulation of abiotic stress responses in Cotton. Life 2022, 12, 1410. [Google Scholar] [CrossRef]
- Li, W.; Zhong, J.; Zhang, L.; Wang, Y.; Song, P.; Liu, W.; Li, X.; Han, D. Overexpression of a Fragaria vesca MYB transcription factor gene (FvMYB82) increases salt and cold tolerance in Arabidopsis thaliana. Int. J. Mol. Sci. 2022, 23, 10538. [Google Scholar] [CrossRef]
- Mijiti, M.; Wang, Y.; Wang, L.; Habuding, X. Tamarix hispida NAC transcription factor ThNAC4 confers salt and drought stress tolerance to transgenic Tamarix and Arabidopsis. Plants 2022, 11, 2647. [Google Scholar] [CrossRef]
- Kim, J.H.; Nguyen, N.H.; Jeong, C.Y.; Nguyen, N.T.; Hong, S.W.; Lee, H. Loss of the R2R3 MYB, AtMyb73, causes hyper-induction of the SOS1 and SOS3 genes in response to high salinity in Arabidopsis. J Plant Physiol. 2013, 170, 1461–1465. [Google Scholar] [CrossRef]
- Tang, J.; Liu, Q.Q.; Yuan, H.Y.; Zhang, Y.X.; Huang, S.Z. Molecular analysis of a novel alkaline metal salt (NaCl)-responsive WRKY transcription factor gene IlWRKY1 from the halophyte Iris lactea var. chinensis. Int. Biodeter. Biodegr. 2018, 127, 139–145. [Google Scholar] [CrossRef]
- Yang, S.D.; Seo, P.J.; Yoon, H.K.; Park, C.M. The Arabidopsis NAC transcription factor VNI2 integrates abscisic acid signals into leaf senescence via the COR/RD genes. Plant Cell 2011, 23, 2155–2168. [Google Scholar] [CrossRef] [PubMed]
- Du, M.; Zhao, J.; Tzeng, D.T.W.; Liu, Y.; Deng, L.; Yang, T.; Zhai, Q.; Wu, F.; Huang, Z.; Zhou, M.; et al. MYC2 orchestrates a hierarchical transcriptional cascade that regulates jasmonate-mediated plant immunity in tomato. Plant Cell 2017, 29, 1883–1906. [Google Scholar] [CrossRef]
- Iglesias-Fernandez, R.; Barrero-Sicilia, C.; Carrillo-Barral, N.; Onate-Sanchez, L.; Carbonero, P. Arabidopsis thaliana bZIP44: A transcription factor affecting seed germination and expression of the mannanase-encoding gene AtMAN7. Plant J. 2013, 74, 767–780. [Google Scholar] [CrossRef]
- Tatematsu, K.; Nakabayashi, K.; Kamiya, Y.; Nambara, E. Transcription factor AtTCP14 regulates embryonic growth potential during seed germination in Arabidopsis thaliana. Plant J. 2008, 53, 42–52. [Google Scholar] [CrossRef]
- Oh, J.; Park, E.; Song, K.; Bae, G.; Choi, G. Phytochrome interacting factor8 inhibits phytochrome A-mediated far-red light responses in Arabidopsis. Plant Cell 2020, 32, 186–205. [Google Scholar] [CrossRef]
- Wang, H.; Wei, T.; Wang, X.; Zhang, L.; Yang, M.; Chen, L.; Song, W.; Wang, C.; Chen, C. Transcriptome analyses from mutant Salvia miltiorrhiza reveals important roles for SmGASA4 during plant development. Int. J. Mol. Sci. 2018, 19, 2088. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.; Khurana, J.P. Transcript profiling reveals diverse roles of auxin-responsive genes during reproductive development and abiotic stress in rice. FEBS J. 2009, 276, 3148–3162. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, S.; Du, S.; Wang, G.; Zhang, J.; Jiang, J. Effects of exogenous (K+) potassium application on plant hormones in the roots of Tamarix ramosissima under NaCl stress. Genes 2022, 13, 1803. [Google Scholar] [CrossRef] [PubMed]
- Herzog, M.; Dorne, A.M.; Grellet, F. GASA, a gibberellin-regulated gene family from Arabidopsis thaliana related to the tomato GAST1 gene. Plant Mol. Biol. 1995, 27, 743–752. [Google Scholar] [CrossRef]
- Liscum, E.; Reed, J.W. Genetics of Aux/IAA and ARF action in plant growth and development. Plant Mol. Biol. 2002, 49, 387–400. [Google Scholar] [CrossRef]
- Hao, Y.; Hu, G.; Breitel, D.; Liu, M.; Mila, I.; Frasse, P.; Fu, Y.; Aharoni, A.; Bouzayen, M.; Zouine, M. Auxin response factor SlARF2 is an essential component of the regulatory mechanism controlling fruit ripening in tomato. PLoS Genet. 2015, 11, E1005649. [Google Scholar] [CrossRef]
- Veach, Y.K.; Martin, R.C.; Mok, D.W.S.; Malbeck, J.; Vankova, R.; Mok, M.C. O-glucosylation of cis-zeatin in maize. Characterization of genes, enzymes, and endogenous cytokinins. Plant Physiol. 2003, 131, 1374–1380. [Google Scholar] [CrossRef]
