Hot Is Rich—An Enormous Diversity of Simple Trichal Cyanobacteria from Yellowstone Hot Springs
Abstract
:1. Introduction
2. Materials and Methods
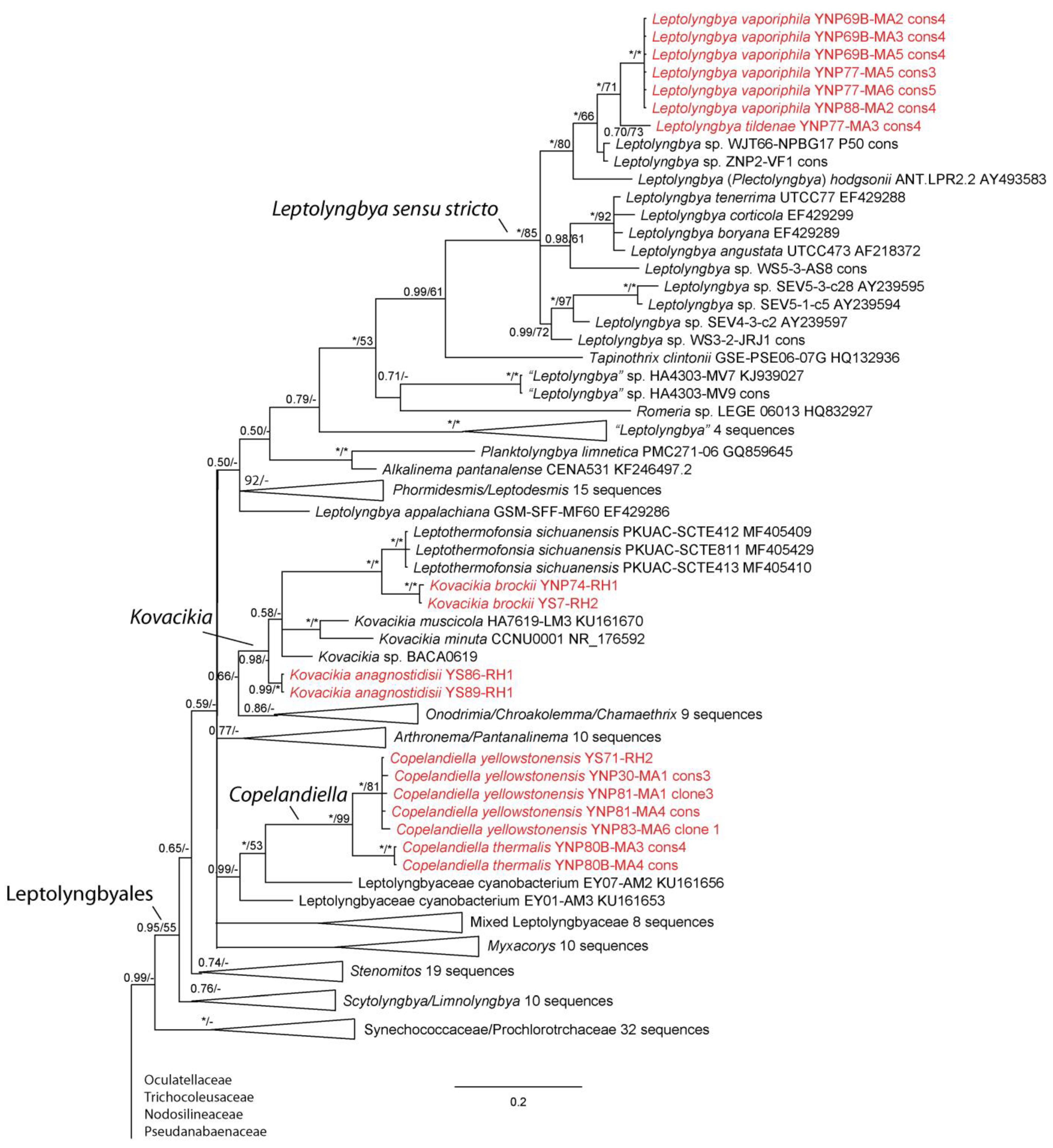
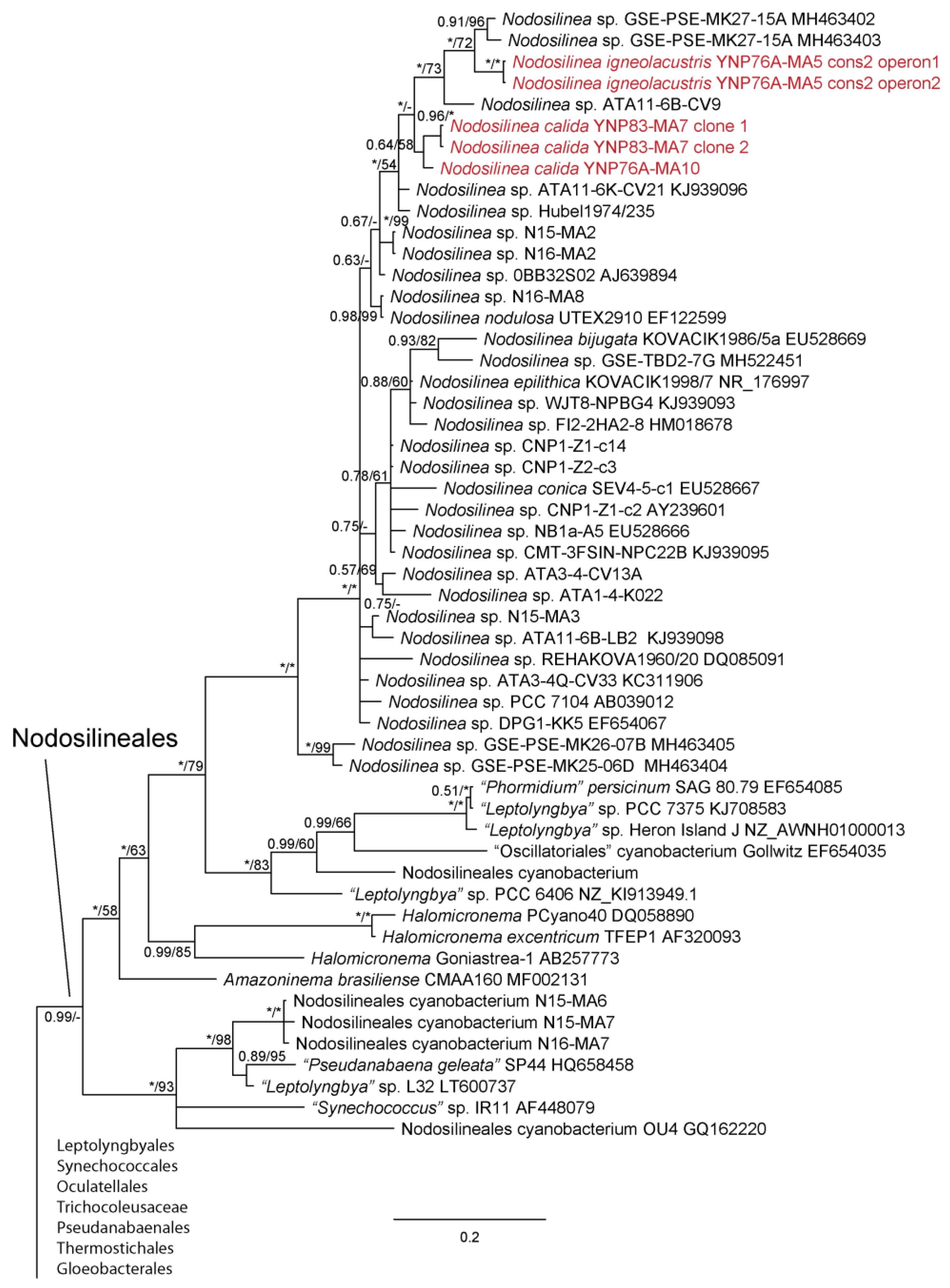
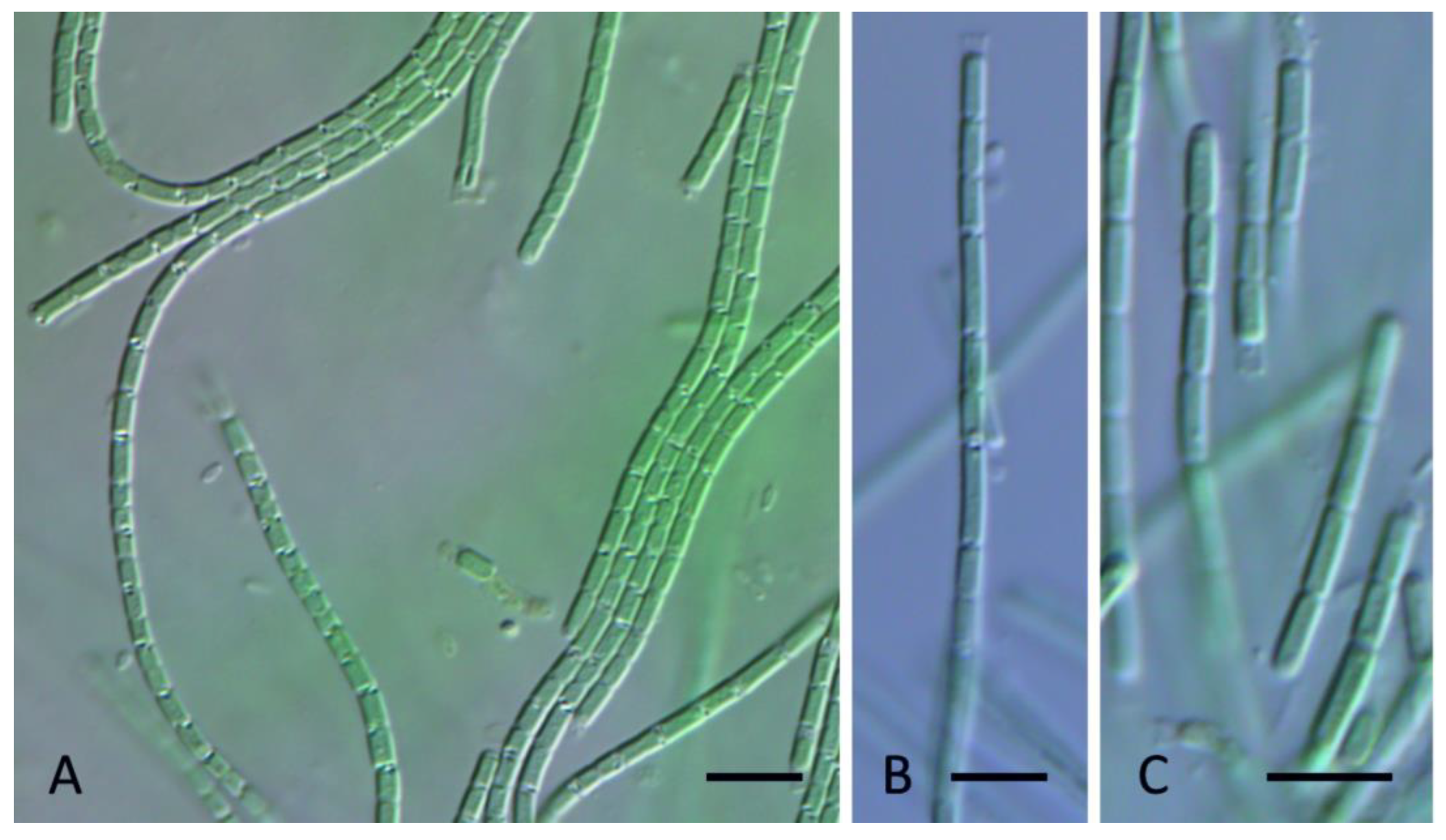
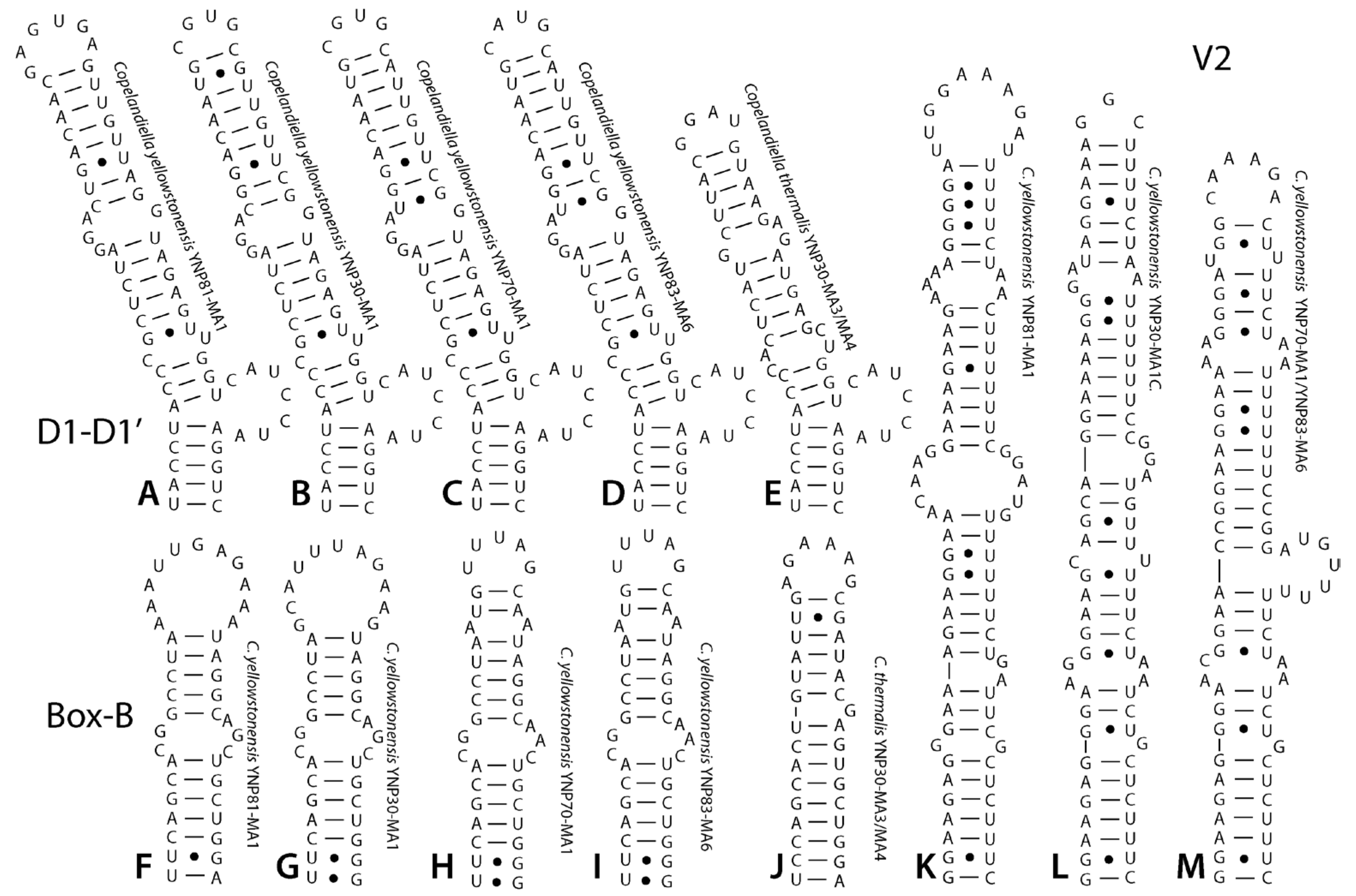
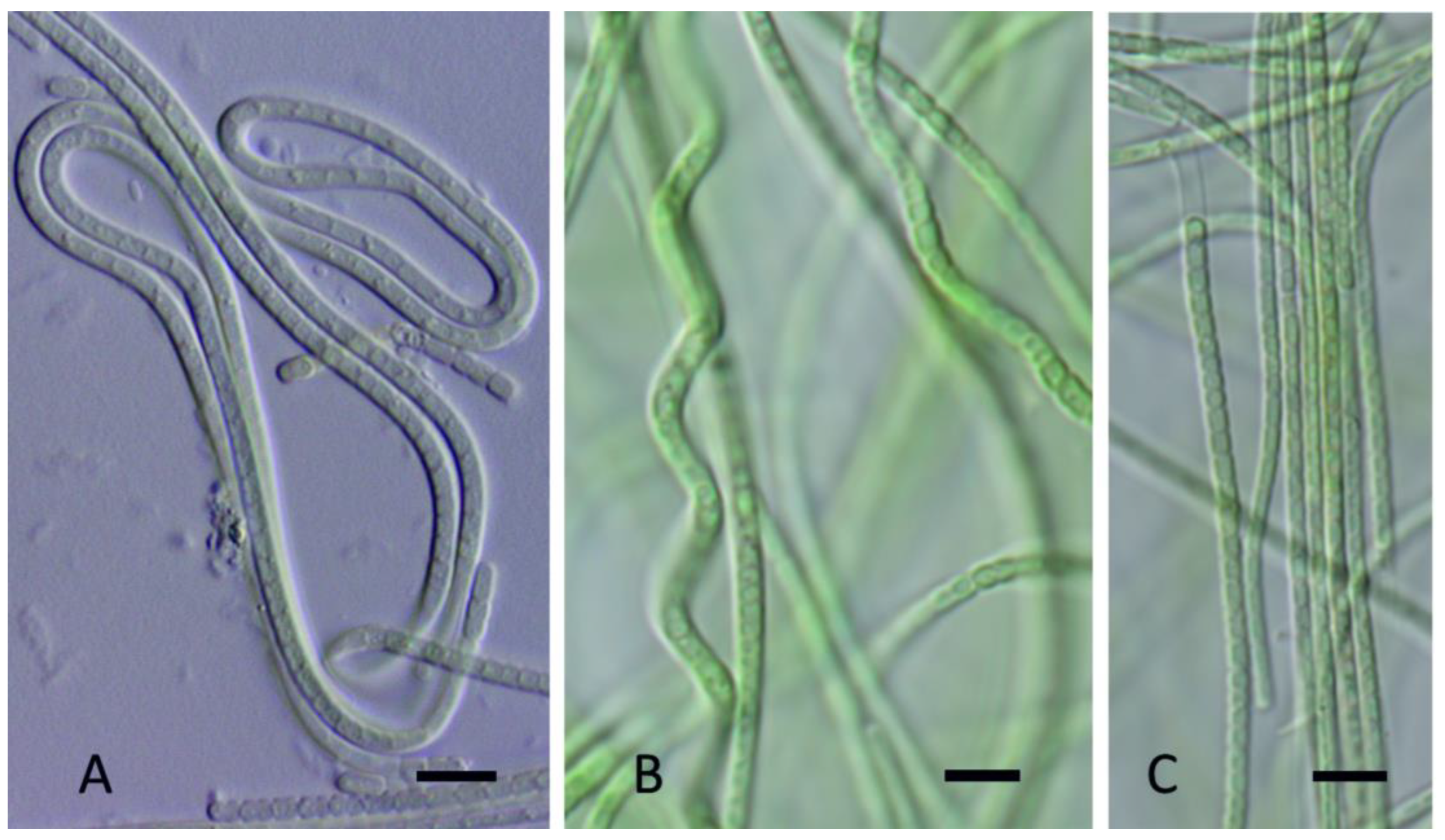
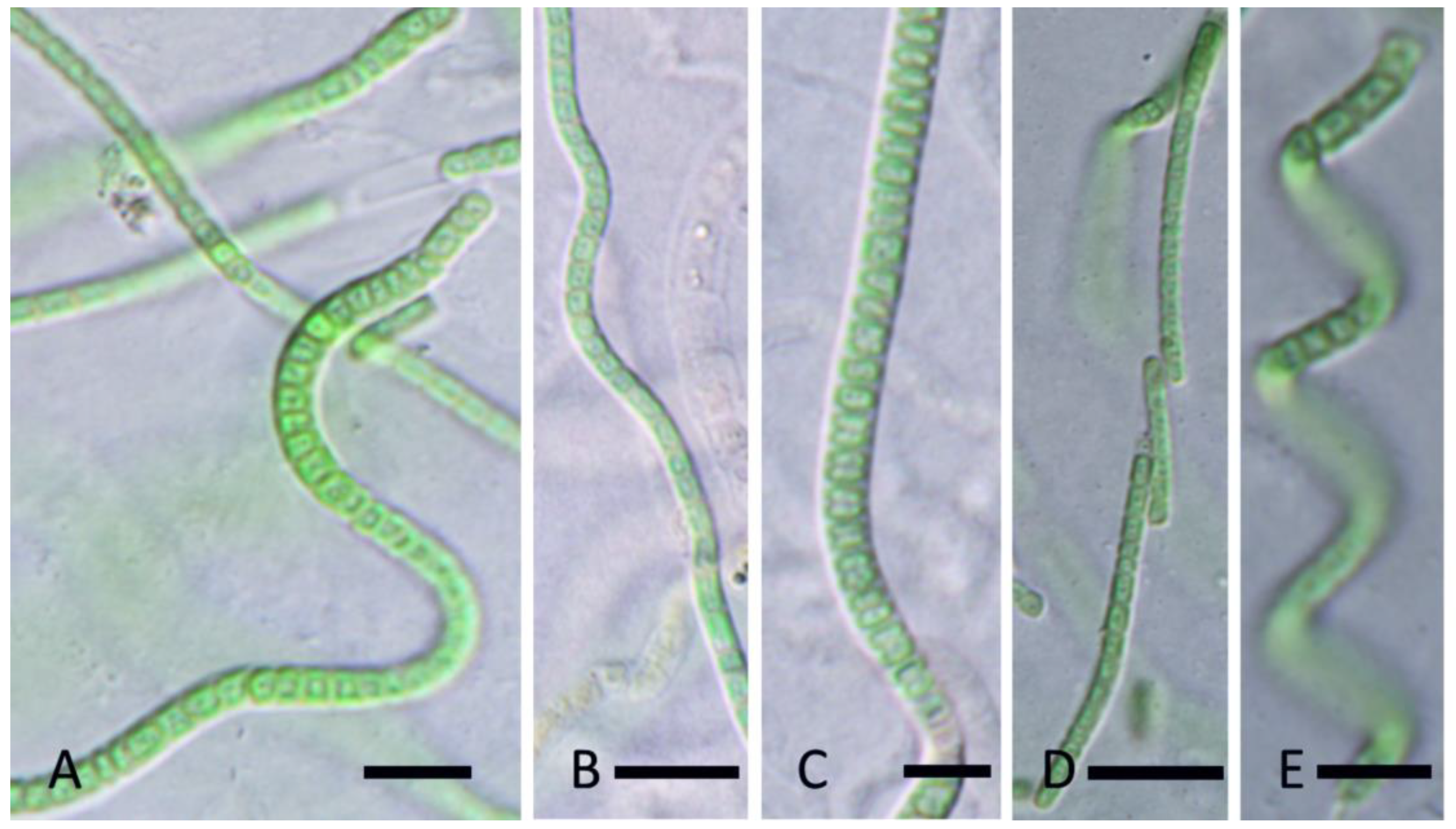
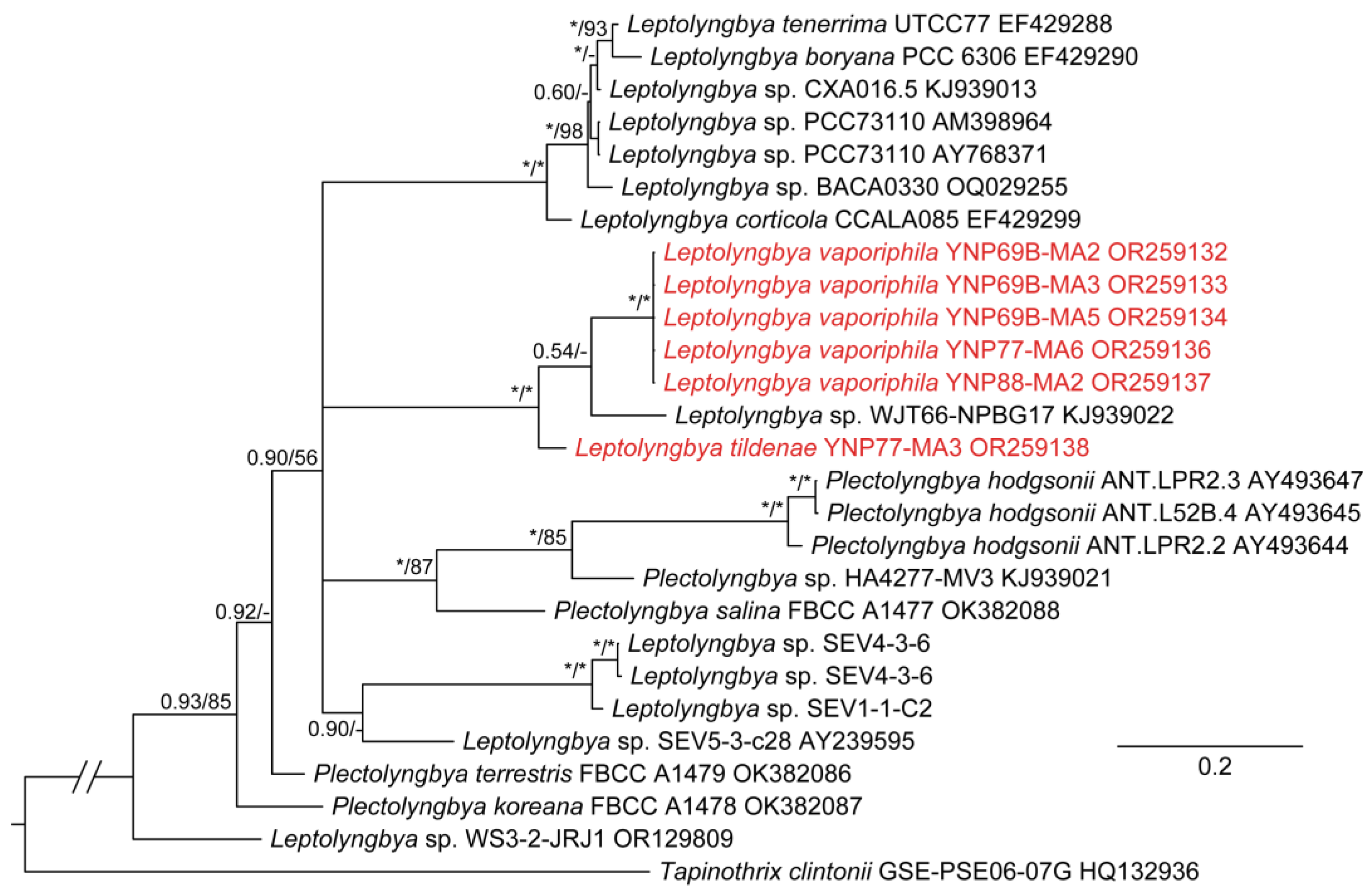
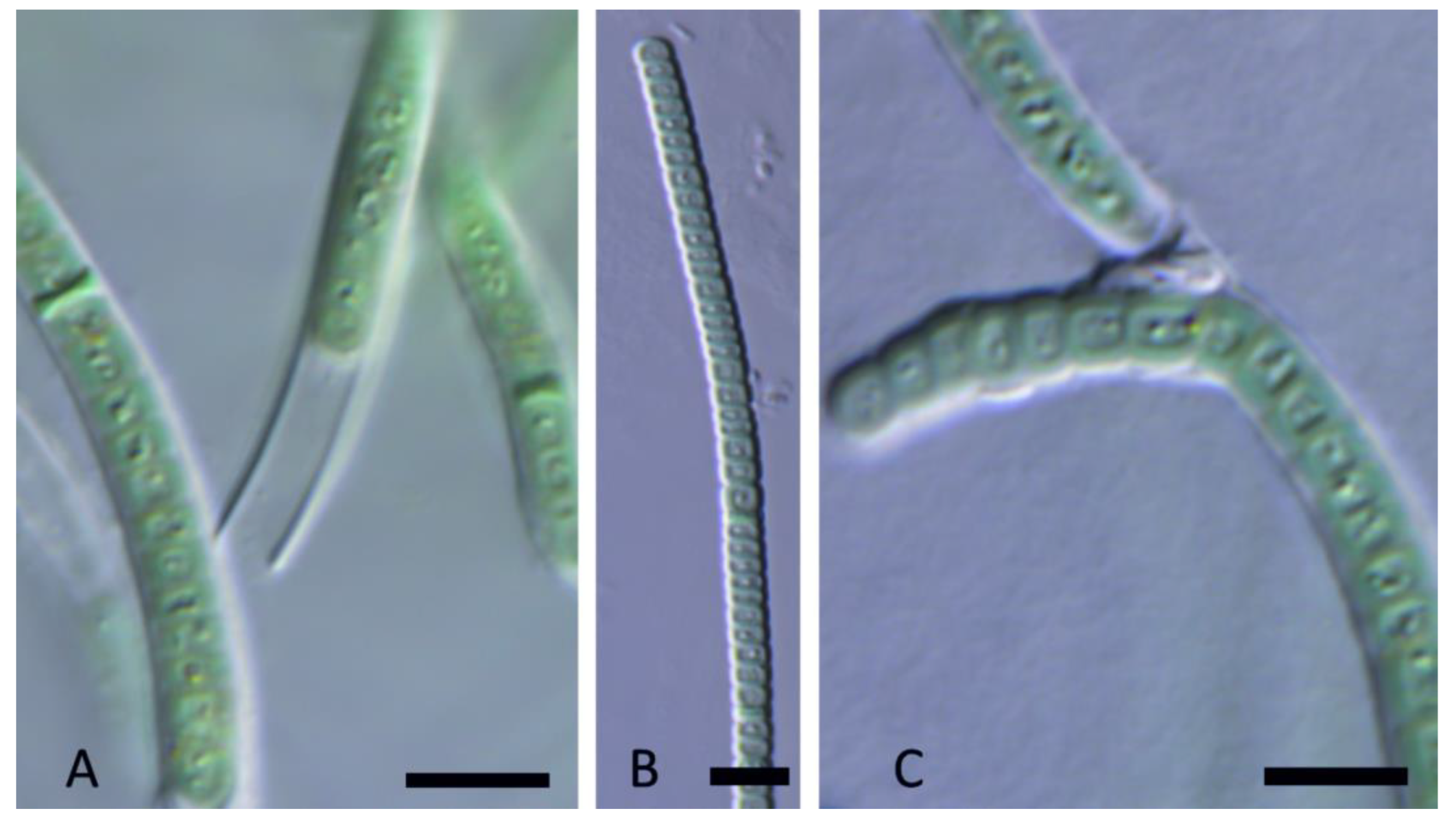
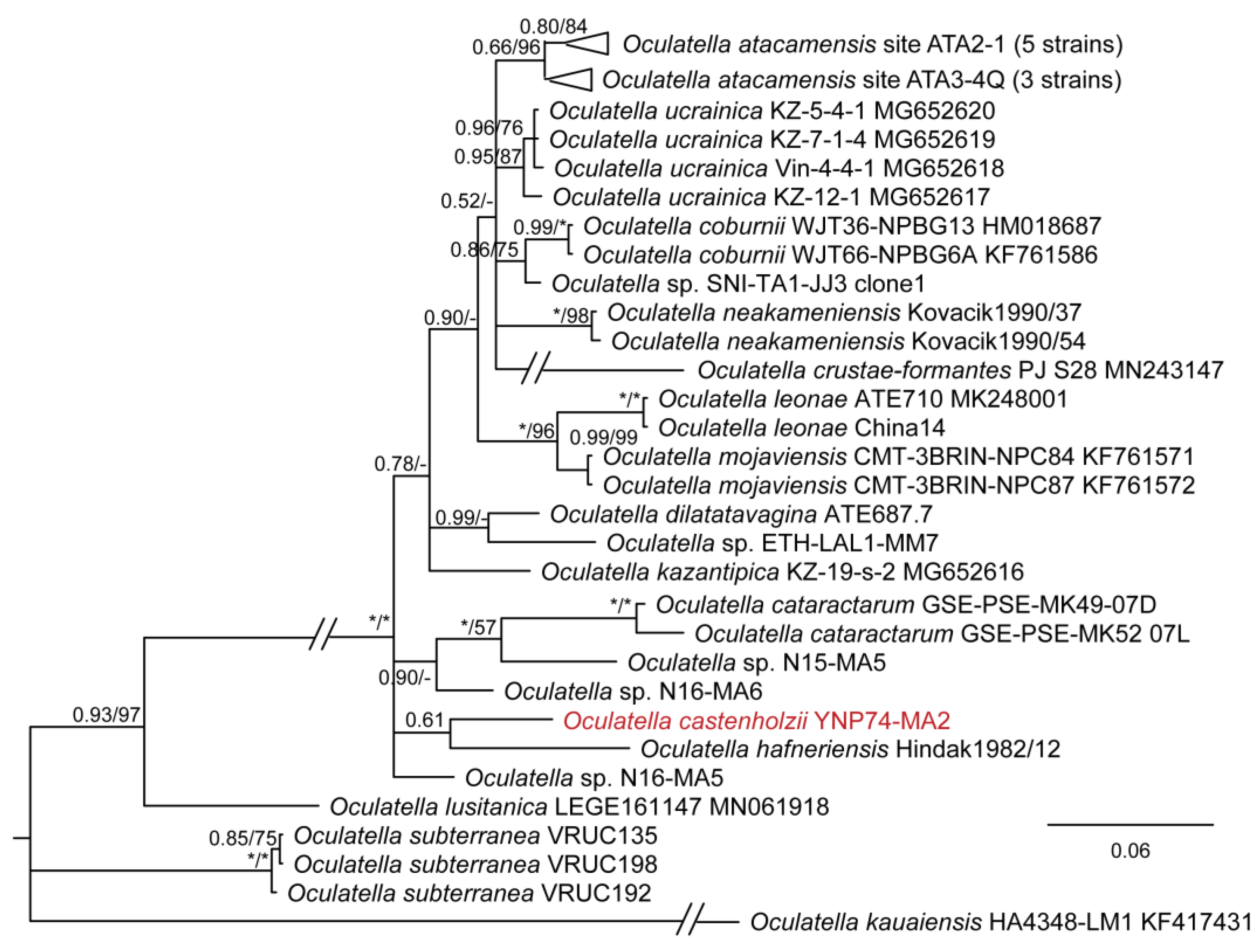
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cohn, F. Über die Algen des Karlsbader Sprudels, mit Rücksicht auf die Bildung des Sprudelsinsters. Abh. Schlesis Gesellsch Vaterländ Kult. 1862, 5, 37–55. [Google Scholar]
- Copeland, J.J. Yellowstone thermal Myxophyceae. Ann. N. Y. Acad. Sci. 1936, 36, 1–232. [Google Scholar] [CrossRef]
- Dyer, D.L.; Gafford, R.D. Some characteristics of a thermophilic blue-green alga. Science 1961, 134, 616–617. [Google Scholar] [CrossRef] [PubMed]
- Brock, T.D. Life at high temperatures. Science 1967, 158, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- Castenholz, R.W. Thermophilic blue-green algae and the thermal environment. Bacteriol. Rev. 1969, 33, 476–504. [Google Scholar] [CrossRef]
- Mullis, K.B.; Ferré, F.; Gibbs, R.A. The Polymerase Chain Reaction; Birkhäuser: Boston, MA, USA, 1994; p. 458. [Google Scholar]
