Identification and Validation of STC1 Act as a Biomarker for High-Altitude Diseases and Its Pan-Cancer Analysis
Abstract
1. Introduction
2. Results
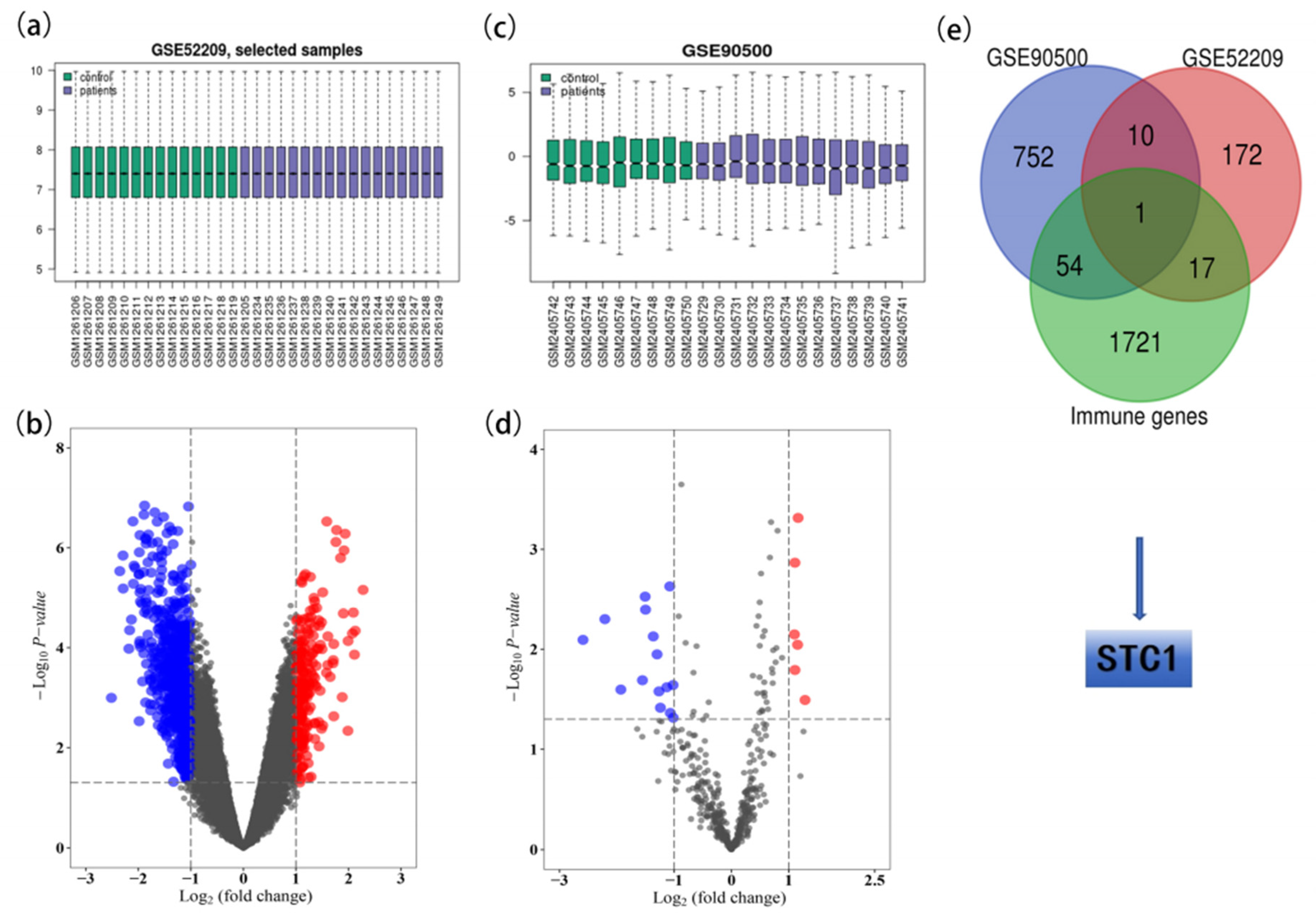
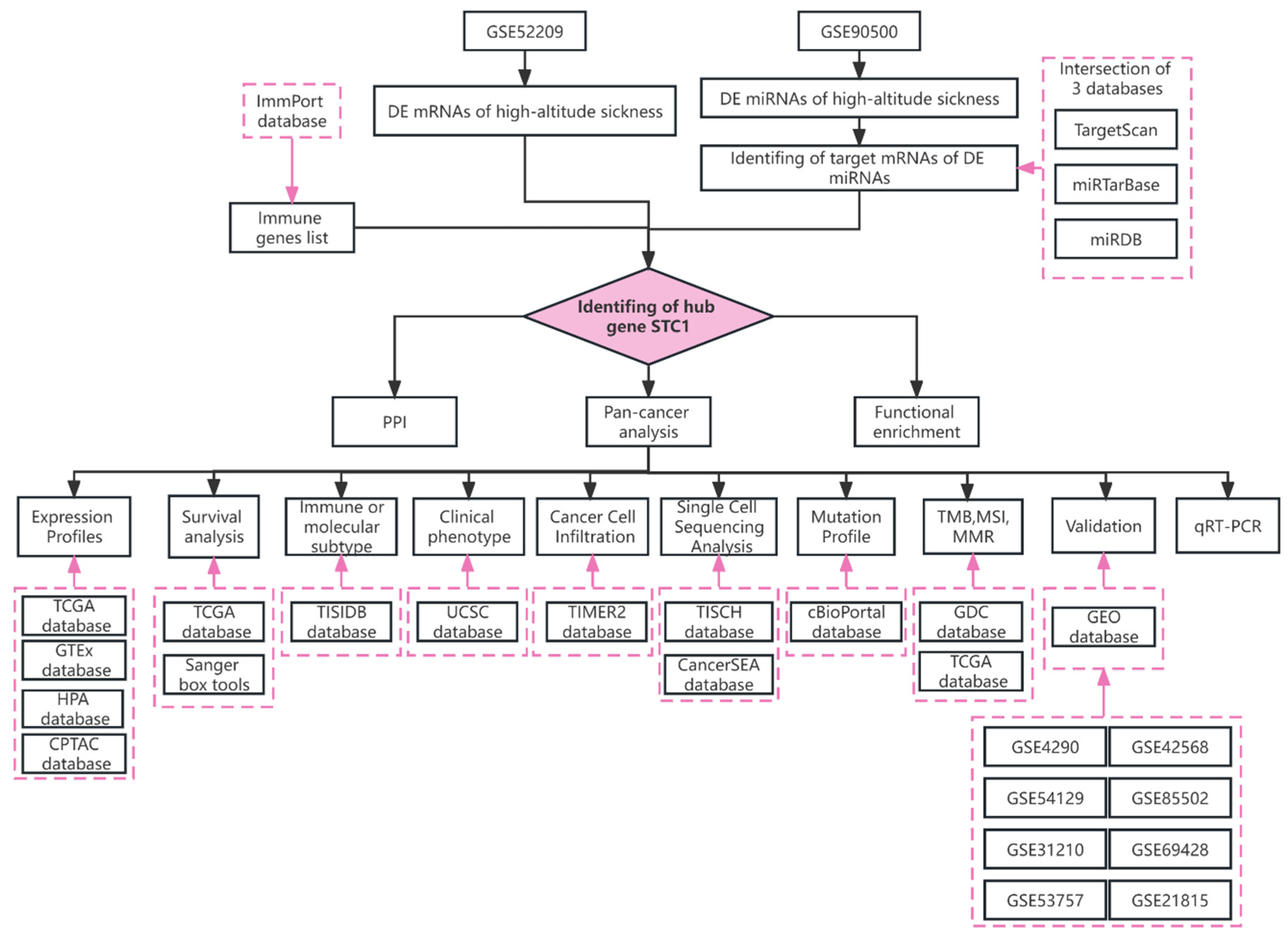
2.1. Identification of the DE-miRNAs and DE-mRNAs
2.2. Identification of the Key Genes
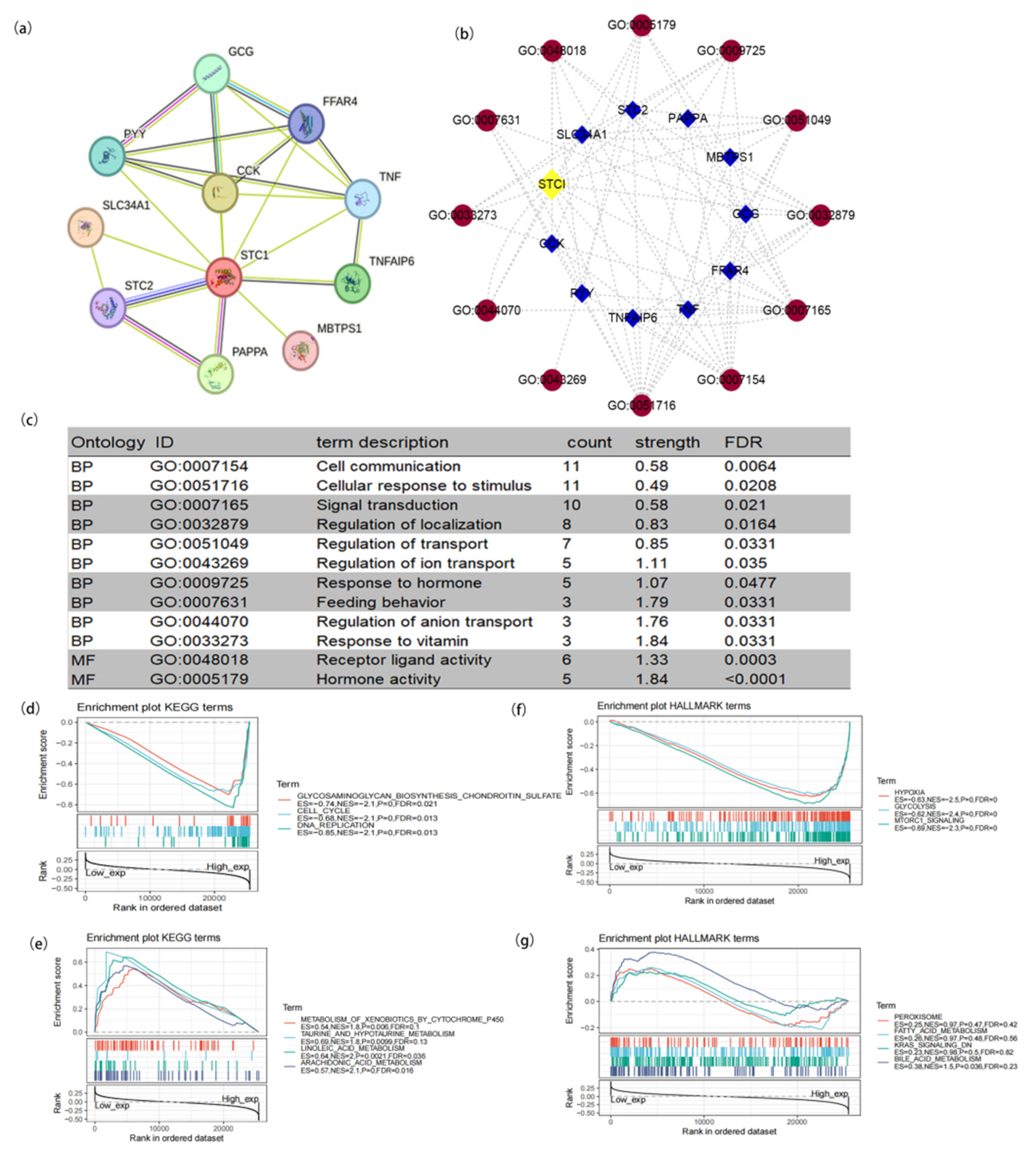
2.3. PPI Network and Function Enrichment of STC1
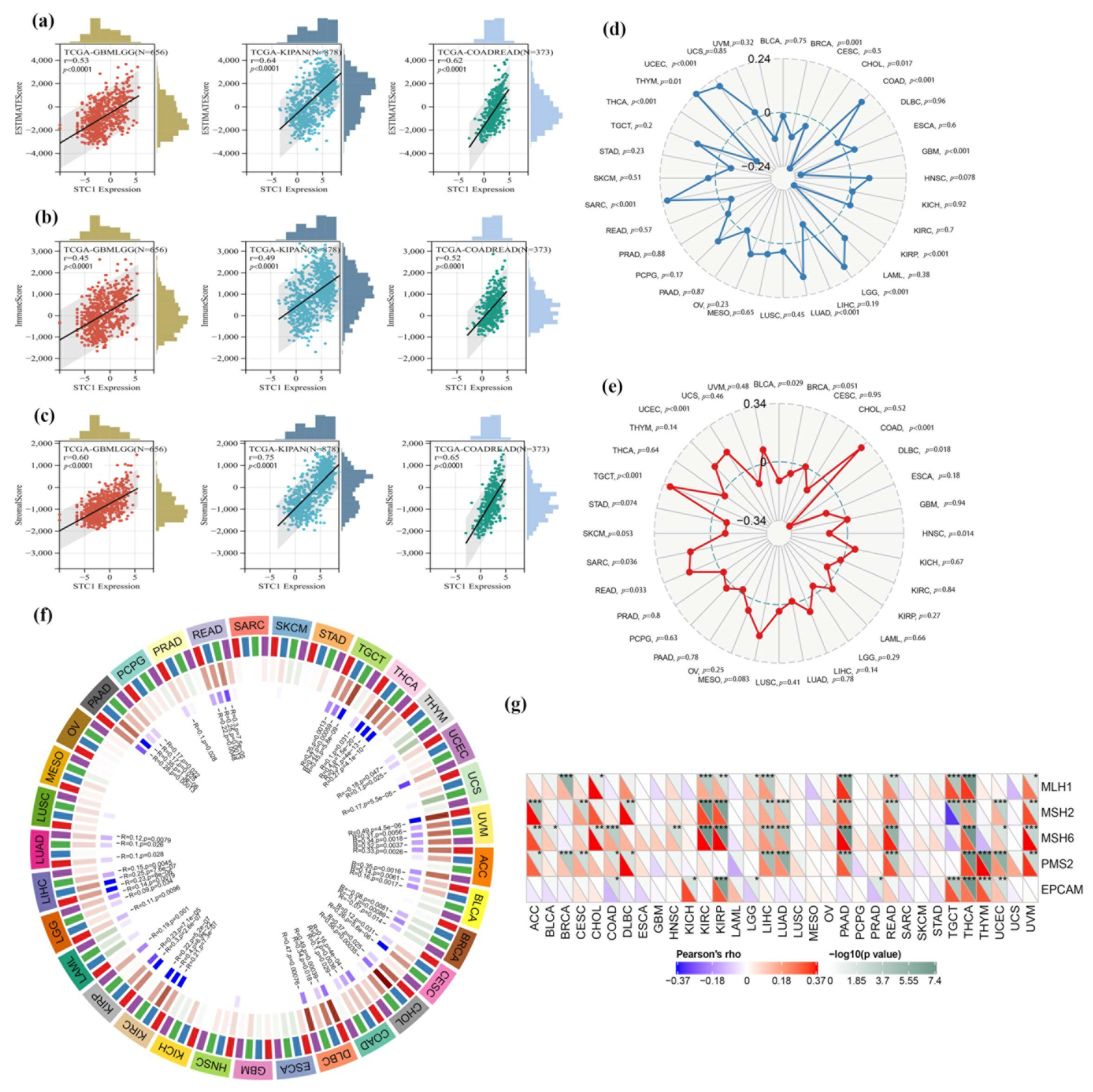
2.4. Analysis of STC1 Expression between Normal and Tumor Samples
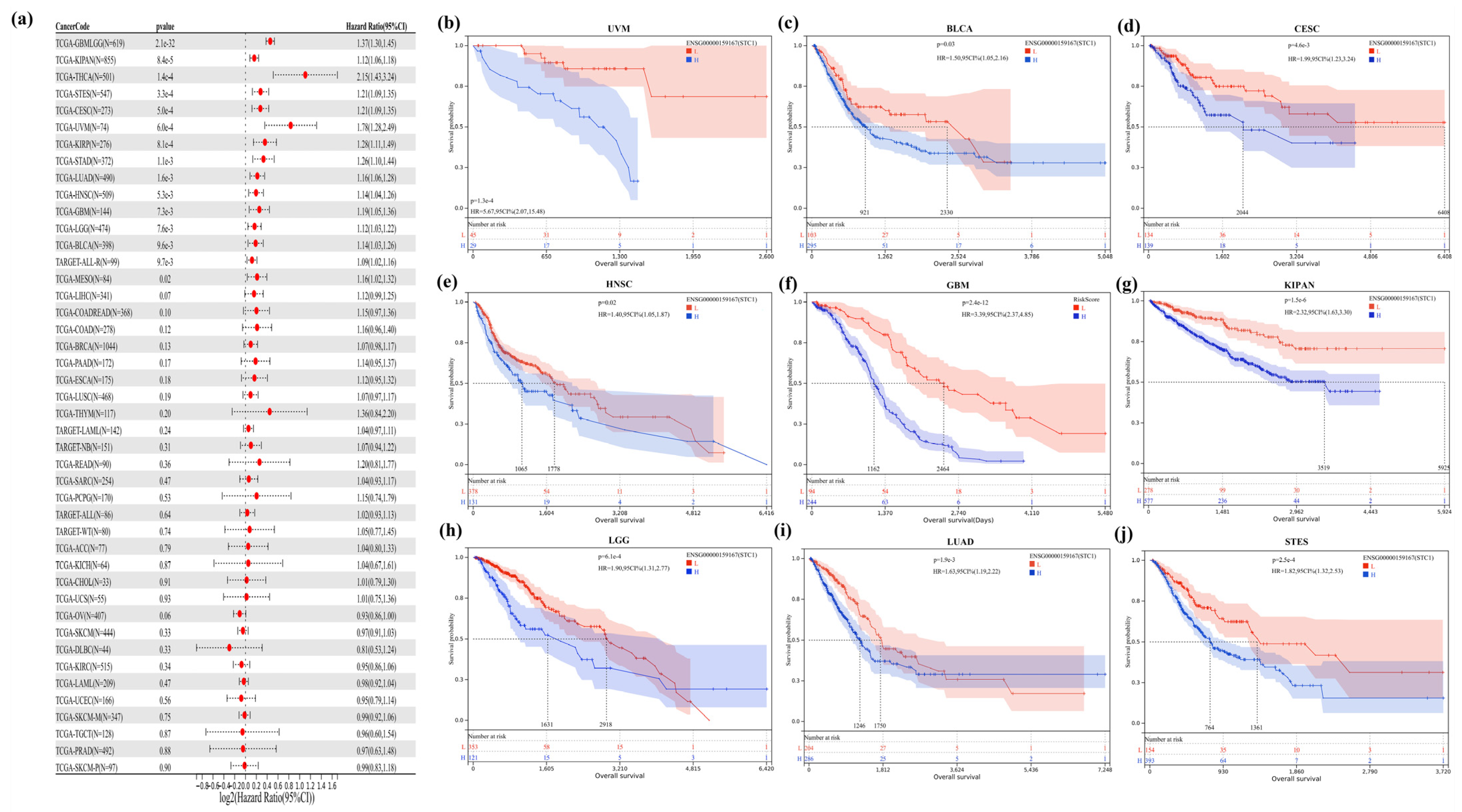
2.5. STC1 Gene Acted as a Survival Prognosis Biomarker
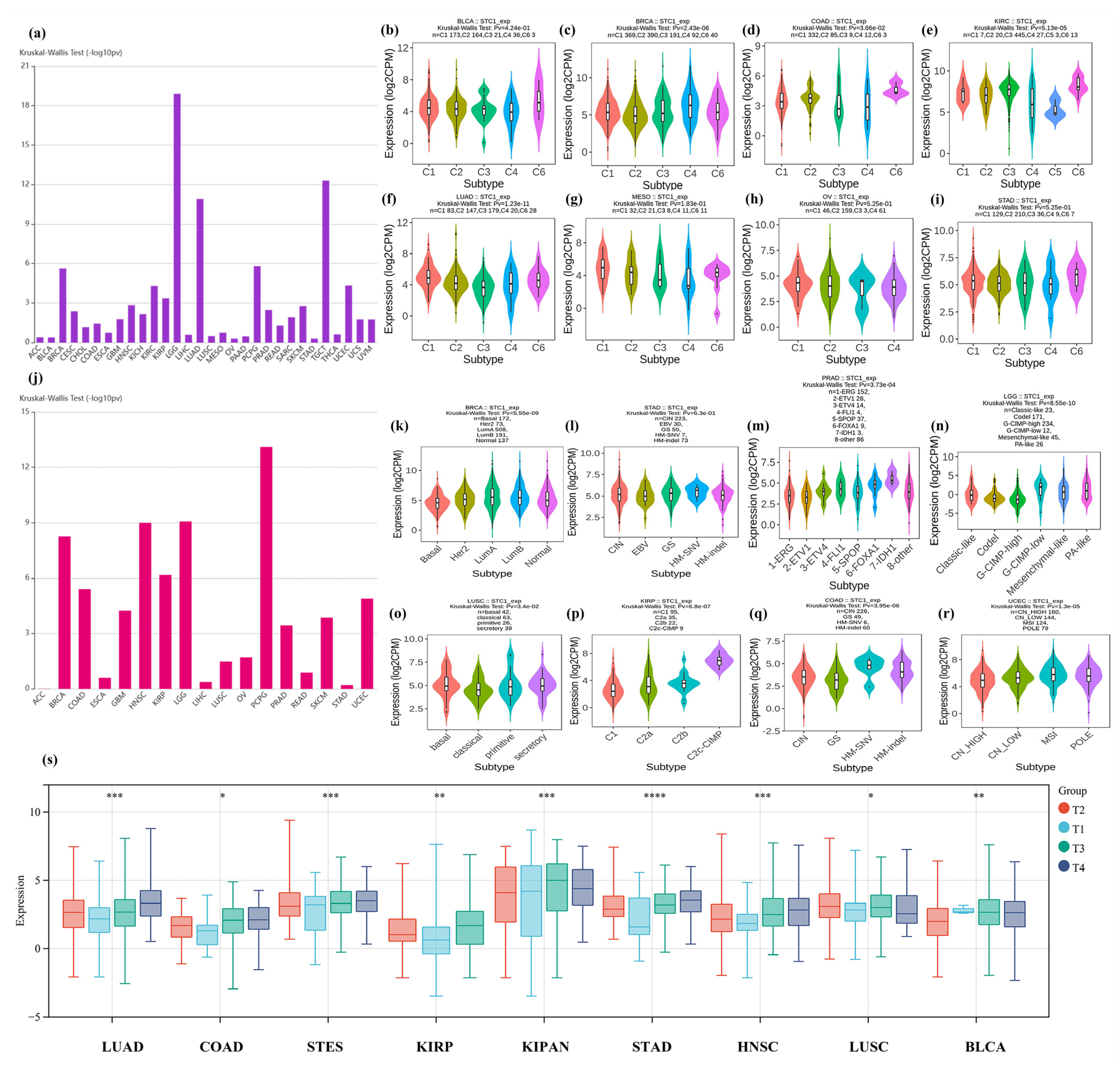
2.6. STC1 Expression in Immune or Molecular Subtypes of Cancers and Clinical Phenotypes
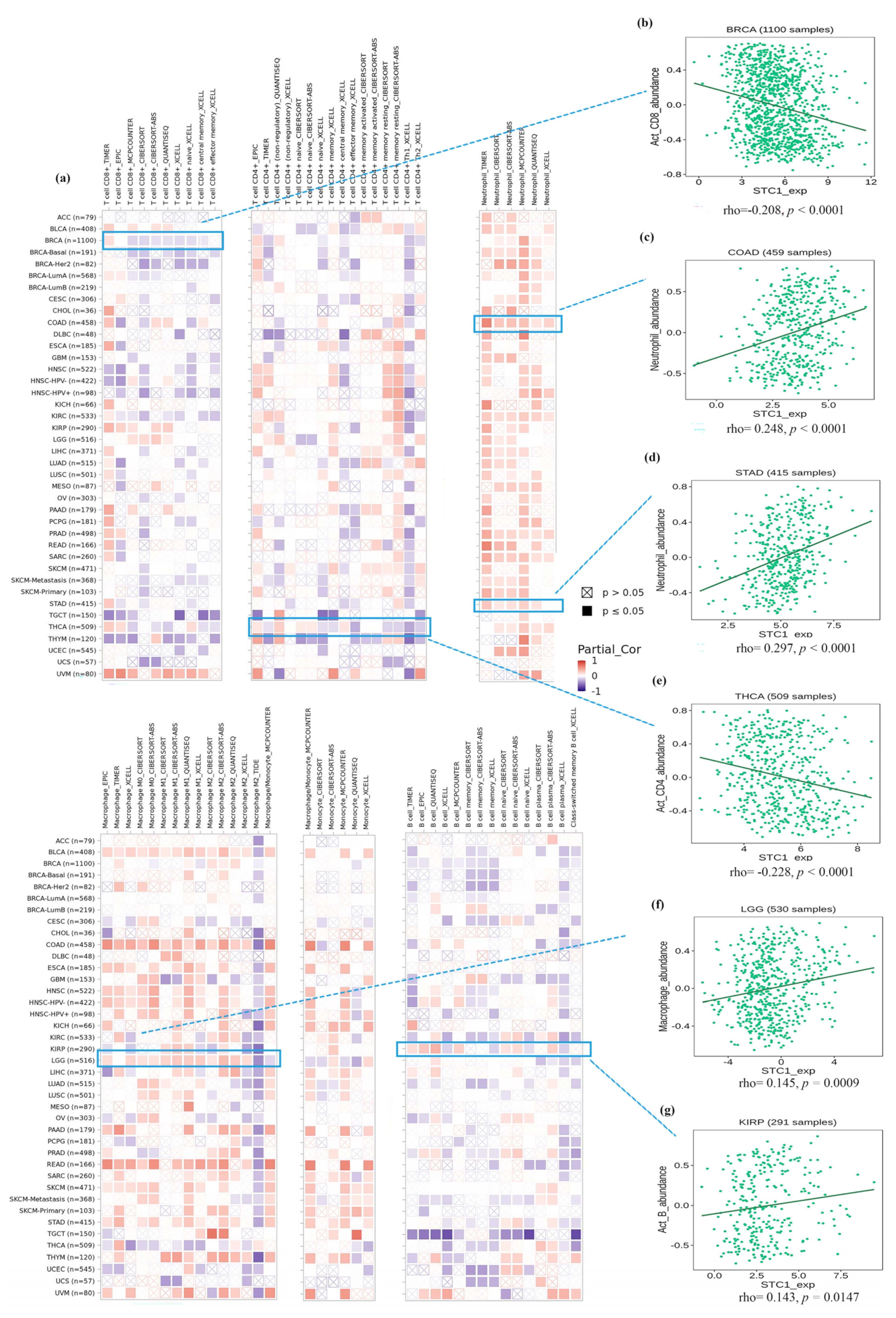
2.7. Immune Cell Infiltration Analysis of STC1 in All Malignancies
2.8. Single-Cell Analysis of STC1 in Cancers
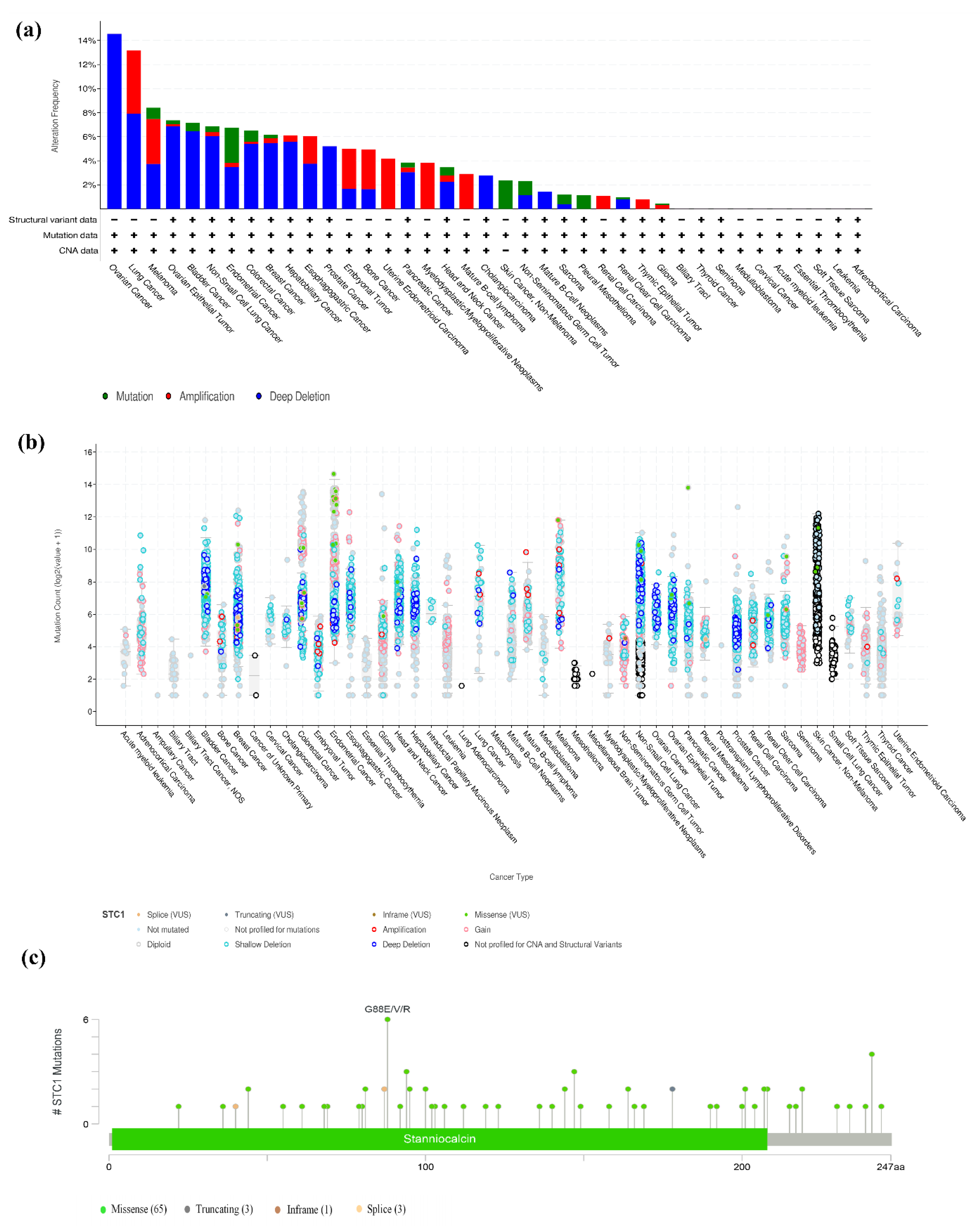
2.9. The Landscape of the STC1 Mutation Profile in Different Tissues
2.10. STC1 Expression in Relation to TMB, MSI, MMR Gene Mutation, and DNA Methylation of STC1
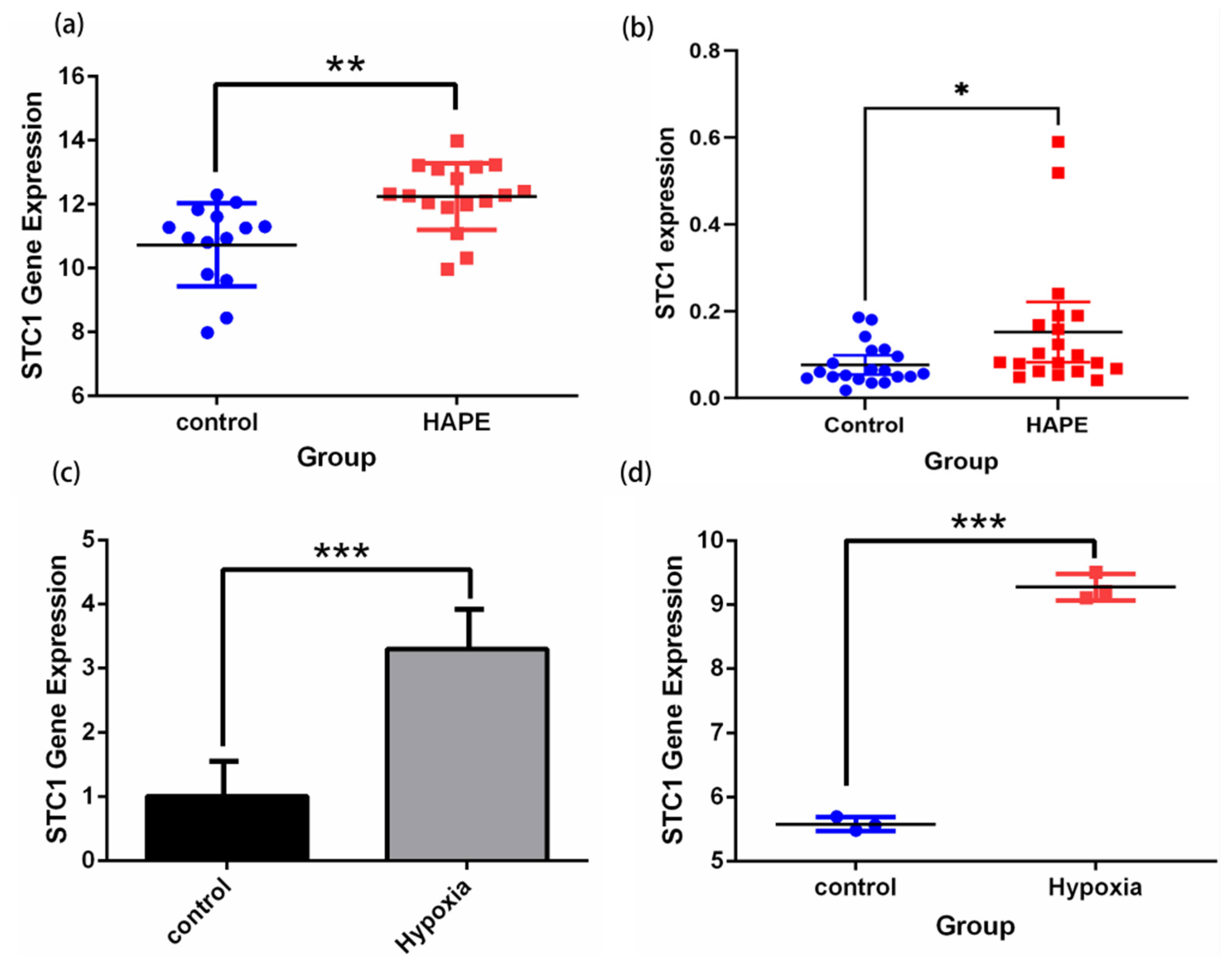
2.11. Validation of STC1 Expression and Its Diagnostic Value in Pan-Cancer GEO Datasets
3. Discussion
4. Materials and Methods
4.1. Data Source and Processing
4.2. STC1 Expression Profiles
4.3. Identification of Target Genes of DE-miRNA
4.4. Protein–Protein Interaction (PPI) Network and Enrichment Analysis of STC1-Related Genes
4.5. Prognosis and Survival Analysis
4.6. Immune or Molecular Subtypes of Cancers and Clinical Phenotypes Analysis
4.7. The Correlation of STC1 Expression with Cancer Cell Infiltration
4.8. Single-Cell Sequencing Analysis of STC1
4.9. Evaluation of Tumor Mutation Burden (TMB), and Tumor Microsatellite Instability (MSI), (Mis-Match Repair) MMR Gene Mutation and DNA Methylation of STC1
4.10. Quantitative RT-PCR (qRT-PCR)
