Sensory Neurons, PIEZO Channels and PAC1 Receptors Regulate the Mechanosensitive Release of Soluble Ectonucleotidases in the Murine Urinary Bladder Lamina Propria
Abstract
:1. Introduction
2. Results
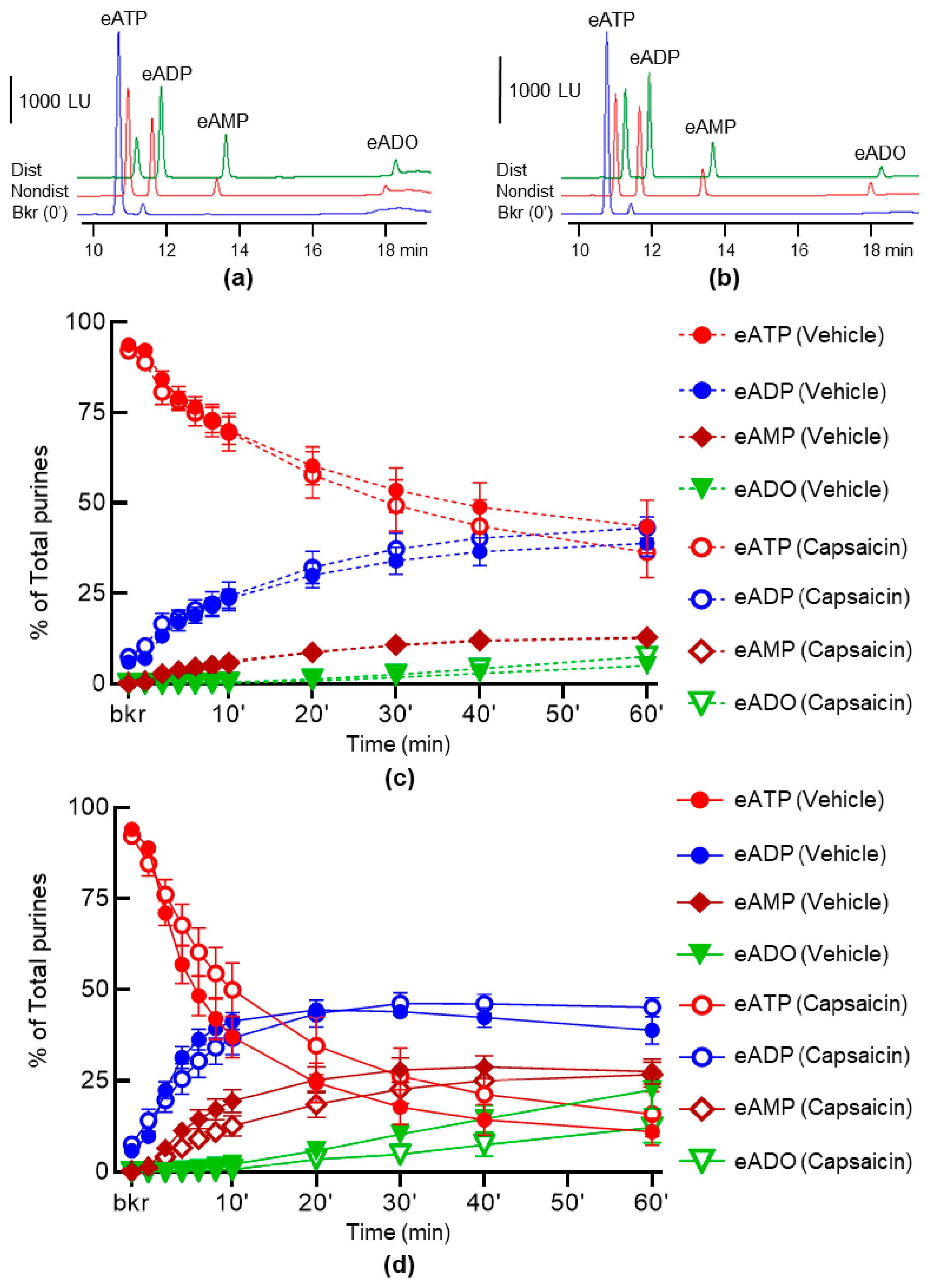
2.1. Capsaicin Does Not Alter the Spontaneous and the Distention-Induced Release of s-ENTDs in LP
2.2. Neural Blockade with Tetrodotoxin (TTX) Has No Effect on the Spontaneous Release of s-ENTDs but Increases the Distention-Induced Release of s-ENTDs in LP
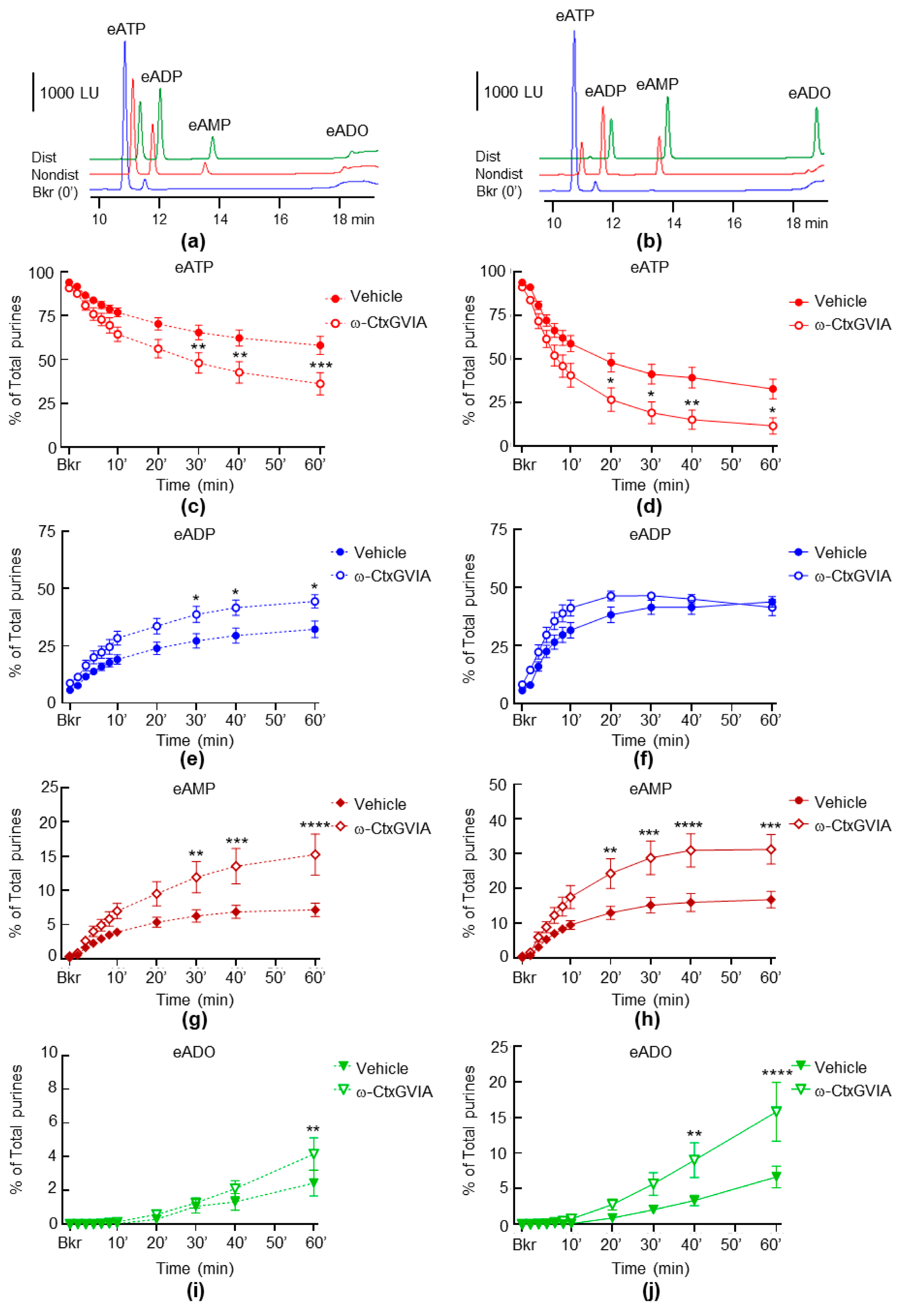
2.3. Neural Blockade with ω-Conotoxin (ω-CtxGVIA) Increases Both the Spontaneous and the Distention-Induced Release of sNTDs
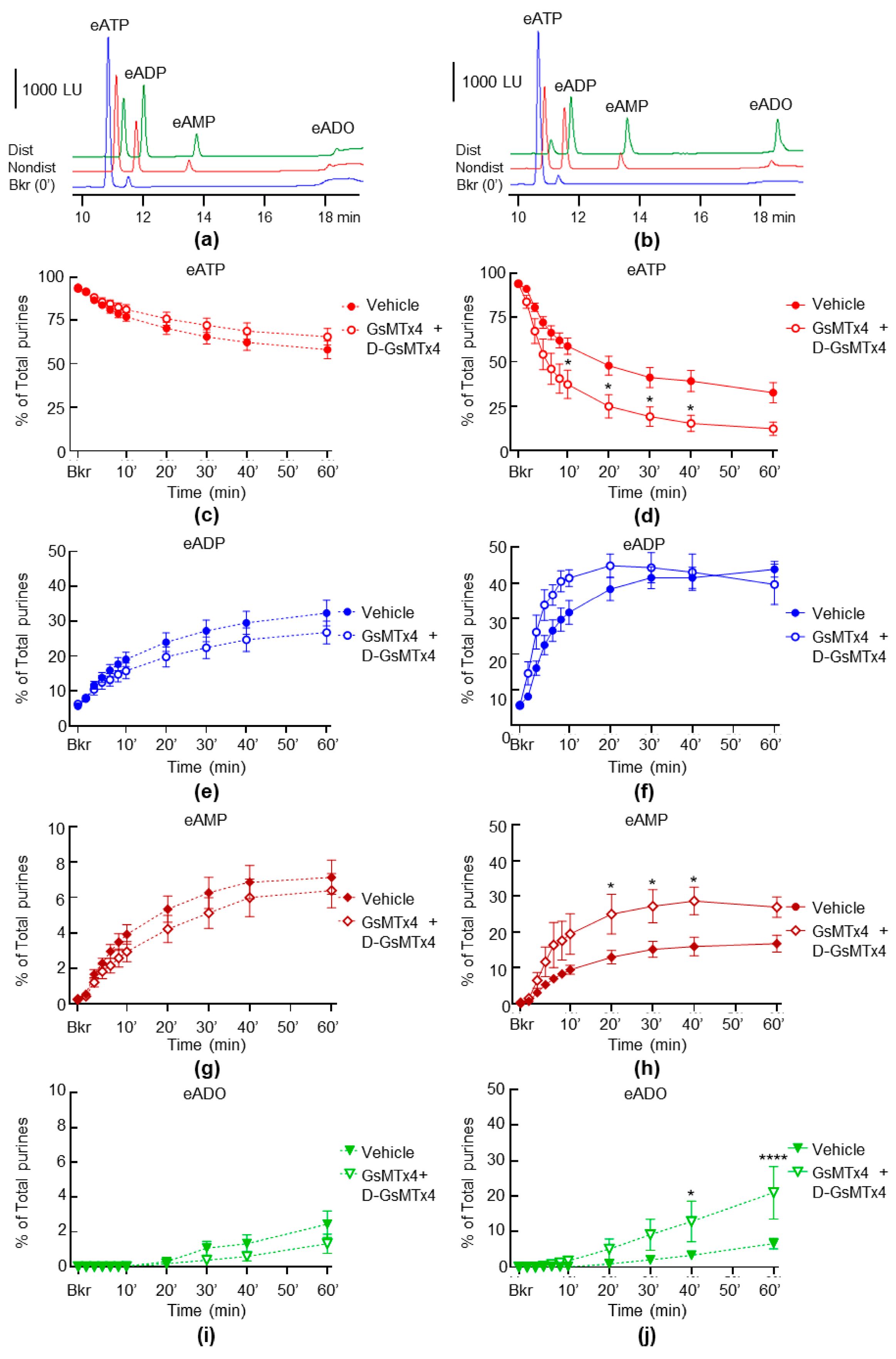
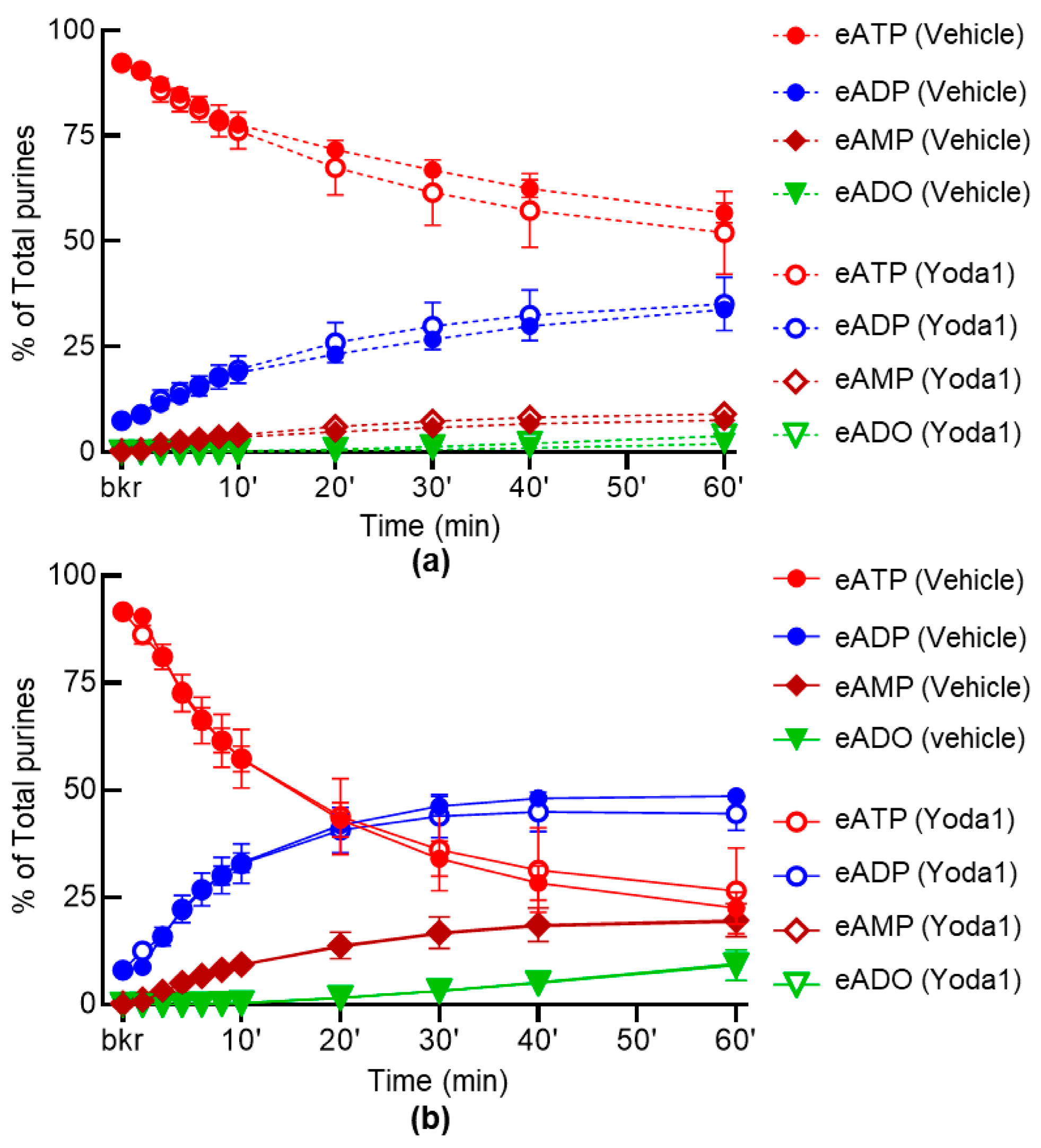
2.4. Inhibition of PIEZO Channels Has No Effect on the Spontaneous Release of s-ENTDs, but Increases the Distention-Induced Release of s-ENTDs in LP
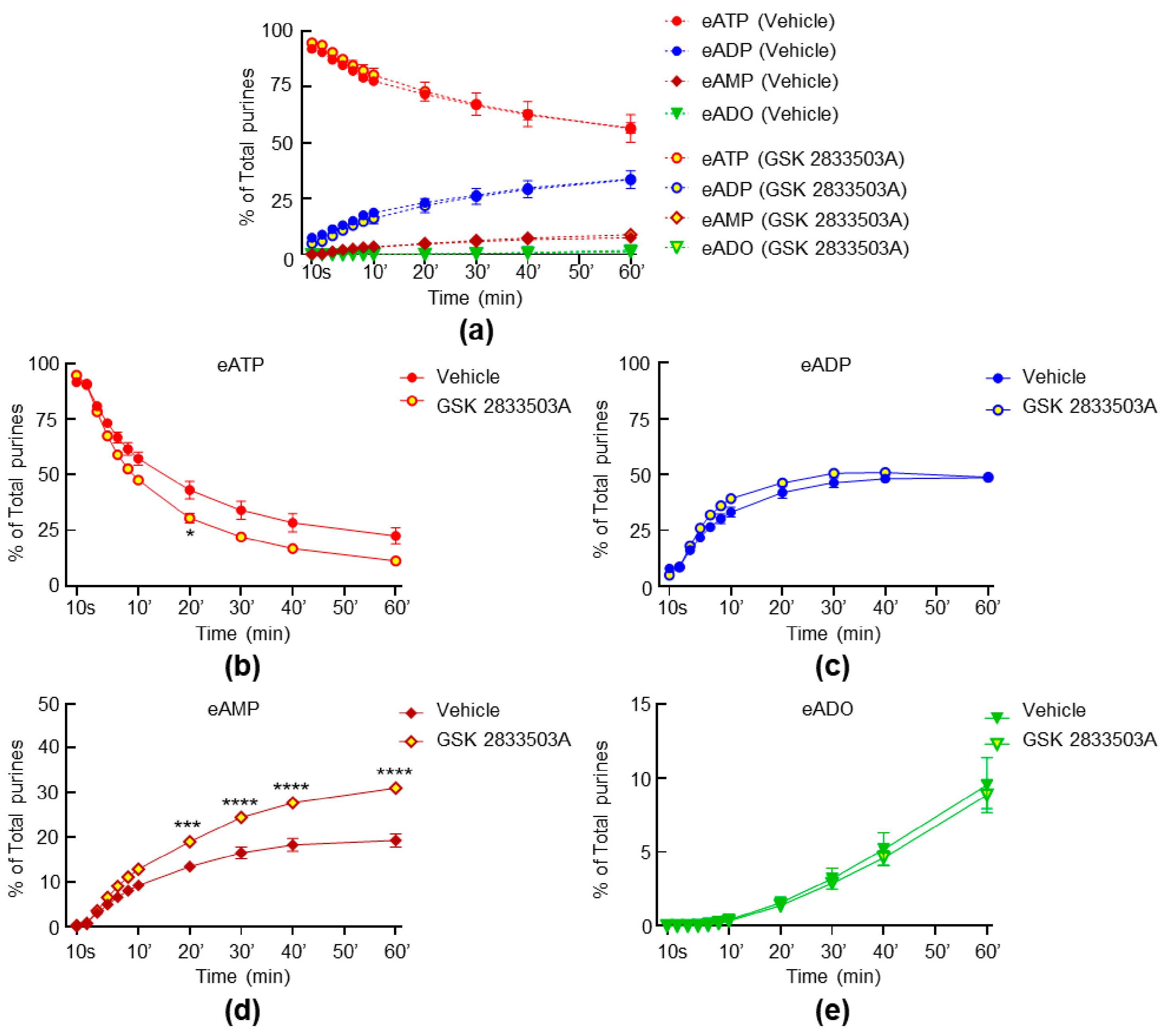
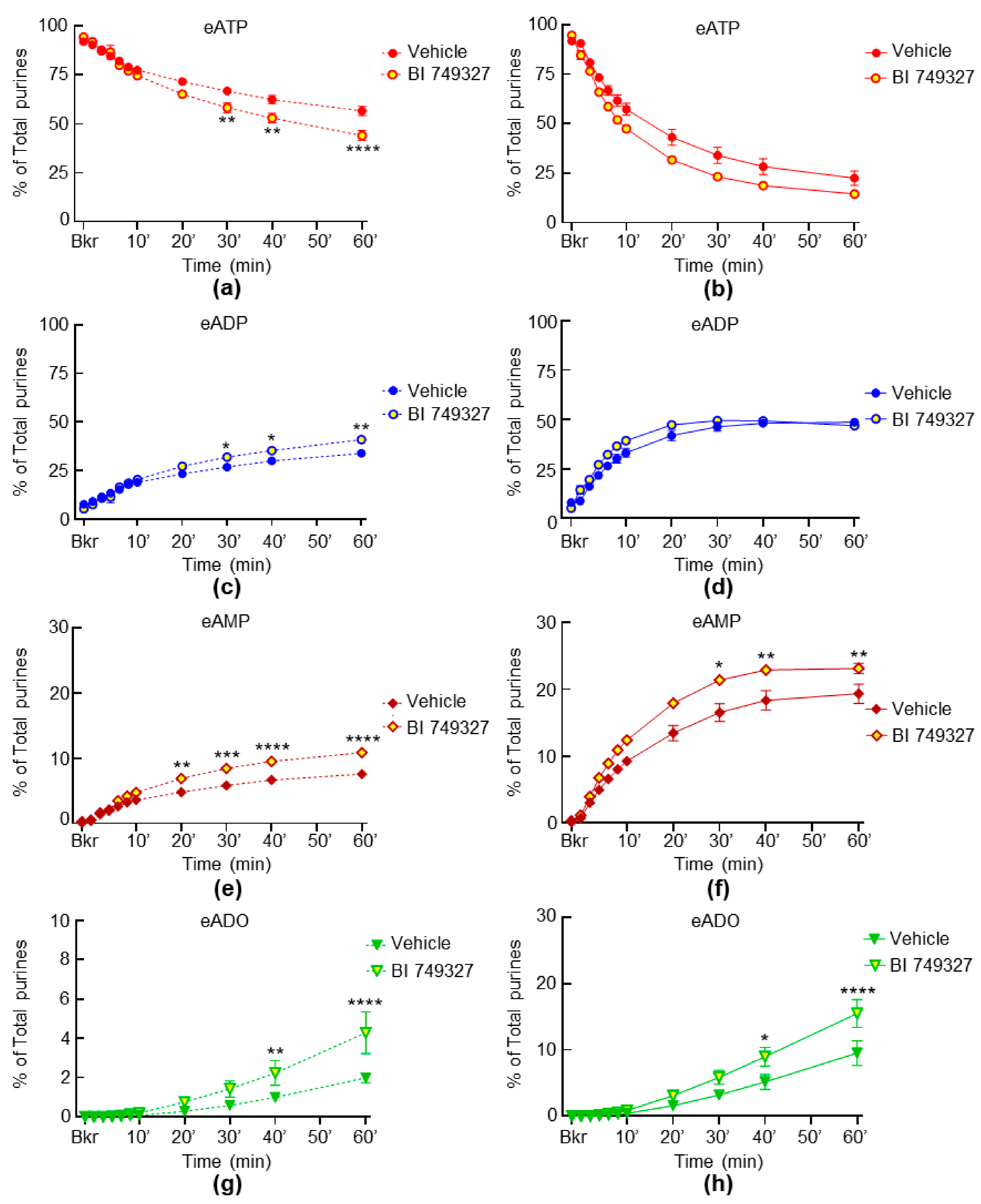
2.5. Inhibition of TRPC1 and TRPC6 Channels Have Weak Effect on the Spontaneous and Distention-Induced Release of s-ENTDs in LP
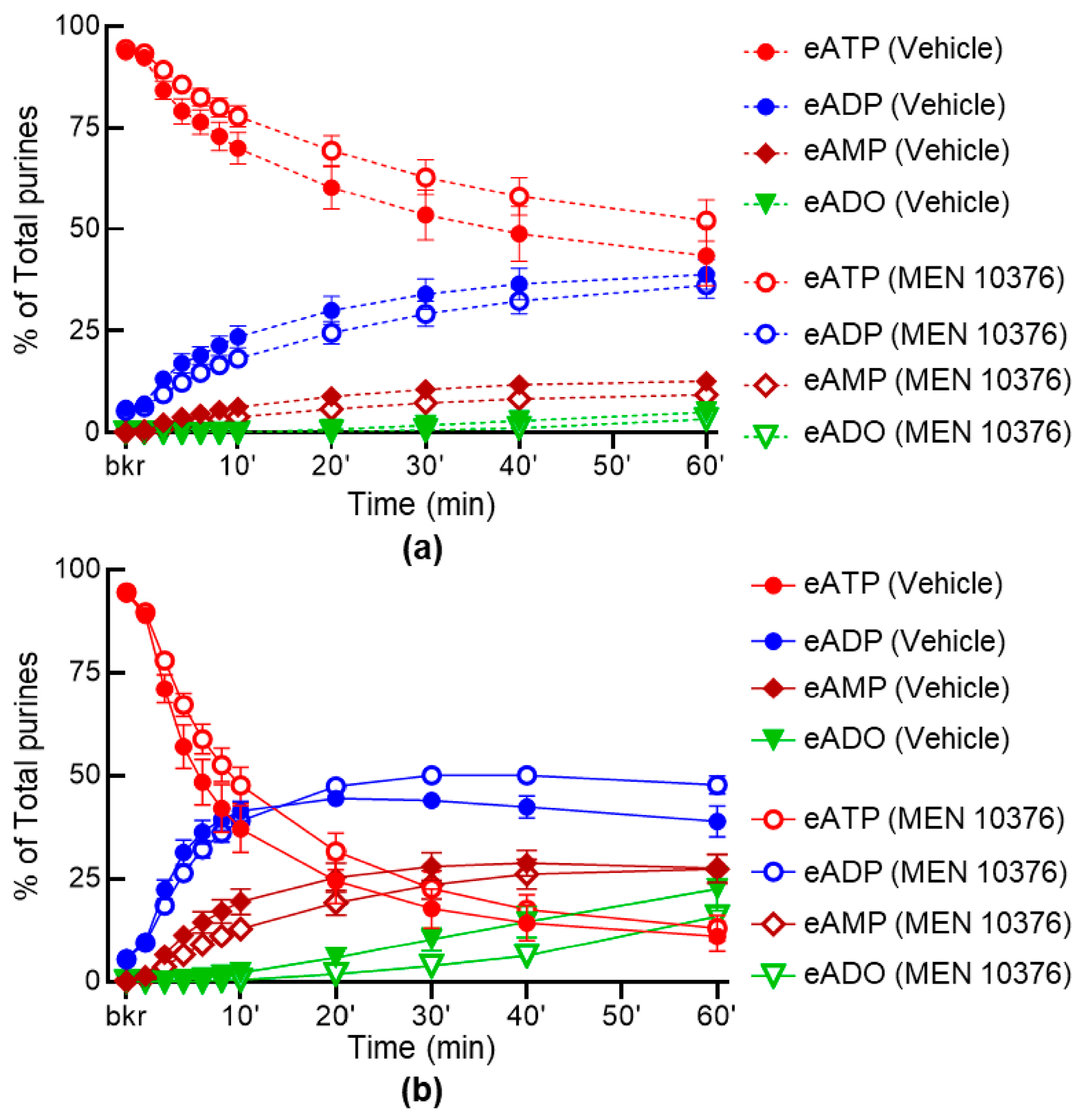
2.6. Inhibition of Receptors for Sensory Neuropeptides Affects Differentially the Release of s-ENTDs in LP
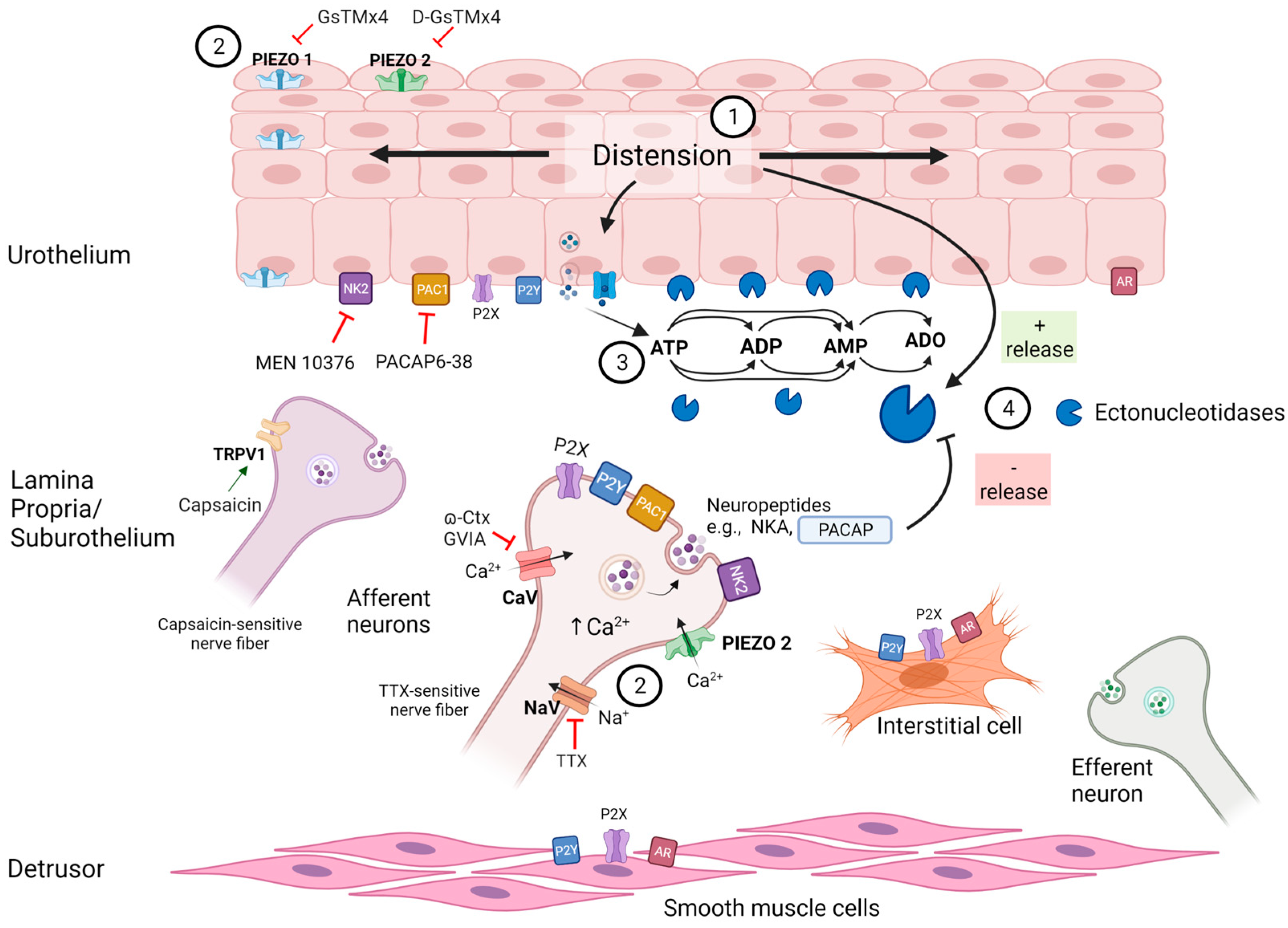
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Ex Vivo Detrusor-Free Bladder Preparation
4.3. General Experimental Protocol
4.4. Preparation of Extraluminal Samples for Measuring Soluble Nucleotidase Activities in the LP
4.5. Time-Course of ATP Hydrolysis in Concentrated ELS of Nondistended and Distended Preparations
4.6. Synthesis of 1,N6-etheno-ATP Substrate
4.7. HPLC Analysis of 1,N6-etheno-nucleotides and 1,N6-etheno-nucleosides
4.8. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Burnstock, G. Purinergic signalling in the urinary tract in health and disease. Purinergic Signal. 2014, 10, 103–155. [Google Scholar] [CrossRef]
- Ferguson, D.R.; Kennedy, I.; Burton, T.J. ATP is released from rabbit urinary bladder epithelial cells by hydrostatic pressure changes—A possible sensory mechanism? J. Physiol. 1997, 505, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Cockayne, D.A.; Hamilton, S.G.; Zhu, Q.M.; Dunn, P.M.; Zhong, Y.; Novakovic, S.; Malmberg, A.B.; Cain, G.; Berson, A.; Kassotakis, L.; et al. Urinary bladder hyporeflexia and reduced pain-related behaviour in P2X3-deficient mice. Nature 2000, 407, 1011–1015. [Google Scholar] [CrossRef] [PubMed]
- Vlaskovska, M.; Kasakov, L.; Rong, W.; Bodin, P.; Bardini, M.; Cockayne, D.A.; Ford, A.P.; Burnstock, G. P2X3 knock-out mice reveal a major sensory role for urothelially released ATP. J. Neurosci. 2001, 21, 5670–5677. [Google Scholar] [CrossRef]
- Namasivayam, S.; Eardley, I.; Morrison, J.F. Purinergic sensory neurotransmission in the urinary bladder: An in vitro study in the rat. BJU Int. 1999, 84, 854–860. [Google Scholar] [CrossRef]