- Brock, T.D.; Freeze, H. Thermus aquaticus gen. n. and sp. n., a nonsporulating extreme thermophile. J. Bacteriol. 1969, 98, 289–297. [Google Scholar] [CrossRef]
- Bunbury, F.; Rivas, C.; Calatrava, V.; Shelton, A.N.; Grossman, A.; Bhayaa, D. Differential Phototactic Behavior of Closely Related Cyanobacterial Isolates from Yellowstone Hot Spring Biofilms. Appl. Environ. Microbiol. 2022, 88, e0019622. [Google Scholar] [CrossRef]
- Momper, L.; Hu, E.; Moore, K.R.; Skoog, E.J.; Tyler, M.; Evans, A.J.; Bosak, T. Metabolic versatility in a modern lineage of cyanobacteria from terrestrial hot springs. Free Radic. Biol. Med. 2019, 140, 224–232. [Google Scholar] [CrossRef]
- Becraft, E.D.; Cohan, F.M.; Kühl, M.; Jensen, S.I.; Ward, D.M. Fine-Scale Distribution Patterns of Synechococcus Ecological Diversity in Microbial Mats of Mushroom Spring, Yellowstone National Park. Appl. Environ. Microbiol. 2011, 77, 7689–7697. [Google Scholar] [CrossRef]
- Bennett, A.C.; Murugapiran, S.K.; Hamilton, T.L. Temperature impacts community structure and function of phototrophic Chloroflexi and Cyanobacteria in two alkaline hot springs in Yellowstone National Park. Environ. Microbiol. Rep. 2020, 12, 503–513. [Google Scholar] [CrossRef]
- Bosak, T.; Liang, B.; Wu, T.-D.; Templer, S.P.; Evans, A.; Vali, H.; Guerquin-Kern, J.-L.; Klepac-Ceraj, V.; Sim, M.S.; Mui, J. Cyanobacterial diversity and activity in modern conical microbialites. Geobiology 2012, 10, 384–401. [Google Scholar] [CrossRef] [PubMed]
