Narrow-Headed Voles Species Complex (Cricetidae, Rodentia): Evidence for Species Differentiation Inferred from Transcriptome Data
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. RNA Isolation, Library Preparation, and Sequencing
2.3. De Novo Transcriptome Assembly
2.4. SNP Calling and Search of Associations
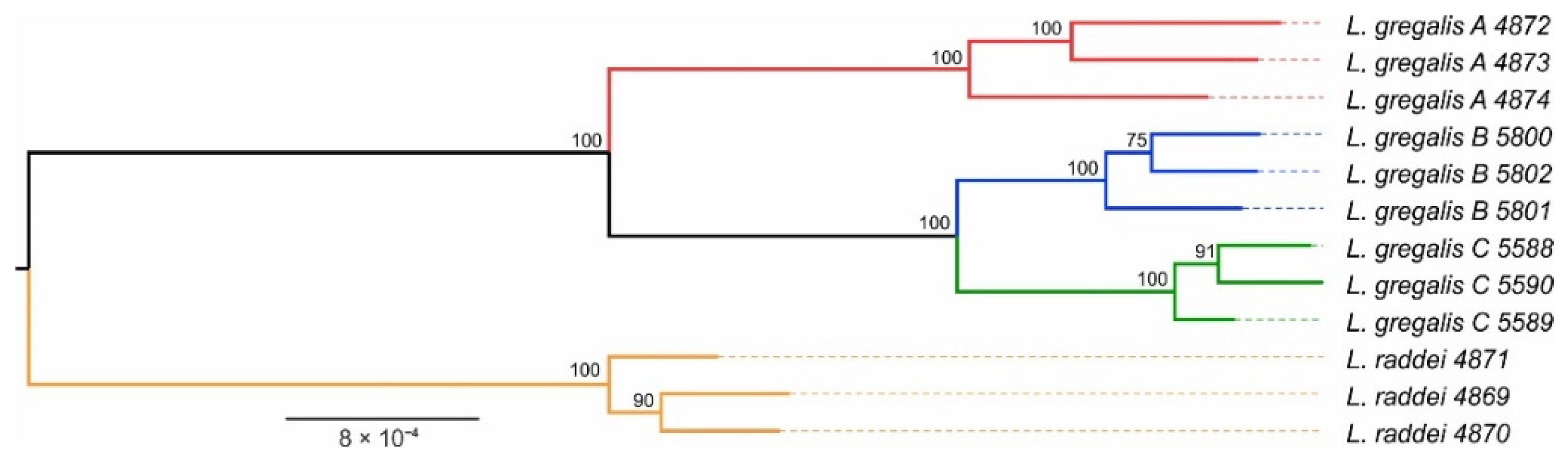
2.5. Phylogenetic Reconstruction
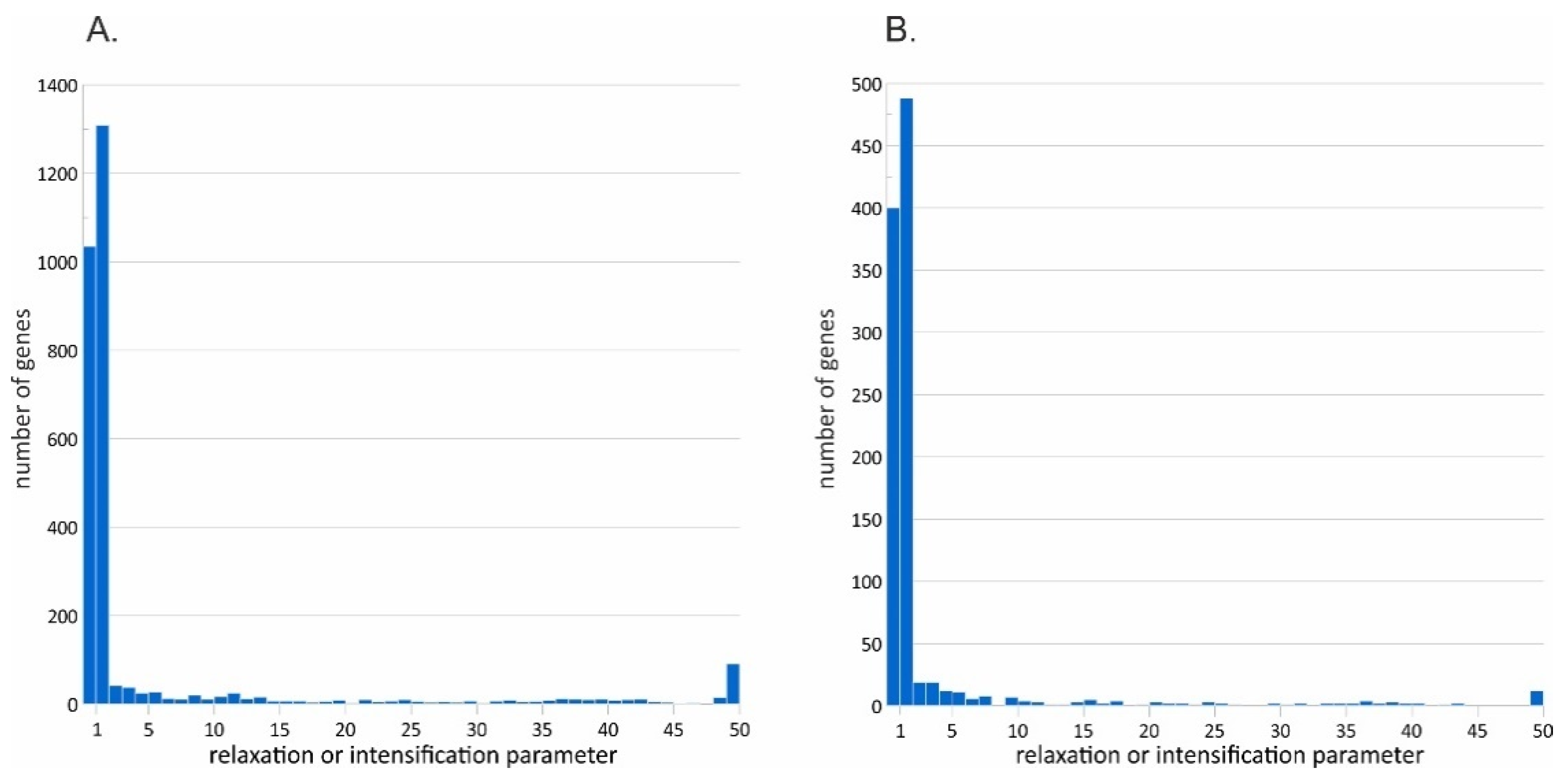
2.6. Natural Selection Estimation
3. Results
3.1. Transcriptome Assembly
3.2. Phylogenetic Reconstruction
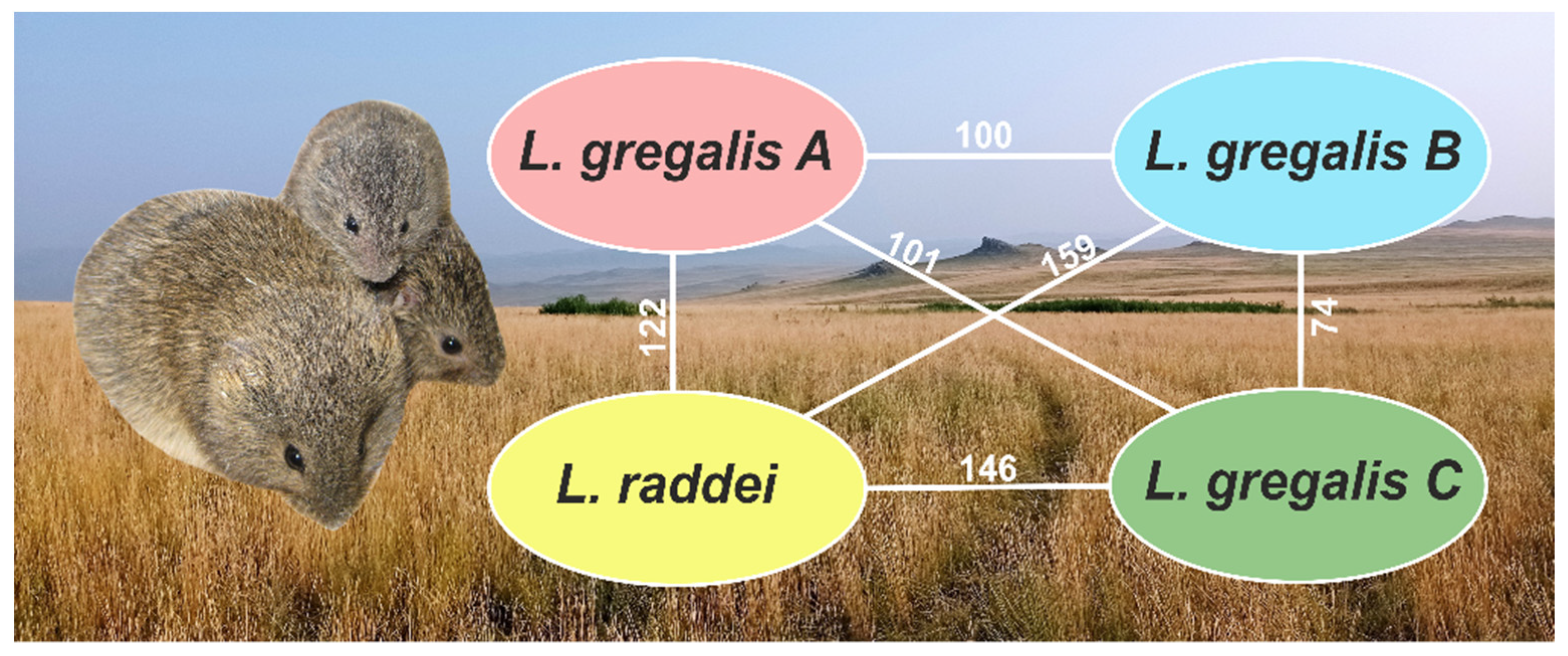
3.3. Variation Calling
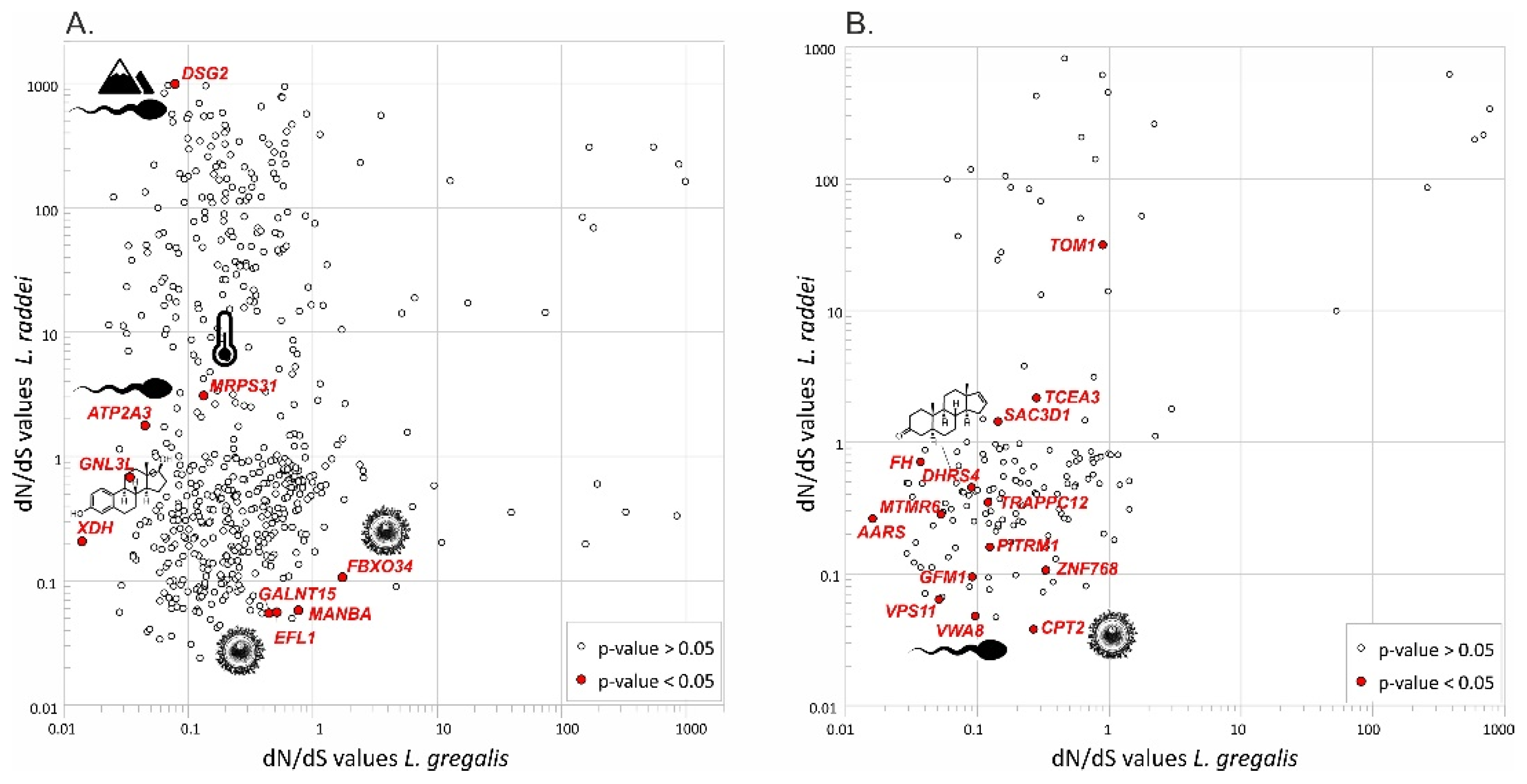
3.4. Signatures of Natural Selection
4. Discussion
4.1. Molecular Mechanisms Underlying Speciation between Cryptic Species L. gregalis and L. raddei
4.2. Traces of Molecular Differentiation at the Early Stages of Speciation within L. gregalis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dupal, T.A. Geographical Variation and Subspecies Systematics of Narrow-Headed Vole Microtus (Stenocranius) Gregalis (Rodentia, Cricetidae). Zool. Zhurnal 2000, 79, 851–858. [Google Scholar]
- Golenishchev, F.N.; Petrovskaya, N.A. Geographic Variation of Narrow-Headed Vole Microtus (Stenocranius) Gregalis Pall., 1779. Theriol. Issled. 2002, 1, 17–34. [Google Scholar]
- Dupal, T.A.; Abramov, S.A. Intrapopulation Morphological Variability of Narrow-Headed Vole (Microtus Gregalis, Rodentia, Arvicolidae). Zool. Zhurnal 2010, 89, 850–861. [Google Scholar]
- Gromov, I.M.; Polyakov, I.Y. Voles (Microtinae); Brill Academic Publishers: Boston, MA, USA, 1992; Volume 3. [Google Scholar]
- Ognev, S.I. Zveri SSSR I Prilezhashhih Stran (The Mammals of the USSR and Adjacent Countries). Vol VII. Gryzuny (Rodentia); Publ. Acad. Nauk USSR: Leningrad, Russia, 1950. [Google Scholar]
- Gromov, I.M.; Erbaeva, M.A. Mammals of the Fauna of Russia and Adjacent Regions. Lagomorphs and Rodents; Zoological Institute RAS: Saint Petersburg, Russia, 1995. [Google Scholar]
- Petrova, T.V.; Zakharov, E.S.; Samiya, R.; Abramson, N.I. Phylogeography of the Narrow-Headed Vole Lasiopodomys (Stenocranius) Gregalis (Cricetidae, Rodentia) Inferred from Mitochondrial Cytochrome b Sequences: An Echo of Pleistocene Prosperity. J. Zool. Syst. Evol. Res. 2015, 53, 97–108. [Google Scholar] [CrossRef]
- Lissovsky, A.A.; Obolenskaya, E.V.; Petrova, T.V. Morphological and Genetic Variation of Narrow-Headed Voles Lasiopo-Domys Gregalis from South-East Transbaikalia. Russ. J. Theriol. 2013, 12, 83–90. [Google Scholar] [CrossRef]
- Petrova, T.V.; Tesakov, A.S.; Kowalskaya, Y.M.; Abramson, N.I. Cryptic Speciation in the Narrow-Headed Vole Lasiopodomys (Stenocranius) Gregalis (Rodentia: Cricetidae). Zool. Scr. 2016, 45, 618–629. [Google Scholar] [CrossRef]
- Kowalskaya, Y.M.; Petrova, T.V.; Abramson, N.I. Preliminary Results of Experimental Hybridization of Mitochondrial Forms of the Narrow-Headed Vole Lasiopodomys (Stenocranius) Gregalis (Rodentia, Arvicolinae); KMK Scientific Press: Moscow, Russia, 2015; p. 92. [Google Scholar]
- Petrova, T.V.; Genelt-Yanovskiy, E.A.; Lissovsky, A.A.; Chash, U.-M.G.; Masharsky, A.E.; Abramson, N.I. Signatures of Genetic Isolation of the Three Lineages of the Narrow-Headed Vole Lasiopodomys Gregalis (Cricetidae, Rodentia) in a Mosaic Steppe Landscape of South Siberia. Mamm. Biol. 2021, 101, 275–285. [Google Scholar] [CrossRef]
- Coyne, J.A.; Orr, H.A. Speciation; Vancouver: Sunderland, MA, USA, 2004. [Google Scholar]