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Richalet, J.P.; Hermand, E.; Lhuissier, F.J. Cardiovascular physiology and pathophysiology at high altitude. Nat. Rev. Cardiol. 2024, 21, 75–88. [Google Scholar] [PubMed]
- Wu, X.H.; He, Y.Y.; Chen, Z.R.; He, Z.Y.; Yan, Y.; He, Y.; Wang, G.M.; Dong, Y.; Yang, Y.; Sun, Y.M.; et al. Single-cell analysis of peripheral blood from high-altitude pulmonary hypertension patients identifies a distinct monocyte phenotype. Nat. Commun. 2023, 14, 1820. [Google Scholar] [PubMed]
- Bigham, A.W.; Lee, F.S. Human high-altitude adaptation: Forward genetics meets the HIF pathway. Genes Dev. 2014, 28, 2189–2204. [Google Scholar]
- McGettrick, A.F.; O’Neill, L.A.J. The Role of HIF in Immunity and Inflammation. Cell Metab. 2020, 32, 524–536. [Google Scholar] [PubMed]
- El Alam, S.; Pena, E.; Aguilera, D.; Siques, P.; Brito, J. Inflammation in Pulmonary Hypertension and Edema Induced by Hypobaric Hypoxia Exposure. Int. J. Mol. Sci. 2022, 23, 12656. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Shrimali, N.M.; Mohammad, G.; Koul, P.A.; Prchal, J.T.; Guchhait, P. Gain-of-function Tibetan PHD2(D4E;C127S) variant suppresses monocyte function: A lesson in inflammatory response to inspired hypoxia. EBioMedicine 2021, 68, 103418. [Google Scholar]
- Liu, P.; Zou, D.; Chen, K.; Zhou, Q.; Gao, Y.; Huang, Y.; Zhu, J.; Zhang, Q.; Mi, M. Dihydromyricetin Improves Hypobaric Hypoxia-Induced Memory Impairment via Modulation of SIRT3 Signaling. Mol. Neurobiol. 2016, 53, 7200–7212. [Google Scholar]
- Yeung, B.H.; Law, A.Y.; Wong, C.K. Evolution and roles of stanniocalcin. Mol. Cell. Endocrinol. 2012, 349, 272–280. [Google Scholar]
- Wan, H.T.; Ng, A.H.; Lee, W.K.; Shi, F.; Wong, C.K. Identification and characterization of a membrane receptor that binds to human STC1. Life Sci. Alliance 2022, 5, e202201497. [Google Scholar]