- Riefler, M.; Novak, O.; Strnad, M.; Schmuelling, T. Arabidopsis cytokinin receptor mutants reveal functions in shoot growth, leaf senescence, seed size, germination, root development, and cytokinin metabolism. Plant Cell 2006, 18, 40–54. [Google Scholar] [CrossRef]
- Smirnova, E.; Marquis, V.; Poirier, L.; Aubert, Y.; Zumsteg, J.; Menard, R.; Miesch, L.; Heitz, T. Jasmonic acid oxidase 2 hydroxylates jasmonic acid and represses basal defense and resistance responses against Botrytis cinerea infection. Mol. Plant 2017, 10, 1159–1173. [Google Scholar] [CrossRef] [PubMed]
- Chini, A.; Fonseca, S.; Fernandez, G.; Adie, B.; Chico, J.M.; Lorenzo, O.; Garcia-Casado, G.; Lopez-Vidriero, I.; Lozano, F.M.; Ponce, M.R.; et al. The JAZ family of repressors is the missing link in jasmonate signalling. Nature 2007, 448, 666–671. [Google Scholar] [CrossRef]
- Tian, X.; Wang, Z.; Li, X.; Lv, T.; Liu, H.; Wang, L.; Niu, H.; Bu, Q. Characterization and functional analysis of pyrabactin resistance-like abscisic acid receptor family in rice. Rice 2015, 8, 28. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Nishimura, N.; Kitahata, N.; Kuromori, T.; Ito, T.; Asami, T.; Shinozaki, K.; Hirayama, T. ABA-hypersensitive germination3 encodes a protein phosphatase 2C (AtPP2CA) that strongly regulates abscisic acid signaling during germination among Arabidopsis protein phosphatase 2Cs. Plant Physiol. 2006, 140, 115–126. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, R.C.; Rodriguez, F.I.; Esch, J.J.; Binder, B.M.; O’Donnell, P.; Klee, H.J.; Bleecker, A.B. Ethylene-binding activity, gene expression levels, and receptor system output for ethylene receptor family members from Arabidopsis and tomato. Plant J. 2005, 41, 651–659. [Google Scholar] [CrossRef]
- Gaudinier, A.; Rodriguez-Medina, J.; Zhang, L.; Olson, A.; Liseron-Monfils, C.; Bagman, A.M.; Foret, J.; Abbitt, S.; Tang, M.; Li, B.; et al. Transcriptional regulation of nitrogen-associated metabolism and growth. Nature 2018, 563, 259–264. [Google Scholar] [CrossRef]
- Blumwald, E. Sodium transport and salt tolerance in plants. Curr. Opin. Cell Biol. 2000, 12, 431–434. [Google Scholar] [CrossRef]
- Park, H.J.; Kim, W.Y.; Yun, A.D.J. A New insight of salt stress signaling in plant. Mol. Cells 2016, 39, 447–459. [Google Scholar] [CrossRef]
- Ren, W.; Wang, Q.; Chen, L.; Ren, Y. Transcriptome and metabolome analyses of salt stress response in Cotton (Gossypium hirsutum) seed pretreated with NaCl. Agronomy 2022, 12, 1849. [Google Scholar] [CrossRef]
- Siddique, M.H.; Babar, N.I.; Zameer, R.; Muzammil, S.; Nahid, N.; Ijaz, U.; Masroor, A.; Nadeem, M.; Rashid, M.A.R.; Hashem, A.; et al. Genome-wide identification, genomic organization, and characterization of potassium transport-related genes in Cajanus cajan and their role in abiotic stress. Plants 2021, 10, 2238. [Google Scholar] [CrossRef]
- Chen, J.; Ran, Q.; Yang, Z.; Zhou, Y.; Yuan, Z.; Lai, H.; Wang, J.; Nie, G.; Zhu, Y. Genome-wide identification and expression profile of the HD-Zip transcription factor family associated with seed germination and abiotic stress response in Miscanthus sinensis. Genes 2022, 13, 2256. [Google Scholar] [CrossRef]
- Hou, C.; Tian, W.; Kleist, T.; He, K.; Garcia, V.; Bai, F.; Hao, Y.; Luan, S.; Li, L. DUF221 proteins are a family of osmosensitive calcium-permeable cation channels conserved across eukaryotes. Cell Res. 2014, 24, 632–635. [Google Scholar] [CrossRef] [PubMed]
- Jojoa-Cruz, S.; Saotome, K.; Murthy, S.E.; Tsui, C.C.A.; Sansom, M.S.; Patapoutian, A.; Ward, A.B. Cryo-EM structure of the mechanically activated ion channel OSCA1.2. Elife 2018, 7, e41845. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, H.R.; Singh, A.K.; Sopory, S.K.; Singla-Pareek, S.L.; Pareek, A. Genome wide expression analysis of CBS domain containing proteins in Arabidopsis thaliana (L.) Heynh and Oryza sativa L. reveals their developmental and stress regulation. BMC Genom. 2009, 10, 200. [Google Scholar] [CrossRef]