- McKinnon, J.S.; Rundle, H.D. Speciation in Nature: The Threespine Stickleback Model Systems. Trends Ecol. Evol. 2002, 17, 480–488. [Google Scholar] [CrossRef]
- Räsänen, K.; Hendry, A.P. Disentangling Interactions between Adaptive Divergence and Gene Flow When Ecology Drives Diversification: Adaptive Divergence and Gene Flow. Ecol. Lett. 2008, 11, 624–636. [Google Scholar] [CrossRef]
- Orr, H.A. The Genetic Basis of Reproductive Isolation: Insights from Drosophila. Proc. Natl. Acad. Sci. USA 2005, 102, 6522–6526. [Google Scholar] [CrossRef] [Green Version]
- Moehring, A.J.; Llopart, A.; Elwyn, S.; Coyne, J.A.; Mackay, T.F.C. The Genetic Basis of Prezygotic Reproductive Isolation Between Drosophila Santomea and D. Yakuba Due to Mating Preference. Genetics 2006, 173, 215–223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rogers, S.M.; Bernatchez, L. The Genetic Basis of Intrinsic and Extrinsic Post-Zygotic Reproductive Isolation Jointly Promoting Speciation in the Lake Whitefish Species Complex (Coregonus Clupeaformis). J. Evol. Biol. 2006, 19, 1979–1994. [Google Scholar] [CrossRef] [PubMed]
- Howard, D.J.; Marshall, J.L.; Hampton, D.D.; Britch, S.C.; Draney, M.L.; Chu, J.; Cantrell, R.G. The Genetics of Reproductive Isolation: A Retrospective and Prospective Look with Comments on Ground Crickets. Am. Nat. 2002, 159, S8–S21. [Google Scholar] [CrossRef] [PubMed]
- Pogson, G.H. Studying the Genetic Basis of Speciation in High Gene Flow Marine Invertebrates. Curr. Zool. 2016, 62, 643–653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poelstra, J.W.; Ellegren, H.; Wolf, J.B.W. An Extensive Candidate Gene Approach to Speciation: Diversity, Divergence and Linkage Disequilibrium in Candidate Pigmentation Genes across the European Crow Hybrid Zone. Heredity 2013, 111, 467–473. [Google Scholar] [CrossRef] [Green Version]
- Janoušek, V.; Wang, L.; Luzynski, K.; Dufková, P.; Vyskočilová, M.M.; Nachman, M.W.; Munclinger, P.; Macholán, M.; Piálek, J.; Tucker, P.K. Genome-Wide Architecture of Reproductive Isolation in a Naturally Occurring Hybrid Zone between Mus Musculus Musculus and M. m. Domesticus: Reproductive Isolation in House Mouse. Mol. Ecol. 2012, 21, 3032–3047. [Google Scholar] [CrossRef] [Green Version]
- Turner, L.M.; Harr, B. Genome-Wide Mapping in a House Mouse Hybrid Zone Reveals Hybrid Sterility Loci and Dobzhansky-Muller Interactions. eLife 2014, 3, e02504. [Google Scholar] [CrossRef]
- Malinsky, M.; Challis, R.J.; Tyers, A.M.; Schiffels, S.; Terai, Y.; Ngatunga, B.P.; Miska, E.A.; Durbin, R.; Genner, M.J.; Turner, G.F. Genomic Islands of Speciation Separate Cichlid Ecomorphs in an East African Crater Lake. Science 2015, 350, 1493–1498. [Google Scholar] [CrossRef] [Green Version]
- Nosil, P.; Egan, S.P.; Funk, D.J. Heterogeneous Genomic Differentiation between Walking-Stick Ecotypes: “Isolation by Adaptation” and Multiple Roles for Divergent Selection. Evolution 2008, 62, 316–336. [Google Scholar] [CrossRef]
- Schmidt, P.S.; Serrão, E.A.; Pearson, G.A.; Riginos, C.; Rawson, P.D.; Hilbish, T.J.; Brawley, S.H.; Trussell, G.C.; Carrington, E.; Wethey, D.S.; et al. Ecological Genetics in the North Atlantic: Environmental Gradients and Adaptation at Specific Loci. Ecology 2008, 89, S91–S107. [Google Scholar] [CrossRef] [Green Version]
- Stinchcombe, J.R.; Hoekstra, H.E. Combining Population Genomics and Quantitative Genetics: Finding the Genes Underlying Ecologically Important Traits. Heredity 2008, 100, 158–170. [Google Scholar] [CrossRef] [Green Version]