- Fecteau, K.M.; Boyd, E.S.; Lindsay, M.R.; Amenabar, M.J.; Robinson, K.J.; Debes, R.V., II; Shock, E.L. Cyanobacteria and algae meet at the limits of their habitat ranges in moderately acidic hot springs. J. Geophysic. Res. Biogeosci. 2022, 127, e2021JG006446. [Google Scholar] [CrossRef]
- Kees, E.D.; Murugapiran, S.K.; Bennett, A.C.; Hamilton, T.L. Distribution and Genomic Variation of Thermophilic Cyanobacteria in Diverse Microbial Mats at the Upper Temperature Limits of Photosynthesis. Environ. Microbiol. 2022, 7, e00317-22. [Google Scholar] [CrossRef] [PubMed]
- Becraft, E.D.; Wood, J.M.; Cohan, F.M.; Ward, D.M. Biogeography of American Northwest Hot Spring A/B′-Lineage Synechococcus Populations. Front. Microbiol. 2020, 11, 77. [Google Scholar] [CrossRef]
- Hamilton, T.L.; Haviga, J. Meet Me in the Middle: Median Temperatures Impact Cyanobacteria and Photoautotrophy in Eruptive Yellowstone Hot Springs. Environ. Microbiol. 2022, 7, e01450-21. [Google Scholar] [CrossRef] [PubMed]
- Reyes, K.; Gonzalez, N.I., III; Stewart, J.; Ospino, F.; Nguyen, D.; Cho, D.T.; Ghahremani, N.; Spear, J.R.; Johnson, H.A. Surface orientation affects the direction of cone growth by Leptolyngbya sp. strain C1, a likely architect of coniform structures Octopus Spring (Yellowstone National Park). Appl. Environ. Microbiol. 2013, 79, 1302–1308. [Google Scholar] [CrossRef]