- Fujimoto, S.Y.; Ohta, M.; Usui, A.; Shinshi, H.; Ohme-Takagi, M. Arabidopsis ethylene responsive element binding factors act as transcriptional activators or repressors of GCC box mediated gene expression. Plant Cell 2000, 12, 393–404. [Google Scholar] [PubMed]
- Aranda-Sicilia, M.N.; Cagnac, O.; Chanroj, S.; Sze, H.; Rodriguez-Rosales, M.P.; Venema, K. Arabidopsis KEA2, a homolog of bacterial KefC, encodes a K(+)/H(+) antiporter with a chloroplast transit peptide. Biochim. Biophys. Acta 2012, 1818, 2362–2371. [Google Scholar] [CrossRef]
- Ni, D.Q.; Zook, J.; Klewer, D.A.; Nieman, R.A.; Soll, J.; Fromme, P. Isolation, folding and structural investigations of the amino acid transporter OEP16. Protein Expr. Purif. 2011, 80, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Viotti, C.; Luoni, L.; Morandini, P.; De Michelis, M.I. Characterization of the interaction between the plasma membrane H-ATPase of Arabidopsis thaliana and a novel interactor (PPI1). FEBS J. 2005, 272, 5864–5871. [Google Scholar] [CrossRef]
- Yokoi, S.; Quintero, F.J.; Cubero, B.; Ruiz, M.T.; Bressan, R.A.; Hasegawa, P.M.; Pardo, J.M. Differential expression and function of Arabidopsis thaliana NHX Na(+)/H(+) antiporters in the salt stress response. Plant J. 2002, 30, 529–539. [Google Scholar] [CrossRef]
- Cheng, N.H.; Pittman, J.K.; Barkla, B.J.; Shigaki, T.; Hirschi, K.D. The Arabidopsis cax1 mutant exhibits impaired ion homeostasis, development, and hormonal responses and reveals interplay among vacuolar transporters. Plant Cell 2003, 15, 347–364. [Google Scholar] [CrossRef]
- Krebs, M.; Beyhl, D.; Goerlich, E.; Al-Rasheid, K.A.S.; Marten, I.; Stierhof, Y.D.; Hedrich, R.; Schumacher, K. Arabidopsis V-ATPase activity at the tonoplast is required for efficient nutrient storage but not for sodium accumulation. Proc. Natl. Acad. Sci. USA 2010, 107, 3251–3256. [Google Scholar] [CrossRef]
- Grotz, N.; Fox, T.; Connolly, E.; Park, W.; Guerinot, M.L.; Eide, D. Identification of a family of zinc transporter genes from Arabidopsis that respond to zinc deficiency. Proc. Natl. Acad. Sci. USA 1998, 95, 7220–7224. [Google Scholar] [CrossRef] [PubMed]
- Stewart, R.R.; Bewley, J.D. Lipid peroxidation associated with accelerated aging of soybean axes. Plant Physiol. 1980, 65, 245–248. [Google Scholar] [CrossRef]
- Hammerschmidt, R.; Nuckles, E.M.; Kuc, J. Association of enhanced peroxidase activity with induced systemic resistance of cucumber to Colletotrichum lagenarium. Physiol. Plant Pathol. 1982, 20, 73–82. [Google Scholar] [CrossRef]
- Chen, X.M. Comparison of three different methods of determining catalase activities in cut flowers. Chin. J. Trop. Agr. 2002, 22, 13–16. [Google Scholar]
- Shen, W.B.; Xu, L.L.; Ye, M.B.; Zhang, R.X. Study on determination of ASP activity. Plant Physiol. Comm. 1996, 32, 203–205. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Li, H.S. Principles and Techniques of Plant Physiological Biochemical Experimental; High Education Press: Beijing, China, 2000; pp. 260–261. [Google Scholar]
- Shi, H.T. Experimental Guidance of Plant Stress Physiology; Science Press: Beijing, China, 2016; pp. 14–15. [Google Scholar]
- Li, M.F.; Sun, P.; Kang, T.L.; Xing, H.; Yang, D.L.; Zhang, J.L.; Paré, P.W. Mapping podophyllotoxin biosynthesis and growth related transcripts with high elevation in Sinopodophyllum hexandrum. Ind. Crops Prod. 2018, 124, 510–518. [Google Scholar] [CrossRef]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef] [PubMed]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.D.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNAseq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Conesa, A.; Gotz, S.; Garcia-Gomez, J.M.; Terol, J.; Talon, M.; Robles, M. Blast2GO: A universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 2005, 21, 3674–3676. [Google Scholar] [CrossRef] [PubMed]
- Willems, E.; Leyns, L.; Vandesompele, J. Standardization of real-time PCR gene expression data from independent biological replicates. Anal. Biochem. 2008, 379, 127–129. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).