- Vincent, B.; Dionne, M.; Kent, M.P.; Lien, S.; Bernatchez, L. Landscape Genomics in Atlantic Salmon (Salmo Salar): Searching for Gene-Environment Interactions Driving Local Adaptation. Evolution 2013, 67, 3469–3487. [Google Scholar] [CrossRef] [PubMed]
- Manthey, J.D.; Moyle, R.G. Isolation by Environment in White-Breasted Nuthatches (Sitta Carolinensis) of the Madrean Archipelago Sky Islands: A Landscape Genomics Approach. Mol. Ecol. 2015, 24, 3628–3638. [Google Scholar] [CrossRef] [PubMed]
- Delport, W.; Scheffler, K.; Seoighe, C. Models of Coding Sequence Evolution. Brief. Bioinform. 2008, 10, 97–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anisimova, M.; Kosiol, C. Investigating Protein-Coding Sequence Evolution with Probabilistic Codon Substitution Models. Mol. Biol. Evol. 2009, 26, 255–271. [Google Scholar] [CrossRef] [Green Version]
- Batsaikhan, N.; Tsytsulina, K.; Formozov, N.; Sheftel, B. Microtus Gregalis. In The IUCN Red List of Threatened Species 2016; The International Union for Conservation of Nature (IUCN): Cambridge, UK, 2016. [Google Scholar]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. 2010. Available online: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 10 April 2020).
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A Flexible Trimmer for Illumina Sequence Data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. Fastp: An Ultra-Fast All-in-One FASTQ Preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef]
- Langmead, B.; Trapnell, C.; Pop, M.; Salzberg, S.L. Ultrafast and Memory-Efficient Alignment of Short DNA Sequences to the Human Genome. Genome Biol. 2009, 10, R25. [Google Scholar] [CrossRef] [Green Version]
- Haas, B.J.; Papanicolaou, A.; Yassour, M.; Grabherr, M.; Blood, P.D.; Bowden, J.; Couger, M.B.; Eccles, D.; Li, B.; Lieber, M.; et al. De Novo Transcript Sequence Reconstruction from RNA-Seq Using the Trinity Platform for Reference Generation and Analysis. Nat. Protoc. 2013, 8, 1494–1512. [Google Scholar] [CrossRef]
- Manni, M.; Berkeley, M.R.; Seppey, M.; Simão, F.A.; Zdobnov, E.M. BUSCO Update: Novel and Streamlined Workflows along with Broader and Deeper Phylogenetic Coverage for Scoring of Eukaryotic, Prokaryotic, and Viral Genomes. Mol. Biol. Evol. 2021, 38, 4647–4654. [Google Scholar] [CrossRef]
- Buchfink, B.; Xie, C.; Huson, D.H. Fast and Sensitive Protein Alignment Using DIAMOND. Nat. Methods 2015, 12, 59–60. [Google Scholar] [CrossRef] [PubMed]
- Lechner, M.; Findeiß, S.; Steiner, L.; Marz, M.; Stadler, P.F.; Prohaska, S.J. Proteinortho: Detection of (Co-)Orthologs in Large-Scale Analysis. BMC Bioinform. 2011, 12, 124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.; Durbin, R. Fast and Accurate Short Read Alignment with Burrows-Wheeler Transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van der Auwera, G.A.; O’Connor, B.D. Genomics in the Cloud: Using Docker, GATK, and WDL in Terra; O’Reilly Media: Sebastopol, CA, USA, 2020. [Google Scholar]
- Cantalapiedra, C.P.; Hernández-Plaza, A.; Letunic, I.; Bork, P.; Huerta-Cepas, J. EggNOG-Mapper v2: Functional Annotation, Orthology Assignments, and Domain Prediction at the Metagenomic Scale. Mol. Biol. Evol. 2021, 38, 5825–5829. [Google Scholar] [CrossRef]
- Mi, H.; Ebert, D.; Muruganujan, A.; Mills, C.; Albou, L.-P.; Mushayamaha, T.; Thomas, P.D. PANTHER Version 16: A Revised Family Classification, Tree-Based Classification Tool, Enhancer Regions and Extensive API. Nucleic Acids Res. 2021, 49, D394–D403. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT Multiple Sequence Alignment Software Version 7: Improvements in Performance and Usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [Green Version]