- Rong, W.; Spyer, K.M.; Burnstock, G. Activation and sensitisation of low and high threshold afferent fibres mediated by P2X receptors in the mouse urinary bladder. J. Physiol. 2002, 541, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Maynard, J.P.; Sfanos, K.S. P2 purinergic receptor dysregulation in urologic disease. Purinergic Signal. 2022, 18, 267–287. [Google Scholar] [CrossRef]
- Zimmermann, H.; Zebisch, M.; Strater, N. Cellular function and molecular structure of ecto-nucleotidases. Purinergic Signal. 2012, 8, 437–502. [Google Scholar] [CrossRef]
- Aresta Branco, M.S.L.; Gutierrez Cruz, A.; Dayton, J.; Perrino, B.A.; Mutafova-Yambolieva, V.N. Mechanosensitive Hydrolysis of ATP and ADP in Lamina Propria of the Murine Bladder by Membrane-Bound and Soluble Nucleotidases. Front. Physiol. 2022, 13, 918100. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Mansfield, K.J.; Allen, W.; Chess-Williams, R.; Burcher, E.; Moore, K.H. ATP during early bladder stretch is important for urgency in detrusor overactivity patients. Biomed. Res. Int. 2014, 2014, 204604. [Google Scholar] [CrossRef]
- Durnin, L.; Hayoz, S.; Corrigan, R.D.; Yanez, A.; Koh, S.D.; Mutafova-Yambolieva, V.N. Urothelial purine release during filling of murine and primate bladders. Am. J. Physiol. Renal Physiol. 2016, 311, F708–F716. [Google Scholar] [CrossRef] [PubMed]
- Todorov, L.D.; Mihaylova-Todorova, S.; Westfall, T.D.; Sneddon, P.; Kennedy, C.; Bjur, R.A.; Westfall, D.P. Neuronal release of soluble nucleotidases and their role in neurotransmitter inactivation. Nature 1997, 387, 76–79. [Google Scholar] [CrossRef]
- Apodaca, G.; Balestreire, E.; Birder, L.A. The uroepithelial-associated sensory web. Kidney Int. 2007, 72, 1057–1064. [Google Scholar] [CrossRef] [PubMed]
- Dalghi, M.G.; Montalbetti, N.; Carattino, M.D.; Apodaca, G. The Urothelium: Life in a Liquid Environment. Physiol. Rev. 2020, 100, 1621–1705. [Google Scholar] [CrossRef]
- de Groat, W.C.; Yoshimura, N. Afferent nerve regulation of bladder function in health and disease. In Sensory Nerves; Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2009; pp. 91–138. [Google Scholar]
- Zagorodnyuk, V.P.; Brookes, S.J.; Spencer, N.J.; Gregory, S. Mechanotransduction and chemosensitivity of two major classes of bladder afferents with endings in the vicinity to the urothelium. J. Physiol. 2009, 587, 3523–3538. [Google Scholar] [CrossRef]
- Birder, L.; Andersson, K.E. Urothelial signaling. Physiol. Rev. 2013, 93, 653–680. [Google Scholar] [CrossRef]
- de Groat, W.C.; Griffiths, D.; Yoshimura, N. Neural control of the lower urinary tract. Compr. Physiol. 2015, 5, 327–396. [Google Scholar] [PubMed]
- Dalghi, M.G.; Clayton, D.R.; Ruiz, W.G.; Al-Bataineh, M.M.; Satlin, L.M.; Kleyman, T.R.; Ricke, W.A.; Carattino, M.D.; Apodaca, G. Expression and distribution of PIEZO1 in the mouse urinary tract. Am. J. Physiol. Renal Physiol. 2019, 317, F303–F321. [Google Scholar] [CrossRef] [PubMed]