- Chakraborty, A.; Brooks, H.; Zhang, P.; Smith, W.; McReynolds, M.R.; Hoying, J.B.; Bick, R.; Truong, L.; Poindexter, B.; Lan, H.; et al. Stanniocalcin-1 regulates endothelial gene expression and modulates transendothelial migration of leukocytes. Am. J. Physiol. Renal Physiol. 2007, 292, F895–F904. [Google Scholar]
- Kanellis, J.; Bick, R.; Garcia, G.; Truong, L.; Tsao, C.C.; Etemadmoghadam, D.; Poindexter, B.; Feng, L.; Johnson, R.J.; Sheikh-Hamad, D. Stanniocalcin-1, an inhibitor of macrophage chemotaxis and chemokinesis. Am. J. Physiol. Renal Physiol. 2004, 286, F356–F362. [Google Scholar]
- Chen, C.; Jamaluddin, M.S.; Yan, S.; Sheikh-Hamad, D.; Yao, Q. Human stanniocalcin-1 blocks TNF-alpha-induced monolayer permeability in human coronary artery endothelial cells. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 906–912. [Google Scholar] [PubMed]
- Wang, Y.; Huang, L.; Abdelrahim, M.; Cai, Q.; Truong, A.; Bick, R.; Poindexter, B.; Sheikh-Hamad, D. Stanniocalcin-1 suppresses superoxide generation in macrophages through induction of mitochondrial UCP2. J. Leukoc. Biol. 2009, 86, 981–988. [Google Scholar] [PubMed]
- Roddy, G.W.; Rosa, R.H., Jr.; Oh, J.Y.; Ylostalo, J.H.; Bartosh, T.J., Jr.; Choi, H.; Lee, R.H.; Yasumura, D.; Ahern, K.; Nielsen, G.; et al. Stanniocalcin-1 rescued photoreceptor degeneration in two rat models of inherited retinal degeneration. Mol. Ther. 2012, 20, 788–797. [Google Scholar]
- Nanou, A.; Lorenzo-Moldero, I.; Gazouleas, K.D.; Cortese, B.; Moroni, L. 3D Culture Modeling of Metastatic Breast Cancer Cells in Additive Manufactured Scaffolds. ACS Appl. Mater. Interfaces 2022, 14, 28389–28402. [Google Scholar]
- Chan, K.K.; Hon, T.C.; Au, K.Y.; Choi, H.L.; Wong, D.K.; Chan, A.C.; Yuen, M.F.; Lai, C.L.; Lo, R.C. Stanniocalcin 1 is a serum biomarker and potential therapeutic target for HBV-associated liver fibrosis. J. Pathol. 2022, 257, 227–238. [Google Scholar]