- Trifinopoulos, J.; Nguyen, L.-T.; von Haeseler, A.; Minh, B.Q. W-IQ-TREE: A Fast Online Phylogenetic Tool for Maximum Likelihood Analysis. Nucleic Acids Res. 2016, 44, W232–W235. [Google Scholar] [CrossRef] [Green Version]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the Ultrafast Bootstrap Approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef]
- Yang, Z.; Nielsen, R.; Goldman, N.; Pedersen, A.-M.K. Codon-Substitution Models for Heterogeneous Selection Pressure at Amino Acid Sites. Genetics 2000, 155, 431–449. [Google Scholar] [CrossRef]
- Yang, Z. PAML 4: Phylogenetic Analysis by Maximum Likelihood. Mol. Biol. Evol. 2007, 24, 1586–1591. [Google Scholar] [CrossRef] [Green Version]
- Huerta-Cepas, J.; Serra, F.; Bork, P. ETE 3: Reconstruction, Analysis, and Visualization of Phylogenomic Data. Mol. Biol. Evol. 2016, 33, 1635–1638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Wertheim, J.O.; Murrell, B.; Smith, M.D.; Kosakovsky Pond, S.L.; Scheffler, K. RELAX: Detecting Relaxed Selection in a Phylogenetic Framework. Mol. Biol. Evol. 2015, 32, 820–832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lopez, J.V.; Culver, M.; Stephens, J.C.; Johnson, W.E.; O’Brien, S.J. Rates of Nuclear and Cytoplasmic Mitochondrial DNA Sequence Divergence in Mammals. Mol. Biol. Evol. 1997, 14, 277–286. [Google Scholar] [CrossRef] [Green Version]
- Von Kopylow, K.; Kirchhoff, C.; Jezek, D.; Schulze, W.; Feig, C.; Primig, M.; Steinkraus, V.; Spiess, A.-N. Screening for Biomarkers of Spermatogonia within the Human Testis: A Whole Genome Approach. Hum. Reprod. 2010, 25, 1104–1112. [Google Scholar] [CrossRef] [Green Version]
- Pease, J.B.; Driver, R.J.; de la Cerda, D.A.; Day, L.B.; Lindsay, W.R.; Schlinger, B.A.; Schuppe, E.R.; Balakrishnan, C.N.; Fuxjager, M.J. Layered Evolution of Gene Expression in “Superfast” Muscles for Courtship. Proc. Natl. Acad. Sci. USA 2022, 119, e2119671119. [Google Scholar] [CrossRef]
- Elokil, A.A.; Abouzaid, M.; Magdy, M.; Xiao, T.; Liu, H.; Xu, R.; Li, S. Testicular Transcriptome Analysis under the Dietary Inclusion of L-Carnitine Reveals Potential Key Genes Associated with Oxidative Defense and the Semen Quality Factor in Aging Roosters. Domest. Anim. Endocrinol. 2021, 74, 106573. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.-W.; Sun, S.-M.; Xu, K.; Li, Y.-Y.; Lei, W.-L.; Li, L.; Liu, S.-L.; Ouyang, Y.-C.; Sun, Q.-Y.; Wang, Z.-B. FBXO34 Regulates the G2/M Transition and Anaphase Entry in Meiotic Oocytes. Front. Cell Dev. Biol. 2021, 9, 647103. [Google Scholar] [CrossRef] [PubMed]
- Wollenberg Valero, K.C.; Garcia-Porta, J.; Irisarri, I.; Feugere, L.; Bates, A.; Kirchhof, S.; Jovanović Glavaš, O.; Pafilis, P.; Samuel, S.F.; Müller, J.; et al. Functional Genomics of Abiotic Environmental Adaptation in Lacertid Lizards and Other Vertebrates. J. Anim. Ecol. 2021, 91, 1163–1179. [Google Scholar] [CrossRef]
- Wu, D.-D.; Yang, C.-P.; Wang, M.-S.; Dong, K.-Z.; Yan, D.-W.; Hao, Z.-Q.; Fan, S.-Q.; Chu, S.-Z.; Shen, Q.-S.; Jiang, L.-P.; et al. Convergent Genomic Signatures of High-Altitude Adaptation among Domestic Mammals. Natl. Sci. Rev. 2020, 7, 952–963. [Google Scholar] [CrossRef] [Green Version]
- Yasumoto, H.; Meng, L.; Lin, T.; Zhu, Q.; Tsai, R.Y.L. GNL3L Inhibits Activity of Estrogen-Related Receptor γ by Competing for Coactivator Binding. J. Cell Sci. 2007, 120, 2532–2543. [Google Scholar] [CrossRef] [Green Version]