- Marshall, K.L.; Saade, D.; Ghitani, N.; Coombs, A.M.; Szczot, M.; Keller, J.; Ogata, T.; Daou, I.; Stowers, L.T.; Bönnemann, C.G.; et al. PIEZO2 in sensory neurons and urothelial cells coordinates urination. Nature 2020, 588, 290–295. [Google Scholar] [CrossRef]
- Heppner, T.J.; Hennig, G.W.; Nelson, M.T.; May, V.; Vizzard, M.A. PACAP38-Mediated Bladder Afferent Nerve Activity Hyperexcitability and Ca(2+) Activity in Urothelial Cells from Mice. J. Mol. Neurosci. 2019, 68, 348–356. [Google Scholar] [CrossRef]
- Grundy, L.; Chess-Williams, R.; Brierley, S.M.; Mills, K.; Moore, K.H.; Mansfield, K.; Rose’Meyer, R.; Sellers, D.J.; Grundy, D. NKA enhances bladder afferent mechanosensitivity via urothelial and detrusor activation. Am. J. Physiol. Renal Physiol. 2018, 315, F1174–F1185. [Google Scholar] [CrossRef] [PubMed]
- Girard, B.M.; Wolf-Johnston, A.; Braas, K.M.; Birder, L.A.; May, V.; Vizzard, M.A. PACAP-mediated ATP release from rat urothelium and regulation of PACAP/VIP and receptor mRNA in micturition pathways after cyclophosphamide (CYP)-induced cystitis. J. Mol. Neurosci. 2008, 36, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Cinar, E.; Zhou, S.; DeCourcey, J.; Wang, Y.; Waugh, R.E.; Wan, J. Piezo1 regulates mechanotransductive release of ATP from human RBCs. Proc. Natl. Acad. Sci. USA 2015, 112, 11783–11788. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Chen, Z.; Liu, L.; Ding, N.; Wen, J.; Liu, J.; Wang, W.; Ge, N.; Zu, S.; Song, W.; et al. Functional Expression of Transient Receptor Potential and Piezo1 Channels in Cultured Interstitial Cells of Human-Bladder Lamina Propria. Front. Physiol. 2021, 12, 762847. [Google Scholar] [CrossRef]
- Bahns, E.; Halsband, U.; Jänig, W. Responses of sacral visceral afferents from the lower urinary tract, colon and anus to mechanical stimulation. Pflügers Arch. 1987, 410, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Daly, D.M.; Collins, V.M.; Chapple, C.R.; Grundy, D. The afferent system and its role in lower urinary tract dysfunction. Curr. Opin. Urol. 2011, 21, 268–274. [Google Scholar] [CrossRef]
- Arms, L.; Vizzard, M.A. Neuropeptides in lower urinary tract function. In Urinary Tract; Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2011; pp. 395–423. [Google Scholar] [CrossRef]
- Fry, C.H.; McCloskey, K.D. Purinergic signalling in the urinary bladder—When function becomes dysfunction. Auton. Neurosci. 2021, 235, 102852. [Google Scholar] [CrossRef]
- Silva-Ramos, M.; Silva, I.; Oliveira, O.; Ferreira, S.; Reis, M.J.; Oliveira, J.C.; Correia-de-Sa, P. Urinary ATP may be a dynamic biomarker of detrusor overactivity in women with overactive bladder syndrome. PLoS ONE 2013, 8, e64696. [Google Scholar] [CrossRef]
- Sun, Y.; Keay, S.; De Deyne, P.G.; Chai, T.C. Augmented stretch activated adenosine triphosphate release from bladder uroepithelial cells in patients with interstitial cystitis. J. Urol. 2001, 166, 1951–1956. [Google Scholar] [CrossRef]