- Long, N.; Xu, X.; Lin, H.; Lv, Y.; Zou, S.; Cao, H.; Chen, X.; Zhao, Y.; Qi, X.; Yang, H.; et al. Circular RNA circPOSTN promotes neovascularization by regulating miR-219a-2-3p/STC1 axis and stimulating the secretion of VEGFA in glioblastoma. Cell Death Discov. 2022, 8, 349. [Google Scholar] [PubMed]
- Zhao, F.; Yang, G.; Qiu, J.; Liu, Y.; Tao, J.; Chen, G.; Su, D.; You, L.; Zheng, L.; Zhang, T.; et al. HIF-1alpha-regulated stanniocalcin-1 mediates gemcitabine resistance in pancreatic ductal adenocarcinoma via PI3K/AKT signaling pathway. Mol. Carcinog. 2022, 61, 839–850. [Google Scholar]
- Sengun, S.; Korkmaz, H.; Ciris, M.; Yuceer, R.O.; Boyluboy, S.M.; Kiran, M. Diagnostic and prognostic value of Stanniocalcin 1 expression in papillary thyroid cancer. Endocrine 2022, 78, 95–103. [Google Scholar]
- Lin, F.; Li, X.; Wang, X.; Sun, H.; Wang, Z.; Wang, X. Stanniocalcin 1 promotes metastasis, lipid metabolism and cisplatin chemoresistance via the FOXC2/ITGB6 signaling axis in ovarian cancer. J. Exp. Clin. Cancer Res. 2022, 41, 129. [Google Scholar] [PubMed]
- Yu, S.; Hu, C.; Cai, L.; Du, X.; Lin, F.; Yu, Q.; Liu, L.; Zhang, C.; Liu, X.; Li, W.; et al. Seven-Gene Signature Based on Glycolysis Is Closely Related to the Prognosis and Tumor Immune Infiltration of Patients With Gastric Cancer. Front. Oncol. 2020, 10, 1778. [Google Scholar]
- Li, R.; Liu, R.; Wu, S.; Zheng, S.; Ye, L.; Shao, Y. Prognostic value of STC1 in solid tumors: A meta-analysis. Biomark. Med. 2022, 16, 253–263. [Google Scholar]
- Law, A.Y.; Ching, L.Y.; Lai, K.P.; Wong, C.K. Identification and characterization of the hypoxia-responsive element in human stanniocalcin-1 gene. Mol. Cell. Endocrinol. 2010, 314, 118–127. [Google Scholar]