- Fournier, R.O. Geochemistry and dynamics of the Yellowstone National Park hydrothermal system. Ann. Rev. Earth Planet. Sci. 1989, 17, 13–53. [Google Scholar] [CrossRef]
- Carmichael, W.W. Isolation, culture, and toxicity testing of toxic freshwater cyanobacteria (blue-green algae). In Fundamental Research in Homogenous Catalysis; Shilov, V., Ed.; Gordon & Breach: New York, NY, USA, 1986; Volume 3, pp. 1249–1262. [Google Scholar]
- Stanier, R.Y.; Kunisawa, R.; Mandel, M.; Cohen-Bazire, G. Purification and properties of unicellular blue-green algae (Order Chroococcales). Bacter. Rev. 1971, 35, 171–205. [Google Scholar] [CrossRef] [PubMed]
- Komárek, J.; Kaštovský, J.; Mareš, J.; Johansen, J.R. Taxonomic classification of cyanoprokaryotes (cyanobacterial genera) 2014 using a polyphasic approach. Preslia 2014, 86, 235–295. [Google Scholar]
- Strunecký, O.; Ivanova, A.P.; Mareš, J. An updated classification of cyanobacterial orders and families based on phylogenomic and polyphasic analysis (Review). J. Phycol. 2023, 59, 12–51. [Google Scholar] [CrossRef]
- Guiry, M.D.; Guiry, G.M. AlgaeBase; World-Wide Electronic Publication, National University of Ireland: Galway, Ireland, 2009; Available online: https://www.algaebase.org (accessed on 28 May 2023).