- Taidi, Z.; Mansfield, K.J.; Bates, L.; Sana-Ur-Rehman, H.; Liu, L. Purinergic P2X7 receptors as therapeutic targets in interstitial cystitis/bladder pain syndrome; key role of ATP signaling in inflammation. Bladder 2019, 6, e38. [Google Scholar] [CrossRef]
- Mutafova-Yambolieva, V.N.; Durnin, L. The purinergic neurotransmitter revisited: A single substance or multiple players? Pharmacol. Ther. 2014, 144, 162–191. [Google Scholar] [CrossRef] [PubMed]
- Durnin, L.; Kwok, B.; Kukadia, P.; McAvera, R.; Corrigan, R.D.; Ward, S.M.; Zhang, Y.; Chen, Q.; Koh, S.D.; Sanders, K.M.; et al. An ex vivo bladder model with detrusor smooth muscle removed to analyse biologically active mediators released from the suburothelium. J. Physiol. 2019, 597, 1467–1485. [Google Scholar] [CrossRef] [PubMed]
- Breen, L.T.; Smyth, L.M.; Yamboliev, I.A.; Mutafova-Yambolieva, V.N. β-NAD is a novel nucleotide released on stimulation of nerve terminals in human urinary bladder detrusor muscle. Am. J. Physiol. Renal Physiol. 2006, 290, F486–F495. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez Cruz, A.; Aresta Branco, M.S.L.; Perrino, B.A.; Sanders, K.M.; Mutafova-Yambolieva, V.N. Urinary ATP Levels Are Controlled by Nucleotidases Released from the Urothelium in a Regulated Manner. Metabolites 2022, 13, 30. [Google Scholar] [CrossRef] [PubMed]
- Schueth, A.; Spronck, B.; van Zandvoort, M.; van Koeveringe, G.A. Computer-assisted three-dimensional tracking of sensory innervation in the murine bladder mucosa with two-photon microscopy. J. Chem. Neuroanat. 2017, 85, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Heppner, T.J.; Hennig, G.W.; Nelson, M.T.; Herrera, G.M. Afferent nerve activity in a mouse model increases with faster bladder filling rates in vitro, but voiding behavior remains unaltered in vivo. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2022, 323, R682–R693. [Google Scholar] [CrossRef]
- Jackson, E.K.; Gillespie, D.G.; Cheng, D.; Mi, Z.; Menshikova, E.V. Characterization of the N(6)-etheno-bridge method to assess extracellular metabolism of adenine nucleotides: Detection of a possible role for purine nucleoside phosphorylase in adenosine metabolism. Purinergic Signal. 2020, 16, 187–211. [Google Scholar] [CrossRef]
- Andersson, K.E. Bladder activation: Afferent mechanisms. Urology 2002, 59, 43–50. [Google Scholar] [CrossRef]
- Chai, T.C.; Russo, A.; Yu, S.; Lu, M. Mucosal signaling in the bladder. Auton. Neurosci. 2016, 200, 49–56. [Google Scholar] [CrossRef]
- Grundy, L.; Erickson, A.; Caldwell, A.; Garcia-Caraballo, S.; Rychkov, G.; Harrington, A.; Brierley, S.M. Tetrodotoxin-sensitive voltage-gated sodium channels regulate bladder afferent responses to distension. Pain 2018, 159, 2573–2584. [Google Scholar] [CrossRef]
- Olivera, B.M.; Miljanich, G.P.; Ramachandran, J.; Adams, M.E. Calcium Channel Diversity and Neurotransmitter Release: The -Conotoxins and -Agatoxins. Annu. Rev. Biochem. 1994, 63, 823–867. [Google Scholar] [CrossRef]
- Li, X.; Hu, J.; Zhao, X.; Li, J.; Chen, Y. Piezo channels in the urinary system. Exp. Mol. Med. 2022, 54, 697–710. [Google Scholar] [CrossRef] [PubMed]