- Liu, R.; Muliadi, V.; Mou, W.; Li, H.; Yuan, J.; Holmberg, J.; Chambers, B.J.; Ullah, N.; Wurth, J.; Alzrigat, M.; et al. HIF-1 stabilization in T cells hampers the control of Mycobacterium tuberculosis infection. Nat. Commun. 2022, 13, 5093. [Google Scholar]
- Xie, H.; Zhou, J.; Liu, X.; Xu, Y.; Hepperla, A.J.; Simon, J.M.; Wang, T.; Yao, H.; Liao, C.; Baldwin, A.S.; et al. USP13 promotes deubiquitination of ZHX2 and tumorigenesis in kidney cancer. Proc. Natl. Acad. Sci. USA 2022, 119, e2119854119. [Google Scholar]
- Fang, Z.; Tian, Z.; Luo, K.; Song, H.; Yi, J. Clinical significance of stanniocalcin expression in tissue and serum of gastric cancer patients. Chin. J. Cancer Res. 2014, 26, 602–610. [Google Scholar] [PubMed]
- Shirakawa, M.; Fujiwara, Y.; Sugita, Y.; Moon, J.H.; Takiguchi, S.; Nakajima, K.; Miyata, H.; Yamasaki, M.; Mori, M.; Doki, Y. Assessment of stanniocalcin-1 as a prognostic marker in human esophageal squamous cell carcinoma. Oncol. Rep. 2012, 27, 940–946. [Google Scholar] [PubMed]
- Li, H.; Li, Q.; Lian, J.; Chu, Y.; Fang, K.; Xu, A.; Chen, T.; Xu, M. MLL2 promotes cancer cell lymph node metastasis by interacting with RelA and facilitating STC1 transcription. Cell. Signal. 2020, 65, 109457. [Google Scholar]
- Zhang, R.; Liu, Q.; Zhou, S.; He, H.; Zhao, M.; Ma, W. Mesenchymal stem cell suppresses the efficacy of CAR-T toward killing lymphoma cells by modulating the microenvironment through stanniocalcin-1. eLife 2023, 12, e82934. [Google Scholar] [PubMed]
- Murdaca, G.; Greco, M.; Tonacci, A.; Negrini, S.; Borro, M.; Puppo, F.; Gangemi, S. IL-33/IL-31 Axis in Immune-Mediated and Allergic Diseases. Int. J. Mol. Sci. 2019, 20, 2856. [Google Scholar] [CrossRef]
- Zhou, Z.; Lu, Z.R. Molecular imaging of the tumor microenvironment. Adv. Drug Deliv. Rev. 2017, 113, 24–48. [Google Scholar]
- Patel, A.; Sant, S. Hypoxic tumor microenvironment: Opportunities to develop targeted therapies. Biotechnol. Adv. 2016, 34, 803–812. [Google Scholar]