- Gewiss, R.; Topping, T.; Griswold, M.D. Cycles, Waves, and Pulses: Retinoic Acid and the Organization of Spermatogenesis. Andrology 2020, 8, 892–897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grindflek, E.; Berget, I.; Moe, M.; Oeth, P.; Lien, S. Transcript Profiling of Candidate Genes in Testis of Pigs Exhibiting Large Differences in Androstenone Levels. BMC Genet. 2010, 11, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Yang, Y.; Li, W.; Zhang, Y.; Yang, Y.; Li, H.; Geng, Z.; Ao, H.; Zhou, R.; Li, K. NRDR Inhibits Estradiol Synthesis and Is Associated with Changes in Reproductive Traits in Pigs: LIU et al. Mol. Reprod. Dev. 2019, 86, 63–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsunaga, T.; Endo, S.; Maeda, S.; Ishikura, S.; Tajima, K.; Tanaka, N.; Nakamura, K.T.; Imamura, Y.; Hara, A. Characterization of Human DHRS4: An Inducible Short-Chain Dehydrogenase/Reductase Enzyme with 3β-Hydroxysteroid Dehydrogenase Activity. Arch. Biochem. Biophys. 2008, 477, 339–347. [Google Scholar] [CrossRef]
- Ran, X.; Hu, F.; Mao, N.; Ruan, Y.; Yi, F.; Niu, X.; Huang, S.; Li, S.; You, L.; Zhang, F.; et al. Differences in Gene Expression and Variable Splicing Events of Ovaries between Large and Small Litter Size in Chinese Xiang Pigs. Porc. Health Manag. 2021, 7, 52. [Google Scholar] [CrossRef]
- Chi, W.; Reinke, V. Promotion of Oogenesis and Embryogenesis in the C. Elegans Gonad by EFL-1/DPL-1 (E2F) Does Not Require LIN-35 (PRB). Development 2006, 133, 3147–3157. [Google Scholar] [CrossRef] [Green Version]
- Paczkowski, M.; Schoolcraft, W.B.; Krisher, R.L. Fatty Acid Metabolism during Maturation Affects Glucose Uptake and Is Essential to Oocyte Competence. Reproduction 2014, 148, 429–439. [Google Scholar] [CrossRef] [Green Version]
- Sharma, M. Mitochondrial Fatty Acid Transport System and Its Relevance to Ovarian Function. J. Infertil. Reprod. Biol. 2018, 6, 1–3. [Google Scholar] [CrossRef]
- Gentile, L.; Monti, M.; Sebastiano, V.; Merico, V.; Nicolai, R.; Calvani, M.; Garagna, S.; Redi, C.A.; Zuccotti, M. Single-Cell Quantitative RT-PCR Analysis of Cpt1b and Cpt2 Gene Expression in Mouse Antral Oocytes and in Preimplantation Embryos. Cytogenet. Genome Res. 2004, 105, 215–221. [Google Scholar] [CrossRef]
- Nenicu, A.; Lüers, G.H.; Kovacs, W.; Bergmann, M.; Baumgart-Vogt, E. Peroxisomes in Human and Mouse Testis: Differential Expression of Peroxisomal Proteins in Germ Cells and Distinct Somatic Cell Types of the Testis. Biol. Reprod. 2007, 77, 1060–1072. [Google Scholar] [CrossRef] [Green Version]
- Hussain, S.; Tuorto, F.; Menon, S.; Blanco, S.; Cox, C.; Flores, J.V.; Watt, S.; Kudo, N.R.; Lyko, F.; Frye, M. The Mouse Cytosine-5 RNA Methyltransferase NSun2 Is a Component of the Chromatoid Body and Required for Testis Differentiation. Mol. Cell. Biol. 2013, 33, 1561–1570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fu, C.; Rojas, T.; Chin, A.C.; Cheng, W.; Bernstein, I.A.; Albacarys, L.K.; Wright, W.W.; Snyder, S.H. Multiple Aspects of Male Germ Cell Development and Interactions with Sertoli Cells Require Inositol Hexakisphosphate Kinase-1. Sci. Rep. 2018, 8, 7039. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meneses, M.J.; Borges, D.O.; Dias, T.R.; Martins, F.O.; Oliveira, P.F.; Macedo, M.P.; Alves, M.G. Knockout of Insulin-Degrading Enzyme Leads to Mice Testicular Morphological Changes and Impaired Sperm Quality. Mol. Cell. Endocrinol. 2019, 486, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Hekim, N.; Gunes, S.; Asci, R.; Henkel, R.; Abur, U. Semiquantitative Promoter Methylation of MLH1 and MSH2 Genes and Their Impact on Sperm DNA Fragmentation and Chromatin Condensation in Infertile Men. Andrologia 2021, 53, e13827. [Google Scholar] [CrossRef]