- Murthy, S.E.; Loud, M.C.; Daou, I.; Marshall, K.L.; Schwaller, F.; Kühnemund, J.; Francisco, A.G.; Keenan, W.T.; Dubin, A.E.; Lewin, G.R.; et al. The mechanosensitive ion channel Piezo2 mediates sensitivity to mechanical pain in mice. Sci. Transl. Med. 2018, 10, eaat9897. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.Z.; Marshall, K.L.; Min, S.; Daou, I.; Chapleau, M.W.; Abboud, F.M.; Liberles, S.D.; Patapoutian, A. PIEZOs mediate neuronal sensing of blood pressure and the baroreceptor reflex. Science 2018, 362, 464–467. [Google Scholar] [CrossRef]
- Dalghi, M.G.; Ruiz, W.G.; Clayton, D.R.; Montalbetti, N.; Daugherty, S.L.; Beckel, J.M.; Carattino, M.D.; Apodaca, G. Functional roles for PIEZO1 and PIEZO2 in urothelial mechanotransduction and lower urinary tract interoception. JCI Insight 2021, 6, 19. [Google Scholar] [CrossRef] [PubMed]
- Coste, B.; Mathur, J.; Schmidt, M.; Earley, T.J.; Ranade, S.; Petrus, M.J.; Dubin, A.E.; Patapoutian, A. Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science 2010, 330, 55–60. [Google Scholar] [CrossRef]
- Noguri, T.; Hatakeyama, D.; Kitahashi, T.; Oka, K.; Ito, E. Profile of dorsal root ganglion neurons: Study of oxytocin expression. Mol. Brain 2022, 15, 44. [Google Scholar] [CrossRef]
- Miyamoto, T.; Mochizuki, T.; Nakagomi, H.; Kira, S.; Watanabe, M.; Takayama, Y.; Suzuki, Y.; Koizumi, S.; Takeda, M.; Tominaga, M. Functional role for Piezo1 in stretch-evoked Ca(2+) influx and ATP release in urothelial cell cultures. J. Biol. Chem. 2014, 289, 16565–16575. [Google Scholar] [CrossRef]
- Suchyna, T.M.; Johnson, J.H.; Hamer, K.; Leykam, J.F.; Gage, D.A.; Clemo, H.F.; Baumgarten, C.M.; Sachs, F. Identification of a peptide toxin from Grammostola spatulata spider venom that blocks cation-selective stretch-activated channels. J. Gen. Physiol. 2000, 115, 583–598. [Google Scholar] [CrossRef]
- Bae, C.; Sachs, F.; Gottlieb, P.A. The mechanosensitive ion channel Piezo1 is inhibited by the peptide GsMTx4. Biochemistry 2011, 50, 6295–6300. [Google Scholar] [CrossRef]
- Suchyna, T.M. Piezo channels and GsMTx4: Two milestones in our understanding of excitatory mechanosensitive channels and their role in pathology. Prog. Biophys. Mol. Biol. 2017, 130, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Kerstein, P.C.; Jacques-Fricke, B.T.; Rengifo, J.; Mogen, B.J.; Williams, J.C.; Gottlieb, P.A.; Sachs, F.; Gomez, T.M. Mechanosensitive TRPC1 channels promote calpain proteolysis of talin to regulate spinal axon outgrowth. J. Neurosci. 2013, 33, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Spassova, M.A.; Hewavitharana, T.; Xu, W.; Soboloff, J.; Gill, D.L. A common mechanism underlies stretch activation and receptor activation of TRPC6 channels. Proc. Natl. Acad. Sci. USA 2006, 103, 16586–16591. [Google Scholar] [CrossRef]