- Ellard, J.P.; McCudden, C.R.; Tanega, C.; James, K.A.; Ratkovic, S.; Staples, J.F.; Wagner, G.F. The respiratory effects of stanniocalcin-1 (STC-1) on intact mitochondria and cells: STC-1 uncouples oxidative phosphorylation and its actions are modulated by nucleotide triphosphates. Mol. Cell. Endocrinol. 2007, 264, 90–101. [Google Scholar]
- Zhao, F.; Yang, G.; Feng, M.; Cao, Z.; Liu, Y.; Qiu, J.; You, L.; Zheng, L.; Zhang, T.; Zhao, Y. Expression, function and clinical application of stanniocalcin-1 in cancer. J. Cell. Mol. Med. 2020, 24, 7686–7696. [Google Scholar]
- Zhang, Q.; Hu, H.; Chen, S.Y.; Liu, C.J.; Hu, F.F.; Yu, J.; Wu, Y.; Guo, A.Y. Transcriptome and Regulatory Network Analyses of CD19-CAR-T Immunotherapy for B-ALL. Genom. Proteom. Bioinform. 2019, 17, 190–200. [Google Scholar]
- Chen, Y.; Wang, X. miRDB: An online database for prediction of functional microRNA targets. Nucleic Acids Res. 2020, 48, D127–D131. [Google Scholar]
- Huang, H.Y.; Lin, Y.C.; Cui, S.; Huang, Y.; Tang, Y.; Xu, J.; Bao, J.; Li, Y.; Wen, J.; Zuo, H.; et al. miRTarBase update 2022: An informative resource for experimentally validated miRNA-target interactions. Nucleic Acids Res. 2022, 50, D222–D230. [Google Scholar]
- McGeary, S.E.; Lin, K.S.; Shi, C.Y.; Pham, T.M.; Bisaria, N.; Kelley, G.M.; Bartel, D.P. The biochemical basis of microRNA targeting efficacy. Science 2019, 366, eaav1741. [Google Scholar] [PubMed]
- Smoot, M.E.; Ono, K.; Ruscheinski, J.; Wang, P.L.; Ideker, T. Cytoscape 2.8: New features for data integration and network visualization. Bioinformatics 2011, 27, 431–432. [Google Scholar]
- Shen, W.; Song, Z.; Zhong, X.; Huang, M.; Shen, D.; Gao, P.; Qian, X.; Wang, M.; He, X.; Wang, T.; et al. Sangerbox: A comprehensive, interaction-friendly clinical bioinformatics analysis platform. iMeta 2022, 1, e36. [Google Scholar]