- Venditti, M.; Donizetti, A.; Aniello, F.; Minucci, S. EH Domain Binding Protein 1-like 1 (EHBP1L1), a Protein with Calponin Homology Domain, Is Expressed in the Rat Testis. Zygote 2020, 28, 441–446. [Google Scholar] [CrossRef]
- Venditti, M.; Minucci, S. Differential Expression and Localization of EHBP1L1 during the First Wave of Rat Spermatogenesis Suggest Its Involvement in Acrosome Biogenesis. Biomedicines 2022, 10, 181. [Google Scholar] [CrossRef]
- El-Raey, M.; Geshi, M.; Somfai, T.; Kaneda, M.; Hirako, M.; Abdel-Ghaffar, A.E.; Sosa, G.A.; El-Roos, M.E.A.A.; Nagai, T. Evidence of Melatonin Synthesis in the Cumulus Oocyte Complexes and Its Role in Enhancing Oocyte Maturation In Vitro in Cattle. Mol. Reprod. Dev. 2011, 78, 250–262. [Google Scholar] [CrossRef]
- Sakaguchi, K.; Itoh, M.T.; Takahashi, N.; Tarumi, W.; Ishizuka, B. The Rat Oocyte Synthesises Melatonin. Reprod. Fertil. Dev. 2013, 25, 674. [Google Scholar] [CrossRef]
- Xue, Q.; Li, G.; Cao, Y.; Yin, J.; Zhu, Y.; Zhang, H.; Zhou, C.; Shen, H.; Dou, X.; Su, Y.; et al. Identification of Genes Involved in Inbreeding Depression of Reproduction in Langshan Chickens. Anim. Biosci. 2021, 34, 975–984. [Google Scholar] [CrossRef]
- Huang, D.; Li, J.; He, L.-Q. Influence of Tripterygium Wilfordii on the Expression of Spermiogenesis Related Genes Herc4, Ipo11 and Mrto4 in Mice. Hered. Beijing 2009, 31, 941–946. [Google Scholar] [CrossRef]
- Xiang, W.; Wen, Z.; Hu, L.; Li, H.; Xiong, C. Expression of NOSTRIN in the testis tissue of azoospermia patients. Zhonghua Nan Ke Xue Natl. J. Androl. 2011, 17, 38–42. [Google Scholar]
- Bansal, S.K.; Gupta, N.; Sankhwar, S.N.; Rajender, S. Differential Genes Expression between Fertile and Infertile Spermatozoa Revealed by Transcriptome Analysis. PLoS ONE 2015, 10, e0127007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doran, J.; Walters, C.; Kyle, V.; Wooding, P.; Hammett-Burke, R.; Colledge, W.H. Mfsd14a (Hiat1) Gene Disruption Causes Globozoospermia and Infertility in Male Mice. Reproduction 2016, 152, 91–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, R.K.-K.; Tseng, H.-C.; Hwu, Y.-M.; Fan, C.-C.; Lin, M.-H.; Yu, J.-J.; Yeh, L.-Y.; Li, S.-H. Expression of Cystatin C in the Female Reproductive Tract and Its Effect on Human Sperm Capacitation. Reprod. Biol. Endocrinol. 2018, 16, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Major, A.T.; Hogarth, C.A.; Young, J.C.; Kurihara, Y.; Jans, D.A.; Loveland, K.L. Dynamic Paraspeckle Component Localisation during Spermatogenesis. Reproduction 2019, 158, 267–280. [Google Scholar] [CrossRef]
- Gispert, S.; Key, J.; Kohli, A.; Torres-Odio, S.; Koepf, G.; Amr, S.; Reichlmeir, M.; Harter, P.N.; West, A.P.; Münch, C.; et al. ClpP-Deletion Causes Azoospermia, with Meiosis-I Delay and Insufficient Biosynthesis of Spermatid Factors, Due to Mitochondrial Dysfunction with Accumulation of Perrault Proteins ERAL1, PEO1, and HARS2 2022. Preprints 2022, 2022040245. [Google Scholar] [CrossRef]
- Grant, K.E. Molecular and Cellular Correlates of Sperm Viability Associated with Male Fertility. Theses Diss 2013, 3178. Available online: https://scholarsjunction.msstate.edu/td/3178 (accessed on 12 May 2022).