- Bowman, C.L.; Gottlieb, P.A.; Suchyna, T.M.; Murphy, Y.K.; Sachs, F. Mechanosensitive ion channels and the peptide inhibitor GsMTx-4: History, properties, mechanisms and pharmacology. Toxicon 2007, 49, 249–270. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, P.A.; Sachs, F. Piezo1: Properties of a cation selective mechanical channel. Channels 2012, 6, 214–219. [Google Scholar] [CrossRef]
- Alcaino, C.; Knutson, K.; Gottlieb, P.A.; Farrugia, G.; Beyder, A. Mechanosensitive ion channel Piezo2 is inhibited by D-GsMTx4. Channels 2017, 11, 245–253. [Google Scholar] [CrossRef]
- Lee, W.; Leddy, H.A.; Chen, Y.; Lee, S.H.; Zelenski, N.A.; McNulty, A.L.; Wu, J.; Beicker, K.N.; Coles, J.; Zauscher, S.; et al. Synergy between Piezo1 and Piezo2 channels confers high-strain mechanosensitivity to articular cartilage. Proc. Natl. Acad. Sci. USA 2014, 111, E5114–E5122. [Google Scholar] [CrossRef]
- Syeda, R.; Xu, J.; Dubin, A.E.; Coste, B.; Mathur, J.; Huynh, T.; Matzen, J.; Lao, J.; Tully, D.C.; Engels, I.H.; et al. Chemical activation of the mechanotransduction channel Piezo1. Elife 2015, 4, e07369. [Google Scholar] [CrossRef]
- Kress, M.; Karasek, J.; Ferrer-Montiel, A.V.; Scherbakov, N.; Haberberger, R.V. TRPC channels and diacylglycerol dependent calcium signaling in rat sensory neurons. Histochem. Cell Biol. 2008, 130, 655–667. [Google Scholar] [CrossRef]
- Maroto, R.; Raso, A.; Wood, T.G.; Kurosky, A.; Martinac, B.; Hamill, O.P. TRPC1 forms the stretch-activated cation channel in vertebrate cells. Nat Cell Biol. 2005, 7, 179–185. [Google Scholar] [CrossRef]
- Liu, Q.; Sun, B.; Zhao, J.; Wang, Q.; An, F.; Hu, X.; Yang, Z.; Xu, J.; Tan, M.; Li, L. Increased Piezo1 channel activity in interstitial Cajal-like cells induces bladder hyperactivity by functionally interacting with NCX1 in rats with cyclophosphamide-induced cystitis. Exp. Mol. Med. 2018, 50, 1–16. [Google Scholar] [CrossRef]
- Michishita, M.; Yano, K.; Tomita, K.I.; Matsuzaki, O.; Kasahara, K.I. Piezo1 expression increases in rat bladder after partial bladder outlet obstruction. Life Sci. 2016, 166, 1–7. [Google Scholar] [CrossRef]
- Prakasam, H.S.; Herrington, H.; Roppolo, J.R.; Jackson, E.K.; Apodaca, G. Modulation of bladder function by luminal adenosine turnover and A1 receptor activation. Am. J. Physiol. Renal Physiol. 2012, 303, F279–F292. [Google Scholar] [CrossRef] [PubMed]
- Kitta, T.; Chancellor, M.B.; de Groat, W.C.; Kuno, S.; Nonomura, K.; Yoshimura, N. Roles of adenosine A1 and A2A receptors in the control of micturition in rats. Neurourol. Urodyn. 2014, 33, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, H.A.; Santer, R.M. Distribution and changes with age of calcitonin gene-related peptide- and substance P-immunoreactive nerves of the rat urinary bladder and lumbosacral sensory neurons. Eur. J. Morphol. 2002, 40, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Merrill, L.; Gonzalez, E.J.; Girard, B.M.; Vizzard, M.A. Receptors, channels, and signalling in the urothelial sensory system in the bladder. Nat. Rev. Urol. 2016, 13, 193–204. [Google Scholar] [CrossRef]