- Burke, D.J.; Kretzer, A.M.; Rygiewicz, P.T.; Topa, A.M. Soil bacterial diversity in a loblolly pine plantation: Influence of ecotomycorrhizas and fertilization. FEMS Microbial. Ecol. 2006, 57, 409–419. [Google Scholar] [CrossRef]
- Wilmotte, A.; Van Der Auwera, G.; Dewachter, R. Structure of the 16-S ribosomal RNA of the thermophilic cyanobacterium Chlorogloeopsis HTF (Mastigocladus laminosus HTF) strain PCC 7518, and phylogenetical analysis. FEBS Lett. 1993, 317, 96–100. [Google Scholar] [CrossRef]
- Boyer, S.L.; Flechner, V.R.; Johansen, J.R. Is the 16S–23S rRNA internal transcribed spacer region a good tool for use in molecular systematics and population genetics? A case study in cyanobacteria. J. Mol. Biol. Evol. 2001, 18, 1057–1069. [Google Scholar] [CrossRef]
- Nübel, U.; Garcia-Pichel, F.; Muyzer, G. PCR primers to amplify 16S rRNA genes from cyanobacteria. Appl. Environ. Microbiol. 1997, 63, 3327–3332. [Google Scholar] [CrossRef]
- Řeháková, K.; Johansen, J.R.; Bowen, M.B.; Martin, M.P.; Sheil, C.A. Variation in secondary structure of the 16S rRNA molecule in cyanobacteria with implications for phylogenetic analysis. Fottea 2014, 14, 161–178. [Google Scholar] [CrossRef]
- Taton, A.; Grubisic, S.; Brambilla, E.; De Wit, R.; Wilmotte, A. Cyanobacterial diversity in natural and artificial microbial mats of Lake Fryxell (McMurdo dry valleys, Antarctica): A morphological and molecular approach. Appl. Environ. Microbiol. 2003, 69, 5157–5169. [Google Scholar] [CrossRef]
- Miller, M.; Schwartz, T.; Pickett, B.; He, S.; Klem, E.; Scheuermann, R.H.; Passarotti, M.; Kaufman, S.; O’Leary, M.A. A RESTful API for Access to Phylogenetic Tools via theCIPRES Science Gateway. Evol. Bioinform. 2015, 11, 43–48. [Google Scholar] [CrossRef]
- Drummond, A.J.; Ho, S.Y.W.; Philips, M.J.; Rambaut, A. Relaxed phylogenetics and dating with confidence. PLoS Biol. 2006, 4, e88. [Google Scholar] [CrossRef]
- Gelman, A.; Rubin, D.B. Inference from iterative simulation using multiple sequences. Stat. Sci. 1992, 7, 157–511. [Google Scholar] [CrossRef]
- Zuker, M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res. 2003, 31, 3406–3415. [Google Scholar] [CrossRef]
- Bevilacqua, P.C.; Blose, J.M. Structures, kinetics, thermodynamics and biological functions of RNA hairpins. Ann. Rev. Physic. Chem. 2008, 59, 79–103. [Google Scholar] [CrossRef]
- Miscoe, L.H.; Johansen, J.R.; Vaccarino, M.A.; Pietrasiak, N.; Sherwood, A.R. The diatom flora and cyanobacteria from caves on Kauai, Hawaii. II. Novel cyanobacteria from caves on Kauai, Hawaii. Biblioth. Phycol. 2016, 123, 75–152. [Google Scholar]
- Anagnostidis, K.; Komárek, J. Modern approach to the classification system of cyanophytes. 3. Oscillatoriales. Arch. Hydrobiol. Suppl. 1988, 80, 327–472. [Google Scholar]
- Zammit, G. Systematics and biogeography of sciophilous cyanobacteria; an ecological and molecular description of Albertania skiophila (Leptolyngbyaceae) gen. & sp. nov. Phycologia 2018, 57, 481–491. [Google Scholar]
- Zammit, G.; Billi, D.; Albertano, P. The subaerophytic cyanobacterium Oculatella subterranea (Oscillatoriales, Cyanophyceae) gen. et sp. nov.: A cytomorphological and molecular description. Eur. J. Phycol. 2012, 47, 341–354. [Google Scholar] [CrossRef]
- Perkerson, R.B.; Johansen, J.R.; Kovácik, L.; Brand, J.; Kaštovský, J.; Casamatta, D.A. A unique pseudanabaenalean (cyanobacteria) genus Nodosilinea gen. nov. based on morphological and molecular data. J. Phycol. 2011, 47, 1397–1412. [Google Scholar] [CrossRef]
- Yarza, P.; Yilmaz, P.; Pruesse, E.; Glöckner, F.O.; Ludwig, W.; Schleifer, K.H.; Whitman, W.B.; Euzéby, J.; Amann, R.; Rosselló-Móra, R. Uniting the classification of cultured and uncultured bacteria and archaea using 16S rRNA gene sequences. Nat. Rev. Microbiol. 2014, 12, 635–645. [Google Scholar] [CrossRef]
- Erwin, P.M.; Thacker, R.W. Cryptic diversity of the symbiotic cyanobacterium Synechococcus spongiarum among sponge hosts. Mol. Ecol. 2008, 17, 2937–2947. [Google Scholar] [CrossRef]