- Feugang, J.M.; Kaya, A.; Page, G.P.; Chen, L.; Mehta, T.; Hirani, K.; Nazareth, L.; Topper, E.; Gibbs, R.; Memili, E. Two-Stage Genome-Wide Association Study Identifies Integrin Beta 5 as Having Potential Role in Bull Fertility. BMC Genom. 2009, 10, 176. [Google Scholar] [CrossRef] [Green Version]
- Velho, A.; Wang, H.; Koenig, L.; Grant, K.E.; Menezes, E.S.; Kaya, A.; Moura, A.; Memili, E. Expression Dynamics of Integrin Subunit Beta 5 in Bovine Gametes and Embryos Imply Functions in Male Fertility and Early Embryonic Development. Andrologia 2019, 51, e13305. [Google Scholar] [CrossRef]
- Yamaguchi, Y.L.; Tanaka, S.S.; Yasuda, K.; Matsui, Y.; Tam, P.P.L. Stage-Specific Importin13 Activity Influences Meiosis of Germ Cells in the Mouse. Dev. Biol. 2006, 297, 350–360. [Google Scholar] [CrossRef] [Green Version]
- Arraztoa, J.A.; Zhou, J.; Marcu, D.; Cheng, C.; Bonner, R.; Chen, M.; Xiang, C.; Brownstein, M.; Maisey, K.; Imarai, M.; et al. Identification of Genes Expressed in Primate Primordial Oocytes. Hum. Reprod. 2005, 20, 476–483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.; Cui, P.; Sun, Q.; Du, Z.; Chen, Z.; Li, Z.; Liu, C.; Cao, Y.; Yang, Z.; Liu, R.; et al. PSPC1 Regulates CHK1 Phosphorylation through Phase Separation and Participates in Mouse Oocyte Maturation. Acta Biochim. Biophys. Sin. 2021, 53, 1527–1537. [Google Scholar] [CrossRef]
- Moura, L.B.S.; Magalhães-Padilha, D.M.; Morais, A.N.P.; Aguiar, F.L.N.; Geisler–Lee, J.; Wischral, A.; Gastal, M.O.; Fonseca, G.R.; Geisler, M.; Figueiredo, J.R. Folliculogenesis-Related Genes Are Differently Expressed in Secondary and Tertiary Ovarian Follicles. Zygote 2021, 29, 503–506. [Google Scholar] [CrossRef]
- Jiborn, T.; Abrahamson, M.; Wallin, H.; Malm, J.; Lundwall, Å.; Gadaleanu, V.; Abrahamsson, P.-A.; Bjartell, A. Cystatin C Is Highly Expressed in the Human Male Reproductive System. J. Androl. 2013, 25, 564–572. [Google Scholar] [CrossRef]
- Chakraborty, S.; Ain, R. NOSTRIN: A Novel Modulator of Trophoblast Giant Cell Differentiation. Stem Cell Res. 2018, 31, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Parkkila, S.; Parkkila, A.K.; Kaunisto, K.; Waheed, A.; Sly, W.S.; Rajaniemi, H. Location of a Membrane-Bound Carbonic Anhydrase Isoenzyme (CA IV) in the Human Male Reproductive Tract. J. Histochem. Cytochem. 1993, 41, 751–757. [Google Scholar] [CrossRef] [Green Version]
- Hynninen, P.; Hämäläinen, J.M.; Pastorekova, S.; Pastorek, J.; Waheed, A.; Sly, W.S.; Tomas, E.; Kirkinen, P.; Parkkila, S. Transmembrane Carbonic Anhydrase Isozymes IX and XII in the Female Mouse Reproductive Organs. Reprod. Biol. Endocrinol. 2004, 2, 73. [Google Scholar] [CrossRef] [Green Version]
- Rengaraj, D.; Gao, F.; Liang, X.-H.; Yang, Z.-M. Expression and Regulation of Type II Integral Membrane Protein Family Members in Mouse Male Reproductive Tissues. Endocrine 2007, 31, 193–201. [Google Scholar] [CrossRef] [Green Version]
- Rengaraj, D.; Liang, X.-H.; Gao, F.; Deng, W.-B.; Mills, N.; Yang, Z.-M. Differential Expression and Regulation of Integral Membrane Protein 2b in Rat Male Reproductive Tissues. Asian J. Androl. 2008, 10, 503–511. [Google Scholar] [CrossRef] [Green Version]
- Rodriguez, T.A.; Sparrow, D.B.; Scott, A.N.; Withington, S.L.; Preis, J.I.; Michalicek, J.; Clements, M.; Tsang, T.E.; Shioda, T.; Beddington, R.S.P.; et al. Cited1 Is Required in Trophoblasts for Placental Development and for Embryo Growth and Survival. Mol. Cell. Biol. 2004, 24, 228–244. [Google Scholar] [CrossRef] [Green Version]
- Sha, Q.; Yu, J.; Guo, J.; Dai, X.; Jiang, J.; Zhang, Y.; Yu, C.; Ji, S.; Jiang, Y.; Zhang, S.; et al. CNOT 6L Couples the Selective Degradation of Maternal Transcripts to Meiotic Cell Cycle Progression in Mouse Oocyte. EMBO J. 2018, 37, e99333. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Silva, G.; Caballero-Campo, P.; Chirinos, M. Sperm MRNAs as Potential Markers of Male Fertility. Reprod. Biol. 2022, 22, 100636. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y. The Ancestral Gene for Transcribed, Low-Copy Repeats in the Prader-Willi/Angelman Region Encodes a Large Protein Implicated in Protein Trafficking, Which Is Deficient in Mice with Neuromuscular and Spermiogenic Abnormalities. Hum. Mol. Genet. 1999, 8, 533–542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dumasia, K.; Kumar, A.; Deshpande, S.; Balasinor, N.H. Estrogen, through Estrogen Receptor 1, Regulates Histone Modifications and Chromatin Remodeling during Spermatogenesis in Adult Rats. Epigenetics 2017, 12, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Davila, R.A.; Spiller, C.; Harkins, D.; Harvey, T.; Jordan, P.W.; Gronostajski, R.M.; Piper, M.; Bowles, J. Deletion of NFIX Results in Defective Progression through Meiosis within the Mouse Testis. Biol. Reprod. 2022, 106, ioac049. [Google Scholar] [CrossRef]