- Rahnama’i, M.S.; Biallosterski, B.T.; Van Kerrebroeck, P.E.; van Koeveringe, G.A.; Gillespie, J.I.; de Wachter, S.G. Distribution and sub-types of afferent fibre in the mouse urinary bladder. J. Chem. Neuroanat. 2017, 79, 1–11. [Google Scholar] [CrossRef]
- Lecci, A.; Giuliani, S.; Tramontana, M.; Santicioli, P.; Criscuoli, M.; Dion, S.; Maggi, C.A. Bladder distension and activation of the efferent function of sensory fibres: Similarities with the effect of capsaicin. Br. J. Pharmacol. 1998, 124, 259–266. [Google Scholar] [CrossRef]
- Shaker, H.S.; Tu, L.M.; Kalfopoulos, M.; Hassouna, M.; Dion, S.; Elhilali, M. Hyperreflexia of the urinary bladder: Possible role of the efferent function of the capsaicin sensitive primary afferents. J. Urol. 1998, 160, 2232–2239. [Google Scholar] [CrossRef]
- Maggi, C.A.; Giuliani, S.; Ballati, L.; Lecci, A.; Manzini, S.; Patacchini, R.; Renzetti, A.R.; Rovero, P.; Quartara, L.; Giachetti, A. In vivo evidence for tachykininergic transmission using a new NK-2 receptor-selective antagonist, MEN 10,376. J. Pharmacol. Exp. Ther. 1991, 257, 1172–1178. [Google Scholar]
- Heppner, T.J.; Tykocki, N.R.; Hill-Eubanks, D.; Nelson, M.T. Transient contractions of urinary bladder smooth muscle are drivers of afferent nerve activity during filling. J. Gen. Physiol. 2016, 147, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Sculptoreanu, A.; Artim, D.E.; de Groat, W.C. Neurokinins inhibit low threshold inactivating K+ currents in capsaicin responsive DRG neurons. Exp. Neurol. 2009, 219, 562–573. [Google Scholar] [CrossRef]
- Liao, C.; May, V.; Li, J. PAC1 Receptors: Shapeshifters in Motion. J. Mol. Neurosci. 2019, 68, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Shivers, B.D.; Görcs, T.J.; Gottschall, P.E.; Arimura, A. Two high affinity binding sites for pituitary adenylate cyclase-activating polypeptide have different tissue distributions. Endocrinology 1991, 128, 3055–3065. [Google Scholar] [CrossRef]
- Hernández, M.; Barahona, M.V.; Recio, P.; Benedito, S.; Martínez, A.C.; Rivera, L.; García-Sacristán, A.; Prieto, D.; Orensanz, L.M. Neuronal and smooth muscle receptors involved in the PACAP- and VIP-induced relaxations of the pig urinary bladder neck. Br. J. Pharmacol. 2006, 149, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Ojala, J.; Tooke, K.; Hsiang, H.; Girard, B.M.; May, V.; Vizzard, M.A. PACAP/PAC1 Expression and Function in Micturition Pathways. J. Mol. Neurosci. 2019, 68, 357–367. [Google Scholar] [CrossRef] [PubMed]
- May, V.; Vizzard, M.A. Bladder dysfunction and altered somatic sensitivity in PACAP−/− mice. J. Urol. 2010, 183, 772–779. [Google Scholar] [CrossRef]
- Girard, B.M.; Campbell, S.E.; Beca, K.I.; Perkins, M.; Hsiang, H.; May, V.; Vizzard, M.A. Intrabladder PAC1 Receptor Antagonist, PACAP(6-38), Reduces Urinary Bladder Frequency and Pelvic Sensitivity in Mice Exposed to Repeated Variate Stress (RVS). J. Mol. Neurosci. 2021, 71, 1575–1588. [Google Scholar] [CrossRef]