- Osorio-Santos, K.; Pietrasiak, N.; Bohunická, M.; Miscoe, L.H.; Kováčik, L.; Martin, M.P.; Johansen, J.R. Seven new species of Oculatella (Pseudanabaenales, Cyanobacteria): Taxonomically recognizing cryptic diversification. Eur. J. Phycol. 2014, 49, 450–470. [Google Scholar] [CrossRef]
- Tang, J.; Shah, M.R.; Yao, D.; Jiang, Y.; Du, L.; Zhao, K.; Li, L.; Li, M.; Waleron, M.M.; Waleron, M.; et al. Polyphasic Identification and Genomic Insights of Leptothermofonsia sichuanensis gen. sp. nov., a Novel Thermophilic Cyanobacteria Within Leptolyngbyaceae. Front. Microbiol. 2022, 13, 765105. [Google Scholar] [CrossRef]
- Shen, L.Q.; Zhang, Z.-C.; Shang, J.L.; Li, Z.-K. Kovacikia minuta sp. nov. (Leptolyngbyaceae, Cyanobacteria), a new freshwater chlorophyll f-producing cyanobacterium. J. Phycol. 2022, 58, 424–435. [Google Scholar] [CrossRef]
- Anagnostidis, K. Untersuchen über die Cyanophycen einiger Thermen in Griechenland. Inst. Syst. Bot. Pflanzengeogr. Univ. Thessalon. 1961, 7, 1–322. [Google Scholar]
- Bohunická, M.; Johansen, J.R.; Fučíková, K. Tapinothrix clintonii sp. nov. (Pseudanabaenaceae, Cyanobacteria), a new species at the nexus of five genera. Fottea 2011, 11, 127–140. [Google Scholar] [CrossRef]
- Tilden, J. Observations on some west American thermal algae. Bot. Gaz. 1898, 26, 89–105. [Google Scholar] [CrossRef]
- Strunecký, O.; Raabová, L.; Bernardová, A.; Ivanova, A.P.; Semanova, A.; Crossley, J.; Kaftan, D. Diversity of cyanobacteria at the Alaska North Slope with description of two new genera: Gibliniella and Shackletoniella. FEMS Microbiol. Ecol. 2022, 96, fiz189. [Google Scholar] [CrossRef] [PubMed]
- Prát, S. Studie o Biolithogenesi; Česká Akademie věd a Umění: Praha, Czech Republic, 1929; 187p. [Google Scholar]
- Kaštovský, J. Welcome to the jungle!—An overview of modern taxonomy of Cyanobacteria. Hydrobiologia, 2023; in press. [Google Scholar]
- Rigonato, J.; Sant’Anna, C.L.; Giani, A.; Azevedo, M.T.P.; Gama, W.A.; Viana, W.L.F.; Fiore, M.F.; Werner, V.R. Sphaerocavum: A coccoid morphogenus identical to Microcystis in terms of 16S rDNA and ITS sequence phylogenies. Hydrobiologia 2003, 811, 35–48. [Google Scholar] [CrossRef]
- Aguilera, A.; Berrendero, E.G.; Kaštovský, J.; Echenique, R.; Salerno, G.L. The polyphasic analysis of two native Raphidiopsis isolates supports the unification of the genera Raphidiopsis and Cylindrospermopsis (Nostocales, Cyanobacteria). Phycologia 2018, 57, 130–146. [Google Scholar] [CrossRef]
- Taton, A.; Wilmotte, A.; Šmarda, J.; Elster, J.; Komárek, J. Plectolyngbya hodgsonii: A novel filamentous cyanobacterium from Antarctic lakes. Polar Biol. 2011, 34, 181–191. [Google Scholar] [CrossRef]
- Turland, N.J.; Wiersema, J.H.; Barrie, F.R.; Greuter, W.; Hawksworth, D.L.; Herendeen, P.S.; Knapp, S.; Kusber, W.-H.; Li, D.-Z.; Marhold, K.; et al. (Eds.) International Code of Nomenclature for algae, fungi, and plants (Shenzhen Code) adopted by the Nineteenth International Botanical Congress Shenzhen, China, July 2017. In Regnum Vegetabile; Koeltz Botanical Books: Glashütten, Germany, 2018; Volume 159. [Google Scholar]
- Casamatta, D.A.; Johansen, J.R.; Vis, M.L.; Broadwater, S.T. Molecular and morphological characterization of ten polar and near-polar strains within the Oscillatoriales (Cyanobacteria). J. Phycol. 2005, 41, 421–438. [Google Scholar] [CrossRef]
- Johansen, J.R.; Kováčik, L.; Casamatta, D.A.; Fučíková, K.; Kaštovský, J. Utility of 16S–23S ITS sequence and secondary structure for recognition of intrageneric and intergeneric limits within cyanobacterial taxa: Leptolyngbya corticola sp. nov. (Pseudanabaenaceae, Cyanobacteria). Nova Hedwig. 2011, 92, 283–302. [Google Scholar] [CrossRef]
- Anagnostidis, K. Nomenclatural changes in cyanoprokaryotic order Oscillatoriales. Preslia 2001, 73, 359–375. [Google Scholar]
- Kim, D.-H.; Lee, N.-J.; Lee, J.-H.; Yang, E.-C.; Lee, O.-M. Three new Plectolyngbya species (Leptolyngbyaceae, Cyanobacteria) isolated from rocks and saltern of the Republic of Korea. Diversity 2022, 14, 1013. [Google Scholar] [CrossRef]