- Linnemannstöns, K.; Ripp, C.; Honemann-Capito, M.; Brechtel-Curth, K.; Hedderich, M.; Wodarz, A. The PTK7-Related Transmembrane Proteins Off-Track and Off-Track 2 Are Co-Receptors for Drosophila Wnt2 Required for Male Fertility. PLoS Genet. 2014, 10, e1004443. [Google Scholar] [CrossRef] [Green Version]
- Moon, S.; Lee, O.-H.; Lee, S.; Lee, J.; Park, H.; Park, M.; Chang, E.M.; Park, K.-H.; Choi, Y. STK3/4 Expression Is Regulated in Uterine Endometrial Cells during the Estrous Cycle. Cells 2019, 8, 1643. [Google Scholar] [CrossRef] [Green Version]
- Gura, M.A.; Mikedis, M.M.; Seymour, K.A.; de Rooij, D.G.; Page, D.C.; Freiman, R.N. Dynamic and Regulated TAF Gene Expression during Mouse Embryonic Germ Cell Development. PLoS Genet. 2020, 16, e1008515. [Google Scholar] [CrossRef]
- Obbard, D.J.; Welch, J.J.; Kim, K.-W.; Jiggins, F.M. Quantifying Adaptive Evolution in the Drosophila Immune System. PLoS Genet. 2009, 5, e1000698. [Google Scholar] [CrossRef] [Green Version]
- McTaggart, S.J.; Obbard, D.J.; Conlon, C.; Little, T.J. Immune Genes Undergo More Adaptive Evolution than Non-Immune System Genes in Daphnia Pulex. BMC Evol. Biol. 2012, 12, 63. [Google Scholar] [CrossRef] [Green Version]
- Shultz, A.J.; Sackton, T.B. Immune Genes Are Hotspots of Shared Positive Selection across Birds and Mammals. eLife 2019, 8, e41815. [Google Scholar] [CrossRef] [PubMed]
- Paterson, H.E.H. The Recognition Concept of Species. In Species and Speciation; Vrba, E., Ed.; Transvaal Museum: Pretoria, South Africa, 1985; pp. 21–29. [Google Scholar]
- Petrova, T.V. Narrow-Headed Vole Lasiopodomys (Stenocranius) Gregalis (Pallas, 1779): Taxonomic Structure, Phylogenetic Position and Evolution; Zoological Institute RAS: Saint Petersburg, Russia, 2017. [Google Scholar]
- Wallace, A.R. Darwinism. An Exposition of the Theory of Natural Selection with Some of Its Applications; Macmillan & Co.: London, UK; New York, NY, USA, 1889. [Google Scholar]
- Feoktistova, N.Y.; Kropotkina, M.V.; Potashnikova, E.V.; Gureeva, A.V.; Kuznetsova, E.V.; Surov, A.V. Speciation in Allopatric Species of the Hamster Subfamily Cricetinae (Rodentia, Cricetidae). Biol. Bull. Rev. 2019, 9, 230–242. [Google Scholar] [CrossRef]

| Enriched GO-Terms | Genes Involved |
|---|---|
| interspecies interaction between organisms (GO:0044419) | UBB, NUP43, VPS4B, ARF1 |
| defense response, defense response to other organism (GO:0006952, GO:0098542) | VAMP7, DDX41, EXOSC2, FADD, CITED1, SSC5D, CD40, COLEC12 |
| response to biotic stimulus, external biotic stimulus, and to other organism (GO:0009607, GO:0043207, GO:0051707) | TPT1, DDX41, EXOSC2, FADD, CITED1, GSTA2, SSC5D, CD40, ARF1, ATG10 |
| regulation of response to stimulus, positive regulation of response to stimulus (GO:0048583, GO:0048584) | PIAS1, YAF2, KMT2D, STK3, TPT1, SCUBE2, UBQLN2, RNF43, VAMP7, PDGFC, ZFAND6, MYLK, USP12, SKI, NUDT6, FADD, TNNT1, PTK7, RB1, SRI, SLC35C2, RASGEF1B, TAF9B, EIF4A3, MAP3K1, MYH7, CD40, TJP2, COLEC12, UBA2 |
| response to external stimulus (GO:0009605) | TPT1, RBP1, POU6F1, MYBBP1A, DDX41, EXOSC2, EXOC6, FADD, CITED1, PTK7, ATG14, NRP2, MAP3K1, SSC5D, SIK3, CD40, PLXNA4, TTC8, ARF1 |
| response to stress (GO:0006950) | PIAS1, AATF, STK3, TPT1, ISY1, UBQLN2, HPS6, UBA6, VAMP7, GNAO1, PDGFC, FAM129A, CNOT6L, MYLK, HK2, MYBBP1A, ND1, ZRANB3, DDX41, PITHD1, POLR2C, EXOSC2, FADD, CITED1, PTK7, ATG14, RCSD1, EHD1, HERC2, MAP3K1, SSC5D, MYH7, SIK3, CD40, TJP2, ALDOC, COLEC12, ATG10 |
| sensory perception of chemical stimulus and smell (GO:0007600, GO:0007606, GO:0007608) | ITGB2, SLC24A3, POU6F1, TTC8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrova, T.; Skazina, M.; Kuksin, A.; Bondareva, O.; Abramson, N. Narrow-Headed Voles Species Complex (Cricetidae, Rodentia): Evidence for Species Differentiation Inferred from Transcriptome Data. Diversity 2022, 14, 512. https://doi.org/10.3390/d14070512
Petrova T, Skazina M, Kuksin A, Bondareva O, Abramson N. Narrow-Headed Voles Species Complex (Cricetidae, Rodentia): Evidence for Species Differentiation Inferred from Transcriptome Data. Diversity. 2022; 14(7):512. https://doi.org/10.3390/d14070512
Chicago/Turabian StylePetrova, Tatyana, Maria Skazina, Aleksander Kuksin, Olga Bondareva, and Natalia Abramson. 2022. "Narrow-Headed Voles Species Complex (Cricetidae, Rodentia): Evidence for Species Differentiation Inferred from Transcriptome Data" Diversity 14, no. 7: 512. https://doi.org/10.3390/d14070512