- Ru, B.; Wong, C.N.; Tong, Y.; Zhong, J.Y.; Zhong, S.S.W.; Wu, W.C.; Chu, K.C.; Wong, C.Y.; Lau, C.Y.; Chen, I.; et al. TISIDB: An integrated repository portal for tumor-immune system interactions. Bioinformatics 2019, 35, 4200–4202. [Google Scholar] [PubMed]
- Shahamatdar, S.; He, M.X.; Reyna, M.A.; Gusev, A.; AlDubayan, S.H.; Van Allen, E.M.; Ramachandran, S. Germline Features Associated with Immune Infiltration in Solid Tumors. Cell Rep. 2020, 30, 2900–2908.e4. [Google Scholar] [PubMed]


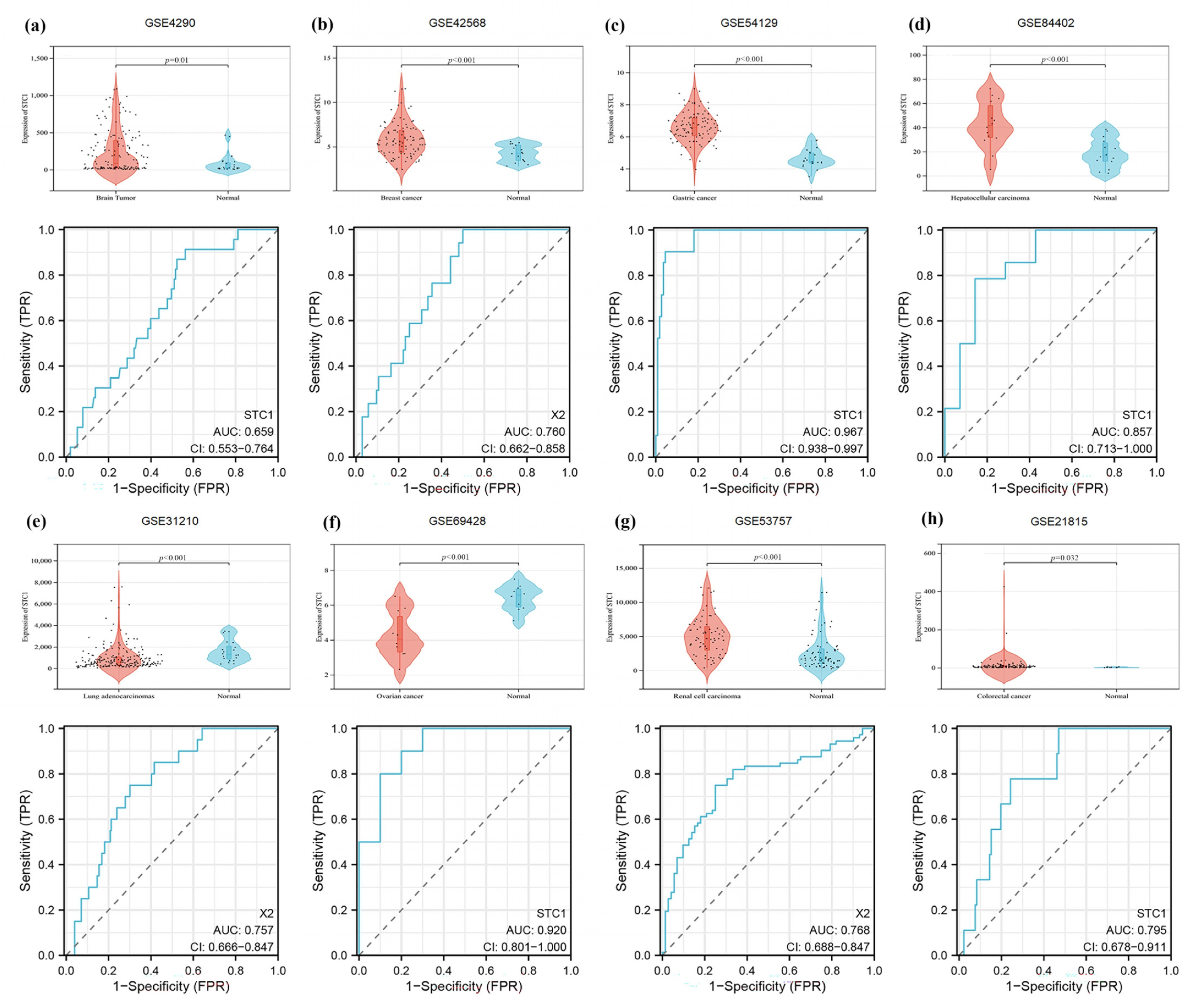
| GEO Dataset | Application | Title | Platform |
|---|---|---|---|
| GSE90500 | Test set | Circulating microRNAs as a signature predicting the occurrence of acute mountain sickness before exposure to high altitude | GPL18058 (Exiqon miRCURY LNA microRNA array, 7th generation) |
| GSE52209 | Test set | Gene expression profiling under exposure to a high-altitude hypoxic environment | GPL9365 (Ocimum Biosolutions Human 40k OciChip) |
| GSE145935 | Validation set | Transcriptomic analysis of human astrocytes in vitro reveals hypoxia-induced mitochondrial dysfunction, modulation of metabolism, and dysregulation of the immune response | GPL570 [HG-U133_Plus_2] Affymetrix Human Genome U133 Plus 2.0 Array |
| GSE4290 | Validation set | Expression data for glioma samples from Henry Ford Hospital | GPL570 [HG-U133_Plus_2] Affymetrix Human Genome U133 Plus 2.0 Array |
| GSE42568 | Validation set | Breast cancer gene expression analysis | GPL570 [HG-U133_Plus_2] Affymetrix Human Genome U133 Plus 2.0 Array |
| GSE54129 | Validation set | Global gene expression analysis of gastric cancer by oligonucleotide microarrays | GPL570 [HG-U133_Plus_2] Affymetrix Human Genome U133 Plus 2.0 Array |
| GSE85502 | Validation set | Genome-wide profiling of H3K27me3 in Drosophila primary spermatocytes | GPL15057NimbleGen Drosophila melanogaster Whole Genome 2.1M tiling array [DM_5_Catalog_tiling_HX1; DesignID 6725] |