- Gomont, M. Monographie des Oscillariées (Nostocacées Homocystées). Deuxième partie—Lyngbyées. Ann. Sci. Natur. Bot. Série 7 1893, 16, 91–264. [Google Scholar]
- Gomont, M. Monographie des Oscillariées (Nostocacées homocystées). Ann. Sci. Natur. Bot. Série 7 1892, 15, 263–368. [Google Scholar]
- Mai, T.; Johansen, J.R.; Pietrasiak, N.; Bohunická, M.; Martin, M.P. Revision of the Synechococcales (Cyanobacteria) through recognition of four families including Oculatellaceae fam. nov. and Trichocoleaceae fam. nov. and six new genera containing 14 species. Phytotaxa 2018, 365, 1–59. [Google Scholar] [CrossRef]
- Phillipson, J. Some algae of Victorian soils. Proc. R. Soc. Vic. 1935, 47, 262–287. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota. 2. Teil: Oscillatoriales. In Süsswasserflora von Mitteleuropa; Büdel, B., Krienitz, L., Gärtner, G., Schagerl, M., Eds.; Elsevier Spektrum Akademischer Verlag: München, Germany, 2005; Volume 19/2, pp. 1–759. [Google Scholar]
- Pietrasiak, N.; Reeve, S.; Osorio-Santos, K.; Lipson, D.A.; Johansen, J.R. Trichotorquatus gen. nov.—A new genus of soil cyanobacteria discovered from American drylands. J. Phycol. 2021, 57, 886–902. [Google Scholar] [CrossRef]




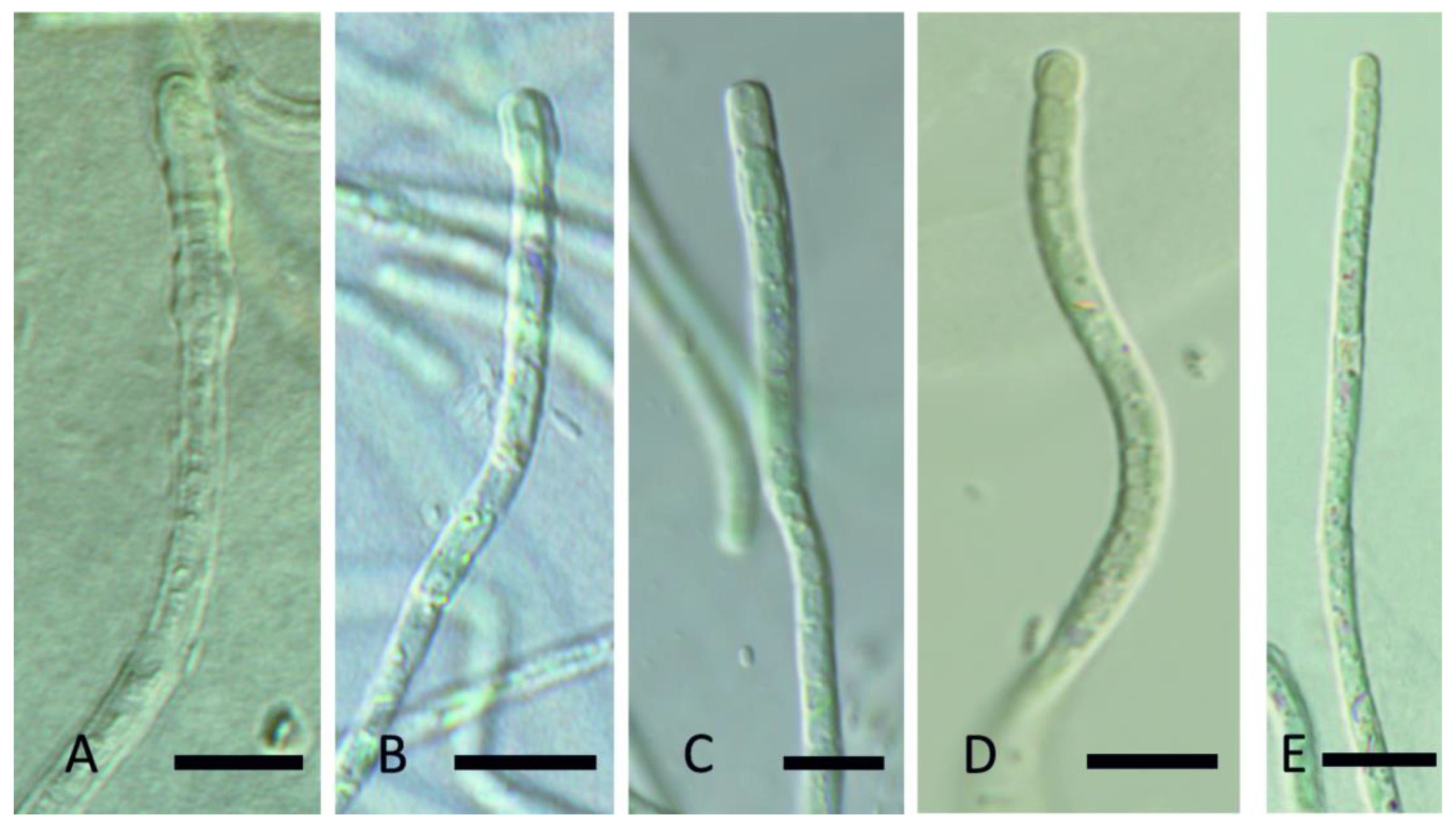
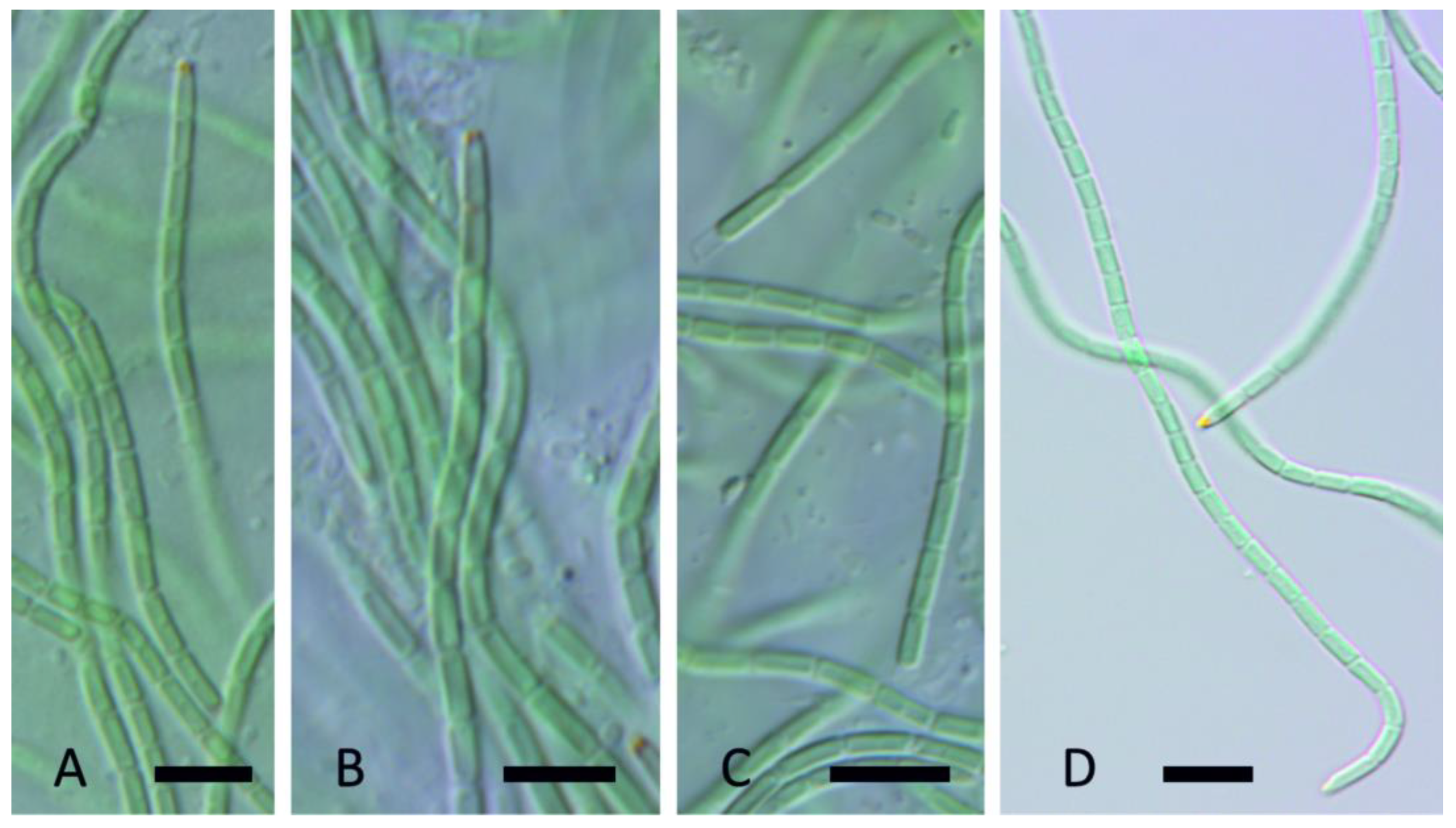
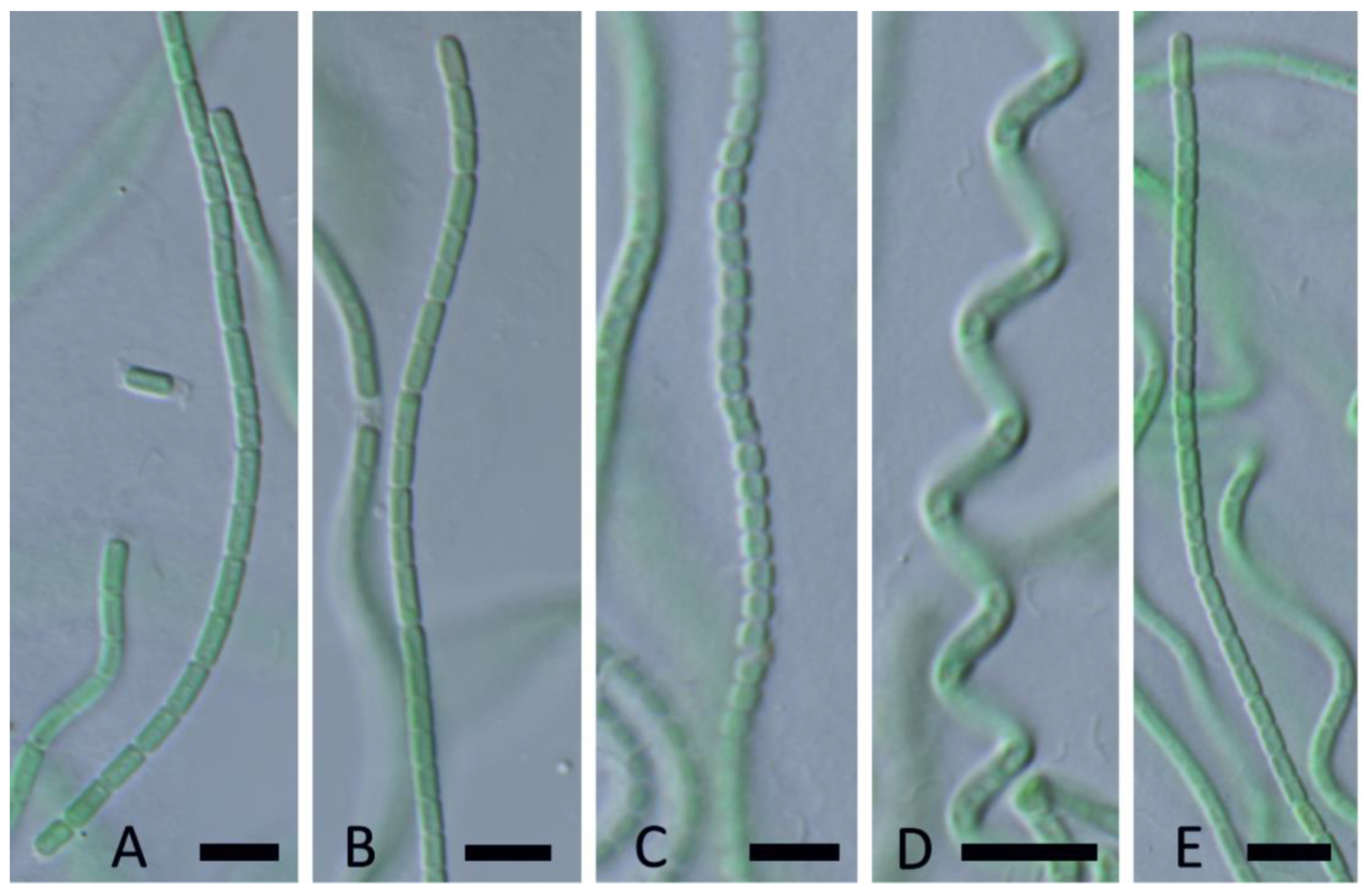
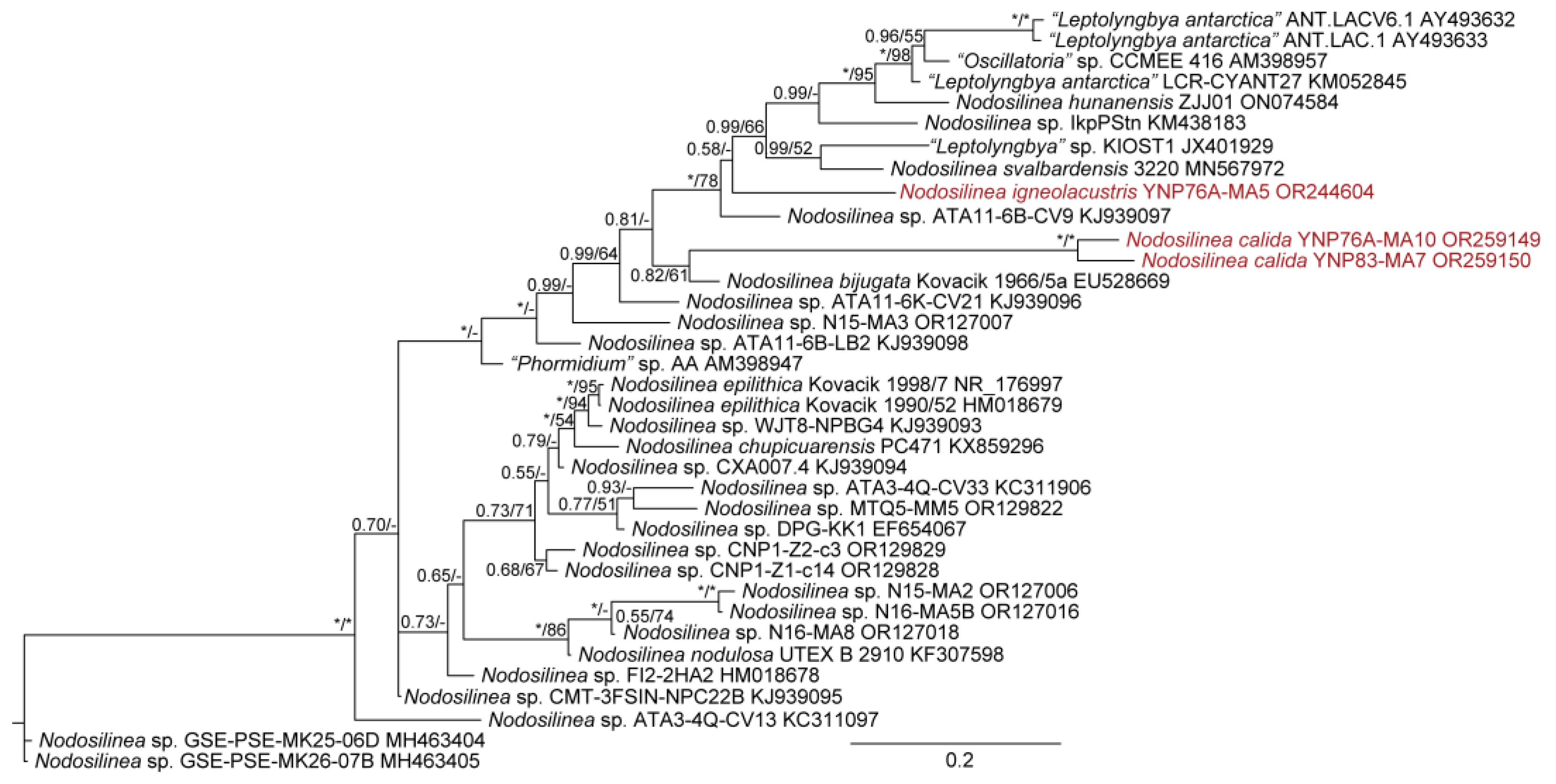
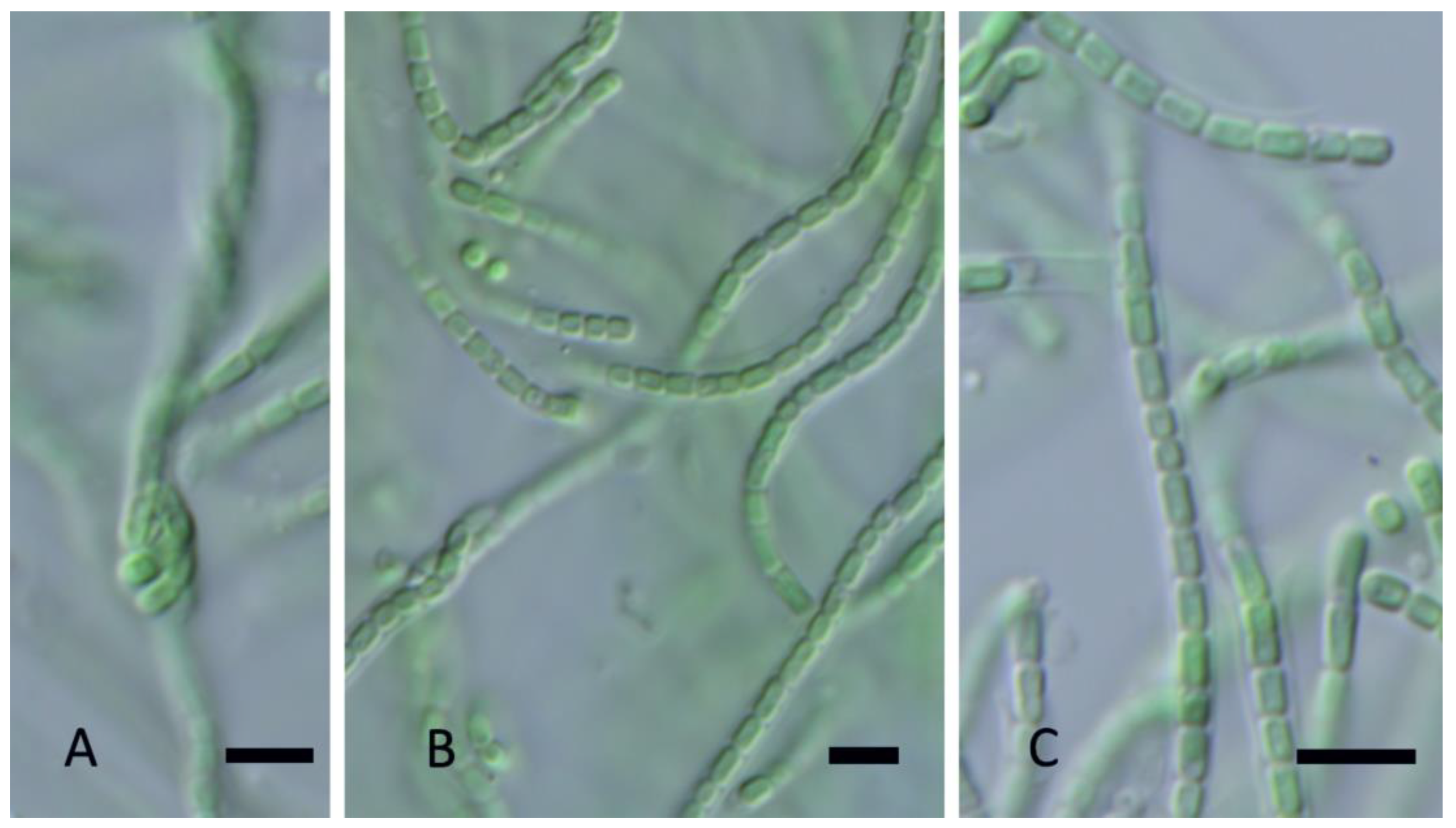
| YNP30-MA1 | YS71-RH2 | YNP81-MA3 | YNP81-MA4 | YNP83-MA6 | YN80B-MA3 | YN80B-MA4 | EY07 AM2 | |
|---|---|---|---|---|---|---|---|---|
| Copelandiella yellowstonensis YNP30-MA1 | ||||||||
| Copelandiella yellowstonensis YS71-RH2 | 99.8 | |||||||
| Copelandiella yellowstonensis YNP81-MA3 | 99.7 | 99.8 | ||||||
| Copelandiella yellowstonensis YNP81-MA4 | 99.7 | 99.8 | 99.8 | |||||
| Copelandiella yellowstonensis YNP83-MA6 | 99.6 | 99.7 | 99.6 | 99.7 | ||||
| Copelandiella thermalis YN80B-MA3 | 97.8 | 97.8 | 97.8 | 97.8 | 97.7 | |||
| Copelandiella thermalis YN80B-MA4 | 97.8 | 97.8 | 97.8 | 97.8 | 97.7 | 100.0 | ||
| Leptolyngbyaceae cynobacterium EY07 AM2 | 91.5 | 91.5 | 91.5 | 91.5 | 91.4 | 90.9 | 90.9 | |
| Leptolyngbyaceae cynobacterium EY01 AM3 | 95.2 | 95.2 | 95.1 | 95.3 | 95.1 | 94.9 | 94.9 | 92.1 |
| YNP81-MA1 | YNP81-MA4 | YNP70-MA1 | YNP83-MA6 | YNP30-MA1 | YNP80B-MA3 | |
|---|---|---|---|---|---|---|
| Copelandiella yellowstonensis YNP81-MA1 | ||||||
| Copelandiella yellowstonensis YNP81-MA4 | 0.0 | |||||
| Copelandiella yellowstonensis YNP70-MA1 | 6.9 | 6.9 | ||||
| Copelandiella yellowstonensis YNP83-MA6 | 7.6 | 7.6 | 1.1 | |||
| Copelandiella yellowstonensis YNP30-MA1 | 7.5 | 7.5 | 4.4 | 3.6 | ||
| Copelandiella thermalis YNP80B-MA3 | 20.2 | 20.2 | 20.3 | 20.1 | 20.8 | |
| Copelandiella thermalis YNP80B-MA4 | 20.2 | 20.2 | 20.3 | 20.1 | 20.8 | 0.0 |
| Leptothermofonsia sichuanensis | Leptolyngbyaceae CENA37 | Leptolyngbyaceae CENA37 | Leptolyngbyaceae CENA35 | Leptolyngbya Greenland 10 | Kovacikia brockii YS7-RH2 | Kovacikia brockii YNP74-RH1 | Kovacikia muscicola HA7619-LM3 | Kovacikia anagnostidisii YS86-RH1 | Kovacikia anagnostidisii YS89-RH1 | Kovacikia minuta CCNU0001 | Kovacikia atmophytica BACA0619 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Leptothermofonsia sichuanensis | ||||||||||||
| Leptolyngbyaceae CENA37 | 95.4 | |||||||||||
| Leptolyngbyaceae CENA37 | 95.5 | 99.9 | ||||||||||
| Leptolyngbyaceae CENA35 | 95.3 | 99.7 | 99.8 | |||||||||
| Leptolyngbya Greenland 10 | 96.4 | 96.6 | 96.7 | 96.7 | ||||||||
| Kovacikia brockii YS7-RH2 | 98.0 | 95.5 | 95.6 | 95.4 | 96.2 | |||||||
| Kovacikia brockii YNP74-RH1 | 97.9 | 95.5 | 95.6 | 95.4 | 96.2 | 99.9 | ||||||
| Kovacikia muscicola HA7619-LM3 | 95.2 | 98.8 | 98.9 | 98.7 | 95.8 | 95.4 | 95.4 | |||||
| Kovacikia anagnostidisii YS86-RH1 | 96.1 | 96.8 | 96.9 | 96.7 | 94.8 | 96.0 | 96.0 | 97.3 | ||||
| Kovacikia anagnostidisii YS89-RH1 | 96.0 | 96.8 | 96.9 | 96.7 | 94.8 | 96.0 | 96.0 | 97.3 | 100 | |||