- Durnin, L.; Corrigan, R.D.; Sanders, K.M.; Mutafova-Yambolieva, V.N. A Decentralized (Ex Vivo) Murine Bladder Model with the Detrusor Muscle Removed for Direct Access to the Suburothelium during Bladder Filling. J. Vis. Exp. 2019, 153, e60344. [Google Scholar] [CrossRef]
- Bobalova, J.; Bobal, P.; Mutafova-Yambolieva, V.N. High-Performance Liquid Chromatographic Technique for Detection of a Fluorescent Analogue of ADP-Ribose in Isolated Blood Vessel Preparations. Anal. Biochem. 2002, 305, 269–276. [Google Scholar] [CrossRef]
- Levitt, B.; Head, R.J.; Westfall, D.P. High-pressure liquid chromatographic-fluorometric detection of adenosine and adenine nucleotides: Application to endogenous content and electrically induced release of adenyl purines in guinea pig vas deferens. Anal. Biochem. 1984, 137, 93–100. [Google Scholar] [CrossRef] [PubMed]


| Drug | Mechanism of Action | Working Concentration (µM) | Vehicle | Vendor |
|---|---|---|---|---|
| BI-749327 | TRPC6 inhibitor | 1 | DMSO 0.2% | MedChemExpress, Monmouth Junction, NJ, USA |
| Capsaicin | TRPV1 channel agonist | 1 | DMSO 0.1% | Tocris Biosciences, Minneapolis, MN, USA |
| ω-CtxGVIA | Cav2.2 (N-type) Ca2+ channel inhibitor | 0.1 | KBS | Tocris Biosciences |
| D-GsMTx4 | PIEZO inhibitor | 1 | KBS | Tocris Biosciences |
| GSK 1702934A | TRPC3/6 activator | 3 | DMSO 0.2% | Tocris Biosciences |
| GSK 2833503A | TRPC3/6 inhibitor | 1 | DMSO 0.2% | Tocris Biosciences |
| GsMTx4 | PIEZO inhibitor | 1 | KBS | Tocris Biosciences |
| MEN 10376 | NK2 receptor antagonist | 10 | KBS | Cayman Chemicals, Ann Arbor, MI, USA |
| TTX | Fast Na+ channel inhibitor | 0.5 | KBS | Alomone Labs, Jerusalem, Israel |
| PACAP6-38 | PAC1 receptor antagonist | 0.3 | KBS | Tocris Biosciences |
| Pico145 | TRPC1,4,5 inhibitor | 1 | DMSO 0.2% | MedChemExpress |
| Yoda1 | PIEZO1 activator | 40 | DMSO 0.2% | Tocris Biosciences |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aresta Branco, M.S.L.; Gutierrez Cruz, A.; Borhani Peikani, M.; Mutafova-Yambolieva, V.N. Sensory Neurons, PIEZO Channels and PAC1 Receptors Regulate the Mechanosensitive Release of Soluble Ectonucleotidases in the Murine Urinary Bladder Lamina Propria. Int. J. Mol. Sci. 2023, 24, 7322. https://doi.org/10.3390/ijms24087322
Aresta Branco MSL, Gutierrez Cruz A, Borhani Peikani M, Mutafova-Yambolieva VN. Sensory Neurons, PIEZO Channels and PAC1 Receptors Regulate the Mechanosensitive Release of Soluble Ectonucleotidases in the Murine Urinary Bladder Lamina Propria. International Journal of Molecular Sciences. 2023; 24(8):7322. https://doi.org/10.3390/ijms24087322
Chicago/Turabian StyleAresta Branco, Mafalda S. L., Alejandro Gutierrez Cruz, Mahsa Borhani Peikani, and Violeta N. Mutafova-Yambolieva. 2023. "Sensory Neurons, PIEZO Channels and PAC1 Receptors Regulate the Mechanosensitive Release of Soluble Ectonucleotidases in the Murine Urinary Bladder Lamina Propria" International Journal of Molecular Sciences 24, no. 8: 7322. https://doi.org/10.3390/ijms24087322