| GSE31210 | Validation set | Gene expression data for pathological stage I-II lung adenocarcinomas | GPL570 [HG-U133_Plus_2] Affymetrix Human Genome U133 Plus 2.0 Array |
| GSE69428 | Validation set | Transformation of human fallopian tube stem cells and high-grade serous ovarian cancer | GPL570 [HG-U133_Plus_2] Affymetrix Human Genome U133 Plus 2.0 Array |
| GSE53757 | Validation set | Gene array analysis of clear cell renal cell carcinoma tissue versus matched normal kidney tissue | GPL570 [HG-U133_Plus_2] Affymetrix Human Genome U133 Plus 2.0 Array |
| GSE21815 | Validation set | Gene expression profiles in 132 laser microdissected colorectal cancer tissues | GPL 6480Agilent-014850 Whole Human Genome Microarray 4x44K G4112F (Probe Name version) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Q.; Xu, Z.; Gong, Q.; Shen, X. Identification and Validation of STC1 Act as a Biomarker for High-Altitude Diseases and Its Pan-Cancer Analysis. Int. J. Mol. Sci. 2024, 25, 9085. https://doi.org/10.3390/ijms25169085
Li Q, Xu Z, Gong Q, Shen X. Identification and Validation of STC1 Act as a Biomarker for High-Altitude Diseases and Its Pan-Cancer Analysis. International Journal of Molecular Sciences. 2024; 25(16):9085. https://doi.org/10.3390/ijms25169085
Chicago/Turabian StyleLi, Qiong, Zhichao Xu, Qianhui Gong, and Xiaobing Shen. 2024. "Identification and Validation of STC1 Act as a Biomarker for High-Altitude Diseases and Its Pan-Cancer Analysis" International Journal of Molecular Sciences 25, no. 16: 9085. https://doi.org/10.3390/ijms25169085
APA StyleLi, Q., Xu, Z., Gong, Q., & Shen, X. (2024). Identification and Validation of STC1 Act as a Biomarker for High-Altitude Diseases and Its Pan-Cancer Analysis. International Journal of Molecular Sciences, 25(16), 9085. https://doi.org/10.3390/ijms25169085