| Kovacikia minuta CCNU0001 | 95.1 | 97.8 | 97.9 | 97.9 | 95.9 | 95.1 | 95.1 | 97.6 | 96.8 | 96.8 | ||
| Kovacikia atmophytica BACA0619 | 95.9 | 96.8 | 96.9 | 96.7 | 95.6 | 95.4 | 95.4 | 97.1 | 98.2 | 98.2 | 97.1 | |
| Leptolyngbyaceae MTQ1-MM2 | 94.3 | 94.3 | 94.4 | 94.3 | 93.7 | 94.6 | 94.6 | 94.8 | 96.1 | 96.1 | 94.0 | 95.3 |
| Leptothermofonsia sichuanensis | Kovacikia brockii YNP74-RH1 | Kovacikia brockii YS7-RH2 | Kovacikia muscicola HA7619-LM3 | Kovacikia anagnostidisii YS86-RH1 | Kovacikia anagnostidisii YS89-RH1 | Kovacikia atmophytica BACA0619 | |
|---|---|---|---|---|---|---|---|
| Leptothermofonsia sichuanensis | |||||||
| Kovacikia brockii YNP74-RH1 | 18.8 | ||||||
| Kovacikia brockii YS7-RH2 | 18.8 | 0.0 | |||||
| Kovacikia muscicola HA7619-LM3 | 23.9 | 20.6 | 20.6 | ||||
| Kovacikia anagnostidisii YS86-RH1 | 28.5 | 24.0 | 24.0 | 18.5 | |||
| Kovacikia anagnostidisii YS89-RH1 | 28.5 | 24.0 | 24.0 | 18.5 | 0.0 | ||
| Kovacikia atmophytica BACA0619 | 26.5 | 21.2 | 21.2 | 12.0 | 18.2 | 18.2 | |
| Kovacikia minuta CCNUW1 | 26.1 | 21.2 | 21.2 | 17.0 | 24.1 | 24.1 | 17.3 |
| A. prattii YNP74-MA3.1 | A. prattii YNP74-MA3.2 | A. skiophila SA373 | A. alaskaensis L1 | Albertania N14-MA1 | Albertania N14-MA3 | Albertania EcFYyy-200 | Albertania CT103 | Albertania CT115 | Albertania CT305 | |
|---|---|---|---|---|---|---|---|---|---|---|
| A. prattii YNP74-MA3.1 | ||||||||||
| A. prattii YNP74-MA3.2 | 99.4 | |||||||||
| A. skiophila SA373 | 96.6 | 96.6 | ||||||||
| A. alaskaensis L1 | 98.1 | 98.0 | 96.6 | |||||||
| Albertania N14-MA1 | 98.0 | 97.9 | 98.1 | 97.9 | ||||||
| Albertania N14-MA3 | 96.6 | 96.5 | 96.9 | 97.8 | 97.7 | |||||
| Albertania EcFYyy-200 | 97.5 | 97.4 | 97.2 | 98.8 | 97.9 | 98.2 | ||||
| Albertania CT103 | 97.8 | 97.7 | 96.9 | 97.1 | 97.7 | 96.1 | 97.0 | |||
| Albertania CT115 | 95.1 | 95.0 | 93.6 | 95.2 | 95.2 | 94.8 | 94.8 | 95.0 | ||
| Albertania CT305 | 96.5 | 96.4 | 97.0 | 97.8 | 97.4 | 98.6 | 98.4 | 96.6 | 94.7 | |
| Albertania CT315 | 96.2 | 96.1 | 96.1 | 97.0 | 96.6 | 97.6 | 97.5 | 95.9 | 95.8 | 97.5 |
| A. prattii YNP74-MA3.1 | A. prattii YNP74-MA3.2 | A. skiophila SA373 | A. alaskaensis KV23 | Albertania sp. N14-MA1 | Albertania sp. N14-MA3 | Albertania sp. CT103 | Albertania sp. CT305 | Albertania sp. CT315 | |
|---|---|---|---|---|---|---|---|---|---|
| Albertania prattii YNP74-MA3.1 | |||||||||
| Albertania prattii YNP74-MA3.2 | 16.7 | ||||||||
| Albertania skiophila SA373 | 13.1 | 22.5 | |||||||
| Albertania alaskaensis KV23 | 15.2 | 18.7 | 19.4 | ||||||
| Albertania sp. N14-MA1 | 13.7 | 16.3 | 15.0 | 15.3 | |||||
| Albertania sp. N14-MA3 | 14.0 | 16.7 | 12.5 | 17.5 | 8.0 | ||||
| Albertania sp. CT103 | 14.7 | 16.1 | 13.8 | 17.3 | 7.8 | 8.1 | |||
| Albertania sp. CT305 | 11.5 | 17.2 | 10.6 | 13.5 | 9.8 | 12.0 | 9.6 | ||
| Albertania sp. CT315 | 14.3 | 19.6 | 15.3 | 20.8 | 14.5 | 11.8 | 12.9 | 11.2 | |
| Albertania sp. CT115 | 11.2 | 26.3 | 20.6 | 14.8 | 15.8 | 13.2 | 13.8 | 16.2 | 23.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaštovský, J.; Johansen, J.R.; Hauerová, R.; Akagha, M.U. Hot Is Rich—An Enormous Diversity of Simple Trichal Cyanobacteria from Yellowstone Hot Springs. Diversity 2023, 15, 975. https://doi.org/10.3390/d15090975
Kaštovský J, Johansen JR, Hauerová R, Akagha MU. Hot Is Rich—An Enormous Diversity of Simple Trichal Cyanobacteria from Yellowstone Hot Springs. Diversity. 2023; 15(9):975. https://doi.org/10.3390/d15090975
Chicago/Turabian StyleKaštovský, Jan, Jeffrey R. Johansen, Radka Hauerová, and Mildred U. Akagha. 2023. "Hot Is Rich—An Enormous Diversity of Simple Trichal Cyanobacteria from Yellowstone Hot Springs" Diversity 15, no. 9: 975. https://doi.org/10.3390/d15090975
APA StyleKaštovský, J., Johansen, J. R., Hauerová, R., & Akagha, M. U. (2023). Hot Is Rich—An Enormous Diversity of Simple Trichal Cyanobacteria from Yellowstone Hot Springs. Diversity, 15(9), 975. https://doi.org/10.3390/d15090975






